Abstract
Objective
There is debate about the effectiveness of intra-articular (IA) hyaluronic acid (HA) injections. We asked the following questions: (1) What is the epidemiology of IA HA use in knee arthroplasty (KA) patients? (2) Is Synvisc associated with a delay to KA? (3) Is there a difference in the delay to KA with the number of HA courses?
Design
The Optum Clinformatics data set (2006–June 2016) was used to identify patients with knee osteoarthritis (OA). Patients who did not receive IA HA were identified, along with those who received Synvisc or non-Synvisc HA. The median duration from knee OA to KA was compared using quantile regression modeling with propensity score weighting. The trend in time to KA with each HA course was evaluated.
Results
Of 4,027,848 knee OA patients, 141,305 patients underwent KA. Overall median time from knee OA diagnosis to KA was 1.2 years (average: 1.9 ± 1.9 years; interquartile range of 0.4-2.8 years). HA patients had significantly longer adjusted median time to KA by at least 7 months (P < 0.001). After adjusting for the time to HA and number of injections, Synvisc patients had 1.7 months longer time to KA than non-Synvisc HA patients (P < 0.001). There was a trend toward longer time to KA with more HA courses.
Conclusions
Most KA patients did not use HA (73.7%) and when received, it was associated with a longer median time to KA by at least 7 months, though the cause and effect could not be examined. The delay to KA increased with more HA courses.
Introduction
Knee osteoarthritis (OA) affects a large number of the population, contributing significantly to healthcare costs and leading to the rise of costlier surgical interventions, such as knee arthroplasty (KA). Today more than 50 million Americans have arthritis and about 43% of them have arthritis-attributable activity limitations. 1 By 2040, these numbers are projected to increase 49% to 78.4 million patients with arthritis (25% of all adults) and 52% to 34.6 million with arthritis-related limitations (11% of all adults). As of 2015, the prevalence of OA was estimated in 30.8 million adults. Knee OA is one of the leading causes of disability among noninstitutionalized elderly adults, 2 affecting an estimated 14 million individuals in the United States. 1 Knee OA contributes more than $27 billion in health care expenditures annually. 1 Patients suffering from knee OA can show signs of severe pain with impaired activity, loss of work productivity, and quality of life.3-5 Annually, around 658,000 Americans undergo surgery for end-stage OA. 1 The rate of total knee replacements in many industrialized countries around the world has grown rapidly, with a 6.8% historical compound annual growth rate in the United States. 6 It has been estimated that the annual number of primary total knee replacement in the United States will exceed 1.3 million in 2020. 7
Knee OA patients have the option to undergo intra-articular (IA) hyaluronic acid (HA) injection treatment; however, there is ongoing debate about its effectiveness.8-19 IA HA administration is intended to restore the decrease in HA concentration and average molecular weight distribution in the knee joint from OA progression. 20 It is believed that there are several mechanisms in which IA HA act on the structure and function of the knee. Chondroprotection is most frequently cited, whereby chondrocyte apoptosis is reduced, while chondrocyte proliferation is elevated. 20 Enhanced proteoglycan and glycosaminoglycan synthesis and anti-inflammatory effects from suppression of interleukin-1β expression are also commonly described mechanisms of action for HA treatment.
With questions being raised about the effectiveness of IA HA, we asked the following research questions: (1) What is the epidemiology of IA HA use in KA patients? (2) Is the use of IA Synvisc associated with a delay to KA? (3) Is there a difference in the delay to KA with the number of HA courses?
Methods
Study Design
This study used a retrospective, observational study design based on the Optum Clinformatics data from 2006 until the end of the second quarter of 2016. Patients with knee OA were identified based on whether they were coded with principal/secondary diagnosis code of knee OA ( Table A1 in the appendix). If patients were coded with nonspecific OA and knee pain, they were also included in the study. 21 The first knee OA diagnosis was verified with a look-back period of 6 months with no prior knee OA diagnosis.
Study Cohort
The study cohort was limited to those aged at least 18 years. Patients were further limited to those who (1) had undergone KA (total knee arthroplasty [TKA] or unicondylar knee arthroplasty [UKA]) and had at least 6 months of follow-up after knee OA diagnosis. Patients that had IA HA treatment prior to the knee OA diagnosis were excluded. Patients were then divided in 3 different cohorts: (1) no HA cohort who did not receive any IA HA following knee OA diagnosis; (2) non-Synvisc cohort who received either multiple types of HAs during the study period or who used only one type of non-Synvisc HA during the study period; and (3) Synvisc cohort who received only Synvisc HA (Sanofi US, Inc.).
After their diagnosis of knee OA, patients were identified for having used IA HA treatment based on the presence of Healthcare Common Procedure Coding System (HCPCS) Q and J codes for HA ( Table A1 in the appendix). Patients who subsequently received a KA were further identified based on the Current Procedure Terminology codes for KA ( Table A1 in the appendix). The KA patients were then stratified into those who received at least one IA HA (HA group) and those who did not (no HA group) before the KA.
Statistical Analysis
To evaluate the effect of the covariates on the median duration from knee OA to KA, a quantile regression model was used, adjusting for the different clinical confounding factors. The use of median time rather than average time was due to the non-normal distribution of times to KA. Confounding factors, such as Charlson score, age, race, region (state), gender, treatment group, and year of knee OA diagnosis were evaluated. The time to HA and the number of HA injections were also included as covariates for the HA patients. The use of IA corticosteroid injection, prescription of nonsteroidal anti-inflammatory drug and opioid use (from pharmacy claims), knee arthroscopy, ultrasound/fluoroscopy use, or physical therapy (PT) use between the knee OA diagnosis and KA were also considered as confounding factors. These were identified using either HCPCS codes or brand names ( Table A2 in the appendix).
Since the baseline characteristics of treated subjects may differ systematically from those of untreated subjects, propensity score weighting was used to account for such differences, to minimize the confounding effects when using observational data, and to account for misspecification of the relationship between the risk factors and outcome. 22 The propensity scores were estimate from the probability of receiving IA HA, conditional on covariates of the patient group mentioned above. The propensity scores were used to weight responses for each subject as an inverse measure to their probability of receiving treatment that the subject actually received. SAS, version 9.4 (SAS Institute Inc., Cary, NC), was used for all statistical analyses. The trend in time to KA for the HA cohort with each additional course of HA was also evaluated. Each course of injection was evaluated based on an interval of 3+ months between each IA HA use.
Results
Epidemiology of KA Cohort
From the initial cohort of 4,027,848 knee OA patients, 141,305 KA patients who met the inclusion/exclusion criteria were identified ( Fig. 1 ). The distribution by the different treatments received are as follow: 104,145 patients (73.7%) without any HA injection; 22,523 patients (15.9%) who received at least 1 non-Synvisc HA injection; and 14,637 patients (10.4%) who received Synvisc HA injections. There was clear disparity between states of the distribution of KA patients who used HA (see Fig. A1 in the appendix). The 3 states with the lowest percent of KA patients who used HA were Hawaii (14.5%), Indiana (16.3%), and Arkansas (16.7%), while the 3 states with the highest percent of KA patients who used HA were New Jersey (39.1%), Louisiana (33.9%), and Mississippi (31.8%). The total number of HA injections in the KA cohort (n = 125,445) during the time period considered in this study showed an increasing trend of annual utilization from 10,178 in the first full year of the study period (2007) to a peak in 2012 (n = 16,741), followed by decline in utilization to 9,223 in 2015. The prevalence of HA injections was higher in the 55- to 69-year age groups compared to the other age groups and slightly higher in the female population ( Fig. 2 ). There was a large distribution of HA injections among white patients when compared with other races. In terms of comorbidity history, our results suggested that most patients presented none (0) or low (1-2) scores for their comorbid conditions before their knee OA diagnosis.
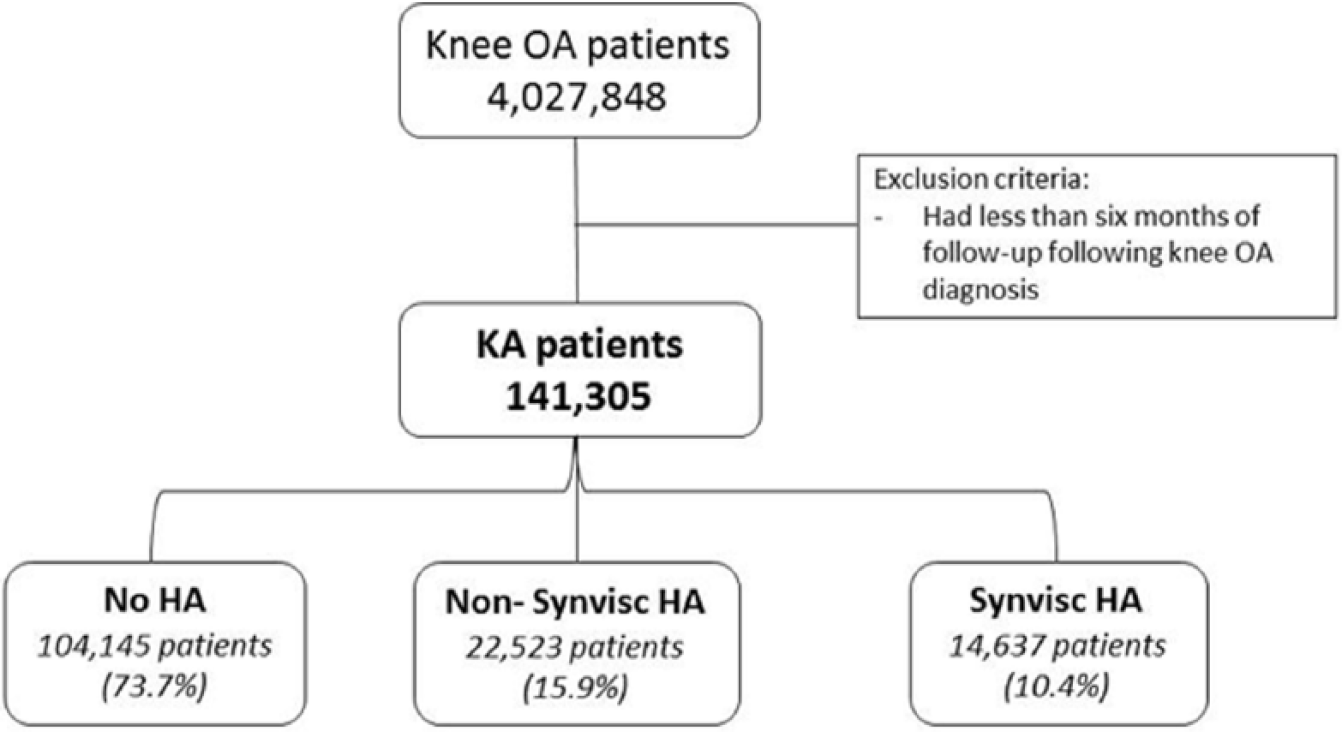
Study design. HA = hyaluronic acid; OA = osteoarthritis; KA = knee arthroplasty.
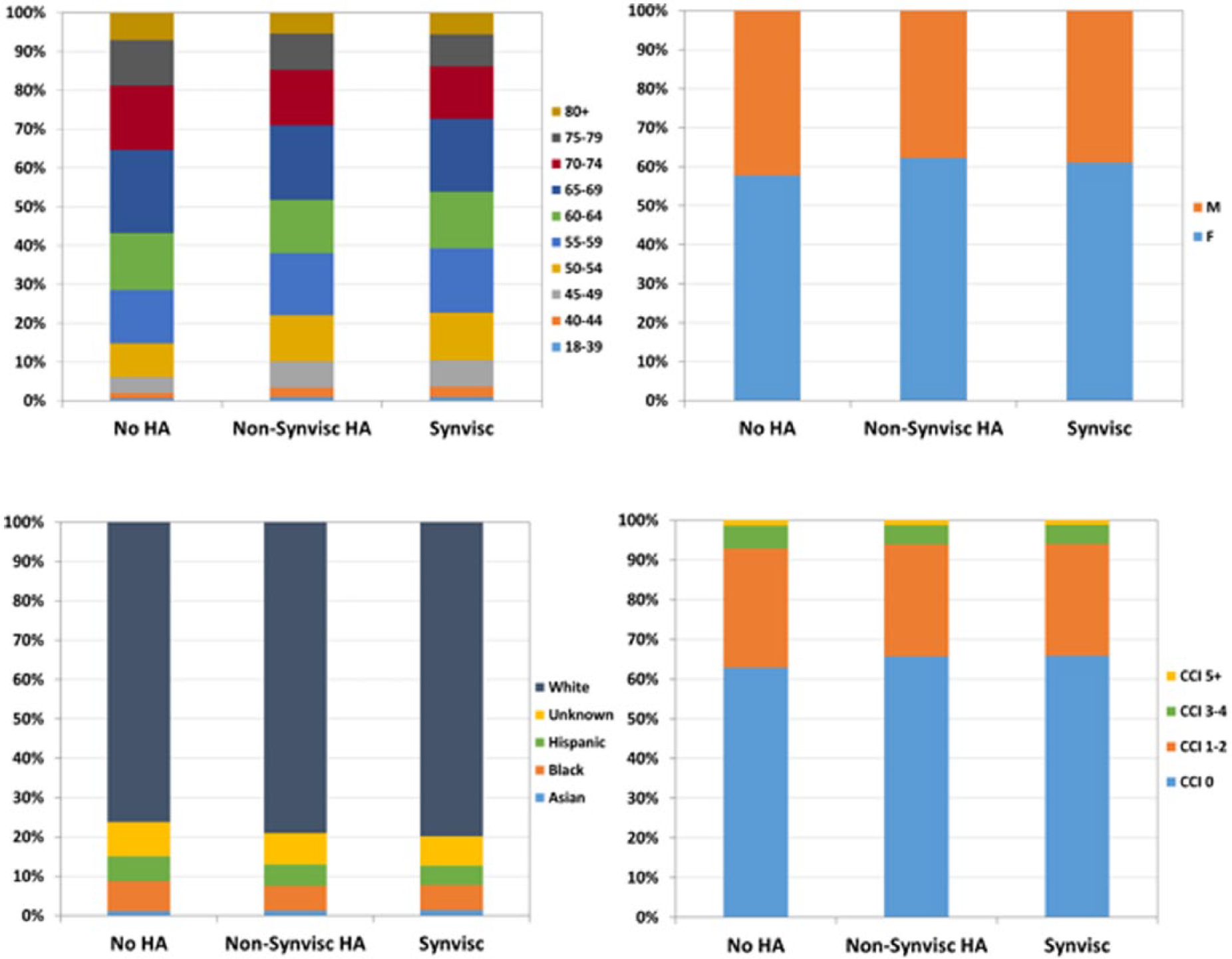
Distribution of age (top left), gender (top right), race (bottom left), and Charlson comorbidity index (bottom right) for each cohort. HA = hyaluronic acid; CCI = Charlson comorbidity index.
Delay to KA
The overall median time from the first knee OA diagnosis to KA was 1.2 years (average: 1.9 ± 1.9 years) with a corresponding interquartile (25th and 75th percentile) range of 0.4 to 2.8 years. The median times to KA were also longer for the HA cohort. The unadjusted median time to KA for the no HA, non-Synvisc, and Synvisc cohorts were 0.9 years (interquartile range [IQR] 25th to 75th percentile): 0.3-2.3 years), 2.1 years (IQR: 1.1-3.8 years), and 2.0 years (IQR: 1.0-3.7 years), respectively ( Fig. 3 ). After propensity score adjustment, the quantile regression showed that HA patients had significantly longer median time to KA by at least 7 months (P < 0.001; Table 1). When the HA cohort was compared after adjusting for the time to HA and number of injections, a longer time was observed for Synvisc patients than non-Synvisc HA patients by close to 2 months (P < 0.001).
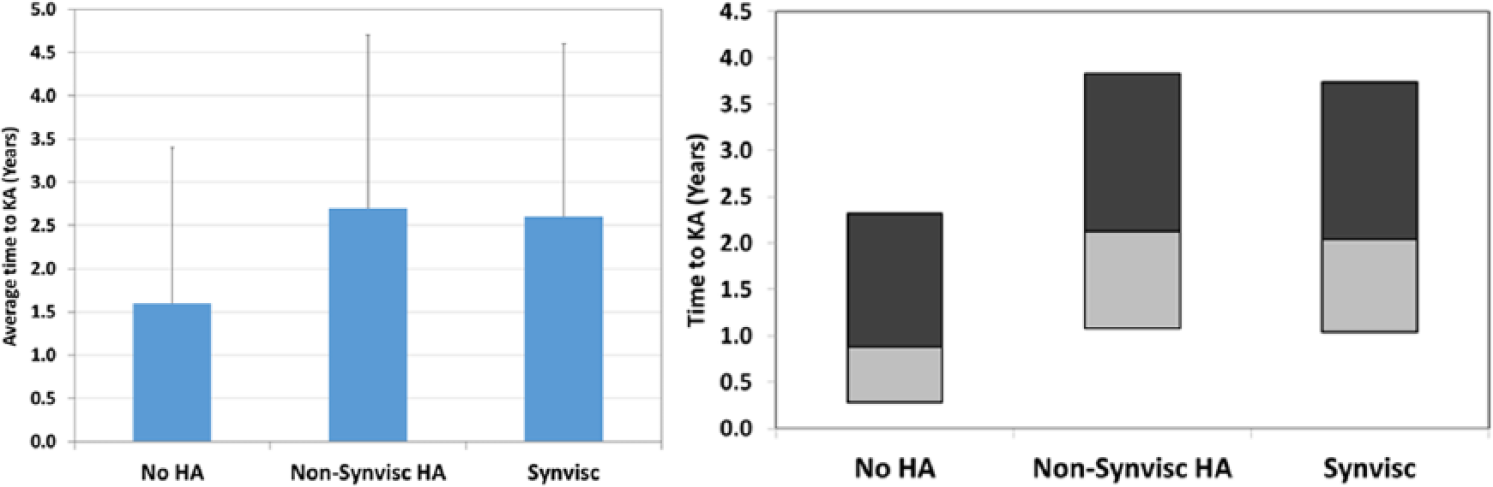
Average (left) and median (right; top and bottom of each bar corresponds to the 75th percentile and 25th percentile, respectively) time to knee arthroplasty (KA) for each cohort. HA = hyaluronic acid.
Differences in Median Time to Knee Arthroplasty (KA).
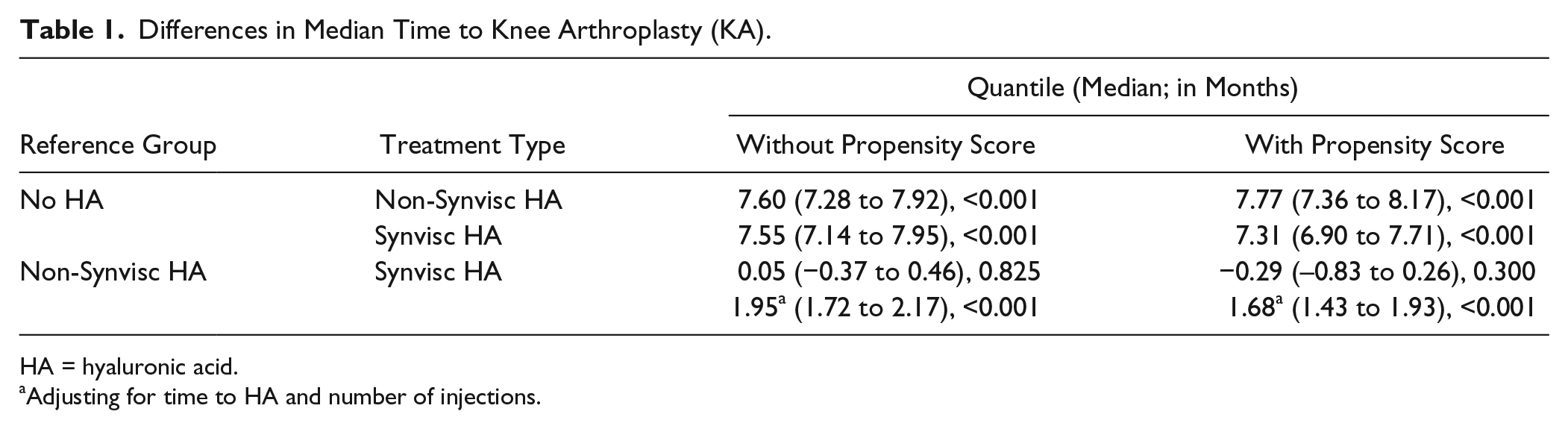
HA = hyaluronic acid.
Adjusting for time to HA and number of injections.
Number of HA courses
The majority of the KA patients received at least 1 course of HA injection ( Table 2 ). There were still a large percentage of patients who underwent multiple courses of HA injections (25.6% of non-Synvisc HA patients, 19.7% of Synvisc patients). There was a trend toward longer time to KA the more HA treatment courses the patient underwent ( Fig. 4 ). For example, in the non-Synvisc HA group, the unadjusted median time to KA increase from 20 months (1.7 years) for those who had 1 treatment course to 61 months (5.1 years) for those who had five or more treatment courses. Similarly, the unadjusted median time to KA increased from 21 months (1.8 years) to 59 months (4.9 years) as patients in the Synvisc group increased their treatment courses from 1 to 5 or more.
Distribution of Hyaluronic Acid (HA) Patients by the Number of HA Courses.

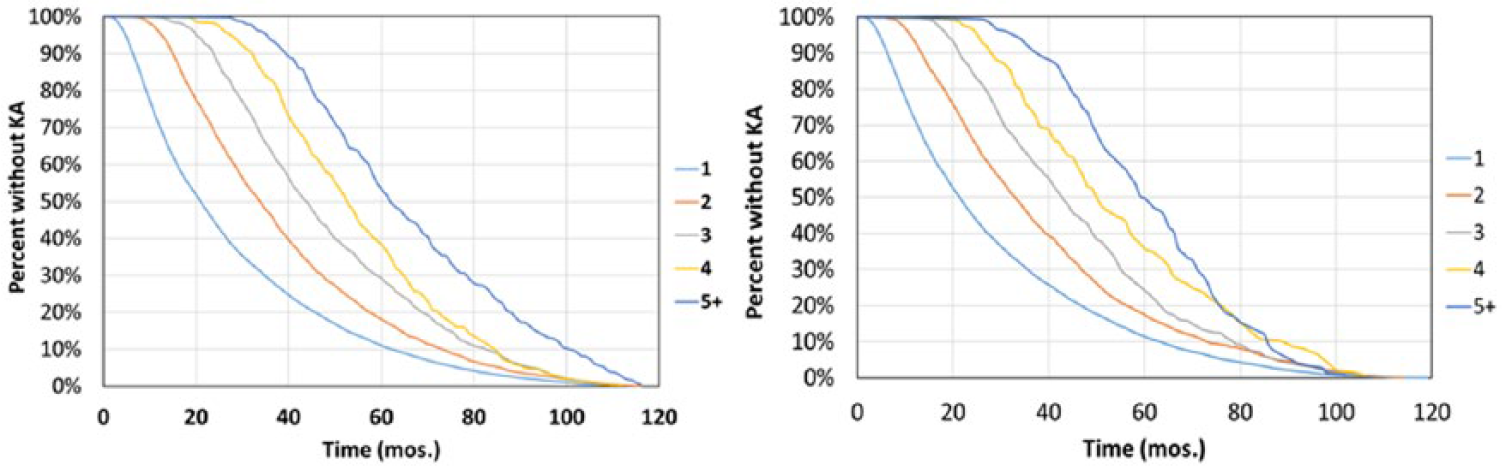
Kaplan-Meier curves with knee arthroplasty (KA) as the endpoint by the number of hyaluronic acid (HA) courses for the non-Synvisc HA cohort (left) and Synvisc cohort (right).
Discussion
This study evaluated the delay to KA following IA HA injections using a large data set of patients of both young and elderly groups from Optum Clinformatics. Our analysis indicated that most of KA patients did not use HA injection (73.7%) and the ones that did receive it were associated with a longer median time to KA by at least 7 months. Such trend of longer time to KA was shown to increase with more HA courses.
Our data revealed that the majority of KA patients did not use HA, along with substantial regional variation in utilization in our KA cohort. There was a 2.7-fold difference between the state with the highest (New Jersey; 39.1%) and the state with the lowest (Hawaii; 14.5%) percent of KA patients who used HA. Yet, the regional disparity and relatively small proportion of KA patients who had not used HA provide an opportunity for more patients to potentially benefit from the promising results of an extended time to KA when receiving HA. The delay to KA suggests a possible benefit of using this kind of therapy, although causation cannot be proven with observational data. When compared to non-HA patients, the HA patients in this study exhibited an additional delay to KA of at least 7 months (median). In terms of actual time between knee OA and KA, our cohort of HA patients had median time to KA of about 2 years, while the non-HA patients had a time to KA of 0.9 years. This was consistent with the reported delay to KA of about 1.8 years for 225 KA patients treated with Synvisc. 23 Ong et al. 24 also found a delay to KA of 1.3 to 1.9 years in the elderly Medicare population, but only when IA-HA was used with corticosteroid therapy. Otherwise, the time to KA was 0.6 to 1.2 years for those elderly patients. Since the non-Synvisc HA cohort included a variety of HA products with different molecular weights and formulations, it is possible that the time to KA may be affected by these factors as shown in several studies. Berenbaum et al. 25 found that intermediate molecular weight HA might be superior to low molecular weight HA on knee OA symptoms over 6 months, with similar safety profiles. Similarly, Maheu et al. 26 described a slightly lower efficacy for low molecular weight preparations versus intermediate and high molecular weight HA, although they suggested further investigations were needed. A recent meta-analysis by Altman et al. 27 also identified HA products with molecular weight of 3,000 kDa or more having superior efficacy. Despite the fact that preparations with high molecular weight have regularly shown a superior effect on relieving pain, according to the European League Against Rheumatism (EULAR) Recommendations 28 the management plan for knee OA patients has to be individualized and adjusted to the patient’s response. Moreover, these studies have not compared the association between molecular weight and the delay to KA.
In our data set, a large percentage of the patients who underwent multiple courses of HA injection presented considerably longer times to KA. The beneficial effects of extended use of HA have also been reported in several studies, which corroborates with our results. In a systematic review on repeated treatment courses of SUPARTZ injections, it was shown that multiple courses were safer than the first course with lower adverse effect rates. 29 In a multicenter and randomized study on the efficacy and safety of multiple injections of HA, it was shown that repeated cycles of IA HA injections improved the knee OA symptoms during the in-between cycle period for at least 1 year. 30 However, they could not demonstrate if such changes translated into true OA remission or a modification of the disease. A prospective cohort study evaluated the effectiveness of repeated cycles of IA HA injections during a 54-month follow-up period, demonstrating the benefit in the function of delaying time to total knee replacement (TKR) and even reducing the number of these procedures in elderly population. 31 The dose response was also observed by Altman et al., 32 who found that the median time to KA was 1.4 years following 1 course of HA, compared to 3.6 years following 5 or more HA courses.
Our study presents some limitations. The severity of OA was unknown in these patients due to the use of claims data. It is unclear to what extent the non-HA group might have more rapid evolution or more advanced disease compared with the HA groups. Our study focused on those who had KA to attempt to control for differences in baseline conditions and other potential confounding factors, including patient demographics, propensity score, and other clinical factors such as surgeon’s decision, surgeon’s availability, and patient access to care, and so on. The reasons to delay surgery are unknown for both the HA and non-HA groups due to the use of claims data. The cause-and-effect of HA use with the delay to KA cannot be examined due to the use of observational data, but this study, along with others, provides compelling data that HA use, especially with more courses of HA, may have beneficial effect in terms of delaying KA. Moreover, it is unclear if the increased delay to KA for the Synvisc HA group with more HA courses may be due to the therapy being used in patients with slower evolution of the disease. It is uncertain if the use of HA treatment slows down the evolution of OA or if the slow evolution of the disease may allow repeated cycles of HA use. We also found that the Synvisc group had 1.7 months longer delay to KA compared to the non-Synvisc group after accounting for propensity score, time to HA and number of injections, but it is unclear if further stratification of the non-Synivsc group by molecular weight would continue to demonstrate similar differences. Additionally, the further delay of 1.7 months was statistically significant, its clinical significance is unclear. Because the research questions of our study centered around KA patients, we did not evaluate whether HA-treated patients were able to avoid KA. Despite such limitations, our data has the advantage of being from a real-world large sample size, which includes both young and elderly knee OA patients.
Conclusions
The association of HA injections, including those specific to Synvisc, with the delay to KA was evaluated in this study. We concluded that a substantial percentage of patients use other therapies prior to KA and it varied by state. We also found that the time to KA was longer for patients who used HA prior to surgery when compared with the ones who did not receive any injection. The time to KA was also longer for Synvisc than non-Synvisc patients, after adjusting for the time to HA and number of injections. The clinical significance of the delay to KA should continue to be investigated.
Footnotes
Appendix
HCPCS Codes or Brand Name for the Clinical Factors Considered.

| Clinical Factor | CPT Code | |
|---|---|---|
| Physical therapy (PT) service | Range of motion/strength training | 97110 |
| Gait training | 97116 | |
| Mechanical traction | 97012, 97140, 97150 | |
| Electrical stimulation | 97014, 97032 | |
| Hydrotherapy including aquatic therapy | 97022, 97034, 97036, 97113 | |
| Vasopneumatic device use | 97016 | |
| Ultrasound | 97035 | |
| Neuromuscular re-education of movement and balance | 97112 | |
| Therapeutic activities, use of dynamic activities | 97530 | |
| PT services in home health setting | G0151 | |
| Corticosteroids (CS) | J0702, J0704 (expired after Dec 31, 2010), J1020, J1030, J1040, J1094, J1100, J1700, J1710, J1720, J2650, J2920, J2930, J3300, J3301, J3302, and J3303 (only effective from Jan 1, 2009) | |
| Knee arthroscopy | 29866 to 29868, 29870 to 29871, 29873 to 29877, 29879 to 29887, 29999, G0289 | |
| Ultrasound/fluoroscopy | 20611 (effective 2015 onward), 76942 or 77002 (have to be coded at same time/on same claim as HA injection) | |
| Clinical Factor | Brand Name | |
| Nonsteroidal anti-inflammatory drug (NSAID) | Celecoxib | |
| Diclofenac | ||
| Etodolac | ||
| Fenoprofen | ||
| Flurbiprofen | ||
| Ibuprofen | ||
| Indomethacin | ||
| Ketoprofen | ||
| Ketorolac | ||
| Lecoprofen | ||
| Nabumetone | ||
| Naproxen | ||
| Oxaprozin | ||
| Clinical Factor | Drug Name | Trade Name |
| Opioid | Alfentanil hydrochloride or alfentanyl or alfentanil | |
| Anileridine | ||
| Alphaprodine | ||
| Bromadol | ||
| Buprenorphine | Butrans, Belbuca, Buprenex | |
| Butorphanol | ||
| Codeine | ||
| Dextropropoxyphene | ||
| Dihydroetorphine | ||
| Dihydrocodeine | Synalgos | |
| Diamorphine | ||
| Etorphine | ||
| Fentanyl | Fentora, Lazanda, Subsys, Abstral, Actiq | |
| Fentanyl citrate | ||
| Fentanyl patch (fentanyl transdermal system) | Duragesic | |
| Heroin | ||
| Hydrocodone | Zohydro, Hysingla Vicoprofen | |
| Hydrocodone bitartrate | ||
| Hydromorphone | Exalgo, Dilaudid | |
| Hydromorphone hydrochloride | Exalgo, Palladone | |
| Levorphanol | ||
| Meperidine | Demerol | |
| Methadose | ||
| Methadone | Dolophine, Methadose | |
| Methadone hydrochloride | Dolophine | |
| Morphine | MS Contin, Oramorph, Embeda, Kadian, Infumorph, Astramorph, Duramorph | |
| Morphine sulfate | Avinza, Embeda, Kadian, MS Contin | |
| Naltrexone | Embeda | |
| Nalbuphine | ||
| Oxycodone | Xtampza, OxyContin, Xartemis, Percocet, Oxecta, Roxicodone, Targiniq | |
| Oxymorphone | Opana | |
| Oxymorphone Hydrochloride | Opana | |
| Pethidine | ||
| Propoxyphene | ||
| Remifentanyl or Remifentanil | ||
| Sufentanyl or Sufentanil | Sufenta | |
| Tramadol | Conzip, Ultram, Ultracet | |
| Tapentadol | Nucynta | |
| 7-Hydroxymitragynine | ||
Acknowledgments and Funding
This study was supported by Sanofi US.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KO, EL, MR are employees of Exponent, Inc., a scientific and engineering consulting firm. Exponent received funding from Sanofi US for this study. KO: Exponent has been paid fees by companies and suppliers for my consulting services on behalf of such companies and suppliers (Stryker Orthopaedics, Zimmer Biomet, Ethicon, Ferring Pharmaceuticals, Paradigm Spine, Medtronic, Pacira Pharmaceuticals, DJO, Ossur). RA is a paid consultant for Ferring Pharmaceuticals, Norvartis, Pfizer, and unpaid consultant for Sanofi-Aventis.
Ethical Approval
Ethical approval was not sought for the present study because this study was based on publicly available data sets, did not use private health identifiable information, and did not represent human subject research, and therefore did not require oversight by our institutional review boards.
Informed Consent
Informed consent was not sought for the present study because the manuscript does not contain any studies with human participants or animals performed by any of the authors. For this type of retrospective study, formal consent is not required.
Trial Registration
Not applicable.
