Abstract
Meniscal degeneration is a common finding even in young patients’ knees, and it is regarded as a predictor for the onset of early osteoarthritis (OA). When symptomatic, it represents a challenge since arthroscopic surgery provides unpredictable results: recent evidence has shown that partial meniscectomy is not better than conservative management up to 2 years of follow-up, and the removal of meniscal tissue may accelerate OA progression toward OA. Intra-articular injection of corticosteroids or hyaluronic acid may help in providing temporary symptomatic relief, but no influence should be expected on the quality of the meniscal tissue. Biologic agents have been adopted to treat a variety of degenerative musculoskeletal pathologies, and the use of platelet-derived growth factors (GFs) has become routine. Preclinical studies have documented that platelet-derived GFs may play a beneficial role in stimulating meniscal repair and regeneration by triggering anabolic pathways and stimulating local mesenchymal stem cells from synovium. Furthermore, also mechanical stimulation (e.g., arthroscopic trephination or percutaneous needling) in the red-red or red-white zone may further promote tissue healing. The purpose of the present brief report is to describe the clinical outcomes at 18 months’ follow-up in a cohort of patients affected by symptomatic medial meniscus degeneration and treated by percutaneous needling plus intra- and perimeniscal injection of autologous conditioned plasma (ACP). The procedure was shown to be safe and provided significant pain reduction and improvement in subjective scores. This treatment option deserves further investigation in a comparative setting, to establish whether it could offer advantage over isolated intra-articular injections.
Introduction
Meniscal degeneration is a common finding even in young patients’ knees, and it is regarded as a predictor for the onset of early osteoarthritis (OA). 1 When symptomatic, it represents a challenge since arthroscopic surgery provides unpredictable results: recent evidence has shown that partial meniscectomy is not better than conservative management up to 2 years of follow-up,2,3 and the removal of meniscal tissue may accelerate OA progression. 1 Intraarticular injection of corticosteroids or hyaluronic acid may help in providing temporary symptomatic relief, but no influence should be expected on the quality of the meniscal tissue.4,5 Biologic agents have been adopted to treat a variety of degenerative musculoskeletal pathologies and the use of platelet-derived growth factors (GFs) has become routine. Preclinical studies have documented that platelet-derived GFs may play a beneficial role in stimulating meniscal repair and regeneration by triggering anabolic pathways and stimulating local mesenchymal stem cells from synovium.6,7 Furthermore, also mechanical stimulation (e.g., arthroscopic trephination or percutaneous needling) in the red-red or red-white zone may further promote tissue healing. 8 The purpose of the present brief report is to describe the clinical outcomes at 18 months’ follow-up in a cohort of patients affected by symptomatic medial meniscus degeneration and treated by percutaneous needling plus intra- and perimeniscal injection of autologous conditioned plasma (ACP). The procedure was shown to be safe and provided significant pain reduction and improvement in subjective scores. This treatment option deserves further investigation in a comparative setting, to establish whether it could offer advantage over isolated intra-articular injections.
Methods, Results, and Discussion
Twelve patients (10 men, 2 woman; mean age 51.7 ± 19.1; mean body mass index 23.8 ± 2.2) were included according to the following inclusion/exclusion criteria: (1) magnetic resonance imaging (MRI) evidence of medial degenerative meniscopathy (body and posterior horn); (2) pain and tenderness located in the medial joint line; (3) Kellgren-Lawrence grade 0 or 1; (4) no MRI evidence of chondral/osteochondral lesion more than grade II according to the International Cartilage Repair Society scale; (5) absence of mechanical symptoms such as locking or catching; (6) absence of imaging-documented bucket handle and flap tears; (7) failure of previous conservative treatment (pharmacological and physiotherapy); (8) no other intraarticular injection in the previous 6 months; (9) no previous surgery on the index knee; and (10) absence of systemic and oncological diseases and ongoing infections.
All the patients were treated by 3 ultrasound (US)-guided intra- and perimeniscal injections of approximately 2 cc of ACP (Arthrex, Naples, FL) at 2 weeks interval.
9
The injections were performed under US guidance using a linear probe, which was placed perpendicular to the medial joint line with the knee at about 70° flexion. Then a 22-g needle was introduced with an “in-plane” technique (
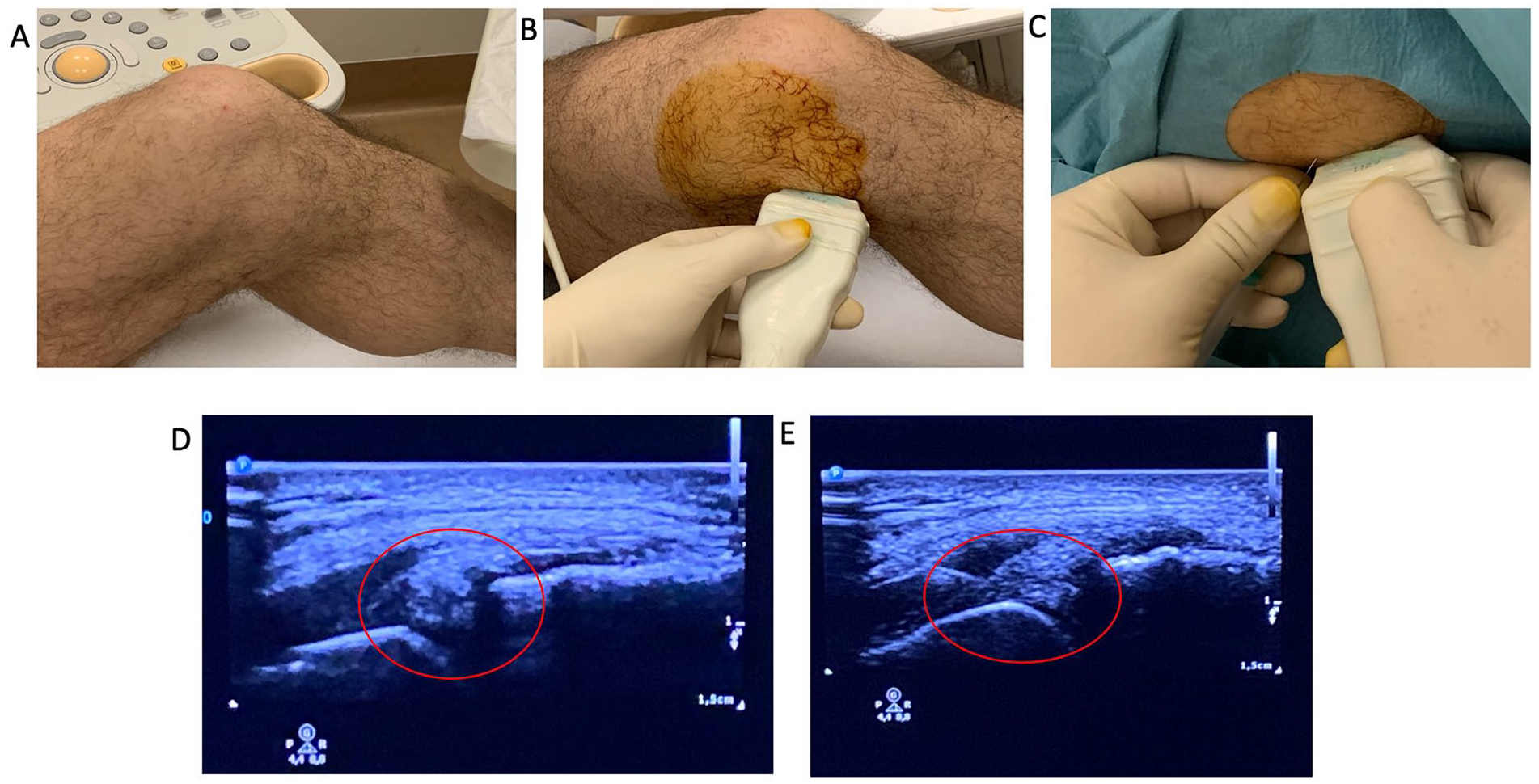
Operative procedure: (
No severe adverse events were reported: the most common adverse event was pain in the injection site, which was treated by cryotherapy and rest on complete resolution. IKDC-subjective score increased from 50.1 ± 18.0 to 71.5 ± 14.2 at 6 months (P < 0.001), with a further significant improvement at 12 months’ evaluation (78.6 ± 11.9; P = 0.03 vs. 6 months) and then stable values up to the final 18 months’ follow-up (81.6 ± 13.9; P = n.s.;
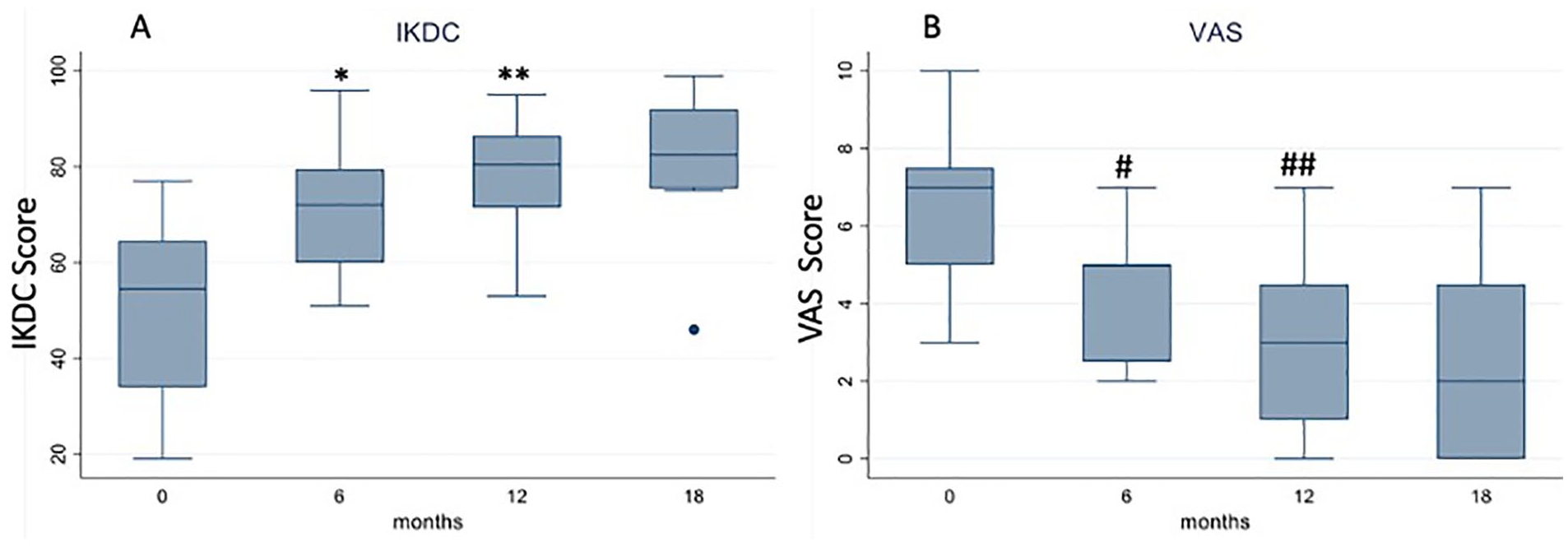
Trend of (
Biologic agents, 10 in particular platelet-derived GFs, have gained a role in the treatment of many orthopedic conditions: although therapeutic protocols involving platelet-rich products still lack standardization and no guidelines have been yet released, positive results have been documented, especially in the treatment of OA. 11 Looking at in vitro evidence, autologous GFs play a role in promoting cartilage anabolism, type II collagen deposition, extracellular matrix remodeling, and they also contribute to reducing inflammatory and catabolic distress. This complex pattern of actions justifies their use in meniscal degeneration,6,7 which is a routine clinical finding even in young or middle-aged patients. 12 Although often accompanied by chondral damage, degenerative meniscal injuries have peculiar clinical features, and that may justify a therapeutic approach specifically targeted to the meniscus. 13 Furthermore, clinical experience, supported by data coming from well-designed randomized controlled trials, suggests that meniscectomy should not be considered as a first-line approach in patients with meniscal degeneration, and only the presence of mechanical symptoms, such as catching and locking, may indicate arthroscopic treatment. 1 Therefore, nonoperative management should be preferred, and among the options available, injective treatments are routinely performed. To this regard, US, which is nowadays largely available in an outpatient setting, allows to asses both intra- and periarticular structures and may offer a significant advantage to selectively treat meniscal pathology. In fact, one potential limitation of intra-articular injections is that the substance distributes in the whole knee and, likely, only a limited amount reaches the degenerated meniscus. This is particularly relevant when using biologic products, such as ACP, whose action is to stimulate tissue repair and healing locally. Therefore, the use of US allows the release of GFs selectively in the meniscal damaged area, and the liquid shape of ACP is another advantage, since it makes intra-meniscal injection effortless compared with more viscous substances. Recent studies14,15 on cadaveric specimens have highlighted that intra- and perimeniscal delivery of therapeutics is feasible, does not require any special equipment, and the use of anatomical landmarks makes the procedure easier and more accurate. 14 Moreover, the presence of degenerated tissue allows the majority of the injected substance to remain inside the meniscus. 14 Anyway, in vivo, it could be difficult to inject all the product inside the meniscus due to the increase in tissue pressure, so a peri-meniscal administration can be also performed, 15 maintaining the needle in the meniscocapsular junction in order to reduce the risk of product leakage in the intra-articular space, which may however occur, especially if the meniscus has torn areas. Under a technical point of view, particular care should be reserved when injecting the lateral meniscus due to its greater mobility which makes needle positioning more challenging. 14
The present trial further confirmed the feasibility of the procedure and its safety, and it also revealed positive clinical outcomes up to 18 months’ follow-up in patients with no or minimal signs of OA (Kellgren-Lawrence 0 or 1). In regard to the therapeutic protocol, we opted for a 3-injection regimen based on our previous experience with intra-articular injections, even if there is no conclusive data to support multiple injections versus a single administration in this specific clinical setting, as well as there is no evidence on the optimal amount of ACP to administer.
Some major limitations must be acknowledged: (1) the small sample size included; (2) the lack of a control group (e.g., intraarticular injections); (3) the concurrent use of needling, which may have itself a therapeutic potential, thus impeding a clear evaluation of the contribution of the sole ACP; and (4) the impossibility of collecting post-treatment MRI to assess the evolution of meniscal signal over time.
These preliminary data suggest that a “precision” approach, aimed at treating the “trigger” tissue rather than the whole joint, can provide a timely and lasting clinical improvement. The use of biologic products has given us a new strategy to enhance tissue healing, but further insights are needed to understand how to fully take advantage of their therapeutic potential: among the factors to consider, administration modalities play a significant role because they can allow the release of biologic agents where they are most needed, thus maximizing their effects. Based on these premises, US-guided meniscal injections might represent a valuable treatment option for meniscal degeneration, worthy of future investigations in the context of a properly designed randomized trial.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Berardo Di Matteo has been a paid speaker for Arthrex. The other authors have nothing to disclose.
Ethical Approval
Ethical approval for this study was obtained from the IRCCS Humanitas Research Center Ethical Committee (Approval number: 1851).
Or
Ethical approval for this study was waived by *NAME OF ETHICS COMMITTEE OR INSTITUTIONAL REVIEW BOARD* because *REASON FOR WAIVER*.
Or
Ethical approval was not sought for the present study because *REASON*.
Informed Consent
Written informed consent was obtained from all subjects before the study.
