Abstract
Objective
Mouse models are commonly used in research applications due to the relatively low cost, highly characterized strains, as well as the availability of many genetically modified phenotypes. In this study, we characterized an
Design
Femurs from euthanized mice were removed and clamped in a custom multidirectional vise to create cylindrical osteochondral defects 0.5 mm in diameter and 0.5 mm deep in both condyles. The IPFP contains progenitors that are a promising cell source for the repair of osteochondral defects. For proof of concept, human IPFP-derived progenitor cells, from osteoarthritic (OA) patients, cultured as pellets, were implanted into the defects and cultured in serum-free medium with TGFβ3 for 3 weeks and then processed for histology and immunostaining.
Results
The custom multidirectional vise enabled reproducible creation of osteochondral defects in murine femoral condyles. Implantation of IPFP-derived progenitor cells led to development of cartilaginous tissue with Safranin O staining and deposition of collagen type II in the extracellular matrix.
Conclusions
We showed feasibility in creating
Introduction
Osteochondral lesions can be painful, result in loss of function, and eventually lead to secondary osteoarthritis (OA).1,2 There are numerous surgical treatments for osteochondral lesions, such as arthroscopic lavage and debridement, microfracture, osteochondral auto-grafting and allografting, and autologous cell techniques. 3 To date, autologous chondrocyte transplantation and matrix-assisted chondrocyte implantation are the only Food and Drug Administration–approved cell-based treatments for focal cartilage defects.4,5 Unfortunately, autologous chondrocytes are in limited supply, their chondrogenic potential varies between donors, and diminishes after multiple cell passages.6,7 As a possible alternative, there has been an increased interest in the use of stem cells for the treatment of both focal lesions as well as generalized OA.8,9
Several studies have assessed the chondrogenic capacity of various cell sources, such as bone marrow-derived mesenchymal stem cells (MSCs), adipose tissue-derived MSCs, synovial tissue-derived MSCs, peripheral blood-derived progenitor cells, and umbilical cord blood-derived MSCs.10-12 Cells harvested from the infrapatellar fat pad (IPFP) also have chondrogenic capacity.13-15 The human IPFP is a readily accessible source of progenitor cells for tissue engineering or other cell-based therapies to treat osteochondral defects.16,17 Also, differentiation of IPFP-derived cells toward a chondrogenic and osteogenic lineage results in higher chondrogenic and osteogenic gene expression relative to cells derived from subcutaneous fat. 14 This makes IPFP-derived progenitor cells an attractive source for the treatment of osteochondral lesions in the knee.
It is essential to show initial proof of concept of novel cell therapies in experimental animal models. Rabbits are the most common small animal model for the study of osteochondral repair.
18
Larger animals (e.g., dogs, sheep, goats, pigs, horses) are often used as preclinical models.18-22 However, mice are affordable, easy to breed and maintain in-house, and useful for screening purposes for treatments in large animal models.18,23 The entire mouse genome has been sequenced, and genetically modified mice are very useful in studying molecular processes and pathways involved in cartilage repair and regeneration.18,24 However, the creation of articular defects in mice is technically challenging due to the small size of the joint and the narrow thickness of the articular cartilage of mice.
18
The practicality of using
Our primary objective was to develop a murine model of
Materials and Methods
IPFP Tissue and Cell Isolation
Human IPFP tissue was obtained from human donors (
IPFP Cell Pellets
High-density pellets of human IPFP cells were formed by centrifuging cells in 0.5 mL of chondrogenic medium with 10 ng/mL TGFβ3 at 900 rpm for 5 minutes. The pellets were cultured in 0.5 mL serum-free chondrogenic medium (Dulbecco’s Modified Eagle Medium [DMEM], Mediatech Inc., Manassas, VA), 1× ITS+1 supplement (Sigma-Aldrich, St. Louis, MO), 100 nM dexamethasone (Sigma), 1.25 mg/mL human serum albumin (Bayer, Leverkusen, Germany), 100 µM ascorbic acid 2-phospahate (Sigma), 1% penicillin/streptomycin/gentamycin (PSG; Gibco, Carlsbad, CA), and 10 ng/mL TGFβ3 (PeproTech, Rocky Hill, NJ). The pellets were cultured for 3 days in an incubator before implantation. To optimize fit of cell pellet constructs into the 0.5 mm defects, 3 pellet sizes (consisting of 40,000, 45,000, and 50,000 cells each) were tested in trial implantations.
Murine Osteochondral Defect
C57BL/6 mice (

(
The defects were created 3 to 4 hours after harvesting, after which the isolated femurs were maintained in culture to assess viability. Immediately after creating the defect, IPFP cell pellets were implanted into the drilled defects (empty defects were used as control) using a standard plastic pipette under stereo microscope control. The femoral condyles where then isolated by cutting the distal femur with sharp scissors 1 to 2 mm proximal to the condyles. Bone wax (Surgical Specialties, Wyomissing, PA) was applied to seal off the cut-off end of the femur. The samples were then placed in a well of a 48-well plate, cultured in 1 mL of serum-free ITS+ medium (Sigma-Aldrich) supplemented with 10 ng/mL TGFβ3 (PeproTech), with medium changes every 2 to 3 days for 3 weeks.
Cell viability was assessed in femurs with osteochondral defects (without cell pellet implantation) on days 0, 4, and 21. A live/dead kit consisting of Calcein-AM and Ethidium Homodimer-1 (Invitrogen, Waltham, MA) was used for viability assessments using a laser confocal microscope (LSM-510; Zeiss, Jena, Germany) as previously described. 30 Briefly, Z-scanned image stacks were collected in order to image the full depth of field encompassed by the rounded surface of the condyle. Stacks were then flattened using a maximum intensity Z projection using ImageJ. 31
Flow Cytometry
IPFP cells were detached, using Accutase (Innovative Cell Technologies, Inc., San Diego, CA), counted and suspended in FACS buffer consisting of Hank’s buffered saline solution (Gibco) and 1% bovine serum albumin (Sigma) at a concentration of 0.2 × 106 cells per 100 µL. A panel of CD molecules were used, each with the following fluorochromes: CD34-FITC, CD73-APC, CD90-PE-Cy7, CD140b-BV421, CD146-BV510, and CD105-BV650. Corresponding IgGs with the same fluorochrome conjugates were also used for background gating. To compensate for spectral overlap, unstained cells and individual tubes with each fluorochrome were incubated with Dynabeads to distinguish positive and negative fluorochromes signal. A Novocyte flow cytometer (ACEA Biosciences Inc., San Diego, CA) was used for data acquisition, and FlowJo software (version 10; FlowJo, LLC, Ashland, OR) was used to determine percentage positive signal for each molecule relative to isotype controls.
Histology and Immunohistochemistry
Mouse osteochondral explants from each group were fixed in Z-Fix (3.7% formaldehyde, Anatech Ltd., Battle Creek, MI) and decalcified using Shandon TBD-2 (Thermo Fisher Scientific, Waltham, MA). The samples were processed for embedding in paraffin and cut into 4-µm-thick sections. Slides were stained with Safranin O and Fast Green to visualize the distribution of proteoglycans in the tissue.
For detection of collagen types I and II, all sections were pretreated with pepsin (Digest-All 3, Thermo Fisher Scientific) for 9 minutes at 37°C in a humid chamber. Rabbit anti-human collagen type I antibody (Ab 34710; Abcam, Cambridge, MA) was used at a concentration of 1 µg/mL. Mouse anti-human collagen type II (II-II6B3; Hybridoma Bank, University of Iowa) was used at 2 µg/mL. Primary antibody incubation was conducted at 4°C for 12 to 16 hours using a humid chamber. The ImmPRESS secondary DAB kit (Vector Laboratories, Burlingame, CA) was used for color development. Isotype controls were used to monitor nonspecific staining.
Results
Viability of Mouse Femoral Explants
Cell viability remained high up to 3 weeks in culture ( Fig. 2 ). Some cells at the border of the osteochondral defect were dead, presumably due to the surgical trauma induced by the drill bit, which has been reported previously. 32
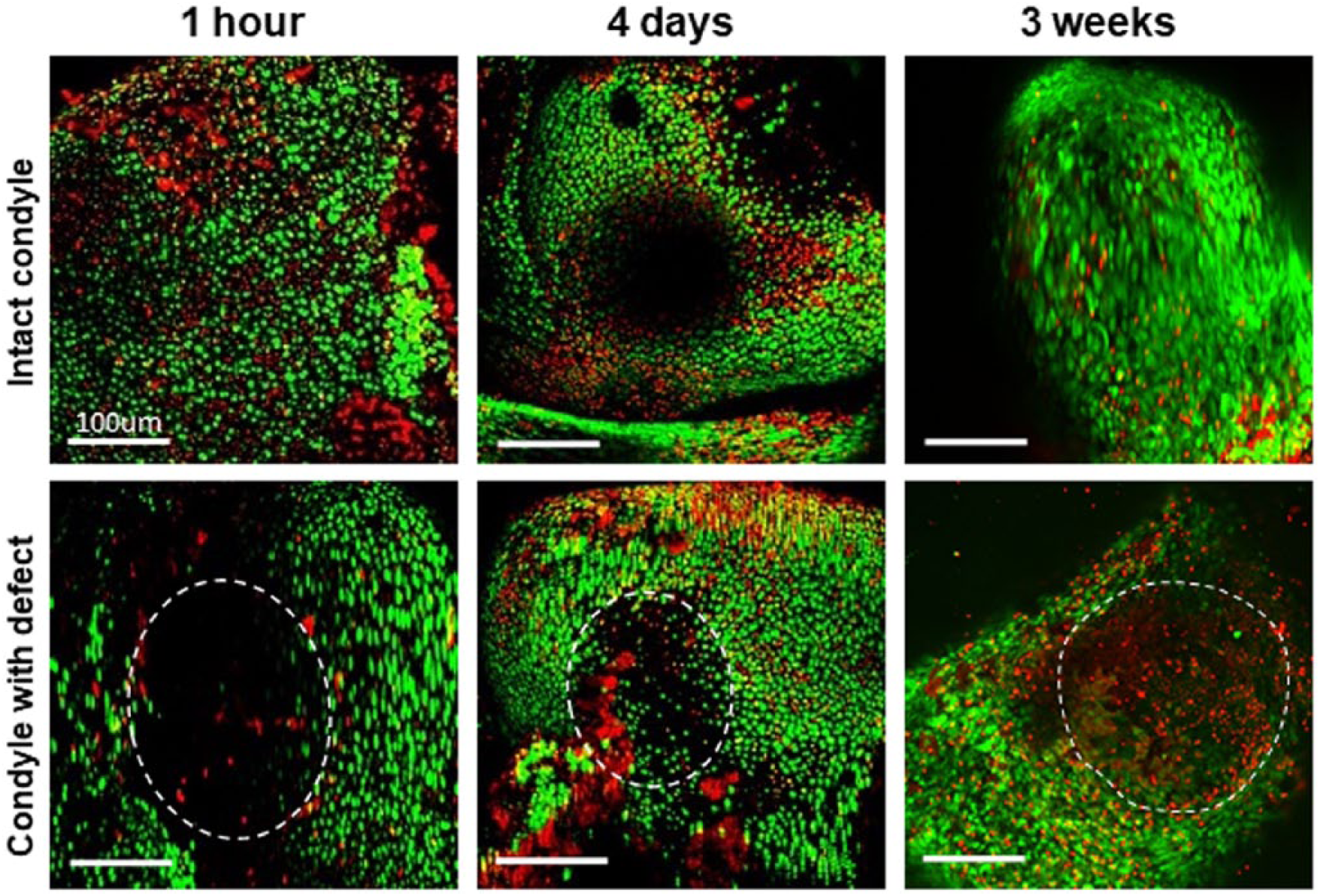
Confocal images showing cell viability of condyles (
IPFP Cells Have a Stem Cell–Like Phenotype
Cells isolated from human IPFP revealed a typical MSC-like profile and were triple positive for CD73, CD90, and CD105 ( Fig. 3A ) and negative for CD34 and CD45. Approximately 50% of the cells were additionally positive for CD140b, a marker for vascular pericytes ( Fig. 3B ). 33
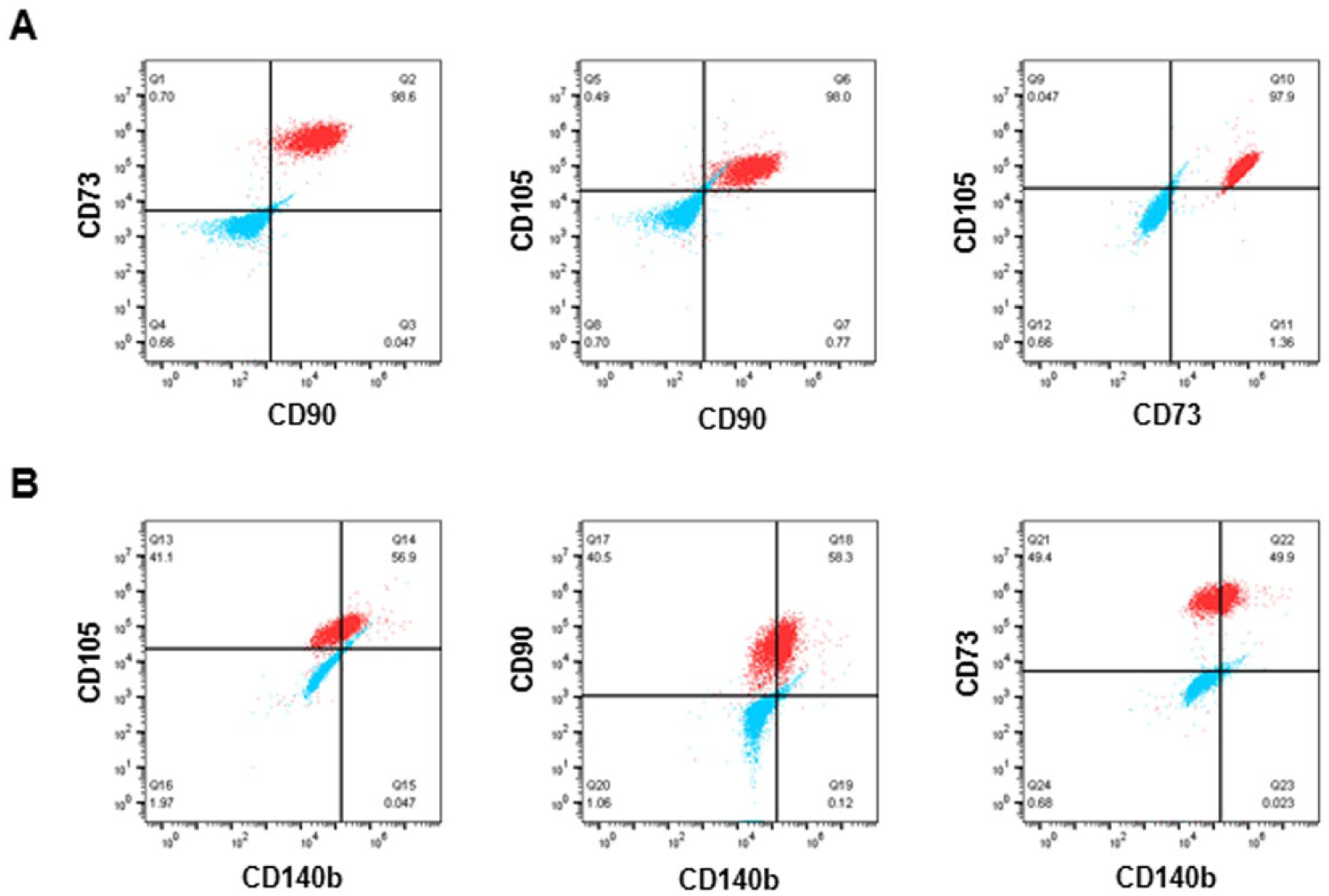
Flow cytometry for isolated human IPFP cells. (
IPFP Pellets
Cell pellet size could be effectively controlled by cell number ( Fig. 4 ) with average pellet diameters ranging from 540 to 670 µm for cell numbers ranging from 40,000 to 50,000, respectively. The pellet size finally chosen for implantation into the 0.5 mm osteochondral defects was approximately 540 µm (40,000 cells; Fig. 4 ) as these pellets consistently filled the entire defect. Larger pellets (50,000 cells) generated exuberant neotissue that covered the entire mouse condyle.
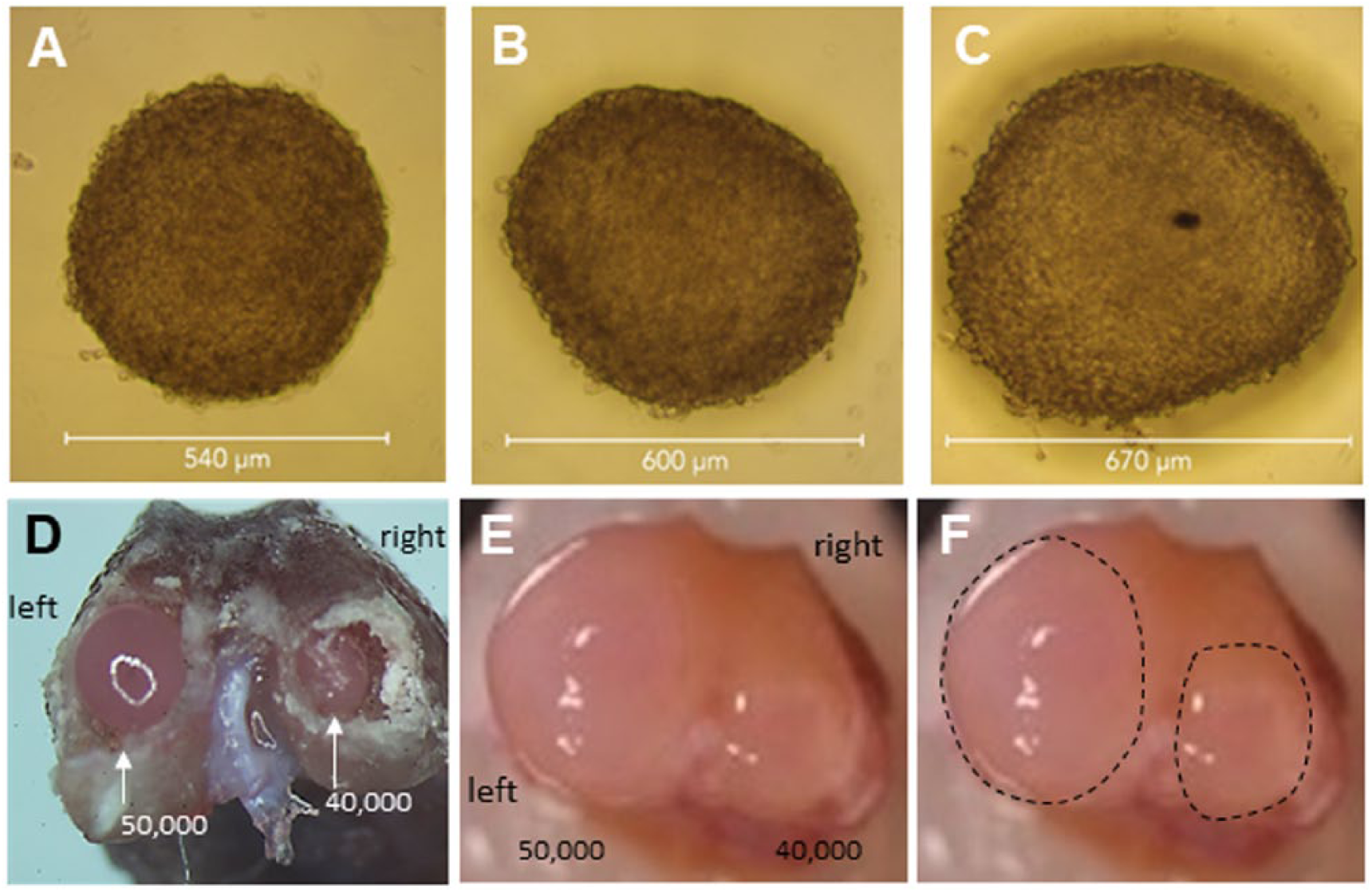
IPFP pellet cultures were formed with a range of cells numbers to assess pellet sizes for implantation into mouse femur defects. (
Cartilaginous Repair Tissue Created by IPFP Cells
No histologic evidence of repair tissue was found in the untreated osteochondral defects. The implanted cells generated tissue that covered the entire femoral condyle. The histologic sections of the condyles implanted with IPFP pellets revealed hyaline-like cartilage repair neotissue, with intense Safranin O staining, and with extracellular matrix staining positive for collagen types I and II ( Fig. 5 ). Isotype control staining for each collagen was negative ( Fig. 5 ).
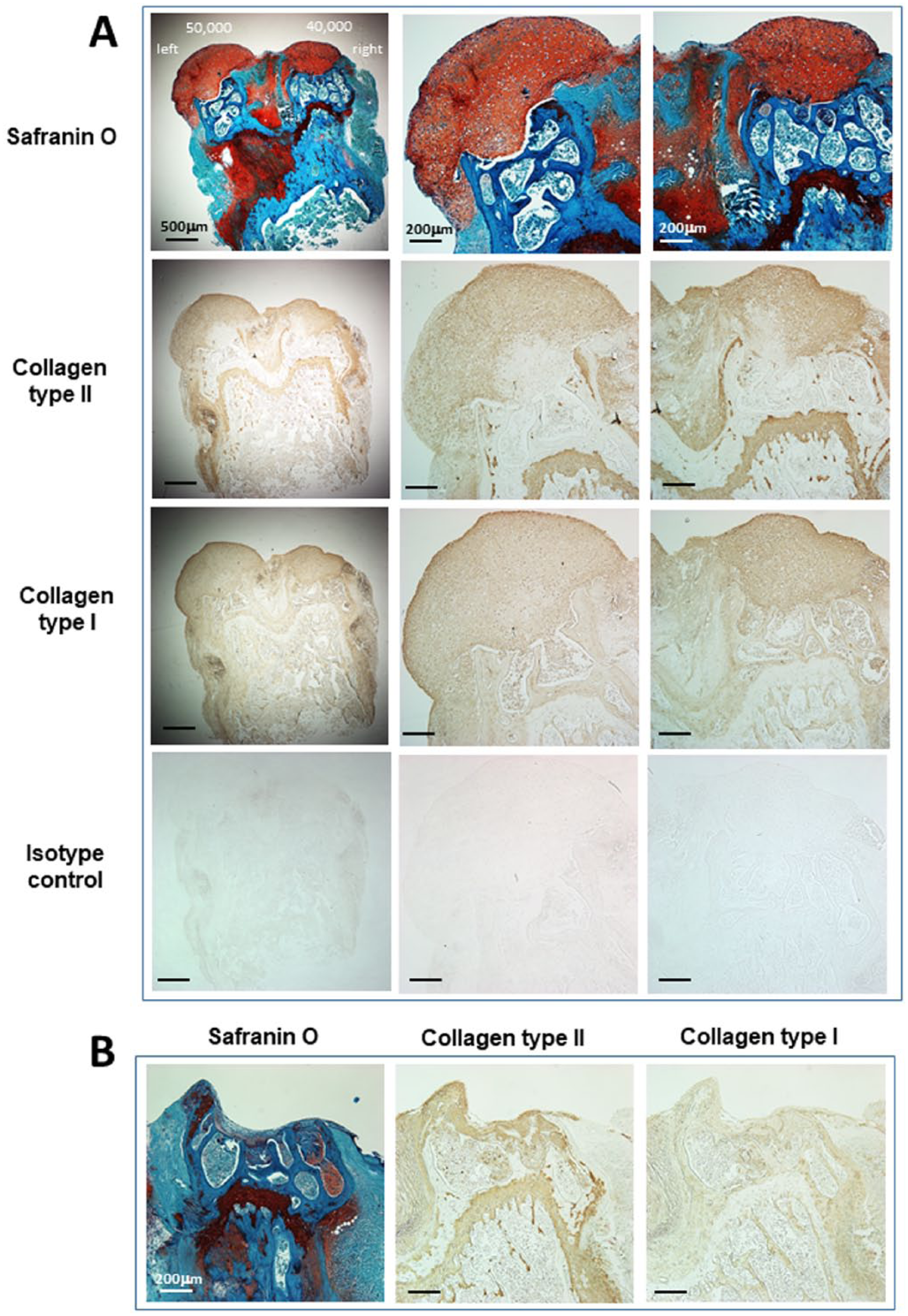
(
Discussion
We showed feasibility of creating
Hydrogels are commonly used in cartilage tissue engineering to suspend the cells and to provide structural integrity. Given our interest in scaffold-free tissue engineering, we decided to analyze repair using cell pellets without hydrogels. We found that 40,000 cells formed a pellet of appropriate size within 3 to 4 days in culture to fit the 0.5-mm diameter defect. Histology of repair tissue generated by implanted pellets revealed a rich glycosaminoglycan-positive neotissue that completely filled the defect. Larger pellets (50,000 cells) generated neotissue that extended beyond the surgical defect and covered the full width of the femoral condyle, which opens the potential for regenerating an entire condyle. However, lower cell densities may be appropriate if cells are to be suspended in hydrogels or other scaffolds. Longer-term culture, beyond 3 weeks, may reveal evidence of tissue remodeling into the characteristic zones of articular cartilage. The potential to create even larger defects may enable the
Our primary objective was not to prove regenerative potential for IPFP cells but for proof of concept of an
One weakness of
In summary, this
Footnotes
Acknowledgments and Funding
The authors thank Merissa Olmer, Peter Gracheck, and Nick Glembotski for technical support; Dr. Nico Verdonschot for constructive comments on this article; and Judy Blake for manuscript formatting and copyediting. Funding was provided by NIH/NIAMS P01 AG007996, CIRM PC1-08128, and by the Shaffer Family Foundation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from the Scripps Institutional Review Board, Protocol No. IRB-14-6320.
Animal Welfare
Ethical Approval for this study was obtained from The Scripps Research Institute, Institutional Animal Care and Use Committee (IACUC), Protocol No. 09-0130.
