Abstract
Objective
Amnion products are used in various musculoskeletal surgeries and as injections for joint pain with conflicting reports of cell viability and protein contents. The objective of this study was to determine the full proteome and examine cell viability in 9 commercial amnion products using an unbiased bottom-up shotgun proteomics approach and confocal microscopy.
Design
Products were subjected to liquid chromatography–tandem mass spectrometry (LC-MS/MS) analysis and searched against a UniProt Homo sapiens database. Relative protein abundance was determined for each sample. Based on proteomics results, lumican was measured by enzyme-linked immunosorbent assay (ELISA) and Western blot analysis was performed for interleukin-1 receptor antagonist (IL-1Ra) and tissue inhibitor of matrix metalloproteinase-2 (TIMP-2). Cell viability was determined by calcein AM (live) and ethidium homodimer (dead) staining and confocal microscopy.
Results
Proteomic analysis revealed 919 proteins in the nine products. Proteins were primarily collagens, keratin, and albumin. Lumican, a small leucine-rich proteoglycan (SLRP) was found in all samples. Western blot analysis for IL-1Ra and TIMP-2 indicated presence of both proteins, with nonspecific antibody binding also present in all samples. No live cells were identified in any product.
Conclusions
Several novel proteins were identified through proteomics that might impart the beneficial effects of amnion products, including SLRPs, collagens, and regulators of fibroblast activity. IL-1Ra and TIMP-2 were identified, but concentrations measured by ELISA may be falsely increased due to nonspecific antibody binding. The concept that the amnion tissues provide live cells to aid in tissue regeneration cannot be supported by the findings of this study.
Introduction
Amniotic membrane has been used since the early 20th century as wound dressings and sources of transplantation tissue.1,2 More recently, biomedical products have been developed from amnion and other birth tissues for orthopedic applications. Various preparations and combinations of amniotic membrane, umbilical cord, umbilical cord blood, and amniotic fluid have been used to treat patients with musculoskeletal pathology with observed outcomes of decreased inflammation3-5 and pain,3,6 reduced incidence of postsurgical fibrous adhesions,7-9 scarring,7,10,11 and reduced wound healing time.11,12 These products are available in liquid or sheet form and can be comprised of single or a combination of tissues. Tissues are further processed to result in a variety of formulations, including cryopreserved or lyophilized liquids for intra-articular injection, and nonviable, dehydrated, or viable cryopreserved sheets of amnion ± chorion or umbilical cord. 13
Both viable and nonviable products have been reported as effective treatments in animal models of wound healing, bone repair, and osteoarthritis (OA).5,7,9,12 In rabbits, acellular human amniotic fluid injected subperiosteally into bone lesions resulted in an increased rate of ossification compared to saline treatment. 14 In another study, acellular amniotic fluid injected adjacent to perichondrial grafts in rabbits led to thicker neochondrogenesis compared with saline-treated controls. 15 The authors of these studies suggested that the benefits of amniotic fluid were due to its inherently high concentration of hyaluronic acid and growth factors, even though neither was measured in the studies. In a rat model of OA, particulate amniotic membrane/umbilical cord product was injected two weeks after induction of OA resulting in thicker cartilage and less synovial inflammation than saline controls. 16 Also in a rat OA model, a micronized, dehydrated amnion/chorion membrane product was injected into knees 24 hours after induction of OA leading to fewer cartilage erosions suggesting that the amnion/chorion product slowed cartilage degeneration. 17 In clinical patients, a multicenter level 1 randomized controlled clinical trial (n=200) compared the efficacy of amniotic suspension allograft (ASA; Organogenesis, Canton, MA), saline, or hyaluronic acid (HA) for the treatment of knee pain. 18 The ASA-treated patients reported significant improvement in overall pain at 3 and 6 months on the visual analogue scale compared with HA or saline patients. Using the Knee Osteoarthritis Outcome Scoring system, ASA-injected patients at 6 months were significantly less painful and symptomatic for knee osteoarthritis than HA patients.
Combined, these studies provide proof-of-concept that birth tissue products are effective in orthopedic applications. Despite these promising preclinical and clinical data, their biological mechanism of action is unknown. Growth factors, tissue inhibitors of metalloproteinases (TIMPs), stem cells, and extracellular matrix proteins acting as a biological scaffold are often mentioned as potential mechanisms of action. Bioactive factors commonly cited as present include transforming growth factor β1 (TGF-β1), platelet-derived growth factor (PDGF), hepatocyte growth factor (HGF), interleukin-1 receptor antagonist (IL-1Ra), vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), TIMPs, hyaluronan, and IL-10.4,19-25
The objective of this study was to determine the full proteome and examine cell viability in nine commercial amnion products using an unbiased bottom up shotgun proteomics approach and confocal microscopy. Our hypothesis was that birth derived tissues such as amnion contain numerous proteins other than growth factors that could be responsible for their mechanism of action and that no live cells are present in any product.
Methods
Product Identification and Sample Preparation
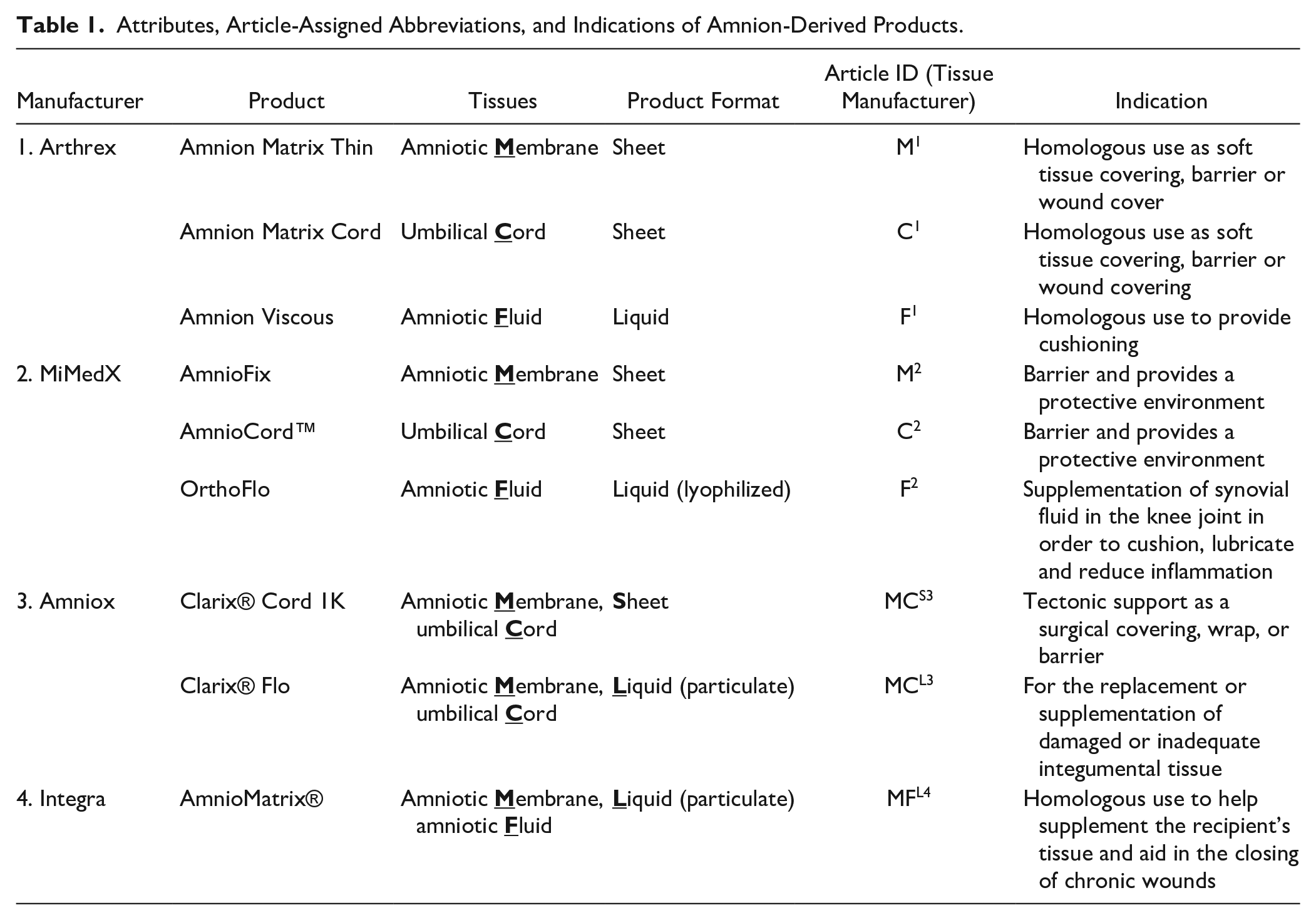
Nine products from 4 manufacturers, including 5 sheet and 4 liquid products, which are intended for injection, were the subject of this study. Products were classified according to their tissue content as reported by the manufacturers ( Table 1 ). Variable claims of cell viability (from no claims to cryopreserved cells) and growth factor content accompanied the products.
Attributes, Article-Assigned Abbreviations, and Indications of Amnion-Derived Products.
Lyophilized fluid samples were reconstituted in lysis buffer (7 M urea, 2% CHAPS, 20 mM dithiothreitol (DTT)) containing protease inhibitors. 26 Sheet samples were snap frozen in liquid nitrogen, pulverized, weighed, and resuspended in lysis buffer. Liquids were rapidly thawed, then centrifuged to pellet cells. Lysis buffer was added to the supernatant, and cell pellets were used for cell culture experiments. Samples were incubated in lysis buffer for three hours at room temperature with agitation then centrifuged at 15,000 × g for 5 minutes at 4 °C to pellet debris. Supernatants were submitted for bottom-up shotgun proteomics to the Cornell University Proteomic and Metabolomics Facility to determine the entire protein make-up of each sample.
Protein Identification by Liquid Chromatography–Tandem Mass Spectrophotometry (LC-MS/MS)
A Bradford assay was used to determine total protein concentration of each sample so that comparisons between samples could be made. Samples were resolved on a sodium dodecyl (lauryl) sulfate–polyacrylamide gel (SDS-PAGE) and stained with Coomassie blue to visualize size and distribution of proteins. A heavily stained protein band with a mass of approximately 66 kDa, suspected to be albumin, was excised from the gel in 3 samples (
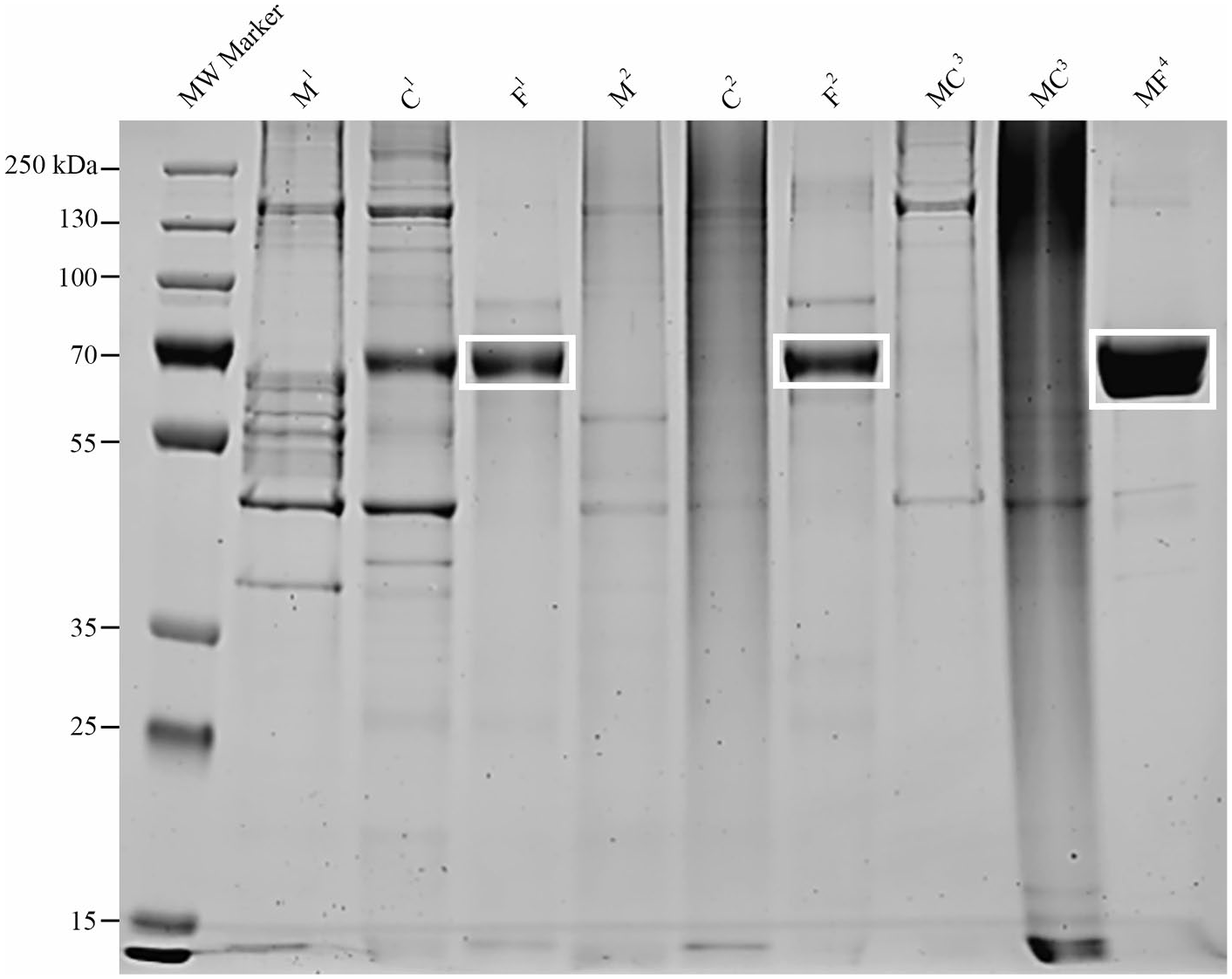
Protein distribution in birth tissue–derived products. Proteins were resolved on a sodium dodecyl sulphate–polyacrylamide gel (SDS-PAGE) and stained with Coomassie to visualize protein distribution prior to proteomics. In amniotic fluid samples, bands at 69 kDa (white boxes) were excised prior to in-gel trypsin digestion in order to improve detection of less abundant proteins by liquid chromatography–tandem mass spectrometry (LC-MS/MS). M = amniotic membrane, C = umbilical cord, F = amniotic fluid, MC = amniotic membrane/umbilical cord, MF = amniotic membrane/amniotic fluid1 = manufacturer 1, 2 = manufacturer 2, S3 = sheet product manufacturer 3, L3 = liquid product manufacturer 3, L4 = liquid product manufacturer 4.
Cell Viability
Cell Culture
Fluid samples (F1 and MF4) were cultured for evidence of adherent cells. Cells were cultured in MSC culture media (1 g/dL glucose Dulbecco’s modified Eagle medium [DMEM], 20% fetal bovine serum [FBS], 2 mM
Confocal Microscopy
Only cryopreserved products (MC3, F1, and MF4) with the potential to contain viable cells were imaged by confocal microscopy. Fresh bovine amnion and passage 1 equine bone marrow-derived mesenchymal stromal cells served as live controls. A devitalized, dehydrated product (M1) served as a negative control. Samples were stained with calcein AM (2 μM; 650853-81, Invitrogen, Carlsbad, CA) and ethidium homodimer (1 μM; L3224, Thermo-Fisher Scientific) for live/dead visualization, respectively, and imaged on a Leica SP5 confocal microscope (Leica Microsystems Inc., Buffalo Grove, IL) as previously described. 28 Additional images for MC3 and MF4 were acquired to visualize collagen in the extracellular matrix.
Lumican ELISA
Proteomics revealed that lumican was in the top 10 most abundant proteins of 7 products. To determine if lumican could elute from products, sheet samples (M1, C1, MC3) were snap frozen in liquid nitrogen and pulverized. Fluid samples were not further processed. Products with remaining sample (M1, C1, F1, F2, MC3, MCL3) were incubated in phosphate-buffered saline (PBS) at 37 °C for 48 hours and analyzed by enzyme-linked immunosorbent assays (ELISAs; DuoSet Lumican ELISA; R&D Systems, Minneapolis, MN).
Western Blot Analysis of Interleukin-1 Receptor Antagonist Protein (IL-1Ra) and Tissue Inhibitor of Matrix Metalloproteinase-2 (TIMP-2)
Proteomic results did not indicate the presence of IL-1Ra or TIMP-2 in any sample. This conflicts with published ELISA and immunohistochemical data on similar products.29-31 In an attempt to reconcile these differences, western blot analysis was performed to determine if IL-1Ra or TIMP-2 was detectable in lysates or eluates from the products. These assays could only be performed on products with sufficient sample remaining after proteomics and lumican ELISA (M1, C1, F1, MC3). Samples were resolved on an SDS-PAGE gel and transferred to polyvinylidene difluoride (PVDF) membranes for Western blot analysis for IL-1Ra (AF-280-SP; R&D Systems) and TIMP-2 (MAB 971; R&D Systems).
Results
Composition of Amnion-Derived Products
Obvious particulate matter was present in 2 of the 4 samples intended for use as liquid injections (

Gross appearance of liquid injection amniotic products. Samples were prepared for proteomic analysis. After centrifugation, abundant particulate matter (black boxes) was observed in 2 of the 4 products. F = amniotic fluid, MC = amniotic membrane/umbilical cord, MF = amniotic membrane/amniotic fluid1 = manufacturer 1, 2 = manufacturer 2, L3 = liquid product manufacturer 3, L4 = liquid product manufacturer.
Number of Proteins in each Amnion-Derived Product.
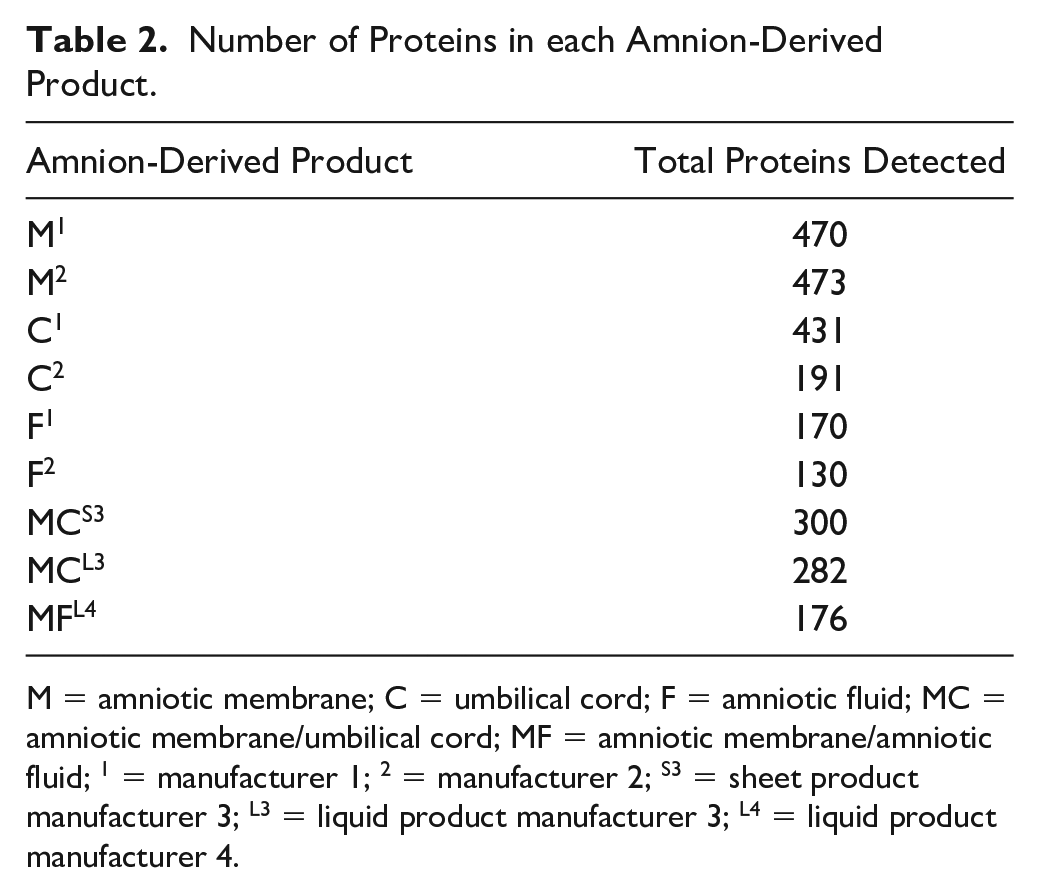
M = amniotic membrane; C = umbilical cord; F = amniotic fluid; MC = amniotic membrane/umbilical cord; MF = amniotic membrane/amniotic fluid; 1 = manufacturer 1; 2 = manufacturer 2; S3 = sheet product manufacturer 3; L3 = liquid product manufacturer 3; L4 = liquid product manufacturer 4.
Proteins detected in the products were not what are typically considered as anabolic or anti-catabolic in joint homeostasis, but neither were there pro-inflammatory proteins detected. The 10 most abundant proteins (excluding albumin) in all products were primarily extracellular matrix proteins such as collagen VI and XII and keratins 6A, 8, 14, and 19 ( Table 3 ). No growth factors were identified in any of the samples. In the anti-catabolic category, TIMP-1 was identified in one sample at low abundance (F2, 0.05%), as was TIMP-3 (M2, 0.03%). One unique peptide of TIMP-2 was detected in F2 and 1 unique peptide of IL-1Ra was detected in F1, but the threshold for protein detection used in this study was 2 unique peptides, and therefore this was not considered valid evidence of TIMP-2 or IL-1Ra.
Top 10 Most Abundant Proteins in Amnion-Derived Samples (Albumin Excluded). a
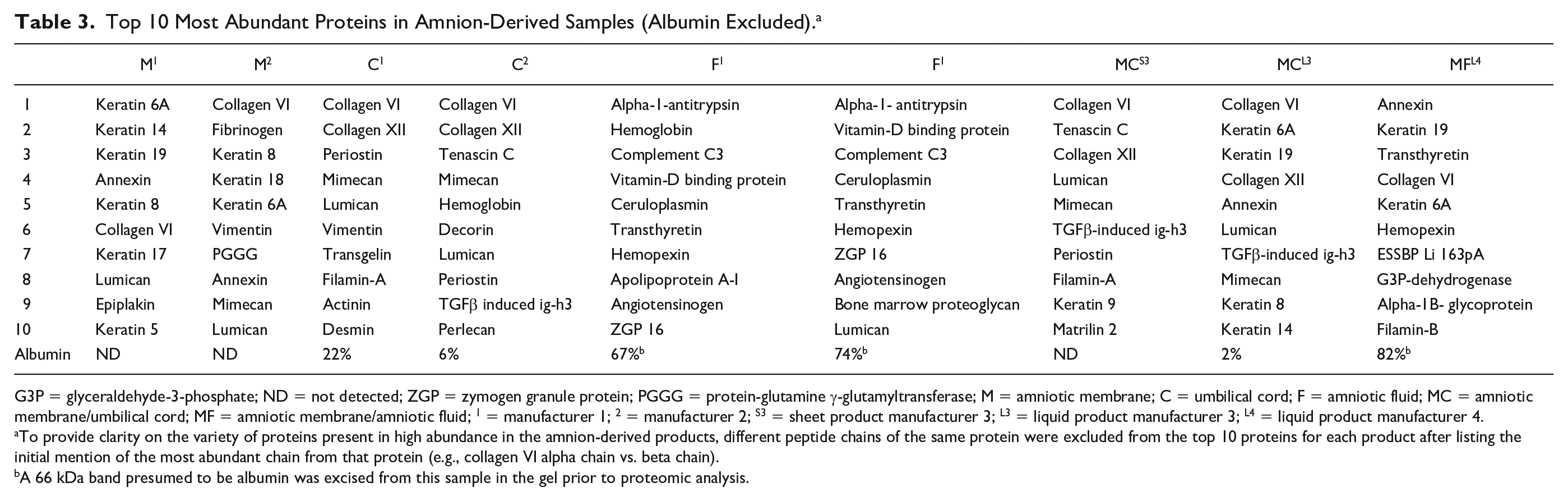
G3P = glyceraldehyde-3-phosphate; ND = not detected; ZGP = zymogen granule protein; PGGG = protein-glutamine γ-glutamyltransferase; M = amniotic membrane; C = umbilical cord; F = amniotic fluid; MC = amniotic membrane/umbilical cord; MF = amniotic membrane/amniotic fluid; 1 = manufacturer 1; 2 = manufacturer 2; S3 = sheet product manufacturer 3; L3 = liquid product manufacturer 3; L4 = liquid product manufacturer 4.
To provide clarity on the variety of proteins present in high abundance in the amnion-derived products, different peptide chains of the same protein were excluded from the top 10 proteins for each product after listing the initial mention of the most abundant chain from that protein (e.g., collagen VI alpha chain vs. beta chain).
A 66 kDa band presumed to be albumin was excised from this sample in the gel prior to proteomic analysis.
The top 10 proteins in all products were reviewed for relevancy to orthopedic tissue repair as might be indicated by their ability to enhance wound repair by inducing cell migration, epithelialization, or angiogenesis, or decreasing inflammation. Several proteins were identified including the protease inhibitor α-1-antitrypsin, which was the most abundant (nonalbumin) protein in both amniotic fluid samples. Periostin was identified in 3 of 4 samples that contained umbilical cord. Additional proteins of greatest interest due to their known importance in cartilage homeostasis and relative abundance in all samples included small leucine-rich proteoglycans (SLRPs) such as lumican and mimecan. These SLRP were found to be in the top 10 components in 7 (lumican) and 5 (mimecan) of the nine products tested ( Table 3 ). Despite removal of a presumptive albumin (69 kDa band) prior to proteomics, all amniotic fluid samples were primarily composed of albumin ( Table 3 ).
Cell Culture and Confocal Microscopy
Light microscopy performed up to day 8 of cell culture showed no evidence of adherent cells. Similarly, on confocal microscopy, no live cells were found in F1, MC3, or MF4 (
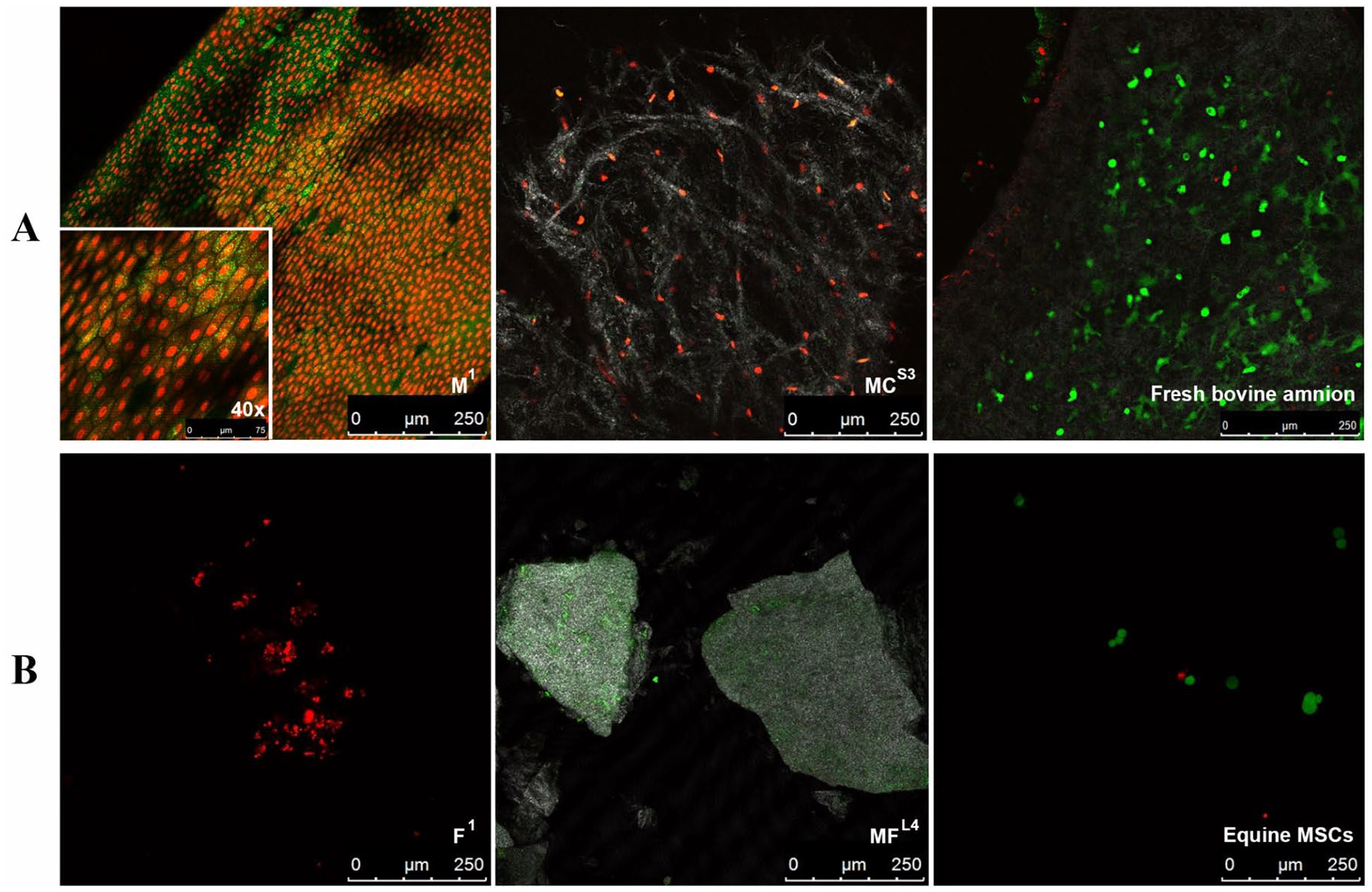
Live/dead cell imaging of (
Lumican Concentration
Approximately 10 times more lumican eluted from dehydrated sheet samples (M1, C1) compared with the cryopreserved sheet product (MC3;
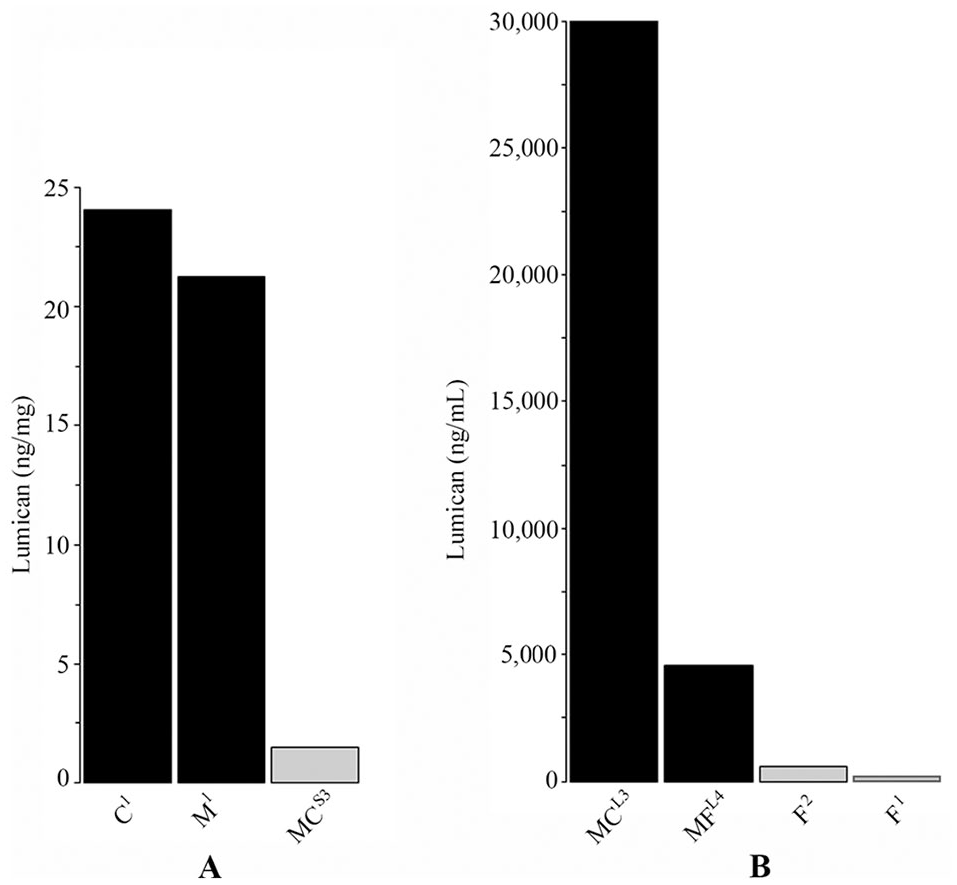
Lumican elution from amnion products. (
Western Blot Analysis of IL-1Ra and TIMP-2
IL-1Ra protein was detected in eluates from M1, C1, and F1 (
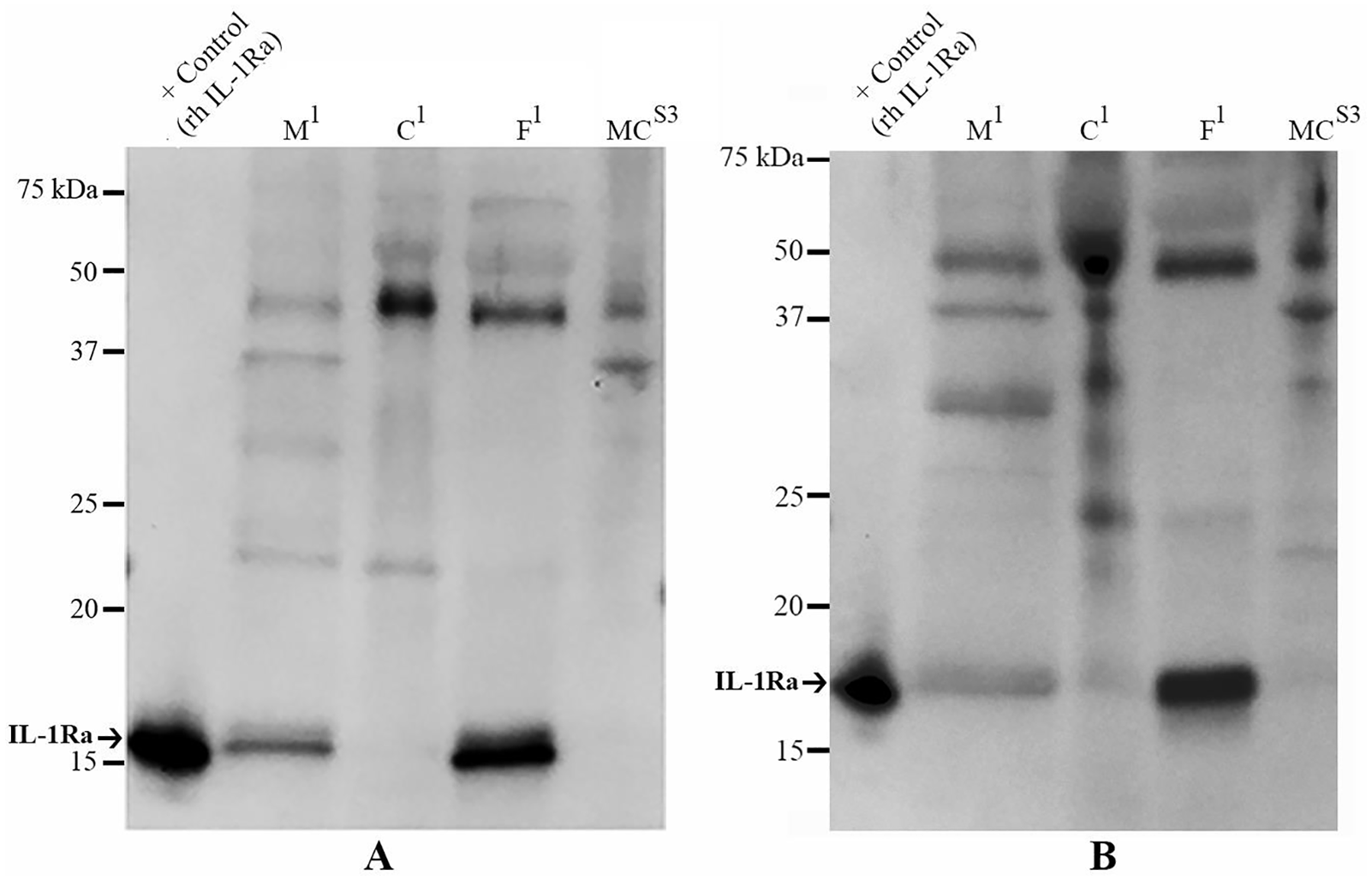
Western blot analyses of IL-1Ra proteins in 24-hour eluates and tissue lysates from amnion-derived samples. Arrows on each scale bar denote the protein of interest in each blot. IL-1Ra was detected in
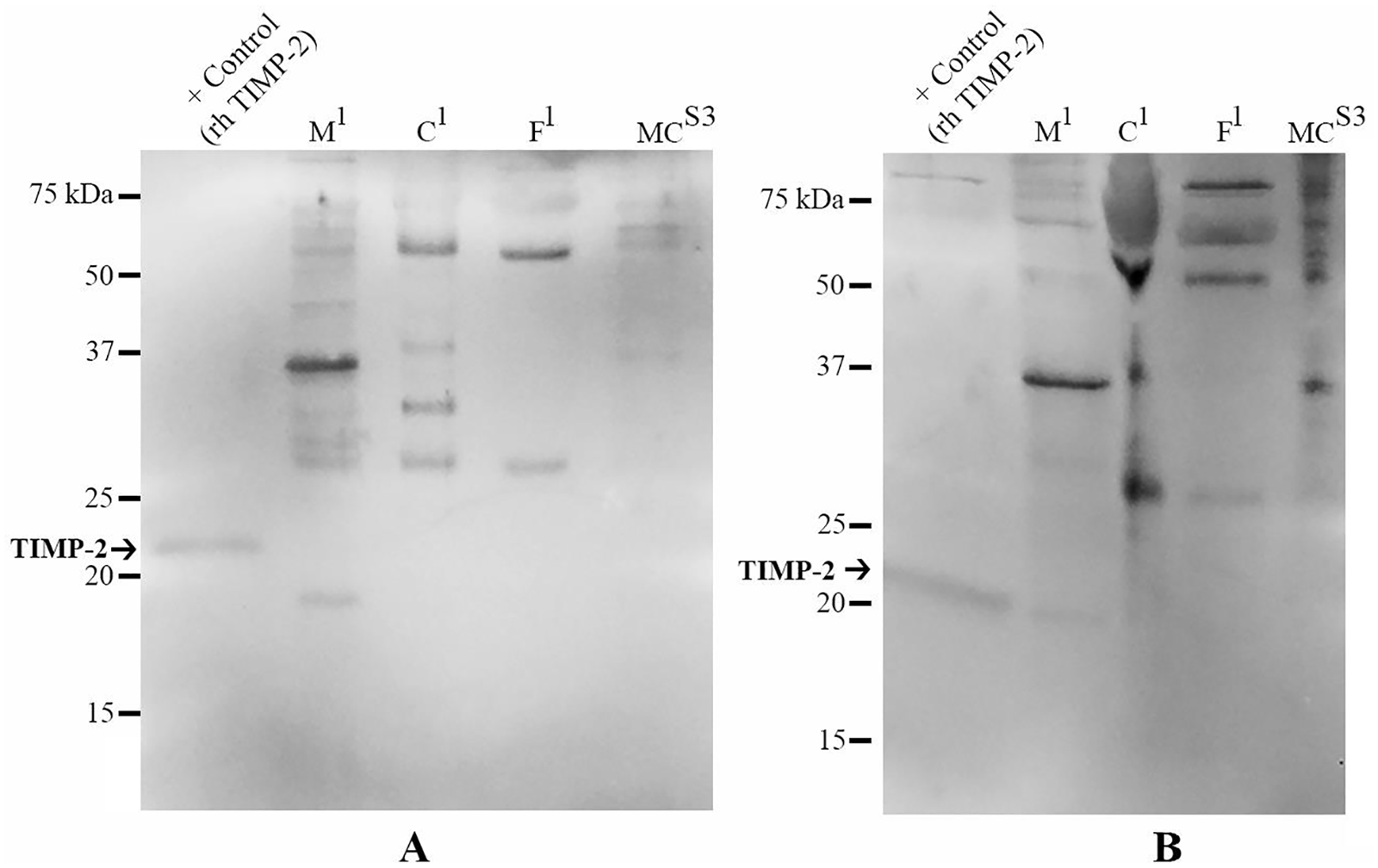
Western blot analyses for TIMP-2 proteins in
Discussion
This study identified 919 proteins among 9 commercial amnion products using bottom-up shotgun proteomics. No growth factors or live cells were identified. It is unlikely that the proteomic technique used missed low abundance or small growth factors or TIMPs. The technique used in this study detects peptides down to 1 femtomole (10−15 moles) in abundance, so even very low concentration proteins should have been detected. 32 The smallest protein detected in this study was 10.17 kDa (fatty acid binding protein; UniProt S4R3A2). IL-1Ra (17 kDa), TIMP-2 (24 kDa), and TIMP-4 (23 kDa), are proteins commonly cited by manufacturers in amnion-derived products and would have fallen into the detectable size range in this study. However, identification of low abundance proteins is a recognized limitation of proteomics, and enrichment methods such as excision of abundant protein bands as performed in the present study to remove albumin, are accepted methods to increase detection of low-abundance proteins.33,34 These methods can also lead to loss of low abundance proteins, especially in the case of albumin, which acts as a sponge, binding small proteins and peptides. 35 Furthermore, excising the albumin band in this study did not fully remove albumin and it was still detected in 6 of 9 samples with ranging from 2% to 82% of total proteins ( Table 3 ). In support of the findings in this study, proteomic studies of fresh human 26 and equine31,36 amniotic membrane where no enrichment techniques were used similarly found no evidence of growth factors or TIMPs, and these studies detected a similar magnitude of proteins as in this study (between 100 and 500 proteins per sample). In contrast, after removal of 14 high-abundance proteins using immunoaffinity depletion, analysis of fresh human amniotic fluid was found to contain 2,881 proteins, including hepatocyte growth factor (0.03% abundance), connective tissue growth factor (0.01% abundance), and TIMP-1 (0.006% abundance). 37 Combined, these studies suggest that growth factors are likely present in amniotic fluid, and possibly in other amnion-derived tissues, but in very low abundance.
Growth factors, TIMPS 1-4, and IL-1Ra are commonly identified in amniotic membrane, umbilical cord, and amniotic fluid products by ELISA.4,19-21,30 The discrepancy between reported ELISA results and the proteomic results of this and other proteomic studies prompted the use of western blot analysis to visualize detection of IL-1Ra and TIMP-2. Interestingly, IL-1Ra and TIMP-2 were identified in some products despite not being identified by proteomic analysis. Cytokines can be labile in samples of fresh and preserved amniotic membrane as confirmed by ELISA with decreasing concentrations detected over time. 38 It is possible that processing and storage, in addition to experimental manipulation of products in this study, diminished growth factor and TIMP concentrations beyond detectable limits of proteomics. Furthermore, only one lot of each product was tested and typically, the lot of a product corresponds to a single donor, and donor variability is known to exist. For example, the protein content of amniotic fluid has been shown to change with maternal hydration status and uterine perfusion, 39 as well as maternal prolactin levels. 17 Growth factor content in multiple amniotic membrane preparations (unprocessed, lyophilized, cryopreserved, or nitrogen-frozen) is also influenced by donor and gestational age. 21 Inter- and intradonor levels of EGF can also vary in amniotic membrane based on gestation age and maternal attributes.22,38 Regardless, the Western blot results indicate nonspecific binding of primary antibodies to TIMP-2 and IL-1Ra, which may result in falsely increased concentrations measured by ELISA depending on the antibodies used for ELISA.
Albumin was the most abundant protein identified in four of the nine products tested in this study (C1, F1, F2, MF4). Even when depleted using a filtration column or by gel excision as in the present study, albumin has repeatedly been identified as the most abundant protein in amniotic fluid.37,40,41 A 5% low-molecular-weight albumin solution has been investigated as a treatment for osteoarthritic knee pain in a phase II study. 42 Patients receiving albumin injections experienced significantly less joint pain at 20 weeks compared to saline controls suggesting that albumin might be therapeutic for knee pain. The mechanism for this putative therapeutic effect of albumin is unknown, but would make an interesting future direction for research—particularly given the high proportion of albumin in amnion-derived products.
Collagens such as collagen VI (ColVI) and keratins were the next most abundant proteins in all samples and have been previously identified in human and equine amniotic membrane by proteomics.26,30,31,43 ColVI connects cells to surrounding tissues and binds with other collagens and proteoglycans to link cells and ECM components in a variety of tissues. 44 It plays an important role in chondrocyte maintenance and proliferation, as well as providing scaffolding for osteoblasts, preosteoblasts, and chondrocytes during bone development. 44 Among proteins known to interact with ColVI resulting in downregulation of inflammation and upregulation of tissue regeneration are collagen XII, matrilin 2, lumican, and decorin, all of which were present in the top 10 detected proteins in the proteomic analysis of this study. Keratins could also explain the positive effects of amnion products. Biological scaffolds made from keratin proteins have been observed to increase cell-adhesion 45 and induce polarization of inflammatory M1 macrophages to their anti-inflammatory M2 phenotype. 46 α-1-Antitrypsin was the most abundant protein after albumin in both amniotic fluid products. However, concentrations of α-1-antitrypsin are unchanged from normal to OA samples, making it less interesting for further study. 47 Periostin was present in 3 of 4 samples that contained umbilical cord, but in no other samples. It is interesting from a mechanism of action perspective because it is a potent proangiogenic factors and plays a critical role in organization of the extracellular matrix particularly in collagen cross-linking. 48
One or more small SLRPs, including lumican, mimecan, and decorin were identified in the top 10 proteins of 7 of 9 samples, and in the top 16 proteins of all samples. SLRPs are subdivided into multiple families that either bind directly to the extracellular matrix or remain as soluble factors that interact with growth factors and cytokines directly. 49 For example, decorin and lumican have been observed to promote wound healing by binding TGF-β, upregulating collagen deposition, and stimulating production and bioactivity of growth factors.50-52 Decorin also binds to and inhibits PDGF released by injured cells, preventing collagen deposition and fibrosis. 53 In a sheep model of osteoarthritis, significant fragmentation and protein malformation of the SLRPs lumican, decorin, aggrecan, and biglycan were observed in joints with the most damaged cartilage. 54 Additionally, lumican is involved in promoting wound healing by regulation of collagen fibril formation and deposition, as has been observed in experiments with lumican knockout mice.55-57
No viable cells were identified through tissue culture or confocal microscopy. The concept that live amniotic epithelial cells (AECs) cells might be present in cryopreserved amniotic membrane samples is predicated on studies showing that in fresh, unprocessed amniotic membrane, AECs that express several cell surface markers of embryonic stem cells, exhibit tri-lineage differentiation, and secrete anti-inflammatory cytokines such as interleukin 10 (IL-10), IL-11, and IL-6 can be isolated.10,58-60 Clinical benefits of AECs might be inferred from studies where culture expanded AECs promoted angiogenesis in a neonatal mouse model of hyperoxia-induced lung injury, and in an adult mouse lung fibrosis model. 61 In a large animal ovine model, AECs significantly upregulated collagen I deposition, which is important for formation of organized extracellular matrix. 62 However, in amniotic fluid, most cells are nonviable squamous epithelial cells. 25 Panero et al. 63 reported cell viability findings of 3 commercial amniotic fluid allograft products as well as unprocessed amniotic fluid; none of which were investigated in the present study. In their study, viable nucleated cells were detected in two commercial amniotic fluid products, but none of the cells displayed characteristics of MSCs such as plastic adherence or colony unit formation. Additionally, any viable cells in commercial products were present in too few numbers to analyze with flow cytometry, and the unprocessed amniotic fluid in their study was found to be devoid of viable cells. The importance of viable cells in amnion products to effectuate a clinical response is unknown because products containing viable cells appear to have some of the same biological effects as those without viable cells. In an in vitro study, both viable and devitalized amniotic membrane inhibited production of pro-inflammatory cytokines IL-1α and tumor necrosis factor alpha from lipopolysaccharide-activated peripheral blood mononuclear cells. 23 In another study, amniotic fluid that had been centrifuged and filtered to remove cells was still capable of inducing angiogenesis, suggesting that a component in the liquid fraction as opposed to the cells in amniotic fluid was responsible for the mechanism of action. 64
The results of this study were unable to refine indications for use as stated by each manufacturer for 9 products. The proteome of the samples is just too complex with a range of 130 to 470 proteins detected per sample and 65% of identified proteins overlapping between samples, and the indications for use are largely nondescript. Further studies, possibly using fractionated amnion samples to identify the specific proteins that are imparting the positive effects of amnion, would refine indications of a more defined formulation, possibly improving patient outcomes.
Limitations
A limitation of this study is its inclusion of only one sample (lot) per commercial product. This was due to logistical difficulties in obtaining tissues. Future directions should take into account the high degree of interdonor variability, which would contribute to lot and sample variability.
Conclusions
Several novel proteins were identified through proteomics that might impart the beneficial effects of amnion products, including SLRPs, collagens, and regulators of fibroblast activity. However, the concept that live cells aid in tissue regeneration cannot be supported by the findings of this study.
Supplemental Material
sj-pdf-1-car-10.1177_1947603520976767 – Supplemental material for Proteomic Analysis and Cell Viability of Nine Amnion, Chorion, Umbilical Cord, and Amniotic Fluid–Derived Products
Supplemental material, sj-pdf-1-car-10.1177_1947603520976767 for Proteomic Analysis and Cell Viability of Nine Amnion, Chorion, Umbilical Cord, and Amniotic Fluid–Derived Products by Liliya Becktell, Andrea M. Matuska, Stephanie Hon, Michelle L. Delco, Brian J. Cole, Laila Begum, Sheng Zhang and Lisa A. Fortier in CARTILAGE
Footnotes
Author Contributions
LBecktell—experiments, data analysis and interpretation, generation and review of manuscript. AMM—study concept, planning of experiments, and manuscript editing. SH—experiments, data analysis, and manuscript editing. MLD—experiments, data analysis, manuscript editing. BJC—study concept, sample procurement, manuscript editing. LBegum—experiments, review of manuscript. SZ—planning, performing, and interpretation of proteomics, review of manuscript. LAF—study concept and oversight, interpretation of data, generation and editing of manuscript. All authors have read and approved the final submitted manuscript.
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Arthrex, Inc.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Fortier and Dr. Cole have received research funding from and are paid consultants to Arthrex, Inc., manufacturer of 3 products used in this study. Dr. Matuska is an employee of Arthrex, Inc. Ms. Becktell, Drs. Hon, Delco, Begum, and Zhang declare that they have no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
