Abstract
Objective
To compare the effects of the complex triamcinolone acetonide-hydroxypropyl-β-cyclodextrin (TA-CD) on
Design
Changes in cell viability and pro-inflammatory cytokines gene expression were analyzed in human chondrocytes using an
Results
The TA-CD viability was enhanced by the presence of the HA-CTL mixture in chondrocyte cultures. The exposure of cells to CM significantly increased interleukin-1β and interleukin-6 gene expression, and when the complex TA-CD was added to the inflamed cells, gene transcription of cytokines was restored to near baseline values, both in the presence or in the absence of HA-CTL mixture.
Conclusion
The addition of HA-CTL mixture significantly attenuated cytotoxicity induced by TA and preserved the anti-inflammatory effects, thus confirming the chondroprotective role of the HA-CTL mixture.
Osteoarthritis (OA) is an inflammatory condition that leads to articular disability and pain. It is associated with mononuclear cells’ infiltration that cause the release of pro-inflammatory mediators such as interleukin (IL)-1β and IL-6, catabolic enzymes that accelerate the degradation of extracellular matrix (ECM) molecules.
1
The most widely used pharmacological treatments to contain inflammation and to reduce symptoms associated with OA are nonsteroidal and corticosteroidal (CSs) anti-inflammatory drugs, such as hydrocortisone, methylprednisolone, dexamethasone, and triamcinolone.
2
Although discrepancies in their efficacy are reported, adverse effects on cell viability and cartilage loss remained a concern. In particular, some authors found that triamcinolone acetonide (TA), one of the most used CSs, exerts its cytotoxicity action in a dose-dependent manner,
3
whereas others demonstrated their chondral protective effects when administrated
The purpose of the present study was to evaluate the viability and anti-inflammatory effects of TA-CD complex in the presence or absence of the HA-CTL mixture on an inflammatory model of human primary articular chondrocytes treated with the conditioned medium (CM) of U937 macrophage induced by phorbol 12-myristate 13-acetate (PMA) lipopolysaccharides (LPS). We used hydroxypropyl-β-cyclodextrin and triamcinolone-acetonide from Cavitron W7 HP7, (Ashland, OR) and a HA-CTL mixture composed of 1.25 mg/mL sodium HA (molecular weight 1000-1600 kDa) and different concentrations of CTL (molecular weight 800-2500 kDa), pH 7.4.
TA and CD Induce Changes in Chondrocyte Viability That Are Reduced by the Presence of the Mixture HA-CTL
First, we tested the effect of HA-CTL mixture on cell viability cultivating the cells with the combination of HA and CTL in the same ratios (1.7:1) as that used in a previous
To determine whether the complex TA-CD alone or in combination with HA-CTL mixture affected the viability of chondrocytes (using a modified Denizot
10
method), the cells obtained from joint cartilage biopsies of 3 different donors were separately cultured in the presence or in the absence of different concentrations of CD, of the complex TA-CD and of TA-CD and HA-CTL mixture. These compounds were initially added to cell cultures, without performing medium changes during the observation time. The experiments were performed in triplicate for each cell preparation and the results are reported in
Figure 1
. CD at the concentrations up to 0.5 mg/mL did not affect cell proliferation (
Fig. 1A
), whereas a significant reduction of viability was found after 6 days of treatment, in the presence of all the tested TA-CD combined concentrations (CD from 0.05 to 15 mg/mL and TA from 0.7 to 212 µg/mL;
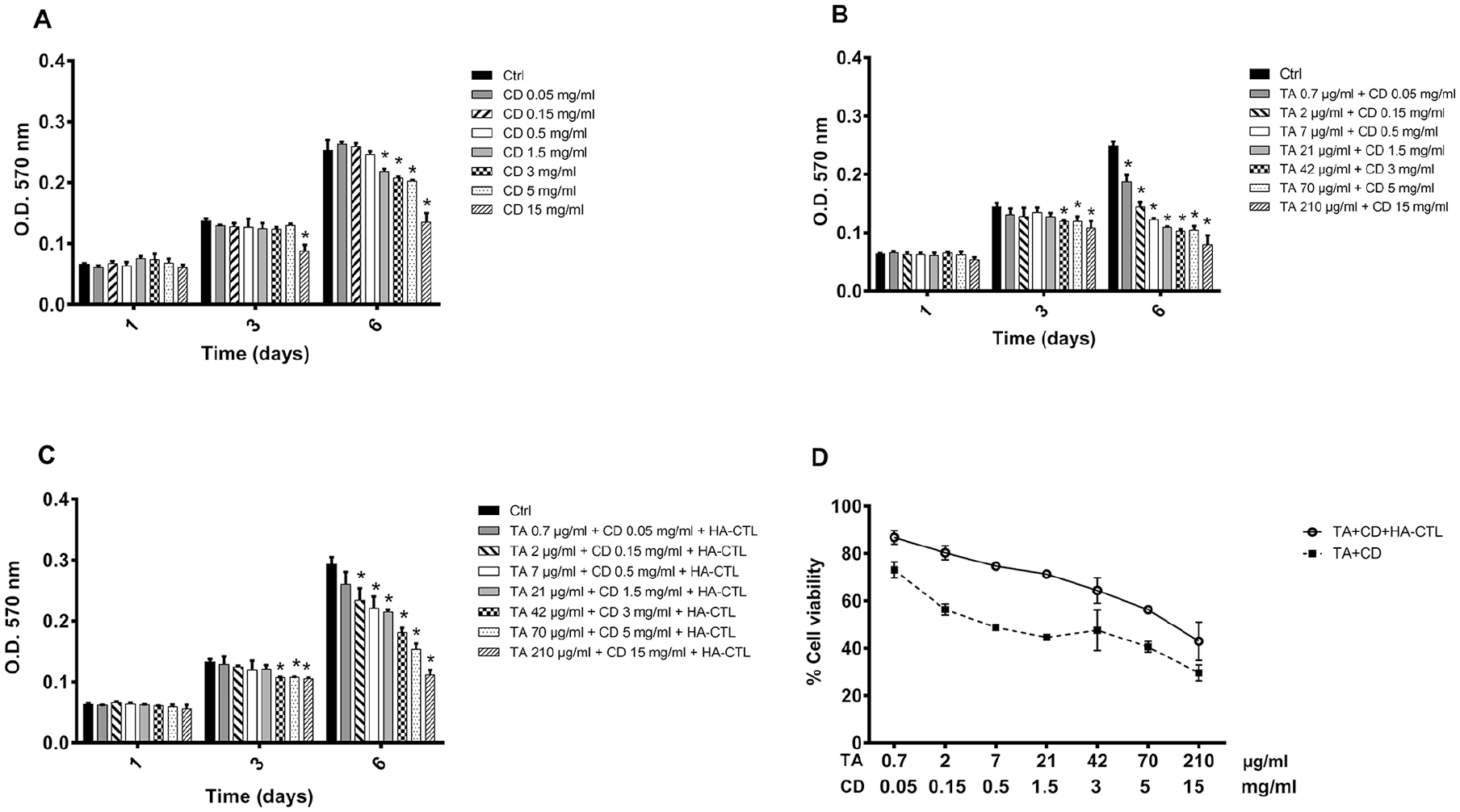
Time-dependent effects of TA and CD, alone or in combination with HA-CTL mixture on human chondrocyte viability. A total of 7000/cm2 cells were seeded in 24-well culture dishes and treated with TA, CD, or their combination, in the presence or absence of HA-CTL (
HA-CTL Mixture with TA Did Not Affect the Reduction of Pro-Inflammatory Molecules’ Expression in Chondrocytes
Human monocytes U937 (Thermo Scientific; Wilmington, DE) were differentiated to macrophages by the addition of PMA (Sigma-Aldrich) at a final concentration of 50 ng/mL for 48 hours followed by the exposure to 1 µL/mL LPS (Sigma) for 1 hour. Cells were then washed and cultivated with complete RPMI for 24 hours to produce the inflammatory CM collected. The PMA/LPS activation of U937 was confirmed by a significant (
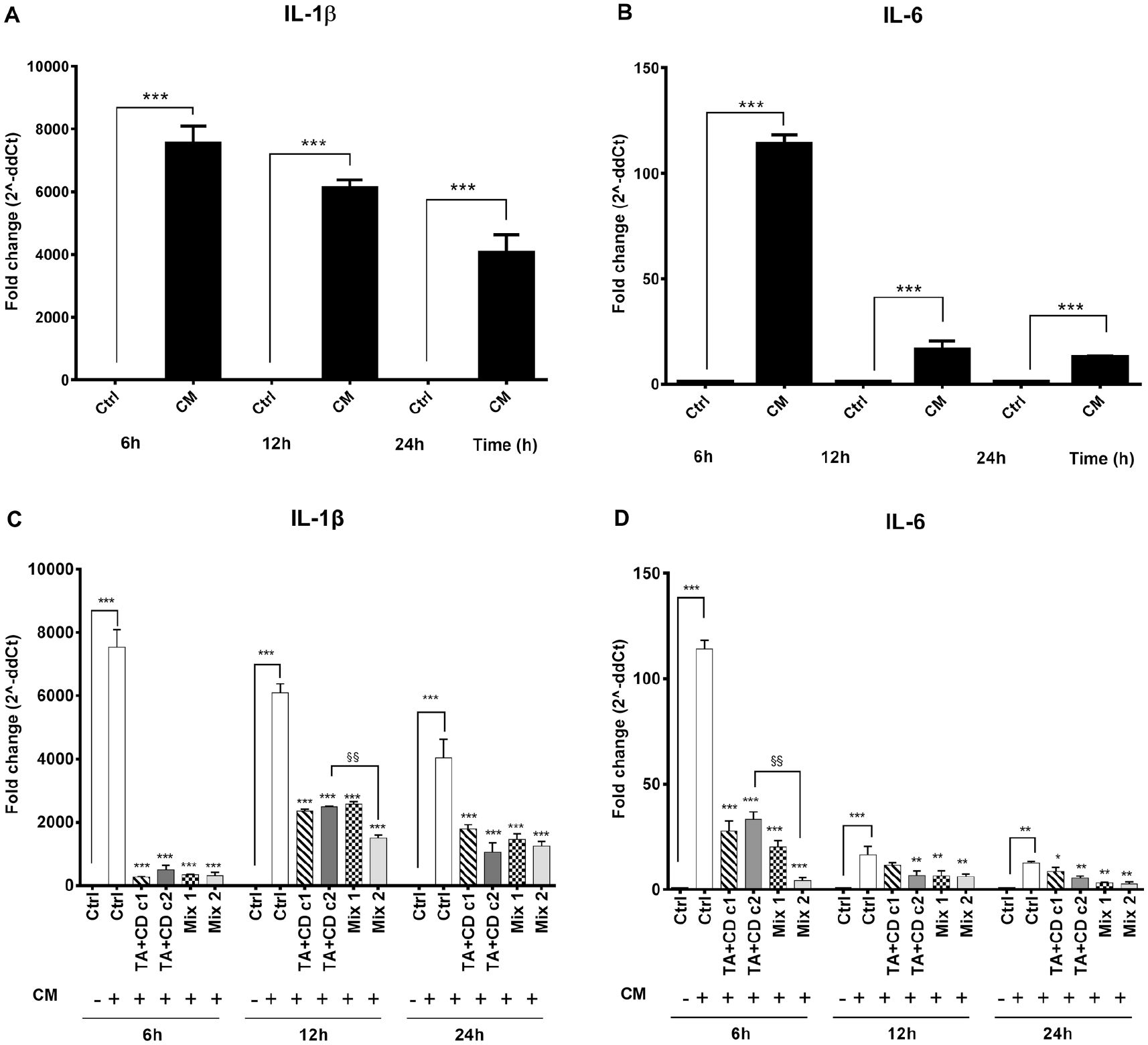
Chondrocyte expression of pro-inflammatory molecules after exposure to TA + CD in the presence or absence of HA-CTL mixture. Human chondrocytes were exposed for 24 hours to CM of activated U937 and then cultured with the studied molecules. RNA transcript levels specific for IL-1β and IL-6 were evaluated by qPCR. Activated U937 cell expression of (
Taken together, the results of the present study demonstrated that the presence of the mixture HA-CTL in primary human articular chondrocyte cultures partially counteracted the cytotoxic effect of TA while maintaining the well-known anti-inflammatory activity of this compound.
11
In fact, when the complex TA-CD was added to cells exposed to the CM of activated U937 human monocytes, gene transcription for IL-1β and IL-6 were restored to near baseline values, both in the presence or in the absence of HA-CTL mixture. Changes in cell viability induced by the complex TA-CD are reduced by the presence of HA-CTL mixture, thus suggesting that, combining corticosteroids with chondroprotective polysaccharides, such as HA, may be effective in maintaining viability of normal chondrocytes and in restoring survival of cells while providing a steroidal anti-inflammatory ingredient.6,12 Our results also validate the findings of the recent
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Jointherapeutics s.r.l. (Padua, Italy). Jointherapeutics did not take part in the study design, data analysis and interpretation, and in the writing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Local Ethical Committee of University of Padova (4744/AO/19).
Informed Consent
All patients signed written informed consent.
