Abstract
Objective
To compare the distal femoral cartilage thickness of patients with sickle cell disease (SCD) with those of healthy subjects using ultrasonography.
Methods
The study comprised 30 patients with SCD (16 male, 14 female; mean age, 30.1 years) and 30 age- and sex-matched healthy subjects. Demographic features and medications of the patients were recorded. With the knees held in maximum flexion, the femoral cartilage thickness was measured bilaterally with a 7- to 12-MHz linear probe. Using ultrasonography, 3 midpoint measurements were taken from both knees: lateral femoral condyle (LFC), intercondylar area (ICA), and medial femoral condyle (MFC).
Results
Patients with SCD had thinner femoral cartilage thickness values at LFC (P = 0.004), at MFC (P = 0.000), and ICA (P = 0.002) when compared with those of the healthy subjects. Patients with SCD also had lower Hb levels (P = 0.000) levels. Weak positive correlations were determined between Hemoglobin (Hb) levels and ultrasonographic measurements in the SCD group at MFC (r = 0.331, P = 0.010), and ICA (r = 0.289 , P = 0.025 ). Low levels of Hb seem to affect the femoral cartilage thickness.
Conclusion
These preliminary findings of decreased femoral cartilage thickness in SCD patients should be complemented with future studies. The possibility of early knee joint degeneration and eventual osteoarthritis in SCD should be kept in mind.
Introduction
Sickle cell disease (SCD) is one of the most globally common inherited blood disorders due to homozygosity for abnormal hemoglobin. 1 The disease is characterized by vaso-occlusive phenomena and hemolysis. Several organs can be affected with alterations in the central nervous system, heart and circulatory system, respiratory system, gastrointestinal system, kidneys, bones, and joints, resulting in increased morbidity and mortality in this group of patients.2,3 Although osteoarticular involvement (osteonecrosis, osteomyelitis, and arthritis) in SCD has been studied, 4 to the best of our knowledge, there have been no studies to date of cartilage involvement in SCD.
Cartilage, which is an avascular tissue, has low metabolic activity and cell density, and gains most of its nutrition and oxygen supply via diffusion from the synovial fluid. There are several factors that control the cartilage integrity such as anabolic or catabolic growth factors and enzymes. 5 In a study by Schumacher et al. 6 using light microscopy, erosion and chondrocyte loss were shown with deep clones in the cartilage and congested vessels with extravasation of red blood cells and mononuclear cells in the synovium. Hypoxia is also accepted as one of the permanent stresses that affect adult articular cartilage. 5 However, to the best of our knowledge, the ultrasonographic knee cartilage thickness in SCD has not been investigated in literature. Accordingly, the aim of this study was to compare the distal femoral cartilage thickness in patients with SCD with those of healthy subjects.
Methods
A total of 30 patients with SCD (16 male, 14 female) and 30 age- and sex-matched healthy subjects (16 male, 14 female) were enrolled in this cross-sectional study. Informed consent was obtained from all the participants. Approval for the study protocol was granted by the Local Ethics Committee.
The demographic and clinical characteristics of the patients, including age, sex, body mass index (BMI), exercise and smoking status, and laboratory data (complete blood count, total bilirubin) were recorded. If the patients performed physical activity of moderate to high-intensity for at least 30 min/d and at least 3 times per week, the patients were evaluated as exercising.
Patients with a history of knee trauma, any systemic and/or chronic disease, including diabetes mellitus, thyroid abnormalities, or rheumatoid arthritis were excluded.
Ultrasonographic Evaluation
Ultrasonographic femoral cartilage thickness measurements were performed on all patients by the same physiatrist (M.T.Y.) using a linear probe (7-12 MHz, Logiq P5, GE Medical Systems, Waukesha, WI, USA) while the patients were in a supine position with their knees in maximum flexion. The probe was placed in an axial position on the suprapatellar region. 7 The cartilage thickness was interpreted as the distance between the thin hyperechoic line at the synovial space/cartilage interface and the sharp hyperechoic line at the cartilage-bone interface.8,9 The femoral cartilage thickness was assessed bilaterally from the midpoints of lateral femoral condyle (LFC), intercondylar area (ICA), and medial femoral condyle (MFC) using ultrasonography ( Fig. 1 ).
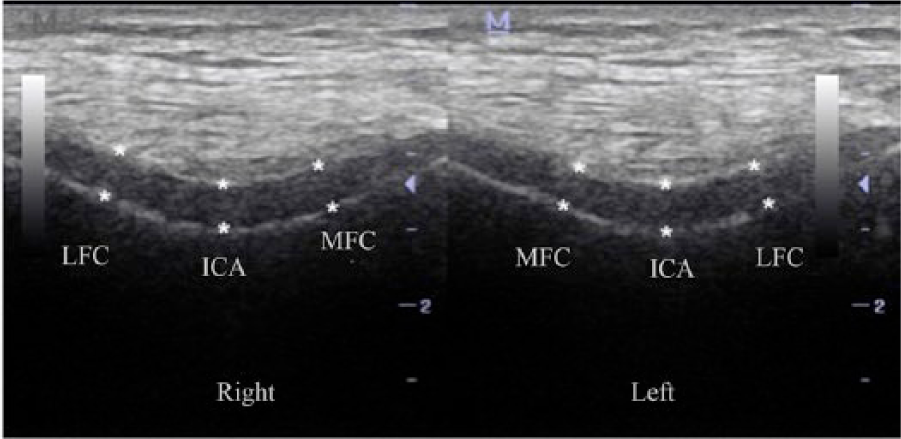
Ultrasonographic image illustrating the distal femoral cartilage measurements in both right and left knee. LFC, lateral femoral condyle; ICA, intercondylar area; MFC, medial femoral condyle.
Statistical Analysis
Statistical analysis was performed using SPSS 22.0 software. Data were expressed as mean ± standard deviation or percentage. Conformity to normal distribution was assessed with the Kolmogrov-Smirnov test. Comparisons of the mean values were made using the independent-samples t test and the chi-square test was used for comparisons between groups where appropriate. Pearson coefficients were used for correlation analyses. A value of P < 0.05 was accepted as statistically significant.
Results
The demographic and clinical characteristics of the subjects are shown in Table 1 . The patient and control groups were similar in respect of mean age, sex, BMI, exercise status, and the number of smokers (all P > 0.05). The comparisons of the femoral cartilage thickness measurements of 60 knees of 30 patients are given in Table 2 . A weak positive correlation was determined between the hemoglobin (Hb) levels and the cartilage thickness values of patients with SCD.
Clinical Characteristics of Study (Sickle Cell Disease) and Control Groups. a
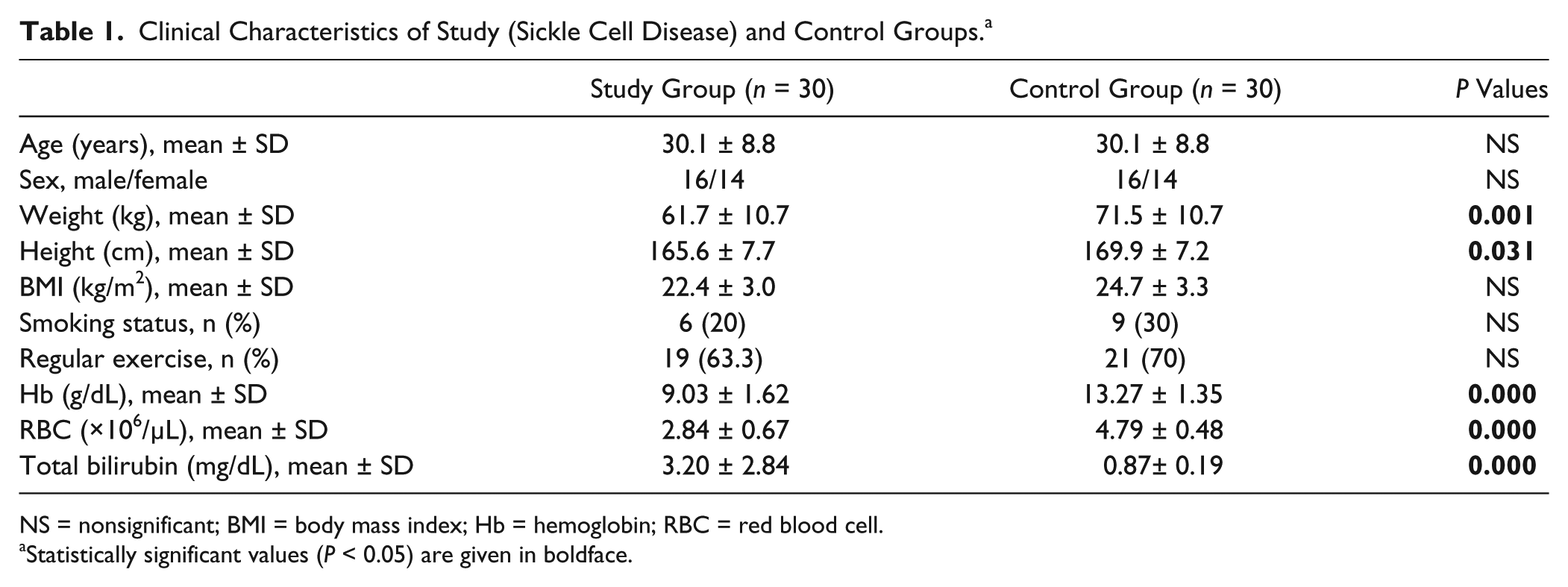
NS = nonsignificant; BMI = body mass index; Hb = hemoglobin; RBC = red blood cell.
Statistically significant values (P < 0.05) are given in boldface.
Comparison of Femoral Cartilage Thickness (mm) Measurements of 60 Knees.
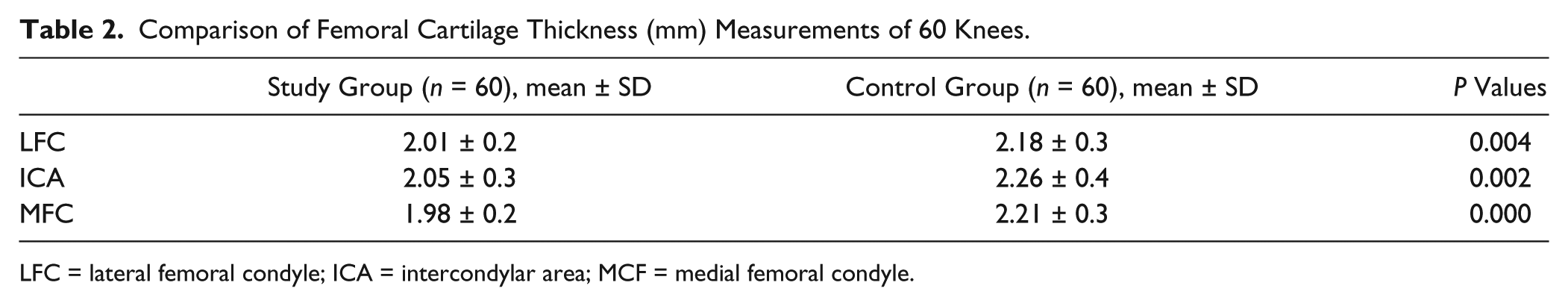
LFC = lateral femoral condyle; ICA = intercondylar area; MCF = medial femoral condyle.
Discussion
In the present study, an assessment was made of the distal femoral cartilage thickness of patients with sickle-cell disease, which to the best of our knowledge, is the first in literature. The results of this study have shown that patients with SCD have thinner distal femoral cartilage.
The risks of mortality and morbidity related to anemia are well known and generally refer to organ failure and even death in severe cases, as a result of anemia-induced tissue hypoxia. 10 Two permanent stress factors on articular cartilage are hypoxia and mechanical loading. Although there is consensus that loading and compression are potent regulators of chondrocyte physiology,11,12 there is still room for debate on the impact of hypoxia on chondrocyte function. As cartilage develops in a hypoxic environment all through life,13-16 the response of chondrocytes to hypoxia may differ from that of other tissues. It has been estimated that oxygen tension within the tissue ranges from approximately 7% on the joint surface to as low as 1% close to subchondral bone. There have been in vitro studies, which have shown that by mimicking the physiological avascular microenvironment of articular cartilage, there is a significant influence of hypoxia or low oxygen tension on cartilaginous matrix production by chondrocytes. Furthermore, Sox9, the chondrogenic marker gene required for the initiation of chondrogenesis, has been shown to be regulated by factors such as hypoxia-inducible transcription factor (HIF), which is known to be vital to chondrogenesis and cartilage development during skeletogenesis.17-22 The expression of collagen type-II has been reported to be significantly higher and type-I lower under hypoxic conditions compared to normoxia. 23 It has also been suggested in recent studies that hypertrophy differentiation of chondrocytes and multipotent stromal cells are suppressed in hypoxia.19,24,25 However, other studies which have examined various conditions affecting cartilage such as degenerative diseases, inflammatory arthritis, aging population, and the increasing prevalence of obesity, many differences have been seen in the measurements of oxygen in synovial fluid, 26 which would indicate that a certain level of oxygen is essential for normal healing and repair in the joint. 27
According to the results of this study, patients with SCD had thinner distal femoral cartilage values when compared with those of healthy subjects. Since the groups were age and gender matched, this difference can be attributed to SCD, on the assumption that the mechanism is associated with possible inhibition of chondrocyte proliferation and phagocytic cells. Lower weight, height, and Hb levels were also associated with thinner cartilage. Initially, these data may suggest that those patients are at high risk of knee osteoarthritis as a thinner cartilage may indicate osteoarthritis. The inflammatory process of SCD that is initiated at birth on the vascular endothelium is suppressed to some extent. Chronic endothelial damage–induced tissue ischemia and infarctions occur all over the body. This condition may be associated with chondrogenesis inhibition. 28 In addition, a previous study of distal femoral cartilage thickness and Hb levels has shown that lower Hb levels had a negative effect on the protection of joint integrity. 29 Therefore, the findings of this current study seem to be reasonable and require further evaluations.
Hemoglobin carries oxygen between blood and tissue. Hb has the capacity to bind nitric oxide (NO) with its high affinity through its iron moiety. Although, NO can be regulatory and even protective at high concentrations, this unstable radical gas and its metabolites can exert profound toxic effects.30,31 Hemoglobin has the capacity to behave as a scavenger to reduce the NO. This data may indicate that lower Hb levels may have a negative effect on the protection of the joint integrity. In patients with SCD, electron microscopy of the synovium has disclosed partially occluded blood vessels and phagocytic cells containing red blood cell debris and crystalline hemoglobin-like material. These observations suggest a role for the phagocytic cells in joint destruction. 6
Sickle cell disease is a systemic microangiopathic process that is characterized by sickle-shaped red blood cells. In this form, the red blood cells change from the usual elastic, biconcave disc-shaped structures to hard forms. Furthermore, the continuous inflammatory process of SCD on the vascular endothelium, which begins at birth, is suppressed to some extent. Chronic endothelial damage-induced tissue ischemia and infarctions occur all over the body. There may be an association of this condition with chondrogenesis inhibition. 28
Hemolysis causes the bilirubin levels in SCD to appear elevated. In a study by Vassilopoulou-Sellin et al., 32 it was reported that bilirubin (and heme) produce strong, dose-dependent, partially reversible inhibition of chondrocyte proliferation in culture without any effect on cell viability. In the current study, the SCD patients were seen to have increased bilirubin (total and direct).
There are some significant limitations to this study. First, the number of patients with SCD was low. Second was the lack of a detailed laboratory investigation of vitamin D, thyroid/parathyroid, and sex hormones, which may affect the femoral cartilage thickness. Third, the treatment of patients with SCD (hydroxyurea), which may have affected cartilage integrity was not taken into consideration. Fourth, the method of cartilage assessment used in the study could be disputed. There have been studies in literature that have recommended volumetric cartilage measurements using magnetic resonance imaging. Nonetheless, relevant literature has also shown ultrasonographic thickness measurements to be acceptable.
In conclusion, the findings of this study have shown that patients with SCD have thinner distal femoral cartilage. The inflammatory process of SCD, lower Hb levels and higher bilirubin levels appear to have a negative effect on femoral cartilage thickness. These data suggest that those patients may be at high risk of knee osteoarthritis as thinner cartilage may indicate osteoarthritis. Further studies are required to provide a better understanding of the clinical relevance of these finding and whether hydroxyurea treatment or blood transfusion could reverse the process. Larger sample sizes, long-term follow-up, and the consideration of histological evaluations will provide credible data.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from Mustafa Kemal University, Medical School Ethics Committee (30/06/2015/107)
Informed Consent
Written informed consent was obtained for anonymized patient information to be published in this article.
