Abstract
Objectives
Ulcerative colitis is a systemic inflammatory disease which primarily involves the gut but presented by numerous extraintestinal manifestations. The effect of ulcerative colitis on knee cartilage has not been evaluated up to the present. In the current study, we aimed to investigate the possible relationship between the presence of ulcerative colitis and femoral cartilage thickness.
Design
Sixty-two patients with confirmed diagnosis of ulcerative colitis and 70 healthy controls aged 18 to 50 years referred to the gastroenterology outpatient department between January 2018 and January 2019 participated in this cross-sectional study. The measurements were made by ultrasonography with the patient in a supine position and the knees in complete flexion. Demographic, clinical, endoscopic and laboratory data were collected for all the subjects.
Results
The groups of ulcerative colitis and control group were similar with regard to sex, mean age, weight, height, body mass index, extremity dominancy, and existence of knee pain (
Conclusion
Knee cartilage was thinner in subjects with mild activity ulcerative colitis than in healthy controls. Decreased knee cartilage thickness may be an indicator of extraintestinal manifestation in patients with mild activity ulcerative colitis. This association between ulcerative colitis and knee cartilage degeneration may be effective in early detection of possible risk factors and potential treatment strategies for both ulcerative colitis and specific subtypes of knee osteoarthritis.
Keywords
Introduction
Ulcerative colitis is a systemic inflammatory disease that primarily involves the gut but is accompanied by numerous extraintestinal manifestations (EIM). 1 Approximately 50% of patients with inflammatory bowel disease (IBD) deal with at least 1 EIM during the course of disease. 2 The development of EIM decreases the quality of life, increases the morbidity and may result in disability. 2
Rheumatological and musculoskeletal manifestations cover a remarkable portion of EIMs, which have a complex pathogenesis and have not been fully understood. These mechanisms include ectopic expression of gut-specific chemokines and adhesion molecules, T cell flowing conducted by non-specific adhesion molecules, microbial antigen translocation and/or cross-reactivity, circulating antibodies, or self-directed inflammatory events sharing prevalent genetic or environmental risk factors with IBD.
3
The role of inflammation in the ethiopathogenesis of osteoarthritis (OA) has contradictory results with a general acceptance that the well-balanced quantities of inflammatory pathways are necessary for cartilage homeostasis and tissue repairs. The increased inflammatory mediators contribute to ethiopathogenesis of cartilage degeneration and OA.
4
So we hypothesize that the probability of early femoral cartilage degeneration and eventual OA may be an extraintestinal manifestation of ulcerative colitis or
As far as we know, the effect of ulcerative colitis on knee joint cartilage has not been assessed up to present. Ultrasonographic (US) evaluation of knee joint cartilage thickness appears to be an effective and valid method to evaluate cartilage degeneration. The aim of the current study was to investigate the possible relationship between the presence of ulcerative colitis and knee cartilage thickness.
Materials and Methods
Patient Selection
Sixty-two patients with confirmed diagnosis of ulcerative colitis and 70 healthy controls aged 18 to 50 years referred to the gastroenterology outpatient department between January 2018 and January 2019 participated in this cross-sectional study. Demographic and clinical properties of the subjects including sex, age, height, weight, body mass index (BMI), and extremity dominancy were recorded. Patients diagnosed with ulcerative colitis by clinical, endoscopic, and histopathological findings, who have been followed up for at least 1 year, and who did not use immunosuppressive drugs—antitumor necrosis factor (TNF), steroids, azothiopurines, and so on—were included in the study. Participants with a background of another systemic disease (and chronic medication use), knee injury, septic/inflammatory arthritis, any surgeries of lower extremities or ulcerative colitis, smoking, varus and valgus alignment or any intra-articular treatment within the last 6 months were not included.
Clinical activity assessment was made by modified Truelove and Witts criteria of the ulcerative colitis. According to these criteria only patients with mild activity were included in the study. Patients with moderate and severe activity (the patients who needed immunosuppressive therapies) were excluded.
Written informed consents form were filled up by participants in the study. Approval of the study was obtained from local ethics committee of Health Sciences University/Istanbul, Turkey.
Measurements of Femoral Articular Cartilage
Linear probe was used to evaluate femoral articular cartilage thickness (M Turbo, Sonosite) by the same physiatrist who had at least 5 years of experience in musculoskeletal US and was blind to patient groups.
General rules on US assessment of OA pathology were applied, which included the following: choosing the highest frequency that allows the visualization of the target area, adopting a multiplanar scanning technique to document US findings indicative of OA on at least 2 perpendicular planes of scanning and performing dynamic examination during flexion–extension movement.5,6 Scanning protocol for a tailored US assessment included the evaluation of the articular cartilage involvement, the identification and measurement of the osteophytes and the detection of joint inflammation. The weight-bearing surfaces of the femoral condyle were scanned in the suprapatellar region with the knee fully flexed.
Femoral articular cartilage thickness was evaluated with the subject in supine position and knees in complete flexion. The probe was located (in the transverse plane) axially over the exterior side of patella. Right and left knee cartilages were evaluated from the middle area of lateral condyle (LFC), medial condyle (MFC), and intercondylar area (ICA). The measurement of cartilage thickness performed by the extent of the straight hyperechoic edge at the bone-cartilage interface and the thin hyperechoic edge at the cartilage–synovial space interface (
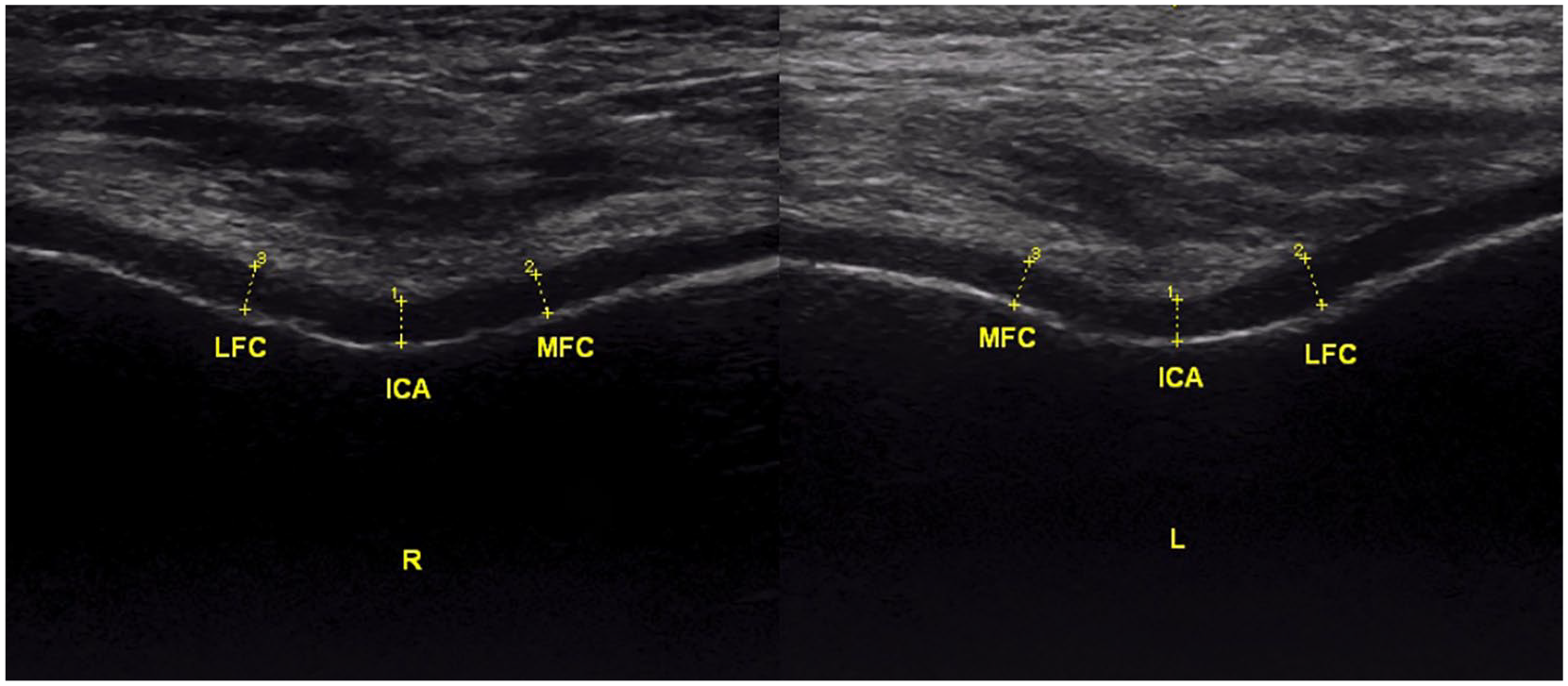
Ultrasonographic view (suprapatellar transverse view) demonstrating the knee cartilage evaluations. Right and left knee cartilage thickness was measured from the middle area of lateral femoral condyle (LFC), medial femoral condyle (MFC), and intercondylar area (ICA). Cartilage thickness evaluated by the extent of the straight hyperechoic edge at the bone-cartilage interface and the thin hyperechoic edge at the cartilage–synovial space interface. L, left; R, right.
Statistical Methods
Statistical analysis was done using SPSS version 15.0. (IBM SPSS Statistics, IBM Corp, Armonk, NY, USA). The power of the study was 90%. Descriptive statistics (percentage, mean, median, distributions) of the study group were determined. Kolmogorov-Smirnov test was used to determine conformity to normal distribution. Student
Results
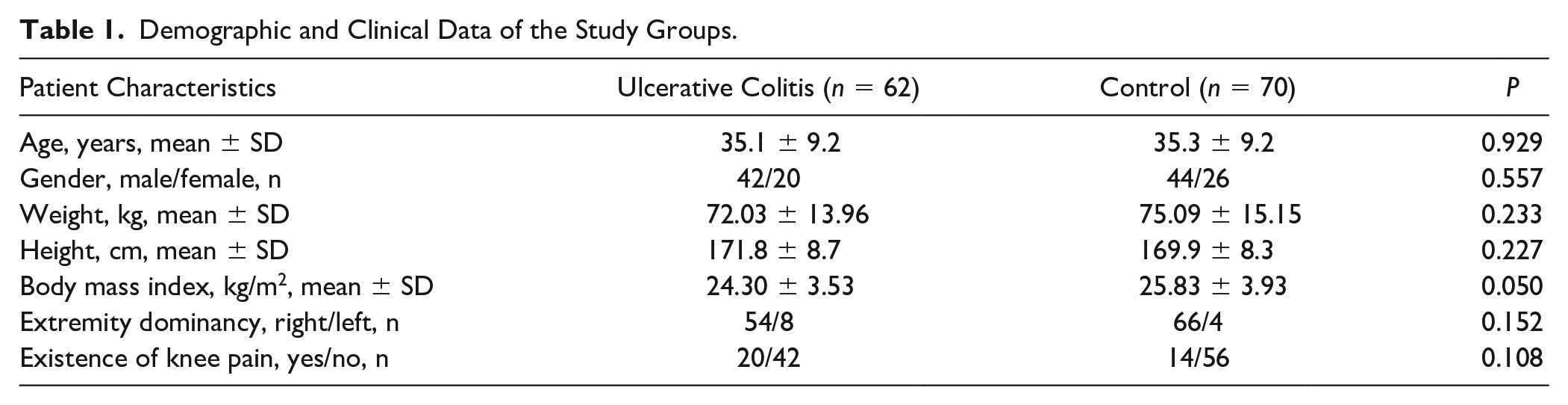
Demographic and clinical data of the participants are presented in
Table 1
. The groups of ulcerative colitis and control group were similar with regard to sex, mean age, weight, height, BMI, extremity dormancy, and existence of knee pain (
Demographic and Clinical Data of the Study Groups.
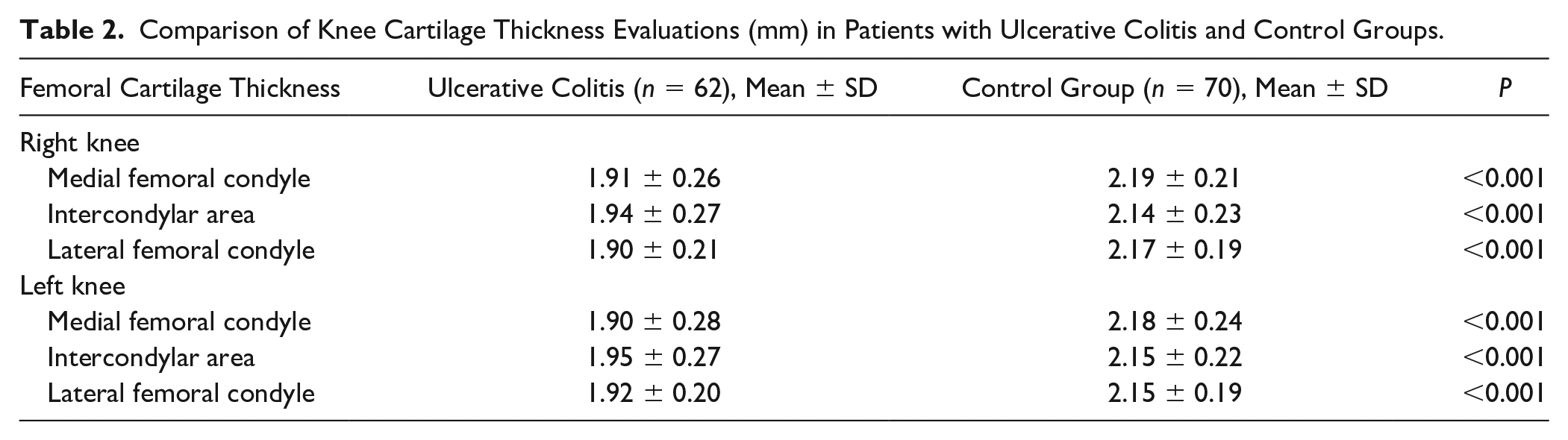
The comparisons of the knee cartilage thickness evaluations of groups are given in Table 2 . Medial femoral condyles, ICAs, and LFCs of both right and left knees had thinner cartilage thickness in ulcerative colitis group than control group (p<0,001).
Comparison of Knee Cartilage Thickness Evaluations (mm) in Patients with Ulcerative Colitis and Control Groups.
A representative image of US from control and ulcerative colitis patients for knee cartilage is presented in Fig. 2 .
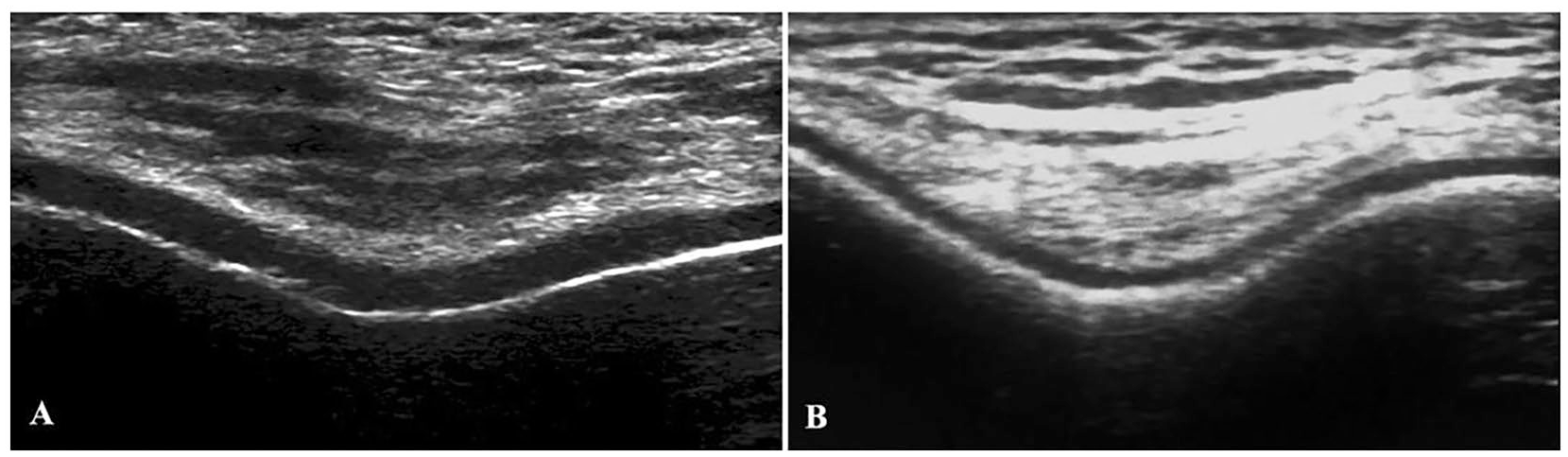
A representative ultrasonographic image from control and ulcerative colitis patients: (
Signs of joint inflammation were also compared between the ulcerative colitis and control groups. Both joint effusion (
Discussion
The aim of the present study was to evaluate the effects of ulcerative colitis on knee cartilage thickness. Knee cartilage thickness was evaluated with US, which has previously been shown to be practical in the determination of cartilage degeneration and knee OA.7,8 US also provides a quantitative assessment of joint cartilage thickness and indicates loss in the sharpness of the cartilage margin, which is practical in early determination of knee OA.
The conclusions of the present work are supported by data of a single technique—US. Therefore, we believe it is important to state its advantages over limitations. US has limitations such as the lack of a standardized measuring method and validated scoring system, operator dependent imaging and reliability, establishment of adequate acquisition parameters. On the other hand, US has major advantages over the other modalities in that it is a highly accurate, reliable, reproducible, fast, economic, and patient-friendly method.9
-11 Also US has lower demands for processing, lack of radiological exposure, and provides valuable practical information about the current status of the knee joint.
9
Maeguchi
Joint and musculoskeletal involvements have well-known associations with IBD.
12
EIM of IBD is defined as an inflammatory entity that is triggered by the inflammation in the intestine but is observed outside of the gut.
13
Karmiris
Nevertheless, pathogenesis of musculoskeletal manifestations in IBD is very complex and unclear. 16 For example, environmental factors may influence development and outcomes of inflammatory bowel disease. 17 Yet, immunological mechanisms and independent inflammatory conditions with similar genetic or environmental risk factors with IBD plays the major role. 3 Upregulation of inflammatory adhesion molecules and chemokines allows capture of effector cells and facilitates their recruitment into extraintestinal areas. Also, T cells from the intestinal mucosa of IBD patients express chemokine receptors that provide entrance to other areas. Low-grade inflammatory process in the extraintestinal areas ensures the uptake of effector cells produced in the gut and moreover intensifies the inflammation. In addition to that, circulating antibodies, immune complex mediated inflammation, increased levels of interleukin-1-β (IL-1-β) and TNF-α has been proposed to contribute to EIM. 3
On the other hand, cartilage degeneration and osteoarthritis are also believed to have immunologic mechanisms besides others. The key inducers of catabolic processes in OA are IL-1 and TNF-α. 18 These inflammatory cytokines are associated with disease progression in both ulcerative colitis and OA. Both IL-1 and TNF-α trigger the synthesis of chondrocyte for degrading enzymes in cartilage, which are essentially ADAMTS metalloproteinases (MMP-1, MMP-3, MMP-13).
Patients with ulcerative colitis frequently suffer from skeletal abnormality, such as reduced bone mineral density, increased fracture risk, and/or joint inflammation. This pathological process is characterized by altered immune cell activity and elevated inflammatory cytokines in the bone marrow microenvironment due to disrupted gut immune response. Ulcerative colitis is recognized as an immune malfunction driven by multiple factors, including cytokines and signaling molecules. However, the mechanism by which intestinal inflammation magnified by gut-residing actors stimulates skeletal abnormality remains to be elucidated. On the other hand, osteoarthritis is a complex, chronic disease that affects primarily the weight-bearing joints of humans and other mammals. 19 During progression of the disease, all compartments of the joints undergo structural, functional, and metabolic changes that involve cellular elements as well as components of the extracellular matrix. The current evidence clearly suggests a link between increased levels of chemokines, cytokines and IBD pathogenesis. Several studies have confirmed the link between joint and gut inflammation. 19 It seems likely that both bacterial antigens and reactive T-cell clones, activated into the gut home the joint. However, the exact immunological mechanisms linking gut and joint inflammation are not fully understood. Various environmental (gut bacteria-dysbiosis) and host factors (migration of activated gut-T cells and macrophages) leading to initiation of inflammation in genetically predisposed individuals may act as triggers of inflammatory responses against gut and joints components. Additionally, it has been shown that macrophages from the gut of IBD patients are able to adhere in endothelial cells of synovial tissue, further enhancing the activation of T cells locally.
Subchondral bone and the synovium play an important role in the initiation and progression of OA.4,18 The subchondral bone in OA undergoes an uncoupled remodeling process, which is notably characterized by macrophage infiltration and osteoclast formation. Metabolic and mechanical factors can lead to synovitis in OA. Synovial tissue is highly vascularized and thus exposed to systemic influences such as low-grade inflammation. Synovitis can induce cartilage damage by fibrin deposition, pannus formation, or via inflammatory cytokine expression and triggers osteopenia, notably in early OA stages. Thus, besides mechanical impairment, underlying systemic factors trigger bone remodeling and might be taken into consideration in the treatment of OA.
The effectiveness of biological agents in EIM treatment may contribute to reveal the obscure inflammatory pathways. Anti-TNF-α drugs have high treatment success for arthritis, cutaneous and ocular EIM. This brought speculation about TNF-α-related mechanisms in EIM pathogenesis.
3
Peyrin-Biroulet
Identification and management of EIMs are an important challenge in IBD practice. We consider it is unclear that whether our current findings are directly related to intestinal inflammation or these 2 entities share a genetic sensitivity with an impaired immune response to external stimuli. 1 Prospective studies with answers to these questions may introduce more effective treatment options.
Several anti-cytokines licensed for use in ulcerative colitis have also been studied in OA. Such cytokines may implicate innate immunity, including macrophages in OA development. 24 Multidirectional research into the mechanisms of IBD and OA has been rapidly growing in the recent years. 25 New inflammatory pathways and possible mechanisms of action have been disclosed, potentially leading to new-targeted therapy. However, the great disease heterogeneity and the overwhelming contribution of environmental risk factors has not modified yet the disease management. Despite a large amount of data regarding the pathogenesis, the translational results are still poor and there is a significant gap in understanding the direct consequence of the identified pathways. However, the discovered mechanisms explain only a small portion of disease variance. The possibility for the future of a better prediction of disease course, response to therapy and therapy-related adverse events may allow a more efficient and personalized strategy.
The relatively small sample size was the first limitation of this study. In addition, the current study was planned as a cross-sectional study that has its own limitation, which is the lack of causality assessment between factors. Also, it would have been better if we had the data of disease durations of patients with ulcerative colitis for investigating the correlations between femoral cartilage thickness and disease durations. Nevertheless, we think our results are significant to imply the relationship between ulcerative colitis and femoral cartilage degeneration.
In conclusion, the current study showed that knee cartilage is thinner in subjects with mild activity ulcerative colitis then in healthy controls. This association between ulcerative colitis and cartilage degeneration may be useful to identify targeted prevention and potential treatment strategies for ulcerative colitis and specific knee OA subtypes. Further studies are needed to evaluate the mechanisms and causality assessment between these entities.
Footnotes
Authors’ Note
The datasets generated and/or analyzed during the current study are not publicly available due ethical statement, but a limited and fully anonymized dataset containing the individual patient data that support the main analyses is available from the corresponding author on reasonable request.
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
We are confirming that the protocol for the research project has been approved by Ethics Committee of Health Science University, Istanbul within which the work was undertaken and that it conforms to the provisions of the Declaration of Helsinki (as revised in Brazil 2013).
Informed Consent
All participants provided written informed consent.
Trial Registration
Not applicable.
