Abstract
Injuries to articular cartilage of the knee are increasingly common. The operative management of these focal chondral lesions continues to be problematic for the treating orthopedic surgeon secondary to the limited regenerative capacity of articular cartilage. The pericellular matrix (PCM) is a specialized, thin layer of the extracellular matrix that immediately surrounds chondrocytes forming a unit together called the chondron. The advancements in our knowledge base with regard to the PCM/chondrons as well as interterritorial matrix has permeated and led to advancements in product development in conjunction with minced cartilage, marrow stimulation, osteochondral allograft, and autologous chondrocyte implantation (ACI). This review intends to summarize recent progress in chondrocytes with matrix research, with an emphasis on the role the PCM/extracellular matrix (ECM) plays for favorable chondrogenic gene expression, as a barrier/filtration unit, and in osteoarthritis. The bulk of the review describes cutting-edge and evolving clinical developments and discuss these developments in light of underlying basic science applications. Clinical applications of chondrocytes with matrix science include Reveille Cartilage Processor, Cartiform, and ACI with Spherox (which was recently recommended for the treatment of grade III or IV articular cartilage defects over 2 cm2 by the National Institute of Health and Care Excellence [NICE] in the United Kingdom). The current article presents a comprehensive overview of both the basic science and clinical results of these next-generation cartilage repair techniques by focusing specifically on the scientific evolution in each category as it pertains with underlying chondrocytes with matrix theory.
Basic Science of Chondrons, Pericellular Matrix, and Interterritorial Matrix
In normal articular cartilage, chondrocytes are embedded within an abundant extracellular matrix (ECM). The solid matrix of the ECM is composed of a crosslinked network of type II collagen, proteoglycans, and several important other collagens (e.g., VI, IX, X, XI) and noncollagenous proteins.1,2 The ECM consists of discrete regions based on proximity to the chondrocytes, composition, and collagen fibril diameter and organization. The ECM can be divided into pericellular matrix (PCM), territorial, and interterritorial regions.
3
The PCM is a specialized, thin layer of the ECM that immediately surrounds chondrocytes
4
forming a unit together called the chondron.
5
This PCM (mainly type VI collagen) has an important role in the metabolic activity of the chondrocyte and the mechanical signaling from and to the ECM.6-8 The PCM has a patent structure, defined molecular composition, and unique physical properties that support the chondrocyte (
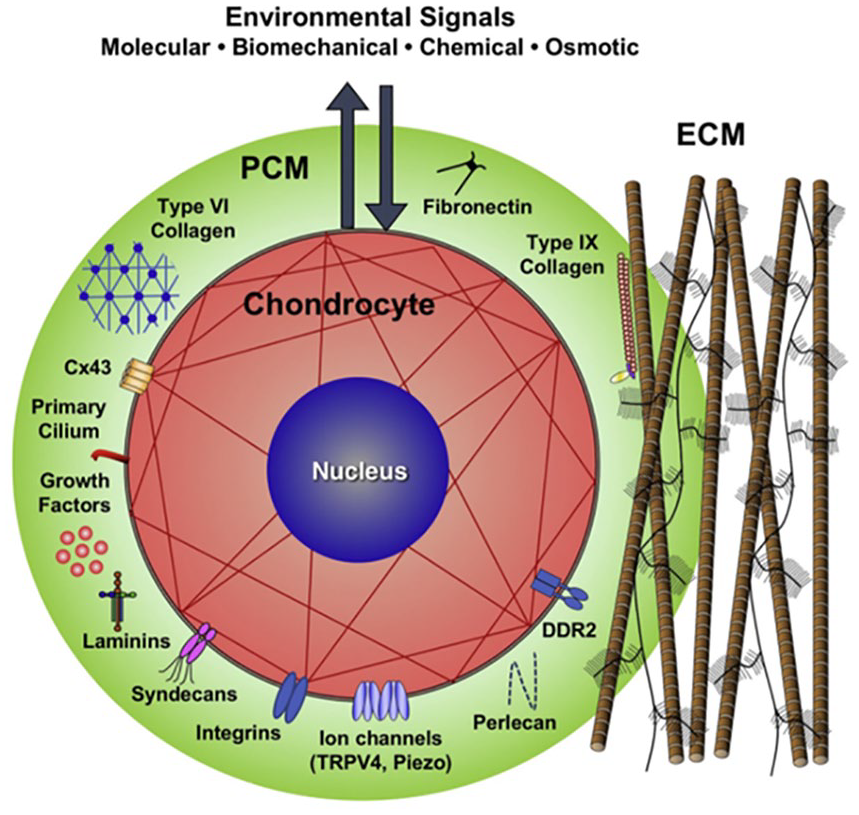
Schematic of a chondron (formed by the chondrocyte together with its surrounding pericellular matrix (PCM). The PCM is a specialized, thin layer of the extracellular matrix. The PCM has a patent structure, defined molecular composition, and unique physical properties that support the chondrocyte. Image used with permission from Guilak
Interterritorial ECM
The PCM integrates with the surrounding tissue via the territorial matrix connecting the PCM to the interterritorial matrix. 9 The interterritorial region of the ECM is the largest matrix region, and its hallmark is randomly oriented bundles of large collagen fibrils. These fibers are arranged based on location in articular cartilage: parallel to the surface of the superficial zone, obliquely in the middle zone, and perpendicular to the joint surface in the deep zone. 3
The interterritorial matrix contains abundant proteoglycans and contributes to the biomechanical properties of articular cartilage. It has elongated parallel ridges with significantly greater surface roughness versus than the PCM. The average Young’s modulus of the interterritorial matrix is significantly greater than the PCM. 10 Thus, the interterritorial matrix appears to possess not only distinct microtopographic contours in comparison with the PCM, but also significantly greater mechanical stiffness. “These distinctive nanostructural and nanomechanical properties may have implications in nutrient diffusion and fluid dynamics, both of which are of vital importance for cartilage health and function.” 10
Biomechanical Importance of the PCM
The PCM has been found to have biomechanical importance to support the chondrocyte. First, within the chondron, the PCM allows the construct to be stiffer versus chondrocytes alone. One study found the PCM allowed for retaining the width and volume of the enclosed chondrocytes via collagen fibrils during compression. 11 Second, through the molecular complex, the PCM has a role in transduction of a mechano-biological signal. This molecular complex, consisting of type VI collagen, biglycan, decorin, and matrilins, interact with other components of the PCM. Specifically, matrilin-1 and -3 attach to the cell membrane, via integrin, and other ECM molecules in order to receive extracellular signals. 12 Another example localized in the chondrocyte PCM, is fibroblast growth factor 2, which is a growth factor that participates in mechanical signal transduction. 13 Finally, the role of transduction translates to gene expression. Mechanical forces regulate chondrocyte differentiation and proliferation via gene expression, that is, Indian hedgehog gene. The upregulation for the expression of Indian hedgehog gene (a vital signal in chondrocyte fate determination and proliferation) 14 was found to be dependent on domain A of matrilin-3. 15 These studies help illustrate a key role of PCM to translate mechanical signals to biological events.
Importance of the PCM as Barrier/Filtration Unit
For the molecules that enter or exit the chondrocytes, the PCM acts as a barrier. An intact PCM helps balance the anabolic and catabolic processes of chondrocytes. Studies have shown cultured Chondrons expressed less matrix metalloproteinase proteins (MMP) in comparison with isolated chondrocytes.16,17 When chondrons were challenged with varied osmolarities, the chondrocyte volumes in the chondrons varied less compared with that of the isolated chondrocytes displaying the most efficient volume regulation.
18
Additionally, acting as a barrier to harmful pathways, chondrons have shown resilience to apoptosis induction versus isolated chondrocytes. Peters
Favorable Chondrogenic Gene Expression and Growth Factors within the PCM
The PCM has a meaningful impact on the functionality of chondrocytes.
22
Zhang
The Role of the PCM in Osteoarthritis
Growing evidence suggests that many of the features of osteoarthritis (OA) are possibly initiated in the PCM. 9 In healthy articular cartilage, the ECM is maintained in homeostasis (a balance of matrix synthesis and degradation). The PCM acts as a transitional zone between the interterritorial matrix and chondrocytes to modulate environmental signals before they reach the chondrocytes. Changes to PCM properties, such as in a pathologic state like OA, may not only represent the disease state but may also influence the regulatory function of PCM and therefore chondrocyte activity. 9 Multiple studies have suggested that associated change in PCM composition, structure, and mechanical function is present in OA.
Studies examining advanced stages of OA show composition changes of collagen expression including significant upregulated expression of collagen type VI 30 and overlapping localization of collagens I, II and III in the PCM. 31 Via the chondrocyte’s primary cilium (organelle that projects from the cell surface into the PCM) these changes in composition can significantly affect mechanotransduction in chondrocytes. 9 Distorted cilia length and incidence has been reported with OA severity, implying that there is altered cilia-mediated signaling in degenerative cartilage. 32 The cilium has mechanosensors, a variety of ion channels such as the transient receptor potential vanilloid 4 (TRPV4), and an ability to interact with matrix proteins such as collagens type II and VI. One study showed TRPV4 provokes intracellular Ca2+ signaling cascades in response to mechanical stimuli. 33
Degradation of PCM structure may be one of the earliest events during OA onset
9
secondary to elevated serine proteases
34
that digest several major PCM components
35
leading to the chondrocyte’s increased contact with type II collagen fibrils. Binding of cell receptors, such as DDR2, to type II collagen has been shown to upregulates production of MMP-13 in chondrocytes leading to degradation of type II collagen in cartilage matrix.
36
This highlights the feedback loop of serine proteases (HtrA1) to cell receptors (DDR2) and the MMP13 degradative pathway in OA progression.
37
In summary, a mechanical force on the articular cartilage activates HtrA1 which in turn degrades PCM proteins, thus leading to exposure of the chondrocyte’s surface receptors to type II collagen fibrils in the ECM. Then, binding of type II collagen fibrils to DDR2 upregulates MMP13, thus leading to degradation of collagen (
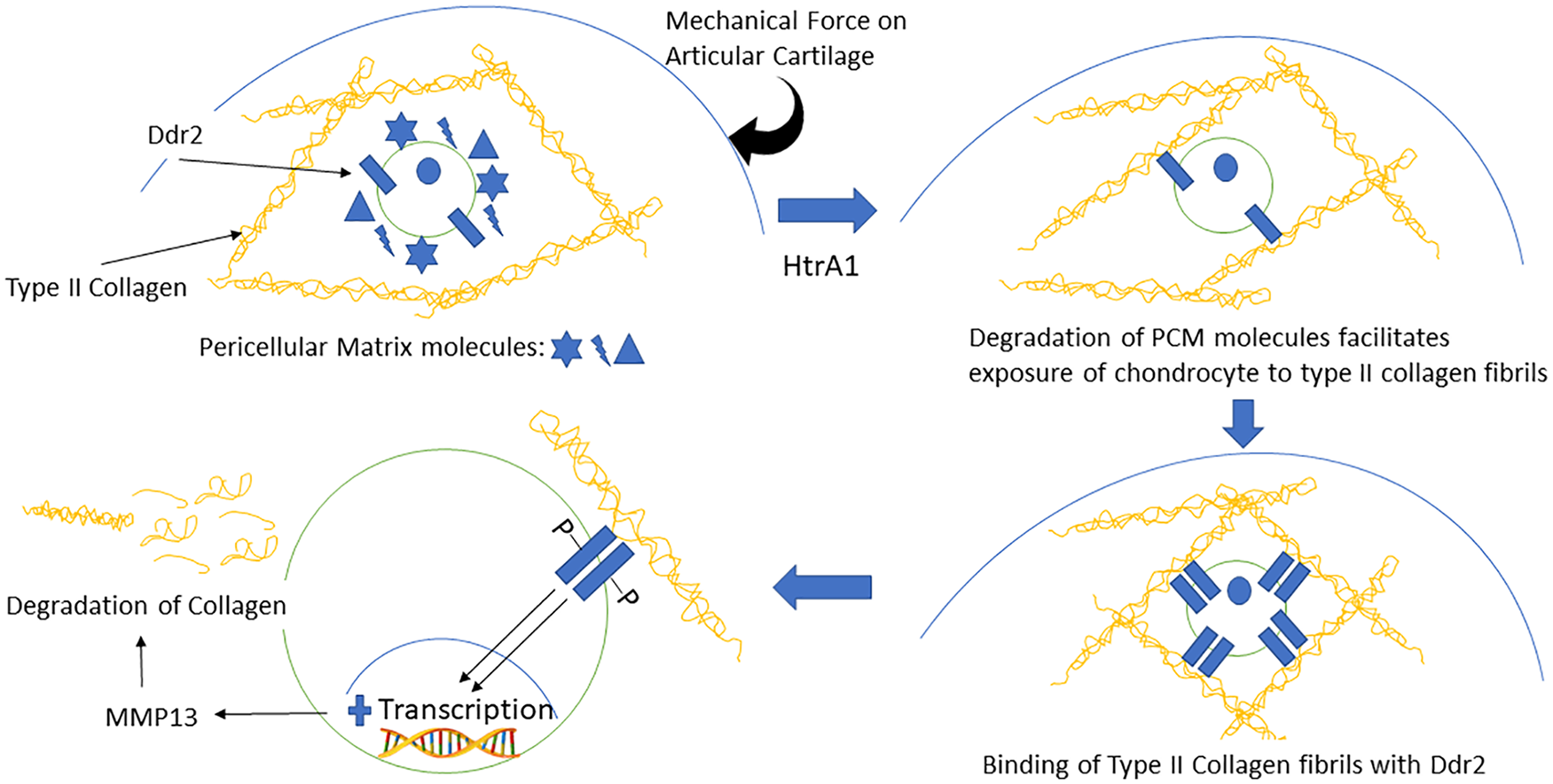
Feedback loop of serine proteases (HtrA1) to cell receptors (DDR2) and the matrix metalloproteinas-13 (MMP13) degradative pathway in osteoarthritis progression. A mechanical force on the articular cartilage activates HtrA1, which in turn degrades pericellular matrix (PCM) molecules, thus leading to exposure of the chondrocyte’s surface receptors to type II collagen fibrils in the extracellular matrix (ECM). Then, binding of type II collagen fibrils to DDR2 upregulates MMP13, thus leading to degradation of collagen.
Clinical Applications Evolving from Chondron and Chondrocytes with Matrix Basic Science
The PCM plays a role in biomechanical transduction, chondrogenic phenotype expression, induction of OA disease processes, and as a filtration unit. Underlying principles regarding the importance of the matrix, including both PCM and interterritorial matrix, has brought about change in multiple cartilage repair procedures including minced cartilage, marrow stimulation, osteochondral allograft, and autologous chondrocyte implantation (ACI).
It has been proposed that the outgrowth of embedded chondrocytes (limited chondrocyte migration is contributing factor to poor cartilage self-healing) can be achieved through increased tissue surface area by mincing the cartilage tissue mechanically into small tissue fragments that covers a large area for new tissue formation.
39
Lu
Multipotent mesenchymal stromal cells (MSCs) describe the nonhematopoietic adult cell population present in various tissues such as bone marrow and adipose tissue that have chondrogenic differentiation capabilities.
41
Marrow stimulation (MS) techniques induce an influx of marrow substrates (MSCs, growth factors, and cytokines) to repopulate a cartilage defect.
42
Stimulation of cartilage matrix production and upregulation of cartilage-specific matrix genes were observed when chondrocytes were combined with MSCs,43,44 thus suggesting that successful combination with freshly isolated articular chondrocytes could promote chondrocytic differentiation of MSC in a 1-step procedure. In a goat model, Bekkers
Growth factors in the surrounding ECM may play an important role in cartilage regeneration. Thus, combining the underlying principles of minced cartilage (migration of chondrocytes and resulting cartilaginous matrix [PCM/ECM] deposition beyond that seen in chondrons) along with scaffolding, the Cartilage Autograft Implantation System (CAIS) Harvester and Dispenser (DePuy Mitek, Raynham, MA) was developed. CAIS is a point of care surgical procedure where cartilage is mechanically minced and then affixed using fibrin glue on a synthetic, absorbable scaffold (designed to keep the tissue fragments in place) that is reinforced with a mesh within the defect. 48 Using a low load-bearing surface (i.e., lateral wall of the intercondylar notch) or during debridement of a cartilage defect (often containing viable cartilage cells), 49 cartilage and accompanying ECM can be harvested. Multiple animal models have shown success with a cartilage fragment seeded scaffold demonstrating high cellularity and intense ECM production as well as hyaline-like repair tissue in terms of morphological, mechanical, and histological assessments.50,51 Another study showed CAIS achieved the highest score compared with ACI in a horse model in terms of arthroscopic, histologic, and immunohistochemistry results. 52 A level II randomized controlled trial (RCT) compared CAIS with MFx in 29 patients with 2-years of follow-up. 48 The multicenter study demonstrated CAIS demonstrated significantly improved International Knee Documentation Committee (IKDC) scores and KOOS compared with MFx at 24 months. There was no difference in terms of fill of the graft bed, tissue integration, or presence of subchondral cysts on MRI.
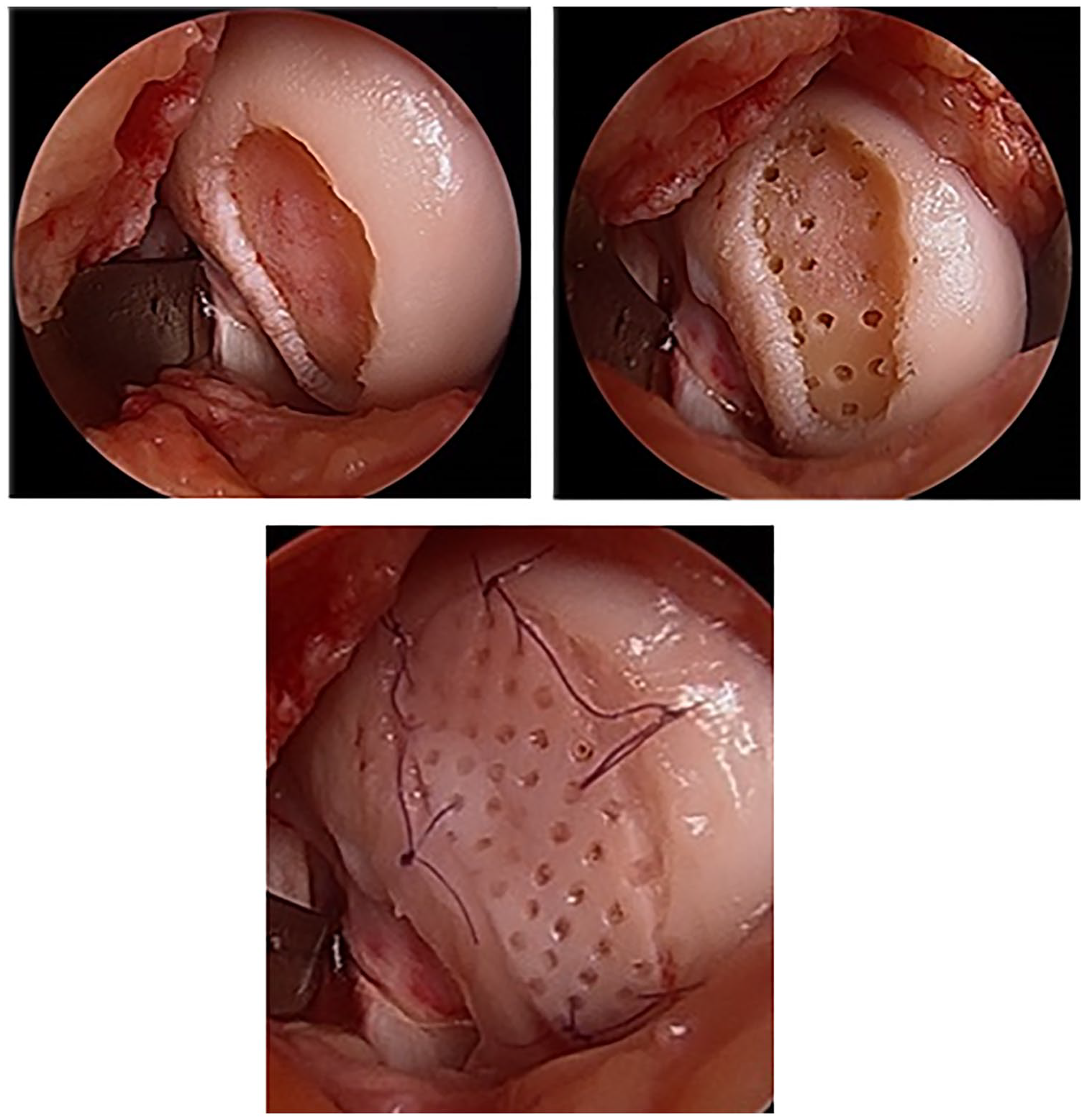
Reveille Cartilage Processor (Exactech, Gainesville, FL) is a single-stage surgical technique that utilizes autologous cartilage combining the underlying principles of minced cartilage (migration of chondrocytes and resulting cartilaginous PCM/ECM deposition analogous to chondrons) along with MS (

Reveille Cartilage Processor technique. (
Multiple cartilage repair products have been developed building on the advances made in the fields of chondrons combined with osteochondral allografts and MS, including a cryopreserved, viable osteochondral allograft with pores spanning the thickness of the graft called Cartiform (Arthrex Inc., Naples, FL, USA). Harvested from human cadaveric specimens, the allograft consists of full-thickness articular cartilage and a thin layer of subchondral bone with perforations in the articular cartilage that allows for flexible conformity and improved integration to the underlying subchondral bone.
55
Of note, the implant has a long shelf life (frozen storage for up to 2 years) secondary to pores which enable the cryopreservation solution to penetrate the tissue to maintain cell viability.
56
The chondrocytes along with extracellular matrix proteins, chondrogenic factors (PCM plus surrounding ECM [beyond that seen in chondrons]), and a thin osseous portion are cryopreserved at −80°C.
55
Following MS, Cartiform serves as a scaffold for the MSCs while the viable chondrocytes simultaneously release PCM/ECM inducing the MSCs to undergo chondrogenesis and produce hyaline cartilage. Nonmetallic and nonbone reactive suture anchors may be used to fix Cartiform in place
57
or secured to the surrounding healthy cartilage using absorbable sutures.
58
Fibrin glue is applied between the graft periphery and the native cartilage walls, avoiding the holes in the graft, as a final step to secure the graft in place (
Cartiform (Arthrex Inc., Naples, FL, USA) is a cryopreserved, viable osteochondral allograft (frozen storage for up to 2 years) harvested from human cadaveric specimens characterized by a minimal amount of bone and pores spanning the thickness of the graft. Following marrow stimulation, Cartiform serves as a scaffold for the mesenchymal stem cells (MSCs) while the viable chondrocytes simultaneously release growth factors into the adjacent microenvironment theoretically inducing the MSCs to undergo chondrogenesis. Surgical images courtesy of senior author (KM).
Similarly, ProChondrix (AlloSource, Centennial, CO) is a laser-etched, fresh cryopreserved osteochondral allograft that provides functional cells and PCM plus surrounding ECM (beyond that seen in chondrons) to a defect site. Following MS, ProChondrix works as a scaffold for the MSCs while the viable chondrocytes concurrently release PCM/ECM to help promote chondrogenesis. In a unpublished series, Mehta
Finally, combining ACI with knowledge learned from studying the PCM has led to advances in technologies. Chondrocyte de-differentiation,
61
during
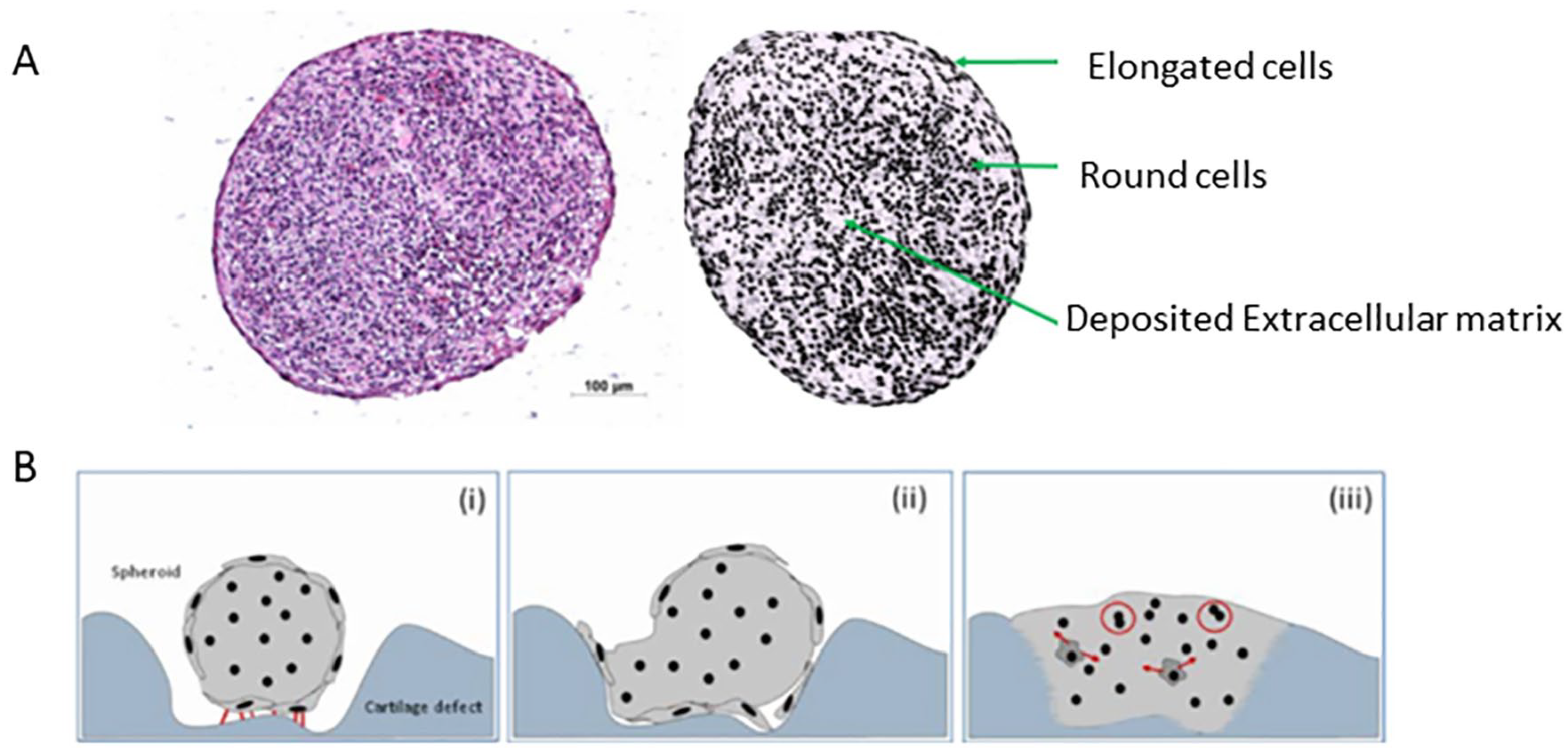
Autologous chondrocyte implantation with Spherox (formerly called chondrosphere). (
Conclusion
In summary, the use of isolated chondrons as a model for studying the chondrocyte microenvironment including the ECM has enhanced our grasp of cartilage biology. Execution and improvement of cartilage engineering protocols with emphasis on the microenvironment of chondrocytes/MSCs and ECM could lead to the production of more robust and functional cartilage. Lesions in articular cartilage can cause considerable musculoskeletal morbidity including pain and loss of function. With that comes significant economic implications, especially when considering its progression to OA. 70 The ambition of a chondrocytes with matrix therapy aims to bring about a repair without the need for further long-term surgery. Bearing this in mind, we have highlighted the ongoing research encompassing novel tissue sources and cell types that are being developed and employed for cartilage repair utilizing underlying chondrocytes with matrix theory.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
