Abstract
Objective
The process of anterior cruciate ligament (ACL) injury-induced meniscal tear formation is not fully understood. Clinical studies have shown that ACL reconstruction (ACLR) reduces the development of secondary meniscal tears, but it is difficult to gain insight into the protective effects of ACLR from clinical studies alone. Using rat ACL transection (ACLT) and ACLR models, we aimed to reveal (1) the formation process of meniscal tears secondary to ACLT and (2) the protective effects of ACLR on secondary meniscal tears.
Design
ACLT surgery alone or with ACLR was performed on the knees of rats. Histomorphological and histopathological changes were examined in the posteromedial region of the meniscus in intact rats and in rats that received ACLT or ACLR up to 12 weeks postsurgery. In addition, anterior-posterior joint laxity was measured using the universal testing machine to evaluate the effects of ACLT and ACLR on joint laxity.
Results
AAnterior-posterior laxity was significantly increased by ACLT compared to the intact knee. This ACLT-induced joint laxity was partially but significantly reduced by ACLR. Meniscal proliferation and hyaline cartilage–like tissue formation were detected in the medial meniscus at 4 weeks post-ACLT. At 12 weeks post-ACLT, hyaline cartilage–like tissue was replaced by ossicles and meniscal tears were observed. These ACLT-induced abnormalities were attenuated by ACLR.
Conclusions
Our results suggest that ACLT-induced joint laxity induces secondary medial meniscal tears through meniscal proliferation and ossicle formation via endochondral ossification. Joint re-stabilization by ACLR suppresses meniscal proliferation and ossicle formation and consequently prevents secondary meniscal tears.
Keywords
Introduction
Anterior cruciate ligament (ACL) injuries are one of the most common knee injuries associated with sports. 1 Secondary tears, proliferation (increase of the size), and ossification in the meniscus are often observed in the human and animal knees after ACL injuries.2-9 The meniscus has important roles in knee function, such as load bearing and transmission, shock absorption, stability, lubrication, and congruity. 10 Therefore, meniscal damage in ACL-injured knees likely disrupts joint homeostasis and consequently increases the risk of developing osteoarthritis (OA) in both human and animals.2,11 Prevention of secondary meniscal tears is thus considered a critical issue in the management of patients with ACL injuries. Abnormal mechanical stresses due to joint laxity are considered to play an important role in the development of meniscal tears,12-15 but the mechanisms for the formation of meniscal tears following ACL injury are not fully understood. Moreover, the interactions between meniscal tears, proliferation, and ossification are not well known.
ACL reconstruction (ACLR) surgery is the primary surgical treatment for ACL injury. 1 Although ACLR reduces joint laxity and abnormal mechanical stresses on the meniscus,13,16,17 relatively few studies have examined the protective effects of ACLR on secondary meniscal tears. Previous clinical studies have shown that early ACLR (within 6-12 months after the injury) reduces secondary meniscal tears.4,18 In addition, children who received early ACLR may have a lower number of meniscal tears at the time of ACLR compared with those who received ACLR after failed nonsurgical treatments. 19 However, there is a lack of high-quality prospective studies investigating the outcomes of surgical and nonsurgical treatment for ACL injuries. 19 It is difficult to gain insight into the protective effects of ACLR on secondary meniscal tears at the tissue and cell levels from human clinical studies alone, because research methods in humans are limited to arthroscopic examination and magnetic resonance imaging.4-7
Our aims in this study were (1) to reveal the formation process of meniscal tears secondary to ACL injury and (2) to examine in greater detail the protective effects of ACLR on secondary meniscal tears. To achieve these goals, we examined time-dependent histomorphological and histopathological changes in the meniscus after ACL transection (ACLT) or ACLR in the rat knee. We hypothesized that (1) secondary meniscal tears occur following meniscal proliferation and ossification and (2) restabilization of the knee joint by ACLR prevents tears as well as proliferation and ossification in the meniscus.
Materials and Methods
Experimental Animals
All experimental designs were approved by the animal experimentation committee of Hiroshima International University (Approval Number: AE18-018). In total, 54 male Wistar rats (Japan SCL, Shizuoka, Japan) were used in this study. Forty-three rats (8 weeks old, 190-230 g) were randomly divided into pre-surgery (intact, n = 4), ACLT (n = 19), and ACLR (n = 20) groups. In the pre-surgery group, data from the right and left knees were analyzed as individual samples; therefore, we obtained data from 8 knees in 4 rats. The remaining 11 rats (8-20 weeks old, 210-480 g) were used only for knee joint laxity testing to evaluate the effects of ACL transection and reconstruction on joint laxities. Rats were housed in standard cages in a temperature-controlled room (20-25 °C) with a 12-hour light/dark cycle. Standard rodent food and water were provided ad libitum.
ACL Transection and Reconstruction
ACL transection and reconstruction were performed on the right knee of rats in the ACLT and the ACLR groups, using previously described methods. 20 In brief, anesthesia was induced with an intraperitoneal injection of ketamine (80 mg/kg) and xylazine (10 mg/kg), then the knee joint was opened via a medial parapatellar approach. The ACL was transected using a surgical knife. In the ACLT group, the joint capsule and skin were then closed by nonabsorbable silk sutures. In the ACLR group, bone tunnels were made from the anteromedial side of the proximal tibia to the lateral side of the distal femur using a 0.8 mm diameter Kirschner wire, and were widened with a 21-gauge needle. Then, a quadruple-bundle tail tendon autograft was inserted through the bone tunnels and fixed to the femoral tunnel using a stainless steel interference screw (0.8 mm diameter and 2.0 mm length, TE-00001; Matsumoto, Chiba, Japan). The knee was flexed, and the graft was manually tensioned by pulling, and fixed to the tibial tunnel using a stainless steel interference screw. Finally, the joint capsule and skin were closed. Left knees were untreated. After surgery, rats were allowed to move freely in the cage.
Histological Analysis
Rats were sacrificed by exsanguination under diethyl ether anesthesia at pre-surgery (n = 4), or at 1, 4, or 12 weeks post-surgery (n = 6 or 7 per group per time point). The knees were sampled and immersion-fixed in 0.1 M phosphate-buffered 4% paraformaldehyde (pH 7.4) for 2 days at 4 °C. Then, knees were decalcified by 17.7% ethylenediaminetetraacetic acid (pH 7.2, Osteosoft; Merck Millipore, Darmstadt, Germany) and embedded in paraffin. Sagittal sections (4 µm) were prepared at the medial midcondylar level. We focused our analyses on the posteromedial region of the meniscus, because secondary meniscal tears following ACL injury are most frequently observed in this region. 4 Moreover, meniscal degeneration after ACLT has been observed in the medial meniscus, but not the lateral meniscus, in pigs. 21
Measurement of Meniscus Size
Sections were stained with aldehyde fuchsin-Masson Goldner dye, and the posterior region of the knee was photographed at 2× magnification. The height (i.e., the distance between top and bottom of the outer edge), length (i.e., the distance between the inner edge and central region of the outer edge), and the area of the posteromedial meniscus were measured using ImageJ software (National Institutes of Health, Bethesda, MD; Fig. 1 ).
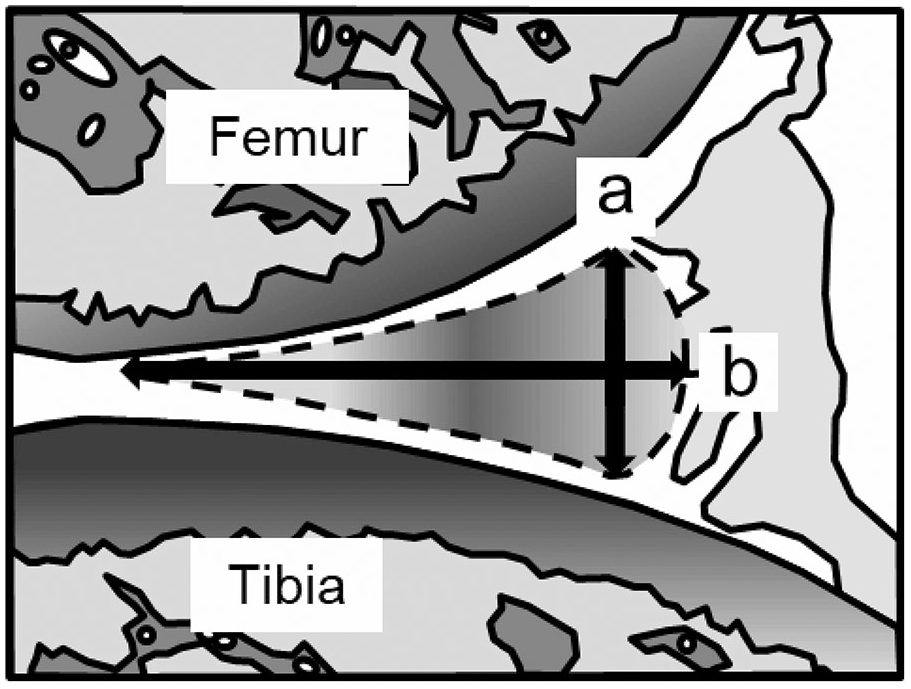
Histomorphometric analysis of the posteromedial meniscus. The height (a), length (b), and area (surrounded by the dotted line) of the posteromedial meniscus were measured.
Histopathologic Scoring
Sections were stained with Safranin-O Fast Green to detect proteoglycans, and a light microscope was used to observe the meniscus. Histopathological scoring in the posteromedial region of the meniscus was performed using Pauli’s scoring system. 22 Six criteria—(1) tissue surface characteristics on the femoral side; (2) tissue surface characteristics on the tibial side; (3) tissue surface characteristics in the inner edge; (4) cellularity; (5) matrix and collagen fiber organization; and (6) Safranin-O Fast Green matrix staining intensity—were scored 0 to 3, where 0 represents normality and 3 represents the worst possible outcome. Following evaluation of each criterion, a total score was calculated.
Immunohistochemistry for Types I and II Collagens
After deparaffinization and rehydration, sections for type I collagen staining were treated with proteinase K (0.05 mol/L; Dako Japan, Tokyo, Japan) for 5 minutes at room temperature, while sections for type II collagen staining were treated with hyaluronidase (25 mg/mL; Sigma-Aldrich, St. Louis, MI) for 60 minutes at room temperature. Then, all sections were rinsed with 0.01 M phosphate-buffered saline (PBS) and incubated with methanol containing 0.3% H2O2 for 30 minutes to quench endogenous peroxidase activity. Sections were incubated with a blocking solution of PBS containing 1% normal horse serum for 30 minutes, and were incubated with either an anti-type I collagen antibody (C2456, 1:2000 dilution; Sigma-Aldrich, St. Louis, MI) or an anti-type II collagen antibody (M193, 25 mg/mL; Takara, Shiga, Japan) overnight at 4 °C. After rinsing with PBS, sections were incubated with the secondary antibody (horse biotinylated anti-mouse IgG, 1:250 dilution; BA-2001, Vector Laboratories, Burlingame, CA) for 30 minutes, followed by incubation with a streptavidin-biotin complex (1:50 dilution; Elite ABC) for 30 minutes. After rinsing with PBS, immunoreactivity was visualized with Dako EnVision + kit/HRP (DAB).
Joint Laxity Testing
To evaluate the effects of ACL transection and reconstruction on joint laxities, anterior-posterior joint laxity testing was performed based on previously described methods. 23 After rats were sacrificed by exsanguination under diethyl ether anesthesia, unilateral knees were harvested and stored at −20 °C until testing. On the testing day, samples were thawed at room temperature (24 °C), and the muscles around the knee were removed. The femur and tibia were inserted into the aluminum pipes (diameter 8 mm) and were rigidly fixed using Kirschner wires. The sample was placed to the universal testing machine (AUTOGRAPH AGS-X 500N; resolution of 0.001 mm, Shimadzu, Kyoto, Japan) at 90° knee flexion ( Fig. 2A ). In humans, the ACL experiences the greatest stress at about 30° knee flexion. 24 In the rodent knees, however, anterior-posterior joint laxity measured at 30° and 90° of flexion is similar. 25 Therefore, we chose 90° of knee flexion as the testing position to test joint laxity. The starting position was set where the force was 0 N. Five loading cycles to a target force of ±5 N at a rate of 0.5 mm/s were applied to the femur ( Fig. 2A ). During testing, the force-displacement data were recorded at a sampling rate of 100 Hz ( Fig. 2B ). The maximum anterior and posterior displacements of the femur in each loading cycle were measured, and means were calculated as anterior (i.e., the posterior displacement of femur with respect to the tibia) and posterior (i.e., the anterior displacement of femur with respect to the tibia) laxities. Joint laxity measurements were repeated immediately following ACL transection and reconstruction ( Fig. 2C ).
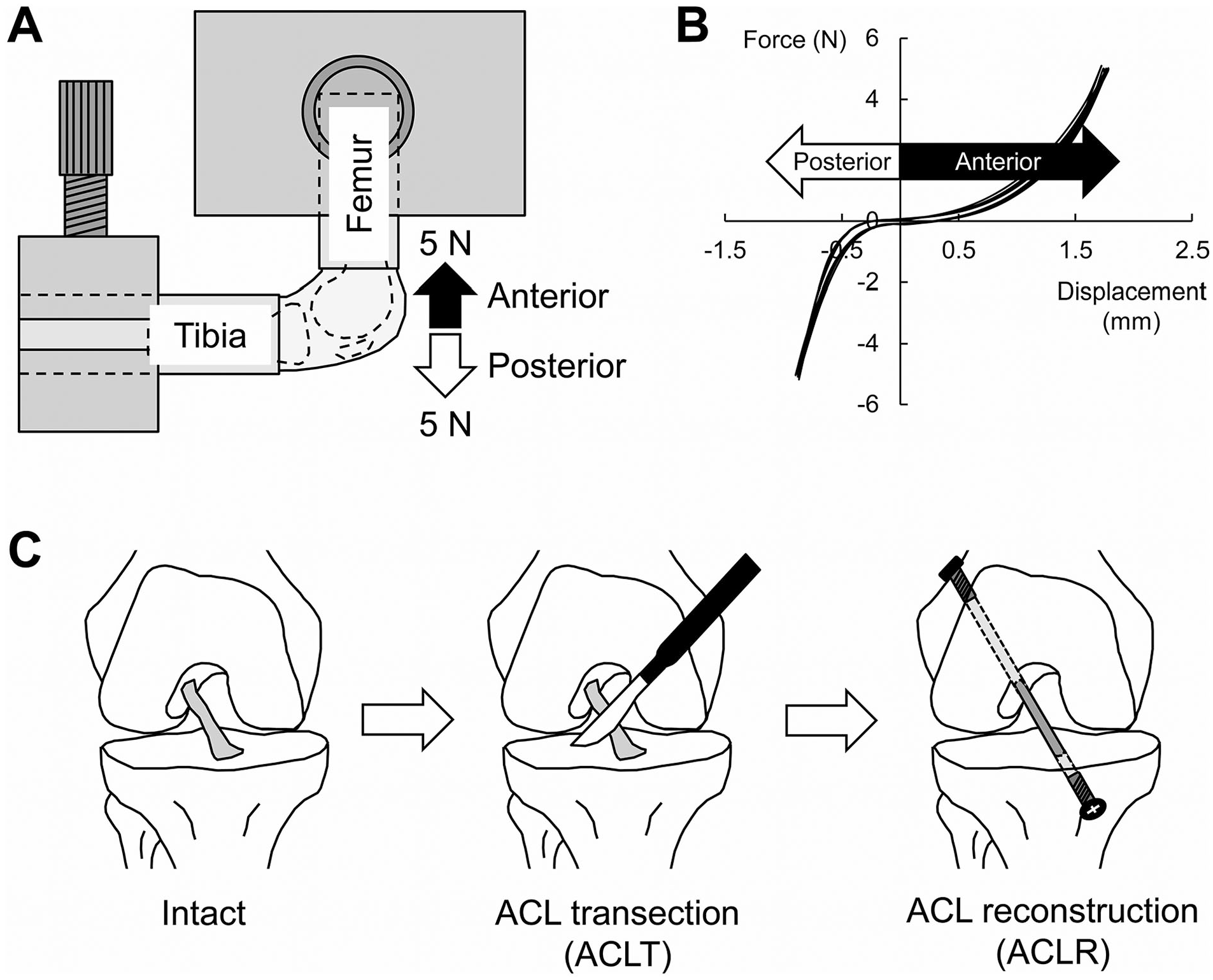
Joint laxity testing. (
Statistical Analysis
Statistical analyses were performed using Dr. SPSS II for Windows (SPSS Japan, Tokyo, Japan). Two-way analyses of variance (ANOVAs) were used to detect significant direct effects or interactions on meniscus sizes. Bonferroni tests were performed post hoc to localize the significant effects, where present. The nonparametric Kruskal-Wallis test followed by the Mann-Whitney test with a Bonferroni adjustment was used to analyze the histopathological score. To analyze joint laxity, the normality of the data was first checked using the Shapiro-Wilk test. Then, one-way repeated-measures ANOVAs, followed by Bonferroni tests, were applied. For all tests, a P value of <0.05 was considered statistically significant.
Results
Joint Laxity
Anterior and posterior laxities in the intact knee were 0.3 ± 0.0 and 0.3 ± 0.1 mm, respectively ( Fig. 3A and D ). After ACLT, both anterior and posterior laxities were significantly greater compared to the intact knee (2.0 ± 0.4 and 1.0 ± 0.3 mm, respectively, P < 0.001, Fig. 3B and D ). ACL reconstruction partially, but significantly, reduced mean anterior laxity to 1.5 ± 0.3 mm (P = 0.005, ACLR vs. ACLT), but there was no change in posterior laxity (1.0 ± 0.4 mm, P = 1.000, ACLR vs. ACLT, Fig. 3C and D ).
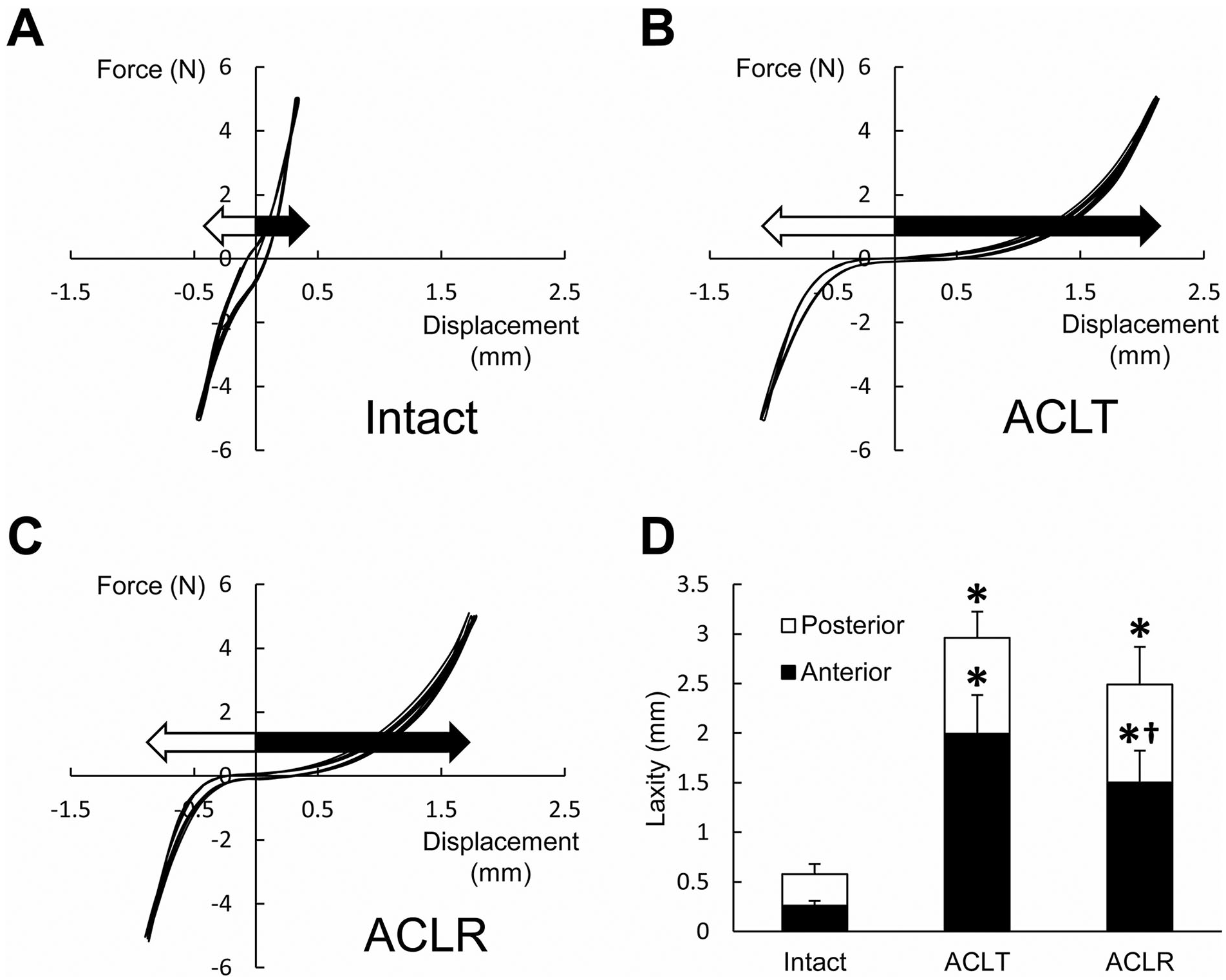
Representative force-displacement curves showing anterior-posterior joint laxity in the (
Meniscus Size
In the pre-surgery group, only the central region of the outer edge of the meniscus was attached to the joint capsule ( Fig. 4A ). However, the entire outer edge of the meniscus was attached to the joint capsule at 1 week post-surgery in the operated (right) knee in both the ACLT and the ACLR groups ( Fig. 4B and E ). The height of the meniscus was unchanged in the ACLT and ACLR groups at 1 week post-surgery (P ≥ 0.468 compared with pre-surgery, Fig. 4B , E , and H ), but rapidly increased at 4 weeks post-surgery (P < 0.001 compared to the pre-surgery group, P < 0.001 compared to 1 week post-surgery, Fig. 4C , F , and H ) and was maintained thereafter ( Fig. 4D , G , and H ). Compared to the ACLT group, meniscus height in the ACLR group was significantly lower at 4 and 12 weeks post-surgery (P = 0.002 and P < 0.001, respectively).
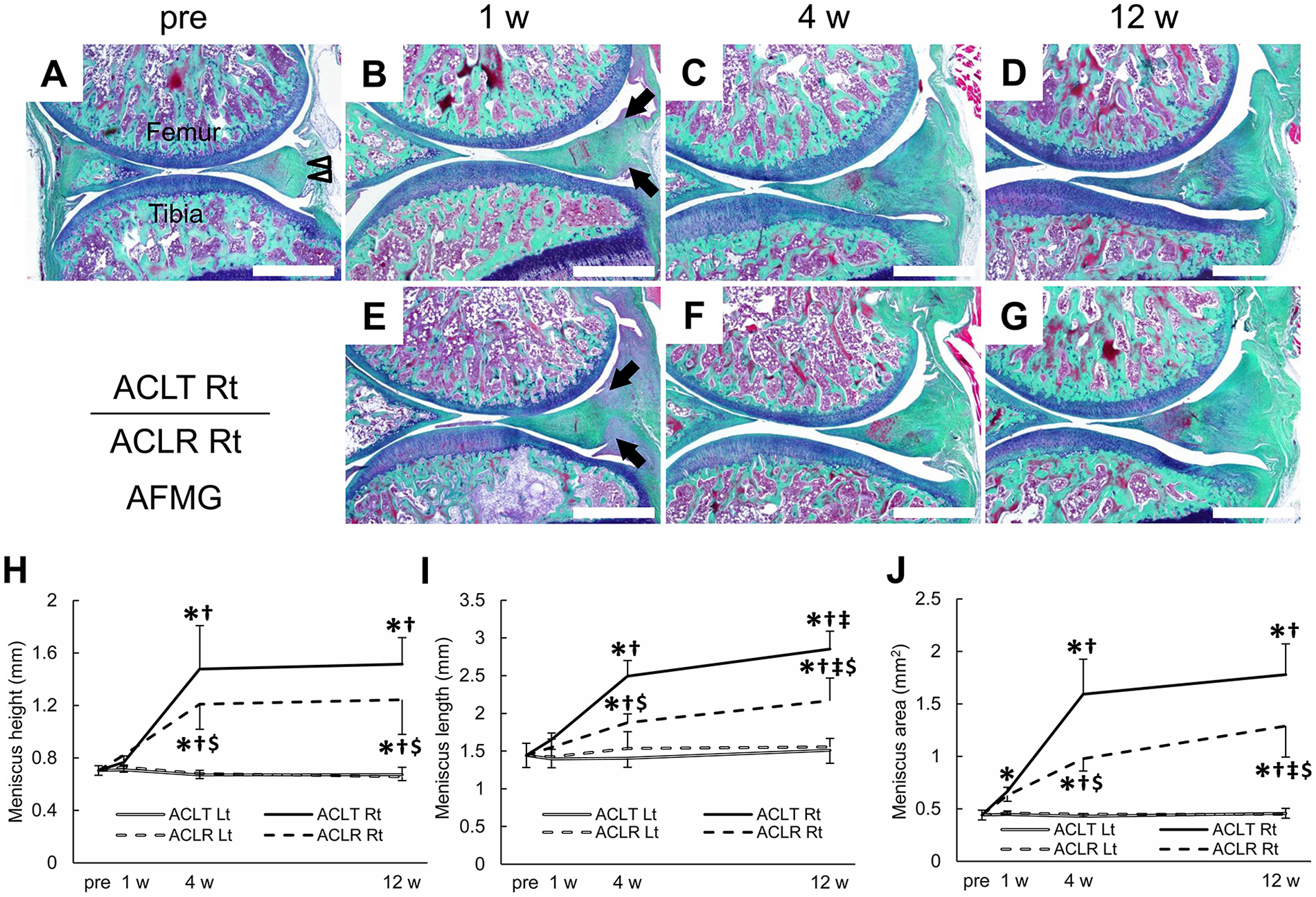
Representative images of the aldehyde fuchsin–Masson Goldner (AFMG)-stained posterior knee joint showing meniscus size for the (
Compared to the pre-surgery group, the meniscus length in the ACLT and the ACLR groups was unchanged at 1 week post-surgery (P ≥ 0.104 compared to pre-surgery, Fig. 4A , B , E , and I ). At 4 weeks post-surgery, the meniscus length in the operated (right) side in the ACLT and the ACLR groups was significantly longer when compared to the pre-surgery group (P < 0.001) and when compared to 1 week post-surgery (P < 0.001 and P = 0.004 in the ACLT and ACLR groups, respectively, Fig. 4C , F , and I ). At 12 weeks post-surgery, meniscus length further increased in the ACLT and the ACLR groups when compared to 4 weeks post-surgery (P = 0.002 and P = 0.015 in the ACLT and ACLR groups, respectively, Fig. 4D , G , and I ). At 4 and 12 weeks post-surgery, the meniscus length in the ACLR group was significantly smaller when compared to the ACLT group (P < 0.001).
At 1 week post-surgery, the meniscus area in the ACLT group (P = 0.014, Fig. 4B ), but not the ACLR group (P = 0.068, Fig. 4E ), was significantly larger compared to the pre-surgery group ( Fig. 4A and J ). At 4 weeks post-surgery, the meniscus area in both groups was significantly larger than that in pre-surgery (P < 0.001) and at 1 week post-surgery (P < 0.001). At 12 weeks post-surgery, the meniscus area in the ACLR group, but not the ACLT group, further increased from 4 weeks post-surgery (P = 0.001 and P = 0.140, ACLR and ACLT, respectively). The meniscus area in the ACLR group was significantly smaller than that of the ACLT group at both 4 and 12 weeks post-surgery (P < 0.001).
The height, length, and area of the meniscus in the untreated contralateral (left) side of the ACLT and ACLR groups were unchanged throughout the experimental period (P = 1.000 compared with pre-surgery, Fig. 4H–J ).
Histopathological Changes
At 4 and 12 weeks post-surgery, the outer region (proximal to the posterior joint capsule) of the proliferated meniscus in the operated side of the ACLT and the ACLR groups was composed of proteoglycan-rich matrix ( Fig. 5A–D ). At 4 weeks post-surgery, spindle-shaped fibroblast-like and round chondrocyte-like cells were both visible in this region in both groups ( Fig. 5E and F ). At 12 weeks post-surgery, chondrocyte-like cells became predominant, and the matrix around the chondrocyte-like cells was intensely stained with Safranin-O in both groups ( Fig. 5G and H ). There were no apparent differences in the pattern of Safranin-O Fast Green staining, or in cell shape, between the ACLT and ACLR groups at both 4 and 12 weeks post-surgery.
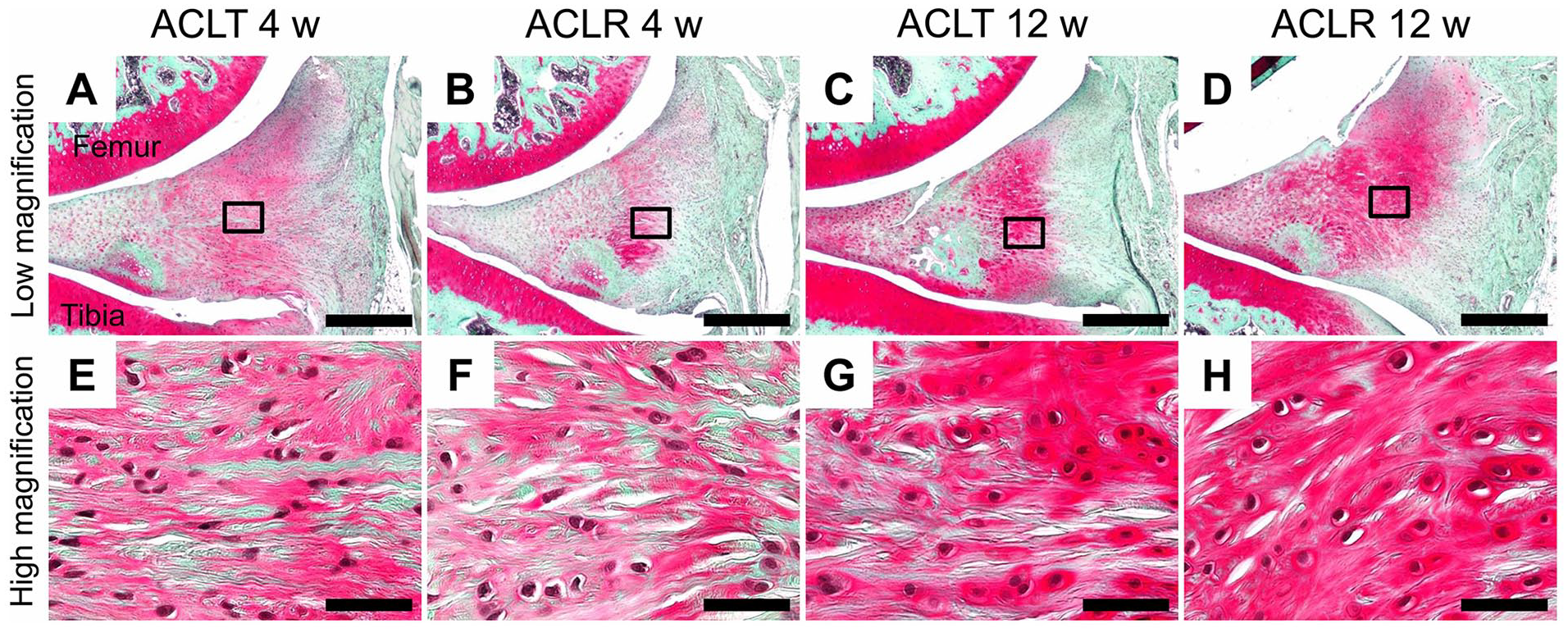
Histopathological features in the outer region of the proliferated meniscus. Representative images of the Safranin-O Fast Green–stained outer region of the proliferated meniscus for (
The meniscus surface of the inner to middle region of the pre-surgery group was smooth, with well-organized collagen fibers ( Fig. 6A ). Compared to the pre-surgery group, there was increased expression of proteoglycans on the tibial side in the ACLT group at 1 week post-surgery ( Fig. 6B ), even though the histopathological scores were not different (P = 1.000, Table 1 ). The histopathological features of the meniscus were nearly identical in the pre-surgery group and the ACLR group at 1 week post-surgery ( Fig. 6E ), with no differences in the histopathological scores (P = 1.000, Table 1 ). At 4 weeks post-surgery, the collagen organization in tibial side of the meniscus in both the ACLT and the ACLR groups was disrupted by formation of hyaline cartilage–like tissue expressing proteoglycan and type II collagen ( Fig. 6C , F , J , and M ). Histopathological scores in the ACLT group was significantly higher (P = 0.018, Table 1 ) than the pre-surgery group, while there was a trend for an increase in the ACLR group (P = 0.120, Table 1 ). At 12 weeks post-surgery, hyaline cartilage–like tissue in the ACLT group was replaced by an ossicle characterized by expression of type I collagen and presence of marrow cavities, and tears adjacent to the ossicle were observed ( Fig. 6D and R , Suppl. Fig. 1). In addition, hypocellular regions were detected around these tears ( Fig. 6D ). Consequently, the histopathological score in the ACLT group at 12 weeks post-surgery was significantly higher than the scores at pre-surgery and at 1 week post-surgery (P = 0.006 for both comparisons), and tended to be higher than the score at 4 weeks post-surgery (P = 0.090, Table 1 ). Hyaline cartilage–like tissue was also observed in the ACLR group at 12 weeks post-surgery ( Fig. 6G and N ), but ossicle and tears were not detected throughout the experimental period. The histopathological score in the ACLR group tended to be higher at 12 weeks post-surgery compared to the pre-surgery group (P = 0.054, Table 1 ). Furthermore, histopathological scores at 12 weeks post-surgery were significantly lower in the ACLR group than the ACLT group (P = 0.015).
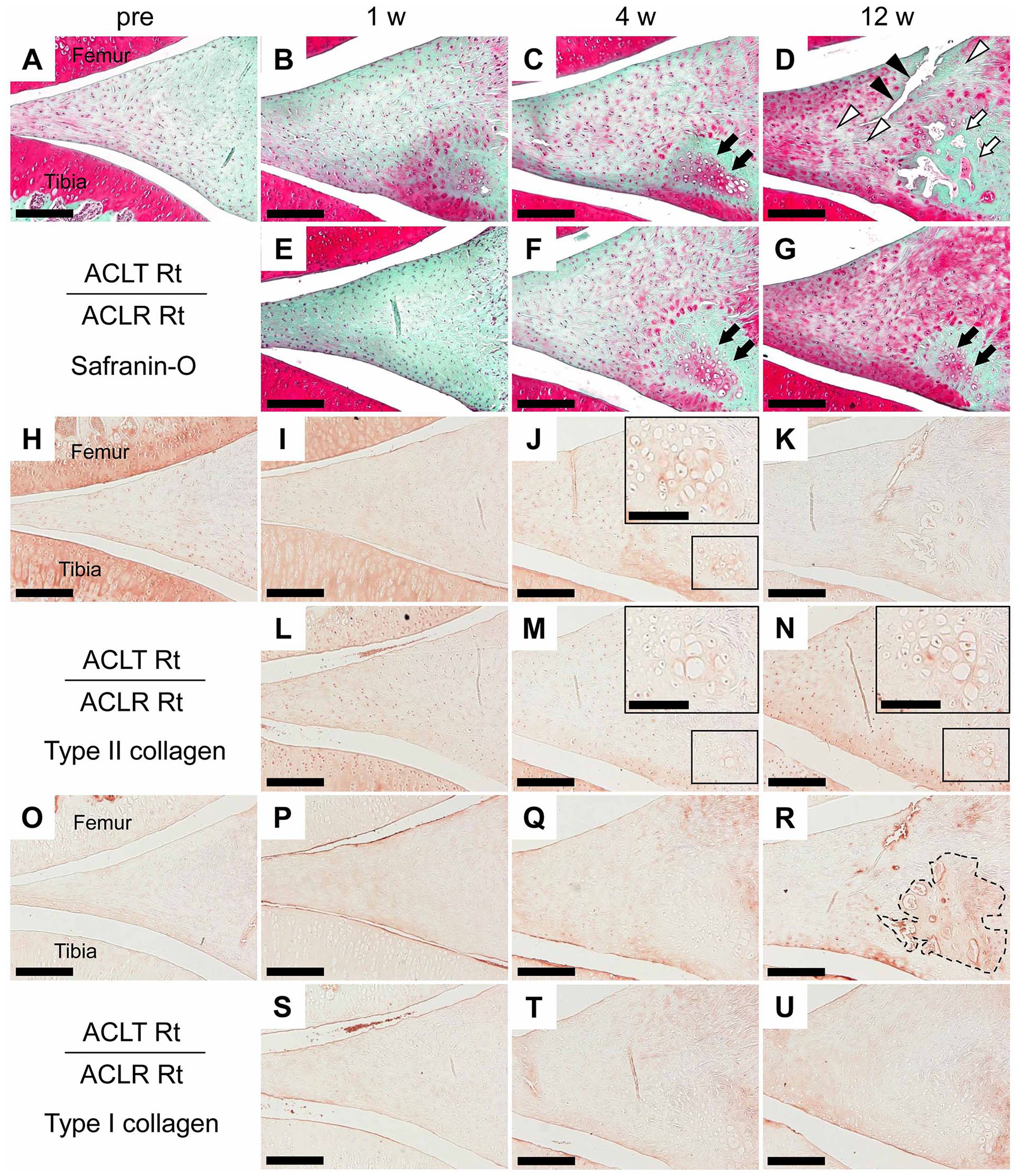
Histopathological features in the inner to middle region of the meniscus. Representative images of the Safranin-O Fast Green–stained sections from the (
Histopathological Scores in the ACLT and ACLR Groups a .
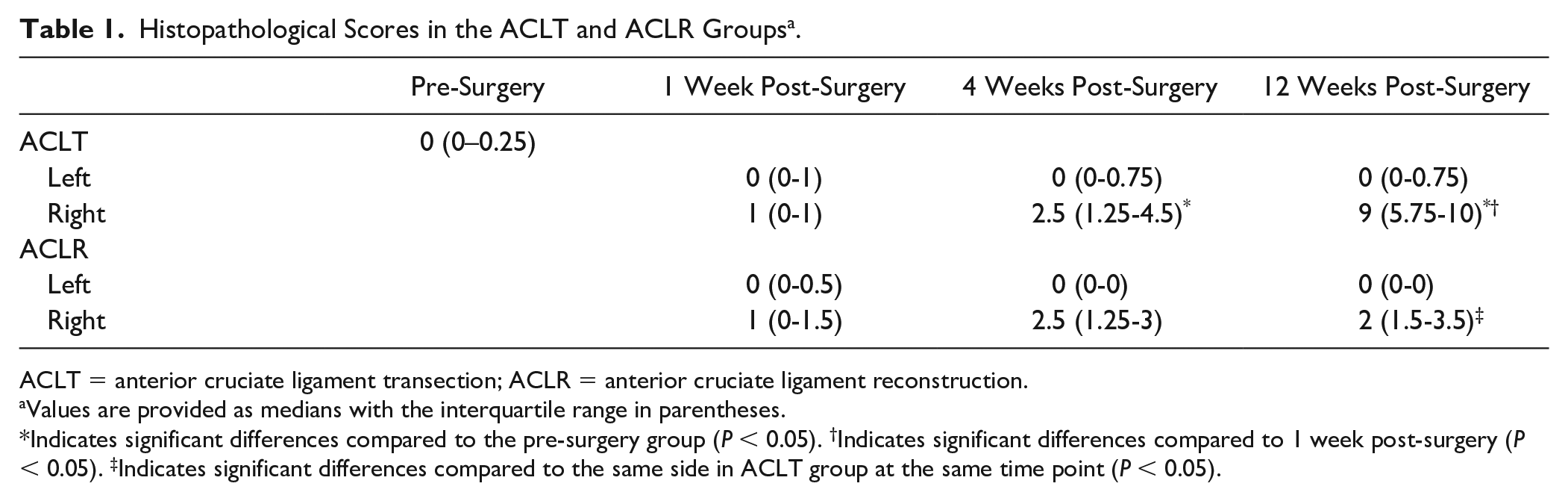
ACLT = anterior cruciate ligament transection; ACLR = anterior cruciate ligament reconstruction.
Values are provided as medians with the interquartile range in parentheses.
Indicates significant differences compared to the pre-surgery group (P < 0.05). †Indicates significant differences compared to 1 week post-surgery (P < 0.05). ‡Indicates significant differences compared to the same side in ACLT group at the same time point (P < 0.05).
Histopathological features in the untreated contralateral (left) side of the ACLT and ACLR groups were unchanged throughout the experimental period (data not shown), and histopathological scores in the contralateral side were similar to that of pre-surgery (P = 1.000, Table 1 ).
Discussion
The most important finding of this study was that secondary meniscal tears after ACLT in a rat knee occurred serially or in parallel to meniscal proliferation and ossicle formation. Our results suggest that ACLT results in secondary meniscal tears through laxity-induced meniscal proliferation and ossicle formation.
The ACL is a critical stabilizer of the knee, 26 and its injury increases anterior-posterior laxity. 27 Using mechanical testing, we confirmed that ACLT induced an increase in anterior-posterior laxity. Joint laxity induced by ACL injury increases mechanical stress on the meniscus.12-15 For example, using freshly amputated human knees, Sakamoto et al. demonstrated that contact pressure (i.e., compressive stress) in the posteromedial regions of the meniscus that are under anterior load to the tibia is increased by ACLT. 15 In addition, previous studies reported that abnormal compressive stress induces proliferative changes in the meniscal after ACLT in guinea pig knees in vivo and bovine meniscus explants in vitro. 2 Meniscal proliferation after ACLT was also detected in our study; the meniscus area was enlarged via increases in both height and length at 4 weeks post-surgery. This meniscal proliferation was not due to growth, because meniscus sizes did not change in the untreated contralateral side throughout the experimental period. Meniscal proliferation would induce further increases in compressive stress.
In our study, the outer edge of the meniscus was attached to the joint capsule in the narrow area at pre-surgery. Similarly, in human cadaveric knee without pathologies, the posterior joint capsule does not attach directly to the superior margin of the medial meniscus and attaches inferior to the superior margin of the medial meniscus at a mean depth of 36.4% of the total meniscus height. 28 However, the amount of contact between the outer edge of the meniscus and joint capsule substantially increased at 1 week post-ACLT, and meniscal proliferation progressed rapidly thereafter. ACL injury is frequently accompanied by meniscal ramp lesion, which is tear of the posteromedial meniscus at the meniscocapsular attachment. 29 Ramp lesion increases anterior laxity in the ACL-injured knees, and repair of ramp lesion with ACLR decreases anterior laxity. 30 These imply that attachment between the meniscus and joint capsule acts as a secondary stabilizer for anterior laxity and receives the tensile stress in ACL-injured knee. This tensile stress might activate fibroblast proliferation and subsequent production of fibrous tissue, 31 which increased contact between the meniscus and joint capsule. In the outer region (proximal to the posterior joint capsule) of the proliferated meniscus, spindle-shaped fibroblast-like and chondrocyte-like cells were equally prevalent at 4 weeks post-surgery, but chondrocyte-like cells predominated at 12 weeks post-surgery. This outer region was composed of a proteoglycan-rich matrix. These results suggest that increased mechanical stress stimulated the differentiation of synovial fibroblast-like cells located in the border between the meniscus and joint capsule into chondrocytes, which produced matrix components, such as proteoglycans, that in turn contributed to meniscal proliferation. Nevertheless, the origin of the cells participating in the meniscal proliferation after ACLT is not well understood. We speculate that synovial fibroblast-like cells, including mesenchymal stem cells, contributed to meniscal proliferation. These cells can differentiate into chondrocytes and take part in the meniscal regeneration.32-34 Mechanical stress is known as a stimulus for the differentiation of synovial fibroblast-like cells into chondrocytes.32,33 For example, Sakao et al. reported that intermittent hydrostatic pressure facilitated cultured rabbit fibroblast-like cells differentiation into chondrocytes. 33
In our study, ossicle formation characterized by type I collagen expression and presence of marrow cavities was observed 12 weeks post-ACLT on the tibial side of the meniscus, which experiences high compressive stress in the ACL-transected knee. 14 Although the prevalence of the meniscal ossicle in patients with ACL injury is unknown, the majority of patients with meniscal ossicle have abnormal ACL or evidence of prior ACLR. 35 Previously, Xie et al. observed increase in type X collagen and runt-related transcription factor 2 proteins and alkaline phosphatase activity, which are associated with ossification, in the murine meniscus after ACLT. 36 Du et al. revealed that meniscal ossification is induced after ACLT in guinea pig knees. 2 They also demonstrated that abnormal compressive stress upregulates calcification-related factors in bovine meniscus explants, and thus suggested that abnormal compressive stress induced by an ACLT results in meniscal ossification. 2 In our study, prior to ossicle formation, hyaline cartilage–like tissue expressing proteoglycan and type II collagen was detected in the same region of the meniscus at 4 weeks post-ACLT. A series of these changes indicated that abnormal mechanical stress induced by ACLT results in ossicle formation through endochondral ossification. Hyaline cartilage–like tissue and ossicle formation observed after ACLT was not due to aging, because these histopathological abnormalities were not detected in the untreated contralateral side throughout the experimental period. In human, it has been proposed that endochondral ossification due to metaplasia of fibrocartilage contributes to ossicle formation. 35 Osteophyte formation in the peripheral regions of the bones is an important feature in osteoarthritic joints. Although the mechanisms of osteophyte formation in OA are not fully understood, endochondral ossification induced by mechanical stress is suggested to contribute osteophyte formation.37,38 Thus, abnormal mechanical stress may be a common mechanism in ossicle formation in the meniscus after ACL injury and osteophyte formation in OA.
In parallel with ossicle formation, we observed that meniscal tears occurred adjacent to the ossicle. Mohankumar et al. previously reported that similar adjacent tears were found in most of the meniscus containing an ossicle in humans. 35 It is considered that calcification of the meniscus alters the biomechanical properties, possibly increasing the susceptibility to tears. 39 Therefore, mechanical stresses attributed to joint laxity on the meniscus containing an ossicle would trigger the development of tears, although we could not demonstrate the causal relationship between ossicle formation and meniscal tears. Schematic diagrams of the formation processes of meniscal tears after ACLT suggested by our results are shown in Figure 7 .
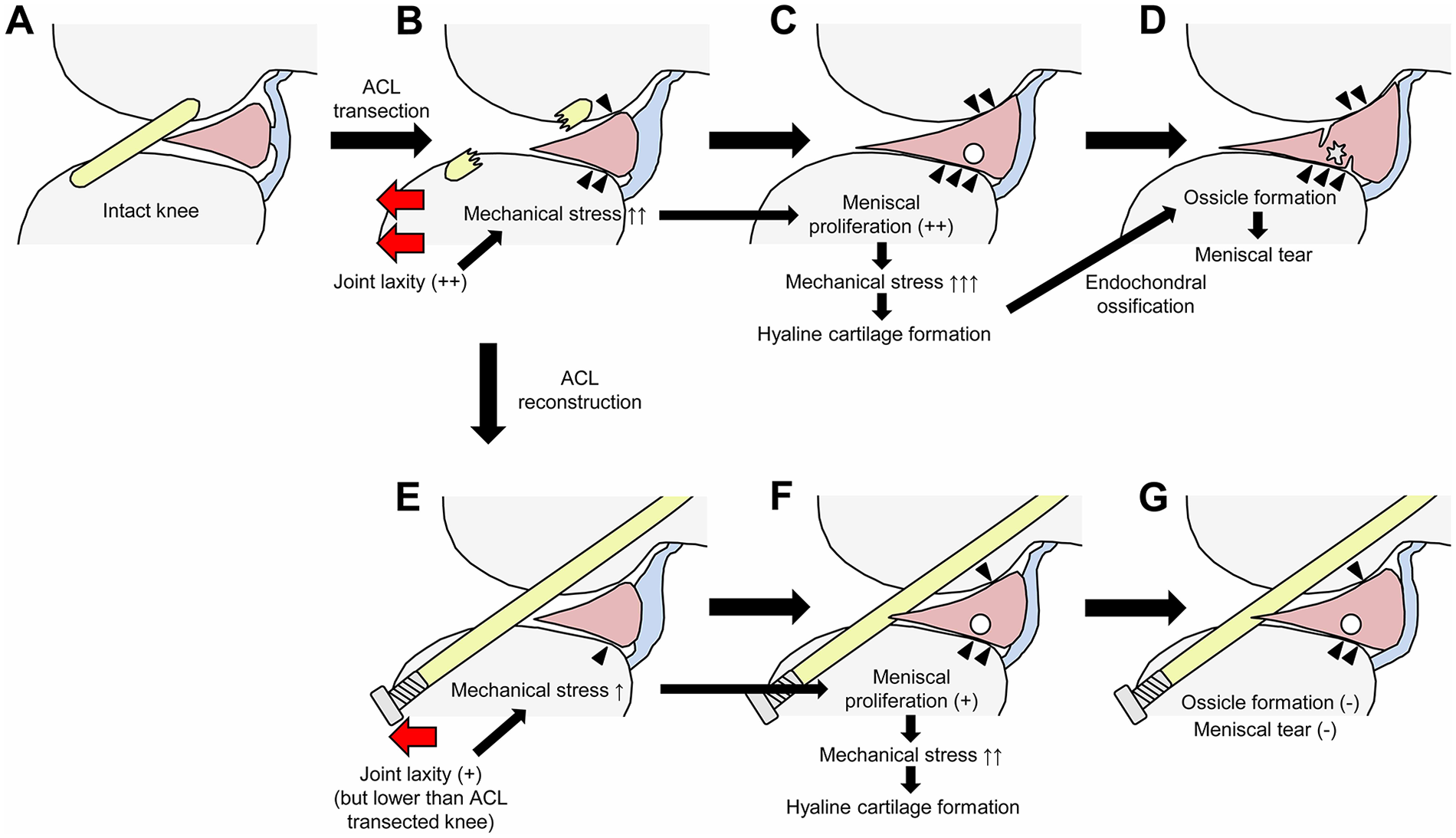
Schema illustrating the processes of anterior cruciate ligament (ACL) injury-induced meniscal tears. ACL transection (ACLT) induces joint laxity, which increases mechanical stress (indicated by arrowheads) on the meniscus (
The second aim of this study was to closely examine the protective effects of ACLR, which is the most common surgical treatment for ACL injury, 1 on secondary meniscal tears using histomorphological and histopathological methods. ACLR reduces joint laxity, which induces abnormal mechanical stresses on the meniscus,13,15-17 and consequently decreases the risk of secondary meniscal tears.4,18 ACL injury-induced joint laxity in humans is almost completely reversed by ACLR, 16 whereas the small body size of rats poses some technical difficulties to surgical precision. 40 ACLT-induced joint laxity was significantly reduced by ACLR in our study, but residual laxity remains even after ACLR. Nevertheless, ACLR partially attenuated ACLT-induced meniscal proliferation, and prevented ossicle formation and secondary meniscal tears. These results indicate that even partial restabilization of the knee joint may prevent meniscal tears following ACL injury ( Fig. 7 ).
Meniscal ossification is associated with development of OA, and when ossicle is accompanied by tear, surgical excision may be needed to prevent OA. 41 Thus, suppression of meniscal tears following ossification is important to prevent OA. Although evidence from the literature regarding the effectiveness of ACLR in preventing the development of knee OA is controversial, ACLR may nevertheless reduce the risk of knee OA.18,42 Suppression of secondary meniscal tears by ACLR may contribute to the prevention of OA in the ACL-injured knee.
This study has limitations. First, the follow-up period was limited to 12 weeks post-surgery. Longer follow-up studies will be needed to determine whether ossicle and tears in the meniscus eventually also developed in knees that underwent ACLR. Second, there was residual laxity even after ACLR in the rat knees, as mentioned above. This residual laxity would contribute at least in part to pathological changes observed after ACLR. Third, it is a small animal model so its clinical relevance to the natural history of humans is not certain. Fourth, we did not perform analyses of the lateral region of the meniscus. To confirm whether the responses of the meniscus after ACLT are different between the medial and lateral regions in our animal model, further studies are needed. Fifth, ACLT increased not only the anterior laxity, but also the posterior laxity. We consider that increase in posterior laxity was due to change in the starting position of the mechanical testing (i.e., the posterior displacement of femur with respect to the tibia) rather than posterior cruciate ligament injury.
In conclusion, our results suggest that ACLT in the rat knee induces secondary meniscal tears through meniscal proliferation and ossicle formation via endochondral ossification. Joint restabilization by ACLR suppressed further development of meniscal proliferation and ossicle formation and consequently prevented secondary meniscal tears.
Supplemental Material
sj-jpg-1-car-10.1177_19476035211014588 – Supplemental material for The Natural History of Medial Meniscal Tears in the ACL Deficient and ACL Reconstructed Rat Knee
Supplemental material, sj-jpg-1-car-10.1177_19476035211014588 for The Natural History of Medial Meniscal Tears in the ACL Deficient and ACL Reconstructed Rat Knee by Akinori Kaneguchi, Junya Ozawa, Kengo Minamimoto and Kaoru Yamaoka in CARTILAGE
Footnotes
Authors’ Note
This study was performed in the Hiroshima International University.
Author Contributions
AK and JO contributed to the study design, analysis, and interpretation of data. AK, JO, and KM contributed to the acquisition of data. AK, JO, and KY contributed to the manuscript preparation. All authors read and approved the final manuscript.
Acknowledgments and Funding
We acknowledge technical assistance from Mayuki Arataki, Yudai Ishikawa, Yusuke Ueno, Takahiro Uchiyama, Yoshiko Kato, and Kei Takemaru. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by JSPS KAKENHI Grant Number 18H06427.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All experimental designs were approved by the animal experimentation committee of Hiroshima International University (Approval Number: AE18-018).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
