Abstract
Objective
The purpose of this study was to describe the current practice trends for managing symptomatic cartilage lesions of the knee with microfracture among ICRS (International Cartilage Regeneration & Joint Repair Society) members.
Design
A 42-item electronic questionnaire was sent to all ICRS members, which explored indications, surgical technique, postoperative management, and outcomes of the microfracture procedure for the treatment of symptomatic, full thickness chondral and osteochondral defects of the knee. Responses were compared between surgeons from different regions and years of practice.
Results
A total of 385 surgeons answered the questionnaire. There was a significant difference noted in the use of microfracture among surgeons by region (P < 0.001). There was no association between the number of years in practice and the self-reported proportion of microfracture cases performed (P = 0.37). Fifty-eight subjects (15%) indicated that they do not perform microfracture at all. Regarding indication for surgery, 56% of surgeons would limit their indication of microfracture to lesions measuring 2 cm2 or less. Half of the surgeons reported no upper age or body mass index limit. Regarding surgical technique, 90% of surgeons would recommend a formal debridement of the calcified layer and 91% believe it is important to create stable vertical walls. Overall, 47% of surgeons use biologic augmentation, with no significant difference between regions (P = 0.35) or years of practice (P = 0.67). Rehabilitation protocols varied widely among surgeons.
Conclusions
Indications, operative technique, and rehabilitation protocols utilized for patients undergoing microfracture procedures vary widely among ICRS members. Regional differences and resources likely contribute to these practice pattern variations.
Keywords
Introduction
Between 30% and 40% of knee arthroscopies involve a cartilage procedure.1-4 Full-thickness, symptomatic, chondral defects present challenging treatment dilemmas with a variety of treatment options. Lesion characteristics, patient age, and surgeon experience often guide treatment decisions for these defects. Microfracture (MFX), first described in the 1980s, 5 is among the most widely performed and best documented first-line treatment techniques to address cartilage lesions. This is due to MFX being a relatively low-cost, single-stage, arthroscopic procedure with minimal technical demands in comparison with other surgical techniques.
Recent results of long-term randomized studies demonstrate the deterioration of functional results over time and higher reoperation rates of MFX procedures in comparison with other methods.6-10 In a systematic review of level I and II studies, Goyal et al. 11 stated that by 5 years after surgery, MFX treatment failure could be expected, regardless of the size of the index chondral lesion. Moreover, MFX has been associated with inability to maintain clinically meaningful pain relief at 5 years postoperatively. 12 In a cost-analysis performed by Miller et al., 13 MFX had higher costs than osteochondral autograft transplantation (OAT) as a result of the additional surgeries during short- and long-term follow-up associated with MFX. Another major concern with this procedure is the potential for irreversible damage to the subchondral bone. An increasing body of evidence suggests that MFX is associated with overgrowth of subchondral bone at the lesion site and cyst formation.14-16
Given these observations, cartilage restoration procedures have been the focus of increased interest over the past few decades. Current cartilage restoration procedure options include reconstruction using allograft or autograft tissues, ex vivo amplification and reimplantation of chondrogenic cells using autologous chondrocyte implantation techniques, as well as the local application of exogenous growth factors and mesenchymal progenitor cell-based therapies. As a result, there has been a shift in the utilization of MFX as well as the emergence of biologic augmentation aimed at improving the quality tissue repair, referred to as “microfracture plus.”17-19
There is a considerable amount of literature describing the use of MFX within different countries and specialty societies2,4,20-23; however, there are no recent data investigating current international practice patterns, the frequency with which MFX is being used, or the demographics of patients undergoing this procedure. The International Cartilage Regeneration & Joint Preservation Society (ICRS) is the main forum for international collaboration in cartilaginous tissue research and joint preservation including 1200 members from 65 different countries. Therefore, this group represents a global cross section of knee surgeons treating chondral pathology.
The main purpose of this study was to describe the current practice trends for managing cartilage lesions with MFX among ICRS members in terms of patient selection, surgical technique and post-operative rehabilitation regimens. We hypothesized that (1) given recent data demonstrating inferior clinical outcomes at 2 to 5 years, the overall utilization of MFX has decreased; (2) there will be substantial regional differences among ICRS members, with North American surgeons performing less MFX; and (3) we would observe an increased utilization of biologics among all surgeons.
Methods
A 42-item electronic questionnaire was designed to collect data on orthopedic surgeon’s current utilization of MFX procedures for the treatment of symptomatic, full thickness chondral and osteochondral defects of the knee. Each of the items contained multiple answering possibilities (multiple choice), which could be individually selected using check boxes. There were items where only one answer could be marked, while there were other items with multiple possible answers.
The first 8 questions were aimed at obtaining information regarding surgeon demographics such as subspecialty training, experience level, clinical practice type, and clinical practice region. If the percentage of MFX of a surgeon’s cartilage practice was 0 (question 8), then that individual survey was excluded. Thus, only questionnaires filled out by surgeons regularly performing MFX of the knee were included in the analysis. Questions 9 to 25 were a list of factors that have been previously demonstrated to be linked to outcome after MFX procedures, including patient age, body mass index (BMI), malalignment, site of the chondral defect, and specific depth of lesion as well as surgical technique factors, including instruments used and any type of biologic augmentation. Cartilage defect grading (question 16) was according to the ICRS classification. 24 The subsequent 12 questions then focused on postoperative care, including the use of CPM (continuous passive motion) devices, range of motion restrictions, weightbearing status, and duration until return to activities. Finally, the last 5 questions asked about the perceived success of the techniques employed as well as the management of patients who had failed previous MFX treatment.
ICRS members were chosen as the sample group, therefore ICRS representatives were contacted and subsequent permission was received to conduct this survey. The questionnaire was sent to all 1200 members via email in January 2019, and the survey was completed via the web using Qualtrics (Seattle, WA, USA. A reminder email was sent in March 2019 and the survey was closed to responses in April 2019. Repetitive questionnaire completions by the same surgeon were excluded, utilizing the first complete reply. The completed questionnaires were examined and processed anonymously for further analysis.
Statistical Analysis
Items from the completed questionnaires were summarized and reported using descriptive statistics. Chi-square analyses were performed to test for any associations between the use of biologic treatments and practice region or number of years in practice. One-way analysis of variance was used to test for any associations between the self-reported proportion of microfracture cases performed for cartilage repair and practice region or number of years in practice. Tukey-adjusted pairwise comparisons were performed where appropriate (e.g., for statistically significant analysis of variance omnibus test). All comparisons were 2-tailed and used a threshold of P ≤ 0.05 as statistically significant.
Results
Demographics
Of the 1200 ICRS members, 969 were orthopedic surgeons. The survey response rate was 39.7% (385/969). Almost half of the respondents (49%, n = 188) were in practice for more than 15 years and only 51 (13%) less than 5 years. The majority of surgeons (78%, n = 300) completed subspecialty fellowship training in sports medicine, and 202 (53%) were employed in private practice. Overall, MFX represented on average 44% of the respondent’s cartilage practice. ( Fig. 1 ) shows the regional distribution of the surgeons. The response rate for each region was as follows: North America 26% (73/279), Latin America 74% (105/142), Europe 38% (138/365), and Asia-Pacific 31% (56/179). The mean percentage of different cartilage procedures performed by surgeons within these regions can be seen in ( Fig. 2 ). More than half (58%, n = 171) of surgeons perform MFX surgery less frequently than they did 5 years ago.
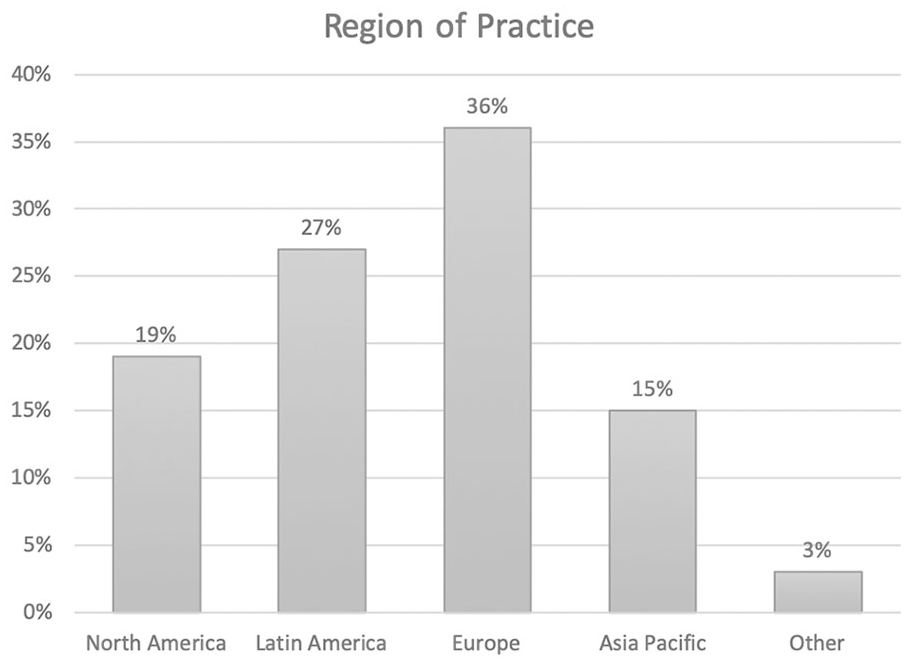
Regional distribution of the respondents (n = 385).
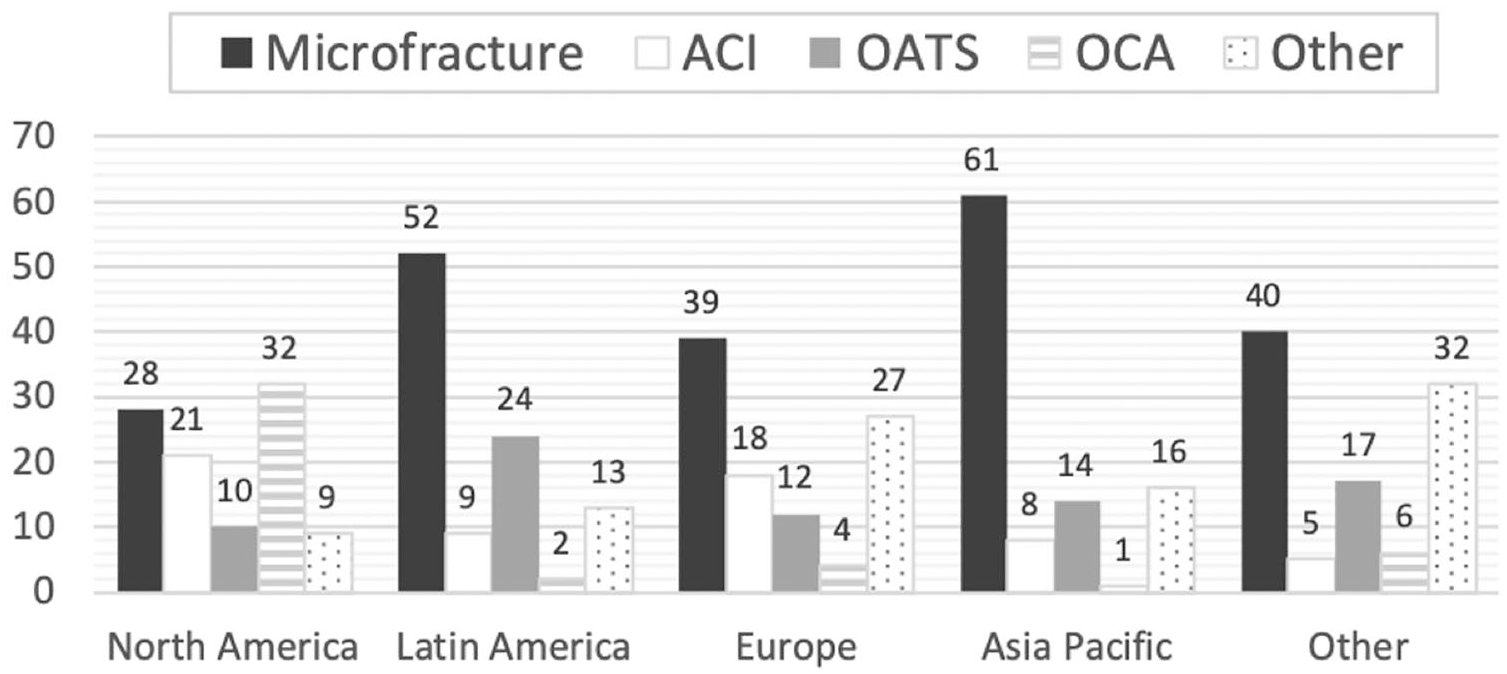
Mean percentage of cartilage practice within regions (n = 385). ACI, autologous chondrocyte implantation; OATS, osteochondral autograft transplantation surgery; OCA, osteochondral allograft transplantation.
There was a significant difference noted in the use of MFX between surgeons in different regions (P < 0.001). Tukey-adjusted pairwise comparisons revealed that surgeons in North America performed a significantly lower proportion of microfracture treatments for cartilage repair than surgeons from Latin America (P < 0.001) and Asia (P < 0.001). Similarly, surgeons in Europe performed a significantly lower proportion of microfracture procedures for cartilage repair than surgeons from Latin America (P = 0.02) and Asia (P < 0.001). There were no differences in the proportion of self-reported percentage of microfracture procedures performed for cartilage repair between surgeons from North America and Europe (P = 0.056), or between surgeons from Latin America and Asia (P = 0.32). In addition, there was no association between the number of years in practice and the self-reported proportion of microfracture cases performed for cartilage repair (P = 0.37).
Of the 385 subjects who completed the questionnaire, 58 (15%) indicated that they do not perform MFX at all and were excluded from the rest of the survey. Most of them were from North America and Europe, 20 (35%) and 24 (41%) respectively. Almost half (43%, n = 25) reported more than 15 years of clinical practice and 24 (42%) less than 10 years.
Indications for Surgery
With regard to lesion size, 183 (56%) surgeons reported that they would limit their indication to MFX cartilage lesions to those lesions measuring 2 cm2 or less, and 115 (35.2%) would extend to 4 cm2. ( Fig. 3 ) shows the maximum size of lesion on which surgeons from different regions would perform MFX surgery. When comparing maximum depth, half of the respondents (50%, n = 163) set their maximum to between 0 and 5 mm below the subchondral plate, followed by no violation of the subchondral plate (37%, n = 122). Using the ICRS classification system, the majority of members would perform MFX surgery on grade 3 and 4 lesions (87% of respondents). There was almost no difference between the indication for traumatic or insidious lesions (49.5% vs. 50.5% of answers) and only 50 (15%) surgeons would consider MFX procedures in cases with associated cystic changes in the subchondral bone.
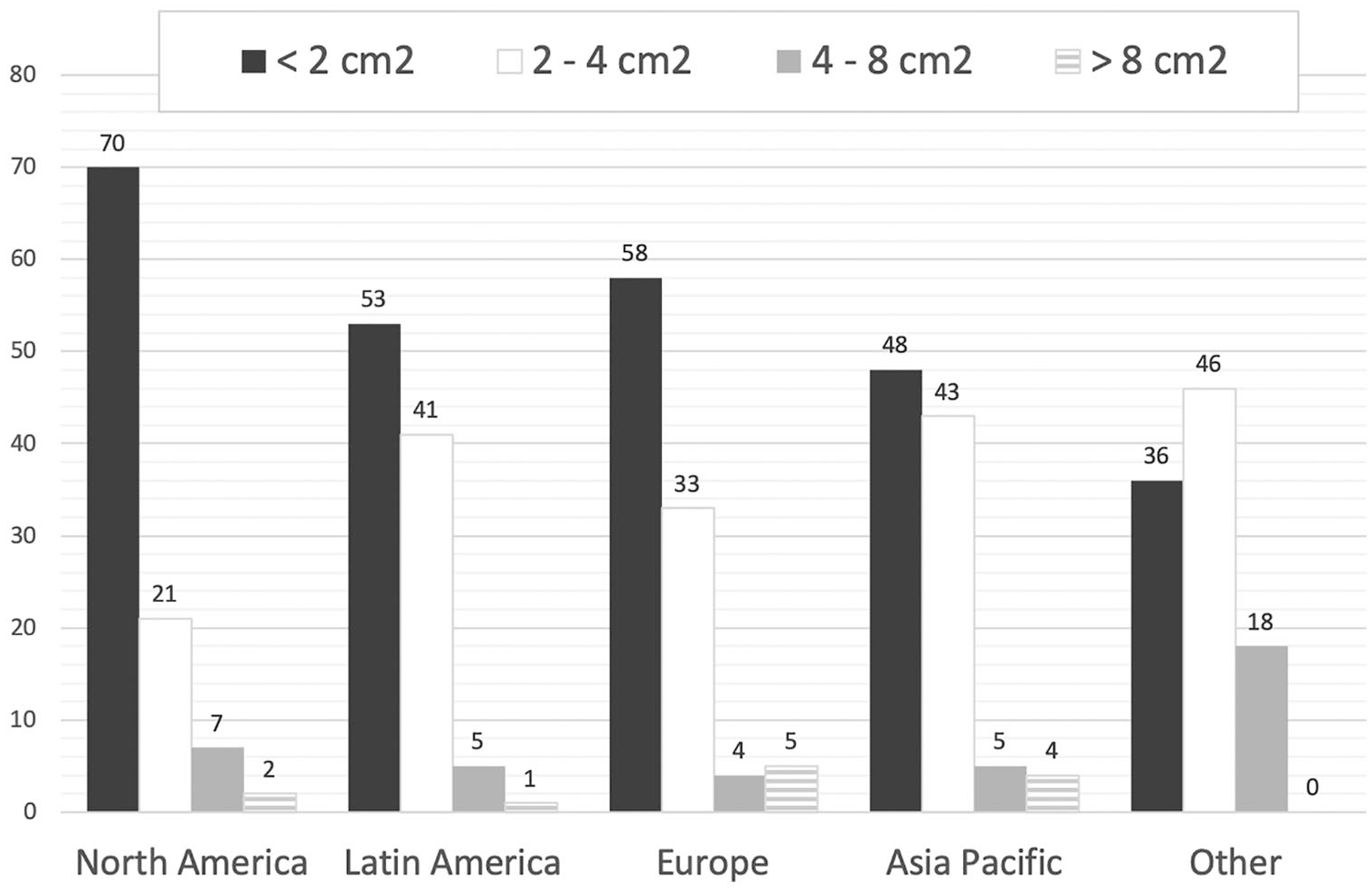
Maximum lesion size on which to perform microfracture (n = 327). Results are shown as mean percentage of responses and sorted by region.
Half of the surgeons (50%, n = 164) reported no upper age limit for the use of MFX and 81 (25%) would use MFX on patients older than 60 years. In a similar way, almost half (49%, n = 159) of the respondents do not have an upper BMI limit and 72 (22%) would perform surgery on patients with BMI >35 kg/m2. With regard to malalignment, 161 (49%) surgeons agreed on 3° to 5° as the maximum degree of uncorrected angle and only 11 (3.3%) would perform MFX with more than 10° of malalignment. Only 40% (n = 132) of surgeons would perform MFX on the tibial plateau and 35% (n = 115) on the patella. Finally, 25% (n = 82) of respondents were not willing to perform MFX on high-level athletes.
Surgical Technique
When subjects were asked about surgical technique, surprisingly 10% (n = 32) of surgeons reported that they do not perform a formal debridement of the calcified cartilage layer and 9% (n = 29) reported that they do not believe that it is important to create stable vertical walls. The most commonly reported MFX instrument was a microfracture awl (70%, n = 229) followed by a Kirschner wire (17%, n = 56). Overall, 47% (n = 150) of surgeons use biologic agents in an attempt to improve the quality of repair tissue and the most common biologic agents were scaffolds (e.g., Biocartilage, Biogide) and injectable agents (e.g., platelet-rich plasma). ( Fig. 4 ) shows the percentage of utilization of biologic agents among different regions. There was no evidence of regional practice differences regarding the use of biologic agents among surgeons from North America, Latin America, Europe, or Asia (P = 0.35). There was also no evidence of differences in the use of biologic agents among surgeons based on the number of years in clinical practice (P = 0.67).
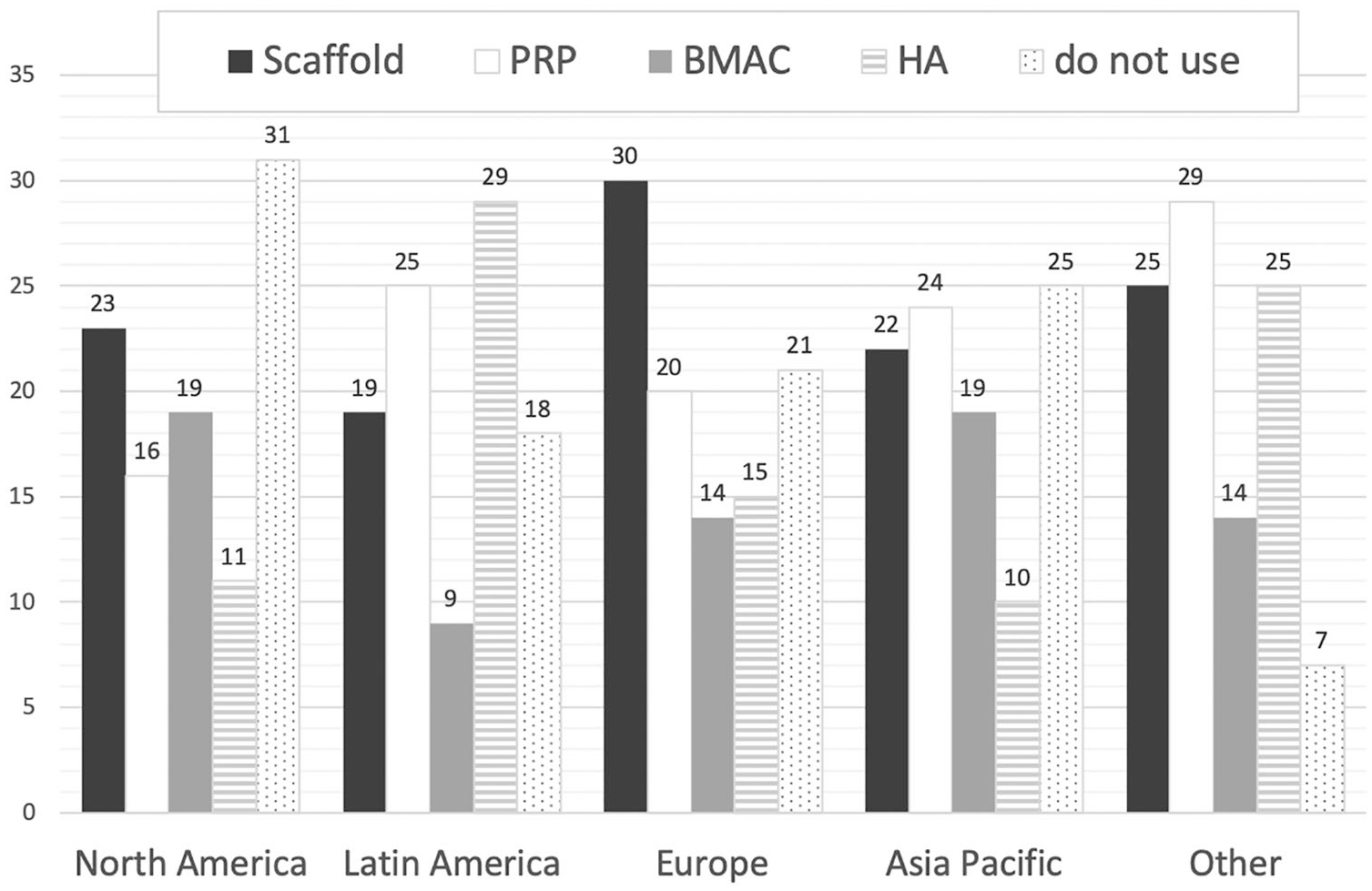
Utilization of biological agents among different regions. Results are shown as mean percentage of responses. PRP, platelet-rich plasma; BMAC, bone marrow aspirate concentrate; HA, hyaluronic acid.
Rehabilitation Protocol
Regarding postoperative management, 171 (52.3%) surgeons reported no concerns with regard to prescribing anti-inflammatory medication following MFX surgery. Rehabilitation protocols varied widely among surgeons, as almost half (51.2%, n = 168) do not recommend bracing regardless of lesion location. The majority (89%, n = 292) would restrict weightbearing following MFX of tibiofemoral lesions, but only 24% (n = 77) of surgeons would restrict weightbearing for patellofemoral lesions. Of those who restrict weightbearing, 64% (n = 200) would initiate partial weight-bearing between 2 and 6 weeks following MFX for tibiofemoral lesions. A total of 208 (64%) surgeons do not restrict range of motion following MFX surgery, and 176 (54%) recommend a CPM device postoperatively. More than half (58%, n = 191) of the respondents allow unrestricted return to activities at 4 to 8 months postoperatively.
Outcomes
Following MFX surgery, approximately half (51%, n = 167) of the surgeons stated that the expected duration of clinical success was 2 and 5 years postoperatively. Only 4% (n = 13) believe that improved outcomes last longer than 15 years, and 14% (n = 46) less than 2 years. In the setting of a failed MFX for a patient who has an isolated 2 cm2 lesion on the femoral condyle with no malalignment, stable ligaments, and an intact meniscus, the next procedure of choice for 52% (n = 170) of surgeons was reported to be OATS, followed by matrix-induced chondrocyte implantation MACI 26% (n = 85). However, if the lesion is located on the trochlea, 45.3% (n = 148) of members would perform MACI and, surprisingly, 15% (n = 49) would consider revision MFX. If this failed MFX is a 4 cm2 lesion on the femoral condyle, 43% (n = 140) of members would perform MACI, and, if it is on the trochlea, the first choice was also reported to be MACI in 53% (n = 174) of respondents.
Discussion
Although MFX is recognized as the gold standard technique to which other cartilage procedures can be compared, many surgeons are considering new first-line treatment approaches as a result of the questionable long-term durability of the fibrocartilage repair tissue, cyst formation, subchondral bone hypertrophy, and the observed decline clinical outcomes at mid-term follow-up.6,8,9,14-16 These considerations correlate well with the findings of this study, as 50% of surgeons believe the benefits of MFX last 2 to 5 years, 58% of surgeons perform less MFX now than they did 5 years ago and 15% of the respondents reported that they do not perform MFX at all.
There was a significant difference in the use of microfracture between regions. The lowest utilization of MFX was observed in North America (28% of practice), where other procedures such as OCA are more commonly used. This compares favorably with previously published U.S. database studies that report utilization of microfracture in 22% of cases.4,23 Moreover, McCormick et al. 2 reported a 3.1% annual incidence growth of restorative procedures during the 2004 to 2011 period in the United States. 2 On the other hand, in Latin America and the Asian-Pacific regions, MFX procedures still represent more than half of cartilage restoration treatments performed by ICRS surgeons. Similarly, we observed that the utilization of biologic agents to improve MFX outcomes in these regions is higher, likely due to decreased availability of other options such as ACI or OCA.
Previously published articles have surveyed orthopedic surgeons within different countries to determine practice patterns for cartilage repair procedures. Elmali et al. 21 found that MFX was the most common cartilage procedure (60%-80%) followed by OATS/mosaicoplasty (20%-40%), among 147 Turkish surgeons. Theodoropoulos et al. 20 published the results of their survey in 2012 regarding the utilization of MFX among Canadian orthopedic surgeons with a response rate of 25%. Similar to our results, the authors found widespread variation among surgeons regarding the indications for MFX, surgical technique, postoperative rehabilitation, and assessment of outcome. Finally, Salzman et al., 22 in a European Survey (242 surgeons from a German-speaking society of arthroscopy), declared MFX was the preferred reparative approach for lesions sized up to 3 cm2. These values correlate well with data from our survey where 90% of European respondents considered 2 to 4 cm2 to be the maximum lesion size on which to perform MFX.
Regarding the indications for MFX, even though it is clear that lesions larger than 4 cm2 are associated with worse outcomes after MFX in comparison with other techniques,9,14,25,26 data from our current survey demonstrate that 9% of surgeons may still attempt MFX in these lesions. Interestingly, half of the surveyed surgeons in our study reported that they do not have an upper age limit and 49% did not report an upper BMI limit for patients being treated with MFX. However, the literature has shown overall better clinical outcomes and MRI-detectable defect fill among younger patients treated with MFX.25,27,28 This finding may be due to the greater prevalence of early degenerative changes among older patients or insufficient numbers of mesenchymal stromal cells (MSCs) being liberated from the subchondral marrow during MFX17,29 procedures performed on older patients. With regard to BMI, Mithoefer et al. 30 reported that a high BMI can adversely affect short-term outcomes, with the worst results being reported among patients with a BMI >30 kg/m2.
Surgical technique is thought to be crucial in order to achieve optimal results. 31 However, 10% of respondents in the current study reported that they do not routinely perform debridement of the calcified cartilage layer and 9% of respondents believe that it is not important to create stable vertical walls at the time of MFX. As previously described, the calcified layer appears to be at least 6 mm beneath the superficial zone, thus, a routine awl will not reach this layer in most knees. When the calcified cartilage layer is not broached, accessing type II collagen fibers will not occur.32,33 Most surgeons (70%, n = 229) reported using an MFX awl as their primary instrument. Although the original surgical technique describes the utilization of an awl 31 rather than a drill or Kirschner wire, current literature supports the use of small-diameter drills for marrow stimulation techniques.15,34-37
Regarding biological augmentation, in our study almost half of the surgeons reported use of biologic agents in an attempt to improve the quality of repair tissue. The most common biologic agents were scaffolds (e.g., Biocartilage, Biogide) and injectable agents (e.g., platelet-rich plasma). There is still considerable controversy surrounding the potential benefit of biologic augmentation since systematic reviews and meta-analysis report mixed results.17,19 In a recent systematic review of basic science literature, Fice et al. 38 noted that a large majority of studies reveal that platelet-rich plasma has beneficial effects on the improvement of the histologic quality of cartilage; however, the lack of standardization of study methodology prevents comparison between studies.
To date, there has also been limited available data regarding rehabilitation protocols after MFX and there is still controversy surrounding the effect of continuous passive motion devices and weightbearing restrictions following cartilage restoration procedures.14,39 Classic rehabilitation protocols described by Steadman et al. 31 promoted the use of a CPM device and, in the case of femoral cartilage lesions, crutch-assisted touchdown weightbearing for 6 to 8 weeks postoperatively, depending on the size of the lesion. For patellofemoral cartilage lesions, the brace was recommended to allow for a range of motion of 0° to 20° for at least 8 weeks after surgery and allowed for weightbearing as tolerated during this period. As expected, rehabilitation protocols varied widely among the surveyed surgeons in the current study. Almost half (51.2%, n = 168) do not recommend bracing regardless of lesion location. The majority of respondents would restrict weightbearing following MFX of tibiofemoral lesions, but only 23% would restrict weightbearing following MFX of patellofemoral lesions. Almost half of the surveyed surgeons would recommend a CPM device after surgery and 60% do not restrict range of motion following MFX procedures.
A clear weakness of this study was the relatively low response rate. Of the 969 ICRS members who identify as orthopedic surgeons, we received a total of 385 responses (39.7%). Additionally, a variable response rate was observed among the 4 regions. Based on these results, it is unclear if this poor low response rate is related to an overall decreased utilization of MFX techniques or not. Second, there was a significant difference in the regional distribution of the respondents, with 36% being from Europe and 27% being from Latin America. Therefore, this data set is most heavily influenced by these 2 regions. Furthermore, the retrospective nature of this study is certainly subject to recall bias. In addition, the availability of biologic therapies between regions is highly variable due to both regulatory and financial factors, thus implementation of certain techniques may be due to a number of other external factors rather than strictly surgeon preference.
In summary, the data of this survey demonstrate that the surgical indications, operative technique, and rehabilitation protocols utilized for patients undergoing microfracture procedures vary widely among different ICRS members. Regional differences appear to contribute to these practice pattern variations. In the future, consensus statements made by organizations such as the ICRS would likely lead to a more uniform management of these injuries based on best practice guidelines, knowing that, unfortunately, the economic health care challenges present in several countries may limit the implementation of non-microfracture-based cartilage restoration procedures.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Informed Consent
Not applicable.
Trial Registration
Not applicable.
