Abstract
Objective:
Case reports of severe acute localized reactions (SALR) following intraarticular (IA) hyaluronic acid (HA) injections for knee osteoarthritis (OA) have been described. We compared surrogate SALR measures between patients using hylan G-F 20 and specific non-hylan G-F 20 HA products.
Design:
Knee OA patients were identified from the Optum Clinformatics dataset (January 2006 to June 2016), stratified into hylan G-F 20 and non-hylan G-F 20 HA users, matched by single or multiple injection products. Occurrences of surrogate SALR measures including inflammation/infection, intraarticular corticosteroid (CS) injections, arthrocentesis/aspiration, arthrotomy/incision and drainage, and arthroscopy were evaluated within 3 days post-HA.
Results:
Based on 694,404 HA injections, inflammation/infection rate was rare within 3 days of HA (up to 0.03%), with no statistical differences between hylan G-F 20 and non-hylan G-F 20 groups (matched by single or multiple injection products). The risk of knee arthrotomy/incision and drainage, arthroscopy, or arthrocentesis for hylan G-F 20 (2 mL) 3 weekly injection patients was lower than Hyalgan/Supartz and Orthovisc patients, but greater than Euflexxa patients. Overall, we found that Hylan G-F 20 (2 mL) 3 weekly injection had lower SALR rates compared to Hyalgan/Supartz and Orthovisc. However, Hylan G-F 20 (2 mL) 3 weekly injection had slightly higher rates of SALR when compared to Euflexxa. Among the single injection products, Hylan G-F 20 (6 mL) single injection had lower rates of SALR than Monovisc and Gel-One.
Conclusions:
This study shows no clear correlation between avian-derived or cross-linked products and SALR and provides evidence against avian-derived products or crosslinking as a source for these reactions.
Introduction
Although the safety of intraarticular hyaluronic acid (HA) has been demonstrated for knee osteoarthritis (OA), 1 case reports of subsequent local reactions have been described.2-22 These patients usually demonstrate symptoms from several hours to 72 hours after the HA injection, though some occur up to 6 days later.2-4,6,8,9,12-14,17-19 The symptoms may include severe pain, hot and/or swollen joint, effusion, and loss of function.2-4,8,9,12-15,17,19 The affected patients may also have similar clinical presentation as infectious arthritis, with blood test results that show generally high C-reactive protein and sedimentation levels.3,17 Along with the variation in the clinical presentations of these patients, the reactions have also been described inconsistently as inflammatory flares, 13 septic arthritis,2,17,20 acute pseudoseptic arthritis,3,8,16,18 pseudosepsis or severe acute inflammatory reaction, 6 acute local reaction,9,14,21 inflammatory reaction, 12 aseptic acute arthritis, 4 pseudogout, 23 acute calcium pyrophosphate dihydrate arthritis, 22 and systemic reaction. 15 Some of the reactions are typically mild and resolve without treatment or with local therapy, but other reactions can include severe inflammation and significant pain, which require clinical intervention, such as arthrocentesis, intraarticular steroid injection, and nonsteroidal anti-inflammatory drugs (NSAIDs). 6
Certain HA products, such as high-molecular-weight, crosslinked HAs, have been implicated in having greater risk of acute reactions. 1 The reactions have also been speculated to be possibly related to the crosslink of hylan or an allergic reaction to avian-derived hyaluronan,5,6 but this is debatable because similar reactions have been reported for non-crosslinked, non-animal, and/or naturally derived HA.3,8,10,16,18 Additionally, the published literature has produced mixed findings for product comparisons. A meta-analysis reported that hylan patients had about twice the risk of experiencing flares and joint effusions compared to patients treated with “standard” HA. 24 The true incidence of local reactions for individual HA products is unknown because most of the data have been limited to case reports. In claims database study of almost 750,000 HA patients, the diagnosis of inflammation or infection within 3 days of a HA injection was extremely rare for both hylan G-F 20 (0.001%) and non-hylan G-F 20 (0.002%) HA patients. 25 The study also concluded that the overall risk of surrogate severe acute localized reaction measures was similar for hylan G-F 20 and non-hylan G-F 20 HA patients. However, the non-hylan G-F 20 HA group was not stratified based on the specific HA product that was used, thus not allowing any further comparisons with hylan G-F 20.
With conflicting reports regarding the incidence of these reactions, termed herein as severe acute localized reactions (SALR), after intraarticular hylan and non-hylan HA use, the objective of the current study was to compare the risk of surrogate SALR outcomes between knee OA patients who used hylan G-F 20 and specific non-hylan G-F 20 HA, based on whether the products were single or multiple injection therapies, avian or non-avian derived, and crosslinked or non-crosslinked.
Methods
The Optum Clinformatics (Eden Prairie, MN) database from January 2006 until the end of the second quarter of 2016 was used as the data source for this study. This US dataset is compiled from medical claims from all 50 states for approximately 13 million lives annually, who are covered by UnitedHealth Group (commercial/private payer). Patient-level anonymized data in the dataset is integrated from physician, facility, and pharmacy claims, collected through affiliated health plans, Optum employer customer health plans, and Optum payer customer health plans. The dataset provides various data elements, such as demographics (age, gender), procedure codes, diagnoses codes, admission and discharge dates, and payments. This data are publicly available for purchase and is exempt from institutional review board approval. The study was designed to evaluate a 10-year period with 2016 being the most recent data available at the initiation of the study. Knee OA and nonspecific OA with knee pain were used to identify the study cohort of knee OA patients (Suppl. Table S1). To identify the first diagnosis of knee OA, a look-back period of 6 months with no previous knee OA diagnosis was used. Consequently, those without at least 6 months of prior claim history were excluded. Patients younger than 18 years old, those who had HA treatment prior to the knee OA diagnosis, and those without at least 6 months of follow-up following knee OA diagnosis were also excluded. The patients who underwent at least 1 treatment of HA were selected from the knee OA cohort, based on the Healthcare Common Procedure Coding System (HCPCS) codes for HA (Suppl. Table S1). The HA patients were then stratified into hylan G-F 20 and specific non-hylan G-F 20 HA cohorts. The Hylan G-F 20 cohort was further stratified into those who only used Hylan G-F 20 (2 mL) 3 weekly injections and those who only used Hylan G-F 20 (6 mL) single injection. Because Hylan G-F 20 (2 mL) 3 weekly injection and Hylan G-F 20 (6 mL) single injection utilize the same HCPCS code, the treatment date, national drug code, brand name, number of units, number of injections, and charge difference were used to differentiate between the 2 products. For the remaining patients who received either multiple types of HAs or only 1 type of non-hylan G-F 20 HA during the study period, the type of non-hylan G-F 20 injections was identified based on their HCPCS codes. Patient data used for this study were de-identified. The use of such data is considered exempt from the Institutional Review Board oversight according to Health Insurance Portability and Accountability Act.
The occurrence of surrogate SALR outcomes or clinical encounters was evaluated for the HA patients within 3 days of each HA use. Surrogate outcomes included office visit, emergency room (ER) visit, urgent care visit, intraarticular corticosteroid (CS) injection, arthrocentesis/aspiration (inpatient and outpatient), knee arthrotomy/incision and drainage (inpatient and outpatient), knee arthroscopy (inpatient and outpatient), and diagnosis of any inflammatory response/infection (Suppl. Table S1). ER visit was included, along with other facility visits, due to previous reports of patients who presented to the ER after they had reactions following HA.12,17 CS injections3,6,9,12-15 and arthrocentesis/aspiration6,9,12,14 are used in the management of HA patients with reactions; thus, these were included as outcomes. Because surgical interventions, such as lavage 16 and incision and drainage, 20 are possible treatment options, arthrotomy/incision and drainage and arthroscopy were also examined. All of the above outcomes or clinical encounters were evaluated when considering claims records that have a corresponding knee OA diagnosis (knee OA related) to those that do not. The requisite of a knee OA diagnosis on the claims records for those outcomes or clinical encounters was to restrict them to being likely related to the knee. However, since claims data were used for this study, a sensitivity analysis was also performed in which the outcomes or clinical encounters did not require a knee OA diagnosis on those corresponding claims data (i.e., with any diagnosis). Thus, any occurrence of the specified outcomes or visits was included in the event of miscoding or lack of knee OA diagnosis coding, even though the outcome or visit was due to the knee.
The surrogate SALR outcomes were compared between Hylan G-F 20 (2 mL) 3 weekly injection or Hylan G-F 20 (6 mL) single injection versus non-hylan G-F 20 HA products, based on whether those products were intended for one injection or multiple injection treatment regimens. Specifically, the outcomes were compared for Hylan G-F 20 (2 mL) 3 weekly injection to Hyalgan/Supartz, Euflexxa, and Orthovisc, while Hylan G-F 20 (6 mL) single injection was compared to Gel-One and Monovisc. A logistic regression model was used to compare the risk of the surrogate SALR outcomes, adjusting for various patient demographics, comorbidities, and other potential confounding clinical factors (SAS, SAS Institute Inc., Cary, NC). A P value of less than or equal to 0.05 was used to determine statistical significance. The patient factors included age, race, census region, and gender, while comorbidities were assessed using the composite Charlson score. Potential confounding clinical factors included (1) use of CS injection during the HA injection; (2) prior use of CS; (3) prior use of knee arthroscopy; (4) use of fluoroscopic/ultrasound imaging during HA injection; (5) physician HA experience/volume in terms of total number of any HA injections; (6) hylan G-F 20 physician experience/volume in terms of total number of hylan G-F 20 injections; (7) use of NSAIDs; (8) use of opioids; (9) use of physical therapy (PT); and (10) year. Prior use of CS or knee arthroscopy were considered within 1 week or 12 months before HA, while use of NSAIDs, opioids, or PT were considered within 12 months before HA. Same time and prior use of CS injection with HA was considered as a potential confounder, as it has been reported that clinicians may inject CS followed by HA 1 week apart to avoid the risk of pseudoseptic arthritis. 26 Moreover, reactions have also been reported following CS injections.17,20 The prior use of NSAIDs and opioids (Suppl. Table S1) required a prescription fill within 7 days following a knee-OA-related office visit to be considered as a knee-OA-related pharmacy claim.
Results
A total of 694,404 HA injections were included in the study, with Hyalgan/Supartz injections representing 35.8%, followed by Euflexxa (24.1%) and Orthovisc (17.0%) ( Table 1 ). Hylan G-F 20 (2 mL) 3 weekly injection and Hylan G-F 20 (6 mL) single injection comprised 15.6% and 6.8% of the HA injections, respectively, while Gel-One (0.5%) and Monovisc (0.2%) each comprised less than 1% of the injections. HA use was more frequent in patients who were 65 to 69 year old, female, white, with Charlson score of 0, and in the South region. Office visits within 3 days following the HA injection was the most common clinical encounter that was examined in the present study, with about 10% to 15% of the patients returning to the physician office ( Figs. 1 and 2 ). When limited to office visits with a knee OA diagnosis, the frequency decreased to about 3% to 6%.
Patient Characteristics.
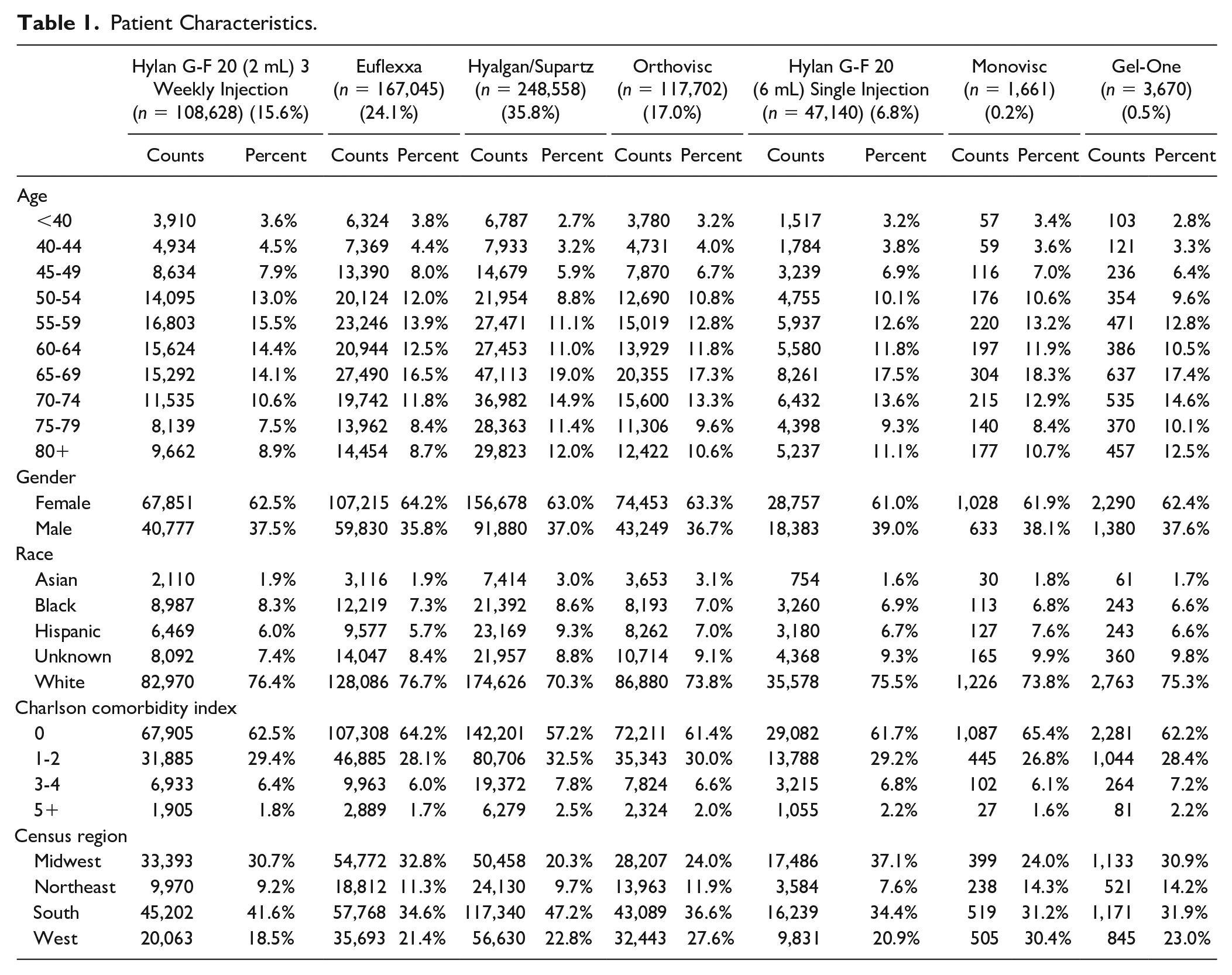
Surrogate SALR outcomes within 3 days post-injection for the Hylan G-F 20 (2 mL) 3 weekly injection, Euflexxa, Hyalgan/Supartz, and Orthovisc patient groups (top: outcomes with any diagnosis on the claims; bottom: outcomes with knee OA diagnosis on the claims). Statistically significant differences are indicated with an asterisk (*P < 0.05; green and red for lower and greater adjusted risks in the Hylan G-F 20 (2 mL) 3 weekly injection group, respectively). SALR = severe acute localized reaction; OA = osteoarthritis; ER = emergency room; CS = corticosteroid; I&D = incision and drainage.
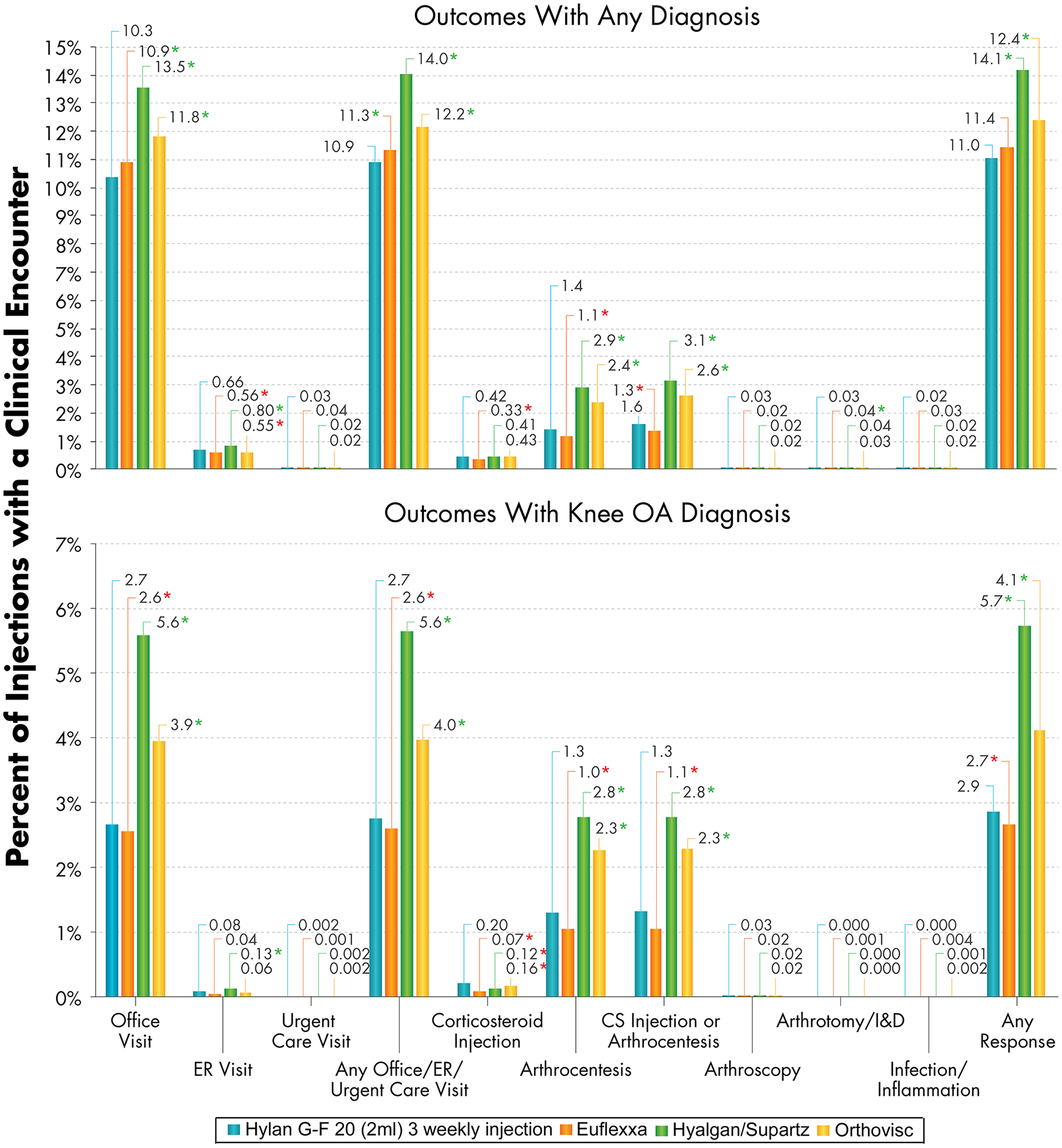
Surrogate SALR outcomes within 3 days post-injection for the Hylan G-F 20 (6 mL) single injection, Monovisc, and Gel-One patient groups (top: outcomes with any diagnosis on the claims; bottom: outcomes with knee OA diagnosis on the claims). Statistically significant differences are indicated with an asterisk (*P < 0.05; green and red for lower and greater adjusted risks in the Hylan G-F 20 (6 mL) single injection group, respectively). SALR = severe acute localized reaction; OA = osteoarthritis; ER = emergency room; CS = corticosteroid.
When compared to patients who received Hyalgan/Supartz HA injections, Hylan G-F 20 (2 mL) 3 weekly injection patients had lower risks of 3-day clinical encounters in terms of office visits, ER visits, any office/ER/urgent care visits, and arthrocentesis ( Figs. 1 and 3 ). Hylan G-F 20 (2 mL) 3 weekly injection patients also had 45% lower adjusted risk of any corticosteroid injection or arthrocentesis (95% confidence interval [CI]: 41% to 48%; P < 0.001) and 47% lower adjusted risk of any arthrocentesis, arthroscopy, or arthrotomy/incision and drainage (95% CI: 44% to 50%; P < 0.001). The risk of corticosteroid injections with a knee OA diagnosis was greater for Hylan G-F 20 (2 mL) 3 weekly injection than Hyalgan/Supartz patients (adjusted hazard ratio [HR]: 1.65; P < 0.001), but there was no significant difference when considering any corticosteroid injections, that is, for any diagnosis (P = 0.294). Similarly, the risk of corticosteroid injections with a knee OA diagnosis was greater for Hylan G-F 20 (2 mL) 3 weekly injection than Orthovisc patients (adjusted HR: 1.47; P < 0.001), but there was no significant difference when considering corticosteroid injections for any diagnosis (P = 0.255) ( Figs. 1 and 3 ). When compared to patients who received Orthovisc HA injections, Hylan G-F 20 (2 mL) 3 weekly injection patients had lower risks of 3-day clinical encounters in terms of office visits, any office/ER/urgent care visit, and arthrocentesis. Compared to Orthovisc patients, Hylan G-F 20 (2 mL) 3 weekly injection patients also had 19% lower adjusted risk of any corticosteroid injections or arthrocentesis (95% CI: 14% to 24%; P < 0.001) and 19% lower adjusted risk of any arthrocentesis, arthroscopy, or arthrotomy/incision and drainage (95% CI: 14% to 25%; P < 0.001).
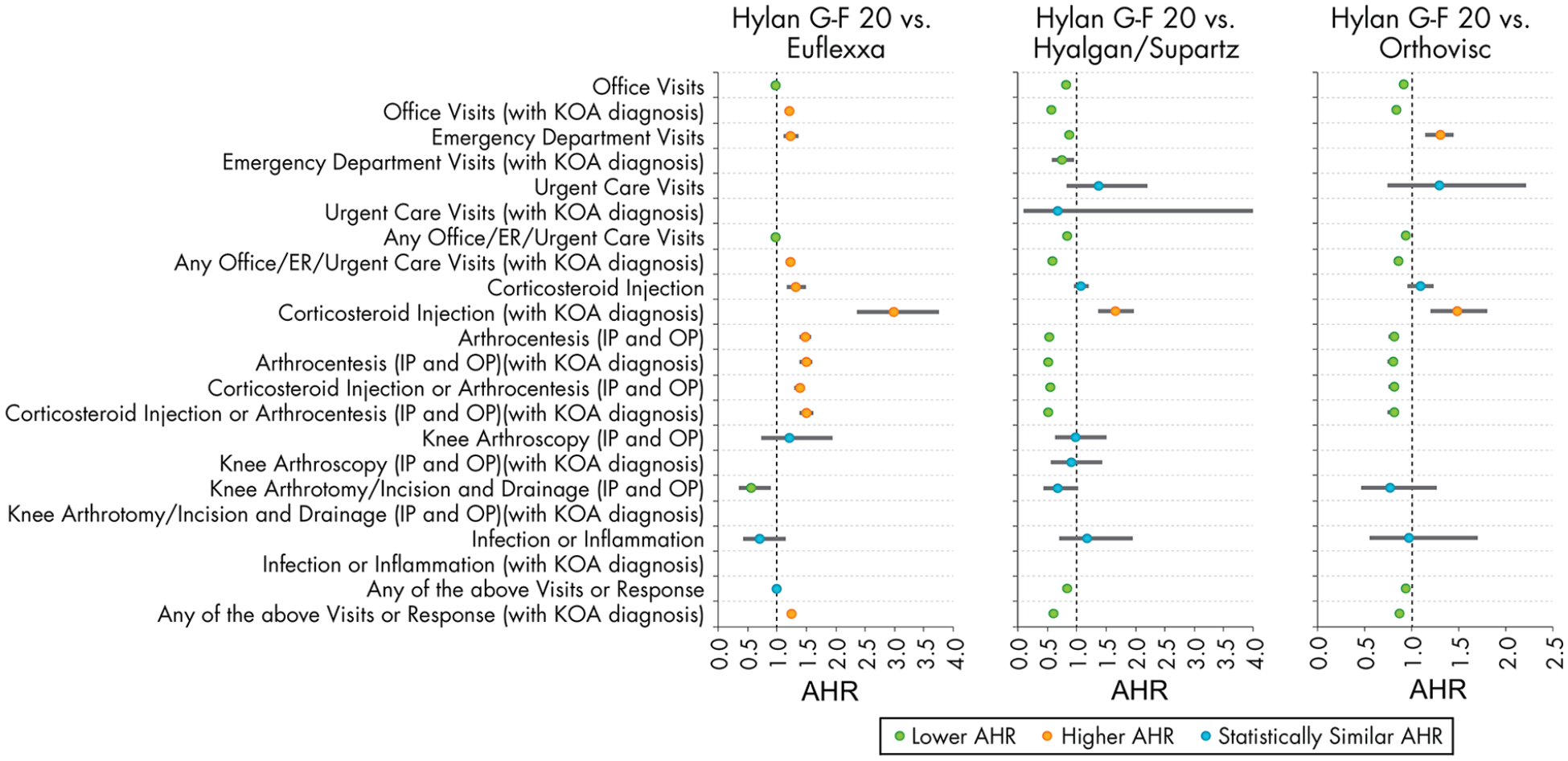
Adjusted likelihood of clinical encounters within 3 days post-injection for the Hylan G-F 20 (2 mL) 3 weekly injection group compared to other non-hylan G-F 20 (multiple injection) product groups. The reference group for each comparison is the non-hylan G-F 20 group. AHR = adjusted hazard ratio; KOA = knee osteoarthritis; ER = emergency room; Urg care = urgent care; CS = corticosteroid; IP = inpatient; OP = outpatient; I&D = incision and drainage.
When compared with patients who received Euflexxa, patients who received a Hylan G-F 20 (2 mL) 3 weekly injection generally had either similar or greater risks of 3-day clinical encounters ( Figs. 1 and 3 ). Corticosteroid injection or arthrocentesis was provided to 1.6% of the Hylan G-F 20 (2 mL) 3 weekly injection patients and 1.3% of the Euflexxa patients, representing a 39% greater adjusted risk (95% CI: 30% to 48%; P < 0.001). Inpatient or outpatient arthrocentesis, arthroscopy, or arthrotomy/incision and drainage was performed in 1.4% of the Hylan G-F 20 (2 mL) 3 weekly injection patients and 1.2% of the Euflexxa patients, which corresponded to a 43% greater adjusted risk (95% CI: 34% to 54%; P < 0.001). Inflammation or infection diagnoses were extremely rare within 3 days of HA injections, with knee OA related ones at 0.004% for the Euflexxa group and none for the Hylan G-F 20 (2 mL) 3 weekly injection group. These frequencies were too low to allow adjusted comparisons between groups via logistic regression. Even when expanded to include those with any diagnosis on the claims, the occurrence rate was 0.03% for the Euflexxa patients and 0.02% for the Hylan G-F 20 (2 mL) 3 weekly injection group, with no significant differences between the groups (P = 0.162). The adjusted risk of the collective occurrence of any of the surrogate SALR outcomes was found to be similar for Hylan G-F 20 (2 mL) 3 weekly injection and Euflexxa cohorts (P = 0.062), but lower for Hylan G-F 20 (2 mL) 3 weekly injection patients compared to Hyalgan/Supartz and Orthovisc patients (both P < 0.001).
Patients who received a Hylan G-F 20 (6 mL) single injection generally had either similar or lower risks of 3-day clinical encounters than those who received a Monovisc or Gel-One injection ( Figs. 2 and 4 ). Adjusted corticosteroid injections or arthrocentesis risk was 34% lower compared to both Monovisc (95% CI: 2% to 41%; P = 0.032) and Gel-One (95% CI: 8% to 37%; P = 0.005) patients. Although the incidence of arthrocentesis, arthroscopy, or arthrotomy/incision and drainage was 2.5% for the Hylan G-F 20 (6 mL) single injection patients compared to 4.5% and 3.4% for the Monovisc and Gel-One patients, respectively, this was not found to be significantly different in the regression analyses. Inflammation or infection diagnoses were extremely rare within 3 days of HA injections for the 1-injection HA groups. There were no knee OA related incidences for all 3 groups. Even when expanded to include those with any diagnosis on the claims, the occurrence rate of inflammation or infection diagnoses was 0.02% for the Hylan G-F 20 (6 mL) single injection patients, while none were diagnosed for the Monovisc and Gel-One groups. These frequencies were too low to allow adjusted comparisons between the groups. The adjusted risk of the collective occurrence of any of the surrogate SALR outcomes was found to be significantly lower for Hylan G-F 20 (6 mL) single injection patients compared to Monovisc patients (adjusted HR: 0.73; P < 0.001) and Gel-One patients (adjusted HR: 0.88; P = 0.012).
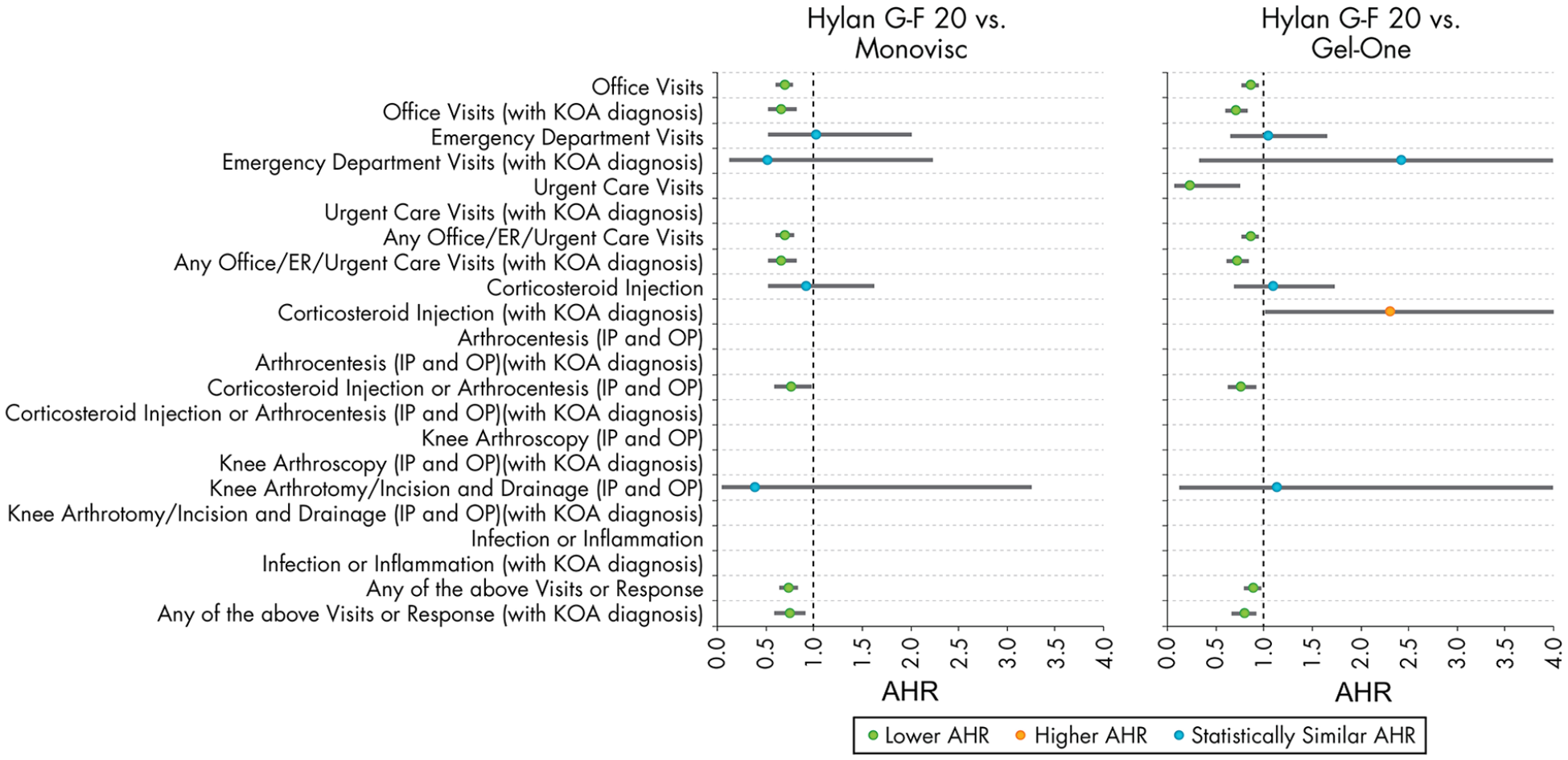
Adjusted likelihood of clinical encounters within 3 days post-injection for the Hylan G-F 20 (6 mL) single injection group compared to other non-hylan G-F 20 (single injection) product groups. The reference group for each comparison is the non-hylan G-F 20 group. AHR = adjusted hazard ratio; KOA = knee osteoarthritis; ER = emergency room; Urg care = urgent care; CS = corticosteroid; IP = inpatient; OP = outpatient; I&D = incision and drainage.
Discussion
In the present real-world study of almost 700,000 HA injections, potential SALR shortly following the HA injections were found to be exceedingly rare. We found no clear correlation between avian-derived products or crosslinking and SALR, which provides evidence against avian-derived or crosslinking products as a source for these reactions and further supporting the multifactorial nature of these reactions. SALRs was also observed in both single and multiple injection product groups. Avian-derived Hylan G-F 20 injections were found to have lower risk of SALR than bacteria-derived HA (Orthovisc and Monovisc), when matched by single or multiple injection types, except when compared to Euflexxa.
Injections of HA products have been reported to cause a rare inflammatory state within the joint. The pathophysiology underlying this response is poorly understood, but often mimics infection, and can place the practitioner in a difficult situation with highly variable diagnostic and therapeutic options. It has been hypothesized that these reactions are immune-mediated and that crosslinked HA products and those from avian origin are more susceptible to these reactions. Previously published findings from the same database as the present study showed that there was no statistically significant difference in SALRs between hylan and non-hylan G-F 20 products. 25 Those findings suggested that crosslinking or avian-derived HA may not play a role in SALRs as had been previously hypothesized, but the analysis was not stratified to be able to provide further comparisons of different products.
The purpose of this study was to determine if different commercially available HA products differ in their risk for developing SALR. Knowing which formulations are predisposed to SALRs may provide insight into the pathophysiology behind these reactions and guide decision making for providers. By comparing the surrogate endpoints for SALR of different HA products, we explored whether the SALR was potentially related to chemical properties (i.e., crosslinking and avian-derived) of the HA. Given the exceedingly rare nature of these reactions, a large database study with surrogate markers for SALR presents an opportunity to find statistically significant differences that would otherwise be unable to be detected with clinical trials.
The presentation of a reaction to HA injection is highly variable and the response of the clinician is even more variable. Some clinicians may have a higher index of suspicion than others for these reactions and elect to treat with an intraarticular CS injection to combat this likely immunologic reaction. Others may have a higher index of suspicion for infection following the injection, this would prompt the clinician to aspirate the knee and potentially perform surgery if the results of the aspirate were consistent with infection. Others may simply schedule a clinic visit and provide reassurance that the inflammation will subside with time. The present study was designed to capture these possible occurrences for each type of injection and determine whether or not there were statistically significant differences between different formulations. These SALRs were collected for both any diagnosis and knee-related diagnosis.
Our findings confirmed that rate of inflammation or infection diagnoses associated with HA injections were extremely rare.3,6 It also demonstrated that Hylan G-F 20 (2 mL) 3 weekly injection has a lower risk of SALR than most HA multiple injections, with the exception of Euflexxa. The comparative results with Euflexxa supported the findings of the 2006 randomized controlled trial by Kirchner and Marshall that showed a higher risk of effusions in patients receiving Hylan G-F 20 (2 mL) 3 weekly injection compared to Euflexxa patients. 21 Although Hylan G-F 20 is an avian-derived HA product and Euflexxa is extracted from bacterial cells, the current evidence could not support an allergic-type reaction associated with avian-derived products may be playing a role, as the risk of SALR was lower for Hylan G-F 20 (2 mL) 3 weekly injection than bacteria-derived Orthovisc 27 and for Hylan G-F 20 (6 mL) single injection than bacteria-derived Monovisc. 27 These reactions have also been documented for products that are not avian derived,3,8,10,16,18 further supporting their multifactorial nature. A randomized controlled trial also reported no difference in local reactions for hylan GF-20 and a medium molecular weight HA product, with no reports of pseudoseptic arthritis in either group. 11
In regard to whether or not crosslinking may play a role, analyses were performed between Hylan G-F 20 (2 mL) 3 weekly injection and Hyalgan/Supartz, all of which are of avian origin, but Hylan G-F 20 being crosslinked while the other two are not. 27 Hylan G-F 20 (2 mL) 3 weekly injection had a lower rate of SALRs when compared to Hyalgan/Supartz. This finding suggests that crosslinking HA may not play a role in increasing SALRs, but SALRs occurred in all HA injection cohorts despite whether or not the products were cross-linked or avian-derived. Our study provides further evidence of the multifactorial nature of these reactions that is not fully understood. Further basic science and clinical trials are needed to confirm these findings, as using surrogate markers of SALRs via a database study can show correlation but not necessarily causation.
Some have also speculated that increased frequency of injections may predispose the patient to greater acute local reaction risk or an “exposure” event. 9 For this reason, we also analyzed HA products that are marketed as a single injection: Hylan G-F 20 (6 mL) single injection, Monovisc, and Gel-One. Hylan G-F 20 (6 mL) single injection was found to have significantly decreased SALRs compared to both Monovisc and Gel-One. SALRs was observed in both single and multiple injection product groups, suggesting that the exposure theory is not the only factor playing a role in these reactions.
As far as we know, this is the first study large-scale real-world study to compare the risk of SALR between different HA products using surrogate endpoints. The limitations of this study largely stem from the nature of data collection from insurance claims database. Although we identified specific diagnoses or office visits that were likely to be related to the patient’s HA injection, this cannot be definitively proven through database claims. An attempt to make these SALRs more reliable was made by specifying whether or not the visit or intervention was knee related, although even those SALRs could have been unrelated to the patient’s HA injection. Another weakness of our study was the lack of objective measures such as laboratory or arthrocentesis results. Knowing the cell count, differential, and culture results could have allowed more specific isolation to the patients of interest. It also could have identified results that are indicative of an immunologic or allergic reaction, such as elevated eosinophils in the synovial fluid. Unfortunately, these data were not available through the database. The dataset also does not contain information about disease progression, severity of disease, range of motion, severity or duration of symptoms, or patient pain scores. It is also unclear if there would be any differences in outcomes associated with the medical specialty of the physician who delivered the HA injection. Although our data suggest that some HAs might have a lower risk of SALR than others, these are simply correlations. It has been speculated that repeated HA injections within brief intervals may be linked to pathogen inoculation. 2 Some have hypothesized that proinflammatory cytokines may play a role in the immunological reactions.3,4 Inappropriate injection technique, inexact needle placement, and block of synovial outflow by the injections have also been considered as potential causes. 6 Finding a direct causation would require basic science or clinical data. Although the dataset was composed of beneficiaries covered by commercial or private payers, patients older than 65 years were still included in the study cohort. However, the findings from the present study may not be representative of wholly Medicare beneficiaries. Despite the limitations of this study, a dataset with close to 700,000 HA injections presents a useful way to detect differences between products with this exceedingly rare complication.
Conclusion
This study shows no clear correlation between avian-derived or crosslinking products and SALR and provides evidence against avian-derived or crosslinking products as a source for these reactions. Overall, among patients who were treated with the single injection products, we found that Hylan G-F 20 (6 mL) single injection had lower rates of SALR than Monovisc and Gel-One. Among those treated with the multiple injection products, Hylan G-F 20 (2 mL) 3 weekly injection had lower rates of SALR when compared to Hyalgan/Supartz and Orthovisc but slightly higher rates of SALR when compared to Euflexxa. Further basic science and large clinical data are needed to validate these findings.
Supplemental Material
sj-docx-1-car-10.1177_19476035211025815 – Supplemental material for Risk of Severe Acute Localized Reactions for Different Intraarticular Hyaluronic Acid Knee Injections in a Real-World Setting
Supplemental material, sj-docx-1-car-10.1177_19476035211025815 for Risk of Severe Acute Localized Reactions for Different Intraarticular Hyaluronic Acid Knee Injections in a Real-World Setting by Kevin L. Ong, Jack Farr, Andrew S. Gudeman, Iain R. Murray, Louis F. McIntyre, Charles D. Hummer, Wilson Ngai, Edmund Lau, Roy D. Altman and Seth L. Sherman in CARTILAGE
Footnotes
Authors’ Note
Work was performed at Exponent, Inc.
Acknowledgment and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Sanofi US.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KLO, EL: Employees of Exponent, Inc., a scientific and engineering consulting firm. Exponent received funding from Sanofi US for this study.
KLO: Exponent has been paid fees for his consulting services on behalf of such companies and suppliers (Medtronic, Stryker Orthopaedics, Sanofi, Ferring Pharmaceuticals, Pacira Pharmaceuticals, St. Jude Medical, Relievant Medsystems, International Society for the Advancement of Spine Surgery, SI-Technology, LLC, Bioventus, Zimmer Biomet, Joerns Healthcare, SpineFrontier, Ethicon, DJO, Ossur, Karl Storz Endoscopy-America, Rex Medical, Smith & Nephew, Covidien).
JF: Research support from Active Implants, Arthrex, Episurf, Fidia, JRF Ortho, Moximed, Norvartis, Organogenesis, Samumed, Vericel, Zimmer Biomet; Paid consultant for Aesculap/B. Braun, Cartiheal, Cook Biotech Inc., Exactech, Moximed, Organogenesis, Regentis, Samumed Inc., ZKR Orthopedics; Editorial or governing board for American Journal of Orthopedics, Cartilage; Paid presenter/speaker for Arthrex, Organogenesis, Vericel; IP royalties for Arthrex Inc., Biopoly LLC, Moximed, Organogenesis; Stocks/stock options for MedShape Inc., Ortho Regenerative Tech Inc.; Publishing royalties, financial or material support for Springer, Thieme Medical Publishers.
ASG: Nothing to disclose.
IRM: Editorial or governing board for Bone and Joint Research, Journal of Bone and Joint Surgery–British.
LFM: Board or committee member for AAOS, Advocacy for Improvement in Mobility, Arthroscopy Association of North America, Delegate Medical Society of the State of New York, Orthopedic Practice Management, Inc., Westchester County Medical Society; Publishing royalties, financial or material support for Embody; Stock/stock options for Embody; Editorial or governing board for Orthopedics Today; Paid presenter or speaker for Sanofi-Aventis, Smith & Nephew; Paid consultant for Smith & Nephew; Employee of US Orthopedic Partners
CDH: Board or committee member for PA Orthopaedic Society; Paid consultant for Sanofi-Aventis.
WN: Employee of Sanofi US.
EL: Exponent has been paid fees for his consulting services on behalf of such companies and suppliers (Medtronic, Stryker Orthopaedics, Sanofi, Ferring Pharmaceuticals, Pacira Pharmaceuticals, Relievant Medsystems, Alcon, Boston Scientific).
RDA: Paid consultant for GlaxoSmithKline, Novartis, Pfizer, Sanofi-Aventis.
SLS: Board or committee member for AAOS, ACL Study Group, American Orthopaedic Society for Sports Medicine, Arthroscopy Association of North America, Epic Bio, International Cartilage Regeneration & Joint Preservation Society, International Society of Arthroscopy, Knee Surgery, Orthopaedic Sports Medicine, Joint Restoration Foundation; Editorial or governing board for American Journal of Orthopedics, Arthroscopy, Vericel; Paid consultant for Arthrex, Bioventus, Ceterix Orthopaedics, CONMED Linvatec, Flexion Therapeutics, GLG Consulting, JRF Ortho, Linvatec, Moximed, Olympus, RTI Surgical, Smith & Nephew, Vericel, Zimmer; Paid presenter or speaker for Arthrex, CONMED Linvatec, Flexion Therapeutics, Joint Restoration Foundation, Smith & Nephew, Vericel; Research support for Arthrex; IP royalties for CONMED Linvatec; Stock/stock options for Epic Bio, Vivorte.
Ethical Approval
Patient data used for this study were de-identified. The use of such data is considered exempt from the Institutional Review Board oversight according to Health Insurance Portability and Accountability Act.
Informed Consent
Informed consent is not required for this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
