Abstract
Objective
Excessive use of glucocorticoids (GCs) may cause adverse effects on the skeletal system in children. However, only a few studies have reported the effects of GCs on the epiphyseal cartilage. This study aimed to uncover the subsequent epiphyseal cartilage changes of immature femoral heads after excessive GC treatment in a mouse model and explain the pathological changes preliminarily.
Design
Female C57BL/6 mice were divided into control and model (excessive GC treatment) groups. The structure of the femoral heads was evaluated by using micro-computed tomography, hematoxylin-eosin staining, and safranin staining analyses. Immunohistochemistry was used to detect angiogenesis and cartilage metabolism. Western blotting and TUNEL staining were used to examine epiphyseal cartilage chondrocyte apoptosis. Primary chondrocytes were isolated from the femoral heads of healthy mice for in vitro studies. The effects of GCs on chondrocyte apoptosis and metabolism were determined by flow cytometry and Western blotting.
Results
The epiphyseal cartilage ossification had started at 4 weeks posttreatment in a portion of mice; the ossification presented as a sequential process in the model group, while the epiphyseal cartilage maintained an unossified state in the control group. Vascular invasion into the epiphyseal cartilage of the model mice was observed at 4 weeks posttreatment. GCs induced chondrocyte apoptosis and altered chondrocyte metabolism in the epiphyseal cartilage.
Conclusions
The epiphyseal cartilage ossification accelerated in the femoral heads of female C57BL/6 mice after excessive GC treatment. Increased chondrocyte apoptosis, altered chondrocyte metabolism, as well as increased vascular invasion, are the potential factors influencing epiphyseal cartilage ossification.
Introduction
Glucocorticoids (GCs) are potent immunosuppressant drugs that are used after organ transplantation and also for treating inflammatory diseases such as asthma, allergic shock, and inflammatory bowel disease, and malignant diseases such as acute lymphoblastic leukemia.1-3 However, large doses or prolonged use of GCs may cause adverse events, including muscle atrophy, glaucoma, cataract, insulin resistance, depression, and severe effects on the skeletal system.1,4,5 Children are especially susceptible to the side effects. A study found that patients with ulcerative colitis, treated with GCs, had higher incidences of adverse events occurring in children than in adults. 6 Furthermore, as the skeletal system of children is immature, the adverse effects of GCs on the bone and cartilage have a significant impact on children’s growth and development.7,8
Growth retardation and osteoporosis are 2 of the most high-profile adverse effects of GCs in children, and many researchers have investigated the underlying mechanisms. Excessive use of GC disturbs the balance between apoptosis and proliferation of chondrocytes in the growth plate, thus disrupting longitudinal growth and resulting in growth retardation.1,9 Besides, the overuse of GC may induce osteoporosis.1,10-12 The pathophysiology of glucocorticoid-induced osteoporosis, including systemic effects and local effects on all cell types in bone tissue, is complicated.13-15 Previous studies have focused on the growth plates and bone mass, but few have paid attention to the epiphyseal cartilage, which is responsible for shaping the epiphysis and is essential for joint plasticity in children.16,17
The epiphyseal cartilage exists under the permanent articular cartilage and above the growth plate. It is composed of chondrocytes and gradually ossifies as the child matures. 18 For instance, on an average, the epiphyseal cartilage of the femoral head of the proximal femur completes ossification at 12.6 and 11.3 years of age in males and females, respectively. 19 The effects of GC on chondrocytes are complex. For example, the growth plate consists of 3 distinct zones, namely, the resting zone, proliferation zone, and the hypertrophic zone. GC inhibits the proliferation of chondrocytes in the resting and proliferation zones. Moreover, GC induces apoptosis in proliferating and hypertrophic chondrocytes, leading to compromised growth plate integrity. However, on the maturation of hypertrophic chondrocytes, contradictory effects of GC have been reported.1,20
Because the effects of GCs on chondrocytes are complicated and the abnormal development of epiphyseal cartilage leads to joint deformity,21,22 it is necessary to investigate the side effects of excessive GC on the epiphyseal cartilage. In the present study, we aimed to uncover the epiphyseal cartilage changes in immature femoral heads following excessive GC treatment in mice to explain the pathological changes preliminarily.
Materials and Methods
Animals
A total of 60 healthy 5-week-old female C57BL/6 mice were purchased from the Hubei Provincial Center for Disease Control and Prevention (Wuhan, China). The mice were housed in cages under standard laboratory conditions with a 12-hour light and 12-hour dark cycle (20 °C, 48% humidity) and fed a standard rodent diet (Agway RMH 3000, Arlington Heights, IL, USA). Food and water were provided ad libitum. Twelve mice were randomly selected for the extraction of chondrocytes. The remaining 48 mice were randomly divided into 2 groups (n = 24 per group), namely, the control and model groups. Investigators were blinded during allocation, animal handling, and endpoint measurements except for the person responsible for grouping and treating the mice. The mice in the model group were subcutaneously injected with 21 mg/kg methylprednisolone (MPS; Pfizer Manufacturing Belgium NV, Brussels, Belgium) for 4 weeks,23,24 while the mice in the control group were injected with equal volumes of phosphate-buffered saline (PBS). Eight mice from each group were euthanized under anesthesia, and the femoral head tissue was collected at 4, 6, and 8 weeks after the start of the treatment. All animal experiments were conducted following the Guide for the Care and Use of Laboratory Animals (2018), and the study was conducted following protocol approval (No. bfr2018003) by the Animal Experimentation Committee of Hubei Provincial Center for Disease Control and Prevention (Wuhan, China).
Chondrocyte Isolation and Treatment
Primary chondrocytes were isolated from femoral heads of female C57BL/6 mice, as described previously.
25
The femurs were dislocated, and the femoral heads were isolated. Pieces of cartilage were collected under a dissecting microscope and rinsed with PBS. Dulbecco’s modified Eagle’s medium supplemented with 2 mmol/L
Flow Cytometry
Flow cytometry was used to detect the rate of chondrocyte apoptosis. Chondrocytes were collected from each group, centrifuged at 225 × g for 5 minutes, and washed twice with PBS. The rate of apoptosis was determined according to the instructions of the Annexin V-FITC Apoptosis Detection Kit (BD Biosciences Pharmingen, San Diego, CA, USA). Briefly, the chondrocytes were resuspended in 500 μL binding buffer, and 5 μL annexin V-FITC was then added followed by 5 μL propidium iodide. The cells were incubated in the dark for 15 minutes at 20 °C. The negative control group was not treated with antibodies. The cells were then analyzed using a flow cytometer (FACSCalibur; BD Biosciences, Beijing, China).
Western Blot Analysis
The cartilage of femoral heads was obtained using methods previously described and washed twice with PBS at 0 °C. 25 Total proteins of the chondrocytes were extracted by incubation with radioimmunoprecipitation assay (RIPA) lysis buffer (P0013B; Beyotime Biotechnology Co., Ltd., Wuhan, China) containing phenylmethane sulfonyl fluoride (PMSF; ST506, Beyotime Biotechnology Co., Ltd.) for 30 minutes. Total protein was also eluted from the culture supernatants. Protein concentrations were measured using a bicinchoninic acid (BCA) Protein Assay Kit (P0010; Beyotime Biotechnology Co., Ltd.). Equivalent amounts of protein (40 μg) were then separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membranes (IPVH00010; EMD Millipore, Billerica, MA, USA). The PVDF membranes were blocked for 2 hours with Tris-buffered saline, and Tween (TBST) buffer containing 5% skim milk. Membranes were then incubated at 4 °C overnight in a solution containing either rabbit anti-caspase-3 polyclonal antibody (1:500; Proteintech Group, Inc., Wuhan, China) for detection of tissue proteins or with rabbit anti-MMP-13 polyclonal antibody (bs-10581R; 1:800; Bioss, Beijing, China) and mouse VEGF-A164 antibody (AF-493-NA; 1:1000; R&D Systems, Shanghai, China) for detection of chondrocyte proteins. Rabbit GAPDH polyclonal antibody (1:1000; Xianzhi Biotech, Hangzhou, China) served as a loading control. Membranes were then washed 6 times with TBST for 5 minutes and incubated at 37 °C for 2 hours with horseradish peroxidase (HRP)-labeled sheep anti-rabbit secondary antibodies (31460; 1:50000, ThermoFisher Scientific, Shanghai, China). Electrochemiluminescence detection reagent (EMD Millipore) was used to detect the antibodies according to the manufacturer’s instructions. The films were scanned, and Bandscan software 5.0 (Glyko, Novato, CA, USA) was used to analyze the grayscale values.
Micro-Computed Tomography Scanning
Femoral heads were scanned using the SkyScan 1172 high-resolution desktop X-ray microtomography (micro-CT) system (Bruker microCT, Kontich, Belgium) with voltage 70 kV, pixel size 9 μm, and filter Al 0.2 mm. We obtained 3-dimensional reconstruction images and the images of femoral heads bisected along the central longitudinal section by using CTvol software (Bruker microCT). The bone volume/tissue volume ratio (BV/TV) of each femoral head was calculated using CTan software (Bruker microCT).
Histology
Femoral head samples were fixed in PBS containing 4% paraformaldehyde and decalcified with 10% ethylenediaminetetraacetic acid (EDTA) solution. The samples were then dehydrated, embedded in paraffin, and longitudinally cut into 4 μm thick sections. Slides were incubated at 56 °C for 60 minutes and immersed in xylol to remove the paraffin. The tissue sections were then rehydrated in a graded alcohol series (100%, 95%, 90%, 80%, and 70%) for 5 minutes each, and rinsed in distilled water. The sections near the round ligament were chosen and stained with hematoxylin-eosin or saffron solid green staining. Three blinded researchers independently assessed the degree of ossification in the epiphyseal cartilage.
Immunohistochemistry
After deparaffination and rehydration of the tissue sections, antigen retrieval and serum blocking were performed. Sections were incubated with rabbit anti-MMP13 polyclonal antibody (bs-10581R; 1:100; Bioss), mouse VEGF-A164 antibody (AF-493-NA; 1:20; R&D Systems, Shanghai, China), and rabbit anti-CD31 antibody (ab28364; 1:50; Abcam, Shanghai, China) followed by incubation with HRP-labeled sheep anti-rabbit secondary antibody (31460; 1:5000, ThermoFisher Scientific) at 37 °C for 20 minutes. The sections were then stained with 3,3′-diaminobenzidine (DAB) using a DAB Staining Kit (C520017; Sangon Biotech, Shanghai, China) and counterstained with hematoxylin.
Terminal Deoxynucleotidyl Transferase dUTP Nick End Labeling (TUNEL) Staining
Paraffin sections (4 μm) were incubated with 20 μg/mL proteinase K at 25 °C for 20 minutes after deparaffinization and washed with PBS. Samples were stained using a TUNEL Apoptosis Detection Kit (40308ES20; Yeasen Biotech Co., Ltd., Shanghai, China) according to the manufacturer’s instructions.
Statistical Analysis
All experiments were repeated at least 3 times, and all quantitative data are presented as mean with 95% confidence interval (95% CI). Student t tests were used to analyze the differences between the 2 groups. A 1-way analysis of variance test was used to compare differences among multiple groups. P value < 0.05 was considered statistically significant. All statistical analyses were performed using the statistical software IBM SPSS Statistics V21.0 (IBM Corp., Armonk, NY, USA), and the graphs were generated by using GraphPad Prism 7.04 software (GraphPad Software Inc., San Diego, CA, USA).
Results
Ossification of Epiphyseal Cartilage Accelerated in Model Mice
Micro-CT images showed that the epiphyseal cartilage (indicated by the white arrow in Fig. 1 ) is lower in density than the bone trabecula. As shown in Fig. 1A-F , there was no ossification in the epiphyseal cartilage at fourth, sixth, and eighth week in the control group. However, in the model group, ossification was observed in the epiphyseal cartilage at 4 weeks posttreatment, and the range of ossification gradually expanded with time (indicated by the red arrows).
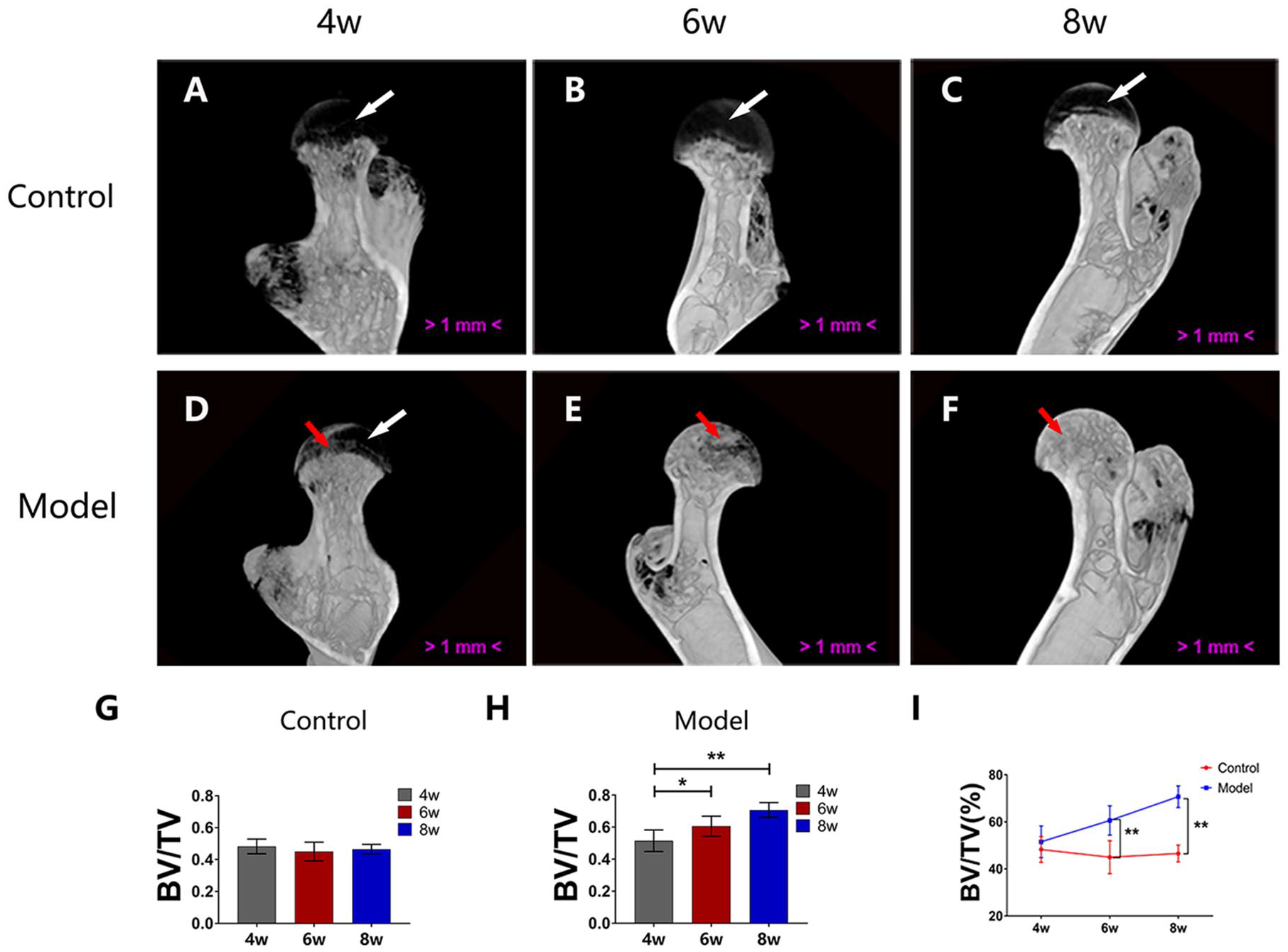
Micro-computed tomography (micro-CT) scanning. (
We found that there was no change in the mean bone volume/tissue volume (BV/TV) of femoral heads at 6 weeks (n = 8; 95% CI, 39.10%-50.88%) or 8 weeks (n = 8; 95% CI, 43.51%-49.53%) compared with that at 4 weeks posttreatment (n = 8; 95% CI, 43.62%-52.85%) in the control group (P = 0.317 and 0.595, respectively; Fig. 1G ). However, in the model group, the mean BV/TV of femoral heads increased at 6 weeks (n = 8; 95% CI, 54.33%-66.89%) and 8 weeks (n = 8; 95% CI, 66.09%-75.35%) compared with that at 4 weeks posttreatment (n = 8; 95% CI, 44.74%-58.27%), P = 0.0068 and P < 0.0001, respectively ( Fig. 1H ). The mean BV/TV in the model group was higher than that in the control group ( Fig. 1I ) at 6 weeks (95% CI, 54.33%-66.89% vs. 95% CI, 39.10%-50.88%; n = 8; P < 0.0001) and 8 weeks posttreatment (95% CI, 66.09%-75.35% vs. 95% CI, 43.51%-49.53%; n = 8; P < 0.0001). In brief, the mean BV/TV of the femoral heads increased in the model mice at 6 and 8 weeks posttreatment, indicating that the ossification of epiphyseal cartilage advanced with time in the model group.
Ossification of Epiphyseal Cartilage in the Model Mice Presented a Sequential Process
The histological results were consistent with the micro-CT results. As shown in Fig. 2A-F, in the control group, epiphyseal cartilage was present under the permanent articular cartilage and was separated from the trabecula by the epiphyseal plate. Ossification in the epiphyseal cartilage was observed in the model group but not in the control group, and the range of ossification gradually expanded with time in the model group.
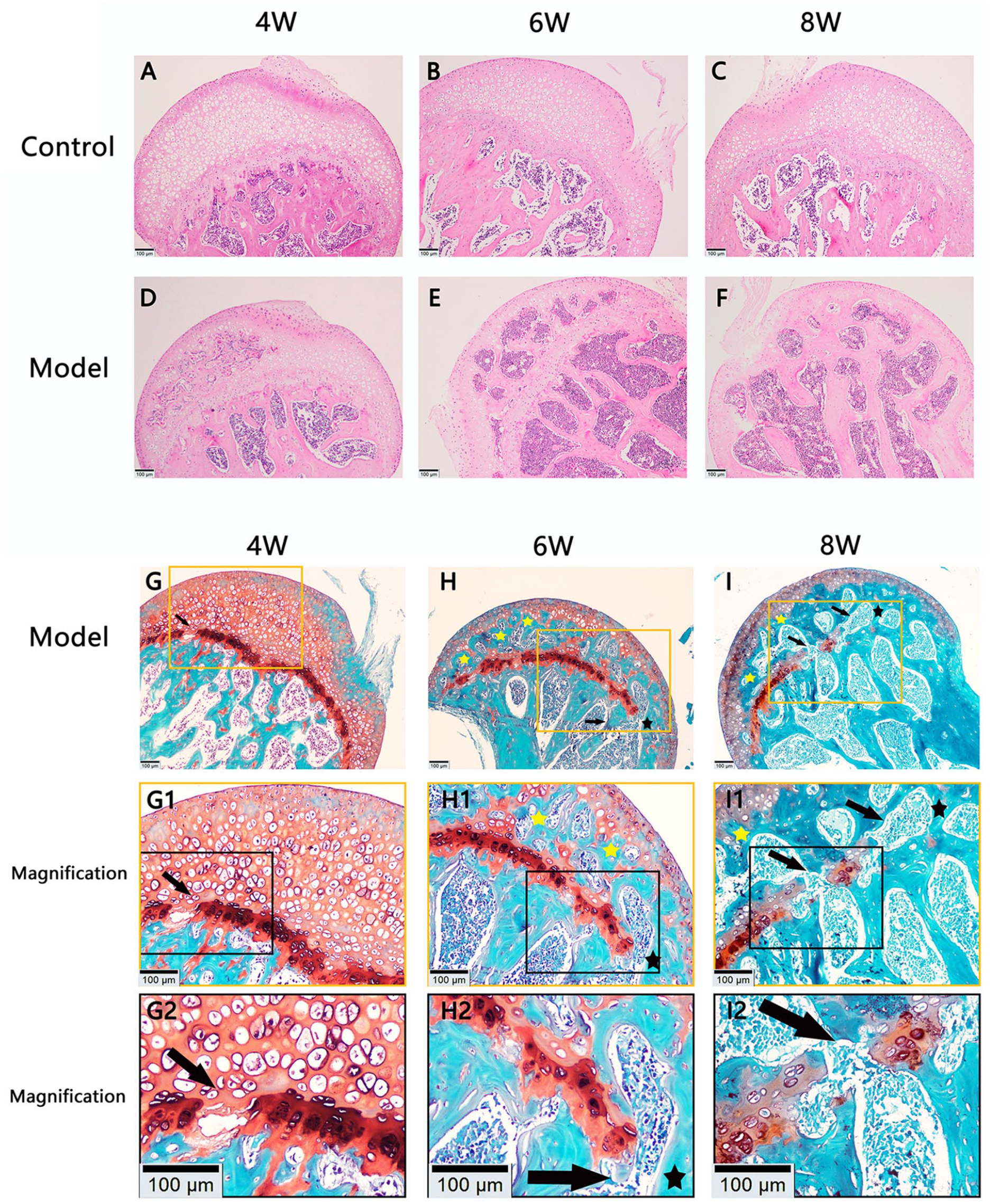
Histological morphology of femoral heads. (
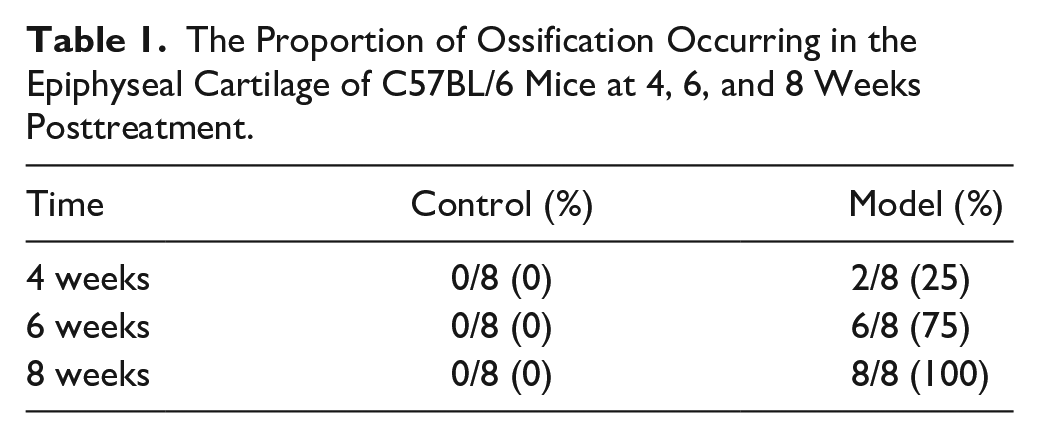
Saffron solid green staining further demonstrated that the epiphyseal cartilage was replaced with trabecular bone in the model group at 6 and 8 weeks posttreatment ( Fig. 2G-I ). The epiphyseal plate was partially absorbed and interrupted at 4 weeks posttreatment, and the absorption became more evident at 6 and 8 weeks, which was accompanied by the appearance of transphyseal osseous bridges and epiphyseal trabeculae. As shown in Table 1 , ossification in the epiphyseal cartilage of the control group did not appear in all the femoral head specimens. However, ossification in the epiphyseal cartilage of the model group was observed in 2 of the 8 mice at 4 weeks posttreatment, and the proportion gradually increased with time. At 8 weeks posttreatment, ossification of epiphyseal cartilage had occurred in all mice of the model group. The above results suggest that the ossification of epiphyseal cartilage accelerated in mice in the model group. The epiphyseal cartilage ossification started at 4 weeks posttreatment in a portion of mice in the model group and presented as a sequential process, which was not observed in the control group.
The Proportion of Ossification Occurring in the Epiphyseal Cartilage of C57BL/6 Mice at 4, 6, and 8 Weeks Posttreatment.
Blood Vessels Increased in the Epiphyseal Cartilage of the Model Group
Vascularization is a crucial step for the ossification of epiphyseal cartilage. As shown in Fig. 3A-C , in the model group, vascular-like structures appeared in the epiphyseal cartilage, with epiphyseal cartilage ossification detected at 4, 6, and 8 weeks posttreatment. Immunohistochemical staining further proved that these structures were blood vessels (CD31 positive, indicated by the red arrows in Fig. 3D1-F1 ). There were no vascular-like structures in the epiphyseal cartilage of the control group ( Fig. 2A-C ).
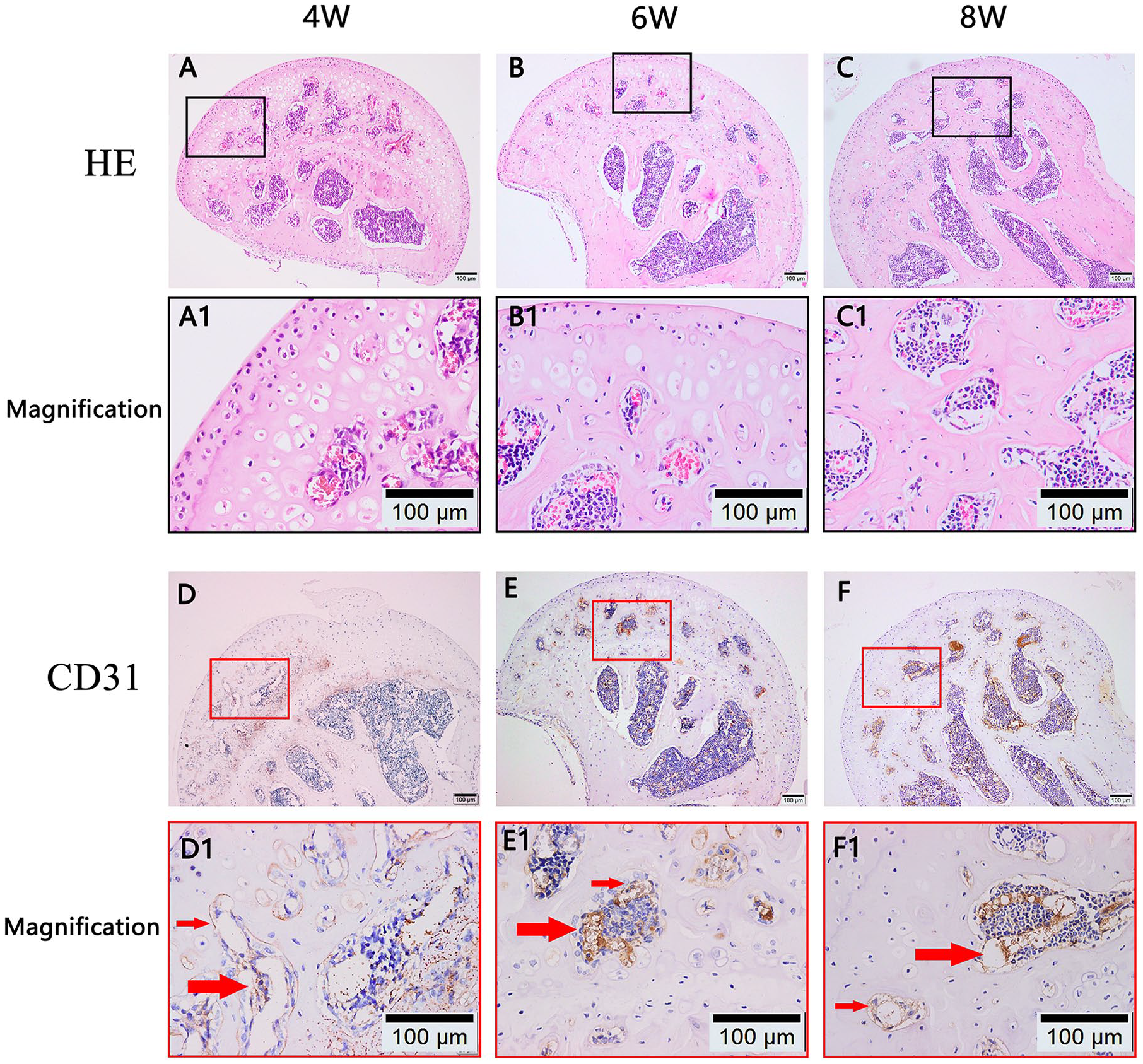
Angiogenesis in the epiphyseal cartilage and bone area ratio of femoral heads. (
Apoptosis of Chondrocytes Increased in the Model Group
At 4 weeks posttreatment, TUNEL staining showed that there were few apoptotic chondrocytes in the epiphyseal cartilage layer in the control group. However, the number of apoptotic chondrocytes increased in the model group ( Fig. 4A ).
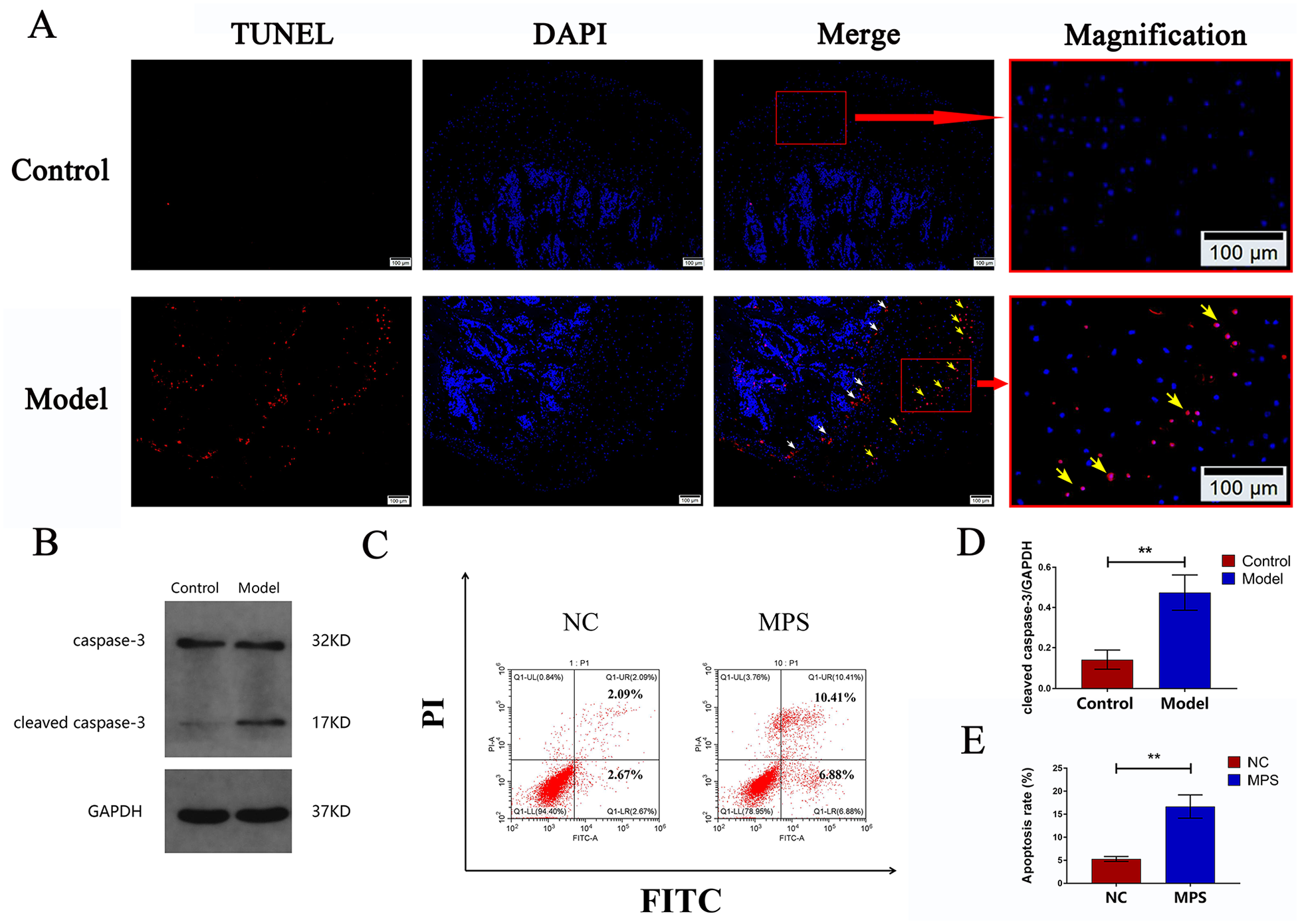
Apoptosis of chondrocytes. (
Caspase-3, a member of the caspase family, plays a central role in cell apoptosis.26,27 Caspase-3 activity was detected in the femoral head by Western blot analysis. As shown in
Fig. 4B
and
Glucocorticoid Caused Changes in Chondrocyte Metabolism
At 4 weeks posttreatment, VEGF-A164 expression decreased in the epiphyseal cartilage, epiphyseal plate, and subchondral bone in the model group compared with that in the control group ( Fig. 5A and B ). In vitro experiments suggested that chondrocytes secreted less VEGF-A164 after GC intervention compared with that in the control group (95% CI, 0.38-0.70 vs. 95% CI, 0.26-0.51; n = 3; P = 0.033) ( Fig. 5D and E ). However, there was local VEGF-A164 aggregation where chondrocyte apoptosis increased in the epiphyseal cartilage ( Fig. 5B2 , B3 , C , C1 , and C2 ).
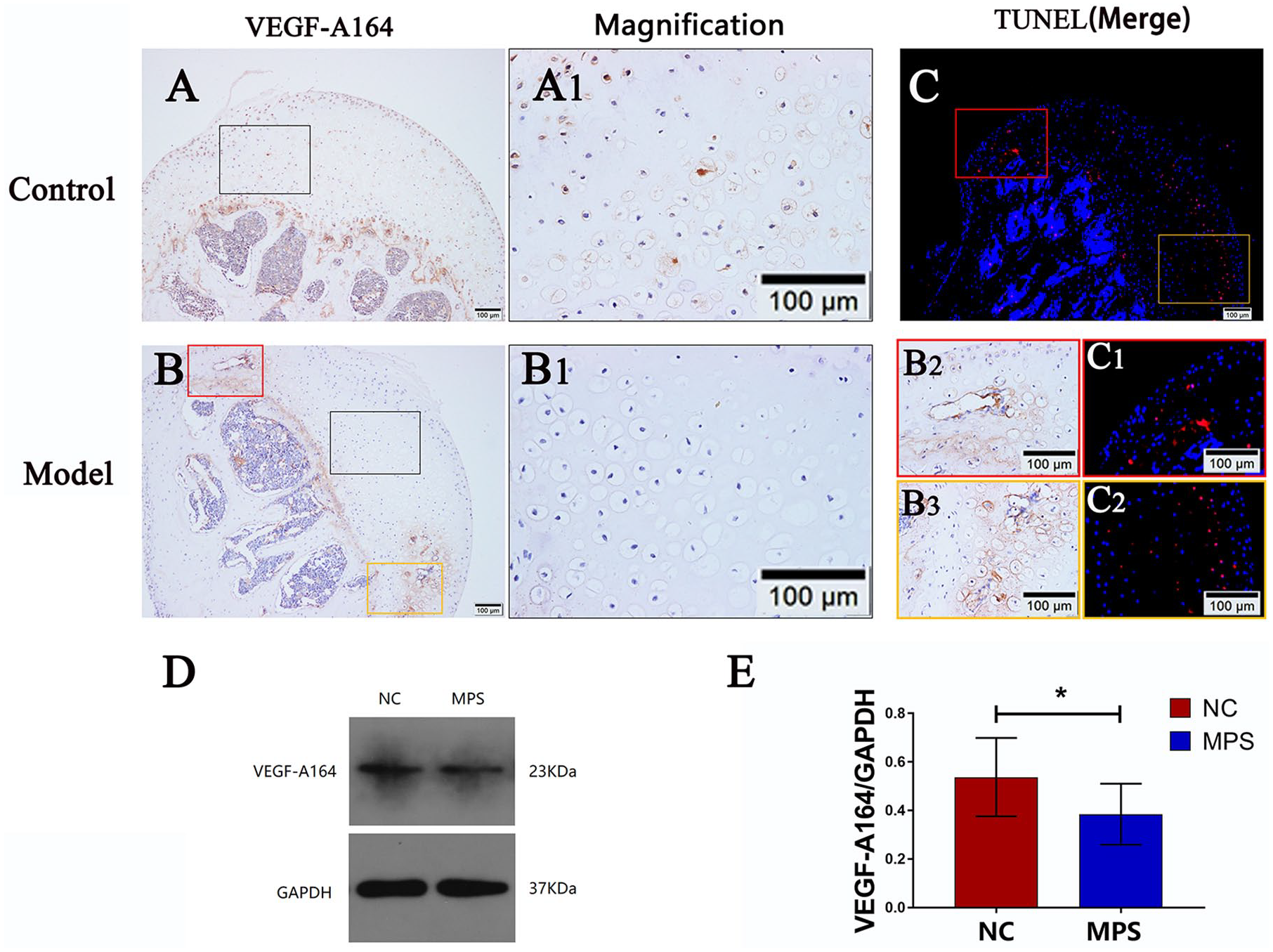
Expression of VEGF-A164 in chondrocytes. (
Matrix metalloproteinase 13 (MMP-13) plays a crucial role in the process by directly or indirectly initiating the degradation of downstream matrix and collagen components.
28
Overexpression of MMP-13 can induce excessive extracellular matrix (ECM) degradation and is crucial for endochondral ossification.
22
As shown in
Fig. 6A
and
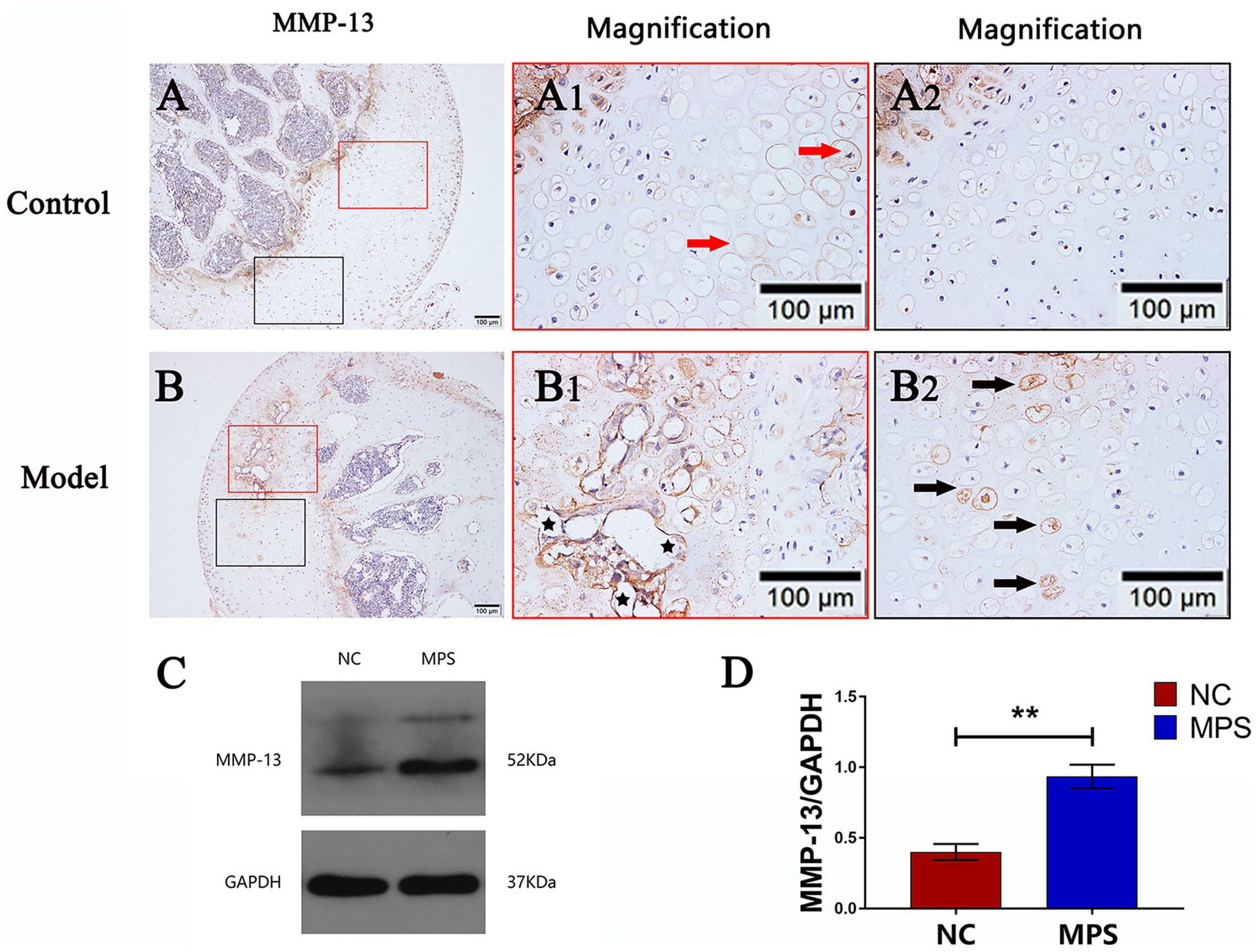
Expression of matrix metalloproteinase 13 (MMP-13) in chondrocytes. (
Discussion
The side effects of GC on the epiphyseal cartilage are different from those on growth plates. High doses or prolonged use of GCs disturb the osteogenic process of growth plates, thus disrupting longitudinal growth and resulting in growth retardation. Here, our study presented a novel observation that excessive GC treatment accelerated epiphyseal cartilage ossification in growing female mice. Our results suggest that the mean BV/TV of the femoral head increased with time in the model mice. This result is not contradictory to previous research where the results indicated that overuse of GC could lead to osteoporosis.1,11,12 As the immature femoral head is partly composed of cartilage, the value of BV/TV is low. As the ossification of the epiphyseal cartilage advanced, the value of BV/TV increased. The function of the BV/TV value here is to indicate an advance in epiphyseal cartilage ossification.
Vascular endothelial growth factor (VEGF) is vital for stimulating angiogenesis.12,29 There are splicing variants of the VEGF protein. VEGF-A is involved in all the essential biology of endothelial cells, from proliferation to vessel function. In humans, 5 isoforms of VEGF-A (VEGF-A121, VEGF-A145, VEGF-A165, VEGF-A189, and VEGF-A206) have been identified with different biological activities. 30 While in mice, VEGF-A is expressed as 3 primary alternative spliced variants (VEGF-A120, VEGF-A164, and VEGF-A188). 31 Among the 3 isoforms in mice, VEGF-A164 is the most abundant isoform, as well as the most potent inducer of endothelial cells sprouting and proliferation. 32 In our study, angiogenesis increased in the epiphyseal cartilage, but the total VEGF-A164 expression decreased in the femoral head of the model group. The phenomena seem contradictory but can be explained. Under normal physiological conditions, a small amount of VEGF-A164 is expressed by chondrocytes in the epiphyseal cartilage; however, epiphyseal cartilage can resist vascular invasion. 33 First, chondrocytes also secrete anti-angiogenic factors such as thrombin sensitive protein-1, chondromodulin-I, and troponin-I. Second, chondrocytes and the ECM around chondrocytes fill the space of epiphyseal cartilage, which has the effect of resisting the invasion of blood vessels. Last, the growth plate isolates the blood circulation of epiphyseal cartilage and metaphysis, making them mutually independent. 33 Thus, there is normally no angiogenesis in the epiphyseal cartilage. However, the break in the balance between angiogenic factors and anti-angiogenic factors can induce vascular invasion. 34 As our results showed that the administration of excessive GC led to chondrocyte apoptosis in the epiphyseal cartilage. Chondrocyte apoptosis could provide the physical basis for the growth of blood vessels into the epiphyseal cartilage.33,34 Furthermore, after the administration of GC, MMP-13 secretion by chondrocytes in the epiphyseal cartilage increased. After apoptosis of chondrocytes in the epiphyseal cartilage, the surrounding ECM may be degraded by MMP-13, providing more room for blood vessels to grow.33,35 Moreover, MMP-13 is also able to release the VEGFA bound to ECM and thereby promote VEGFA-mediated angiogenesis. 36 This process is consistent with our observation that there was a local increase in VEGF-A164 levels where chondrocyte apoptosis increased. Therefore, although VEGF-A164 levels decreased as a whole, the increased chondrocyte apoptosis and secretion of MMP-13 in the epiphyseal cartilage would promote vascular invasion. In addition, we found that the integrity of the epiphyseal plate was gradually destroyed over time, which may further promote the metaphysis vascular invasion.
In the present study, we demonstrated in vitro and in vivo that GC induced apoptosis of chondrocytes, altered chondrocyte metabolism, and induced vascular invasion in the epiphyseal cartilage. However, our explanation of the pathological changes is preliminary. Further research is required to study the exact mechanisms of how chondrocyte apoptosis and metabolic change affect vascular invasion in the epiphyseal cartilage. Besides, if the blood vessels cross the epiphyseal plate from the metaphysis and enter the epiphyseal cartilage prematurely, the entire epiphyseal plate and overall growth of the femoral head will be disrupted, which may cause angular deformities in the developing femoral head. 21 In our study, because the observation time was relatively short, whether the femoral head development of mice in the model group would be malformed must be observed further.
We used female C57BL/6 mice to establish the model. The reason is that proximal femoral physeal senescence in C57BL/6 mice occurs at an average age of 7 and 6 weeks in male and female mice, respectively, while physeal ossification begins around 13 and 25 weeks of age in male and female mice, respectively. 21 This physiological process provided sufficient time to observe epiphyseal cartilage and epiphyseal plate changes. However, our model has some defects. The female C57BL/6 mouse model fails to mimic humans completely. Initially, the proximal femoral epiphysis in both humans and mice consists of avascular unmineralized cartilage. In humans, a secondary ossification center then forms in the femoral head, and periphyseal vessels enter the epiphyseal cartilage at about 6 months of age. In contrast, mice do not develop a secondary ossification center during skeletal development. Once skeletal maturity is reached, humans and mice both develop vessels penetrating the epiphyseal plate associated with epiphyseal cartilage ossification and epiphyseal closure. Therefore, the epiphyseal cartilage of mice undergoes a more prolonged phase of mineralization and ossification through chondrocyte-dependent mechanisms as compared to that of humans. 21 Hence, whether the results of our animal experiments are perfectly applicable to children, remains to be studied.
In summary, our study revealed that epiphyseal cartilage ossification is accelerated in the femoral heads of female C57BL/6 mice after excessive GC treatment. To our knowledge, this is the first study to investigate the sequential changes of epiphyseal cartilage after excessive GC treatment in a mouse model. Increased apoptosis of chondrocytes, changes in chondrocyte metabolism, as well as increased vascular invasion were the potential influencing factors. However, our study was preliminary. The exact connections between chondrocyte apoptosis, metabolic changes, and vascular invasion in epiphyseal cartilage deserve further study. Whether the femoral head development would be malformed after excessive GC treatment and whether the results of our animal study are applicable to children remain to be observed.
Footnotes
Author Contributions
Shengyang Jin and Hong Wang contributed to the idea and design of the study. Yu He was responsible for grouping and treating the mice. Shengyang Jin and Liang Yang performed the experiments and collected the data. Shengyang Jin, Chunqing Meng, and Kaige Ma analyzed the data. Shengyang Jin drafted the paper, Liang Yang and Wei Huang substantially revised the paper. Hong Wang gave the final approval of the version to be submitted. All authors have read and approved the final manuscript. Shengyang Jin takes responsibility for the integrity of the data analysis.
Acknowledgements and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (Grant No. 81672166).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted following protocol approval (No. bfr2018003) by the Animal Experimentation Committee of Hubei Provincial Center for Disease Control and Prevention (Wuhan, China).
Animal Welfare
All animal experiments were conducted following the Guide for the Care and Use of Laboratory Animals (2018).
