Abstract
Objective
Large articular cartilage defects are a challenge to regenerative surgery. Biomaterial scaffolds might provide valuable support for restoration of articulating surface. The performance of a composite biomaterial scaffold was evaluated in a large porcine cartilage defect.
Design
Cartilage repair capacity of a biomaterial combining recombinant human type III collagen (rhCo) and poly-(
Results
The lateral half of the cartilage defect in the operated groups showed better filling than the medial half. The mean overall macroscopic score for the rhCo-PLA, spontaneous, and nonoperated groups were 5.96 ± 0.33, 4.63 ± 0.42, and 10.98 ± 0.35, respectively. The overall histological appearance of the specimens was predominantly hyaline cartilage in 3 of 9 samples of the rhCo-PLA group, 2 of 8 of the spontaneous group, and 9 of 9 of the nonoperated group.
Conclusions
The use of rhCo-PLA scaffold did not differ from spontaneous healing. The repair was affected by the spatial properties within the defect, as the lateral part of the defect showed better repair than the medial part, probably due to different weightbearing conditions.
Introduction
Traumatic injuries to articular cartilage typically affect physically active, young patients. 1 Cartilage defects have a poor intrinsic repair capacity, and full-thickness cartilage defects are an independent risk factor for future total knee arthroplasty. 2 These defects can be repaired surgically in attempt to alleviate symptoms and improve joint function. Biomaterial scaffolds can be used for augmented cartilage repair to mimic tissue architecture and provide the regenerating tissue with structural support 3 and allow for earlier weightbearing. The benefits of biomaterial scaffolds in cartilage repair were assessed by a recent consensus meeting. The consensus on the use of scaffolds in cartilage defects supports scaffold-based therapies in all chondral and osteochondral knee lesions in the absence of joint instability, malalignment, meniscal deficiency, and osteoarthritis, although chondral scaffolds should not be used for deep osteochondritis dissecans lesions. 4 Articular cartilage consists of mainly water and functions as a shock absorber by allowing for fluid flux through the tissue. This movement of fluid together with mechanical stimulus, created by joint movement, provides nutrition to the tissue and promotes the synthesis of its extracellular matrix compounds and fibril organization.5-7 As mechanical stimulus is crucial for cartilage tissue, early weightbearing after cartilage repair procedures has been increasingly studied. Recent clinical studies have shown that accelerated weightbearing program after a cartilage repair procedure allow for an earlier return to sports without hampering the repair result.8,9
Our research group has developed a novel composite scaffold rhCo-PLA.10,11 It is designed to adapt to altering weightbearing conditions in both chondral and osteochondral defects. Its recombinant human collagen (rhCo) and poly(
The mean defect size in recent clinical studies reporting the results of surgical cartilage repair ranges from 2.3 to 6.1 cm.2,12-14 As the defects requiring cartilage repair are large, the performance of the rhCo-PLA scaffold needs to be tested in a large cartilage defect model. Therefore, this study was undertaken to further challenge the scaffold in a large surgically created defect in a porcine model. Aware of the soft nature of porcine subchondral bone that would increase the challenge, the used defect covered a major part of the weightbearing area of the medial femoral condyle. As the nonoperated contralateral limb in our previous study showed changes related to the surgery itself, 10 a healthy nonoperated control group was included in the present study.
Materials and Methods
Pilot Defect Size Testing
To confirm the size of the largest possible lesion to the weightbearing area of medial femoral condyle, porcine femurs were obtained from an abattoir. An arthrotomy was made through a medial parapatellar approach to expose the medial condyle of the femur. The largest possible defect that would still allow suturing its outer rim was created to the condyle. The oval defect measured 11 × 17 mm (1.5 cm2). A custom-made instrument with the aforementioned measurements was then manufactured.
Scaffold Manufacturing
The rhCo-PLA scaffolds were manufactured as previously described10,11 using recombinant human type III collagen (FibroGen Inc, San Francisco, CA, USA) and poly(
Surgical Procedures and Postoperative Care
The study was authorized by the Finnish Animal Experimentation Board (ESAVI/6113/04.10.07/2015) and conducted according to the ethical guidelines and regulations of the Finnish Act on the Protection of Animals Used for Scientific or Educational Purposes (497/2013) and Government Decree on the Protection of Animals Used for Scientific or Educational Purposes (564/2013). A total of 27 female domestic pigs (Sus scrofa domestica) were included in the study. The pigs were obtained from a local farmer at the age of 4 months. The pigs were randomized into 3 groups (n = 9 in each). The rhCo-PLA group receiving a rhCo-PLA scaffold was compared with spontaneous repair (spontaneous group) and healthy cartilage of nonoperated group. After 2 weeks of acclimatization, pigs in the 2 intervention groups were sedated with medetomidine (0.2 mg/kg, subcutaneously) and ketamine (10 mg/kg, subcutaneously). The pigs were then anesthetized with buprenorfin (0.05 mg/kg, subcutaneously), carprofen (3 mg/kg, subcutaneously), and propofol (3 mg/kg or to effect, intravenously). Preoperative antibiotics of cefuroxime (3 g, intravenously) was administered and general anesthesia was continued with isoflurane 2.5%. The animals were placed on supine position on the operating table. Through a medial parapatellar approach, the joint was opened, and the medial condyle of the femur was exposed. The outer rim of the cartilage defect was created with a custom-made punch. Then all cartilage was carefully removed with a curette. The subchondral bone was left intact so that the bone surface just started bleeding. The wound was closed in layers.
The pigs were housed separately after the surgeries. Antibiotic prophylaxis continued for 3 days postoperatively. Analgesia with carprofen 3 mg orally once a day and buprenorphine 0.01 mg/kg subcutaneously thrice a day was continued for 3 days postoperatively. The pigs were allowed free weightbearing and unrestricted movement immediately after the operations and grouped on day 3 after the surgery.
The nonoperated control group of pigs were housed in groups throughout the study period. No surgical interventions were carried out for this group.
Second Operation
Five weeks after the first surgery, the second operation was carried out. The animals were medicated and anesthetized and the joint was approached as described above. The custom-made instrument was fitted to the previously made defect and all scar and repair tissue was carefully removed with a curette. The subchondral bone was left intact so that the bone surface just started bleeding. For the rhCo-PLA group, the rhCo-PLA scaffold was tailored with the custom-made instrument to fit the lesion diameter. The scaffold was pressed to the bleeding bottom of the defect and secured with sutures. The wound was closed in layers.
For the spontaneous group, the defects were debrided but no repair procedures were carried out, and the wound was closed in layers.
Sacrifice and Sample Collection
After 4 months of follow-up, the pigs were anesthetized and sacrificed with medetomidine (0.2 mg/kg, subcutaneously), ketamine (10 mg/kg, subcutaneously), pentobarbital (60 mg/kg, intravenously), and T-61 (4-5 mL/50 kg). Both knees were opened and any possible excess synovial fluid was carefully assessed and documented. Both the femoral and tibial cartilage were assessed for any pathological changes. The distal femurs of the hind legs were then photographed, where after the medial condyle was detached with a diamond saw and stored at –20 °C in a plastic container in which a swab moistened with Ringer acetate solution was placed on the bottom.
Macroscopical Evaluation
A subjective evaluation of the overall repair covered the entire defect area. Using the International Cartilage Repair Society (ICRS) macroscopic scoring system, 15 the macroscopical repair was evaluated from the lateral half of the defect area in order to match the macroscopical evaluation with the histological ICRS evaluation. The assessment was made from blinded, randomized photographs by 3 independent observers (ES, JP, TP) using a web-based survey (SurveyMonkey, San Mateo, CA, USA). The tissue of the nonoperated control group was assessed from the area corresponding the defect area in the 2 intervention groups.
Differences between the medial and the lateral half of the defects in the rhCo-PLA treatment group and the spontaneously healed group were analyzed. A MatLab (The MathWorks, Inc., Natick, MA, USA) program was developed for the defect side comparison. First, the lateral and medial defect sides were selected from the samples’ RGB (red, green, blue) photographs. Then, the photographs were converted to HSV (hue, saturation, value) color space. Comparison between the lateral and medial defect side was made with the saturation channel by calculating the mean values of the selected regions.
X-Ray Micro-Computed Tomography (µCT)
The frozen condyles were thawed at room temperature for 1 hour in PBSI and then imaged with Zeiss Xradia MicroXCT-400 (Zeiss, Pleasanton, CA, USA) µCT scanner. Before the actual imaging, a scout image was taken from the whole sample. Therefore, it was possible to accurately select the defect area as a field of view. The x-ray tube voltage was adjusted to 120 kV and the tube current to 83 µA. A total of 1,200 projections were taken in 360° rotation angle and 5 seconds exposure time was used. With binning 2, the resulted pixel size was 22.32 µm. Projections were reconstructed to the 3-dimensional image stack with the device manufacturer’s XMReconstructor software. Analysis volume was chosen as a cylinder with a diameter of 15 mm and a height of 6 mm. Bone volume was selected from the background by manual thresholding. The bone structural parameters bone volume fraction, trabecular thickness, and spacing were calculated as described in Palmroth et al. 16 Image processing and analysis were done with the Avizo 2019.3 software (Thermo Fisher Scientific, Waltham, MA, USA) and the BoneJ plug-in 17 in the Fiji software. After the imaging, the samples were refrozen at –20 °C.
Histology
The collected condyle specimens were cut around the defect area and halved from the middle of the defect with a diamond saw. The samples were fixed with 10% buffered formalin for 4 weeks, followed by decalcification in a solution containing 10% EDTA, 4% formalin, and 0.1 M phosphate buffer. The samples were halved and embedded in paraffin. The middle part of the defect was analyzed histologically by cutting the samples containing the lateral half of the defect to 5 µm sections and staining them with safranin-O. Blinded safranin-O-stained sections were evaluated by 3 independent observers (JP, ES, AV) using the ICRS II score. 18
For immunohistochemical evaluation of collagen types I and II, the histological sections were prepared as previously described.10,19 Briefly, the sections were digested with hyaluronidase (2 mg/mL, Sigma-Aldrich) and pronase (2 mg/mL, Calbiochem, Merck KGaA). Endogenous peroxidase activity was blocked with hydrogen peroxide (EnVision+ System-HRP [AEC], Dako North America Inc.) Nonspecific staining was blocked with 10% normal goat serum (Dako Denmark A/S, Glostrup, Denmark). Primary antibodies against collagen type II (ab34712, Abcam, Cambridge, UK) and collagen type I (ab34710) were diluted to 4 μg/mL in PBS supplemented with 1% bovine serum albumin (Sigma-Aldrich) and 0.1% Triton X-100. Horseradish peroxidase (HRP)-conjugated goat anti-rabbit secondary antibody (Dako) was applied. Antibody binding was visualized with AEC (3-amino-9-ethylcarbazole) substrate chromogen (Dako).
Histopathological Evaluation
Samples of the synovial membrane of the right and left stifle joint and popliteal lymph nodes were collected at the time of sacrifice. The samples were fixed in 10% buffered formalin, routinely processed and embedded in paraffin and cut into 5 µm tissue sections. The sections were stained with hematoxylin and eosin and evaluated under light microscope by animal pathologists (Finnish Centre for Laboratory Animal pathology, University of Helsinki, Finland).
Statistical Analyses
Differences between groups were analyzed with bootstrap type generalized estimating equations (GEE) using an exchangeable correlation structure. The weight gain of the animals was analyzed with analysis of covariance with baseline weight as covariate. P values less than 0.05 were considered statistically significant. Statistical difference between the saturation values of the medial and lateral part of the photographed defects were analyzed with t test. The statistical analyses were performed with Stata version 16.0 (StataCorp, College Station, TX, USA). The data are presented as mean ± SE unless otherwise stated.
Results
Animal Experiments
During the study, the animals gained weight from 134.1 ± 2.5 to 217.0 ± 4.5 kg. The rhCo-PLA-treated animals gained weight from 123.4 ± 1.8 to 197.4 ± 4.1 kg (Δ = 74 kg). Spontaneously healed animals gained weight from 140.5 ± 5.2 to 226.5 ± 6.7 kg (Δ = 86 kg). Weight gain for the nonoperated group was from 138.8 ± 4.7 to 228.1 ± 8.5kg (Δ = 89.3 kg). Weight gain did not differ between the groups (P = 0.31).
Between the operations, the defects had shrunken by 1 to 2 mm around the rim of the lesion and there was a deposition of soft, fibrous-like tissue macroscopically. The underlying subchondral bone seemed hardened in clinical evaluation.
One of the pigs in the spontaneous group developed a malignant hyperthermia, a condition typical for domestic pigs, and was euthanized on the second postoperative day of the second operation. Otherwise, the animals recovered well from both surgeries. One of the pigs in the rhCo-PLA treatment group showed a joint infection of unknown etiology at the time of the sacrifice but was still included in the analyses.
Macroscopic Evaluation
The repair tissue showed an uneven quality even within the same defect. It was typical for both intervention groups that the lateral edge of the large defect showed good filling but the medial half presented with poor repair ( Fig. 1 , Supplemental Figure S1). None of the defects in either intervention group showed good filling on the medial edge of the defect if the lateral side was poorly repaired. The repair tissue was in level with the surrounding cartilage in 3 of 9 samples in the rhCo-PLA group, in 1 of 8 samples in the spontaneous group, and in 9 of 9 samples in the nonoperated control group.
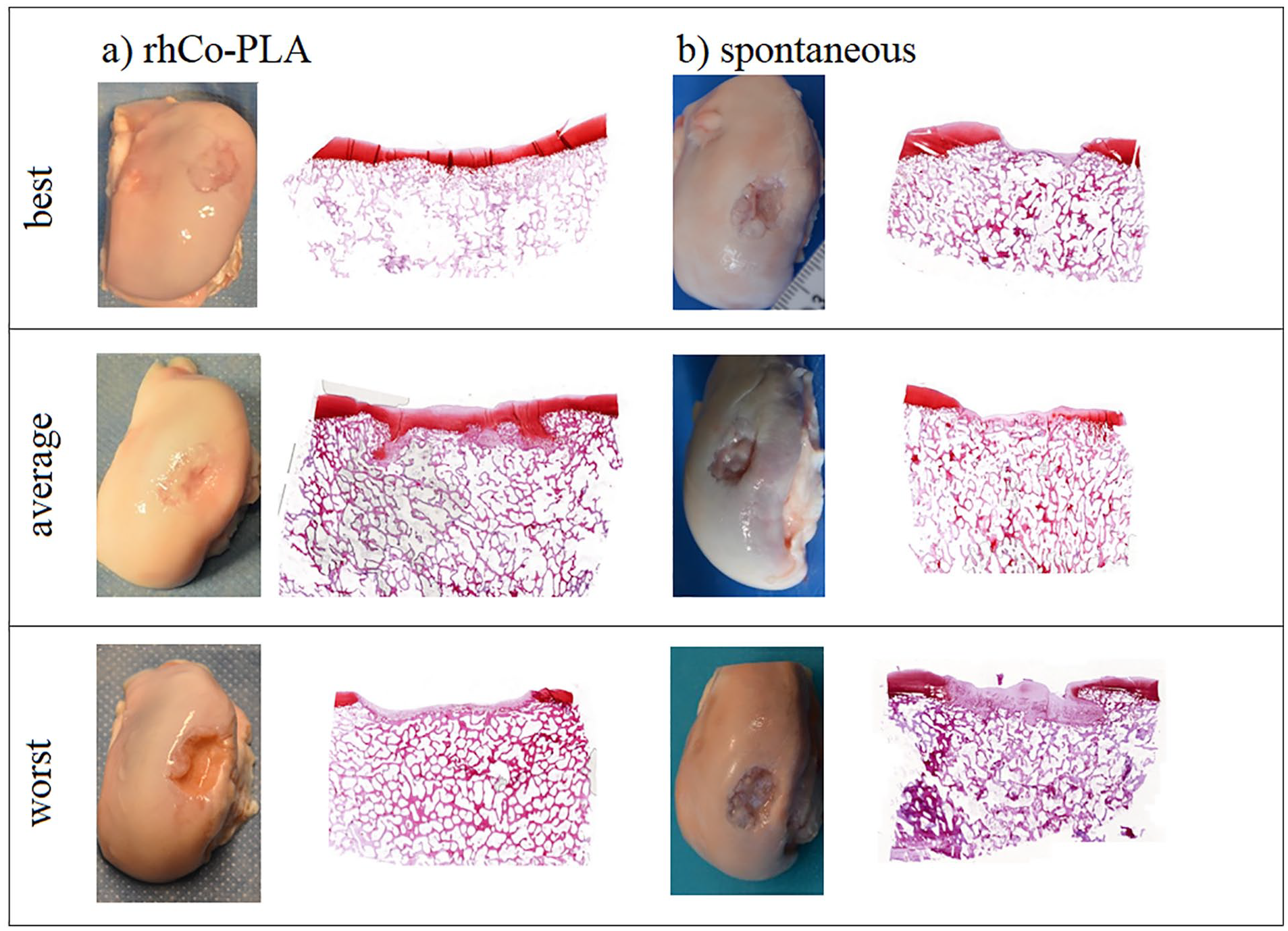
Photographs of the medial condyles of the femur and safranin-O-stained tissue sections of the middle part of the defect showing the best, average, and the worst samples (
The mean overall ICRS score for the rhCo-PLA group was 5.96 ± 0.33 (corresponding to ICRS grade III), for spontaneous group 4.63 ± 0.42 (grade III), and for healthy nonoperated group 10.98 ± 0.35 (grade II). The results showed that the healthy nonoperated group received higher scores than the 2 intervention groups. There was a statistically significant difference in the total ICRS score between the groups and the difference was located between the rhCo-PLA group and the healthy nonoperated group, and between the spontaneous group and the healthy nonoperated group (P < 0.0001) ( Fig. 2 ). Otherwise, no statistically significant differences were detected.
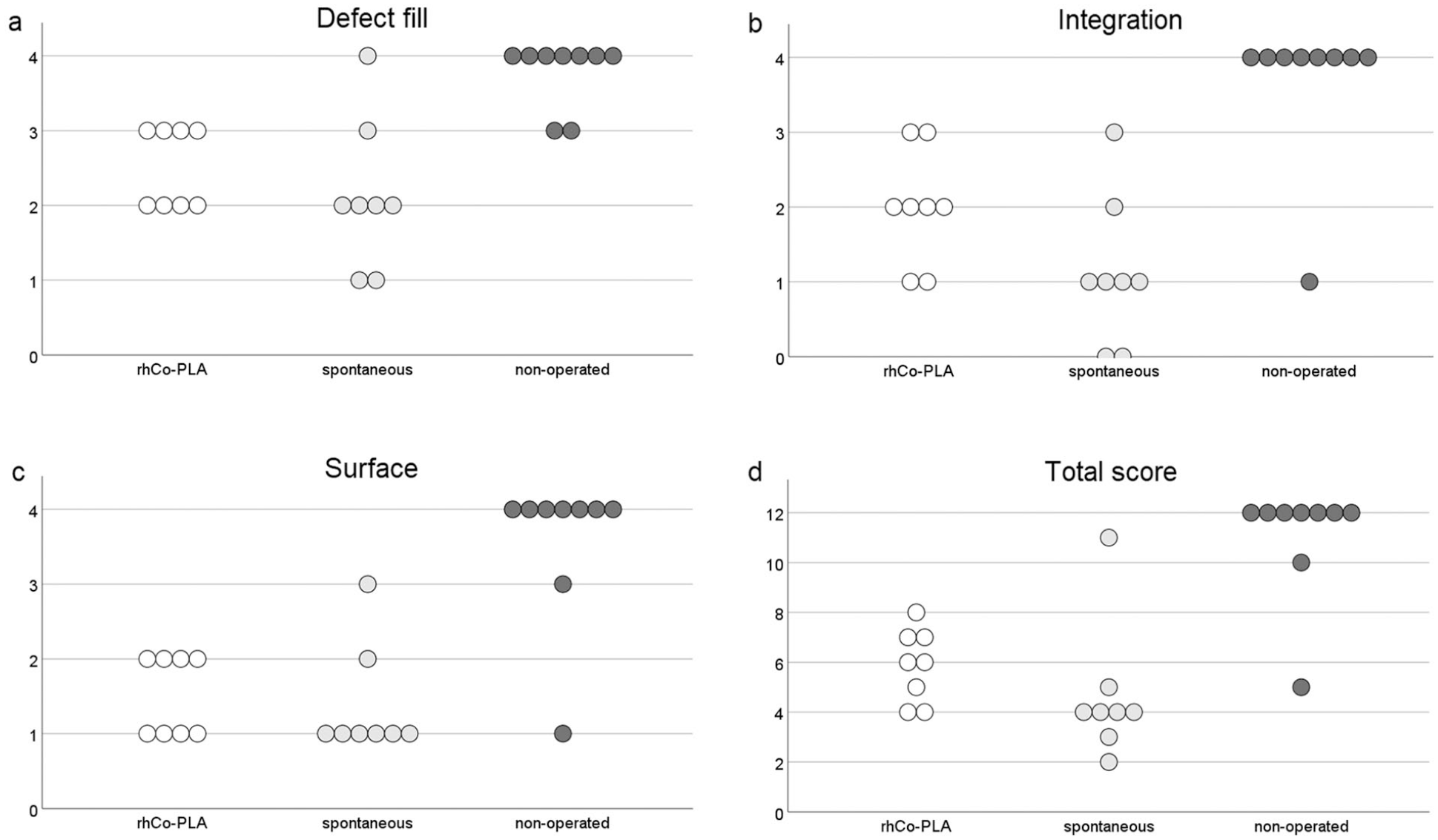
The International Cartilage Repair Society (ICRS) macroscopic score
15
showed large variation between individual animals. The subscores of (
The medial halves of the defect areas showed higher HSV saturation values when compared with the lateral halves ( Fig. 3 ). The mean saturation value in the rhCo-PLA group was 0.49 for the lateral part and 0.57 for the medial part (p = 0.025). The mean saturation value in the spontaneous group was 0.28 for the lateral part and 0.37 for the medial part (p = 0.003).
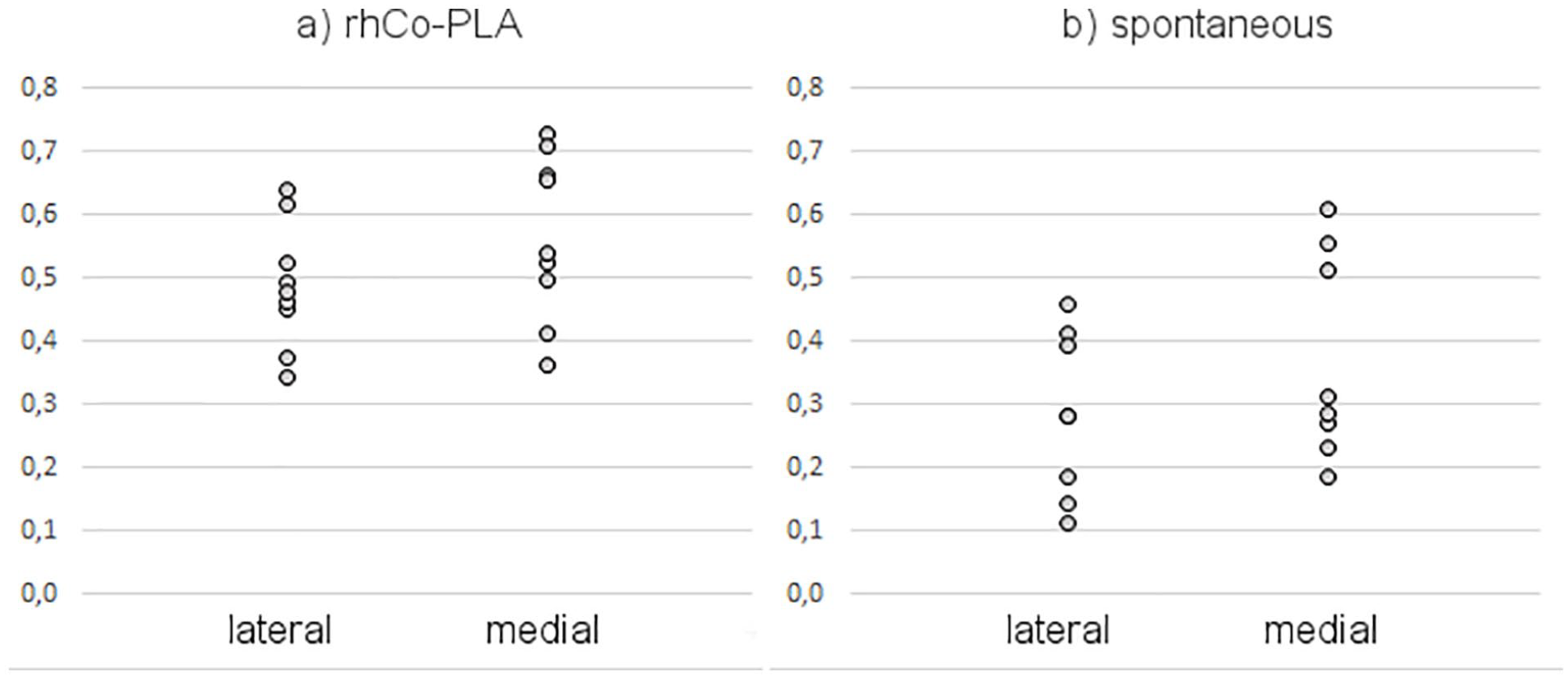
The hue–saturation–values were higher in the medial halves of the defects in both (
Tissue Structure
Safranin-O staining was moderate to good in 4 of 9 specimens in the rhCo-PLA group, in 4 of 8 specimens in the spontaneous group, and in all the specimens in the nonoperated group. The same well-stained samples showed good type II collagen uptake in immunohistochemical evaluation. The type II collagen was found in the pericellular region and was more abundant in the perimeter of the defect than in the middle ( Fig. 4 ). The overall histological appearance of the specimens was predominantly hyaline cartilage in 3 of 9 samples of the rhCo-PLA group, 2 of 8 samples of the spontaneous group, and 9 of 9 samples of the nonoperated group. The rest of the samples in the 2 intervention groups showed predominantly fibrocartilaginous and fibrous repair tissue. Fibrillation and delamination of the cartilage surface was common. The tissue type in the nonoperated control group was hyaline cartilage in all animals. Typically for porcine cartilage, none of the specimens had a tidemark ( Fig. 1 , Table 1 , Supplemental Figure S1).
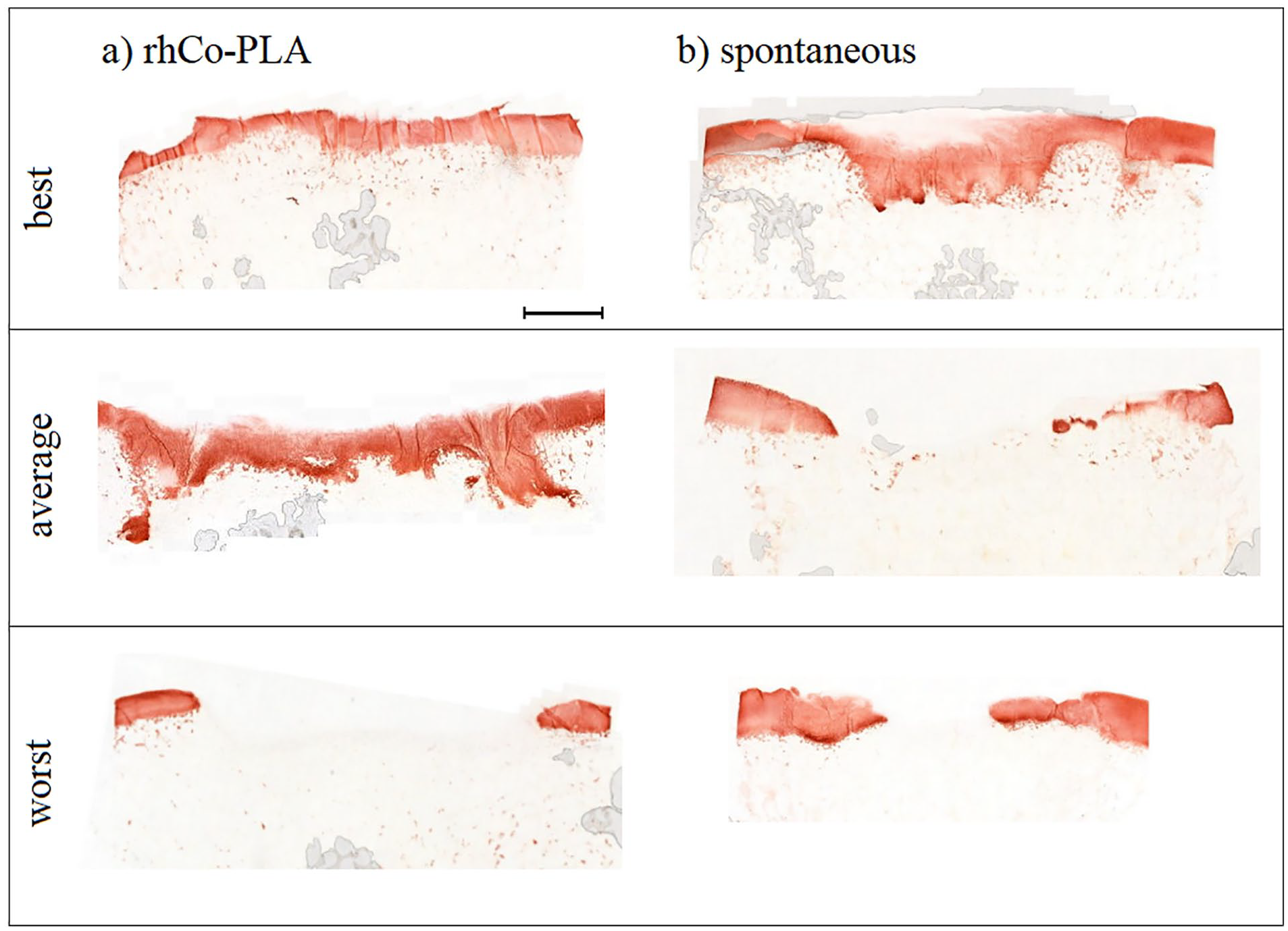
Comparison of immunohistochemical type II collagen staining in best, average, and worst samples in the (
Results of the ICRS II Histological Score 18 for Each Study Group.
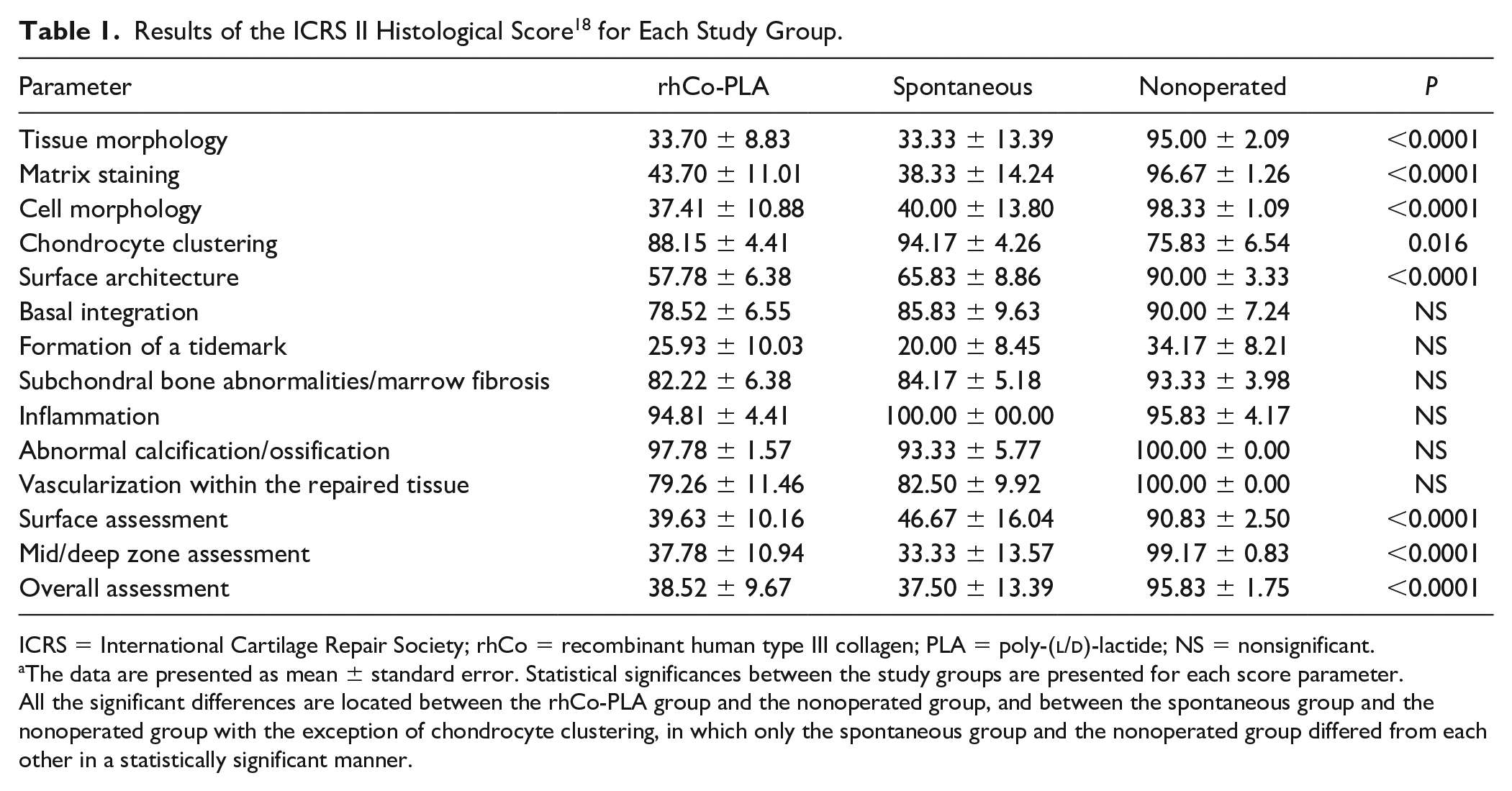
ICRS = International Cartilage Repair Society; rhCo = recombinant human type III collagen; PLA = poly-(
The data are presented as mean ± standard error. Statistical significances between the study groups are presented for each score parameter. All the significant differences are located between the rhCo-PLA group and the nonoperated group, and between the spontaneous group and the nonoperated group with the exception of chondrocyte clustering, in which only the spontaneous group and the nonoperated group differed from each other in a statistically significant manner.
The PLA fibers of the rhCo-PLA scaffold were detected in histological evaluation of all the rhCo-PLA-treated animals, confirming that the scaffolds stayed on place. Histologically, only one sample in the rhCo-PLA group showed a small cyst-like defect in the subchondral bone. Otherwise, no subchondral bone defects were seen in the histological sections of any of the studied groups.
The ICRS II histological score revealed that the healthy nonoperated group received higher score than the 2 intervention groups in all subscores, except for cell clustering and inflammation ( Table 1 ). The rhCo-PLA group and the spontaneous group differed in a statistically significant manner from the nonoperated group in tissue morphology, matrix staining, cell morphology, surface architecture, superficial assessment, middle/deep zone assessment, and overall score (P < 0.0001 for each parameter). Chondrocyte clustering showed a difference between the spontaneous group and the healthy nonoperated group (P = 0.016).
Histopathological Evaluation
The synovium samples exhibited minimal or mild synovitis with multifocal infiltration of lymphocytes and plasma cells in the subintima in 9 of 9 animals in the rhCo-PLA group, in 4 of 9 animals in the spontaneous group, and in 0 of 9 animals in the nonoperated group. Foreign material resembling suture material was observed in three pigs in the rhCo-PLA group. Additionally, foreign material particles were observed in 1 pig in the spontaneous group and 1 in the nonoperated group. One pig in the nonoperated control group had a cyst consisting of organized hemorrhage in the meniscus, likely originating from trauma. No other pathological findings related to the rhCo-PLA scaffolds were observed in the lymph nodes or synovium of these animals.
Minimal or mild reactive follicular hyperplasia of the lymph nodes, which is a frequent and usual finding in young animals, was seen in all samples with the exception of 2 animals in the nonoperated group. In addition, all samples exhibited a mild to moderate infiltration of eosinophils and occasional yellow to brown pigmented granular material (hemosiderin) in the medullary sinuses.
Subchondral Bone
The average size of the subchondral bone defects was slightly larger in the spontaneously healed group (74.9 ± 30.1 mm3) than in the rhCo-PLA group (60.8 ± 51.8 mm3), although the difference was not statistically significant ( Fig. 5 ). The 3 study groups did not show statistically significant differences in the bone structural parameters of trabecular thickness, trabecular separation of bone volume fraction.
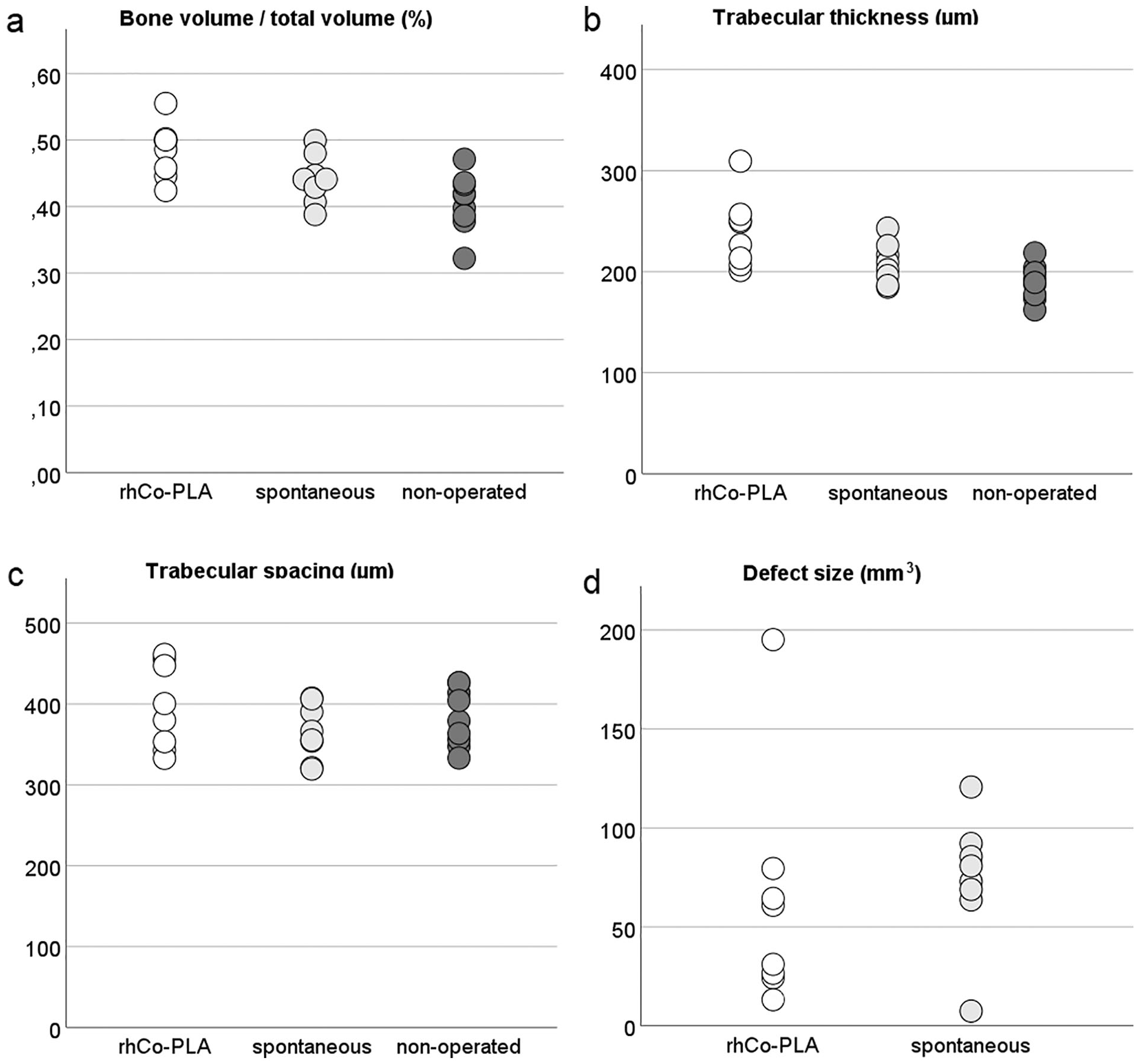
Results of the x-ray micro-computed tomography (µCT) showing (
Discussion
The results of this study show that the lateral part of the medial condyle of the femur has a better healing capacity than the medial part of the condyle. This is probably due to the weightbearing conditions. In both intervention groups, the lateral part of the defect within the medial condyle of the femur showed statistically better repair than the medial part, according to macroscopic analyses. The mechanical stimulus from weight bearing is considered an important factor in cartilage anabolism. Mechanical stimulus improves chondrocyte viability 20 and cartilage extracellular matrix deposition 21 in vivo and ex vivo. The lateral part of the medial condyle bears weight 22 and might therefore have an improved cartilage repair capacity. The rhCo-PLA scaffold has shown weight-adaptive properties, 3 which provides the regenerating tissue with mechanical cues and potentially improves its performance in comparison with other biomaterial scaffolds.
The use of an animal model allows for standardizing the test group as well as the location and the size of the defect, enabling an effective comparison of the 2 intervention groups. The porcine model was chosen as it has, together with the equine model, thick cartilage giving the closest approximation to human cartilage. 23 As seen in our previous study, the nonoperated contralateral limb, which is often used as a control, is also affected by the surgeries. 10 Therefore, it does not represent a normal, healthy porcine cartilage. In this study, a healthy nonoperated group to which no interventions were done was used as a control. Interestingly, not even the healthy group received grade I ICRS macroscopic score, indicative of normal cartilage, suggesting that spontaneous cartilage defects in domestic pigs are common. These animals are bred to gain muscle mass quickly, and they are typically slaughtered at the age of 4 months. The animals used in this study were of 8 months of age at the time of sacrifice, weighing on average 217 kg, which puts a heavy strain on their joints. The healthy, nonoperated group gave valuable information on porcine cartilage and subchondral bone. This study benchmarks normal tissue in porcine cartilage repair studies and can thus later be used as a reference in evaluating repair results in later experiments.
Previous experiments have shown subchondral bone pathologies when repairing porcine cartilage with biomaterial scaffolds.10,24,25 Due to the soft nature of the subchondral bone of domestic pigs, osteochondral—and even chondral—repair in this model is difficult, and sinking of the scaffolds common. In this study, despite the small subchondral bone voids in the 2 intervention groups, the trabecular thickness and spacing of the subchondral bone did not show statistically significant differences from normal porcine subchondral bone. The rhCo-PLA implants did not show signs of detachment or sinking into the bone.
Young animals typically have the capacity of spontaneous cartilage healing, 26 but it has been stated that porcine cartilage does not regenerate in large defects. 23 Although neither of the intervention groups showed repair tissue completely similar to healthy cartilage, the relatively short follow-up period of 4 months produced partial repair in both study groups. The use of the rhCo-PLA scaffold did not hamper the repair capacity of the young porcine cartilage. The rhCo-PLA and the spontaneous groups did not differ from one another when the repair tissue was evaluated with ICRS macroscopic score or ICRS II histological score.
Foreign material, resembling suture material, was found in the synovium of 3 animals in the rhCo-PLA group. Foreign material was also found in 1 pig in the spontaneous group and 1 in the nonoperated group. Suture material used in the 2 intervention groups could explain the foreign material found in their synovium. One animal in the spontaneously healed group showed presence of foreign material surrounded or phagocytized by macrophages in the synovium and joint capsule. Surprisingly, 1 animal in the nonoperated group also exhibited foreign material in the synovium, without a cellular reaction. Although foreign bodies in the knee joint are uncommon, there are case reports that describe foreign bodies migrating into the knee joint after a trauma.27,28 This might be the case with this one animal, although the origin of the particle remains unknown. No other pathological findings related to the rhCo-PLA scaffolds were observed in the lymph nodes or synovium of these animals. The lymph nodes of the animals in all groups showed minimal or mild follicular hyperplasia. As this was seen also in the healthy, nonoperated group, this finding lacks clinical significance. However, the synovium of the operated animals exhibited signs of inflammation. The mild synovitis was seen in all the rhCo-PLA-treated animals and in 4 of 9 of the spontaneously healed animals. The inflammation might be associated with the operation itself, the materials used in the operation, or developing osteoarthritis.29,30
Bioabsorbable aliphatic polyesters, such as poly(lactic-co-glycolic acid) (PLGA) and PLA, are widely used in medicine due to their favorable properties. Their applications include suture materials, drug delivery, and bone screws, as well as biomaterial scaffolds intended for cartilage repair. The wounds of both groups were closed with polyglactin 910 sutures, which are made of a copolymer composed of 90% glycolide and 10%
The heterogeneity of the repair tissue between individual animals and even in the same defect creates a challenge for the evaluation of the defects. This issue was managed by focusing the assessment to the middle part of the defect both in macroscopical and histological scoring. There was large variation in the repair results between individual animals but mostly, the variation seen in the results was due to the spatial variations in the repair capacity within each defect. Further studies are needed to confirm whether this is due to different weightbearing conditions.
Conclusions
In this study of a large full-thickness articular cartilage defect in a porcine model, the use of rhCo-PLA scaffold did not differ from spontaneous healing. Both intervention groups showed inferior cartilage quality compared with healthy nonoperated porcine cartilage. The lateral side of the medial condyle of the femur showed good cartilage repair in both spontaneously healed and rhCo-PLA-augmented groups. Thus, the weightbearing conditions may be of high importance when repairing articular cartilage.
Supplemental Material
sj-tif-1-car-10.1177_19476035211030988 – Supplemental material for Cartilage Repair Capacity within a Single Full-Thickness Chondral Defect in a Porcine Autologous Matrix-Induced Chondrogenesis Model Is Affected by the Location within the Defect
Supplemental material, sj-tif-1-car-10.1177_19476035211030988 for Cartilage Repair Capacity within a Single Full-Thickness Chondral Defect in a Porcine Autologous Matrix-Induced Chondrogenesis Model Is Affected by the Location within the Defect by E. Salonius, A. Meller, T. Paatela, A. Vasara, J. Puhakka, M. Hannula, A.-M. Haaparanta, I. Kiviranta and V. Muhonen in CARTILAGE
Footnotes
Acknowledgements
The authors wish to thank Hannu Kautiainen (Medcare Oy) for his expertise in the statistical analyses and prof. emer. Satu Sankari) at the Finnish Centre for Laboratory Animal Pathology (FCLAP) for her histopathological expertise. Histological images were generated using 3DHISTECH Pannoramic 250 FLASH II digital slide scanner at Genome Biology Unit supported by HiLIFE and the Faculty of Medicine, University of Helsinki, and Biocenter Finland.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A-MH and VM are cofounders, board members, and major shareholders in Askel Healthcare Ltd, a spin-off from Tampere University of Technology and University of Helsinki, aiming to commercialize COPLA cartilage repair technology based on rhCol-PLA scaffold studied in this article. AV is a shareholder in Askel Healthcare Ltd. The other authors have no conflicts of interest to declare.
Ethical Approval
The study was authorized by the Finnish Animal Experimentation Board (ESAVI/6113/04.10.07/2015).
Animal Welfare
This study was conducted according to the ethical guidelines and regulations of the Finnish Act on the Protection of Animals Used for Scientific or Educational Purposes (497/2013) and Government Decree on the Protection of Animals Used for Scientific or Educational Purposes (564/2013).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was conducted with the financial support of Tekes (grant 40171/14), Helsinki University Hospital (grant TYH2015207), and Novo Nordisk Foundation Exploratory pre-seed grant No. 17196. The sponsors of this study had no role in the study design, collection, analysis, or interpretation of the data; in the writing of the manucript; or in the decision to submit the manuscript for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
