Abstract
Objective
Evaluation of collagen orientation and arrangement in articular cartilage can improve our understanding of primary osteoarthritis (OA) progression and targeted therapies. Our goal was to determine if polarized light microscopy (PLM) for collagen organization is useful in identifying early primary OA features in comparison to current standard histopathological methods.
Design
Osteochondral specimens from 90 total knee arthroplasty patients with relatively preserved lateral femoral condyle were scored using (1) histological-histochemical grading system (HHGS); (2) Osteoarthritis Research Society International (OARSI); (3) PLM-Changoor system for repair cartilage, scores ranging between 0 (totally disorganized cartilage) and 5 (healthy adult cartilage); and (4) new PLM system for primary OA cartilage with superficial zone PLM (PLM-SZ) and deep zone PLM (PLM-DZ) scores, each ranging between 0 (healthy adult SZ and DZ collagen organization) and 4 (total loss of collagen organization). Serial sections were stained for collagen I and II antibodies. Spearman correlation coefficients (rs) were determined.
Results
The associations between: (1) PLM-Changoor and HHGS or OARSI were weak (rs = −0.36) or moderate (rs = −0.56); (2) PLM-SZ and HHGS or OARSI were moderate (rs = 0.46 or rs = 0.53); and (3) PLM-DZ and HHGS or OARSI were poor (rs = 0.31 or rs = 0.21), respectively. Specimens exhibiting early and mild OA (HHGS < 5 and OARSI < 8.6) had PLM-SZ and PLM-DZ scores between 0 and 4 and between 0 and 3, respectively, and indicated new histopathological features not currently considered by HHGS/OARSI.
Conclusions
PLM was effective at identifying early SZ and DZ collagen alterations that were not evident in the traditional scoring systems. Incorporating PLM scores and/or additional HHGS/OARSI features can help improve characterization of early primary OA cartilage.
Introduction
Primary osteoarthritis (OA) can be attributed to a number of etiologies that result in varying biological responses that alter structure and function of the joint leading to a similar final pathway of joint degeneration. Overall clinical symptoms of pain, joint instability, and impaired quality of life, during the final late stages of OA warrant treatment with joint replacement. 1 Nonetheless, there is insufficient knowledge and understanding of OA disease progression, particularly during the early phases of OA, which are critical to enable development of therapeutics and treatment procedures at the first-stages of the disease rather than end-stage. 2
Histopathological assessment provides a view of the spectrum of features exhibited by cartilage at various stages of OA progression and is a common method to assess the in situ state of cartilage tissue.3,4 Currently, the associations between histopathological grade, severity of the disease and clinical correlation are weak. 5 The 2 most commonly used histopathology scoring systems for grading primary OA cartilage degeneration are histological-histochemical grading system (HHGS) 6 and Osteoarthritis Research Society International (OARSI). 7 HHGS scores 4 histopathological features, including structural integrity, cell organization, proteoglycan loss, and tidemark integrity as observed in hematoxylin and eosin (HE) and safranin O–fast green (SafO-FG) stained sections of cartilage. 8 OARSI scores combine several histopathological features primarily related to structure degradation under a grade along with a stage factor to represent the spatial extent of the disease. Both these scoring systems are heavily weighted on structural integrity (fissures, crevices, and delamination), which is often a feature of late pathological stage. Prior to the earliest histological change of fibrillation of the superficial zone of articular cartilage, abnormalities are seen within the extracellular matrix (ECM).3,9-11 Degradation of aggrecan and collagen II molecules are some of the early pathological changes that alters chondrocytes activity and mechanical properties at the surface and subchondral bone, eventually leading to irreversible structural damage. To identify early OA pathological features, we need new criteria and assessment methods that focus on detecting these ECM abnormalities.
The accepted gold standard for evaluating collagen fibril organization in cartilage tissue sections has been polarized light microscopy (PLM).12,13 This technique exploits the optical anisotropic properties of fibrillar collagens, allowing inference on collagen orientation and packing density from the presence of birefringent/nonbirefringent regions.14,15 Birefringence occurs from changes to the direction of polarization of light passing through histological sections, due to the orientation, arrangement, packing density, and diameter of collagen fibrils at each point in the section.16-18 Unstained cartilage sections have shown to be more reliable and suitable for PLM than picrosirius red (PSR)–stained sections.13,19
In healthy adult hyaline cartilage, tissue sections imaged with PLM exhibit a thin, uniform layer of birefringence at the articular surface due to parallel alignment of collagen I in the superficial zone (SZ).15,19,20 The random arrangement of collagen II fibrils in the transitional zone (TZ) lead to no birefringence in this zone.15,19,20 The presence of orthogonally arranged thin collagen II fibrils (30-200 nm) in the deep zone (DZ) results in low birefringence, with increasing intensity near the tidemark area due to thicker collagen II fibrils and increasing amount of collagen II fibrils.21-24 Primary OA progression leads to disorganization or degeneration of collagen ECM that can be detected by changes in patterns of birefringence and used as indicators of early and mild OA.25 -27 PLM can be a useful technology to assess collagen architecture within the ECM. 21 Changoor et al. developed a scale for scoring PLM images of repair cartilage (referred here onward as PLM-Changoor). 13 Evaluation of cartilage collagen orientation and arrangement, which is strongly associated with ECM degradation and subchondral bone (SCB) changes in primary OA, may significantly add to our understanding of disease progression and targeted therapies. Given that the original PLM scoring system was developed to analyze repair cartilage, 2 new PLM scoring systems specific to the SZ and DZ (referred here onward as PLM-SZ and PLM-DZ, respectively) for primary OA cartilage were developed and used in this study.
To incorporate new criteria and methods for histopathological assessment, it will be useful to understand how these new PLM scoring techniques compare to existing methods and what the observations imply. Therefore, the primary goal of this study is to correlate OA disease state as assessed by HHGS or OARSI scores with PLM-Changoor, PLM-SZ, and PLM-DZ scores. These assessments may guide whether PLM should be added to routine histopathological OA scoring systems and/or if PLM features can help reveal identification of new HE or SafO features to characterize the disease state of primary early OA specimens. Additionally, it may improve our current understanding of idiopathic OA and potentially recognize different subgroups of disease and enable a better correlation of histopathological features with clinical outcome measurements, including imaging (e.g., magnetic resonance imaging) and patient-reported outcome measures (PROMs).
Methods
This study was approved by the Institutional Review Board committee of the Cleveland Clinic (Protocol: 13641). A total of 90 patients (female = 45) with mean age of 62.4 years (range, 37-84 years), and a mean body mass index of 31.8 kg/m2 (range, 18.1-51.4 kg/m2) with varus knees scheduled for total knee arthroplasty (TKA) were recruited. Inclusion criteria were: idiopathic OA (primarily medial compartment and/or patellofemoral disease) exhibiting a relatively preserved lateral compartment (joint space width [JSW]: 0-10 mm, mean 5.8 mm) based on preoperative weightbearing, anterior-posterior (AP) radiographs taken in full extension and 30° of fixed flexion. Exclusion criteria were: secondary arthritis related to systemic inflammatory arthritis; history of autoimmune disorders, gout or pseudogout, previous surgery to the index knee, current or previous treatment with systemic glucocorticoids or osteotropic medication; cancer within previous 2 years; known or suspected infection; and osteonecrosis.
Human Cartilage Procurement and Processing
The lateral femoral condyle (LFC) was collected after making the distal femoral cut during TKA, and the anterior-posterior orientation was noted ( Fig. 1 ). Osteochondral specimens (4 × 4 × 8 mm) were obtained by placing the condyle in an in-house fabricated miter box in anterior-posterior orientation ( Fig. 1 B and C ). Immediately after surgical retrieval, specimens were collected in 10% neutral buffered formalin containing 0.5% cetylpyridium chloride (preserves proteoglycan and hyaluronan contents in cartilage tissues) 28 and were fixed for 48 hours at 4°C. They were subsequently decalcified for 5 weeks at 4°C using Cal-rite (ThermoScientific, Waltham, MA). The decalcified tissue was dehydrated in an alcohol series into a xylene-substitute and embedded in paraffin with a consistent spatial orientation. The embedded tissue was cut in the plane perpendicular to the surface of the cartilage to obtain a representative overview of the tissue structure and thickness. Five-micrometer thick paraffin sections were cut to obtain unstained (UNS), HE-, SafO-FG-, and PSR)-stained sections. 29 For unstained PLM imaging, sections were deparaffinized using Clear-rite (xylene-substitute), then rehydrated in an alcohol series followed with water. The samples were then cover slipped with Cytoseal XYL mounting media. Stained sections were digitally imaged on a calibrated Leica microscope under bright field (BF).

Lateral femoral condyles (LFC) were obtained from varus knee total knee arthroplasty patients for assessment in the study. (
Semiquantitative Histology Scoring
All the specimens were scored using HHGS and OARSI scoring systems by 3 expert reviewers.3,4,6,7
Semiquantitative PLM-Changoor scoring system developed for repair cartilage was used to assess collagen organization in our specimen cohort ( Table 1 ). 13 The scores range between 0 and 5, with lower score indicating degrading cartilage; a score of 0 describes cartilage specimens that have sparse patches of birefringence that are neither parallel nor perpendicularly orientated indicating disorganized cartilage. A score of 5 describes normal, adult hyaline cartilage, where a distinct and smooth SZ and DZ birefringence is apparent, showing parallel and perpendicularly aligned collagen, respectively.
Description of Polarized Light Microscopy (PLM) Scoring System by Changoor Group for Repair Cartilage. 13

DZ = deep zone; TZ = transitional zone; SZ = superficial zone.
To assess the collagen organization features that are important and appropriate for primary OA cartilage, we developed a new qualitative scoring system ( Table 2 ). Considering the fact that birefringence in adult human cartilage pertains mostly to the SZ and DZ of cartilage (TZ comprises of randomly organized collagen and thus the birefringence is null), a PLM-SZ score and PLM-DZ score were assigned for each specimen.
Description of a New Polarized Light Microscopy (PLM) Scoring System for Primary Osteoarthritis Cartilage.
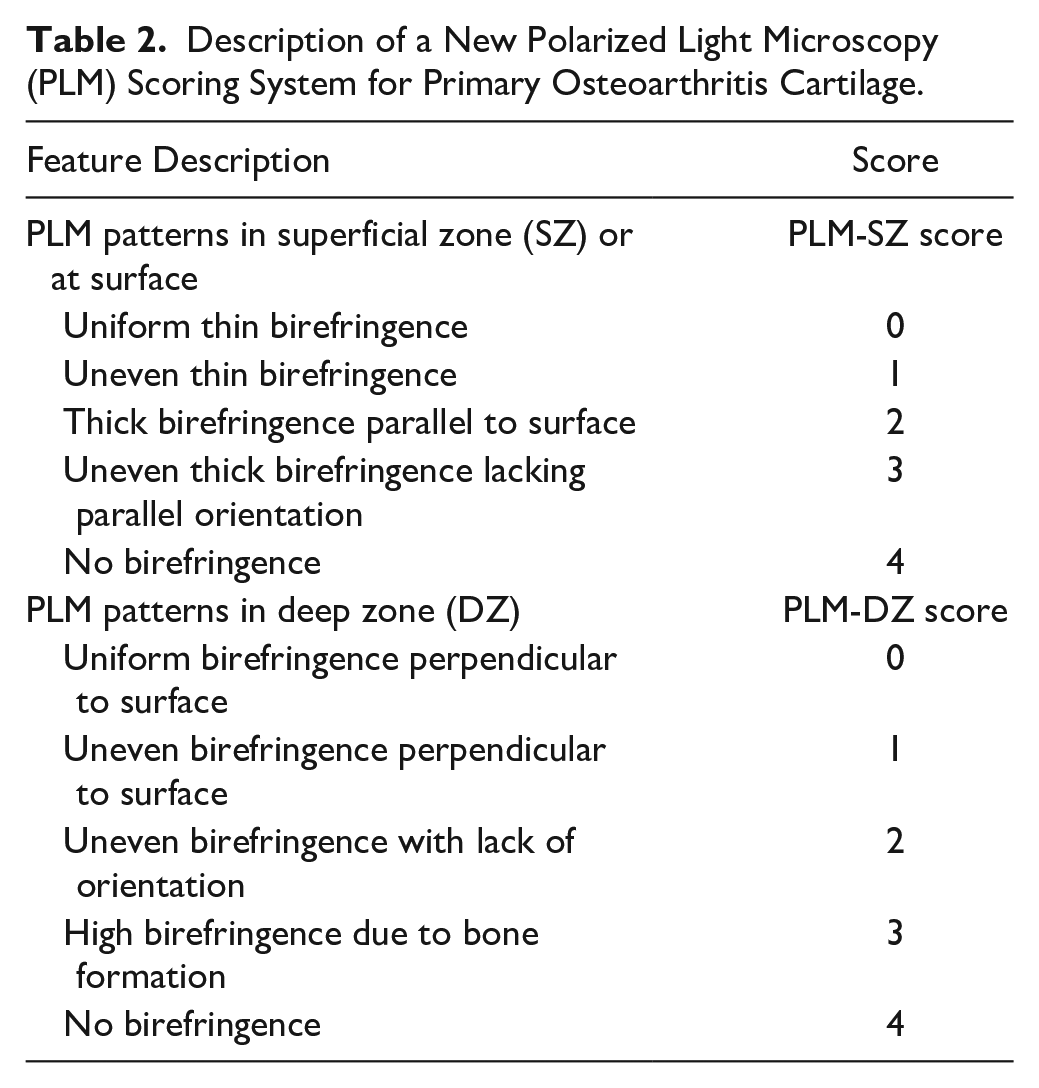
PLM-SZ score ranges between 0 and 4, with higher scores indicating degrading SZ of cartilage. A uniform thin bright birefringence in the SZ indicates healthy adult SZ and receives a PLM-SZ score of 0. As the surface starts to degrade, its uniformity is lost resulting in uneven thin birefringence parallel to the surface (PLM-SZ score: 1). Further SZ cartilage changes may result in thicker birefringence at the SZ (PLM-SZ score: 2). Surface degradation can result in crevices in the surface that show uneven thick birefringence lacking parallel orientation (PLM-SZ score: 3). Complete loss of superficial zone will result in no birefringence at the SZ and receive a PLM-SZ score of 4.15,20,21,30
PLM-DZ scores range between 0 and 4, with higher score indicating degrading DZ of cartilage. A uniform birefringence signal aligned perpendicular to the surface in the DZ indicates healthy adult DZ and receives a PLM-DZ score of 0. In healthy cartilage specimens, it should be noted that the birefringence signal intensity close to the tidemark region may be brighter than in the mid-DZ as the collagen fibers are thicker. Uneven birefringence perpendicular to the surface receives a PLM-DZ score of 1. Further DZ cartilage changes can result in uneven birefringence with lack of orientation (PLM-DZ score: 2). Eruption of blood vessels through the tidemark region can result in formation of bone-like structures in the hyaline cartilage region, which results in high birefringence areas and they receive a PLM-DZ score of 3. No birefringence in the DZ receive a PLM-DZ score of 4.15,20,21,30
New HE/SafO patterns for Association with PLM
To identify new HE/SafO-based histopathological features that were not considered in HHGS and OARSI scoring systems and are potential features that can be associated with early or mild OA pathogenesis and PLM patterns, we identified specimens with HHGS structure score = 0 or 1, but displayed SafO matrix degradation patterns, including (1) partial loss of SafO stain in the interterritorial matrix region that is not associated with obvious signs of surface erosion, (2) loss of SafO stain in the interterritorial matrix, (3) loss of SafO stain in the pericellular matrix region, and (4) loss of SafO stain in the DZ near tidemark, or tidemark changes, including (1) multiple tidemarks, (2) multiple tidemarks that are breached by multiple blood vessels, (3) unspecified tissue composition deposited near the tidemark, and (4) appearance of cartilage-bone-cartilage-bone interleaved tissue layers accompanied by multiple tidemarks or both, as reported in our previous study. 3 Cartilage specimens that exhibited conventional OA progression features by surface degradation (HHGS structure score: 2-4) were included for comparison as well.
Immunostaining with Collagen I and II
Paraffin-embedded sections (5 µm) were dewaxed through xylene and a graded alcohol series. Sections were rinsed three times in phosphate buffered saline (PBS) for 5 minutes each. Endogenous peroxidase activity was quenched by incubation with 3% (v/v) H2O2 for 20 minutes. Tissue sections were washed in PBS for 2 minutes (3 times), blocked with 10% (w/v) bovine serum albumin (BSA) in PBS for 1 hour in a humidified chamber, and then blotted to remove excess BSA. Tissue sections were then incubated for 1 hour with a primary monoclonal antibody against collagen type I (1:100 dilution in blocking solution; Abcam ab21287) or collagen type II (1:100 dilution; Abcam ab34712) and then washed in PBS (3 times) for 2 minutes each. Tissue sections were then incubated with biotinylated goat anti-rabbit IgG (H + L) (1:1000 dilution; Vector Laboratories), for 1 hour in a humidified chamber at room temperature and were then washed 3 times for 2 minutes in PBS. Tissue sections were then incubated in an avidin-conjugated peroxidase complex solution (Vector Laboratories BA-1000) for 1 hour in a humidified chamber at room temperature followed by 3 PBS washes (2 minutes each). Finally, the sections were incubated in diaminobenzedine (DAB) substrate solution (Vector Laboratories H-2200) in a dark chamber for 10 minutes, and rinsed in PBS 3 times for 2 minutes each. Last, DAB enhancer (Vector Laboratories) was added for 20 seconds and washed with PBS; sections were then mounted in xylene-based mounting media and cover-slipped. Sections with absence of primary antibody were used as controls.
Microscopic Methods
BF images of the HE, SafO-FG, PSR, collagen I (Col I-BF), and collagen II (Col II-BF) stained sections were obtained using color QICam camera (QImaging, Surrey, British Columbia, Canada) at 4× and 10× magnification mounted on Leica DM6000 microscope (Leica Microsystems, Wetzlar, Germany). Unstained sections (PLM), and PSR-stained sections (PSR-PLM) were also imaged using PLM (Leitz Ortholux II POL, Leitz, Wetzlar, Germany) mounted on a conventional light microscope (Leica DM4000 B LED). PSR-PLM system consisted of an analyzer (positioned in the light path between the light source and specimen), polarizer (positioned in the light path between the specimen and camera), and a CCD camera (Leica DFC 7000T on C-mount) at 4× and 10× magnification. The analyzer was inserted into the optical path and the polarizer was rotated to 270° with respect to the transmission azimuth, so as to maximize the birefringence signal from collagen fibrils, while keeping the background at minimum light signal intensity.
Statistical Analysis
Spearman’s correlation coefficients were estimated to assess the associations between all histopathological scores: HHGS, OARSI, PLM-Changoor, PLM-SZ and PLM-DZ.
Results
Correlation between HHGS, OARSI, and PLM
Spearman’s correlation coefficient between HHGS and OARSI scores was strong (rs = 0.75, P < 0.001), between HHGS and PLM scores was weak (rs = 0.36, P < 0.001) and between OARSI and PLM scores was moderate (rs = 0.56, P < 0.001) ( Fig. 2 ).
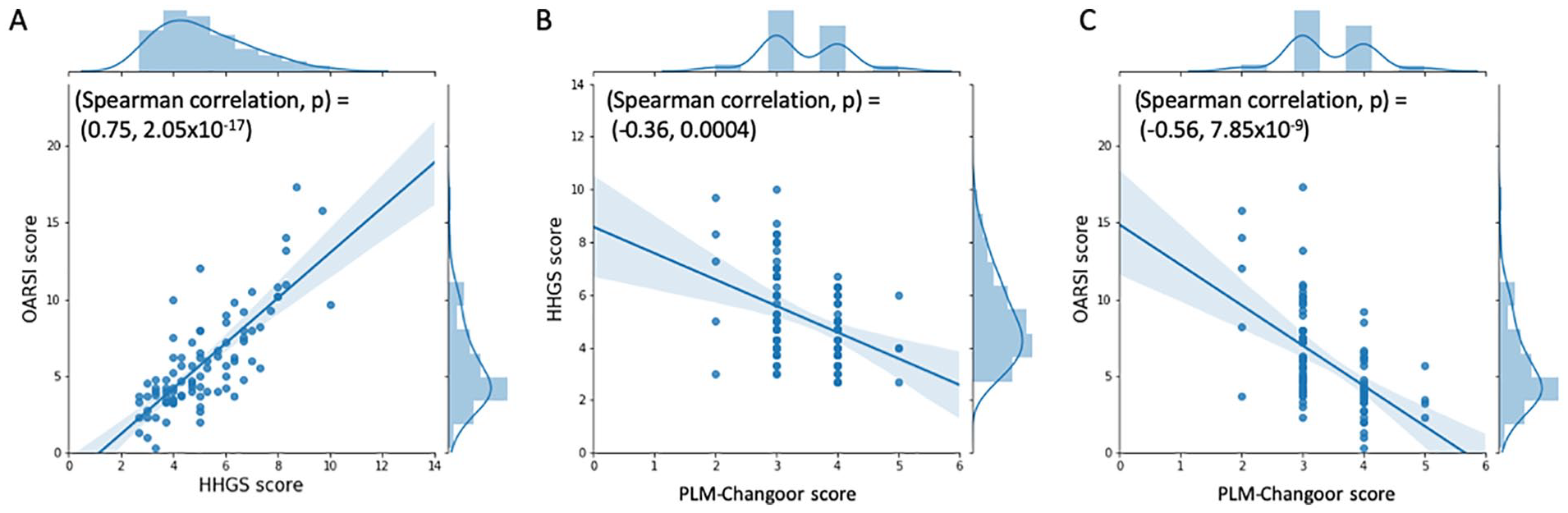
Joint plots showing distribution of traditional histopathological scores (HHGS, OARSI) and polarized light microscopy (PLM) score for repair cartilage (PLM-Changoor) for 90 osteochondral specimens obtained from lateral femoral condyle. (
Spearman’s correlation coefficient between HHGS or OARSI and PLM-SZ scores were moderate (rs = 0.46, P < 0.001 or rs = 0.53, P < 0.001, respectively) ( Fig. 3A and B ). However, Spearman’s correlation coefficient between the HHGS or OARSI and PLM-DZ scores were poor (rs = 0.31, P = 0.003 or rs = 0.21, P = 0.04, respectively) ( Fig. 3C and D ).
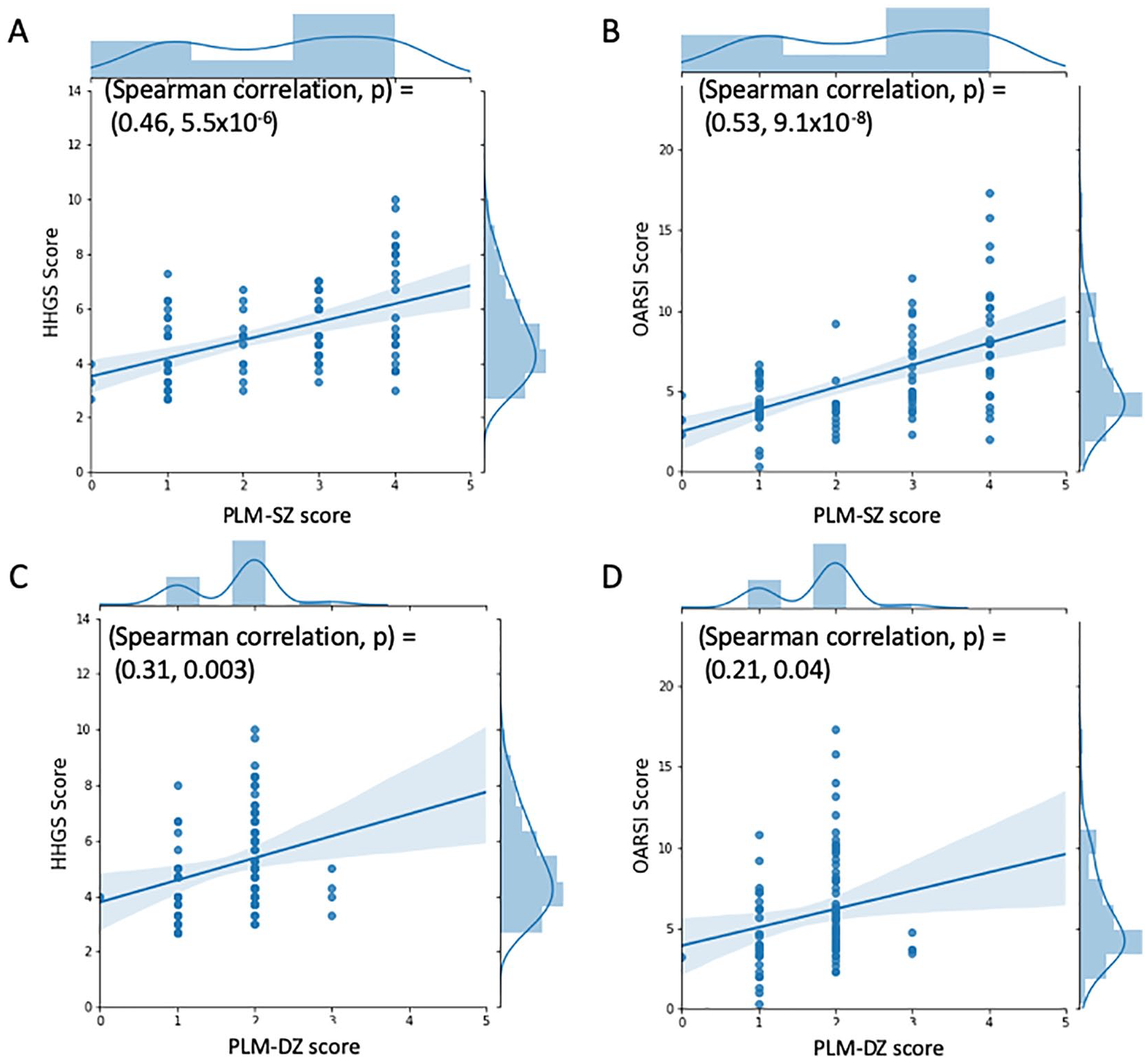
Joint plots showing distribution of traditional histopathological scores (HHGS, OARSI) and polarized light microscopy (PLM) score for primary osteoarthritis cartilage (PLM-SZ and PLM-DZ) for 90 osteochondral specimens obtained from lateral femoral condyle. (
Conventional OA Progression Specimens (Cartilage Degradation Progressing from Surface Down)
Cartilage specimens from 40 out of the 90-patient cohort showed OA progression with surface degradation (44% of the specimens). Figure 4 shows representative histology sections where cartilage degradation initiates at the surface with irregularities ( Fig. 4B1 and C1 ), progresses to development of shallow crevices ( Fig. 4 D1 ), and then deep clefts ( Fig. 4 E1 ). HE-based ( Fig. 4A1-E1 ) and SafO-FG-based ( Fig. 4A2-E2 ) HHGS and OARSI scoring systems, which are heavily weighted on the structural deformation, assessed increasing grade ranging from 2 to 10 for HHGS and from 0 to 17 for OARSI ( Fig. 4A-E ).3,4

Representative human cartilage specimens obtained from lateral femoral condyle displaying traditional human osteoarthritis (OA) progression features (OA degradation progressing from surface down) as seen in hematoxylin and eosin (HE) (column 1:
The corresponding unstained sections when viewed under PLM (
Fig. 4A3-E3
) and immunostained for Col II (
Fig. 4A4-E4
) and Col I (
Fig. 4A5-E5
) showed patterns that correlated with HE (
Fig.4A1-E1
) and SafO-FG (
Fig. 4A2-E2
) observations: PLM-imaged cartilage showed an increase in birefringence zone thickness at the cartilage surface as degradation increased (
Fig. 5
, left panel top: UNS-PLM:
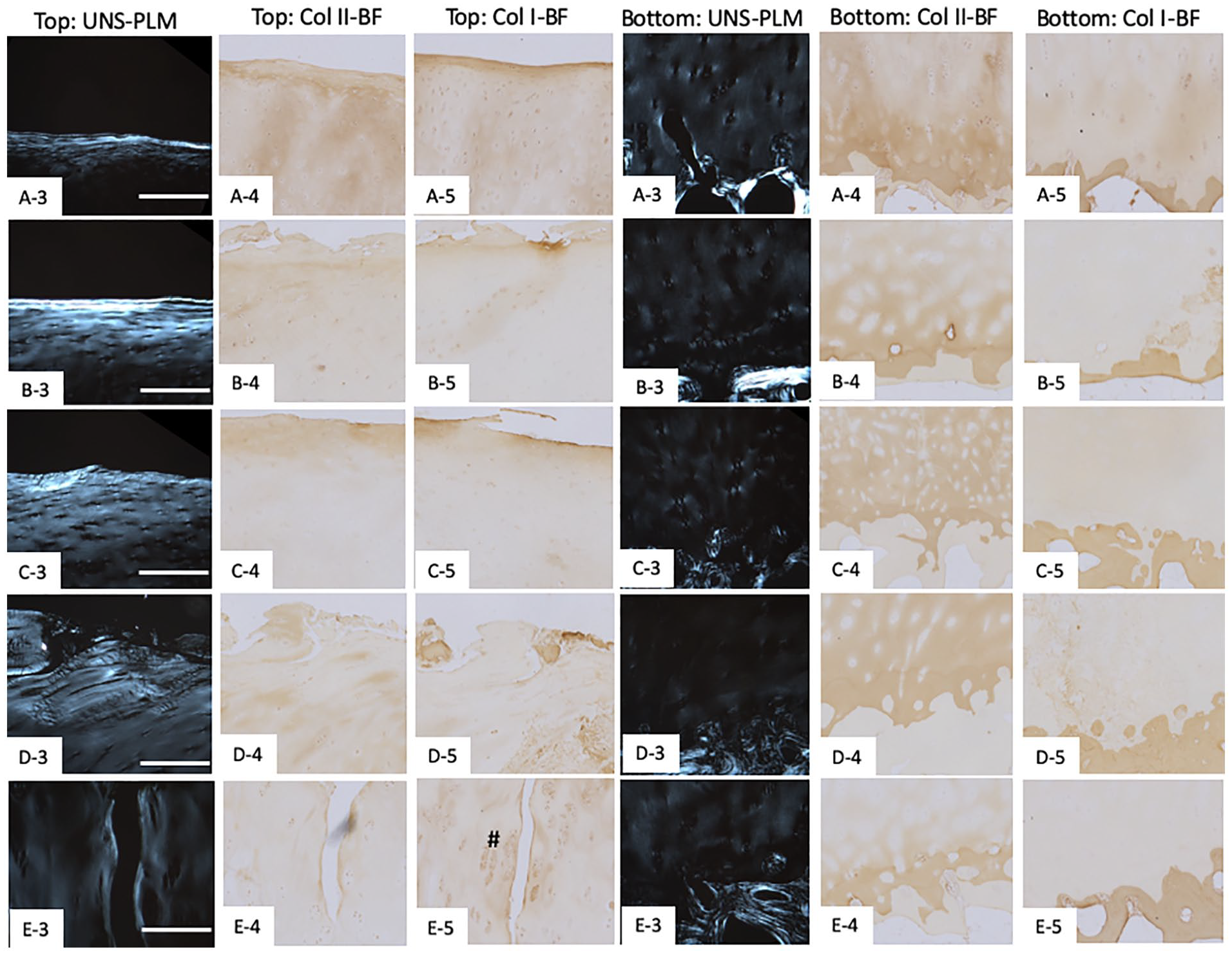
Higher magnification (10×) of the boxed regions shown in Figure 4 to illustrate the osteoarthritic features in detail. Box regions near the superficial zone (top region) for unstained polarized light microscopy, collagen II, and collagen I cartilage sections are shown in columns 1, 2, and 3. Box regions near the deep zone (bottom region) for unstained polarized light microscopy, collagen II, and collagen I cartilage sections are shown in columns 4, 5, and 6. Scale bar represents 200 µm. Note: # indicates Col I-stained regions near the crevice.
Overall, samples exhibiting surface related modifications (surface irregularities, clefts to transitional and deep zone) either HE or SafO-FG stained compared favorably with PLM. However, the histopathological changes occurring in the mid-deep zone of the cartilage were not easily identified using HE and SafO-FG staining and PLM provided more discernable changes with respect to matrix remodeling and tidemark related changes ( Fig. 4 ).
OA Cartilage Specimens with Normal Surface Structure Exhibiting Slight SafO Stain Loss
Cartilage specimens from 37 out of the 90-patient cohort (41% of the specimens) showed OA progression with little to no surface fissures in the cartilage (HHGS structure score = 0 or 1) and showed slight loss in SafO staining (HHGS SafO-FG staining score = 1) ( Fig. 6 ). As per the HHGS scoring system, SafO-FG scores are based on the stained area in the entire cartilage section being assessed and does not consider select regional differences in the SafO staining tissue patterns. These regional staining patterns include interterritorial matrix SafO staining loss ( Fig. 6A2 ), SafO staining loss near the tidemark ( Fig. 6B2 ), localized-matrix degradation ( Fig. 6C2 ), and territorial matrix SafO loss ( Fig. 6D2 ). Thus, for many of the samples included in this study the overall HHGS SafO scores were assessed as slight reductions in SafO staining yet, on closer inspection at higher magnification, there were noticeable differences in SafO staining patterns ( Fig. 7A2-D2 ).

Representative human cartilage specimens displaying normal cartilage structure but exhibiting varying patterns of slight safranin-O stain loss as seen under hematoxylin and eosin (HE) (column 1:
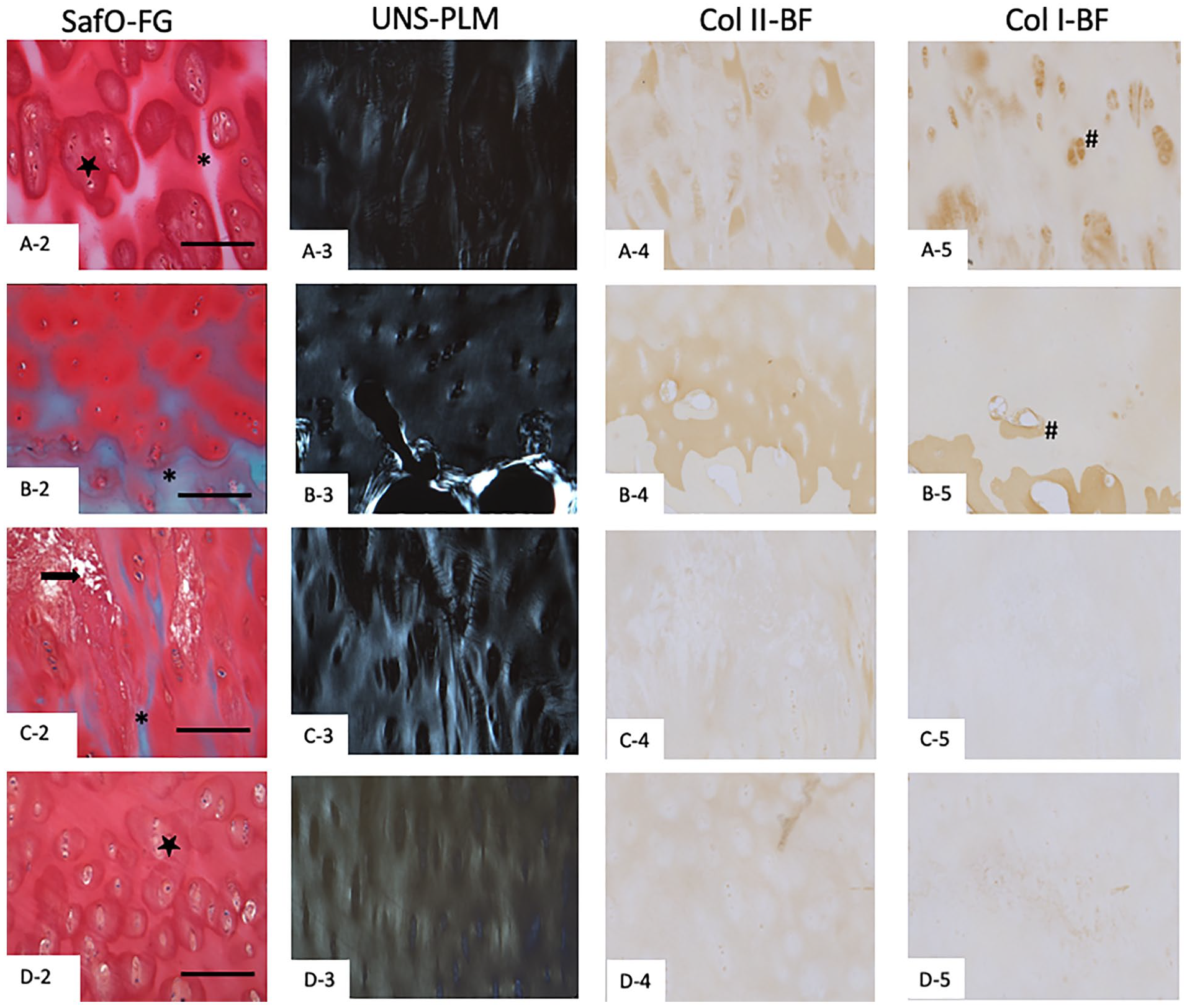
Higher magnification (10×) of the boxed regions shown in Figure 6 to illustrate the osteoarthritic features in detail. Box regions of interest from the cartilage presented in Figure 6 (SafO-FG staining features), as seen under safranin-O/fast green (column 1 SafO-FG), unstained polarized light microscopy (column 2 UNS-PLM), collagen II immunohistochemical staining (column 3 Col II-BF), and collagen I immunohistochemical staining (column 4 Col I-BF). Scale bar represents 200 µm. Note that since the staining was performed on different sections of cartilage, sometimes overlapping of same regions on different sections was not possible. Asterix indicates inter-territorial matrix loss; star indicates territorial matrix loss; arrow indicates matrix necrosis; # indicates some of the Col I stained regions).
The corresponding unstained sections when viewed under PLM displayed even wider variations in birefringence patterns ( Fig. 6A3-D3 ). There were variations seen in the SZ birefringence patterns with Figure 6B3 showing the closest to normal SZ pattern. However, the patterns of birefringence in the DZ were strikingly different suggesting varying degrees of collagen matrix remodeling in the DZ. These observations are in line with our correlation analysis where we found moderate correlation between HHGS or OARSI and PLM-SZ scores indicating worsening superficial zone PLM scores with increasing OA severity. However, the correlation between HHGS or OARSI and PLM-DZ were poor suggesting that deep zone changes as observed under PLM were not being considered by traditional histological scores. In comparison to the PLM birefringence pattern for the “best-quality” cartilage in our cohort ( Fig. 4A3 ), Figure 6A3 , C3 , and D3 showed discontinuous and localized high-intensity birefringence signal. Col II immunostaining was seen throughout the cartilage section, although with the different SafO-FG staining patterns, the staining patterns in the DZ cartilage became patchy and irregular ( Fig. 6A4-D4 ). Col I immunostaining was distinctly seen as a thin layer in the SZ of cartilage in all specimens ( Fig. 6A5-D5 ). However, we also found Col I staining around chondrocytes in hyaline cartilage regions with interterritorial SafO loss ( Fig. 7A5 ) and around blood vessels, suggesting bone formation in hyaline cartilage regions near the tidemark that also showed SafO loss ( Fig. 6B5 ).
For all the specimens, corresponding PSR (Supplementary Fig. 2A1-D1) and PSR-PLM (Supplementary Fig. 2A4-D4) images showed similar enhanced birefringence patterns in comparison to unstained-PLM images. The only cartilage region that differed in the birefringence pattern was near the tidemark, where the unstained-PLM showed normal or high birefringence signal, whereas the PSR-PLM showed a significantly reduced birefringence signal (green). The reason for this difference is not known presently.
Overall, it was noted that even though all the cartilage specimens were scored as slight loss of SafO-FG as per conventional HHGS scoring system ( Fig. 6 ), the different patterns of SafO loss observed suggest varying degrees of collagen matrix remodeling, which was most apparent in the unstained-PLM images.
OA Cartilage Specimens with Normal Surface Structure Exhibiting Modifications Near Tidemark
Cartilage specimens from 46 out of the 90-patient cohort (51% of the specimens) showed OA progression with no surface fissures in the cartilage (HHGS structure score = 0 or 1) but showed varying patterns of OA progression near the tidemark ( Fig. 8 ). As per the HHGS scoring system, tidemark integrity can be scored 0 (tidemark not breached) or 1 (tidemark breached by blood vessels). This scoring system does not take into account various histopathological features as seen in this primary OA cohort like: multiple tidemarks that have not been breached by blood vessels ( Fig. 8A1 ; Fig. 9A1 ), unspecified tissue composition deposited near the tidemark ( Fig. 8B1 ; Fig. 9B1 ), multiple tidemarks that have been breached by blood vessels ( Fig. 8C1 ; Fig. 9C1 ), and formation of bone tissue well within the hyaline cartilage region resulting in the appearance of cartilage-bone-cartilage-bone interleaved tissue layers accompanied by multiple tidemarks ( Fig. 8D1 ; Fig. 9D1 ).

Representative human cartilage specimens displaying normal cartilage structure but exhibiting varying patterns of modifications near tidemark as seen under hematoxylin and eosin (HE) (column 1:
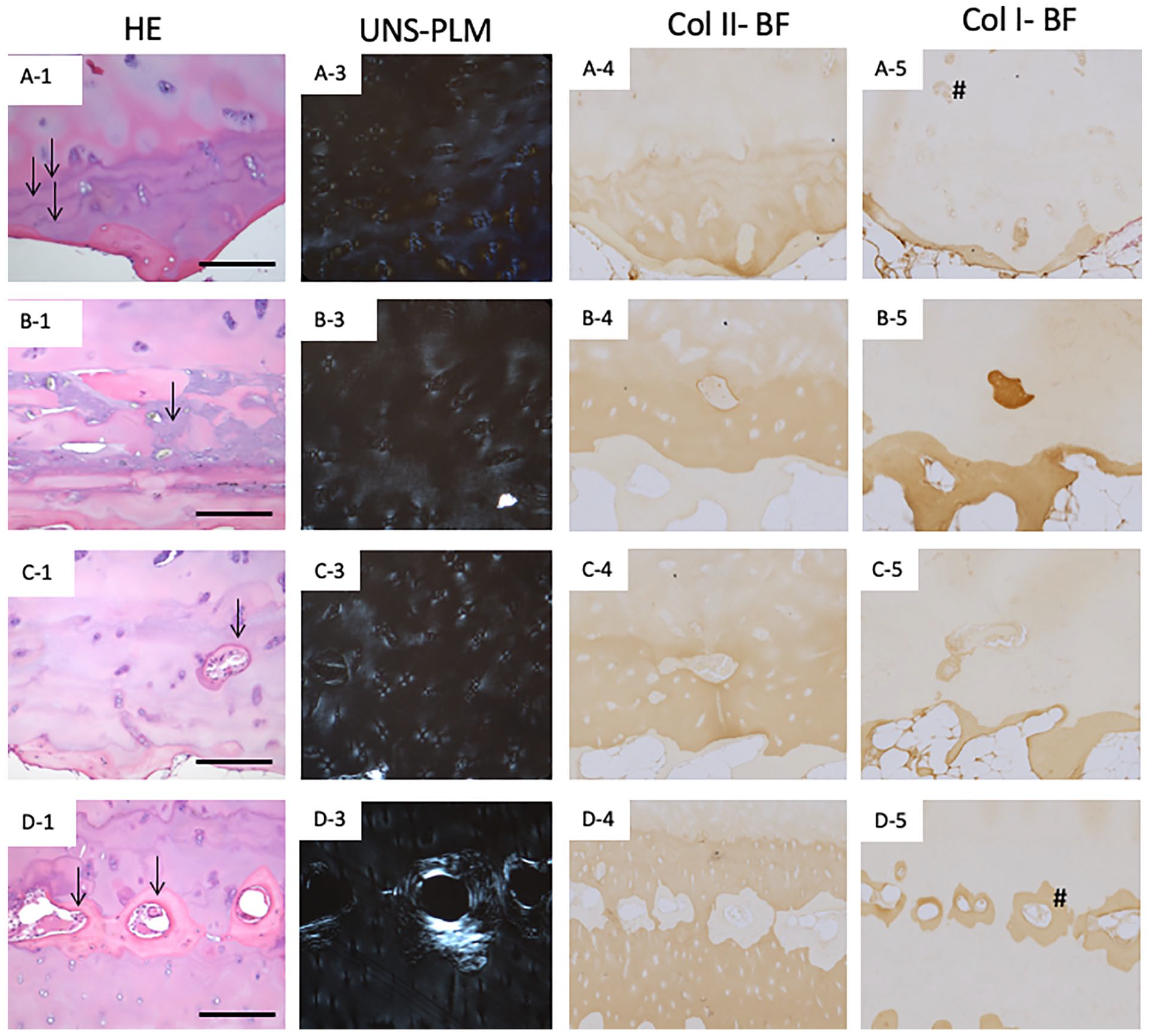
Higher magnification (10×) of the boxed regions shown in Figure 8 to illustrate the osteoarthritic features in detail. Box regions of interest from the cartilage presented in Figure 8 (tidemark features), as seen under hematoxylin and eosin (column 1 HE), unstained polarized light microscopy (column 2 UNS-PLM), collagen II immunohistochemical staining (column 3 Col II-BF), and collagen I immunohistochemical staining (column 4 Col I-BF). Scale bar represents 200 µm. Note: Hash indicates some of the Col I stained regions.
The corresponding unstained sections when viewed under PLM displayed wide variations in birefringence patterns ( Fig. 8A3-D3 ). All the specimens exhibited birefringence near the SZ although they displayed uneven thickness of the SZ with disoriented patchy appearing birefringence not parallel to the articular surface. The TZ cartilage showed varying thickness of low birefringence compared with “best-quality” cartilage ( Fig. 8B3 , C3 , D3 vs. Fig. 4A3 ). Although birefringence was present throughout the DZ cartilage, the patterns varied between upper-DZ, mid-DZ and lower-DZ regions, with granular, patchy, and irregular intensity birefringence. Interestingly, there was a unique pattern of birefringence around the chondrons in the DZ cartilage ( Fig. 9A3-D3 ). In case of specimens like Figures 8D3 and 9D3 , birefringence was observed associated with the bone formed within the hyaline cartilage region.
Col II immunostaining was detected throughout the cartilage tissue with irregular staining patterns seen in the SZ and in the DZ where noticeable loss in SafO staining occurred ( Fig. 8A4-D4 ). Col I immunostaining was distinctly seen as a layer of varying thickness in the SZ of cartilage in all specimens ( Fig. 8A5-D5 ). Col I staining was also seen around chondrocytes in TZ ( Fig. 8A5 ) and DZ ( Fig. 8B5 ) regions of hyaline cartilage. Bone tissue that formed around the blood vessels in the DZ of hyaline cartilage was distinctly stained with Col I ( Figs. 8D5 and 9D5 ).
Overall, it was noted that even though all the cartilage specimens ( Fig. 8 ) showed normal or slight surface irregularities, with slight loss of SafO staining, the different patterns of changes near the tidemark indicated varying patterns of cartilage remodeling leading to OA.
Discussion
Primary OA is a degenerative joint disease that encompasses a spectrum of histopathological features that have not been completely defined and fully understood. 31 Understanding what these histopathological features mean exactly may help define different OA-subtypes, better understand different OA progression mechanisms, and help identify early OA histopathological indicators. The current standard methods for primary OA histopathological assessment are HHGS and OARSI systems that use HE and SafO-FG stained tissue sections to assess the condition of cartilage tissue degradation 8 . HE allows evaluation of overall cartilage tissue features, cell morphology, bone-cartilage interface, abnormal calcification, and tidemark. 32 SafO-FG provides an assessment for presence of sulfated glycosaminoglycans by staining them red/pink and the subchondral bone green. 33
In this study, we demonstrated that both HHGS and OARSI have weak or moderate correlation with PLM-Changoor score. While this scoring system was designed to assess repair cartilage and not native cartilage, it is the only reported PLM scoring system to date and was thus included in our analyses. However, the histopathological features of primary OA, especially mild and moderate OA are different than repair cartilage. We found most of our specimens categorized as a 3 or 4 PLM-Changoor score, since the specimens displayed all three zones of cartilage with varying birefringence patterns in the SZ and DZ cartilage. For this reason, we developed a new semiquantitative PLM-SZ and PLM-DZ scoring system for primary OA specimens that separately scores SZ and DZ birefringence patterns, respectively. Interestingly, we determined a moderate association between PLM-SZ score and HHGS and OARSI, however, a weak association between PLM-DZ and HHGS and OARSI scores. This suggests that cartilage degradation as determined by HHGS and OARSI is associated more strongly with the collagen alterations in the SZ but not with the DZ. In addition, it was also noted that specimens exhibiting early and mild OA (HHGS scores <5 and OARSI scores <8.6) had PLM-SZ scores between 0 and 4 and PLM-DZ scores between 0 and 3. These results suggest that for early or mild OA cartilage specimens, PLM may be more effective at detecting SZ and DZ collagen changes, which may not be evident by the traditional HHGS and OARSI scoring systems using HE/SafO stains. In addition, effort was made to identify additional HE- and SafO-based features that may be able to better inform collagen changes (PLM birefringence patterns). We will have to test this on a bigger sample cohort to confirm our findings.
Based on the observations in our prior work3,4 and in this study, we found that traditional scoring methods like HHGS and OARSI using stains HE and SafO-FG provide incomplete histological assessments of primary OA cartilage especially at earlier stages of the disease when cartilage structural modifications are subtle or minimal. In specimens with substantial evidence of tissue remodeling or damage, the grade of cartilage matrix alterations determined by HE and SafO-based HHGS and OARSI correlated well with the level of cartilage matrix alterations identified by PLM (44% of specimens in our cohort). However, this correlation diminished when structural modifications were minimal (56% of specimens in our cohort) and OA changes occurring mainly interterritorial and territorial ECM and near the tidemark and SCB.3,20,27 The results from this study suggest that articular cartilage histopathological assessments may be improved by (1) adding a PLM score for primary OA cartilage degeneration in the existing scoring system and/or (2) refining the existing scoring systems to include the defined SafO and tidemark features observed in our sample cohort. The first option would demand an additional mode of imaging and expertise in PLM. The second option to modify the existing HHGS and OARSI systems to add new histopathological features would require including better definitions in cartilage tissue alterations that are not currently validated. Both of these approaches would require further testing on larger sample cohorts and more detailed definition of the histological parameters to score.
These unrecognized histopathological features are important to consider as they may be the result of different possible mechanisms of OA progression and, if validated, may serve as additional indicators of early primary OA. Scanning electron microscopy studies have demonstrated that collagen fiber disassembly occurs at an early stage of the disease when the articular cartilage surface is still smooth and intact.22,34 Disruption or disorganization of collagen fibers has been associated with an eventual loss of the proteoglycans, which would accelerate the rate of damage accrual in cartilage.13,21,35,36 Contrarily, some studies have reported that OA first affects proteoglycans followed by changes in the collagen meshwork.37-39 Other studies have demonstrated that cartilage tissues with increasing Outerbridge grade demonstrate collagen fiber rearrangement and disorganization involving increased entanglement of thin collagen II fibrils and collagen network breakdown.20,22 The OA progression observations in our study may be explained by (1) influence of inflammatory signals from the surrounding tissues, which result in the production of collagen I instead of collagen II by chondrocytes along the fissures38,39; (2) increased synthesis and packing density of collagen in OA cartilage40-42; (3) disorganization, derangement, reorganization, or reorientation of collagen fibrils resulting from physiological adaptive responses to injury all of which can lead to changes in collagen birefringence43-45; (4) degradation of cartilage matrix aggrecan and collagen mediated by proteinases46,47; and (5) changes in SCB and calcified cartilage zone that alter load distribution patterns at the bone-end leading to cartilage/bone thickness changes, collagen modifications, and altered metabolic cell responses.40-43
Based on our findings, we would recommend using PLM rather than PSR-PLM for assessing OA cartilage. We found that although PSR enhances the birefringence of collagen, giving better visualization of OA changes, there were discrepancies in the birefringence patterns between PSR-PLM and PLM sections, especially in the DZ regions of cartilage adjacent to the tidemark ( Supplementary Figs. 1 and 2 ). The PSR-PLM showed very low levels of birefringence (green) in this region. This however, was not true with PLM, where we observed normal or high intensity birefringence, which is appropriate and corresponds to observations by other groups reporting collagen fibrils tend to become thicker near the DZ.22,44 The reason for low PSR staining in the DZ is unclear, but there have been contradicting studies reporting on the stoichiometric binding of PSR stain to collagen molecules suggesting that PSR binding to OA cartilage may still be incompletely understood and needs further assessment.45 -47 Some studies suggest PSR-PLM should be performed on sections after removing proteoglycans, as they often obscures the Col II epitopes, as seen in our study by reducing IHC Col II staining intensity in cartilage tissue sections. 19 Consensus explanations for the different colors of birefringence in PSR-PLM (red, orange, yellow, green) is lacking. Previous studies have shown that these colors may be more representative of collagen disorganization, which includes rearrangement in orientation or thickness of collagen fibers rather than composition of collagen.16,47-50 Normal human cartilage when viewed under PSR-PLM should show a thin band of yellow to red birefringence corresponding to the dark-red stained region in PSR-BF, parallel to the surface in the SZ of articular cartilage. 51 On the other hand, DZ cartilage typically exhibits green or no birefringence orthogonal to the surface.52,53 While the specimen shown in Figure 4A is not a completely normal cartilage, it represents the closest to normal collagen orientation in our sample cohort. Similar birefringence patterns were reported by Changoor et al., who found these patterns in normal unstained cartilage samples from elderly populations.21,48 Importance of PLM has been established in grading repair cartilage.13,54 Thus, it may be valuable for the assessment of primary OA cartilage degeneration as well.
Col I and II immunostaining provides additional information on the composition of collagen to compare to PLM imaging. The results from this study along with others demonstrate that PLM is more representative of the collagen organization than its collagen type composition. In collagen organization, the intensity of birefringence is predicted to be best correlated to collagen age and crosslinking between collagen fibrils. More crosslinking between fibrils can lead to higher birefringence in cartilage.13,16,22,47 Less mature fibrils are more hydrated and less uniformly aligned than more mature collagen, and so they exhibit less birefringence compared to more mature collagen. This could explain the appearance of weaker birefringence in Figures 6A5 and 8D5 . This Col I deposition could indicate newly formed, less mature Col I fibers that would result in less birefringence than more mature Col I fibers.
Although articular cartilage is composed primarily of collagen type II, many studies have reported the importance of less abundant collagen molecules like type VI, IX, and XI that play an important role in maintaining collagen II structural integrity. Future investigations should focus on these molecules as well to possibly find an explanation for which combination of collagen types can be best correlated to the PLM birefringence patterns. Scanning electron microscopy, transmission electron microscopy, and second harmonic generation microscopy may be other imaging techniques that could be used to potentially study the organization and composition of the collagen molecules in cartilage.
It has been shown that in varus aligned knees, the risk of OA progression in the medial femoral condyle (MFC; biomechanically stressed compartment) is increased, while it is decreased in the lateral femoral condyle (LFC; unloaded compartment). 55 However since OA is a whole joint disease, both MFC and LFC present in the same joint share the same synovial fluid and potentially the same inflammatory milieu. By collecting samples from the lateral femoral condyle (less loaded compartment) of varus knees, we contend to have collected representative samples exhibiting more early to intermediate stages of OA.3,4 Although the exact sequence of events during OA in either MFC or LFC are unknown, the histological features reported by our previous studies and other studies in LFC, suggest a decrease in proteoglycan content, alterations at the bone-cartilage interface, clonal grouping of chondrocytes, hypercellularity, and/or hypocellularity, all of which are all histological outcomes of OA progression. Additionally, numerous studies have reported progression of OA in the lateral compartment as a source of failure in medial unicompartment arthroplasty,56-59 suggesting continued OA disease progression due to a compromise of LFC structural and mechanical integrity despite the “normal” radiographic appearance of this knee subcompartment in the early phases.60,61
Our study is not without limitations. Even though the LFC cartilage from varus knee TKA patients were relatively preserved, these were collected from the OA joint space and thus would not represent normal cartilage features. We lacked “true normal” controls for comparative assessment in this study. Additionally, for PLM assessment, we could not control for some differences that may have been introduced during specimen preparation. In particular, specimen fixation has been reported to alter birefringence signal intensity due to changes in bulk optical properties of the material (cross-linking of proteins), as well as change in the total optical path length (tissue shrink during fixation). Since all the tissue sections in this study were prepared using the same standardized protocol, then the differences we observed and noted in this study’s specimens are unlikely affected by this issue. Nevertheless, it becomes problematic to consider our findings fully comparable to those of other studies investigating in situ cartilage structure. Finally, the scoring system is non-parametric, which weakens the correlation analysis for a 90-patient cohort size. In addition, other uncontrolled patient variables or covariates might have influenced the regression analysis. For example, loss of PLM-SZ may also be related to age or BMI, given that “wear and tear” is hypothesized to play a role in OA progression.
Conclusions
PLM was effective at identifying early SZ and DZ collagen alterations that were not evident in the traditional histopathology scoring systems. Incorporating PLM scores and/or additional HE/SafO features assisted us in better characterization of early/mild primary OA specimens. PLM was able to distinguish specimens which exhibited minimal surface degradation but showed considerable collagen modifications/degradation. On the other hand, both HHGS and OARSI poorly assesses early/mild OA where surface degradation is minimal and is unable to distinguish the subtle patterns of early OA development. Substantial cartilage tissue alterations (proteoglycan loss, collagen matrix remodeling and subchondral bone changes) can be initiated without obvious mechanical surface disruption, and these alterations may play roles in the initiation or propagation of primary OA. Addition of a PLM score in the existing HHGS scoring system, and/or modifying the existing HHGS scoring systems to include the additional SafO and tidemark features that can correlate with PLM-observed collagen modifications should help us refine and advance our understanding of early and mild primary OA progression. Further investigation of PLM may allow a better understanding in classifying OA-subtypes, OA progression and identification of early OA histopathological features.
Supplemental Material
suppl1 – Supplemental material for Comparative Assessment of Primary Osteoarthritis Progression Using Conventional Histopathology, Polarized Light Microscopy, and Immunohistochemistry
Supplemental material, suppl1 for Comparative Assessment of Primary Osteoarthritis Progression Using Conventional Histopathology, Polarized Light Microscopy, and Immunohistochemistry by V.P. Mantripragada, W. Gao, N.S. Piuzzi, C.D. Hoemann, G.F. Muschler and R.J. Midura in CARTILAGE
Supplemental Material
suppl2 – Supplemental material for Comparative Assessment of Primary Osteoarthritis Progression Using Conventional Histopathology, Polarized Light Microscopy, and Immunohistochemistry
Supplemental material, suppl2 for Comparative Assessment of Primary Osteoarthritis Progression Using Conventional Histopathology, Polarized Light Microscopy, and Immunohistochemistry by V.P. Mantripragada, W. Gao, N.S. Piuzzi, C.D. Hoemann, G.F. Muschler and R.J. Midura in CARTILAGE
Supplemental Material
suppl3 – Supplemental material for Comparative Assessment of Primary Osteoarthritis Progression Using Conventional Histopathology, Polarized Light Microscopy, and Immunohistochemistry
Supplemental material, suppl3 for Comparative Assessment of Primary Osteoarthritis Progression Using Conventional Histopathology, Polarized Light Microscopy, and Immunohistochemistry by V.P. Mantripragada, W. Gao, N.S. Piuzzi, C.D. Hoemann, G.F. Muschler and R.J. Midura in CARTILAGE
Footnotes
Authors’ Note
All the work was performed at Lerner Research Institute, Cleveland Clinic.
Acknowledgments and Funding
We would also like to acknowledge the histological processing skills of Katrina Fife in the Histochemistry Core Facility in the Biomedical Engineering Department (Lerner Research Institute, Cleveland Clinic). The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Health grant (R01AR063733) awarded to GFM.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Institutional Review Board committee of the Cleveland Clinic (Protocol: 13641).
Informed Consent
Written informed consent was obtained from all subjects before the study.
Trial Registration
Not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
