Abstract
Objective
The International Cartilage Regeneration and Joint Preservation Society’s (ICRS’s) global registry, aims to be the best source of information for patients and an unbiased resource of evidence-based medicine for scientists and clinicians working to help those unfortunate enough to suffer the pain and disability associated with articular cartilage lesions. This article constitutes the scientific summary of the reports’ main findings.
Design
The article outlines the historical precedents in the development of orthopedic registries from the earliest tumor registries, then local arthroplasty databases that led ultimately to international collaborations between national arthroplasty and soft tissue registries. The ICRS global cartilage registry was designed from the outset as a GDPR (General Data Protection Regulation) compliant, multilingual, multinational cooperative system. It is a web-based user-friendly, live in 11 languages by end 2019, which can be accessed via https://cartilage.org/society/icrs-patient-registry/. Patients and clinicians enter data by smartphone, tablet, or computer on any knee cartilage regeneration and joint preservation treatment, including the use of focal arthroplasty. Knee Injury and Osteoarthritis Outcome Score and Kujala patient-reported outcome measures are collected preoperatively, 6 months, 12 months, and annually for ten years thereafter. EQ-5D data collection will allow cost-effectiveness analysis. Strengths, weaknesses, and future plans are discussed.
Results
Since inception the registry has 264 users across 50 countries. Major findings are presented and discussed, while the entire first ICRS global registry report is available at https://cartilage.org/society/icrs-patient-registry/registry-annual-reports/. Conclusion. A measure of the maturity of any registry is the publication of its findings in the peer reviewed literature. With the publication of its first report, the ICRS global registry has achieved that milestone.
Keywords
Introduction
The International Cartilage Regeneration and Joint Pres-ervation Society’s (ICRS’s) global nonarthroplasty registry aims to be the best source of information for our patients and for ourselves as scientists and clinicians working to help those unfortunate enough to suffer the pain and disability associated with articular cartilage lesions. Stakeholders of clinical registries include the patients, health care providers—both health care professionals and facilities—government and insurers paying for health care delivery, public health and regulatory agencies, industry, the research community, and the media. In its best form, a registry has been said to be a mission-driven, independent stakeholder-registry team collaboration that enables rapid, transparent, and open-access knowledge generation and dissemination. 1 Launched at the ICRS Congress in Sorrento in September 2016, the first report of the ICRS global registry is now available at https://cartilage.org/society/icrs-patient-registry/registry-annual-reports/. This article constitutes the scientific summary of the reports’ main findings. A measure of the maturity of any registry is the publication of its findings in the peer reviewed literature. With the publication of its first report, the ICRS global registry has achieved that milestone.
Patient Registries
Overview of the Trajectory of Patient Registries
Clinical registries may be organized around specific joints, implant type, or disease. There are national registries published for spine, upper limb, hip, knee, and foot and ankle. Some of the earliest registries were set up to gain understanding of the outcomes of musculoskeletal tumors with the earliest results published for the Leeds Bone Tumour Registry where data collection began in 1958. 2 Retrospective analysis of a consecutive, unselected population-based dataset in Sweden started in 1964, 3 at the Balgrist University Orthopedic Clinic in Switzerland in 1970, and the Registry of Rizzoli Orthopaedic Institute in Italy in 1982. 4 Since then, the utilization of clinical registries has continued to grow. Using the search terms “orthopaedic registry data” on PubMed in August 2019 showed over 5,000 results of which 518 were potentially relevant to the subject of this review. On further reading, 40 are referenced in this article.
There are registries for arthroplasty which may be industry led with a postmarket surveillance function or independently run by national or international professional societies. Nonarthroplasty registries exist for hip arthroscopy, knee soft tissue surgery and osteotomy, and articular cartilage treatments. There are military and civilian trauma registries. Pediatric registries may use different scores than their adult counterparts and are used to look at a number of conditions. Diagnosis-specific registries are published for rheumatoid arthritis, musculoskeletal tumors, pain, and rarer conditions such as facioscapulohumeral muscular dystrophy 5 and fibrodysplasia ossificans progressiva. 6
Clinical registries’ data are typically used for patient outcome surveillance to improve patient safety and health care quality. Outcomes of interest include short-term surgical complications, longer-term outcomes usually implant survival or reoperations, and patient-reported outcomes. Local, regional, and national registries have played an important role in the development of hip and knee arthroplasty and the treatment of patients’ pathologies of these joints. The use of large-scale national databases for observational research in orthopedic surgery has grown substantially in the past decade, and the datasets can be grossly categorized as either administrative claims or clinical registries. Both government and private claims to describe temporal trends, geographic variation, disparities, complications, outcomes, and resource utilization associated with both musculoskeletal disease and treatment. 7
A general shortage of evidence was highlighted in 2018, regarding risks and benefits of medical devices following serious safety concerns with metal-on-metal hip replacements and silicone breast implants. The new European medical device regulation will enforce post-market surveillance of existing and new implants. The usefulness of registry data as a source of information for medical device real-world clinical performance and safety has been demonstrated. Whole life span implant surveillance via revision rate monitoring, quality assessment of surgical and perioperative care, and hospital performance assessment. More recently, national and international comparison and benchmarking has been of interest. 8
Four arthroplasty registries led the use of data collection to enable evidence-based medicine. In the United States, The Mayo registry started in 1969 by Mark Coventry, is recognized as the oldest continuing registry for arthroplasty. Along with The Harris Registry at Massachusetts General Hospital, which was started in 1972 by William H. Harris, they have greatly contributed to the advancement of arthroplasty surgery and have identified poorly performing implants and techniques. Sir John Charnley stated in 1972, that “a serious consideration should be given to establishing a central register to keep a finger on the pulse of total implant surgery on a nation-wide basis.” It took until 2002 for the United Kingdom to introduce its National Joint Register. The Swedish Knee Arthroplasty Registry, starting in 1976 by Lars Lidgren, is the oldest national registry dedicated to joint arthroplasty and along with the Swedish Hip Arthroplasty Registry, started in 1978 by Peter Herberts, they have established the infrastructure, analysis, and reporting mechanisms, which have encouraged. other countries to subsequently develop national registries around the world. 9
As more countries have adopted the concept of national registries, a new area of research is possible by pooling the resources of large registries, as is now occurring with the Nordic countries. International cooperation is required between all those responsible for the design of registers as well as those who gather, assess and publish the data. 10 The process of registry globalization is a result of continued efforts over the past 50 years in improving and disseminating the knowledge gained from the early registries. 11 For example, patients with elective primary THR and TKR have been enrolled in the multinational Global Orthopaedic Registry. 12 . In his PhD thesis “On the Importance of Stepwise Introduction of New Implant Technology” in 1995, Henrick Malchau discussed a hypothesis that as we are all obliged to build clinical treatments on evidenced based principles, a more precise and careful evaluation when new implant technology is introduced would reduce the number of patients at risk. He suggested a 4-step process with the initial step being the use of preclinical studies, followed by 3 clinical steps: prospective randomized trials, multicenter studies followed by register studies. While his thesis was addressing the use of hip and knee arthroplasty, the same steps are equally important in the application of any device, technique, or technology. All the steps prior to the final step of registry studies have been very well executed and so it is timely that the ICRS has developed its registry. The ICRS global cartilage registry has been designed from the outset as a multilingual, multinational cooperative. Thus, it has avoided many of the difficulties other organizations have faced when trying to compare results between different systems from different countries and by collecting EQ-5D data will enable cost-effectiveness analysis in different health care settings.
Data Collection Instruments and System
Patient-reported outcome measures (PROMs), often questionnaires, are measures of different aspects of patient perceived outcomes. These measures can be either generic or joint specific. Generic measures are related to a patient’s general health and quality of life, whereas a specific PROM is focused on a particular disease, symptom, or anatomical region. Regardless of type, PROMs are produced by patients. Patients report on their own health status directly without interpretation from a surgeon or other medical professional. This reduces the possible bias that would unavoidably be introduced by a surgeon interpreting their own patients’ outcomes.
PROMs are increasing in use in many national and regional orthopedic arthroplasty registries. While revision surgery is the traditional endpoint of registries, it is a blunt and an insufficient measure of success; PROMs address this shortcoming by expanding beyond implant survival and measuring outcomes that are relevant to patients—relief of pain, restoration of function and improvement in quality of life. PROMs data can provide important information on value-based care, support quality assurance and improvement initiatives, help refine surgical indications, and may improve shared decision making and surgical timing.
There are several practical considerations that need to be considered when implementing PROMs collection, as the undertaking itself may be expensive, a burden to the patient, as well as being time and labor intensive. 13 The development of validated scoring systems or PROMs collection systems lead to the possibility of more detailed reviews. In 2001, the senior author began a local data collection system to study all aspects of soft tissue knee surgery using a paper-based questionnaire while the electronic PC-based patient user interface was developed by the hospital IT system. This was presented at BASK in 2005 and ESSKA in Austria in 2006. Working with colleagues on the steering committee of the UK National Ligament Registry since 2015 gave useful experience in the design of a web-based national registry and cross-fertilization of that aided in the design of the ICRS registry undertaken.
During the development of the ICRS registry great focus was placed on both choosing which PROMs to collect in the core dataset as well as to identifying a data collection system that was user friendly. The main focus when selecting PROMs was to limit the patient burden while maximizing the scientific return. PROMs collected in the ICRS registry are the KOOS (Knee Injury and Osteoarthritis Outcome Score), 14 EQ5-D, 15 and if patellofemoral lesions are treated, Kujala. 16 These are collected preoperatively, 6 months, 12 months, and annually thereafter. Complication data are collected at 6 weeks and at each annual review.
Cost-effectiveness analysis of the most common orthopedic surgery procedures: knee arthroscopy and knee anterior cruciate ligament reconstruction has been carried out. To conduct that analysis, one must have an outcome to measure quality of life (cost of a quality-adjusted life-year [QALY]). The QALY is calculated by multiplying difference in health-related quality of life, before and after treatment, by life expectancy. 17
Furthermore, the inclusion of the EQ-5D data collection in the ICRS global registry will enable cost-effectiveness and health economic analysis which is increasingly required for continued service provision in many countries’ health care systems. PROMs should, of course, be taken in context with their inherent limitations. The PROMs may not accurately reflect the functioning of the patient as recorded by independent objective measures of patient ability to perform tasks 18
Three key issues dictate the design of medical information systems.
Information. The basic function of every database is to store information about the phenomenon under investigation. Strict conformance to the rules of relational database design give consistent data and flexible access to that data.
Intelligence. Flexible access enables the user to harvest additional value from collected data. This value is usually gained via reports defined at the time of database design. Although these reports are indispensable, machine learning, a subdiscipline of artificial intelligence, has been successfully used to extract knowledge from databases of varying size by uncovering a correlation among fields and records. This knowledge can be represented in the form of decision trees, production rules, and probabilistic networks.19-21
Interface. Despite the obvious importance of collecting data and extracting knowledge, current systems may have interfaces which are not user friendly. Successful human-computer interfaces follow “golden” rules of dialog design: consistency, use of shortcuts for frequent users, informative feedback, organized sequence of actions, simple error handling, easy reversal of actions, user-oriented focus of control, and reduced short-term memory load. Visual representation of data and query results, is best as users react much more positively to visual rather than textual information.22,23 Electronic surveys produce higher response rates than paper collection of data, but some still prefer to use a mixture of both. 24 Web-Based data collection platforms can implement effective electronic Patient-Reported Outcome Registry. 25 Given the ubiquity of mobile phone ownership smartphone-friendly data collection has shown to be of use in several areas of the information capture pathway. 26
The ICRS registry is a web-based user-friendly tool that can be accessed via https://cartilage.org/society/icrs-patient-registry/. To further increase accessibility, the ICRS Global Registry is now live in 8 languages English, Italian, Spanish, Portuguese, Japanese, Greek, Dutch, and Polish. The registry is in the final stages of completing Chinese, Swedish, and German translations, which will be available by the end of 2019. Patients and clinicians can enter data by smartphone, tablet, or computer. It can be used to collect data of the outcomes of any knee pathology treatment up to and including the use of focal arthroplasty. Unicompartmental or total knee arthroplasty outcomes are already well served by mature arthroplasty registries and are beyond the scope of this exercise. The ICRS global registry is free to use, requiring minimal input from the clinician, limited to defining the pathology affecting the knee, its management (if any) and concomitant surgical interventions. Patients may self-register with their electronic consent adhering to the most stringent global consenting requirements.
Data are collected, encrypted, anonymized, and stored on servers currently located in the United Kingdom and for future data input, the physical location of data storage will be within certain jurisdictions geographic boundaries in accordance with the highest international data-handling standards and is GDPR (General Data Protection Regulation) compliant. Surgeons have access to a comprehensive real-time report of their own patients’ data, which may be used to aid an individual patient’s clinical follow-up to chart their clinical progress. A surgeon may access their own aggregated data to facilitate annual appraisal. Analysis of pooled anonymized data is undertaken by members of the steering committee to produce an unbiased and comprehensive report which will be produced annually. It is being used to assess the effects of bracing, injection therapies, soft tissue knee surgery of all kinds, focal implants, and periarticular osteotomy about the knee. It can be used to analyze the natural history of untreated or simply debrided articular cartilage lesions. There is a significant advantage to this objective approach to generating reports in comparison to originator unit or industry-controlled databases, which have a widely perceived risk of underreporting of poorer outcomes.
Cartilage-Specific Registries
One of the earliest examples of local knee registry data collection using a standardized format of recording demographics and surgical findings of the soft tissues (including articular cartilage status) in the knee was developed by Professor Iain Smilie of the University of Dundee, Scotland in the 1950s. Findings were recorded on cards and, although labor intensive, these data have allowed one of the longest longitudinal studies published at various time points up to 40 years of the outcome of meniscectomy in adolescents operated in the 1950s. 27 Since then, the earliest development of a specific pilot cartilage surgery register was in Norway in 2004. The Norwegian Registry of Knee Ligament (NKLR) was successfully established and has yielded useful information on the treatment of patients with both knee ligament and combined knee injuries. Patients with focal cartilage defects (FCDs) in their knees have reduced function and the treatment is difficult. The study was designed as a prospective cohort study in the form of a register. Patients with full-thickness FCDs in the knee with The International Cartilage Regeneration and Joint Preservation Society (ICRS) grade 3-4 on arthroscopy were included. The pilot included 2 hospitals; Oslo University Hospital (OUS), Ullevål and Akershus University Hospital (AHUS). They registered 58 patients with isolated FCDs, whereas 16 additional patients with full-thickness FCDs were registered through the NKLR. The compliance for the isolated FCDs were low in both locations, although it reached an acceptable level in one hospital when patients with combined injuries from the NKLR were included. The forms were incompletely filled out by the surgeons postoperatively and needed to be revised prior the establishment of a multination-wide register. 28
The German Cartilage Registry (KnorpelRegister DGOU) was introduced in October 2013 and aimed to evaluate patients who underwent cartilage regeneration for symptomatic cartilage defects. It represents a multination cohort study covering Germany, Austria, and the German-speaking part of Switzerland, which was introduced by a “Tissue Regeneration” working group of the Germany Society of Orthopaedic Surgery and Traumatology. The registry is technically based on a web-based remote data entry (RDE) system from 23 cartilage repair centers with up to 72 patients entered per month. Unlike the patient collective in the majority of prospective randomized controlled trials, the “real-world” patient population within the registry showed a high proportion of patients with accompanying pathologies, aged older than 50 years at the time of treatment and with unfavorably assessed accompanying pathologies such as lesions of the opposing cartilage surface or a previously resected meniscus. The platform proved to be very promising within the first 6 months. Unlike data from other clinical trials, the previous analysis of the patients’ data and therapies reflects successfully the actual medical care situation of patients with cartilage defects of the knee joint. 29 This dataset has been interrogated to produce several peer-reviewed publications.30-33
ICRS Patient Registry Results
The use of the ICRS registry is entirely voluntary and to date there are 264 clinician and delegate users across 50 countries, making it the largest cartilage registry. There is a total of 535 pathways in the registry across injection treatments, cartilage repair or restoration surgery, or other associated surgeries. In its 3 years since launch, it has grown exponentially; there has been a 90% increase in patient pathways added over the past 2 years ( Fig. 1 ).
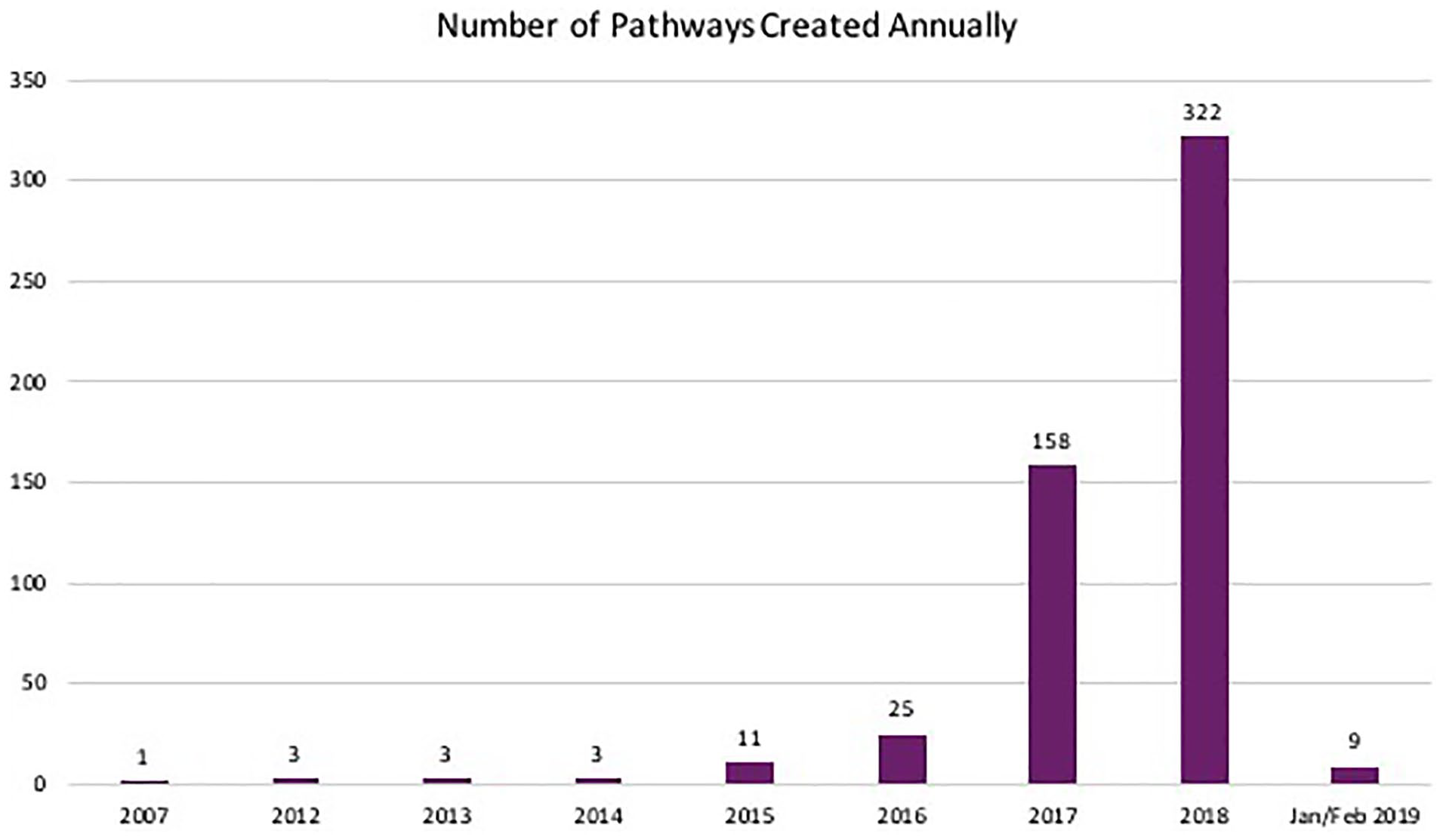
The volume of knee pathways that were created each year.
To date, those patients in the registry receiving cartilage repair or restoration surgery had an average age of 35 years (range 16-57 years) with an average body mass index of 26 kg/m2 (range 18-40 kg/m2). This is slightly older than the average age reported to be 29.8 years in the Norwegian pilot cartilage surgery register 28 and younger than the German Cartilage Registry mean age 37.11 years, 29 although they have reported a high proportion of degenerative lesion treatment in their cohort. 33 Seventy-two percent of them had a single articular cartilage lesion treated, 23% for 2, and 6% for 3 or more. Furthermore, 32% of these patients underwent concomitant surgical procedures. This is keeping with the experience of the German cartilage registry. 33 Two-thirds of the patients in the ICRS registry had postoperative bracing prescribed.
A total of 218 patient pathways currently exist for patients undergoing injection treatments. The commonest injections used being hyaluronic acid, stem cells, platelet-rich plasma, or a combination of these. Those patients receiving injection therapies had an average age of 66 years (range 31-89 years) and were more frequently women (64%). Neither a posttreatment brace nor physiotherapy was prescribed in the majority of the treatment pathways reported. The first annual report provides an overview of the data within the registry. The report can be accessed at https://cartilage.org/society/icrs-patient-registry/registry-annual-reports/.
The creation of the ICRS registry seeks to cultivate a global collection of representative data for patient suffering from articular cartilage defects. The existence of the registry is increasingly recognized as an effective tool for the objective and unbiased follow-up of biological treatment of symptomatic articular cartilage lesions, for example, the UK National Institute for Clinical Care Excellence (NICE) has recommended that any patients treated in the United Kingdom with mosaicplasty procedures should be documented and followed up into the long term using the ICRS registry. NICE recommendations are often taken up by regulatory authorities elsewhere. The confidence that they have shown in the reliability of the registry is a testament to its credibility.
Complications
The ICRS registry can track any reported complications from both the patients and the clinicians. To date, there are no complications recorded in the first ICRS global registry report. However, this is not an uncommon finding in voluntary registry data collection and the collection of complication data via the ICRS registry should not be used as a substitute for the stringent documentation of adverse outcomes in standard prospective studies and required in post-market surveillance studies.
While some may believe the national arthroplasty registers monitoring of adverse outcomes are sufficiently robust that they “have to be used for research and post-market surveillance.” 34 Others report concerns that outline the weaknesses of national registers. Revision is much less sensitive than PROM to measure clinical failure, with one report suggesting that using revision as an end point was exaggerating the success of total knee replacements in comparison to partial knee replacement. 35 In another paper, it was stated that a mature national joint registry with widespread adoption and audit can successfully demonstrate trends and influence future orthopedic practice. But the authors were keen to stress that while correlations can be identified they must not be misinterpreted as causality. It is essential to consider confounding when analyzing observational datasets. 36
Although registry data indicate that most prostheses have similar outcomes, some have a higher than anticipated rate of revision when compared to all other prostheses in their class the Australian Orthopaedic Association National Joint Replacement Registry (AOANJRR) has developed a method to report prostheses with a higher than expected rate of revision or “outlier” prostheses.
They state it is important that registries use a transparent and accountable process to identify an outlier prosthesis. 37 As the ICRS global registry gains traction and gains sufficient data to compare differing treatments we must use the learning curves of the arthroplasty registries to adopt similar fair, unbiased methodology to tease out differences between treatment options. Articular surface replacement (ASR) hip prostheses were recently recalled worldwide by the manufacturer. The Kaplan-Meier method and proportional-hazard modeling were used to compare the revision rate of primary total hip arthroplasties involving the ASR systems with conventional primary hips and all other hip resurfacing prostheses, finding significantly higher revision rates in the ASR cohort. 38 The UK NJR questioned its ability to monitor the introduction and performance of a new prosthetic joint replacement. Despite assessment processes in place, particularly the CE (Conformité Européenne) mark, the National Joint Registry, and the Orthopaedic Data Evaluation Panel. They said that these organizations can only react to poor performance; as monitoring agencies they cannot prevent a poor implant from entering the market. When an implant has been shown to perform badly in the United Kingdom, it is referred by the NJR to the Medicines and Healthcare products Regulatory Agency (MHRA), which then discusses the report with the manufacturer. When it is deemed that there is unequivocal evidence of a serious problem with the implant, the MHRA issues alerts to all hospitals and practitioners. They expressed continuing concerns about how to assess and monitor modifications to already existing devices. 39
Patients are accurate in reporting the absence of complications, but not their presence. The sensitivity of patient-reported complications needs to be improved by better clarity of the questions asked to document complications. 40
Adverse outcomes associated with 180,717 episodes elective knee arthroscopy were studied in a population-based cohort study in Victoria, Australia. 41 The most common adverse outcomes within 30 days were deep venous thrombosis (DVT; 579, 0.32%), effusion and synovitis (154, 0.09%), pulmonary embolism (PE; 147, 0.08%), and hemarthrosis (134, 0.07%). The 30-day orthopaedic readmission rate was 0.77%, and there were 55 deaths (0.03%). Within 90 days of arthroscopy, they identified 655 events of DVT (0.36%) and 179 PE events (0.10%). Overall there were 6.4 adverse outcomes per 1,000 elective knee arthroscopy procedures (0.64%). 41
Administrative claims data benefit from the high patient numbers obtained through a majority of hospitals but their disadvantages include lack of precision of coding schemes. 7 This methodology was used to assess complications following anterior cruciate ligament reconstruction in the English NHS. 42 It was reported, between March 2008 and February 2010 in 13,941 operations, 90-day DVT and pulmonary thromboembolism rates were 0.30% (42) and 0.18% (25) respectively. There were no in-hospital deaths. 0.75% (104) of the consecutive patient cohort had a wound complication recorded. 0.25% (35) underwent a further procedure to wash out the infected knee joint and 1.36% (190) were readmitted to an orthopedic ward within 30 days. 42
The first UK National Ligament Registry Report was published in 2015. 43 There have been annual reports each year since. None of which have recorded complication rates reflecting the findings from the NHS administrative dataset. 42
The absence of complication reports in the first ICRS global registry report is typical of the earlier registry reports for soft tissue procedures but in reality complications will ultimately be shown to be in the frequencies reported for the arthroscopic techniques used for debridement and articular cartilage biopsy harvest and commonly associated procedures such as ACL reconstruction as reported in the publications listed above.
One must conclude that the limitations of registry data as outlined above from each of the areas of non-orthopedic registries, arthroplasty and soft tissue registries documented in recording of complications will be an issue even when those imputing data to the ICRS registry do record complications. Causes of the failure of various implants are inconsistently attributed between registries. Given the importance of accurate adverse event documentation in post-market surveillance, registry data will be insufficient to allow the ICRS registry to act as a postmarket surveillance tool. The information we will generate will complement more traditional data collection systems in postmarket surveillance studies of adverse events to give data of sufficient robustness to convince regulatory authorities of the safety of the procedures we are documenting in the registry.
Future Projects
This first report shows the potential we have to achieve our aim of being the best source of information for our patients and for ourselves as scientists and clinicians working to help those unfortunate enough to suffer the pain and disability associated with articular cartilage lesions. The registry’s translation into 11 languages by the end of 2020 will enable our patients from across the globe to input sufficient data to achieve our aim. They will need encouragement by the teams looking after them to remain engaged in the project long term. Those administering treatments will need to lead by example and ensure the operative and injection therapies are fully documented in the system and that the complications experienced, or absence of such issues, are explicitly documented.
The first tranche of pathways has concentrated on the management of conditions of the knee. New pathways are in process to enable treatments of other joints to be reported such as the hip, foot and ankle. Furthermore, the veterinary members of the ICRS are designing an equine pathway as the first planned multispecies pathway.
There are a number of mature single center cartilage databases that are in the process of being assimilated into ICRS global registry. Once the information importing is completed, we expect the number of patients’ outcomes will be boosted by 1500 cases with follow up periods of up to 10 years.
Footnotes
Acknowledgments and Funding
The authors acknowledge the contributions of our fellow members of the ICRS Steering Committee: Pieter Emans, Laurie Goodrich, Guilherme Gracitelli, Cale Jacobs, Marcel Karperien, Sven Kili, and Kai Mithoefer. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The ICRS Global Registry is part of the ICRS commitment to research and education in cartilage restoration and joint preservation. The registry is indirectly supported by generous donations of the ICRS sponsors toward these aims.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: At the time of initial submission, all authors were involved on either the ICRS registry steering committee or in the managing of the registry.
