Abstract
Objective
The aim of this study is to evaluate the effects of intraarticular insulin on the treatment of chondral defects.
Design
Twenty-four mature New Zealand rabbits were randomly divided into 3 groups as control (Group 1), microfracture (Group 2), and microfracture and insulin (Group 3). Four-millimeter full-thickness cartilage defects were created to the weight-bearing surface on the medial femoral condyles of each rabbit. In the first group, any additional interventions were not performed. Microfracture was performed on defects in groups 2 and 3. Additionally, 10 IU of insulin glargine was administrated into the knee joints of the third group. Three months after surgery, the knee joints were harvested and cartilage quality was assessed according to Wakitani and ICRS (International Cartilage Repair Society) scores histopathologically. Insulin injections were performed into the knees of 2 additional rabbits without creating a cartilage defect to evaluate the potential adverse effects of insulin on healthy cartilage (Group 4).
Results
The total ICRS and Wakitani scores of the insulin group were found to be significantly lower than the microfracture group but similar to the control group. No negative effects of insulin on healthy cartilage were detected. Intraarticular insulin after surgery has led to a statistically significant decrease in systemic blood sugar levels whereas the decrease observed after administration to intact tissues was not statistically significant.
Conclusions
Insulin had a negative influence on the quality of cartilage regeneration and had no effect on healthy cartilage. Intraarticular insulin administration does not cause significant systemic effects in intact tissue.
Introduction
Focal full-thickness cartilage defects may cause pain and swelling, alter normal joint mechanics,1,2 and can lead to osteoarthrosis by causing degradation in the surrounding cartilage tissue.3,4 Although it is difficult to estimate the incidence and prevalence reliably, as many are asymptomatic, it has previously been observed that approximately 60% of patients undergoing arthroscopy had chondral lesions and 20% had focal chondral defects.5-7 The surgical treatment aims to relieve the symptoms, increase range of motion, and generate repair tissue, possibly similar to the original tissue histologically and functionally. To date, several treatment strategies have been developed to enhance the quality of repair tissue. Microfracture is one of the most common techniques based on the stimulation of subchondral bone ensued by the migration of pluripotent mesenchymal stem cells from the bone marrow into the defect area. This technique has been shown to generate fibrocartilage tissue that is mechanically weaker than hyaline cartilage.8,9 Despite the efficacy on symptom relief, the long-term results of microfracture technique are not promising.10-12
Additional studies are being conducted to determine whether the use of complementary biological therapeutics, alone or in conjunction with microfracture, can improve the quality of newly formed tissue.13-17 Insulin is an anabolic hormone, which has the potential to contribute to cartilage healing. The presence of insulin and IGF-1 receptors in healthy and osteoarthritic chondrocytes are known. 18 Insulin can act as a growth factor by binding to insulin receptors, IGF-1 receptors, and hybrid receptors in cartilage cells. It has been shown that insulin stimulates the transformation of cartilage precursor cells into chondroblasts and chondrocytes and increases protein and glycosaminoglycan synthesis as well as collagen 2 synthesis in cartilage tissue cultures.19-21 Previous research has established that insulin has potential as a treatment for osteoarthritis. 22
Despite several in vitro studies have confirmed anabolic effects of insulin on cartilage,19,21 this is the first in vivo study. The purpose of the current study was to evaluate the quality of regenerated tissue after microfracture with and without intraarticular insulin injection. We hypothesized that intraarticular insulin would improve treatment outcomes in a rabbit chondral defect model.
Methods
This study was performed on 26 mature New Zealand white rabbits weighing between 2,500 and 3,500 g. All the procedures were approved by Bezmialem Animal Care and Use Committee with approval number 13249 prior to performing the study.
Twenty-four rabbits were randomly allocated to 3 groups. In the control group (group 1), in which spontaneous healing was evaluated, only osteochondral defects (OCDs) were applied to the femur and not treated ( Fig. 1 ). In the second group (group 2), additional microfracture was performed to the cartilage defect. In the third group (group 3), intraarticular insulin, as well as microfracture, was applied to the formed cartilage defect. We created additionally the fourth group (group 4), consisting of 2 rabbits, to determine the possible negative effects of insulin on healthy cartilage tissue. In the fourth group, 10 IU insulin glargine (Lantus, Sanofi Aventis, Frankfurt, Germany) was administered intraarticularly for consecutive 3 weeks without surgical intervention on the knees of rabbits. Eight rabbits were assigned to the first 3 groups, while 2 rabbits were used for the fourth group.
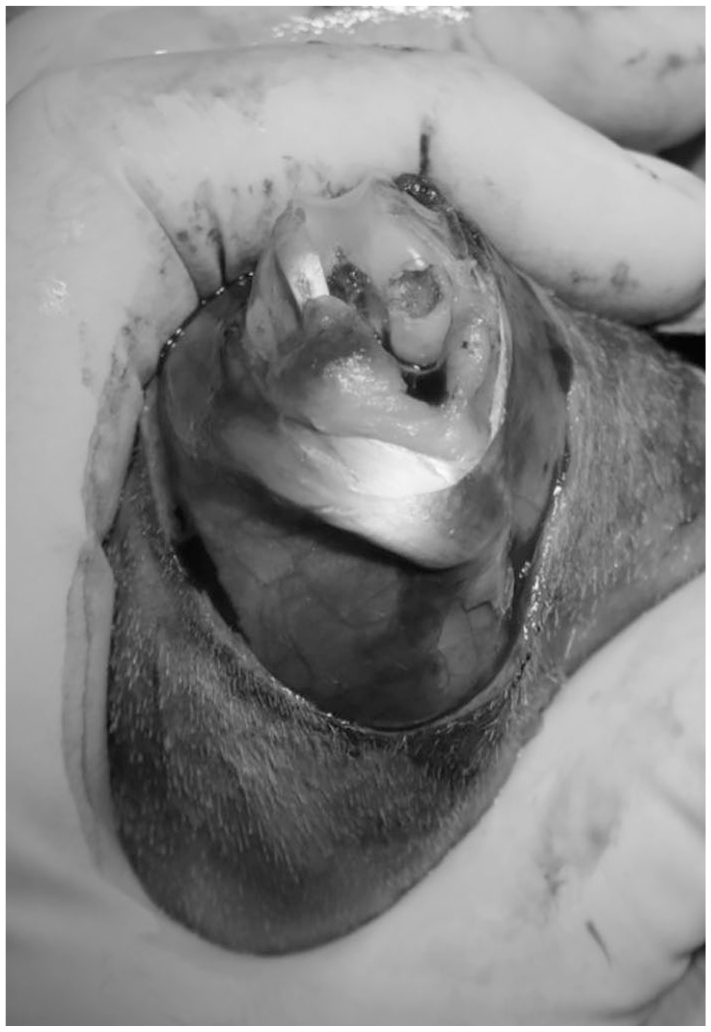
Osteochondral defect created in a rabbit medial femoral condyle.
Rabbits underwent intramuscular anesthesia and sedation with 2.5 to 10 mg/kg xylazine (Rompun, Bayer, Leverkusen, Germany) and 20 to 40 mg/kg ketamine (Ketalar, Zentiva, Kirklareli, Turkey). Preoperative antibiotic prophylaxis with 50 mg/kg cefazolin was performed. The right knee area of the animals was shaved, the area was cleaned with betadine, and after sterile coverage, an anterior midline incision was made. Medial parapatellar arthrotomy was performed, the patella was dislocated laterally, and the femoral condyle exposed. Two-millimeter-deep chondral defect was made without damaging the subchondral bone with a 4-mm dermal biopsy punch on the weight-bearing area of the medial femoral condyle ( Fig. 1 ). No additional intervention was performed to group 1. In the second and three groups, microfractures were created with 0.5-mm Kirschner wire at a depth of 2 mm from the periphery to the center of the defect without damaging the intact tissue between the holes. The capsule was then tightly closed with interrupted 3-0 vicryl sutures. After closing the skin incision with 3.0 prolene, 0.1 mL of physiologic saline (control injection) was injected to the knee of first and second groups and 10 IU insulin glargine was applied to the third group intraarticularly and repeated twice in 1-week intervals in the following period. Postoperatively, the movement of the knee joint was not restricted.
Blood glucose levels of rabbits in the intraarticular insulin injected group was monitored using a blood glucose meter (FreeStyle Optium H, Abbott, IL) before and 48 hours after administration.
Three months after surgery, rabbits were sacrificed according to institutional animal care and use committee protocol with high-dose sodium pentobarbital (>200 mg/kg) and knees were harvested from each animal for histological evaluation.
Histologic Evaluation
The acquired specimens of the distal femur were embedded in 10% formalin for 1 week and decalcified in 10% ethylenediaminetetraacetic acid (EDTA)-buffered saline solution. Then samples were embedded in paraffin blocks and cut into 4-µm sections. Following staining with hematoxylin-eosin, all sections were blindly scored by 2 pathologists according to ICRS (International Cartilage Repair Society) and Wakitani histology scoring systems.23,24
Statistical Analysis
The histological scores and blood glucose levels were analyzed using SPSS software (Version 21.0, IBM). Kolmogorov-Smirnov and Shapiro-Wilk tests administered to evaluate the normality of distributions and according to results total score of both histological classifications and blood glucose levels subjected to parametric tests.
One-way analysis of variance (ANOVA) was used for total scores of both histological classifications followed by Tukey post hoc test to perform the binary comparisons.
Nonparametric data were tested for significant differences by Kruskal-Wallis test and Mann-Whitney U test performed for pairwise group comparisons.
Blood glucose levels before and after insulin administration were tested for significant differences via the paired samples t test. Statistical significance was determined to be P < 0.05 for the 95% confidence interval.
Results
All rabbits completed the study and tolerated the procedures well. Any complications or adverse effects related to surgery or insulin injection were not observed.
Macroscopic evaluation of femoral condyles showed that the defect areas in all groups were filled with repair tissue, which is irregular and rough in varying proportions. In the second group, it was observed that the surface of the newly formed cartilage appeared to be more regular than in the other 2 groups ( Fig. 2 ).

Macroscopic samples of group 1 (
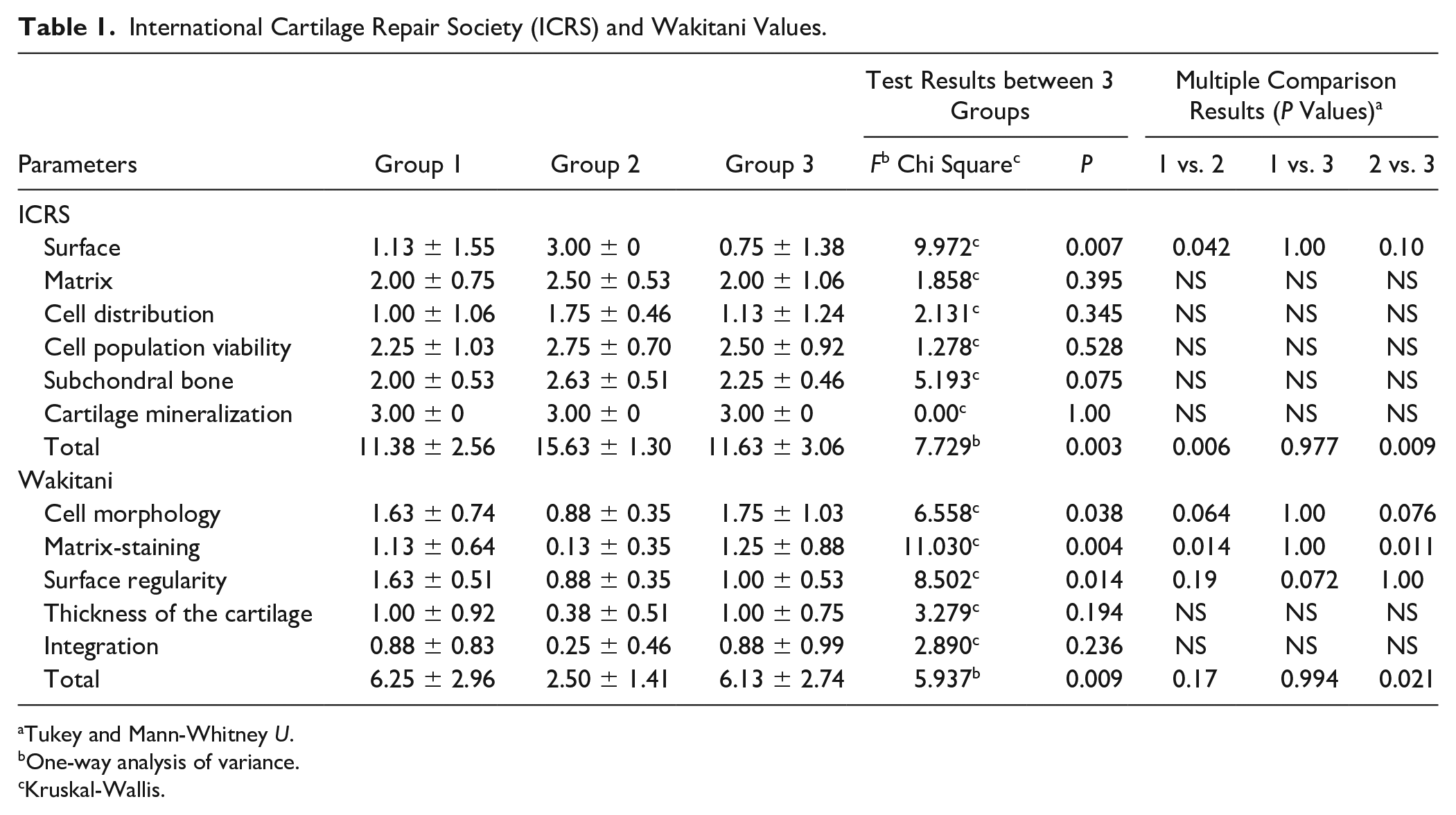
The results of histopathological evaluations according to ICRS and Wakitani scales are shown in Table 1 . What is interesting about the data in this table is that ICRS total scores of the third group in which insulin and microfracture were applied have very close values compared to the first group, which did not receive any treatment (P = 0.977). In contrast, the ICRS total score of the second group, to which only microfracture was applied, was found to be higher than the other 2 groups and the difference was found to be statistically significant (P = 0.006 and 0.009). Evaluating the parameters, the only parameter with a statistically significant difference between the groups according to the ICRS evaluation was the surface (P = 0.007). Treatment of insulin resulted in a mean surface score of 0.75 compared with 1.13 in control group and 3 in the microfracture group. When the groups were compared pairwise, the difference between the first and third groups was not significant, while the difference between the second and other 2 groups was statistically significant (P = 0.01 and 0.042).
International Cartilage Repair Society (ICRS) and Wakitani Values.
Tukey and Mann-Whitney U.
One-way analysis of variance.
Kruskal-Wallis.
Results of Wakitani scores were found to be similar to those obtained with ICRS scale. Insulin and microfracture applied group 3 had a slightly low total score compared to the control group, although the slight difference was not statistically significant (P = 0.994). The Wakitani total score of the second group was statistically significantly lower compared to the other 2 groups (P = 0.017 and 0.021). The Wakitani scores were statistically significant with respect to differences in surface regularity, cell morphology, and matrix staining between the groups ( Table 1 ).
The mean preoperative blood glucose levels of the third group who received insulin treatment were 197.8 ± 51.7 mg/dL, and the postoperative values were 57 ± 20.9 mg/dL, and the decrease was statistically significant (P < 0.05; Table 2 ). Despite the severe decrease in blood glucose levels, no clinical manifestation associated with hypoglycemia was observed. The mean blood glucose values of the same group before the second insulin administration were 120.1 ± 14.4 mg/dL, while the values measured after application were 113.6 ± 26.7 mg/dL and a decrease in blood glucose level of 7 mg/dL, which was not statistically significant (P > 0.05; Table 2 ). Based on the third insulin injection, no statistically significant difference was found among the results of glucose levels. The mean blood glucose level before the third insulin administration was 111.7 ± 10.1 mg/dL, while the mean after administration decreased by 4 mg/dL and was 107.0 ± 17.8 mg/dL ( Table 2 ).
Blood Glucose Levels.
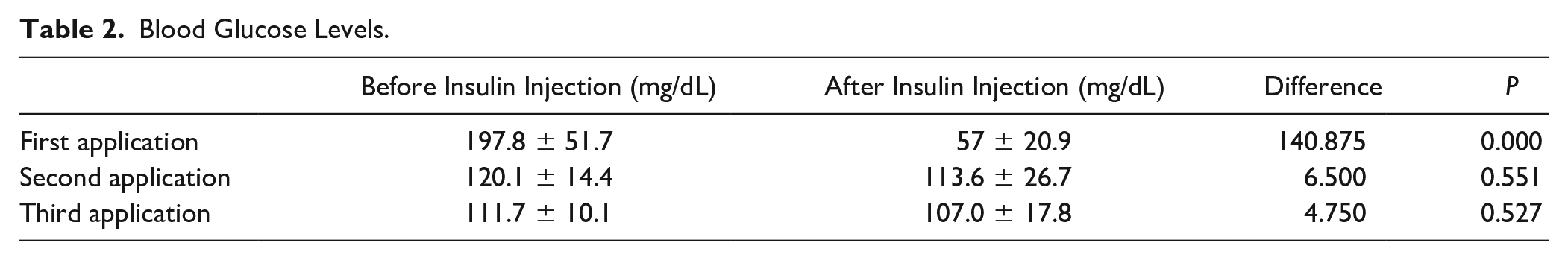
The blood glucose levels in group 4, which did not undergo surgery prior to insulin administration, were 112.5 ± 2.1 mg/dL, 122 ± 15.5 mg/dL, and 105 ± 7 mg/dL, respectively, after intraarticular insulin administration 75 ± 41 mg/dL, 97 ± 38 mg/dL, and 78.5 ± 16.2 mg/dL. As there were 2 rabbits in this group, the blood glucose values before and after insulin administration were not compared statistically. In this group, the cartilage tissue was completely normal and healthy after insulin application.
Discussion
Microfracture is a simple and cost-effective method in which mesenchymal stem cell migration and chondral transformation to the defect area by stimulation of the bone marrow are aimed. Although the symptoms are relieved with the formation of fibrocartilage tissue, alternative strategies for the regeneration of hyaline cartilage are required due to the insufficient long-term results. The aim of this study was to evaluate the effect of microfracture treatment and additional intraarticular insulin administration on the treatment of full-thickness cartilage defects in healthy rabbits. Our hypothesis was that insulin would improve the quality of the newly formed regenerate.
Local insulin application has been previously investigated in animal fracture models, bone defects, spinal fusion, and osteoporotic models.25-29 In the study of lower extremity fracture models in nondiabetic rats, insulin was reported as a potential therapeutic agent for fracture union. It was shown that locally applied insulin has an accelerating effect on fracture union and the newly formed bone tissue is biomechanically stronger. 25 The same study also demonstrated that insulin increases the number of mesenchymal cells and their transformation to chondrocytes in the early stages of enchondral ossification. To date, the effect of insulin on cartilage tissue by intraarticular administration has not been evaluated.
Insulin glargine (Lantus/Sanofi) with the replacement of 3 amino acids of insulin and the addition of zinc was chosen for its subcutaneous activity, which lasts up to 26 hours without a peak. As the weekly administration of intraarticular injections can be tolerated by the patients, 3 injections were given at intervals of 1 week. The weakness of this method is that intraarticular insulin levels were not evaluated due to application problems. In a study investigating the effect of local insulin on a fracture model by Paglia et al. following application of 10 units of Humulin U into the medullary cavity, a significantly higher amount of insulin was found in the first 12 to 48 hours in the insulin-treated femur than on the contralateral side, whereas the local insulin levels were almost equal by 96 hours. 26 In light of the studies to date, it would be appropriate in further studies to reduce the intervals of insulin administration to 4 days or to use slow-release forms.
To evaluate whether insulin was systemically released, systemic blood glucose levels in rabbits with local insulin delivery were monitored 48 hours before and after administration. The lowest blood glucose values were determined after application by the fourth hour and by comparison with the values before application, the possible systemic effects of insulin were determined. It was found that postoperative insulin application caused a significant reduction in systemic glucose levels, but there was no significant difference in the 2 subsequent applications. This preliminary finding suggests that the trauma caused by surgery facilitates the transition of insulin into the systemic circulation, and the systemic effects of intraarticular insulin administration in nontraumatic tissue are quite small. This finding is consistent with Park et al., who found that locally administered insulin has no systemic effect in the fracture model in rats treated with insulin-absorbed calcium sulfate. 25 In contrast, these results differ from the findings presented by Paglia et al., which suggests the mean systemic blood glucose levels decreased from 95 to 67 mg/dL by the 12th hour after application of humulin U into the intramedullary cavity of the rat femur in a study investigating the effect of local insulin on the fracture model. 26
The assessment of cartilage regeneration with ICRS and Wakitani histological cartilage scoring systems has demonstrated better outcomes in the microfracture group in accordance with the previous studies.30,31 Surprisingly, no significant difference was found between ICRS and Wakitani scores of group 3 rabbits treated with insulin additional to microfracture and group 1 rabbits, which did not receive any treatment. We found this result significant because this unexpected result provides a negative relation between insulin and chondral transformation and it can be interpreted that insulin may have inhibited the chondral transformation of mesenchymal stem cells.
Analyzing the literature we could not find any studies that examines the effects of insulin on chondral cartilage repair in vivo. On the other hand, we reached a few in vitro studies that differ from our findings. Prior studies have shown that insulin stimulates the chondrogenic conversion in mesenchymal progenitor cell culture in a dose-dependent manner. 21 In addition, insulin has been shown to have anabolic effects on chondrocytes by increasing proteoglycan synthesis and reducing the effect of IL-1. 22 It can, therefore, be assumed that the negative effect of insulin on cartilage tissue development obtained in our study may be due to dose, insulin formation, or lack of other growth factors that should be present in the environment.
Despite the absence of previous in vivo studies of the application of insulin to cartilage tissue, it has been shown in the fracture model in rats that the formation of enchondral cartilage was lower in the group receiving low doses of insulin than in the control group, while chondral transformation was increased in those receiving high doses of insulin. 25 This finding is consistent with that of Mueller et al., who found in mesenchymal stem cell culture that the chondrogenic differentiation effect of insulin results from direct or indirect stimulation of the IGF-1 receptor. 21 Since it is known that insulin only binds to IGF-1 receptors in high doses, it seems possible that our results could be due to an insufficiency of the intraarticular insulin dose. On the other hand, according to the in vitro study conducted by Ribeiro et al. in 2015, it was stated that high insulin concentration had catabolic effects on chondrocytes. 32 Also, high doses of insulin and IGF-1 cause insulin-like growth factor receptor downregulation. 33 It is therefore likely that insulin, given as bolus to the medium, may also cause IGF-1R downregulation in mesenchymal stem cells, reducing or even inhibiting differentiation. In the animal experimental study evaluating the effect of insulin on the tibial growth epiphysis, it was found that insulin caused an increase in the growth plate width and this effect was increased by increasing the amount of IGF-1 in the environment. 34 In the same study, it was observed that the effect of insulin reached a maximum in a certain dose range and then decreased its effect. With regard to previous studies, it can be assumed that insulin may have a safe dose range and may damage healing tissue concentration-related when the dose is incorrect.
In the study conducted by Petrou et al. in the culture of MSC, it was found that IGF-1 alone does not have any effect on chondrogenic differentiation, whereas it increased chondral differentiation in the presence of TGFβ-1. 35 In our study, it should be taken into consideration that the reason for the poor histological scores of the insulin group may also be due to the lack of growth factors.
It was observed that the cartilaginous tissues of group 4 rabbits, which are intraarticular insulin administered without surgery, were macroscopically and histologically normal. These findings suggest that intraarticular insulin administration had no short-term adverse effects on healthy cartilage tissue.
As an experimental chondral defect model, a 2 mm deep defect in the femoral-medial condyle was created with a 4 mm dermal punch biopsy, taking the previous studies as reference. The biggest problem of these models is not being able to create standard devices to adjust the depth of the defect. Because the most important factors affecting cartilage healing are the size and depth of the defect. This problem was tried to be overcome by performing the surgery of all animals by the same surgeon.
This study has several limitations. First, the injection of insulin into the joint as a bolus may prevent the desired effect. Slow-release test models can provide a more stable intraarticular concentration and may lead to different results. Future studies could address this issue by incorporating the use of drug-delivery systems, which could release insulin in a more controlled manner. Second, the study conducted with a single dose of insulin can be tried at different intervals. Since there are no similar studies in the literature, the relationship between dose and effect is not clear. In addition, due to technical inadequacies, we applied hematoxylin and eosin staining that would only allow us to evaluate the general tissue architecture. If we could stain the samples with Safranin-O, we could detect the proteoglycan content of the healing tissue and perform a more detailed examination.
Conclusions
To date, there are no other in vivo studies in the literature evaluating the effect of insulin on cartilage transformation and proliferation. One of the issues that emerges from these findings is that intraarticular insulin administration does not cause significant systemic effects in intact tissues. We also found that intraarticular insulin administration did not have a negative impact on healthy cartilage tissue in the short term. In our study, the result that insulin had a negative effect on the quality of cartilage regenerate shows that insulin affects the healing process of cartilage tissue. Regarding the positive effects of insulin at the molecular basis and our in vivo results, repetition of the study with slow-release insulin formulations and different dose ranges is inevitable. Further studies with combinations of growth factors need to be conducted.
Footnotes
Authors’ Note
Place of work: Experimental Application and Research Center, Bezmialem University, Istanbul, Turkey.
Acknowledgements and Funding
The authors thank the contributions of Andac Salman in the statistical part of this article and also Ulkan Kılıc for the veterinary support. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Istanbul University Scientific Research Projects Department (Grant Number 39484).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All the procedures were approved by Bezmialem Animal Care and Use Committee with Approval Number 13249 prior to performing the study.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
