Abstract
Mouse models of osteoarthritis and cartilage degeneration are important and powerful tools for investigating the molecular mechanisms of the disease pathology. Because of the vast number of genetically modified mouse models that are available for research, the ability to use these models is particularly attractive for the mechanobiologic interactions in the pathogenesis of osteoarthritis. However, the very small scale of mouse articular cartilage, where the healthy tissue is only 80 µm in thickness, poses challenges in quantifying mechanical characteristics of the tissue. We introduce here a novel approach that combines experimental and analytical methods to quantify the nuanced mechanical changes during cartilage degeneration at this scale. Cyclic reference point indentation is used to directly test the murine articular cartilage to obtain the force-deformation and the phase-shift characteristics of the tissue. The cartilage zonal thicknesses are confirmed from histology. These data are then fitted to a parallel spring model to determine the depth-dependent tissue stiffness and modulus. Using this approach, we investigated the effects of trypsin degradation on the zonal mechanical behavior of mouse articular cartilage. We observe a decline of the superficial zone stiffness coupled with the loss of the superficial layer. Subsequent degradation by trypsin allowed the identification of middle- and deep- zone properties. Taken together, this approach can be a useful tool for understanding the disease mechanisms of cartilage homeostasis and degeneration, and for monitoring of therapies for osteoarthritis.
Introduction
Osteoarthritis affects nearly 100 million people globally. 1 The pain resulting from the degeneration of articular cartilage causes significant loss of mobility and quality of life.2,3 Articular cartilage is hyaline cartilage of complex structure and composition and consists mostly of collagen (90%-95% type II) fibers, proteoglycans, and water. 4 The superficial zone accounts for 10% to 20% of the cartilage thickness and resists shear forces. The middle zone is the next 40% to 60% thickness and it provides modest resistance to compressive forces. The deep zone contains the cartilage-bone transitional interface and consists of high proteoglycan concentrations and vertically aligned collagen fibrils. 4 Chondrocytes are interspersed in the extracellular matrix (ECM), and are responsible for maintenance of the ECM. However, turnover of ECM constituents is remarkably slow, with proteoglycan turnover rate of 25 years 5 and collagen turnover rate of up to centuries. 6
Murine models are particularly attractive for the mechanistic studies of cartilage degeneration. A large number of transgenic, surgical, and injury models are available in mice. 7 Because murine articular cartilage thickness at the lateral tibial plateau is on the order of 80µm, 8 mechanical characterization is challenging at this scale. Moreover, articular cartilage exhibits time-dependent viscoelastic mechanical behavior. 9 The heterogeneous structure of articular cartilage further complicates testing due to zone-dependent mechanical behavior.
This viscoelastic behavior means traditional static testing methods will not capture the time-dependent behavior of articular cartilage. Stress-relaxation and creep methods for testing articular cartilage are well-documented,10,11 but require significant testing times and do not provide specific quantitative insights in the depth-dependent changes of the cartilage. We propose here an approach that combines experimental and analytical methods to quantify the depth-dependent degeneration of the murine articular cartilage.
Methods
Six tibial plateaus were obtained from 12-week old male BALB/c mice with Washington University Institutional Animal Care and Use Committee approval. A scalpel cut was made above midshaft to separate each plateau from the rest of the tibia with the assistance of a dissection scope (M400 Photomakroscop; Wild, Heerbrugg, Switzerland). The cut at the bone was made parallel to the medial plateau surface, ensuring a level surface during mechanical testing. Careful cuts were made to sever the cruciate ligaments between the femoral condyles and tibial plateau. All samples were wrapped in gauze soaked with phosphate-buffered solution (PBS), and stored at −80°C until ready for use.
Biochemical Treatment
Trypsin incubation was used to induce degradation of the articular cartilage. The tibial plateaus were removed from −80°C storage and allowed to thaw for 30 minutes. They were then mechanically tested as a time-0 group. On completion of mechanical testing, the same samples were submerged in 1X trypsin–ethylenediaminetetraacetic acid solution (Sigma-Aldrich, St. Louis, MO) and incubated at 37°C. Trypsin is a serine protease that allows for targeted proteoglycan cleavage. 12 At 5, 20, and 60 minutes of total submersion time, they were removed from trypsin solution and washed with PBS to stop trypsin degradation. They then underwent mechanical testing. The samples were then placed back into fresh trypsin solution and incubated at 37°C until the next time point (not including mechanical testing time), with the exception of the final 60-minute time point.
Mechanical Testing Using Cyclic Reference Point Indentation
The tibial plateaus were fixed to 1 cm × 1 cm × 0.3 cm square aluminum platens using cyanoacrylate adhesive. The platen with attached sample was then placed in a PBS-filled petri dish. The petri dish was placed on a balance (Mettler-Toledo, Columbus, OH) and tared. Mechanical testing was conducted using a BioDent reference point testing apparatus (Active Life Scientific, Santa Barbara, CA), with load cell resolution of 0.001 N and piezo-actuator resolution of 0.01 µm. A nonporous cylindrical flat punch indenter, diameter 0.375 mm, was lowered onto the center of the lateral plateau, until a steady preload of 5 g held for 30 seconds ( Fig. 1A ). The test probe then engages sinusoidally at 0.05 Hz for 5 cycles, with an indentation of 20 µm ( Fig. 1B ). Testing was conducted at 6 additional frequencies up to 1 Hz to determine the frequency-dependent loss tangent of the articular cartilage; the specimen was allowed to return to preload state between frequencies. Pilot study indicated no evidence of mechanical changes from repeated testing, with stiffness and loss tangent remaining consistent. The selected strain is well within the range of previous murine articular cartilage literature. 13
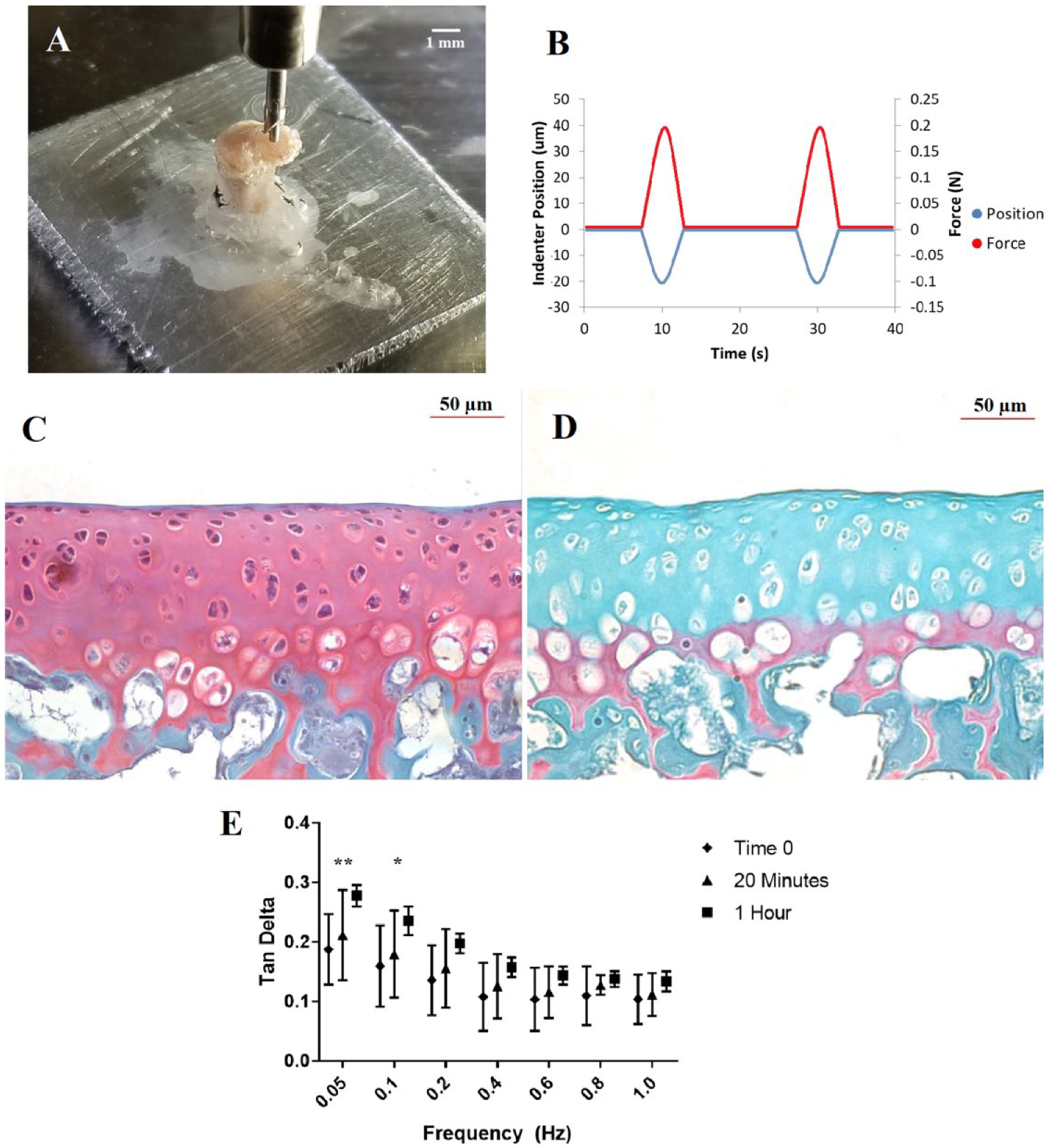
(
The viscoelastic loss tangent (tan δ) was calculated from the force-time and displacement-time curves. The average time delay between the displacement peak and the associated peak force response was determined using a Matlab script and converted to a phase angle in radians. The tan δ is therefore the tangent of the phase angle.
Histology
The histology specimens were fixed in 3% paraformaldehyde (Sigma-Aldrich, St. Louis, MO). They were then decalcified for 10 days in Immunocal (StatLab, McKinney, TX), and then dehydrated in increasing concentrations of ethanol. The specimens were embedded in paraffin and cut into 10−µm sections. Safranin-O and fast green stains were applied to visualize the extent of proteoglycan depletion. The slides were viewed under light microscopy (Zeiss, Oberkochen, Germany).
Data Analysis and Modeling
To account for nonlinearity and depth-dependent behavior, a parallel-spring recruitment model was used ( Fig. 2B ). Parallel spring models have been previously used to model articular cartilage behavior.14,15 The unloading force-displacement data corresponding to the first D1 of indentation distance was fitted to a linear regression, the slope of which is the superficial spring stiffness (K1) in N/mm. This was repeated for the remaining D2 of indentation distance, yielding a total stiffness (Ktotal) that is the linear combination of K1 and K2, the middle spring stiffness. K2 is thus simply Ktotal − K1. Stiffness values for each of the 5 cycles per test were averaged. D1 was determined by the displacement where the unloading curve diverged from linearity, indicating the contribution of an additional spring element. Accordingly, we chose 6 µm as the transition distance, or 30% of the total indentation.
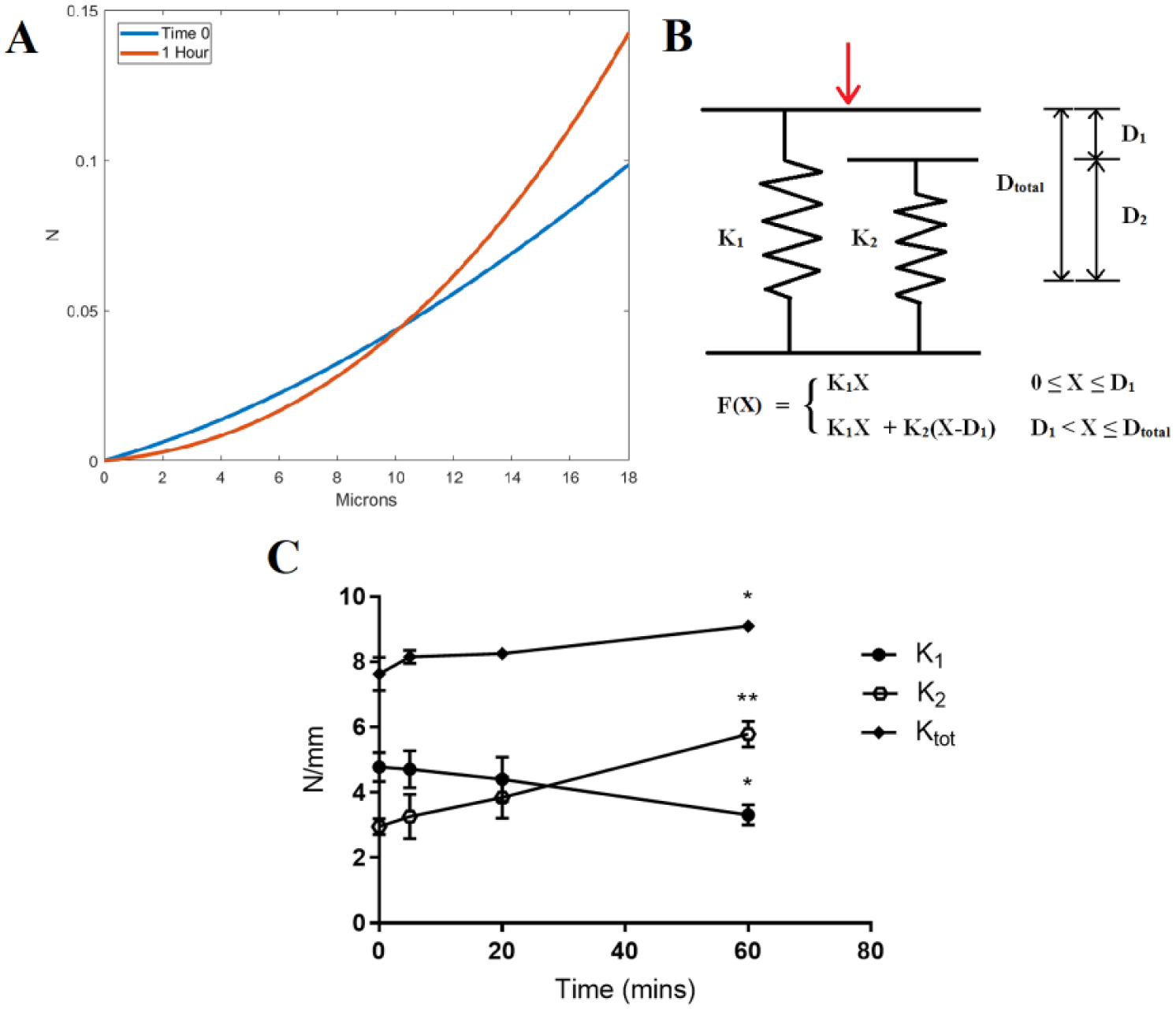
(
Statistical Analyses
Paired Student t tests were conducted to determine any differences in stiffness between time-0 and trypsinated cartilage. Repeated-measures 2-way analysis of variance (ANOVA) with post hoc Tukey’s test was conducted on loss tangent data to determine the effects of treatment time and frequency. All statistics are 2-tailed and were computed using GraphPad Prism 7 (GraphPad, La Jolla, CA).
Results
Histological assessment of the control and trypsin-treated cartilage samples confirm the loss of proteoglycans, as evidenced by the reduction of safranin-O uptake ( Fig. 1C and D ). As determined by repeated-measures 2-way ANOVA, loss tangent data were significantly driven by both trypsination time (P < 0.0001) and frequency (P < 0.0001) factors. Significant differences in loss tangent were observed between the time-0 and 1-hour time points at 0.05 Hz (+47%, P = 0.007), and at 0.10 Hz (+50%, P = 0.02) ( Fig. 1E ). Post hoc Tukey’s tests revealed there were no frequency-dependent differences in tan δ within the time-0 cartilage group, but significant frequency-dependent differences within all trypsin-treated time groups. Changes in K1 and K2 give the 1-hour trypsinated cartilage apparent nonlinear loading behavior (see representative data, Fig. 2A ). Between time-0 and 1-hour trypsination, the superficial stiffness K1 significantly decreased by 30.8% (P = 0.05), the middle stiffness K2 significantly increased by 103.5% (P = 0.005), and the total stiffness Ktotal significantly increased by 19.1% (P = 0.04) ( Fig. 2C ).
Discussion
This method is aimed at rapid phenotype screening for murine articular cartilage stiffness and loss tangent. Additionally, it allows study of depth-dependent heterogeneity without invasive sample preparation, such as microtome sectioning. Cyclic reference point indentation was used to obtain the force-deformation characteristics from murine articular cartilage. This data are then fitted to a parallel spring model to determine local tissue stiffness and modulus.
Ideally, biphasic or triphasic theory–based approaches would be applied for subsequent higher order characterization; these have been extensively studied in bovine models. 16 However, higher order theories have seldom been used with experimental murine cartilage models, 17 and limited mostly to foundational study of healthy murine cartilage, indicating some degree of impracticality for phenotype screening.
Additional spring elements could be included in order to increase complexity within the model, especially for deeper indentations. However, in our study, third spring elements provided negligible improvements in the goodness of fit. For comparison to literature, dynamic modulus can be readily derived from stiffness.18-20 Since most available murine articular cartilage literature probes the superficial zone at a minimum, we use the superficial stiffness of time-0 cartilage for broadest comparison. We find the dynamic (0.05 Hz) modulus of superficial cartilage to be 2.45 MPa ± 0.27 SD. This value is similar to the Young’s modulus of 2.0 ± 0.3 MPa that Cao et al. 8 reported. This study was conducted at the same location in situ, and creep testing was conducted at similar indentation depths as our D1 of 6 µm. In general, there is a wide range of equilibrium and dynamic moduli reported for murine articular cartilage, dependent largely on the testing modality. Studies have reported values as low as 0.05 MPa from dynamic atomic force microscopy, and as high as 2.0 MPa from biphasic microindentation. 13 While atomic force microscopy does allow for dynamic nanoscale testing, 21 it does not allow for mechanical characterization under realistic load conditions. Dynamic modulus can greatly exceed equilibrium compressive modulus, even at physiological strain rates, 22 which may explain why the calculated modulus value is slightly large. Nevertheless, the dynamic modulus of superficial time-0 cartilage (2.45 MPa) appears reasonable.
Proteoglycans help maintain tension of the collagen-proteoglycan network, and proteoglycan cleavage by trypsination is a known mechanism for reducing cartilage stiffness. 23 Collagen fibrils in superficial cartilage are oriented parallel to the surface, and help translate applied compressive forces into tension along the fiber axes. 24 The degradation of this mechanism is represented in our model, with a decrease in superficial spring stiffness due to trypsination, and an increase in contribution from the middle spring. Additionally, glycosaminoglycan-depleted cartilage is known to have a pronounced toe region in its mechanical response. 25 This is corroborated by inspection of force-displacement behavior ( Fig. 2A ) and decreased K1. The increase in tan δ with trypsination corresponds to increased viscous behavior, which is not intuitive given the role of proteoglycans in maintaining hydration and decreasing fluid permeability. However, the flow-dependent mechanism of cartilage viscoelasticity may not be dominant at the relatively low frequencies in our study. 23 Instead, the result may arise from increased flow-independent viscoelasticity of the solid matrix; trypsination has been previously shown to induce disorder in the organization of collagen fibrils. 25 Finally, we find significant decreases in tan-delta with testing frequency, a phenomenon that has been previously reported in bovine cartilage. 22
In conclusion, a combination of cyclic reference point indentation and analytical modeling was used to investigate mechanical changes in trypsinated tibial plateau cartilage, a biochemical model of osteoarthritis. Depth-dependent stiffness and loss tangent were extracted with minimally invasive sample preparation. We believe that this could be a practical method for the mechanical characterization of murine model articular cartilage degeneration.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases (K01AR069116, R21AR069804), National Institute of Aging (R43 060607), and the Washington University Musculoskeletal Research Center (NIAMS P30 AR057235).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Washington University Institutional Animal Care and Use Committee.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
