Abstract
Purpose
To evaluate the clinical and biological outcome of combined bone marrow aspirate concentrate (BMAC) and platelet-rich plasma (PRP) on a collagen scaffold for treating cartilage lesions in the knee.
Methods and Materials
Ten patients (mean age 29.4 years, range 18-36) suffering from large full-thickness cartilage in the knee were treated with BMAC and PRP from January 2015 to December 2016. In a 1-step procedure autologous BMAC and PRP was seeded onto a collagen scaffold and sutured into the debrided defect. Patients were evaluated by clinical outcome scores (IKDC [International Knee Documentation Committee Subjective Knee Form], KOOS [Knee Injury and Osteoarthritis Outcome Score], and pain score using the Numeric Rating Scale [NRS]) preoperatively, after 3 months, and after 1 and 2 years. Second-look arthroscopies were performed (n = 7) with biopsies of the repair tissue for histology. All patients had magnetic resonance imaging (MRI) preoperatively, after 1 year, and after 2 to 3.5 years with MOCART (magnetic resonance observation of cartilage repair tissue) scores evaluating cartilage repair.
Results
After 1 year significant improvements were found in IKDC, KOOS symptoms, KOOS ADL (Activities of Daily Living), KOOS QOL (Quality of Life), and pain at activity. At the latest follow-up significant improvements were seen in IKDC, KOOS symptoms, KOOS QOL, pain at rest, and pain at activity. MRI MOCART score for cartilage repair improved significantly from baseline to 1-year follow-up. Histomorphometry of repair tissue demonstrated a mixture of fibrous tissue (58%) and fibrocartilage (40%).
Conclusion
Treatment of cartilage injuries using combined BMAC and PRP improved subjective clinical outcome scores and pain scores at 1 and 2 years postoperatively. MRI and histology indicated repair tissue inferior to the native hyaline cartilage.
Introduction
Articular cartilage repair is biologically challenging due to its highly complex, avascular, alymphatic, and aneural nature. Cell-based repair techniques with cultured chondrocytes (autologous chondrocyte implantation [ACI]) have proven to lead to superior morphological, biological, and clinical cartilage repair outcomes compared with alternative treatments.1,2 The ACI procedure, however, has a number of drawbacks. It requires 2 separate surgeries, has a limited availability of donor cartilage, and requires an interposed and very costly laboratory chondrocyte culturing step. 3
Mesenchymal stem cells (MSCs) was originally proposed as a cell source for cartilage repair with a less invasive sample harvesting yielding highly proliferative cells able to undergo chondrogenic differentiation in vitro and produce hyaline-like cartilage.4-6 However, use of cultured MSCs has the same high costs as ACI. Therefore, there is great interest in developing new 1-step procedures. The use of bone marrow aspirate concentrate (BMAC) obtained and isolated intraoperatively may be an attractive 1-step method for cartilage repair. BMAC can be used as a cell source in combination with a scaffold as alternative to MACI, as intraarticular injections, or as an adjuvant to other treatments.3,7,8
Currently, bone marrow cells are utilized clinically as a cell source in marrow-stimulating cartilage repair techniques such as microfracture and subchondral drilling. In these techniques the available MSCs for the cartilage repair is much smaller compared with MSCs in bone marrow aspirated from the iliac crest. 8 MSCs represent only 0.001% to 0.02% of the nucleated cells in bone marrow.9-12 In spite of their small proportion, the MSCs may promote healing through a paracrine effect on the surrounding tissue. 13 In vivo studies applying the premise of MSCs as donor cells for differentiation into chondrocytes have been with varying outcomes.5,14-17 More recently, the role of MSCs as differentiating cells has been questioned, and several studies highlight their potential pivotal role in the regenerative microenvironment in vivo as medicinal drugstores for immunomodulation and anabolic stimulation of host cells.6,18-24 Furthermore, BMAC is a rich source of growth factors that promote chondrogenesis.25,26
Platelet-rich plasma (PRP) is an increasingly popular source of autologous growth factors and bioactive proteins released on platelet activation, which are believed to promote cartilage matrix synthesis. 27 Similar to BMAC, PRP can be administered as monotherapy as intraarticular injections 28 or as adjuvant to other cartilage repair treatments. 29
A strategy using BMAC seeded onto a collagen I/III scaffold (Chondro-Gide) with or without PRP has been proposed and has shown promising clinical results with regards to subjective clinical outcome and magnetic resonance imaging (MRI) evaluations.3,30,31 Improvements with regard to the same outcomes have also been seen using other methods to deliver the BMAC.7,32,33 There are, however, only few studies evaluating both the clinical and histological responses of BMAC for cartilage repair and further studies are needed to determine the indications, effects, and the best applications of BMAC.34,35
The aim of this study was to evaluate the clinical and biological outcome of combined BMAC and PRP on a collagen I/III scaffold for cartilage repair in the knee. We hypothesized that the treatment would result in improvement in subjective clinical outcome and result in hyaline and fibrocartilaginous repair tissue evaluated by MRI and histologic evaluation of second-look biopsies.
Materials and Methods
The study was a prospective case series of 11 knees (10 patients) (mean age 29.4 years, range 18-36) with large full-thickness cartilage lesions that underwent surgical cartilage repair treatment at Aarhus University Hospital, using BMAC and PRP on a collagen I/III scaffold. The local ethics committee under the Danish National Committee on Health Research Ethics approved the protocol (#M-2014-389-14).
Patients
Patients suffering from large full-thickness cartilage lesions on patella or the femoral condyles were included in the study. Inclusion criteria were the following: symptomatic focal chondral lesions between 2 and 8 cm2 grade 3 or 4 using the ICRS (International Cartilage Repair Society) classification based on MRI or previous arthroscopy, age between 18 and 50 years, ASA (American Society of Anesthesiologists) group 0 to 2, and patient agreement to follow rehabilitation protocol and to attend follow-up visits.
Exclusion criteria were diagnosed malignancy, tricompartmental arthritis, injury diagnosed as osteochondritis dissecans, untreated meniscal lesion, untreated ruptures of the cruciate and collateral ligaments, knee axis malalignment >5° based on clinical evaluation, rheumatoid arthritis, Mb. Bechterew, poor general health ASA 3 or higher, alcohol or drug abuse, and systemic or intraarticular corticoid steroid treatment within the past 6 months.
Eight patients had the cartilage lesion(s) in the patella, 2 patients in the trochlea, and 1 patient in the medial femoral condyle. Patient demographics and characteristics are shown in Table 1 .
Patient Demographics and Lesion Details (Intraoperatively Measured Defect Sizes).
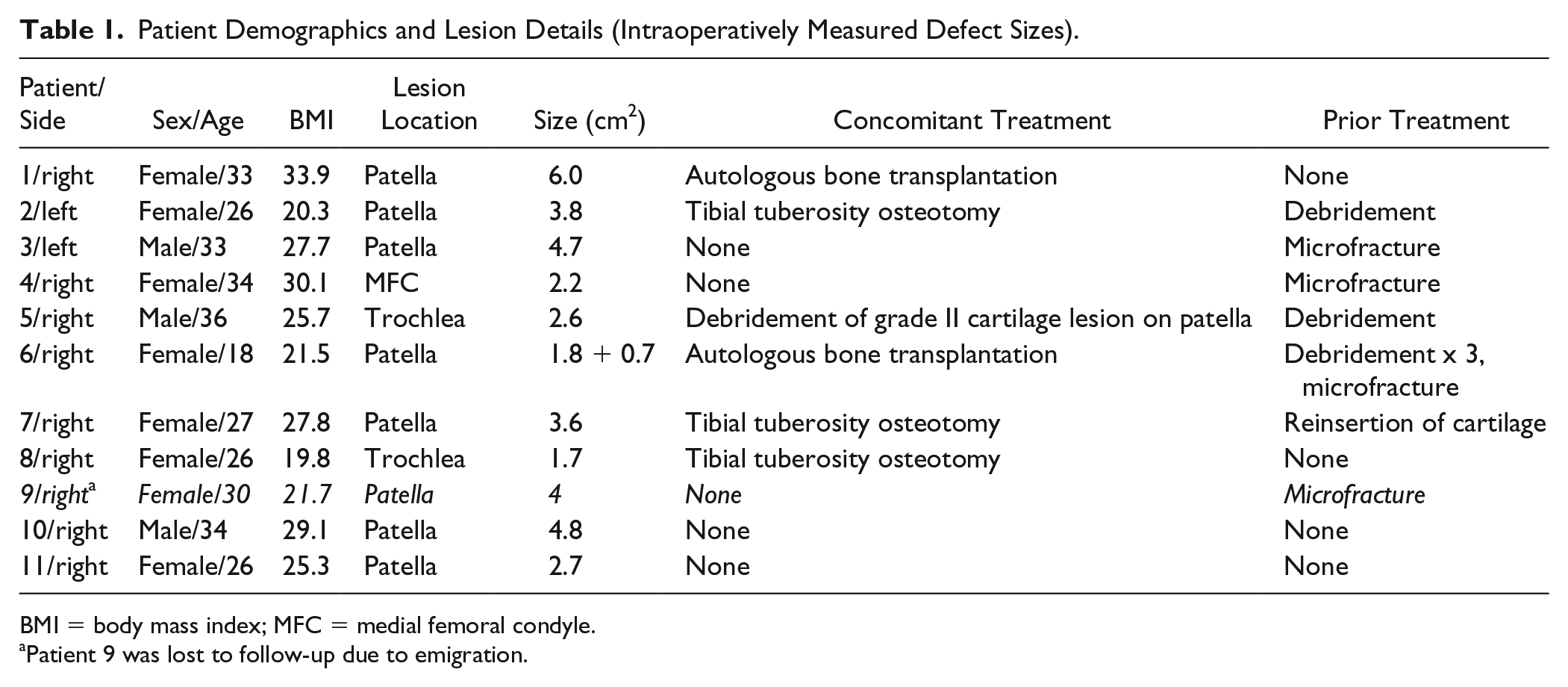
BMI = body mass index; MFC = medial femoral condyle.
Patient 9 was lost to follow-up due to emigration.
Preparation of BMAC, PRP, and Thrombin
BMAC, PRP, and thrombin were prepared according to manufacturer’s instructions (Zimmer Biomet, Winterthur, Switzerland). After the patient was anaesthetized, bone marrow was aspirated from the posterior iliac crest using sterile technique. A small incision was made, and an 11-gauge needle was used to enter the marrow cavity. Twenty-four to 48 mL of bone marrow were aspirated into a heparin-coated (1000 U/mL) syringe containing 6 to 12 mL ACD-A (Anti-coagulant Citrat Dextrose Solution A). The bone marrow aspirate was concentrated using a MarrowStim Mini Concentration System (Zimmer Biomet). Centrifugation was performed for 15 minutes at 3200 rpm.
For PRP preparation, 27 mL of whole blood was aspirated into a heparin-coated (1000 U/mL) syringe containing 3 mL ACD-A and 3 mL PRP was yielded using a Mini GPS III Platelet Separation System (Zimmer Biomet). Centrifugation was performed for 15 minutes at 3200 rpm. For thrombin, 11 mL of whole blood was aspirated into a heparin-coated (1000 U/mL) syringe containing 1 mL ACD-A (Zimmer Biomet). Thrombin was yielded using a Clotalyst Autologous Clotting Factor (Zimmer Biomet).
Surgery
Prior to the open-surgery procedure, a knee arthroscopy was performed to identify the defect and additional joint pathology.
Following arthroscopy, the defect was exposed through a parapatellar arthrotomy. The damaged cartilage as well as the calcified layer was removed using a curette until sharp vertical edges of healthy articular cartilage was achieved. After debriding the defect an aluminum template of the scaffold was used to measure the size of the defect and used to cut out the collagen I/III (Chondro-Gide, Geistlich Pharma, Wolhausen, Switzerland) scaffold in the correct size and shape. The BMAC was seeded onto the scaffold and allowed to adhere to the scaffold for 5 minutes. Subsequently, a mix of thrombin and PRP was placed on top of the BMAC with the purpose of yielding a clot to preserve BMAC in the scaffold. The scaffold with BMAC and PRP was then placed on the defect with the BMAC and PRP/thrombin pointing toward the defect.
After size adjustment the scaffold was sutured to the cartilage in the periphery of the defect using 6-0 vicryl suture and finally fibrin glue was applied along the edge to make a water-proof seal ( Fig. 1 ).
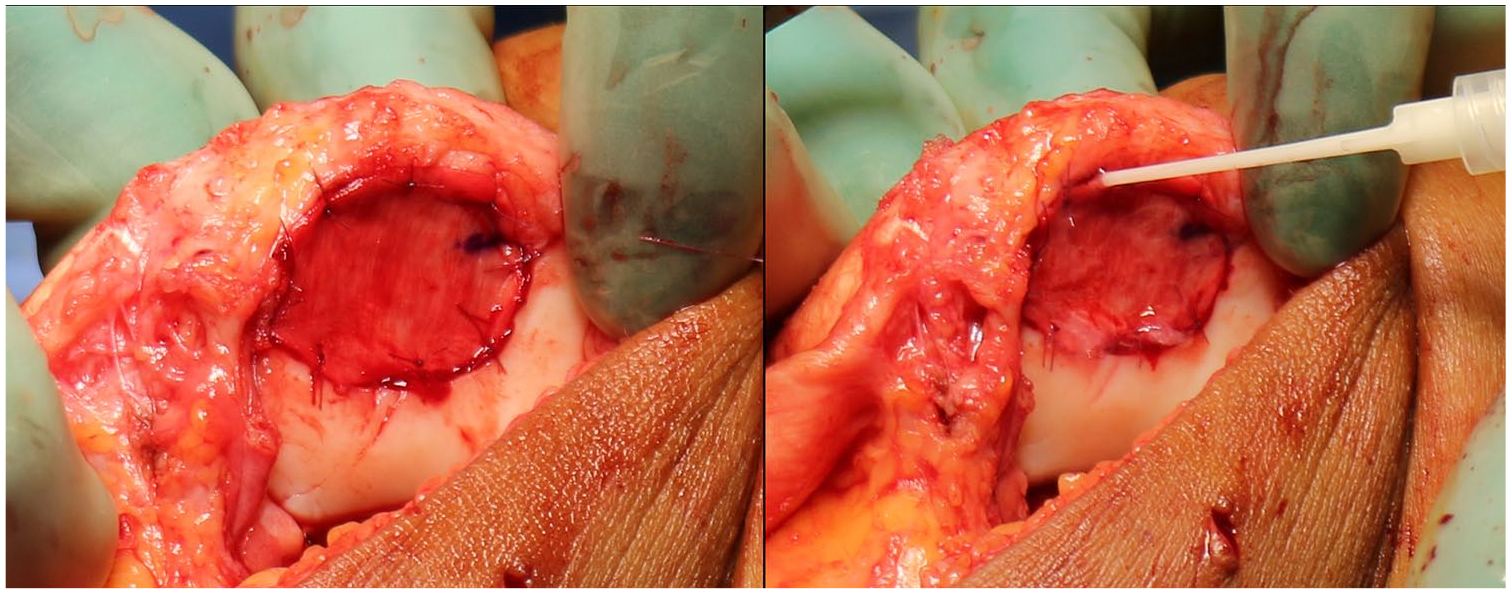
The scaffold is inserted with the BMAC + thrombin/PRP side pointing toward the defect, sutured to the cartilage in the periphery of the defect (left) and sealed with fibrin glue (right).
Postoperative Rehabilitation
All patients attended a stepwise activity progression program for the first 3 months postoperative. The patients were allowed 0° to 20° range of motion for the first 2 weeks in a hinged brace. From 2 to 6 weeks 0° to 90° range of motion and from 6 weeks full range of motion was allowed. No weight bearing was allowed from 0 to 2 weeks. From 2 to 6 weeks, partial weight bearing was allowed. Impact activities were allowed after 6 months. Trained physiotherapists supervised the postoperative rehabilitation. Nonsteroidal anti-inflammatory drugs were prohibited for the initial 3 months.
Clinical Evaluation
The clinical subjective outcome was evaluated using the International Knee Documentation Committee Subjective Knee Form (IKDC) and the Knee injury and Osteoarthritis Outcome Score (KOOS). Pain was scored during rest and activity using the Numeric Rating Scale (NRS) from 0 to 10, with 0 being no pain and 10 being the worst pain. Scores were obtained through patients filling out the questionnaires in writing or via telephone evaluation by a trained medical professional. Scores were obtained preoperatively, and after 1 and 2 years.
Magnetic Resonance Imaging Evaluation
For evaluation of chondral repair, patients had MRI scan prior to surgery, at 1 year and again at 2 to 3.5 years postoperatively. The images were evaluated by an orthopedic surgeon and an experienced radiologist using the 3-dimensional (3D) MOCART (magnetic resonance observation of cartilage repair tissue) score. 36 Images were scored according to 11 parameters: defect fill, cartilage interface, bone interface, repair tissue surface, repair tissue structure, signal intensity, subchondral lamina, chondral osteophytes, bone marrow edema, subchondral bone, and joint effusion. The score ranges from 0 (worst) to 100 (best).
Preoperative MRIs were performed on various 1.5 T MRI scanners with the exception of one patient, who had the preoperative scan on a Magnetom Skyra 3T scanner (Siemens Healthcare, Erlangen, Germany). One-year scans were all performed on ONI Optima 430s 1.5 T extremity scanner (GE Healthcare, Buckinghamshire, UK). The following sequences were acquired for evaluation on this scanner: Axial STIR, coronal FSE T1, sagittal SmartFat-fat water separation technique (FSEfw) PD FS, sagittal water FSEfw PD FS, and sagittal 3D T2* mapping.
MRI scans at 2 to 3.5 years were performed on a Magnetom Skyra 3T scanner with the exception of 2 patients being scanned on 2 different 1.5 T MRI scanners. The following sequences were acquired on the 3 T scanner: T1W coronal, PDW fatsat coronal, PDW fatsat sagittal, PDW fatsat transversal, T2W mDixon sagittal; T2W Water, T2W in phase, T2W out phase, and T2W fat.
Second-Look Arthroscopy
All patients were offered a second-look arthroscopy and biopsy at 1-year follow-up. Seven second-look arthroscopies were performed, and seven 2-mm-diameter osteochondral biopsies were taken from the center of the cartilage repair tissue. The biopsies were obtained at an average of 18.1 months (range 11-32 months) postoperative. One patient had second-look arthroscopy after 11 months due to mechanical symptoms and a loose body in the knee was removed.
Arthroscopy video pictures were available for evaluation and were evaluated semiquantitatively by 2 experienced orthopedic surgeons using the Arthroscopic International Cartilage Repair Society classification. 37 The arthroscopic ICRS (International Cartilage Repair Society) score evaluates the macroscopic appearance according to degree of defect repair, integration to border zone, and macroscopic appearance. Each parameter is scored from 0 to 4. The overall repair tissue is termed as normal with a score of 12, nearly normal with a score of 8 to 11, abnormal with a score of 4 to 7, and severely abnormal with a score of 0 to 3.
Histochemistry, Immunohistochemistry, and Histomorphometry
The biopsies were prepared as previously described. 38 Samples were dehydrated in ethanol of increasing concentration (70% to 96%) and cleared in isopropanol and xylene. Finally, the samples were embedded in methyl methacrylate and cut in 7-µm slices using a hard tissue microtome (Reichert Jung Polycot). All samples were stained with hematoxylin and eosin, safranin O, and toluidine blue according to the protocol published by our group. 39 Immunohistochemical staining with polyclonal rabbit antibodies for collagen type I (Abcam, Ab 34710, Cambridge, UK) and collagen type II (Neomarkers, MS 306-P0, Fremont, CA) was performed as previously described using a Dako Autosatiner (Dako Universal Staining System, Carpinteria, CA). 40 Negative staining controls were labeled with rabbit serum (Dako, X0902) or mouse IgG isotype control (Thermo Fisher Scientific, Camarillo, CA), respectively. Labeling was done using streptavidin–horse radish peroxidase and aminoethyl carbazole according to manufacturer’s instructions (Dako). Slides were counterstained with Mayer’s hematoxylin. A visual analogue scale (0-100) was used to evaluate the samples stained with collagen type I and II according to the percentage of positively stained repair tissue. Quartiles were used to range the amount of positively stained tissue from 0% to 25%, 25% to 50%, 50% to 75%, or 75% to 100%.
Repair tissue was morphologically quantified by histomorphometry as described by Foldager et al. 41 using polarized light for discrimination between tissue types. Sections were cut for every 70 µm, resulting in a total of 5 to 7 sections per biopsy. A 5 × 5 point counting grid was superimposed onto each section at ×10 magnification (newCAST software; Visiopharm). In the repair tissue area, a region of interest was drawn, and tissue was counted according to tissue type (hyaline cartilage, fibrocartilage, fibrous tissue, bone, or vascular tissue).
Statistical Analysis
All data are reported as mean scores (± SD). Data were tested for normality using histograms and Q-Q plots. Paired t test (normally distributed data) and Wilcoxon test (nonnormally distributed data) were performed on the data for MOCART, KOOS, IKDC, and NRS scores to test differences between baseline and follow-up times. Statistical analysis was performed using STATA version 15.0 (StataCorp, College Station, TX).
Results
One patient was lost to follow-up due to emigration (patient 9).
Clinical Evaluation
All patient-reported outcome measures (PROMs) improved from baseline to follow-up ( Table 2 ).
Mean Clinical Subjective Outcome Scores.
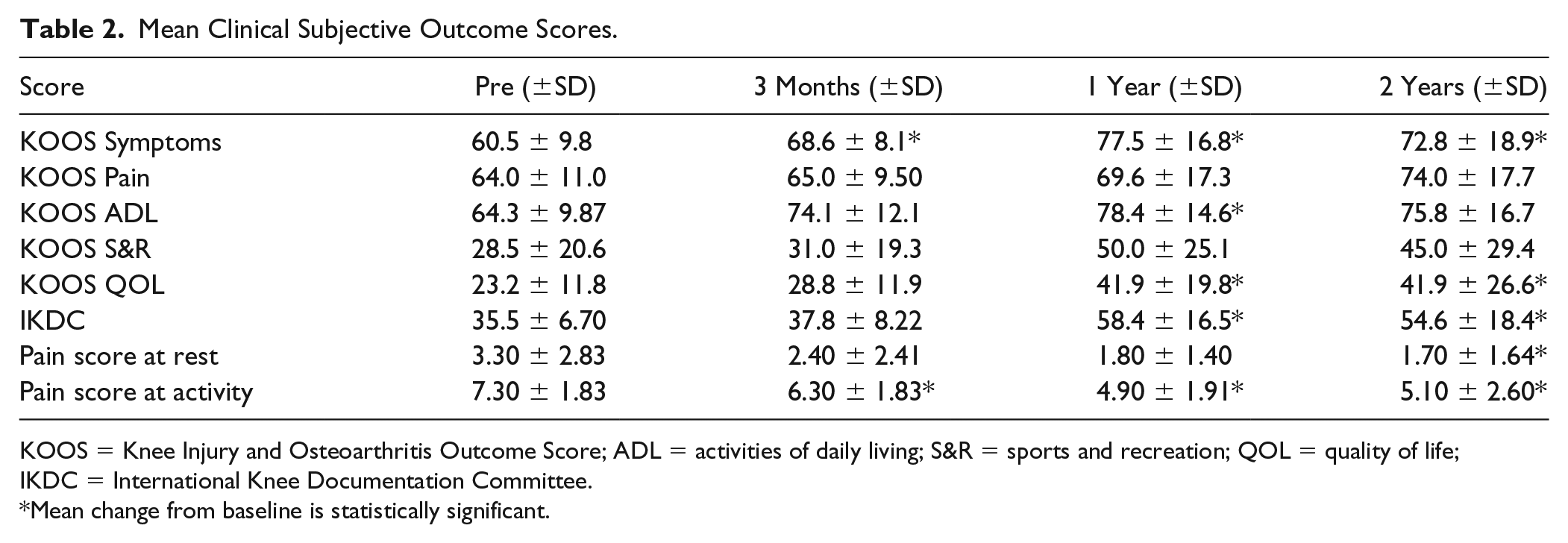
KOOS = Knee Injury and Osteoarthritis Outcome Score; ADL = activities of daily living; S&R = sports and recreation; QOL = quality of life; IKDC = International Knee Documentation Committee.
Mean change from baseline is statistically significant.
At 1 year the following PROMs had improved significantly: IKDC (35.5 to 58.4, P = 0.003), KOOS symptoms (60.5 to 77.5, P = 0.01), KOOS ADL (activities of daily living) (64.3 to 78.4, P = 0.04), and KOOS QOL (quality of life) (23.2 to 41.9, P = 0.04). The pain score during activity also improved significantly (7.3 to 4.9, P = 0.006).
At 2 years the following PROMs had improved significantly: IKDC (35.5 to 54.6, P = 0.009), KOOS symptoms (60.5 to 72.8, P = 0.04), and KOOS QOL (23.2 to 41.9, P = 0.04). The pain scores during rest (3.3 to 1.7, P = 0.02) and activity (7.3 to 5.1, P = 0.007) also improved significantly.
Magnetic Resonance Imaging Evaluation
The pre-operative MOCART score was 17.8 ± 7.2, which improved significantly to 36.3 ± 18.4 after 1 year (P = 0.01). At the latest follow-up the MOCART score was 28 ± 12.1 and demonstrated a tendency for improvement compared with baseline (P = 0.06). In general, patients had no chondral osteophytes and small or no effusion and had the highest scores in these categories. No patients had intact subchondral lamina, subchondral bone, or homogenous structure of the repair tissue and the lowest scores were therefore achieved in these categories.
Second-Look Arthroscopy
Six of 7 second-look arthroscopies showed smooth cartilage in level with the surrounding cartilage and with good integration to the adjacent tissue. Six of 7 patients had normal or nearly normal appearing gross cartilage morphology using arthroscopic ICRS scores ≥8. The average Arthroscopic ICRS score was 8.7 ± 0.9.
Histochemistry, Immunohistochemistry, and Histomorphometry
Safranin O and toluidine blue staining of more than 50% metachromasia was present in 4 of 7 biopsies representing presence of glycosaminoglycans (GAGs). Almost no metachromasia was seen in the remaining 3 biopsies ( Fig. 2 ).
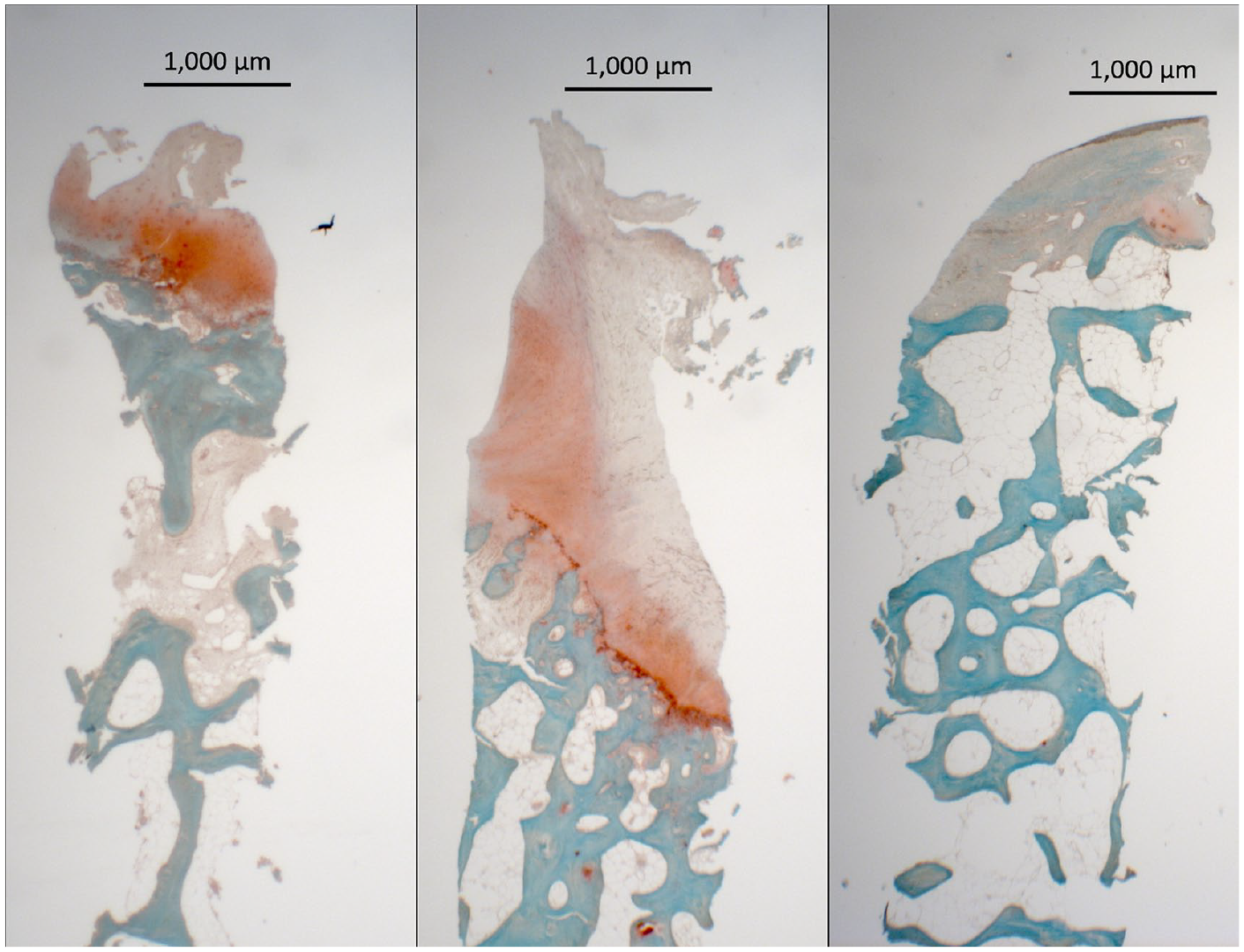
Safranin O–stained biopsy samples representing the best (left), average (middle), and worst (right) outcomes.
Two patients had more than half of the repair tissue staining positively for collagen I. In both patients, 75% to 100% of the repair tissue stained positively for collagen I. No patients had more than 50% positive collagen II staining in the repair tissue ( Fig. 3 ). Collagen II was not systematically distributed in the same area of the biopsies, while collagen I was consistently present in the superficial areas of the biopsies.
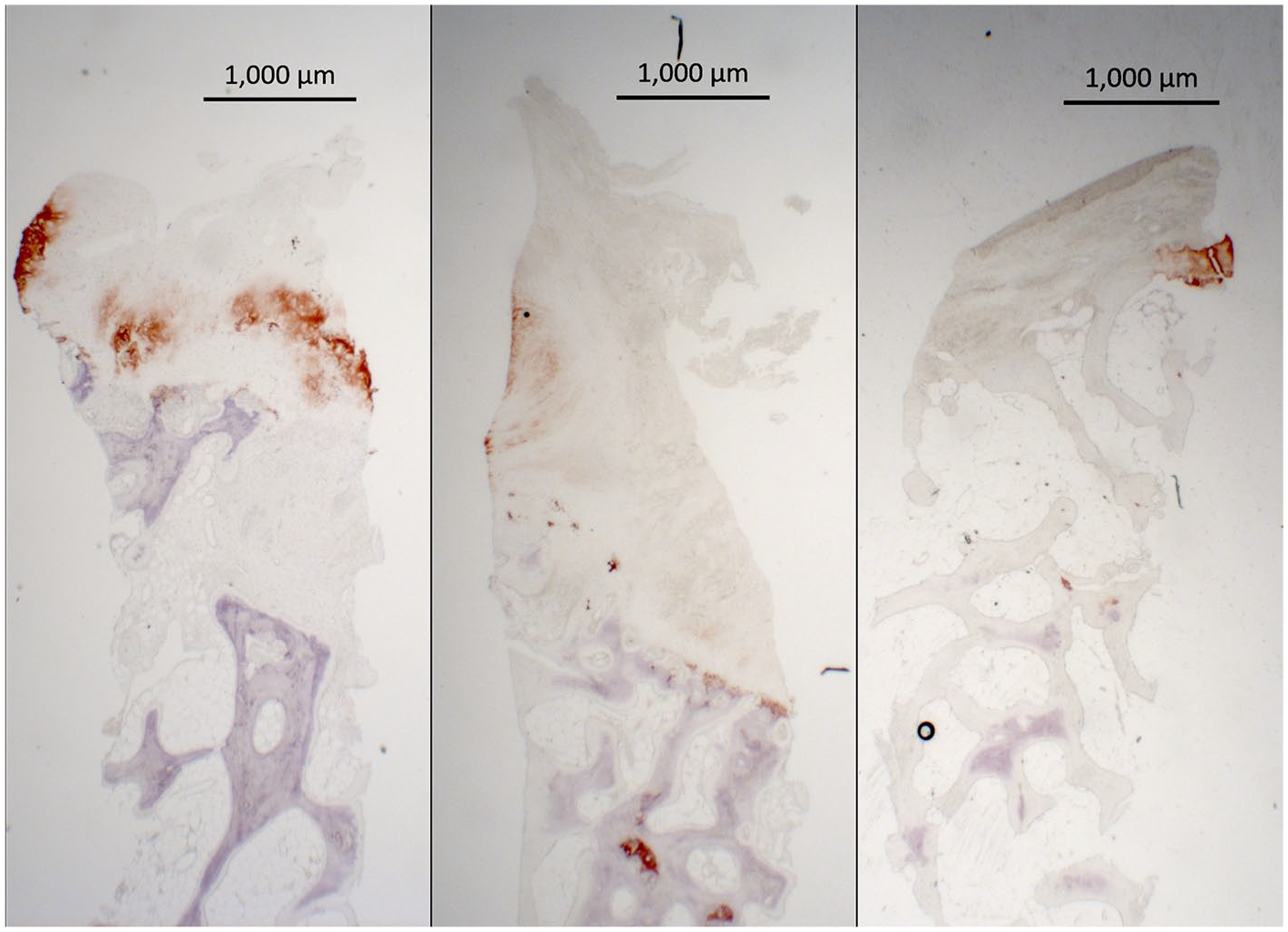
Collagen II stained biopsy samples representing the best (left), average (middle), and worst (right) outcomes.
Biopsies contained on average 1.5% (±3.3%) hyaline tissue, 40.0% (±36.9%) fibrocartilage, and 58.4% (±35.9%) fibrous tissue.
Discussion
The primary findings of the present study were that BMAC and PRP on a collagen I/III scaffold led to an improvement in subjective outcome scores as well as MRI-evaluated biological repair after 1 year. Repair tissue was mainly a mixture of fibrous and fibrocartilaginous tissue.
Improvements were seen in all subjective outcome scores, however not statistically significant for all scores. Using the commonly accepted threshold for the minimal clinically important difference (MCID) of the IKDC of 10 points, 7 out of 10 patients improved over the follow-up period, while the mean improvement in the IKDC exceeds the MCID. 42 Improvements were less marked than what was shown in the few former studies on BMAC for cartilage repair of the knee.3,7,34,42-44
In studies by Gobbi et al., BMAC was also used in combination with a collagen I/III scaffold.3,43 A few differences can, however, be noticed between the studies; no PRP was added and the bone marrow was activated with a batroxobin enzyme (Platelex). In one of Dr. Gobbi’s studies, 6 of the 15 patients (40%) had lesions in their patella, comprising a much lower proportion of patients compared with the present study (70%). These differences in repair stimulation concept could explain some the outcome difference between the present study and the Gobbi study.35,45 However, in another study by Gobbi et al. comparing BMAC in combination with a hyaluronic acid scaffold and MACI, all patients had lesions in their patella or trochlea and this pure patellofemoral cartilage defect cohort had significant improvements in all clinical scores with an increase in mean IKDC from 38.8 ± 19.2 to 82.5 ± 10.7—an increase of 43.7 points—compared to an increase in mean IKDC in our study of 35.5 ± 6.7 to 54.6 ± 18.4—19.1 points. 44 Information about prior surgery and duration of symptoms were not listed in the articles.
Improvements in our study were also lesser than seen in many clinical studies on treatments for cartilage repair. Several factors can have influenced this: (1) large proportion of patella injuries; (2) large proportion of patients with prior surgery, including microfracture; and (3) long duration of symptoms—all factors related to worse outcome after cartilage repair surgery.46-49
Regarding patella lesions, 7 out of 10 patients had lesions in the patella, comprising a larger proportion compared with most other studies done on BMAC for cartilage injuries in the knee. 34 In general, lesions on the patella comprise 20% to 38% of all symptomatic chondral lesions in the knee.50-52 Cartilage injuries in the patella are known to be challenging and have formerly been shown to have worse outcome compared with other lesion sites in the knee.53-57
Regarding prior surgery, 6 out of 10 patients had had prior surgery, which is also known to have negative influence on treatment outcome.48,49 Similar results have been reported with patients undergoing ACI—also a cell-based cartilage repair treatment.48,58 In a study by Pestka et al., a group receiving ACI after they had failed primary microfracture had a mean postoperative IKDC score of 58.4 ± 22.4 compared with 69 ± 19.1 in a group of patients receiving primary ACI. 48 Minas et al. furthermore reported a failure rate of ACI among patients with prior marrow stimulation as high as 26%. 49
Regarding symptom duration all patients included in this study had had symptoms for more than 3 years (average >5 years) before surgery, which have also been shown to have a negative influence on the outcome.46,47 Solheim et al. reported a risk for a poor outcome after knee cartilage repair that was twice as high among patients with symptoms more than 36 months prior to surgery compared with patients with symptoms less than 36 months. 46 Krishnan et al. also reported significantly worse outcome after ACI among patients with a duration of symptoms more than 24 months compared with patients with symptoms less than 24 months. 47
One strength of the present study is that we were able to make a comprehensive follow-up including PROMs, MRI, second-look arthroscopies, and biopsies. We reported second-look arthroscopy data and histological outcome from 7 out of 10 patients available for follow-up. Six of the 7 defects had a normal or near-normal macroscopic appearance using the Arthroscopic ICRS score. Contrary to our hypothesis, repair tissue from biopsies consisted of mainly fibrocartilage and fibrous tissue. Only very small amounts of hyaline cartilage were present. The lack of hyaline cartilage may in part be due to the relatively early biopsies, meaning that some tissue maturation may still occur. Repair tissue quality after cartilage repair surgery has been shown to improve with increasing time from surgery.59,60 In a meta-analysis by DiBartola et al., a correlation between a higher hyaline content and clinical outcome was found. 59 Since hardly any hyaline cartilage was present in our biopsies no correlation between hyaline cartilage and clinical outcome could be seen. Other studies which have included biopsies have reported much larger fractions of the repair tissue being hyaline cartilage compared to our study.59-63 This may in part be due to evaluators not using polarized light when evaluating the biopsies. Failure to use polarized light when determining tissue type may lead to an erroneous characterization of fibrocartilage as hyaline cartilage. 41 The histological composition of the repair tissue in this study seems to resemble the findings after microfracture most in the study by DiBartola et al. 59 Microfracture is also based on a repair response owing to cells from the bone marrow. This treatment may therefore induce a repair tissue similar to that after microfracture while conserving the subchondral bone. As mentioned earlier the damage to the subchondral bone during microfracture has been discussed as potential “bridge-burning,” lowering the chance of successful outcome following revision surgery. 49
For evaluation of the MRI outcome we used the 3D MOCART score. Even though the 3D MOCART score per definition is a score for evaluation of outcome on MRI after cartilage repair, we chose to use a preoperative MOCART score for comparison with the postoperative scores as formerly reported. 64 MOCART scores were relatively low compared with other cartilage repair studies.36,64-66 This is in line with our histological findings showing repair tissue of fibrous and fibrocartilaginous nature—inferior to the native hyaline cartilage. The correlation between histology and MRI is, however, uncertain. Only few studies have correlated findings on MRI with histology, and the evidence of a correlation is conflicting.66-70 As with histology the evidence of correlation between MRI and clinical outcome after cartilage repair is ambiguous.71-75 A recent study, however, found a correlation between MRI at one year and mid- to long-term clinical outcome. 61
Another strength of our study is the pragmatic nature of it. Inclusion criteria were broad compared to most studies on treatments for cartilage injuries. 76 This improves the external validity of our study but may on the other hand have led to a decrease of the improvements seen, as more complex patients were included.
Our study is limited by number of included patients, intermediate follow-up, a lack of control group, different concomitant treatments, and a lack of quantification of BMAC and PRP in terms of total nucleated cells, number of platelets, colony-forming units, and MSCs. However, no studies have shown superiority of one device over another for the preparation of BMAC. 77 There is still a need for more research focusing on which features of the PRP and BMAC are important to improve BMAC for cartilage repair and, in addition, the use of combined PRP and BMAC makes it impossible to distinguish between effects of PRP and BMAC.45,77,78 Theoretically BMAC and PRP may work synergistically, through the ability of PRP to promote cell differentiation and cell metabolism25,27,79; however, there is a lack of studies investigating this effect.80,81 By better characterization of BMAC and PRP it may in the future be possible to correlate specific components of BMAC and PRP to better outcome, which could make it easier to investigate how to optimize BMAC and PRP.
Considering the demographics of our patients, the data from the present study are encouraging for combined BMAC/PRP cartilage repair with confirmation of clinically relevant subjective outcome improvements as well as new MRI and histological data regarding the biological repair response. This 1-step cartilage repair concept remains attractive with low costs compared to other cell-based techniques. There are still several unsolved issues of BMAC cartilage repair such as optimal cell preparation and timing and the efficacy of supplemental products such as PRP. 8 Also, larger, controlled, and long-term studies are needed to validate this treatment option.
In conclusion, treatment of cartilage injuries using combined BMAC and PRP improved subjective clinical outcome scores and pain scores at 1 and 2 years postoperatively. MRI and histology indicated repair tissue inferior to the native hyaline cartilage.
Footnotes
Acknowledgments and Funding
The authors acknowledge the technical assistance of Anette Baatrup and Torsten Grønbech Nielsen. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The local ethics committee under the Danish National Committee on Health Research Ethics approved the protocol (#M-2014-389-14).
Informed Consent
Written informed consent was obtained from all subjects before the study.
