Abstract
Objective
The purpose of this narrative review is to summarize what is currently known about the structural, chemical, and mechanical properties of cartilage-bone interfaces, which provide tissue integrity across a bimaterial interface of 2 very different structural materials. Maintaining these mechanical interfaces is a key factor for normal bone growth and articular cartilage function and maintenance.
Materials and Methods
A comprehensive search was conducted using Google Scholar and PubMed/Medline with a specific focus on the growth plate cartilage–subchondral bone interface. All original articles, reviews in journals, and book chapters were considered. Following a review of the overall structural and functional characteristics of the physis, the literature on histological studies of both articular and growth plate chondro-osseous junctions is briefly reviewed. Next the literature on biochemical properties of these interfaces is reviewed, specifically the literature on elemental analyses across the cartilage–subchondral bone junctions. The literature on biomechanical studies of these junctions at the articular and physeal interfaces is also reviewed and compared.
Results
Unlike the interface between articular cartilage and bone, growth plate cartilage has 2 chondro-osseous junctions. The reserve zone of the mature growth plate is intimately connected to a plate of subchondral bone on the epiphyseal side. This interface resembles that between the subchondral bone and articular cartilage, although much less is known about its makeup and formation.
Conclusion
There is a notably paucity of information available on the structural and mechanical properties of reserve zone–subchondral epiphyseal bone interface. This review reveals that further studies are needed on the microstructural and mechanical properties of chondro-osseous junction with the reserve zone.
Keywords
Introduction
The interface between cartilage and bone is a unique region, which provides tissue integrity across a bimaterial interface of 2 very different structural materials. Maintaining the interface at the chondro-osseous junction is a key factor for normal bone growth and articular cartilage function and maintenance. Unlike the interface between articular cartilage and bone, growth plate cartilage has 2 chondro-osseous junctions. The reserve zone of the mature growth plate is intimately connected to a plate of subchondral bone on the epiphyseal side. This interface resembles that between the subchondral bone and articular cartilage, although much less is known about its makeup and formation. On the other side of the growth plate, the hypertrophic zone attaches to the metaphysis through calcified cartilage, which is formed by endochondral ossification. Although endochondral bone growth occurs at the metaphyseal side of the growth plate, there is evidence of a slower growth front at the interface between the reserve zone and the subchondral bone plate of the epiphysis. The existence of such a growth front may help explain the mechanism of physeal mammillary process development, which creates matching involutions at both cartilage-bone junctions. Although there have been several publications on the elemental distribution and biomechanical properties of the chondro-osseous junction of articular cartilage, there is surprisingly little information on the biochemical, morphology, and biomechanical parameters of growth plate–subchondral bone interface.
This review article focuses on those aspects of the growth plate reserve zone–subchondral bone junction that relate to the development and adaptation of this interface during bone growth. Following a review of the overall structural and functional characteristics of the physis, the literature on histological studies of the articular cartilage–subchondral bone interface is briefly reviewed, followed by histological studies on the growth plate cartilage–subchondral bone interface. Next, the literature on the biochemical properties of these interfaces is reviewed, specifically the literature on elemental analyses across the cartilage–subchondral bone junctions. Finally, the literature on biomechanical studies of these junctions at the articular and physeal interfaces is reviewed and compared.
Growth Plate Zonal Structure and Function
The growth plate is a thin layer of hyaline cartilage, which separates epiphyseal bone from the metaphysis at the end of each long bone and provides bone growth during skeletal development. The growth plate consists of at least 3 major zones: reserve zone, proliferative zone, and hypertrophic zone. Figure 1 shows these histological zones of the growth plate for a yearling bovine cow. In all zones, chondrocytes are enveloped by a thin layer of the pericellular matrix (PCM) consisting of proteoglycans and acting as a diffusion barrier for large molecules. 1 The PCM is characterized by type VI collagen. Several studies suggested that the PCM seems to function as a mechano-transducer and transitional zone between cells and the extracellular matrix (ECM), which converts mechanical signals from and to chondrocytes.1-5 Primary cilia on the surface of the cell can also act as mechanosensory organelles, which transduce mechanical forces into biological signals.5,6 Understanding the sequence of events by which cells and ECM communicate and transduce biomechanical and biochemical signals is still providing a challenge to investigators.7,8
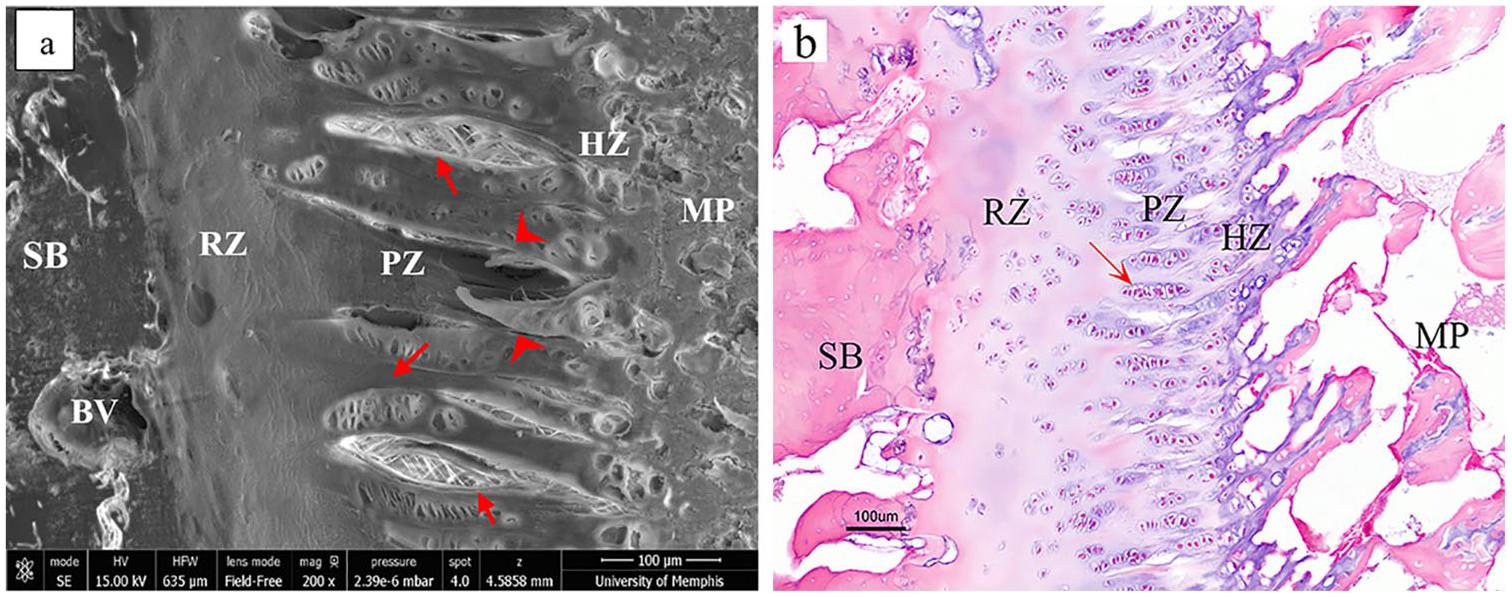
Growth plate images of a bovine (12- to 18-month) growth plate: (
Reserve Zone Structure and Function
Many terms have been used for the reserve zone, such as the germinal layer, resting zone, stem-cell zone, and small-size-cartilage-cells zone.9-12 Reserve zone chondrocytes are scattered irregularly within the ECM. Although the exact function of the reserve zone cells is not quite clear, some potential roles have been postulated.
First, the higher lipid concentration in chondrocyte vacuoles supports the idea that reserve zone cells might store nutrients for later nutritional requirements.10,11,13,14 Early studies mentioned that these cells do not have any proliferative capacity and they are sometimes called undifferentiated cells.10,11,15 Later authors reported that these cells might have the capability to generate new chondrocytes. 16 Cells in the reserve zone may be linked to stem cells, which have the proliferative capacity and can generate clones of chondrocytes for the proliferative zone. Therefore, one of the postulated functions of the reserve zone is to provide a pool of stem-like cells.12,16,17 However, not all chondrocytes in the reserve zone can function as stem cells. 18 The average reserve zone chondrocyte cycle time for 35-day-old rats was measured as 4 to 5 days, which shows reserve zone stem cells rarely divide or proliferate and have a slow proliferation rate, which is one of the characteristics of stem cells.12,19
Second, chondrocytes in the reserve zone also secrete a morphogen that causes the chondrocyte proliferative clones to align to form a chondron by an as yet unknown mechanism.16,18 The reserve zone can be divided into two parts; reserve cartilage, closer to the proliferative zone with flatted chondrocytes, and epiphyseal cartilage, nearer to the epiphyseal bone and chondro-osseous junction with round/elliptical cells. 16 However, most studies have reported the reserve zone cells as being spherical and round.10,11,20,21 With increasing age and decreasing growth rate, the reserve zone chondrocytes become flatter. 14 Reserve zone chondrocytes are randomly distributed as single or paired cells,9,11-13,15 and have about the same dimensions as chondrocytes in the proliferative zone.10,11
Third, chondrocytes may release hypertrophy-inhibiting morphogens to prevents premature hypertrophy of proliferative cells. Therefore, proliferative chondrocytes near the reserve zone cannot differentiate to hypertrophic cells, while toward metaphyseal end of the chondrons, cells not exposed to such factors enlarge and become hypertrophic cells. 18
Proliferative Zone Structure and Function
Chondrocytes and their surrounding PCM and territorial matrix were called chondrons by Benninghof,22,23 who described chondrons as being the basic anatomic and functional units of cartilage in analogy to osteons, which are the basic functional units of Haversian bone. In this context, we also refer to the columns of chondrocytes in the growth plate that consist of the proliferative zone,1,3,7 zone of maturation, and hypertrophic zone as the growth plate chondrons. The chondrons are clustered around nutrient arteries that pass through the subchondral bone plate and reserve zone and end at the base of the proliferative zone of a cluster of chondron columns. It has been noted that after uniaxial tensile testing to failure of isolated growth plate samples, failure occurred within the proliferative zone/reserve zone interface with clusters of chondrons torn out of the reserve zone, but otherwise remaining intact on the side of the metaphysis after separation from the epiphyseal end of the sample; chondrons remained joined to each other in clusters appearing to converge at the reserve zone junction around the source of the vascular supply, 24 suggesting that this connection, as a unit of chondrons with a central blood supply, is relatively strong. This can be seen in the microradiography studies of rabbit (see figs. 11 and 14 in Morgan 25 ) and rat (see plate I, figs. 4 and 5 in Irving 26 ) growth plates and in the freeze-fracture studies of the authors’ laboratory ( Fig. 2 ). Morgan 25 described blood vessels penetrating the subchondral bone plate through openings at the summit of from 8 to 12 columns of proliferating epiphysial cartilage cells, “the rounded tops of the columns inclining toward the central aperture in the calcified zone.” In mammals, the small arteries from the epiphyseal side do not penetrate beyond the proliferative zone, making proliferative and hypertrophic zones dependent on diffusion.9,10,15,27,28 Two main functions of the proliferative zone are matrix production (proteoglycan and collagen) and cell proliferation (chondrocytes), which together with the hypertrophic zone contribute to longitudinal bone growth.9,10,15,18
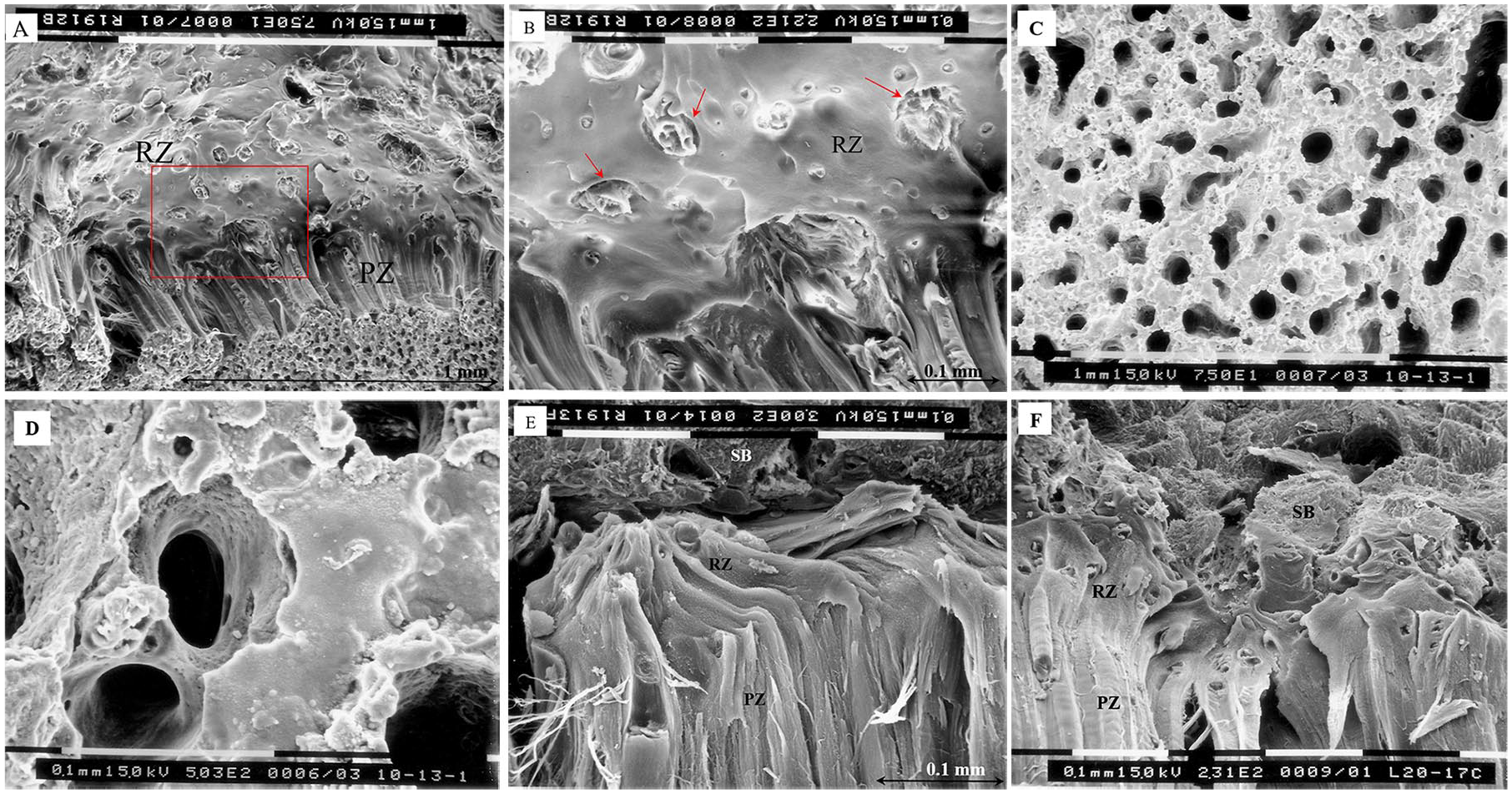
Scanning electron microscopy (SEM; Philips SEM 515) images of a yearling bovine proximal tibial growth plate. (
Chondrocytes migrate from the reserve zone and arrange themselves to align with the longitudinal chondrons.29,30 The flattened chondrocytes near the reserve zone–proliferative zone interface, at the base of the chondron are sometimes called mother cells,9,15,29 which can ultimately give rise to approximately 30 hypertrophic chondrocytes. 17 The linear growth rate of the growth plate is proportional to the rate of daily cell proliferation (cells/day) in the proliferative zone of the mother cells multiplied by the average height of mature chondrocytes (µm/cell) in the hypertrophic zone.10,13,31 The average number of newly formed chondrocytes in each chondron is about 5 per day in the rat model (6-8 weeks old). 32 Another study found the daily rate of cellular turnover in hypertrophic zone to be about 8 cells, which was calculated by dividing daily longitudinal growth by the mean height of hypertrophic cells. 33 This means that every 3 hours, a chondrocyte will be replaced at the end of the hypertrophic zone. The average cell cycle time or chondrocyte replication rate for the proliferative zone was reported to be about 48 hours in 35-day-old-rats, compared with 20 days in 5- to 8-year-old children. 12 These values may be factors of age and the animal species.
Hypertrophic Zone Structure and Function
The hypertrophic zone plays a dominant role in endochondral ossification. Hypertrophic chondrocytes are formed by the final differentiation of the proliferative cells. As chondrocytes progress from flattened cells in the upper proliferative zone close to the reserve zone, they begin to enlarge eventually reaching diameters up to 5 times greater (~50 µm in diameter) in the hypertrophic zone as reported in some studies, including birds.10,34 Other studies have shown that the horizontal and vertical diameters of the cell increase by a factor of 1.6 and 4, respectively, in rats,33,35 which results in an increased cell/matrix ratio in the hypertrophic zone. In contrast, confocal microscope studies of 4-week-old pigs show a higher cell/matrix volume ratio in the proliferative zone compared with the hypertrophic zone. It was measured to be around 11% to 13% in the reserve zone, 17% in the proliferative zone, and 15% in the hypertrophic zone.20,21 These parameters may vary with the species, the age of the animal and perhaps with specimen preparation and measurement technique (2D or 3D). 36 Comparing these values with our histology images of a bovine yearling ( Fig. 1 ) and early pubertal human growth plate (see fig. 2 in Duesterdieck-Zellmer et al. 37 ) in which there are larger cells within a lower matrix volume in the hypertrophic zone, we expect to have higher cell/matrix volume ratio in the hypertrophic zone than the 2 other zones. In addition to the large changes in size and shape, the terminally differentiated cells undergo a change in phenotype. 18 Hypertrophic cells can either undergo cell death through apoptosis or, as some have suggested, transform into osteoblasts through trans-differentiation, which reprograms cells to change their phenotype. Hypertrophic cell differentiation is regulated by the cell’s microenvironment, local factors like matrix metalloproteinases (MMPs), Wnt/β-catenin and the Ihh/PTHrP signaling pathway.3,37-39 Trans-differentiation is a mechanism to generate osteoblasts as osteoprogenitor cells by an unknown mechanism while apoptosis is a mechanism to permit tissue remodeling.18,39,40 A recent study proposed a model in which chondrocytes transform to osteoblasts directly. 41 The main function of the hypertrophic zone is to prepare the matrix for calcification by depositing calcium phosphate mineral in the matrix. 42
Cartilage calcification occurs as the extracellular matrix volume decreases while the relative volume of chondrocytes gradually increases. At the initiation site of mineralization, the concentration of proteoglycan, type II collagen, alkaline phosphatase, and hyaluronic acid are at a maximum.3,43 The concentration of type II collagen might be a function of its binding to hydroxyapatite. 43 However, by the end of the calcification process, proteoglycan and type II collagen have been split to form type X collagen and release hyaluronic acid. MMP-13 degrades type II collagen and contributes to the production of type X collagen (COL10A1: collagen gene) from type II collagen (CoL2a1: collagen gene). Then type X collagen is synthesized in the hypertrophic zone and is regulated by Runx2 factor.3,43,44
Growth Plate Mammillary Processes
Growth plate mammillary processes are the undulations of cartilage-bone interfaces, which form an interlocking interface of hills and valleys that continue to evolve as the bone grows in length and circumference ( Fig. 3 ). These can be considered at various levels: primary, secondary, and tertiary, depending on the relative size of the processes in relation to the epiphyseal dimension. On the metaphyseal side, the mammillary processes are formed by endochondral ossification. On the epiphyseal side, mammillary undulations closely match the 3D topographical pattern of the metaphyseal side forming a mechanical interlocking interface.45-47 After the secondary center of ossification ceases to form bone and leaves a thickened “endplate” of subchondral bone at both the articular and growth plate interfaces, the undulations along the interface with the growth plate reserve zone continue to evolve to match those on the metaphyseal side. The mechanism by which the epiphyseal side achieves this is unknown and will require future study. These 2 chondro-osseous interfaces play a dominant role in the structural integrity of the tissue and successful attachment of the soft and hard tissues and may have relevance to conditions such as slipped capital femoral epiphysis and cam morphology. 48 Therefore, further histological, biochemical, and biomechanical studies of the chondro-osseous junctions are warranted.
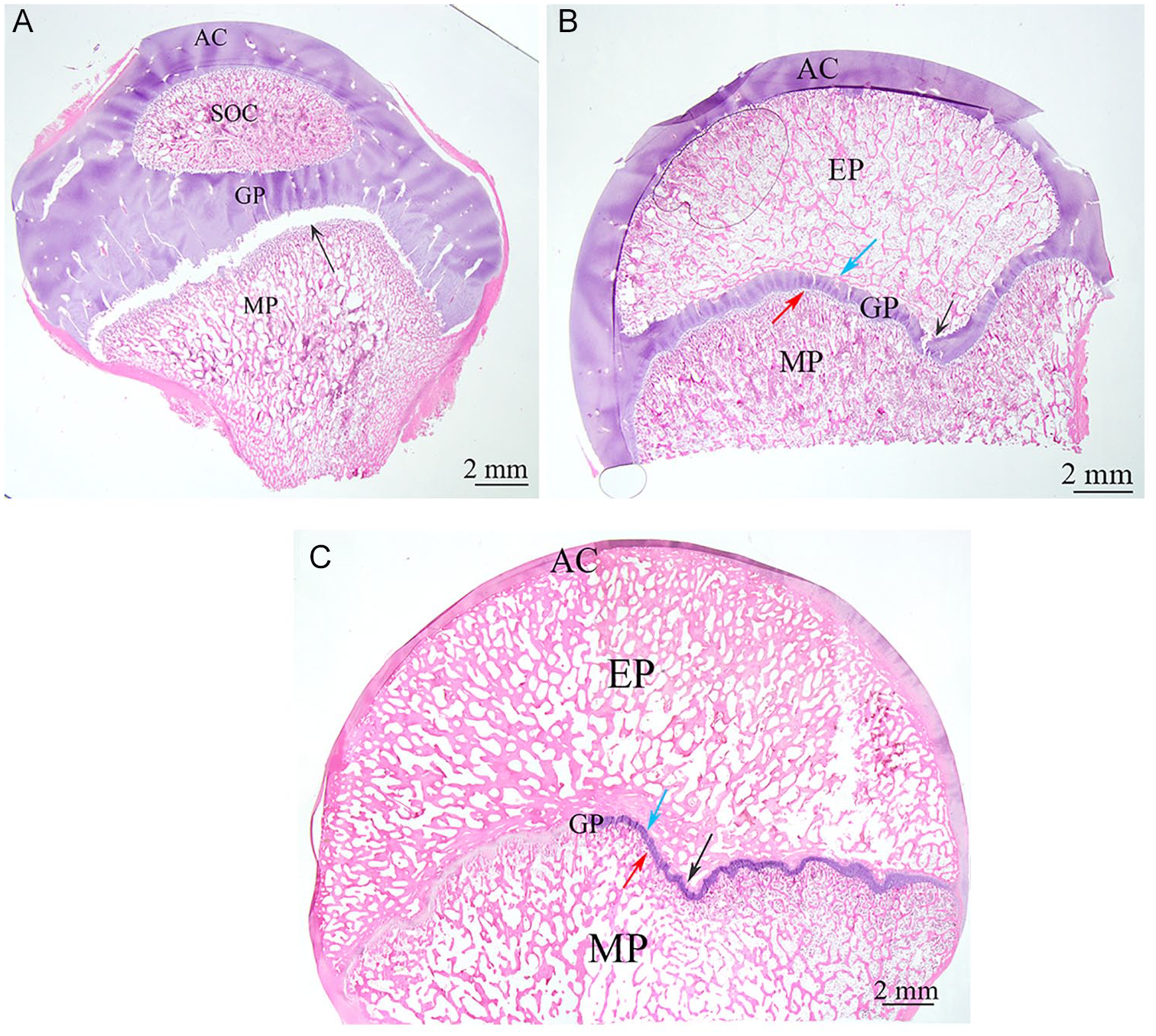
Stereo-microscopy (Olympus, SZX16, Japan) sections cut in the same middle plane relative to pig femoral heads for 3 different age groups, which show the development of mammillary processes with aging. (A) 20-day-old. (B) 35-day-old. (C) 480-day-old. AC, articular cartilage; GP, growth plate cartilage; EP, epiphysis; MP, metaphysis. Black arrows show the undulation of the tubercle in the femoral head, red arrows indicate the metaphyseal secondary mammillary processes, and blue arrows point to the epiphyseal secondary mammillary processes. The scale bar is 2 mm. From the authors’ laboratory.
As the structural properties of the growth plate chondro-osseous junction, specifically the interface on the epiphysis side, are still mostly unexplored, the purpose of this study is to focus on those features. Experimental observations show that there are some structural similarities between the chondro-osseous junction of articular cartilage and that of the growth plate reserve zone with the epiphysis. Although the chemical and mechanical features of the articular cartilage–subchondral bone interface have been studied, almost no attention has been paid to the growth plate–subchondral bone interface. Chemical and mechanical studies of this interface may elucidate the mechanism of the successful attachment of these 2 dissimilar tissues of bone and cartilage and reveal more information related to mammillary process development. To develop a more complete understanding of these cartilage-bone interfaces, which evolve over time, we need more studies that examine the development and evolution of these interfaces during growth, aging, and disease.
Chondro-osseous Junction Structure
Histological studies have revealed details of the collagenous network and orientation and cell shape in the tissue at the chondro-osseous interface. The structural integrity between articular cartilage as a soft tissue and the stiffer calcified cartilage is aided by collagen fibers that cross the tidemark. 49 The anisotropic architecture of collagen fibers within the cartilage matrix has a crucial role in cell protection and biomechanical strength and anchors the articular cartilage into the calcified base.50,51 In the hypertrophic and proliferative zones, collagen fibers are mostly oriented longitudinally.1,52 Collagenous fibers are said to be arranged to give the appearance of arches and make a honeycomb-like network at the base of the proliferative zone, near the reserve zone of growth plate cartilage, which is similar to what has been noted for the superficial zone of articular cartilage.51,53 At the interface of the reserve zone and the epiphyseal subchondral bone plate, collagen fibers orient transversely (radially) and in some parts obliquely. This structure is said to be similar to that in subchondral bone–articular cartilage interface.50,51
Structure of Articular Chondro-osseous Junction
In articular cartilage, the chondro-osseous junction includes the noncalcified cartilage, tidemark, calcified cartilage, cement line, and subchondral bone plate.54-56 The tidemark is a strong hematoxylin-staining border between avascular noncalcified cartilage and vascular calcified cartilage, which forms as a result of an advancing ossification front. Calcified cartilage is a distinct mineralized layer intermediate between compliant articular cartilage and its stiff substrate of the bone plate. It provides a transition zone between soft and hard tissue, which facilitates load transfer at the interface by its intermediate Young’s modulus.57,58 It has been reported that calcified cartilage is 10 to 100 times stiffer than articular cartilage and 10 times softer than bone. 59 The upper boundary on one side is demarcated by the tidemark and on the other boundary by the cement line, which is at the interface between calcified cartilage and lamellar bone.60,61 The tidemark was first named and noted by Fawns and Landells, 61 who described it as a zone of weakness in comparison with the interface between bone and cartilage which is a “double layer of a stain-free line and a granular layer of polysaccharide” somewhat, resembling the tidemark, but without its weakness and never duplicated. It has been shown that there is an increased alignment in collagen fiber orientation, predominantly perpendicular to the tidemark, and an increase in packing density through the tidemark toward calcified cartilage, which provides a smoother transfer of stress from articular cartilage with type II collagen to rigid calcified cartilage with type X collagen.49,62-64 The dynamic structure and irregular geometry of the tidemark provide shearing resistance of the interface and it is frequently duplicated, both as a normal process of aging as well as due to cartilage pathologies.54,62,65 Therefore, the appearance of multiple tidemarks or tidemark duplication is not necessarily a significant identification for joint disorders such as osteoarthritis (OA)66-68 and can be seen in normal tissue ( Fig. 4 ).
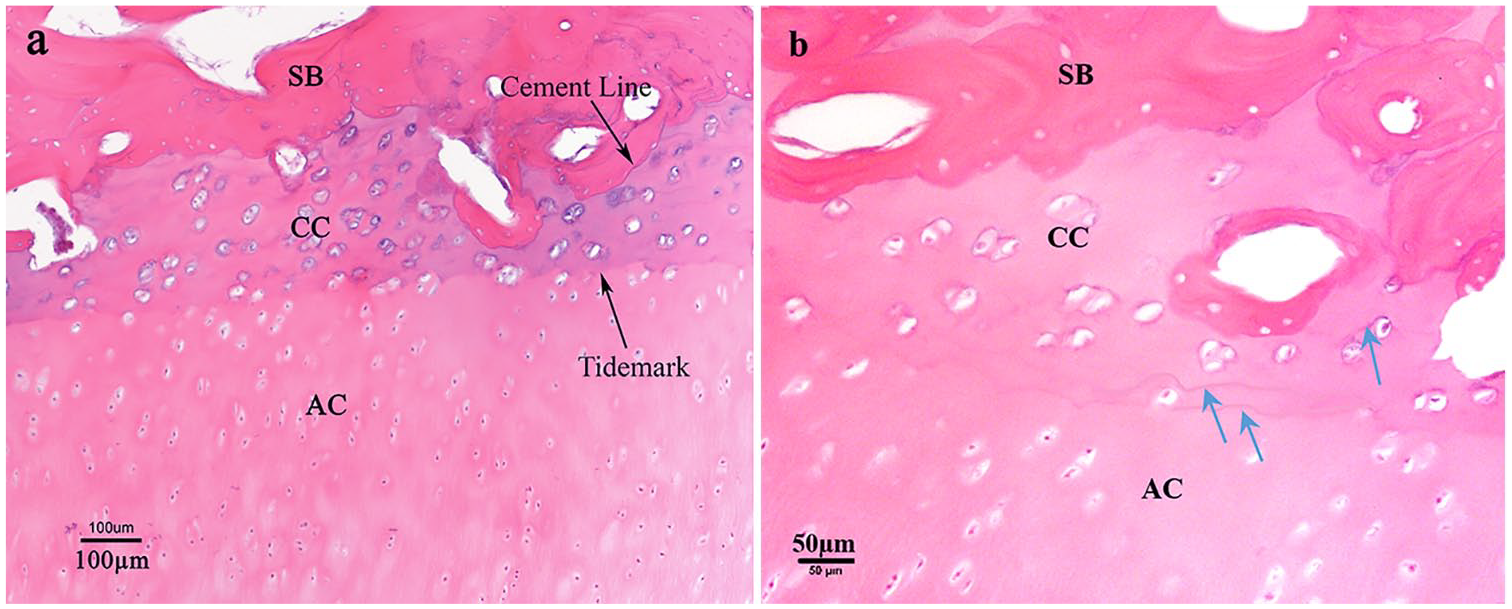
Articular cartilage chondro-osseous junction of a bovine yearling (12-18 months), stained with hematoxylin and eosin (H&E); blue arrows show multiple tidemark lines. SB, subchondral bone plate; CC, calcified cartilage; AC, articular cartilage. From the authors’ laboratory.
In normal tissue, tidemark duplication represents an advance of the calcification front and can be the result of metabolic activities. With cartilage degradation and disease like OA, tidemark duplication also occurs in which the tidemarks become more distinct and are duplicated between chondrocyte lacunae. 59 Cartilage lesions cause the formation of bony spicules with a central canal, originating from Haversian canals within the subchondral bone, which advances calcified cartilage. 69 The higher density of these spicules in OA makes the cement line more irregular and decreases the calcified cartilage thickness by gradual replacement with new less structurally organized Haversian canals.59,69 The tidemark thickness varies from 2 to 10 µm.54,66 The interface surface roughness values (Rs, which is the unitless surface roughness parameter) for the articular calcified cartilage/subchondral bone tidemark and cement line of the normal human femoral condyle were measured to be 1.14 ± 0.04 and 1.99 ± 0.38, respectively. 70 The thickness and shape of the tidemark are related to the loading sustained. Load-bearing areas have thicker tidemarks with more undulations to provide more stability and resistance to shear forces during articulation. 68
There are different definitions for the structure referred to as the subchondral bone plate. Duncan et al. 71 defined it as a region, which separates articular cartilage from bone marrow cavities including the calcified cartilage region. In the current review article, we use the term subchondral bone to refer to the cortical bone-like endplate, lying adjacent to the calcified cartilage. The subchondral bone plate thickness varies in the range of 0.1 to 1.5 mm, with numerous intercommunication spaces, which provide a direct connection between the noncalcified cartilage and the marrow cavity of the trabecular bone.72,73 Bone plate composition, thickness, and perfusion vary with the region of the joint surface. The thickness is determined by joint function, the stress distribution, and changes with the joint disease. 73 For example, OA results in subchondral bone thickening and increases its porosity. 74 Clark et al. 72 showed that the subchondral bone plate is thicker in the center of the adult tibial plateau and a thinner on the lateral side; however, Mila et al. 73 reported a uniform thickness of subchondral bone for the same region.
A 3D reconstruction of samples from histological serial sections of a human knee joint revealed that noncalcified cartilage islands pass beyond the tidemark interface and go through calcified cartilage to reach into the bone marrow. 54 This “invagination” by noncalcified cartilage islands could also provide a nutritional pathway for articular cartilage and allow crosstalk within the chondro-osseous junction. 54 It has been hypothesized that subchondral bone may also protect articular cartilage through endogenous bone-mediated factors for cartilage chondrocyte survival. 75 In explant experiments in which articular cartilage was cultured without subchondral bone, most chondrocytes did not survive, whereas they survived in samples in which the subchondral bone remained attached. 75
Structure of Growth Plate Chondro-osseous Junction
After the growth plate cartilage–subchondral bone plate unit has formed following completion of growth by the secondary center of ossification, the growth plate cartilage is protected with a dense subchondral bone plate, bordering on the reserve zone. 76 The formation of subchondral bone may begin with the mineralization of cartilage on the reserve zone side through a mechanism, which is not completely elucidated. There is no tidemark formation at the metaphyseal growth plate cartilage–bone interface where the hypertrophic zone merges into the zone of provisional calcification; however, there is evidence demonstrating the presence of a tidemark at the reserve zone–subchondral bone interface.76,78 The formation of this tidemark would be the result of cartilage calcification.67,77 Therefore, in addition to an endochondral ossification front on the metaphysis side, which is responsible for longitudinal bone growth, there is a second ossification front on the epiphysis side, which might be responsible for subchondral bone plate formation 76 and modeling of the mammillary processes on the epiphyseal border.
The final stages of growth involve the process of epiphyseal fusion, which was described by Parfitt 79 as the formation of 2 bony plates following the cessation of growth. One bony plate is what we refer to in this review as the subchondral bone plate which arises first and lies at the border of the reserve zone; at a later point in time a second bony plate forms at the border of the hypertrophic zone “arising from compaction of new trabeculae that are lying horizontally rather than vertically.” 79 The time between the formation of the first and second bone plates may vary with species and skeletal sites. In the bovine proximal tibial epiphysis, we have observed the presence of the subchondral bone plate at the reserve zone as early as 4 to 5 months and the absence of formation of the second plate at the metaphyseal border even as late as 12 to 18 months. The age of epiphyseal union varies for different species. Even for the same species, the epiphyseal union order is not the same for all long bones. 80 It is known that fusion occurs earlier in females 81 and is influenced by hormones. Estrogen is the principal hormone stimulating growth in puberty for both sexes; however, in late adolescence, estrogen promotes growth plate fusion.36,80,82,83 Lower levels of estrogen stimulate bone growth, while higher levels promote epiphyseal fusion. 83
Haines 78 found small cartilage nodules close to the subchondral bone, including closely packed cells in a young and still growing dog model following completion of the epiphyseal growth from the secondary center. These cartilage nodules were located between the bone marrow and subchondral epiphyseal bone and suggest the formation of new cartilage from bone marrow. 78 Similar evidence can be seen in histology studies of the authors’ laboratory for a yearling bovine calf ( Fig. 5 ), in which there are some neocartilage cells in the reserve zone–subchondral bone interface. Neocartilage cells, proposed by Haines, 78 seem to be larger than chondrocytes existing in the reserve zone. They are closely packed in a basophilic matrix, which suggests the formation of new cartilage from epiphyseal bone marrow that might participate in subsequent epiphyseal subchondral bone formation and further development of mamillary processes. Their basophilic matrix suggests that these cells are differentiating. The neocartilage cells may be chondroblast or osteoblast precursor cells.
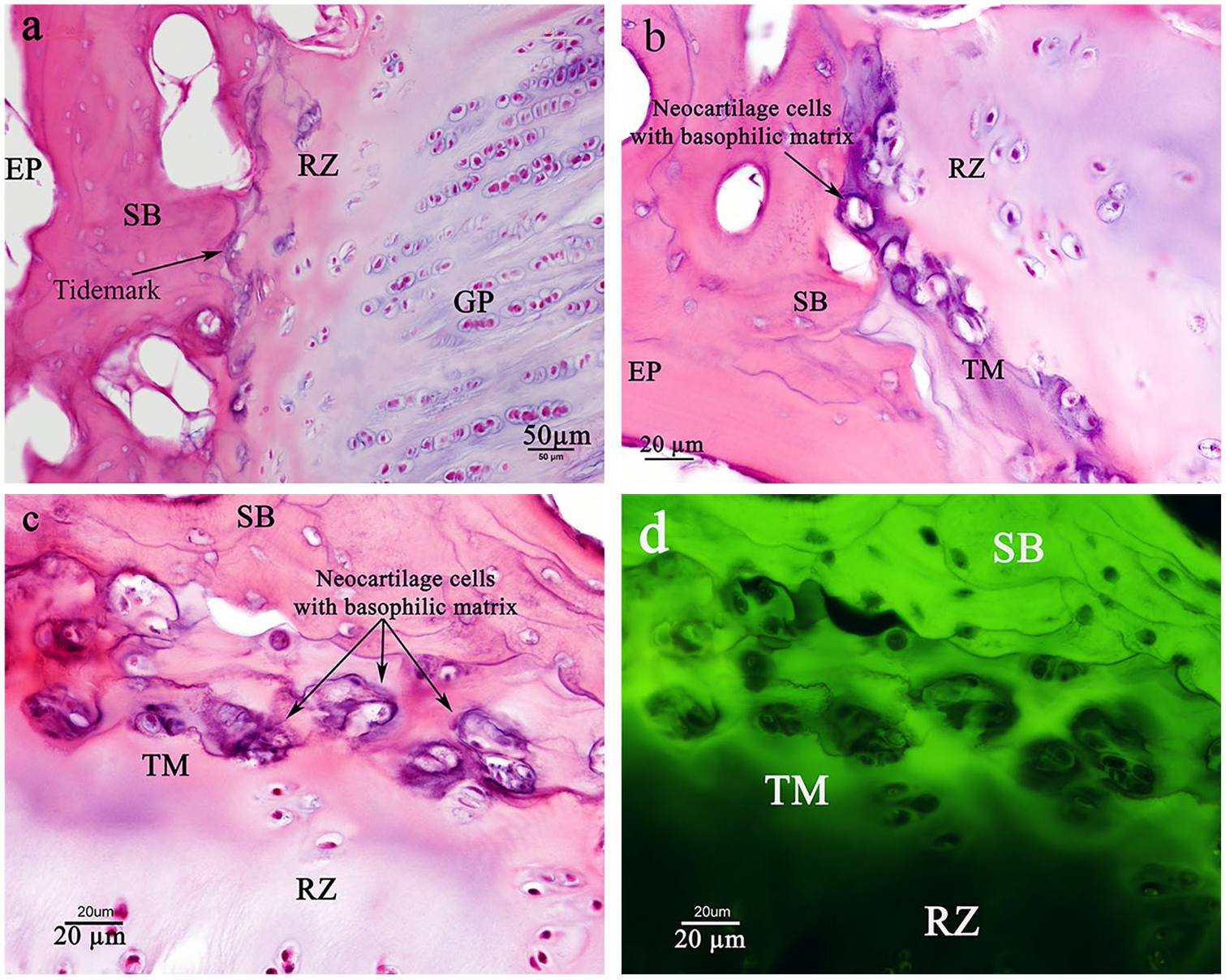
(
At the earlier developmental stage, before the formation of the subchondral bone plate when the secondary center of ossification (SCO) is still developing, there are larger cells in the reserve zone–epiphyseal bone interface of the SCO. These large cells are hypertrophic cells related to the growth of the SCO, which are similar to those at the metaphyseal bone border of the primary center of ossification. Farnum and Wilsman 40 found such cells near the reserve zone–epiphyseal bone interface in their histology study of 4-week-old Yucatan pig (see fig. 1a in Farnum and Wilsman 40 ). A narrow-calcified cartilage plate near the epiphyseal bone plate, which sometimes included a few hypertrophic cells, was also reported by Hansson et al. 13 Candela et al. 19 used immunohistochemistry labeling by thymidine analog 5-ethynyl-2′-deoxyuridine (EdU) to trace the “slow cycle” cells within the articular and growth plate cartilage of mice. EdU-labeled cells were found in a narrow region within the superficial and reserve zone of articular and growth plate cartilage, respectively. These slow cycle cells are mostly stem or progenitor cells, which are able to differentiate to provide new chondrocytes. 19
Alkaline phosphate (ALP) localization within different zones of articular and growth plate cartilage has been used to reveal bone formation sites. 84 In articular cartilage, ALP is localized in the matrix of the deep zone and mature chondrocytes. Histology images of the growth plate (see fig. 2c and d in Miao and Scutt 84 ) showed an increase in the ALP secretion from the proliferative to hypertrophic zones reaching the highest values in the hypertrophic zone. In these images ALP can also be seen to be localized in the reserve zone–subchondral bone interface, which could be a sign of bone formation, 84 Furthermore, an earlier study had reported finding a layer of calcified cartilage between the bone plate and reserve zone. 9 These aforementioned studies and observations show that there is a likely possibility for bone formation at the reserve zone–subchondral bone plate interface that could explain the mechanism of subchondral bone plate formation and mammillary process development. This hypothesis requires more studies and experiments to be fully explored.
Chondro-osseous Junctions’ Chemical Properties
Elemental Distribution across the Articular Chondro-osseous Junction
Bone has a dynamic structure that remodels continuously. Cartilage and bone structural and biomechanical properties are determined by compositional and biochemical variables. Alterations in biochemical properties in the cartilage zones may in part account for the varying zonal mechanical properties.85,86 The measurement of the elemental and ion distributions in healthy and nonhealthy tissue can be used as a diagnostic tool in clinical studies. One example is the evaluation of the chemical composition of calcified cartilage and subchondral bone in studies of degenerative joint disease or OA. There is some evidence that early OA is accompanied by alterations in elemental composition. 2 Pathological changes in the major elemental distributions and the balance of the cartilage components may contribute to structural alteration in the collagen network, which can change mechanical properties and subsequently initiate cartilage degradation. 87 For instance, the local concentration of Pb as a toxic metal in the skeleton targets osteoblast and osteocyte activities, inhibits fracture healing, and results in osteopenia. Therefore, tracing Pb is of considerable interest in cartilage pathology. This toxic element mostly accumulates in the transition zone between the non-calcified cartilage and calcified cartilage (tidemark).88,89
Cartilage ECM is composed of fluid and a small percentage of macromolecules, 2 which control ECM turnover. Growth factors and enzymes, including MMPs and ALP have a key role in collagen formation, cartilage degeneration, and bone remodeling. ALP is involved in cartilage mineralization and bone formation and is produced by osteoblasts. ALP is also produced by chondrocytes in the deep zone of articular cartilage and the growth plate hypertrophic chondrocytes. 90 However, to function, these enzymes require bound metal ions of certain elements such as Ca, P, Zn, and K, known as cofactors.2,87-89,92-94 Among all cofactors, the divalent cation, Zn2+, at the active site of the tidemark, plays a fundamental role in the growth and degeneration process of OA.77,88,95-98 ALP is known as a Zn-dependent enzyme. Although Zn is considered as an essential element for normal skeletal growth and is concentrated at the site of new bone formation,96,97 its accumulation within the tidemark and transition zone suggest increased activity of cartilage-degrading enzymes.2,87,94 Accumulations of other cofactors at the bone-cartilage interface also indicate a raised activity of cartilage degrading enzymes. Therefore, elemental features are expected to provide a good estimation of changes in enzymatic activities, occurring in OA.2,94,95
Elemental analysis has demonstrated that Ca and P reach their maximum values at the calcification front to participate in hydroxyapatite crystal formation. In addition to Ca and P, the presence of Zn is also essential for bone calcification. 93 Localization of S in articular cartilage relates to its contribution in matrix proteoglycans, while its level is decreased within bone. 93 Chemical studies of all essential elements in the articular cartilage–subchondral bone plate interface show a gradient in elemental concentration from cartilage to bone, which might provide a mechanical transition zone between the 2 different tissues of cartilage and bone with very different mechanical properties. Calcified cartilage may function as such a transition zone. Elemental studies of the articular cartilage–subchondral bone interface using various techniques are summarized in Table 1 .
Summary of Elemental Analysis Studies of the Articular Cartilage–Subchondral Bone Interface.

OA = osteoarthritis; XRF = synchrotron X-ray fluorescence; SRm-XRF = synchrotron radiation–induced micro X-Ray fluorescence; PIXE = proton-induced X-ray emission; SAXS = small-angle X-ray scattering; RBS = Rutherford backscattering; µ-PIXE = micro-proton-induced X-ray emission; µ-SXRF = micro-synchrotron X-ray fluorescence; PIGE = proton induced gamma-ray emission.
Elemental Distribution within the Growth Plate
In contrast to the aforementioned studies on the elemental analysis of the articular cartilage subchondral bone interface, to our knowledge, there is no such study of the reserve zone–subchondral bone interface. There have been studies on the elemental distribution across the growth plate and close to the metaphyseal bone border. One of the earliest elemental analyses of the growth plate was reported by Boyde and Shapiro. 99 They studied isolated chondrocytes (extracted from each of the growth plate zones) and ECM in different growth plate zones for 4 species; pig, rabbit, rat, and guinea pig. 99 Their results show that the Ca and P content reached their highest values in the cells and ECM of the hypertrophic zone.99-101 The higher concentration of P and Ca in the hypertrophic zone is related to their roles as precursors to phospholipid biosynthesis and hydroxyapatite formation (Ca10(PO4)6(OH)2). 101 During remodeling, septoclasts solubilize the cartilage ECM and the dissolution of proteins in the matrix releases high local levels of ions, peptides, and glycans and secreted matrix metalloproteins. Some of these can act as potent apoptogens of chondrocytes. 39 In addition, the higher concentration of Ca in the hypertrophic zone is associated with extracellular lymph within the cartilage matrix and chondroitin sulfate or other matrix acid mucopolysaccharides. Phosphorus is mostly stored in hydroxyapatite, extracellular lymph, and intracellular organic and inorganic phosphate, and matrix polyphosphates. 102
In comparison with Ca and P, S content decreases in the hypertrophic zone, where chondrocytes no longer synthesize sulfated proteoglycans to provide more space for deposition of hydroxyapatite. Localization of S in noncalcified cartilage zones is needed for matrix proteoglycans negative chains that bind large amounts of water.99,100,103-105 Potassium (K) and chloride (Cl) are present in all cells in different cartilage zones. 99 By evaluating the weight ratio of trace elements to Ca the percentage of Ca, S, K, Cl, Ni, Cu, Zn, and Sr could be determined in each zone of growth plate. 105 In addition to these elements, C, N, O, Na, Mg, Si, Fe, Sn, and Pb have been detected within the ovine scapula growth plate. 92 Higher percentages of C, N, and O in cartilage are related to protein synthesis. 92 Mg and Cl are involved in cell physiology. 106 Comparing the distribution of the main elements of Mg, P, S, Cl, K, Na, and Ca in the proliferative, prehypertrophic, hypertrophic, and calcification zones showed that S and Cl decreased from proliferative zone to the zone of early calcification. Similarly, the value of Na dropped from the proliferative zone to early hypertrophic zone and then remained constant. While Mg and K stayed the same over all zones. 106 Table 2 summarizes the elemental studies on different zones of the growth plate using various techniques.
Summary of Elemental Analysis Studies of the Growth Plate.

EDX = energy dispersive X-ray spectroscopy; SRIXE = synchrotron radiation induced X-ray emission; RZ = resting zone; PZ = proliferative zone; HZ = hypertrophic zone; PHZ = prehypertrophic zone; CZ = calcification zone.
Chondro-osseous Junction Permeability
Cartilage may be viewed as a poroelastic material with a porous solid phase and a mobile fluid phase. Permeability is measured by how fast the fluid phase can move through the solid phase when subjected to a pressure gradient. A tissue with relatively low permeability (resistance to fluid flow) builds up hydrostatic pressure under compressive loading, which helps articular cartilage support joint loads and avoid collapse of the solid matrix and prolongs the time to reach equilibrium. 107 In addition to hydraulic permeability as a material property, to evaluate the fluid flow throughout an irregular structure like the chondro-osseous junction, it is useful to also look at the hydraulic conductance as a structural property of the tissue. 107 Table 3 shows how the main material and structural properties of the articular unit, which affect the fluid flow, change with cartilage degeneration. Fluid flow and solute transport across the cartilage-bone interface are likely to be key factors in cartilage-bone crosstalk for maintaining normal physiology. 108 Cartilage is, in general, an anisotropic and inhomogeneous tissue and its permeability depends on collagen arrangement, fiber orientation, and fiber volume fraction. 109 It has been shown that under compression, the permeability of articular cartilage is different between the axial and transverse direction and it is measured to be greater in the transverse direction.109-113 Fluid exchange across the joint is also influenced by tissue vascular channels, microcracks, porosity, and subchondral bone thickness. The porosity of human articular subchondral bone has been measured to be 5.8% ± 1.8%. 114 The average pore diameters of the articular calcified cartilage and subchondral bone plate have been measured as 10.7 ± 6.5 nm and 39.1 ± 26.2 nm, respectively. 115 In a healthy joint, the articular subchondral bone plate thickness is highly variable relative to the region of the joint surface. The thickness is a function of joint loading and stress distribution. 73
Effects of Articular Cartilage Material and Structural Properties on Fluid Flow With Osteoarthritis Progression.
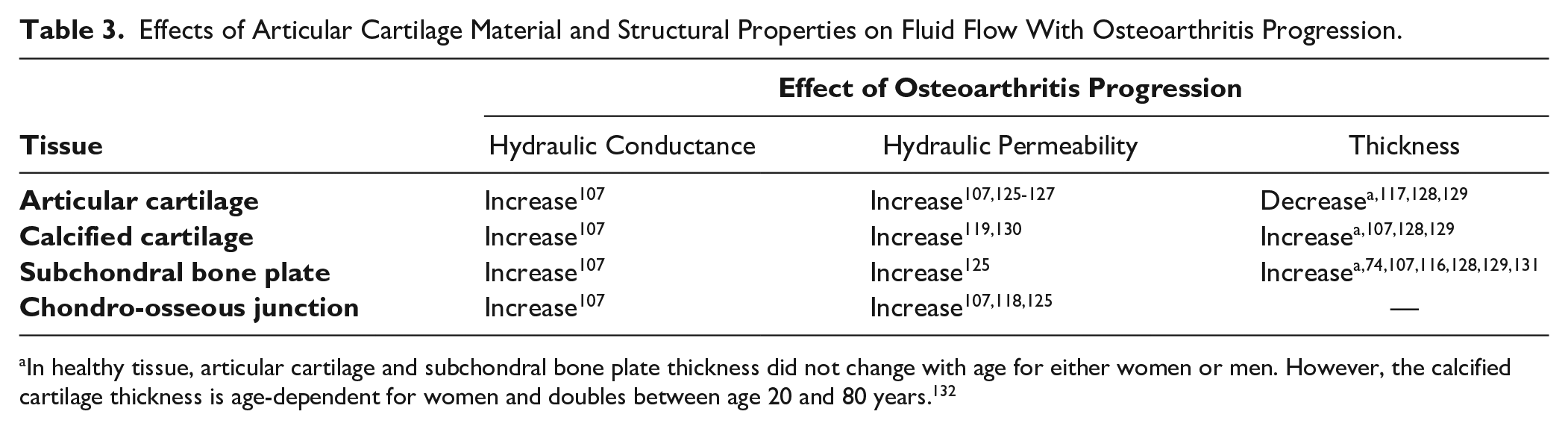
In healthy tissue, articular cartilage and subchondral bone plate thickness did not change with age for either women or men. However, the calcified cartilage thickness is age-dependent for women and doubles between age 20 and 80 years. 132
The subchondral bone plate is a highly vascular tissue. It has cavities of various sizes and shapes. The largest irregular cavities (more than 100 µm wide) relate to marrow containing spaces, while more cylindrical-shaped canals (30-70 µm in diameter) contain marrow cells and blood vessels. The smallest pores (<30 µm) are vascular canals, which hold 1 or 2 blood vessels. 68 The vessels enter calcified cartilage from subchondral bone to provide cartilage nutrition. 116 These arteries contribute to the remodeling of the calcified cartilage and subchondral bone plate and affect tissue permeability. 117 It has been shown that there is an increase in the vessels invading the calcified cartilage and tidemark with OA, which may explain the increase of tissue permeability.116,118,119 Although the mineralization of cartilage in calcified regions acts as a barrier to molecular transport, its nonmineralized patches are considered as molecular pathways for small solutes. 120 The uncalcified cartilage helical patches/prolongations, dipping through the calcified cartilage to subchondral bone marrow spaces, affect chondro-osseous junction permeability and support tissue nutrition. 54 Furthermore, an in vivo study in humans using intravenous injection revealed that gadolinium diethylenetriaminepentaacetic acid (Gd(DTPA)2−) penetrated through the subchondral bone into the articular cartilage. 121 The tidemark and mineralized cartilage are permeable to low-molecular-weight solutes; however, their diffusivity is lower than in uncalcified cartilage.108,122 Depth-dependent cartilage diffusivity could be explained by various glycosaminoglycans distribution, water content, collagen orientation, and other macromolecules through the cartilage.122-124 Molecular transport through the chondro-osseous junction depends on the morphological and microstructural features of calcified cartilage, and subchondral bone.114,122 It has been shown that there is a link between the diffusion behavior of the neutral solute and the micro-architecture of the subchondral plate/calcified cartilage. 114
In contrast to these qualitative and quantitative studies on articular cartilage–subchondral bone permeabilities, little is known about the growth plate cartilage–bone interfaces. Cohen et al. 133 investigated bone-physis-bone permeability using numerical modeling of compressive stress relaxation experiments. They considered 3 different boundary conditions for the chondro-osseous junction permeability; permeable epiphyseal and metaphyseal, permeable metaphysis/impermeable epiphysis and finally, impermeable epiphysis and metaphysis. The best fit to their experimental stress relaxation data was obtained using a permeable metaphysis and impermeable epiphysis boundary condition. Another study investigated the time-dependent volume change of epiphysis–growth plate cartilage–metaphysis interfaces under cyclic compressive loading using magnetic resonance imaging. 134 The results showed that the time constants for physeal cartilage consolidation and recovery were 12% to 30% of the corresponding values for articular cartilage. It was hypothesized that this could be explained by a more permeable cartilage-metaphyseal border since the physeal cartilage–epiphyseal interface is similar to the articular cartilage–epiphyseal interface. It required less time to reach equilibrium and provided less hydrostatic pressure than cyclic compression of the articular cartilage. 134
Chondro-osseous Junctions’ Mechanical Properties
Physiological loading is essential for the maintenance of the composition and structure of normal cartilage. 135 Mechanical loading also modulates processes related to endochondral bone formation, such as mineral deposition and cartilage calcification.42,91 Dynamic loading can also affect solute transport in cartilage extracellular matrix by pumping out the solutes from blood vessels and has a key role in the nutrition of avascular tissues for maintaining their cellular metabolic activities. 136 Many experimental and computational studies have explored the mechanical behavior of growth plate regions at macro- and microscale levels under physiological loads.19,137-142 Although some of the mechanisms of mechano-transduction of the growth plate are known today, there are certainly many others still unrevealed, specifically in the chondro-osseous junction of both articular and growth plate cartilage. Understanding the mechanical and structural features of this junction can give us insights useful for engineering tissue interfaces and help design and develop biomaterial replacements for the treatment of joint disease and lost tissue. 143
The chondro-osseous interface plays a crucial role in maintaining the integrity of the joint and the successful attachment of 2 mechanically dissimilar tissues. In comparison with articular cartilage, the higher impact energy absorbing and greater shock absorbing ability of subchondral bone is an advantage. 144 Higher energy absorption by subchondral bone can protect the overlying cartilage from further damage and in the long term, any microcracks/microfractures resulting from high-stress level can be remodeled because of the greater ability of bone for healing. 144
Physiological loads are transmitted through the junction of cartilage and the comparatively rigid subchondral bone. This material mismatch along the interface of 2 structurally and mechanically different tissues contributes to stresses likely to result in debonding unless mitigated by modifications in the surrounding tissue properties or in the nature of the bond between the dissimilar materials. The bone-cartilage interface is capable of resisting the static and dynamic loads of everyday activities by features that warrant further investigation. 82 In a study comparing the shock-absorbing ability of articular cartilage and subchondral bone, the reported impact-induced microcracks at the cartilage-bone interface under a dynamic load, presented in 3 samples out of 10, may be associated with the shock-absorbing ability mismatch between articular cartilage and subchondral bone. 144 Tensile and shear testing of the growth plate has produced failure planes through different zones of the growth plate, but not at the subchondral bone/reserve zone interface.24,145-147
Mechanical Properties of the Articular Chondro-osseous Junction
Despite the importance of the chondro-osseous junction, there are few published studies on the mechanical properties of the articular cartilage-bone interface.148,149 Ferguson et al. reported values for the human femoral head articular calcified cartilage Young’s modulus of 19.01 ± 3.9 GPa by nanoindentation technique; however, Mente et al. measured it as 0.16 ± 0.09 and 0.58 ± 0.19 GPa for the bovine patella and femur, respectively, using 3-point bending.148,150 Campbell et al. 151 mapped the elastic modulus of articular cartilage across the chondro-osseous interface using atomic force microscopy (AFM) and nanoindentation tests. They revealed that there is a transition zone between the soft tissue and hard tissue with a width of 2.3 ± 1.2 µm for the rabbit femoral head. Gupta et al. 152 evaluated the mechanical properties and mineral content of the cartilage-bone interface to address how the mineralized matrix of articular calcified cartilage remains attached to subchondral bone without debonding. They reported an interface width of 20 to 40 µm using AFM technique, although their results were affected by sample dehydration. As a biphasic tissue, the interaction between the solid and fluid phases gives cartilage its viscous mechanical properties. Dehydration can result in tissue shrinkage, reduced toughness, and increased strength and stiffness. 153 A study of age-related biochemical and biomechanical alterations across the articular cartilage–subchondral bone unit revealed that cartilage thickness decreased with growth, paralleled by a structural condensation of the underlying subchondral bone due to endochondral ossification. 154 Subchondral bone thickness and mineral density increase, and porosity decreases during skeletal development. 155 Table 4 summarizes research studies on the mechanical properties of the articular cartilage–bone interface.
Summary of Studies of Mechanical Properties of the Articular Cartilage–Subchondral Bone Interface During Growth.

AC = articular cartilage; ACC = articular calcified cartilage; SB = subchondral bone plate; E = Young’s modulus; qBSE = quantitative backscattered electron microscopy; AFM = atomic force microscopy.
Mechanical Properties of the Growth Plate Chondro-osseous Junction
Although there have been studies investigating mechano-transduction and characterizing the biochemical properties of the growth plate, we have been unable to find any studies on the mechanical properties of the growth plate–subchondral bone interface, even though it plays a significant role in maintaining growth plate tissue integrity and strength. Most of the research has focused on measuring the biomechanical behavior of the growth plate zones, which are briefly mentioned here. The different zones of the growth plate vary in composition; therefore, each zone behaves mechanically differently under physiological loading conditions. Growth plate cartilage and articular cartilage are similar in that they have a mechanical gradient along their zones, and in both, growth starts with an increase in cell numbers and volumes. 156 The mechanical properties of the zones vary with stage of development and skeletal location. Chondrocytes respond differently depending on the frequency and amplitude and duration of loading. 157 Congdon et al. revealed that articular cartilage and growth plate cartilage respond differently to mechanical stimuli. 158
Using AFM to compare the Young’s modulus of the interterritorial matrix and PCM surrounding the chondrocytes showed that these elastic modulus values were greater for the interterritorial matrix, which has a more solid phase relative to those for the PCM with its more fluid phase. This might be related to their nanostructure, collagen type, and concentration. Growth plate nanostructural and nanomechanical properties may have implications in nutrient diffusion and fluid dynamics, which are vital for cartilage health and function. 159 The indentation-derived Young’s modulus measured in small (2 × 2 µm) areas of the ECM increases from 0.57 MPa in the reserve zone to 1.44 MPa in the zone of calcifying cartilage measured in the longitudinal direction employing AFM. 156 The growth plate is anisotropic with different values for Young’s modulus along longitudinal and transverse directions. 156 An unconfined compression study of 0.4 mm diameter plugs of newborn swine (ulna) growth plate demonstrated 160 that the macroscopic (ECM + cells) Young’s modulus of the reserve zone is double that of the proliferative/hypertrophic zone along the longitudinal direction, in agreement with the study by Cohen et al., 161 while it is 3 times the value in the transverse direction. Tensile properties evaluated by testing 8-week-old rabbit distal radial and ulnar growth plate after excising the perichondrial ring, but otherwise intact, showed that the reserve zone is about 75% stiffer than other 2 zones. 162 These mechanical property values depend on test methods (microindentation vs. macroscopic tension or compression), species, location, and growth plate developmental stage. The macroscopic tensile Young’s modulus is an order of magnitude greater than the compressive Young’s modulus and aggregate modulus. 161
Summary
The chondro-osseous junction in articular cartilage consists of noncalcified cartilage and calcified cartilage layers, a tidemark, and a subchondral bone plate. The mechanical and structural properties of this junction play a key role in skeletal joint diseases like OA. It has been observed that a similar interface exists at the growth plate reserve zone–epiphysis junction, which includes the cartilage reserve zone, calcified cartilage, tidemark region, and subchondral bone plate. Therefore, the growth plate may be considered to consists of 2 ossification fronts, one at the hypertrophic zone–metaphysis interface and another at the reserve zone–subchondral bone epiphysis interface. In this article, we reviewed the biochemical, histological, and mechanical literature on the chondro-osseous junctions of articular cartilage and growth plate cartilage. Our focus was on the reserve zone–subchondral bone region, which has received comparatively less attention than articular cartilage. It is hypothesized that there is a gradual mechanical and chemical transition between the soft reserve zone and the stiff epiphyseal subchondral bone plate, which would enable a stable interface similar to the articular cartilage-subchondral bone interface. In histological preparations of the growth plate, clusters of neocartilage cells have been observed in this transition zone.78,163 These neocartilage cells have not been reported in the tidemark region of articular cartilage. The neocartilage clusters in the reserve zone–epiphysis junction could provide insight into the mechanism of subchondral bone formation. This study highlights a need for an improved understanding of chondro-osseous junction of the growth plate–epiphyseal bone to enable new tissue engineering strategies to be explored for osteochondral defect treatment. The study of the growth plate reserve zone chondro-osseous junction development can also help us understand the mechanism of mammillary process formation and how the undulations on the epiphyseal side continues to match those on the metaphyseal side long after the formation of a compact epiphyseal bone plate.
Footnotes
Acknowledgments and Funding
We thank Dr. Omar Skalli, Dr. Felio Perez, and Lauren Thompson of the University of Memphis Integrated Microscopy Center for their help with histology and electron microscopy. We thank Riley Tusha of Tyson Fresh Meats, Inc. (Dakota Dunes, SD, USA) for providing us with cow bones and Dr. Randal Buddington at the University of Memphis for the piglet bones. We thank Dr. Charles M. Cobb of the University of Missouri at Kansas City School of Dentistry, Kansas City, MO for help with freeze fracture specimen preparation and electron microscopy. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
