Abstract
Objective:
While articular cartilage defects are common incidental findings among adult athletes, the effect of running on the cartilage of adolescent athletes have rarely been assessed. This study aims to assess the variations in the articular cartilage of the knees in healthy adolescent basketball players using quantitative T2 MRI (magnetic resonance imaging).
Design:
Fifteen adolescent basketball players were recruited (13.8 ± 0.5 years old). Girls were excluded to avoid potential gender-related confounding effects. Players underwent a pre-run MRI scan of both knees. All participants performed a 30-minute run on a treadmill. Within 15 minutes after completion of their run, players underwent a second, post-run MRI scan. Quantitative T2 maps were generated using the echo modulation curve (EMC) algorithm. Pre-run scans and post-run scans were compared using paired t test.
Results:
Participants finished their 30-minute run with a mean running distance of 5.77 ± 0.42 km. Pre-run scans analysis found statistically significant (P < 0.05) changes in 3 regions of the knee lateral compartment representing the cartilaginous tissue. No differences were found in the knee medial compartment. Post-run analysis showed lower T2 values in the medial compartment compared to the pre-run scans in several weight-bearing regions: femoral condyle central (pre/post mean values of 33.9/32.2 ms, P = 0.020); femoral condyle posterior (38.1/36.8 ms, P = 0.038); and tibial plateau posterior (34.1/31.0 ms, P < 0.001). The lateral regions did not show any significant changes.
Conclusions:
Running leads to microstructural changes in the articular cartilage in several weight-bearing areas of the medial compartment, both in the femoral and the tibial cartilage.
Introduction
The positive implications of physical activity are indisputable. Youth participation in physical activity and sports has been found to have many merits.1,2 Over the past 3 decades we have witnessed a trend of increasing participation of youth in sport activity and athletics.3,4 Adolescent athletes are often treated as semiprofessional athletes, involving intense training programs, adjusted nutrition plans, and psychological conditioning. Nevertheless, the competitive nature of these activities, together with the high demanding lifestyle associated to it, often leads to a rise in sports related injuries, many of which resemble the patterns of collegiate and professional athletes. 5 In the United States alone it is estimated that more than 7 million high school students participate in athletics each year, with reports of more than 4 million sports or recreational injuries sustained by school-age children annually. 6
Knee injuries are one of the most common sports-related injuries in young athletes, 7 with a rate of 2.98 per 10,000 athlete exposures, 8 most of which involve the knees, which have a propensity toward severe injuries. 9 The occurrence of such injuries is more common during competition than during practice, while the injuries pattern are also dependent on the type of activity and gender. 8 Recent studies report that basketball players have higher frequency of knee injuries compared to their peers. 10
Articular cartilage is a form of hyaline cartilage, composed of chondrocytes and an extracellular matrix, which is, in turn, composed of water (accounting for 65% to 80% of the cartilage mass) and proteins. The protein composition of the cartilage is commonly divided to collagen (10% to 20% of the cartilage mass; 90% to 95% of the collagen is type II), proteoglycans (10% to 15% of the cartilage mass) and non-collagenous proteins. The collagen has a structural role that gives the cartilage its framework and accounts for its tensile strength. The proteoglycans allow the cartilage high-water content due to their negative electric charge (aggrecan has the most hydrophilic features of the cartilage proteoglycans). This, in turn, accounts for the cartilage compressive strength. Its microstructure not only enables the healthy cartilage to serve as a low-friction surface for articulation without losing its tensile strength but also aids in resisting compression. 11
Magnetic resonance imaging (MRI) has evolved into a highly valuable tool for assessment of articular cartilage changes, with sensitivity of over 80% and specificity of over 95%. 12 Different methods of compositional MRI have been shown to allow identification of early and potentially reversible cartilage damage. 13 These include qualitative and quantitative T2 contrast, measuring spin lattice relaxation time in the rotating frame (T1ρ), and the use of delayed gadolinium-enhanced MRI of the cartilage (dGEMRIC).
T2, the spin-spin relaxation time, reflects interactions between water protons and tissue macromolecules, and in turn reflects the structure of the extracellular matrix. Contrary to conventional T2-weighted imaging, quantitative T2 mapping (qT2) enable the detection of small alterations in the water content, as well as variations in the structural integrity of collagen and the proteoglycans.14,15 Articular cartilage signal abnormalities are common incidental findings. Walczak et al. reported signal abnormalities in 50% of asymptomatic professional NBA players off-season. 16
The prevalence of chondral damage among young athletes was found to be 36% higher than in the general population. 17 While cartilage injuries are often asymptomatic, these injuries can be debilitating with potential long-term risk for early osteoarthritis. Even though we do not fully understand the natural history of these injuries, the current available literature demonstrates a progression from cartilage injury to osteoarthritis, 18 even in asymptomatic patients. 19
MRI has shown to be capable of identifying changes of thickness and signal intensity in the articular cartilage under loading conditions, which were associated with volumetric and morphological changes.20,21 Furthermore, studies have also shown the potential merit of parameters such as T2 relaxation time mapping in evaluation of the biochemical composition of the cartilage owing to its sensitivity to changes of water interactions, which in turn may reflect alterations in collagen content, direction, and orientation.22-24 In this study we probed articular cartilage signal changes in healthy knees of adolescent basketball players using qT2 MRI. Using quantitative mapping before and after running we were able to observe and quantify changes in the articular cartilage. This may aid in revealing the effect of running on healthy knee joint cartilage of adolescent basketball players 22 and help optimize adolescent training programs. We hypothesized that the weight-bearing area of articular cartilage of the medial compartment is prone to experience significant alterations as opposed to other weight-bearing areas in the knee.
Methods
Patient Population
Our study was reviewed and approved by a local institutional review board committee. Fifteen healthy volunteers between the ages of 13 and 15 were recruited from local youth basketball teams. Girls were excluded to avoid potential gender-related confounding effects attributed to differences in knee biomechanics and anatomy. The purpose was to recruit healthy volunteers with similar age and physical health parameters. Volunteers were excluded if they reported previous knee surgery, inability to run for 30 minutes, or other known major knee injury (an injury which may cause disability or one that requires medical attention).
Assessment Day Schedule and Protocol
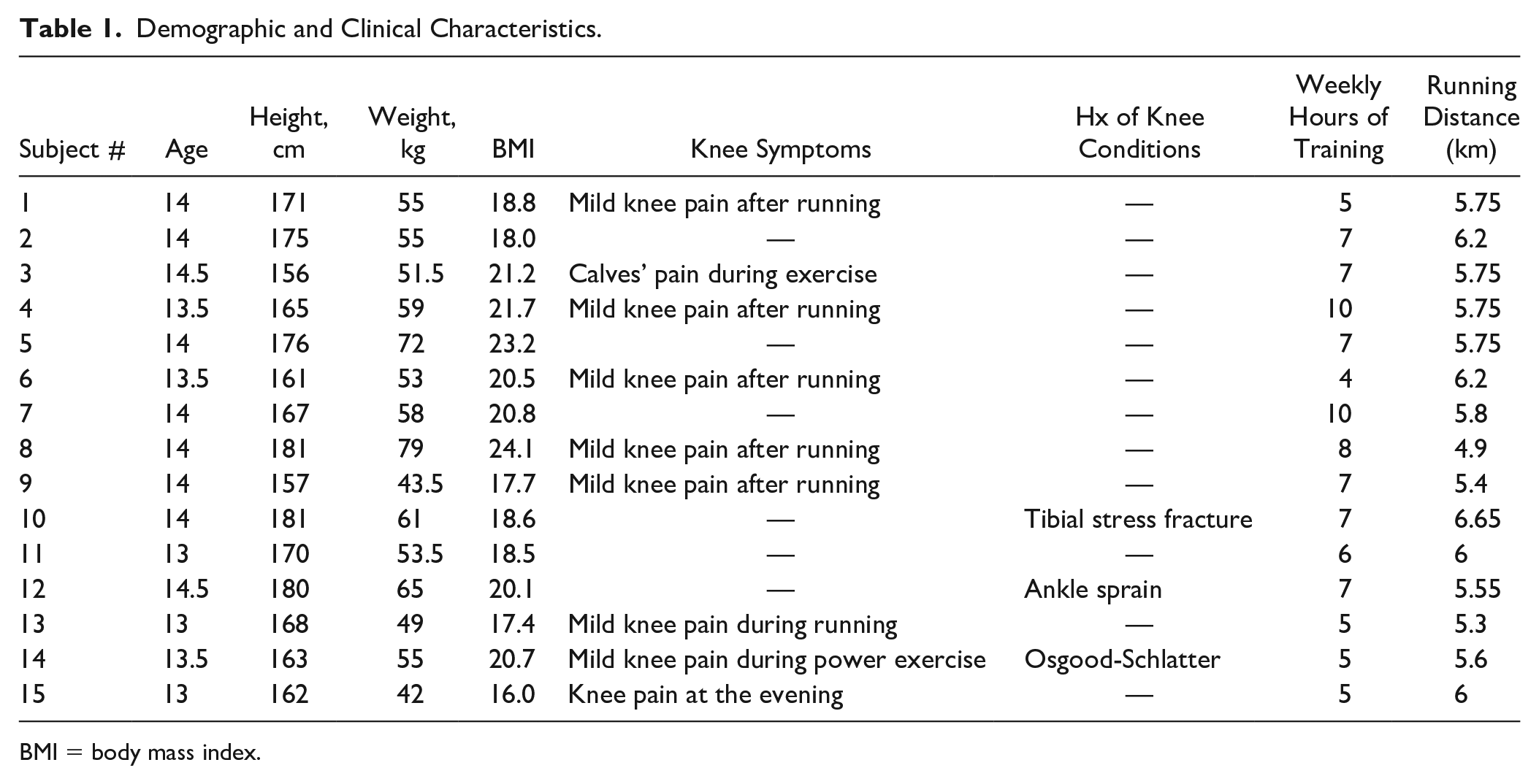
Participants arrived at 08:00 after full-night sleep and were instructed to avoid any physical activity prior to their meeting. After receiving full explanation on the nature of the study, subjects’ parents provided informed consent. Participants’ age, height, and weight were documented, followed by collection of the participants’ medical history, including sports-related injuries and knee-related symptoms. Each participant was also asked about any knee related pain/history of his parents. Participant data are summarized in Table 1 . After further briefing, participants underwent a pre-run MRI scan of both knees (30 knees), followed by a physical examination performed by an orthopedic surgery resident (6th year, PGY-6), in an attempt to identify any sign of instability, meniscal injuries, and other pathologies in the knees ( Table 1 ). Within 30 minutes of completing the pre-run scan, the participants performed a 30-minute run on a treadmill. They were encouraged to run at a fast pace for a total of 30 minutes. Their run was supervised by the coauthor of the paper (YC, orthopedic surgeon). All 15 participants finished their 30-minute run. Mean running distance was 5.77 ± 0.42 km (range 4.9-6.65 km; Table 1 ). Within 15 minutes after completion of their run, participants underwent a second, post-run MRI scan of both knees ( Fig. 1 ). Pre-run MRI scan was 20 minutes long for each knee. Post-run scan was 15 minutes long for each knee. We started from the right-side knee. That means that post-run scan was done within 45 minutes from running end. This could help us minimize variations. Data were organized and analyzed.
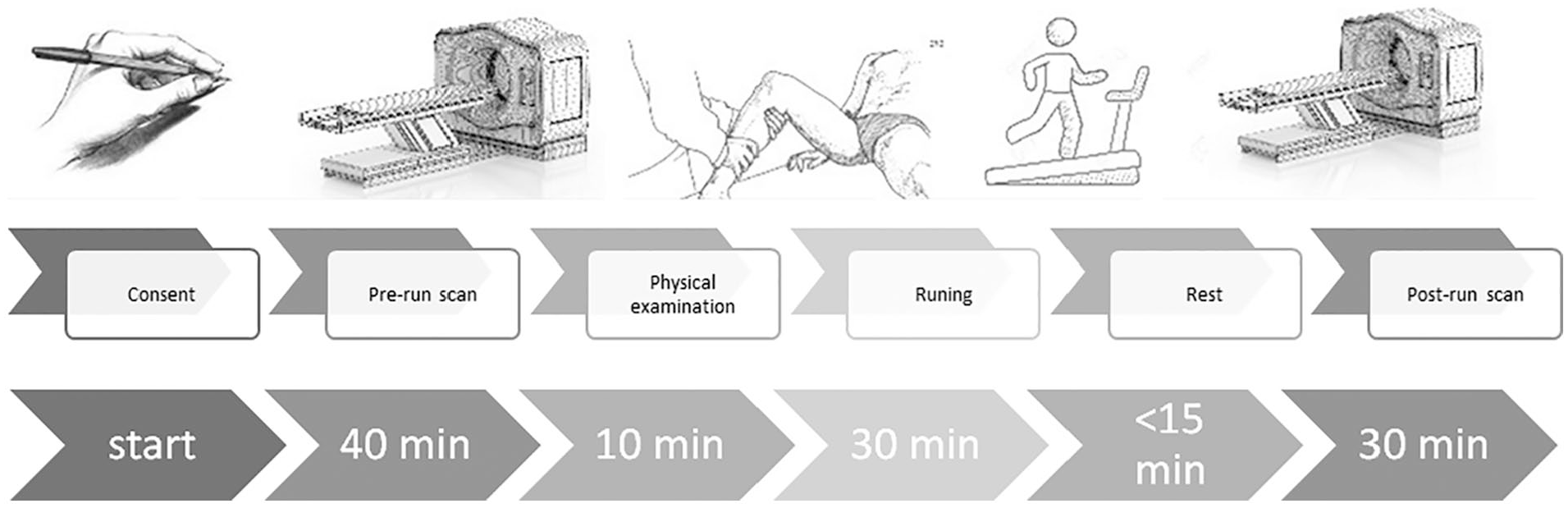
The study flow chart: informed consent, pre-run scan, physical examination, 30 minutes run, post-run scan within 15 minutes of run finish.
MRI Scans
All scans were performed on a 3-T MRI scanner (Skyra, Siemens Healthineers Inc., Erlangen, Germany) using a 15-channel dedicated knee coil. Quantitative mapping of T2 relaxation times was performed using a multi echo spin echo (MESE) imaging protocol. Experimental parameters included: TE/TR = 15/1100 [ms], NEchoes = 6, field-of-view = 160 × 160 mm2, matrix size 384 × 384, BW = 130 [Hz/Px], slice thickness = 4 mm, Nslices = 11 (per one knee). Sagittal images of the medial and the lateral femorotibial joints were acquired (17 slices in each knee). Each knee’s data set included 2 series of images: pre-run and post-run. Pre-run scans included T1- and T2-weighted images, along with quantitative mapping of T2 values, while post-run scan included only T2 mapping. To ensure consistent slice positioning, imaging planes were set to follow the standard anatomical alignment of MRI scans. This allowed to identify more methodically the anatomical regions of interest within series of imaged slices, while removing the need for further image registration procedures.
Data Post Processing
Quantitative T2 maps (exemplified in
Figs. 3
MESE protocols are known to produce highly biased T2 values when fitting the signal to a theoretical exponential decay pattern of the form
Radiologic MRI Analysis
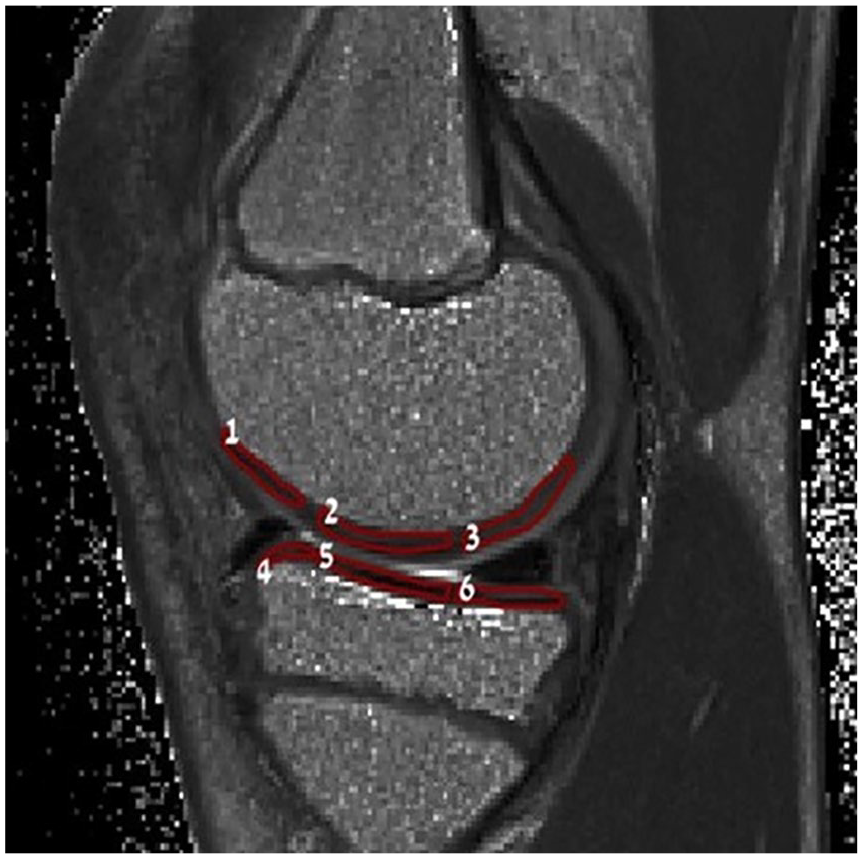
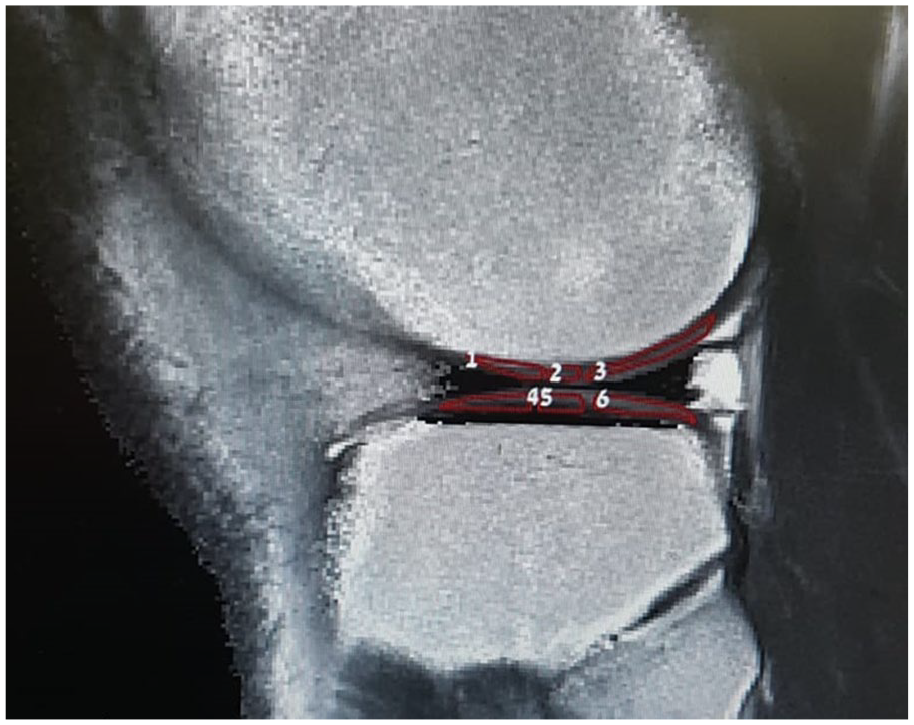
A musculoskeletal (MSK) radiologist (OL) examined the MRI scans of all participants for pathologies. Six sagittal sections were selected out of 17 available sections. Sagittal sections were chosen to represent central areas of medial and lateral parts of the knee joint (3 sections of medial compartment and 3 sections of lateral compartment), as shown in
Figure 2
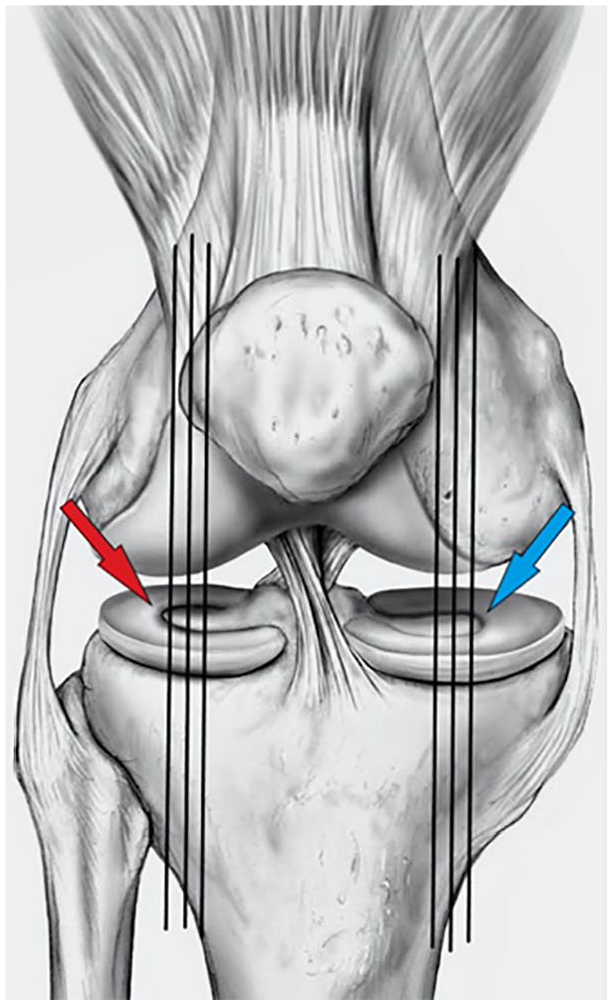
. Thus, 12 sagittal sections were analyzed in each knee (6 sections from pre-run scan, 6 sections from post-run scan). Six regions of interest (ROIs) were manually segmented by a trained orthopedic surgeon, supervised by the MSK radiologist, in each sagittal section. These segments covered the medial and lateral weight-bearing parts of the cartilage (see
Fig. 2
). Femoral and tibial surface of the cartilage were each divided into 3 regions, inner margins of the meniscus were used as a marker for the anterior and posterior borders of the regions (
Figs. 3
Six sagittal sections were selected out of 17 available sections, chosen to represent central areas of medial and lateral parts of the knee joint. Three sagittal sections for each compartment. Each section had to be central and to include meniscus area.
Femoral and tibial surface of the cartilage were each divided into 3 regions using the inner margin of the meniscus as a marker for the anterior and posterior borders of the weight-bearing regions.
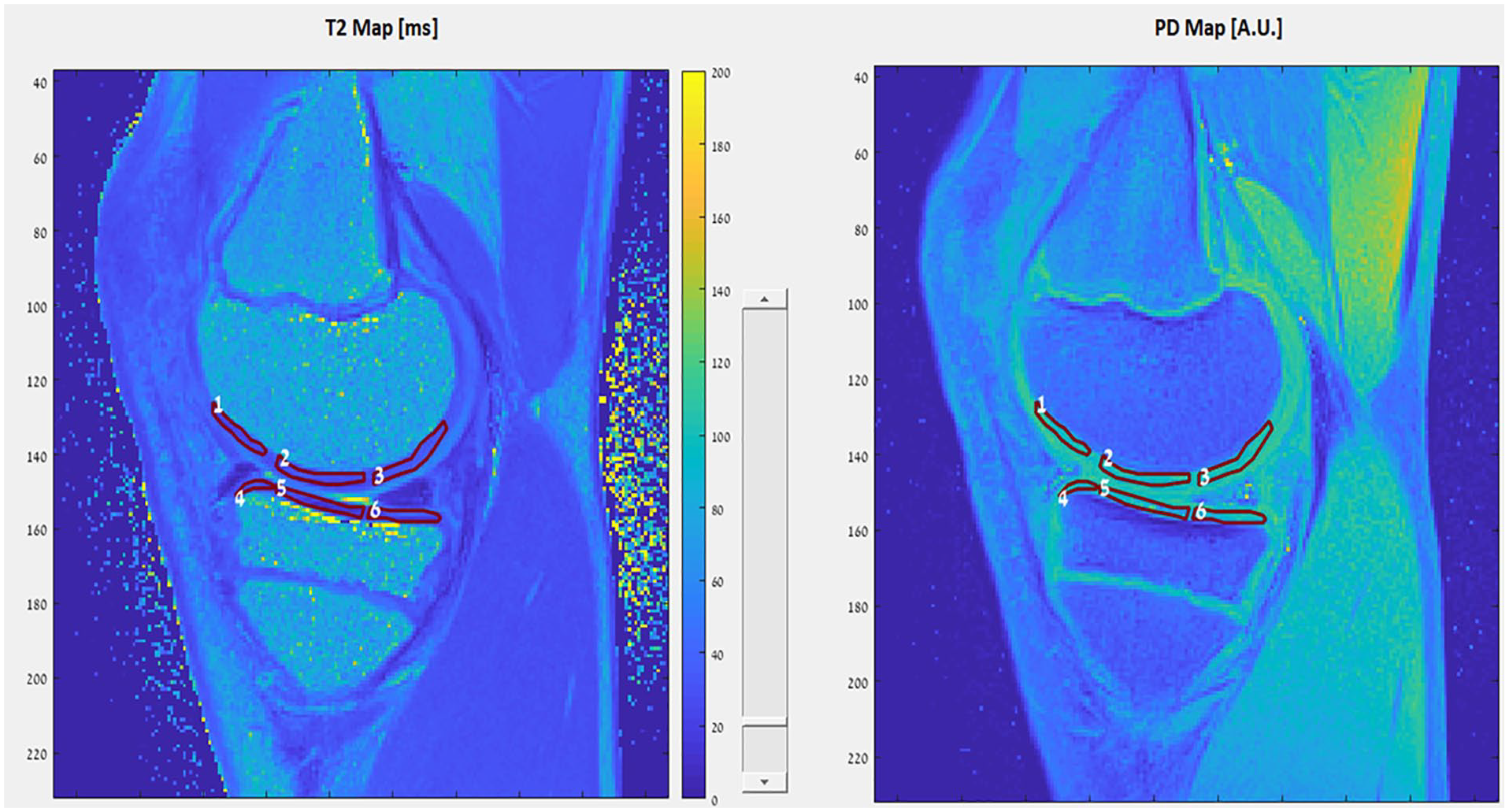
An example of image processing—medial knee compartment.
An example of image processing—lateral knee compartment.
Statistical Analysis
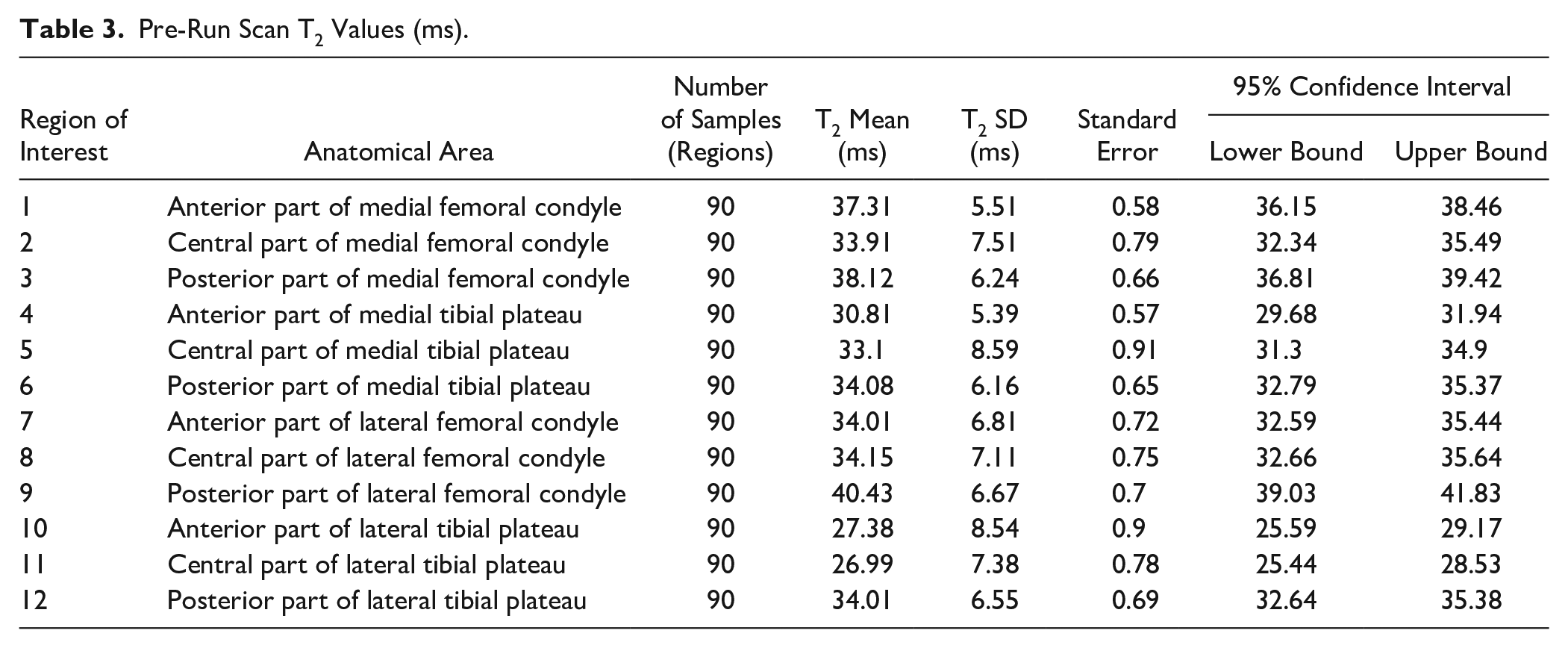
SPSS software (version 2015, IBM) was used for statistical analysis of the T2 mapping scans. Twelve ROIs were analyzed ( Tables 3 and 4 ): 6 from medial compartment and 6 from lateral compartment. Three slices were chosen for each ROI assessment, leading to a total of 90 samples for each ROI (30 knees) in each series. The 2 series, pre- and post-run scans, were compared using a paired t test for each ROI ( Tables 5 and 6 ). P-values less than 0.05 were considered to be statistically significant. ANOVA analysis of all 12 regions with pre-run values was performed ( Table 7 ), using the Bonferroni test with each analysis. Each region was compared to the other 11 regions. Differences which were statistically significant are presented in Table 7 .
Results
Demographics and Patient Characteristics
All the participants were active players in youth basketball teams, with at least 4 practices weekly (6.67 ± 1.76; Table 1 ). Participants’ mean age was 13.8 ± 0.5 years old. Mild leg pain during or after exercise was reported by 9 out of 15 participants (60%). Prior sports-related injury was reported by 3/15 (20%) of the participants ( Table 1 ).
Physical Examination
No major pathological finding or signs of knee instability were found on physical examination. All participants’ knee range of motion was within normal range (full extension, >130° of flexion). Alignment of the knees was normal, except for one knee which showed mild valgus. Tibial tuberosity tenderness was found in 4 knees. All meniscal tests (Lachman test, Bounce home test) were reported as normal. No knee effusion was identified. Mean body mass index (BMI) was 19.81 (±2.25) ( Table 1 ).
Demographic and Clinical Characteristics.
BMI = body mass index.
MRI Findings
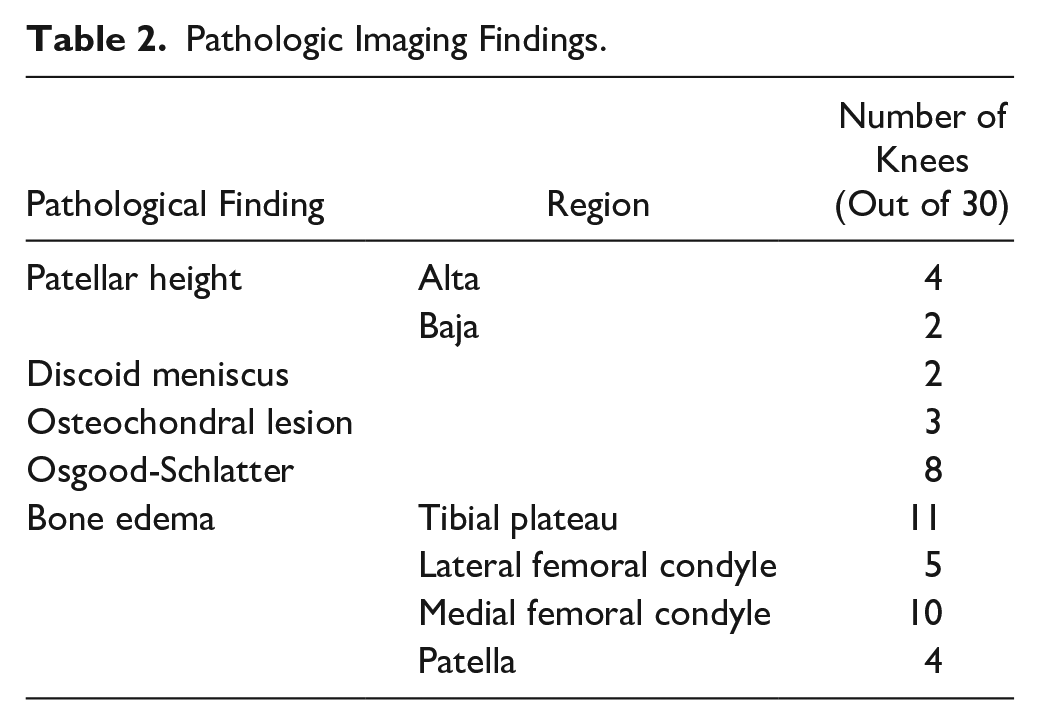
MRI scans of 30 knees (15 participants) were obtained and reviewed by an expert MSK radiologist. Mild edema of tibia was the most common finding (
Table 2
). Pre-run assessment of the knee ROIs was compared to the post-run findings.
Tables 3
and
Pathologic Imaging Findings.
Pre-Run Scan T2 Values (ms).
Post-Run Scan T2 Values (ms).
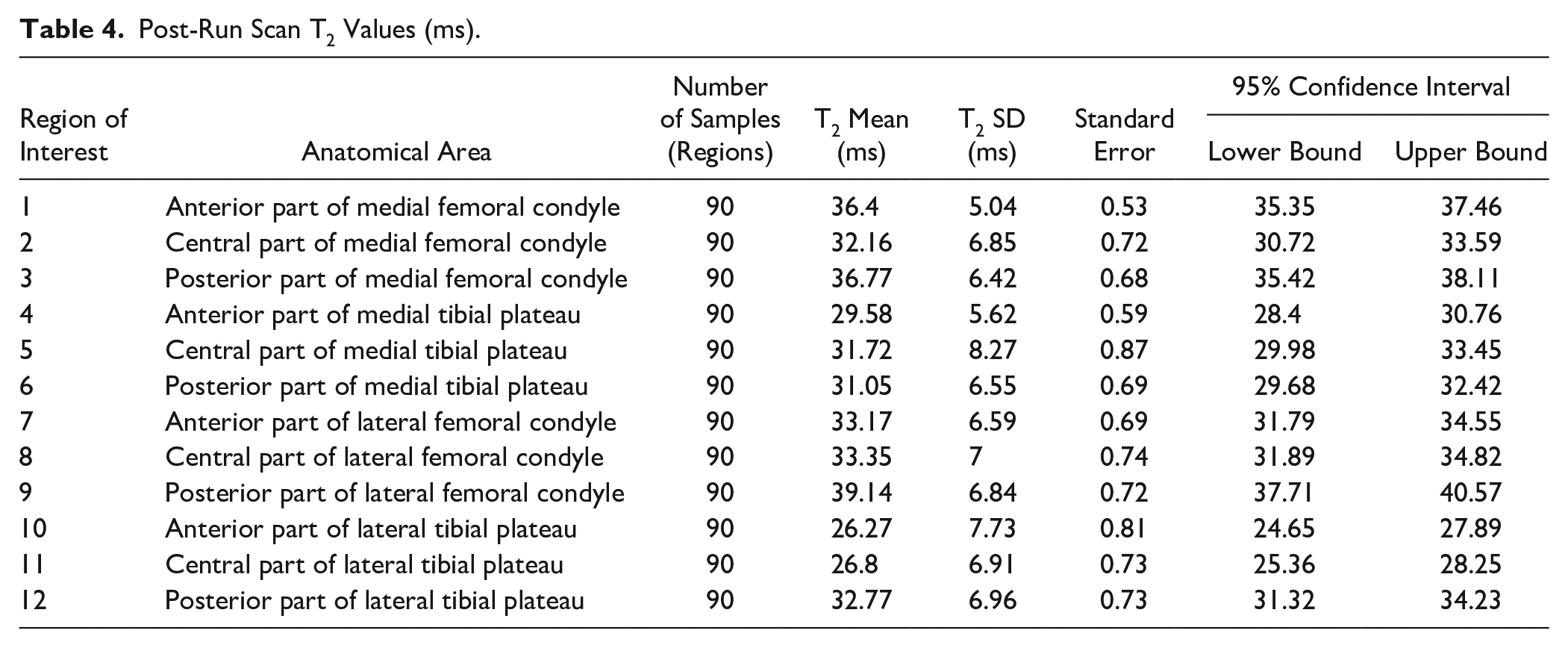
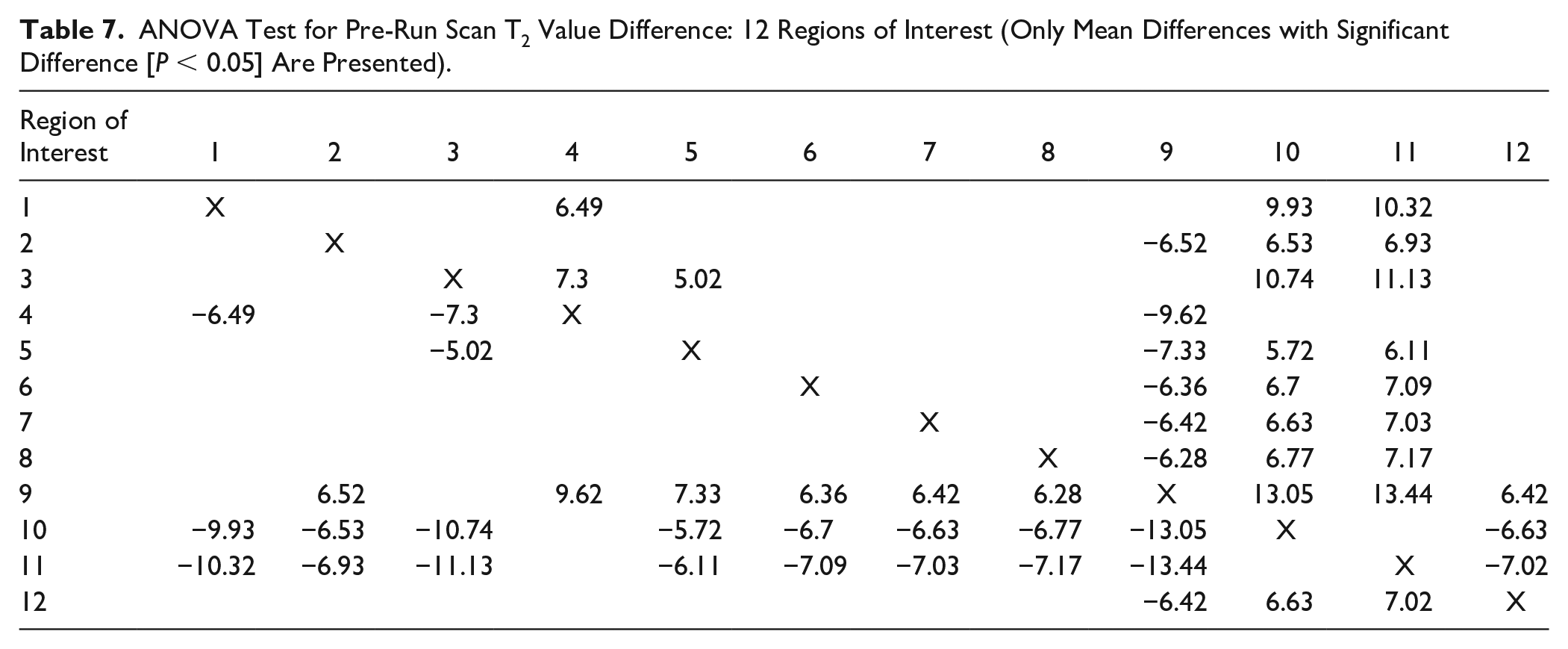
Further analysis was done using ANOVA test and compared 12 ROIs, out of which 3 exhibited statistical trends. Statistically significant difference (P < 0.05) toward lower T2 values in ROI 10 (anterior lateral tibial plateau) and ROI 11 (central lateral tibial plateau) ( Table 7 ). In ROI 9 (posterior lateral femoral condyle) we found a statistical trend toward higher T2 values. We did not reveal significant findings in the medial compartment.
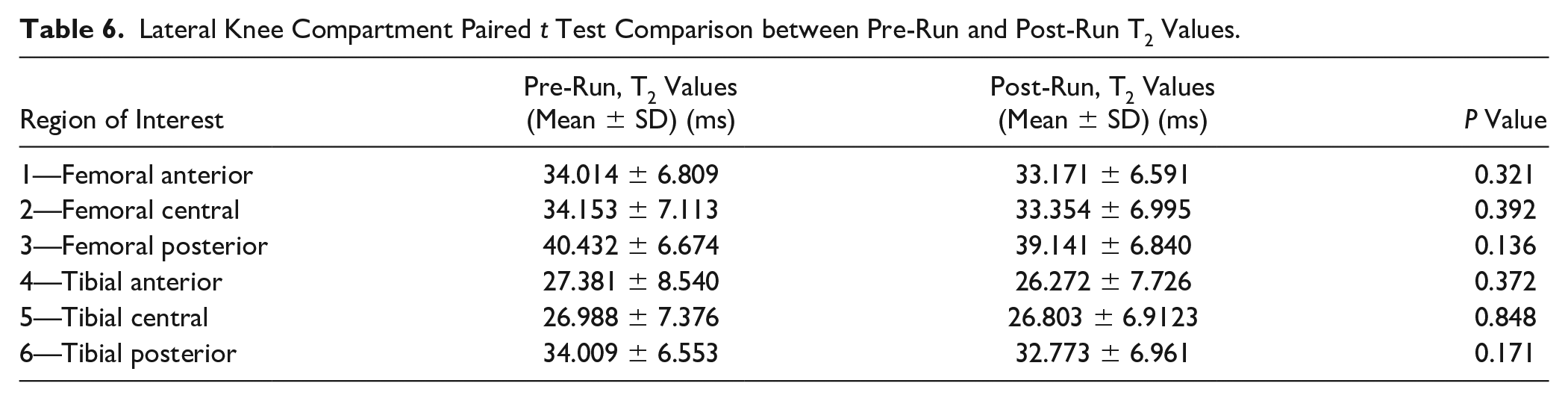
Medial compartment evaluation showed statistically significantly lower T2 values on post-run scans in several ROIs compared to the pre-run scans ( Table 5 ). Significant difference was found in: ROI 2 (pre mean 33.91 ms, post mean 32.16 ms, P = 0.020), representing the cartilage of femoral central weight bearing area of medial compartment (medial femoral condyle); ROI 3 (pre mean 38.12 ms, post mean 36.77 ms, P = 0.038), representing the cartilage of femoral posterior weight-bearing area of medial compartment (medial femoral condyle); ROI 6 (pre mean 34.08 ms, post mean 31.05 ms, P < 0.001), which represents the cartilage of tibial posterior weight-bearing area of medial compartment (medial tibial plateau). Comparison of the lateral regions did not show any statistical significance ( Table 6 ).
Medial Compartment Paired t Test Comparison between Pre-Run and Post-Run T2 Values.
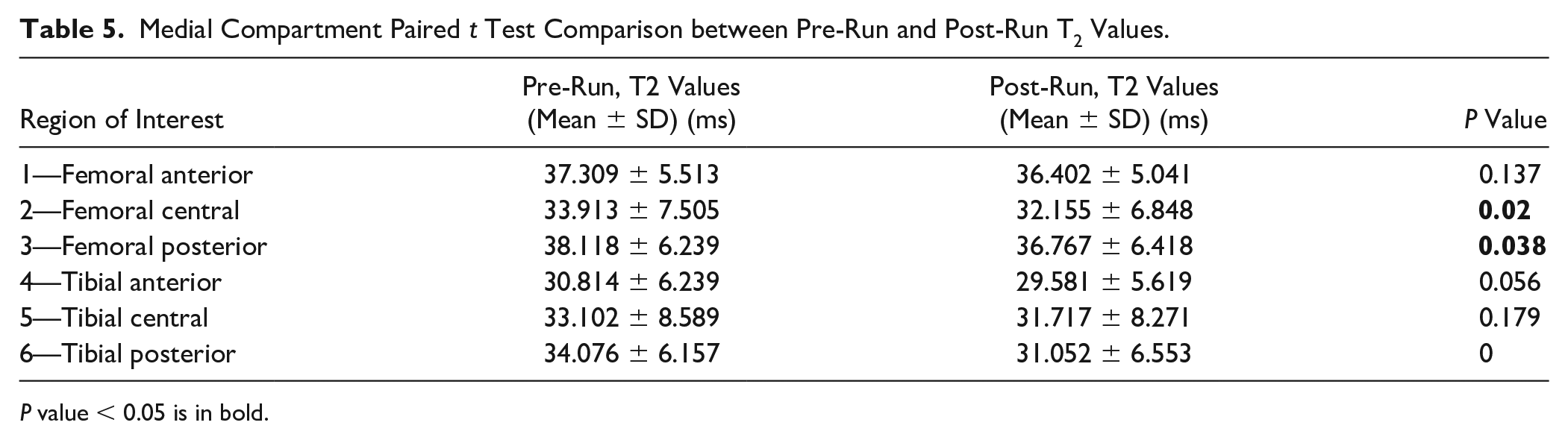
P value < 0.05 is in bold.
Lateral Knee Compartment Paired t Test Comparison between Pre-Run and Post-Run T2 Values.
ANOVA Test for Pre-Run Scan T2 Value Difference: 12 Regions of Interest (Only Mean Differences with Significant Difference [P < 0.05] Are Presented).
Discussion
As the population of young athletes grows, it is important to understand the effect of high-intensity training on their body. Our study assessed the effect of 30 minutes running on the knees of healthy adolescent athletes. To the best of our knowledge this is the first study to explore the latter using quantitative MRI T2 mapping.
The potential of quantitative MRI to serve as a tool for chondral injury detection at an early stage has been demonstrated before. 27 T2 relaxation time is one of the variables that is used to identify articular cartilage abnormalities. 28 Other MRI techniques have also been described as methods to assess articular cartilage changes. For example, Tarabin et al. 29 used the morphological sequences of Double Echo Steady-State (DESS) and True Fast Imaging With Steady-State Precession (TrueFISP) to assess articular cartilage changes after distal radius fracture and showed the potential advantage of this noninvasive method. Advanced MRI techniques were found to be able not only to identify cartilage defects and structural change but also function, reflecting the load-bearing ability of the cartilage. 30
In this study, we identified T2 relaxation time alterations following physical activity, which may represent ultrastructural changes including collagen orientation and water content in the tissue. These changes may be attributed to multiple causes including changes in load distribution during running and chondral alterations.28,31,32
Examination of the pre-run scans revealed differences between ROIs in the lateral compartment (ROIs 9, 10, 11), suggesting that the composition of the articular cartilage differ between regions on the lateral compartment. These findings imply of the intracompartmental structural differences within the cartilage, which are not affected by loading. However, the clinical relevance of such differences remains unclear.
The lack of pre-run differences on the medial compartment increases the reliability of post-run differences in this compartment. The medial-posterior regions of the medial compartment of the knee exhibit the lowest distance of movement during running due to their inherent biomechanical stability (medial posterior root). Following 30 minutes of running we identified significant alterations on three regions of the medial compartment—femoral-central, femoral-posterior, and tibial-posterior ( Table 4 ). These findings were consistent with our hypothesis that the weight-bearing area of articular cartilage of the medial compartment would be prone to experience significant alterations as opposed to other weight-bearing areas in the knee. This can be attributed to the different mechanical loading during running that each region experiences, with the medial compartment experiencing more prominent loads associated with significant mechanical stress. 33
Our findings are consistent with those of Waldenmeier et al., 34 which assessed MRI studies of young professional soccer players following loading tasks. They reported significant alterations of the cartilage in three location: weight-bearing areas of the MFC, the LFC, and on the tibial articular cartilage on the medial compartment. Maximal articular abnormalities were identified on the medial femoral condyle which is also the most common location of chronic cartilage lesions. 34 Although no clinical association can be drawn between our findings and future degenerative changes, it is interesting to acknowledge the articular cartilage on the posterior aspect of the medial condyle was found to be the most affected area in advanced osteoarthritic knees. 35 Our findings are also consistent with a previous study by Goyal et al., 36 who showed that on the tibial surface, the medial, or posteromedial half of the medial tibial plateau, are the most common areas to suffer chondral damage.
Several questions still remain unanswered. First, the ultrastructural changes within the articular cartilage that were reflected by the quantitative T2 values are yet to be understood. The lateral compartment presented changes unrelated to loading, and post-run changes were identified in the medial compartment which are presumably attributed to loading. Second, the long-term effects of the articular changes that occurred in the medial compartment following running are unknown. Third, we do not know how long it takes for the knee and the cartilage to regain their normal appearance and function, and in turn, what is the optimal resting time between practices. Fourth, whether the findings would have been different if the study had examined other patterns of running, such as cutting. Last, is a change to adolescent athlete practices necessary, and if so, what is the optimal training program for these athletes?
Our study has some limitations. First, the study group was not big enough, limiting our understanding of the apparent changes in the articular cartilage. Furthermore, the exclusion criteria for this study were broad, focusing on gender and prior known injuries. This aspect carries a particular risk of T2 distortion due to the inclusion of athletes with unknown cartilage defects. For example, our results showed that several athletes had mild tibial edema in their pre-run exam. Nevertheless, we believe that findings such as these are common even among adolescent athletes and because we focused on assessing the change following a run, the differences are still valid. While it is not suggested that the post-run alterations are necessarily permanent, the biomechanical reasons of these changes may imply or suggest adverse effects of loading in the medial compartment.
In conclusion, our study identified alterations of the articular cartilage in several weight-bearing areas of the medial compartment, both in the femoral and the tibial cartilage, following 30 minutes of running. While these findings do not represent pathology, seeing as no major pathology was identified, they should alert us to abnormal loads that are affecting these areas, potentially making them susceptible to further injury and damage. We believe that as care givers, we owe it to adolescent athletes to protect them as much as we can to avoid acute injuries and long-term disability. Optimization of training programs, enabling adequate recuperation time, and consistent support by trainers and physicians are crucial to achieving this goal. To the best of our knowledge this is the first study to assess the effect of running on the knees of adolescent athletes using quantitative MRI. We encourage further research in order to add to our findings, reaching larger number of examinees, and assess other patterns of physical activity.
Footnotes
Appendix A
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was performed in accordance with the Declaration of Helsinki and the local medical ethical regulations. This study was approved by our institutional review board.
Informed Consent
Informed consent was received from the participants’ parents. No identifiers which could reveal patients’ identities were included.
