Abstract
Objective
We sought to evaluate the effect of different concentrations of ethanol on phenotype and activity of articular chondrocyte synthesis of neonatal rats in 2-dimensional (2D) and 3-dimensional (3D) culture.
Methods
Chondrocytes were cultured in chondrogenic medium with different concentrations of ethanol: 0.0% v/v (control); 0.05% v/v (8.6 mM); 0.25% v/v (42.9 mM), and 0.5% v/v (85.7 mM). Chondrocytes under 2D culture were subjected to MTT assay, while chondrocytes under 3D culture were processed for paraffin inclusion and stained by periodic acid Schiff (PAS) to evaluate mean chondrocyte diameter and percentages of cells, nucleus, cytoplasm, well-differentiated matrix, and PAS+ areas. The expression of gene transcripts for aggrecan, Sox9, and type II collagen was evaluated by real-time quantitative polymerase chain reaction.
Results
There was no difference between groups by the MTT assay. PAS staining revealed that chondrocytes treated with 0.5% v/v ethanol had higher percentages of cytoplasm and nuclear areas, but with a reduction in PAS+ matrix area. The mean diameter of chondrocytes was similar between groups. The expression of aggrecan in the group treated with 0.5% v/v ethanol was lower in comparison to that in the control. In the groups treated with 0.25% v/v and 0.5% v/v ethanol, the percentage of differentiated cartilage was lower in comparison with that in the control. The group treated with 0.05% v/v ethanol was similar to the control in all parameters.
Conclusions
Ethanol acted directly on in vitro cultured articular chondrocytes of newborn rats, altering the chondrocyte phenotype and its synthesis activity, and these effects were dose dependent.
Introduction
Chondrogenesis is a complex phenomenon, involving several stages, being one of the first cellular events that occurs during skeletal development. The formation of the skeleton occurs from intramembranous and endochondral ossifications 1 ; however, most bones originate from the endochondral bone formation, in which the mesenchymal stem cells differentiate into chondrocytes, forming a cartilaginous mold that will be gradually replaced by bone. 2 During the postnatal period, proliferation, differentiation, and maturation of the chondrocytes of the epiphyseal plate and the articular cartilage determine the final length of adult bones. 3 Sox9 is essential for cell survival, conversion of chondroprogenitor cells into chondrocytes, chondrocyte maturation, 4 aggrecan expression, 5 and for type II collagen expression, the latter being the main component of the extracellular matrix. 6
However, if the steps that determine endochondral bone formation and growth are not properly regulated and coordinated, changes in bone growth may arise as a consequence of this lack of control. 7 Among the substances that are able to interfere with endochondral bone formation and growth, ethanol stands out. 8 The consumption of alcoholic beverages by pregnant women is a risk factor for the fetus, since ethanol is able to pass freely across the placenta and enter the fetal circulation because of its low molecular weight and high solubility. 9 Ethanol has the ability to affect maternal physiological mechanisms necessary for fetal development, such as the control of calcemia. 10 In addition, prenatal ethanol exposure (PEE) is capable of inducing a set of fetal changes known as fetal alcohol spectrum disorders (FASD), which are characterized by facial abnormalities, structural or functional dysfunction of the central nervous system, and reduction in bone growth. 11
A limitation of studies in humans is the difficulty in stating that the observed changes in the individuals who suffered PEE are exclusively due to ethanol, since its association with environmental factors and the use of other drugs is frequent. 11 Besides, ethical reasons prevent pregnant women from being included in certain experiments; therefore, murine models have been widely used to clarify the damage caused by ethanol consumption in the prenatal period.9,12 Rats with PEE have decreased bone length as well as disorganization of the epiphyseal plaque, with reduced resting zone thickness and increased hypertrophic zone. 8 In addition, rats with PEE are predisposed to osteoarthritis.9,12 Often, the reduction of bone growth caused by PEE is not attributed to the direct effects of ethanol. The reduction in femur length of rats with PEE, for example, is attributed to increased maternal glucocorticoid levels and not to direct effects of ethanol on the fetus. 13 In addition, it is believed that the reduction in bone growth may be caused by malnutrition due to the diminished suckling ability of the pup 14 associated with milk ejection failure due to the suppressive effect of ethanol on the release of maternal oxytocin. 15
Although there are studies demonstrating the direct effects of ethanol on cultures of osteoblasts, 16 osteoclasts, 17 and mesenchymal stem cells, 18 there are no reports of studies that investigated the effects of ethanol addition on the phenotype, viability, and activity of articular chondrocytes in neonates. We hypothesized that the reduction of endochondral bone growth caused by PEE is due to direct effects of ethanol on chondrocytes. The present work proposes the use of an experimental model of 2- and 3-dimensional (2D and 3D, respectively) cultures, with the objective of evaluating, for the first time, the direct effects of different concentrations of ethanol in the culture medium on chondrocyte phenotype, synthetic activity, as well as expression of gene transcripts involved in chondrogenesis, as represented by Sox9, type II collagen, and aggrecan.
Methods
All procedures were approved by the Ethics Committee on Animal Use of the Universidade Federal de Minas Gerais, under protocol number 119/2016.
Isolation and Culture of Chondrocytes
Ten 3-day-old Wistar rats were used. The animals were subjected to euthanasia using an overdose (150 mg/kg) of Tiopental (Thiopentax, Cristália, Itapira, SP, Brazil) intraperitoneally. Antisepsis was performed on the skin of the hind limbs with 70% alcohol and povidone-iodine (PVPI); then, the femurs were dissected from the muscle and connective tissues with sterile instruments in a laminar flow cabinet and washed in 0.15 M sterile phosphate buffered saline (PBS).
Cartilage sections were immediately harvested from the distal and proximal ends of the femur. For isolation of chondrocytes, the cartilage fragments were washed with 0.15 M PBS. Cartilage was then digested by overnight incubation with 0.4% collagenase type 1 (Sigma-Aldrich, St. Louis, MO, USA) plus Dulbecco’s modified Eagle’s medium low glucose (DMEM–low glucose) (Gibco, Invitrogen, Grand Island, NY, USA) with 5% fetal bovine serum (Invitrogen, Grand Island, NY, USA).19,20
After 12 hours, the chondrocyte suspensions were centrifuged for 10 minutes at 1400 rpm and washed with 0.15 M PBS. After centrifugation, cells were resuspended and cultured in T25 flasks with chondrogenic medium consisting of DMEM–low glucose plus 1% fetal bovine serum (Gibco, Invitrogen) and supplemented with ascorbic acid (Sigma-Aldrich), dexamethasone (Sigma-Aldrich), insulin (Biobras, São Paulo, Brazil), transferrin (Sigma-Aldrich), (Sigma-Aldrich), and bovine serum albumin (Sigma-Aldrich). The cells were incubated at 37°C with 5% CO2. After 24 hours, cultures were washed with 0.15 M PBS to remove nonadherent cells.
After the second passage, depending on the type of assay, the cells were transferred to 24-well plates (2 × 104 cells/well), 6 wells (1 × 105 cells/well), T25 flasks (1 × 106 cells/bottle), or to plastic tapered tubes with capacity for 15 mL (1 × 106 cells/tube), where they were cultured in 2D or 3D systems. After 2 days of culture, the chondrogenic medium was replaced by chondrogenic medium containing different concentrations of ethanol: 0.05% v/v (8.6 mM); 0.25% v/v (42.9 mM), and 0.5% v/v (85.7 mM), similar to a previously established protocol, 16 mimicking the mild, moderate and intense consumption of ethanol, respectively. Thus, three groups were treated with ethanol and one control group was cultured in chondrogenic medium identical to that of the treated groups, but free of ethanol. The culture medium was changed 3 times per week.
All analyses (MTT assay, reverse transcription–polymerase chain reaction [RT-PCR], and pellet morphometry) were performed simultaneously. Chondrocytes from the articular cartilage of 10 newborn rats of 5 different litters were used to obtain heterogeneity. A chondrocyte pool of these cells from all the rats was prepared. The same pool of cells was used for all the analyses (MTT assay, RT-PCR, and pellet morphometry). Each group consisted of 6 wells (MTT) or 6 pellets (RT-PCR and morphometry).
MTT Assay
At the end of 7, 14, and 21 days of culture, the chondrocytes of all groups under 2D culture in 24-well plates were subjected to an assay of the conversion of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) (Invitrogen, Camarillo, CA, USA, catalog number: M6494) into formazan crystals. This is a quantitative method of evaluating succinate dehydrogenase enzyme activity in viable cells, where the enzyme present in mitochondria converts the MTT substrate to formazan crystals. 21 The medium was replaced with 210 μL of medium with 1% fetal bovine serum in each well and 170 μL of MTT (5 mg/mL). The plate was incubated for 2 hours in an incubator at 37°C with 5% CO2. Formazan crystals were observed under a microscope and then 210 μL of SDS (sodium dodecyl sulfate) with 10% HCl was added, which remained overnight in an incubator at 37°C with 5% CO2. Cell debris were cleared from the MTT assay samples before measuring the absorbance at 595 nm. Subsequently, the samples were analyzed in a plate reader at a wavelength of 595 nm. The mean and standard deviation were determined for 6 wells/group.
Histochemical and Morphometric Analysis of Chondrocyte Pellets
At the end of 21 days of culture, the chondrocyte pellets of all groups under 3D culture in 15 mL conical tubes were washed with 0.15 M PBS and fixed with 4% paraformaldehyde. The pellets were then processed into paraffin blocks 22 and subsequently sectioned into 5-μm slices using a microtome. The histological sections were placed under gelatinized glass slides 23 and stained by the periodic acid Schiff (PAS) (Sigma-Aldrich, St. Louis, MO, USA) to determine the percentage of PAS+ cartilaginous matrix. Also, the percentage areas of cytoplasm and nuclei of chondrocytes and well-differentiated cartilage areas were determined. The well-differentiated cartilage was characterized by chondrocyte groupings housed within gaps and surrounded by a large quantity of matrix, similar to articular cartilage tissue. These analyzes were carried out using a graticule with 121 points coupled to the eyepiece of an optical microscope, with a magnification of 40×. The average of 2 distinct fields was used to obtain the final values for each pellet. The size of the chondrocytes was also analyzed by mean diameter measurements.
The measures were carried out by blinded readers. The pellet was serially sectioned, and the central histological section was used for analysis. The central section was used for all the analyses and for all groups. To analyze the percentages of cells, nucleus, cytoplasm, well-differentiated matrix, and PAS + areas, two fields were used under a 40× objective lens, thereby analyzing the entire central section of the pellet. To analyze the chondrocyte diameter, images of five fields of the histological central section of each pellet, under a 100× objective lens, were captured with the aid of a Leica ICC50 camera coupled to a Leica DM500 microscope. The images were analyzed using LAS EZ 3.4 software. Diameters of chondrocytes with well-defined cytoplasmic boundaries were measured.
All morphometric variables were evaluated for 6 pellets per group, that is, 6 pellets were used for each group (n = 6), making it total 24 pellets.
Evaluation of the Expression of Aggrecan, Sox9, and Type II Collagen
Real-time quantitative PCR (RT-qPCR) was performed on 3D cultures of chondrocytes in all groups after 21 days of culture in T25 flasks. Quantitative evaluation of aggrecan, Sox9, and collagen type II expression was performed.
Briefly, 1 μg of RNA was used for cDNA synthesis using the SuperScript III Platinum Two-Step qPCR kit with SYBR Green (Invitrogen, Grand Island, NY, USA). RT-qPCR was performed on a SmartCycler II thermal cycler (Cepheid, Carpinteria, CA, USA). The first step of amplification was initiated with reverse transcription for 120 seconds at 50°C, followed by PCR with the following parameters: 45 cycles of 15 seconds at 95°C and 30 seconds at 60°C. At the end of each assay, the fluorescence data were analyzed for CT values. Gene expression was calculated using the
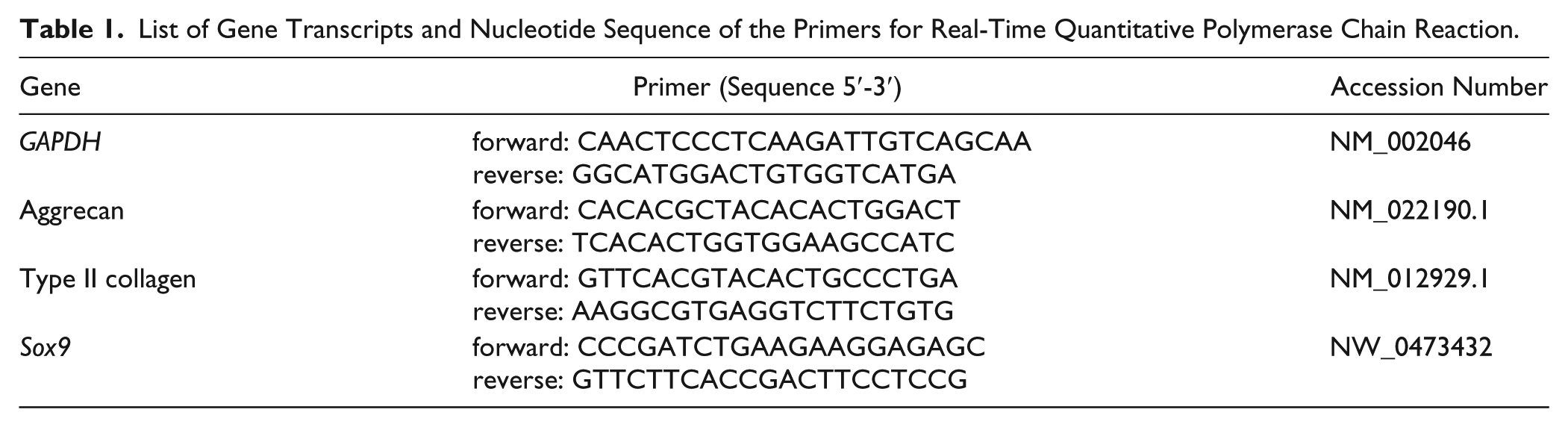
List of Gene Transcripts and Nucleotide Sequence of the Primers for Real-Time Quantitative Polymerase Chain Reaction.
Statistical Analysis
The design was entirely randomized, and for each variable, the mean and standard deviation were determined. Analysis of variance was performed, and comparisons of means were performed by Student t test using the GraphPad computational package. Differences were considered significant if P < 0.05.
Results
MTT Assay
There was no significant difference between the means of any of the groups treated with ethanol and the control group with respect to cell viability as assessed by the MTT assay ( Fig. 1 ).
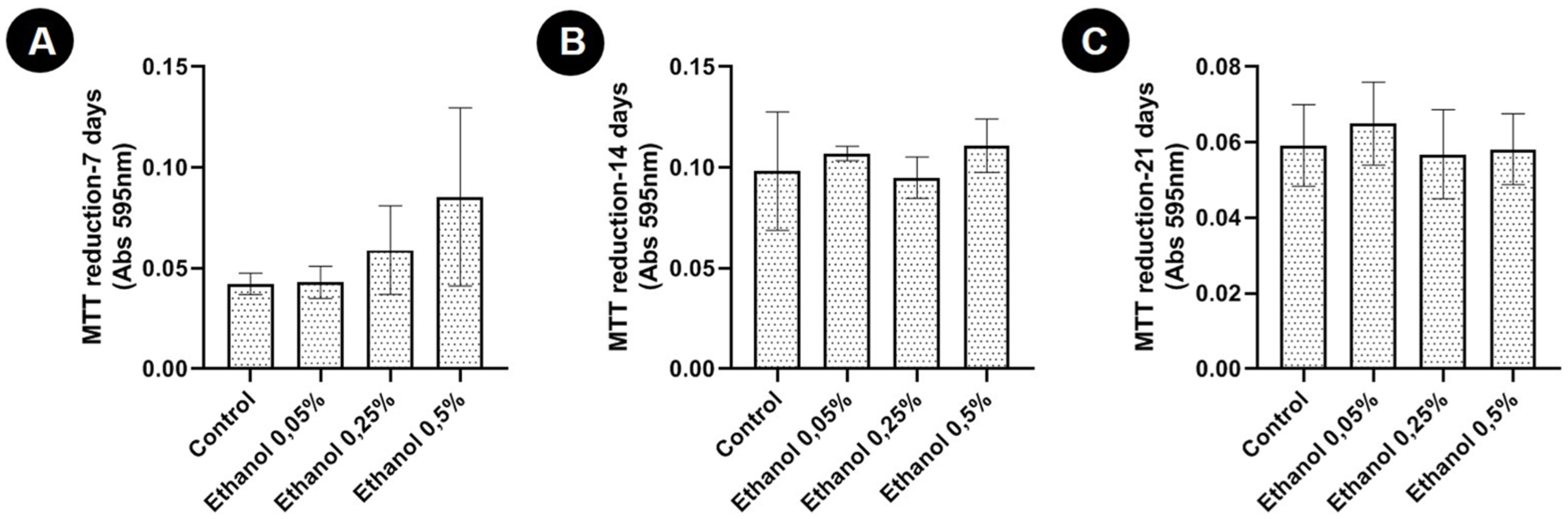
Conversion of MTT to formazan (mean ± standard deviation) in 2-dimensional cultures of articular chondrocytes from newborn rats without ethanol (control) and with different concentrations of ethanol (0.05% v/v [8.6 mM]; 0.25% v/v [42.9 mM], and 0.5% v/v [85.7 mM]) after 7, 14, and 21 days of culture. *P < 0.05 (Student t test).
Histochemical and Morphometric Analysis of Chondrocyte Pellets
Chondrocyte pellets from the treated groups had similar sizes to the control group (data not shown). In all pellets, regardless of group, there were areas of chondrocyte clusters enveloped by a small amount of matrix, as well as well differentiated areas of cartilage similar to articular tissue, where chondrocytes were located within gaps of varying sizes and were enveloped by large quantities of chondrogenic matrix. In all pellets, although the percentages varied according to the group, there were areas of PAS+ and PAS− matrix. Chondrocytes treated with 0.5% ethanol had a significantly lower percentage of PAS+ matrix in comparison with the control ( Fig. 2 ). In this group treated with 0.5% ethanol, it was possible to verify that the percentage of well-differentiated chondrogenic matrix was also significantly lower in comparison with the control ( Fig. 3 ). However, the percentages of nuclei and cytoplasmic areas of the chondrocytes in the pellets of this group were significantly higher in comparison to the percentages observed in the pellets of the control group. However, mean chondrocyte size did not differ significantly between groups ( Fig. 3 ).
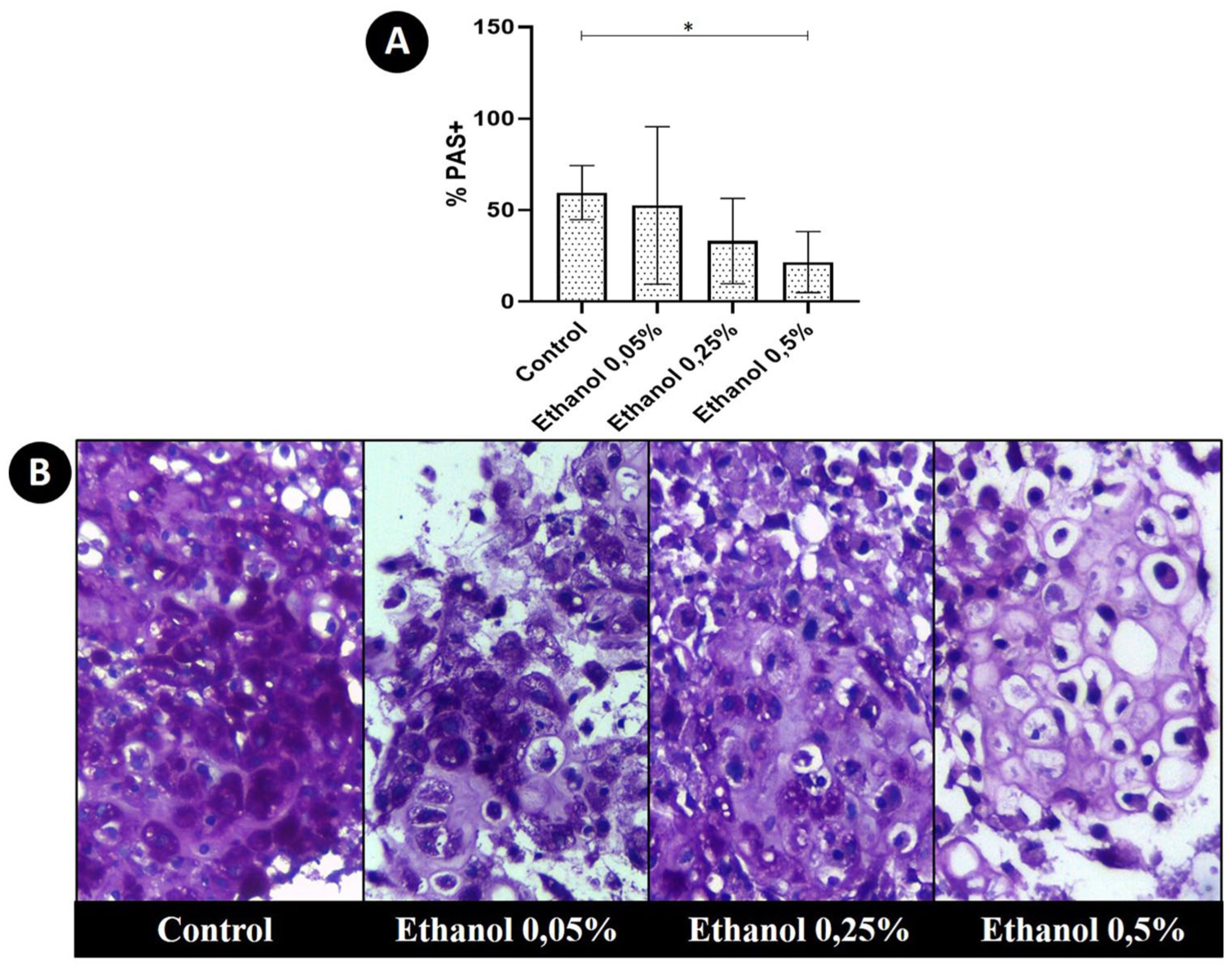
(
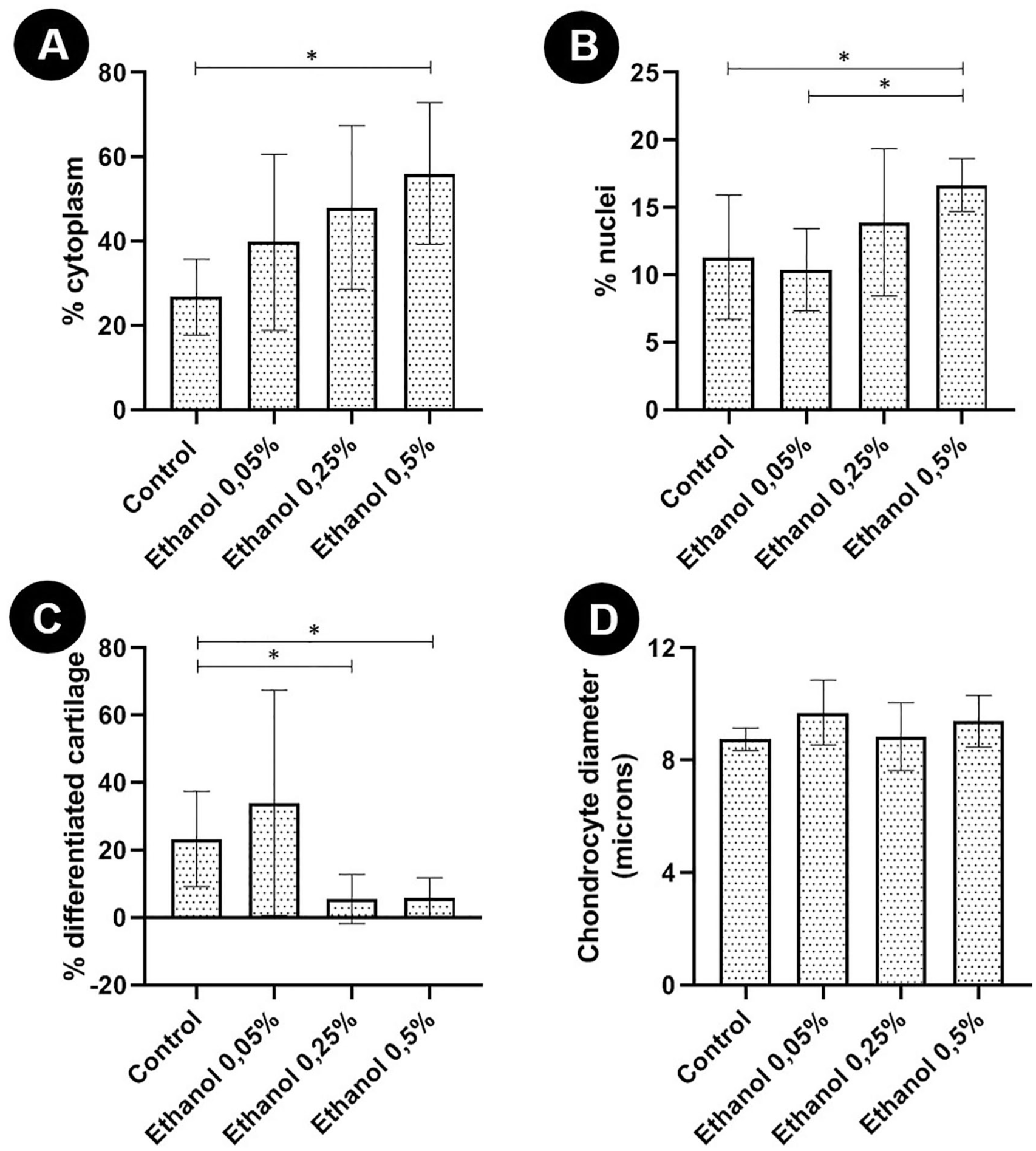
Morphometric analysis of articular chondrocyte pellets of newborn rats cultured without ethanol (control) and with different concentrations of ethanol (0.05% v/v [8.6 mM]; 0.25% v/v [42.9 mM], and 0.5% [85.7 mM]) after 21 days of 3-dimensional culture. (
The percentage of well-differentiated cartilaginous matrix was also significantly lower in the group treated with the moderate concentration of ethanol, that is, 0.25%. However, in this group, the percentage of PAS+ areas, as well as the other parameters evaluated, were similar to those of the control group ( Fig. 2 ). There was no significant difference between the morphometric evaluation of the pellets of the group treated with the lowest concentration of ethanol (0.05%) and the control group.
Expression of Aggrecan, Sox9, and Type II Collagen
In relation to the expression of gene transcripts for aggrecan, Sox9, and collagen type II, performed after 21 days of 3D culture, only chondrocytes treated with the highest concentration of ethanol (0.5%) showed a significant reduction in aggrecan expression in comparison with that in the control group. None of the ethanol concentrations tested significantly altered the expression of Sox9 and type II collagen ( Fig. 4 ).

Expression of gene transcripts (mean ± standard deviation) of aggrecan, Sox9, and type II collagen by the real-time quantitative polymerase chain reaction (RT-qPCR) technique in articular chondrocyte of newborn rats cultured without ethanol (control) and with different concentrations of ethanol (0.05% v/v [8.6 mM]; 0.25% v/v [42.9 mM], and 0.5% v/v [85.7 mM]) after 21 days of 3-dimensional culture. (
Discussion
The results of the present study demonstrated, for the first time, that articular chondrocytes of newborns rats cultured in chondrogenic medium containing 0.5% v/v ethanol present significant reduction in percentage of PAS+ chondrogenic matrix, percentage of well-differentiated cartilage, and expression of aggrecan. The results also showed that such effects depend on the ethanol concentration, with 0.25% v/v exhibiting reduction only of percentage of well-differentiated cartilage, and that the lowest concentrations of ethanol (0.05% v/v) had no effects in terms of the parameters evaluated.
Ethanol interferes in the activity of chondrocyte synthesis in a dose-dependent manner. While low concentrations of ethanol did not significantly alter PAS+ matrix synthesis, chondrocyte pellets treated with the highest ethanol concentration synthesized a lower percentage of PAS+ matrix. PAS staining was chosen as a simple and effective way to demonstrate the presence of proteoglycans. Our study used a 3D culture system, which has the advantage of maintaining the cartilaginous phenotype for several weeks in appropriate medium. 24 A possible reason for the diminished production of proteoglycans in the extracellular matrix may be the interference of ethanol in chondrocyte maturation. It was found evidence that chronic ethanol consumption induces osteogenic differentiation in articular cartilage chondrocytes of adult humans. In addition to promoting dedifferentiation, ethanol also reduced the gene expression of transforming growth factor–β (TGF-β), which is important for chondrogenic differentiation. 25
It was found that the addition of 0.5% v/v ethanol to the culture medium promoted an increase in the percentage of cytoplasm in chondrocytes. At first, this finding may suggest that ethanol induces cellular hypertrophy. However, this suspicion was resolved by measuring the mean diameter of the chondrocytes, which allowed the conclusion that there was no significant difference in chondrocyte size between groups. Thus, it is possible that the increase in the percentage of cytoplasm and also chondrocyte nuclei in the group treated with 0.5% v/v of ethanol occurred because the synthesis of cartilaginous matrix was reduced.
Another finding observed in our study was the smaller number of areas with well-differentiated cartilage in groups treated with 0.25% v/v and 0.5% v/v of ethanol, which is compatible with the finding that ethanol interferes with pathways essential for cartilaginous differentiation. 26 This result suggested that ethanol changes the phenotype of chondrocytes, as evidenced by the lower synthesis of proteoglycans in the extracellular matrix (reduction of PAS+ areas and decreased aggrecan expression) and reduction of chondrocyte maturation (reduction in well-differentiated cartilage areas). In osteoblasts, it was found a reduction in the expression of bone differentiation markers such as osteonectin and type I collagen caused by ethanol. 16
RT-qPCR results revealed that there was less aggrecan gene expression in chondrocytes treated with 0.5% v/v ethanol. Aggrecan is the main structural proteoglycan of the extracellular matrix, and acts by promoting cell-cell and cell-matrix interactions, besides lubricating and conferring resistance to the joint. 27 Thus, reduction in aggrecan expression considerably compromises joint function and may be indicative of loss of chondrocyte phenotypic characteristics, 28 and this may predispose individuals to osteoarthritis. 25 The decrease in aggrecan expression may explain the reduction in the percentage of PAS+ matrix, and consequently of proteoglycans, in the group treated with 0.5% v/v ethanol.
Ethanol did not significantly alter the expression of the gene transcript for Sox9 and type II collagen at any of the studied concentrations. Sox9 is an essential transcription factor for initial chondrogenic differentiation, cell proliferation, and for the transcription of genes that induce the expression of type II collagen and other extracellular matrix constituents. 5 In fetal chondrogenesis, the areas of mesenchymal condensation, which expresses Sox9 are sites of synthesis of cartilage, giving rise to the cartilaginous mold that will be replaced by bone. 29 Thus, as the expression of Sox9 did not differ significantly between groups, it is suggested that ethanol may act directly on later stages of chondrocyte maturation. In addition, it is possible that this is the reason why type II collagen also did not differ between groups, since Sox9 induces the expression of type II collagen.
It was found reduction in the expression of Sox9 and type II collagen genes in chondrocytes extracted from individuals with a history of daily ethanol consumption for a period of 20 years. However, the methodology used by the authors was different from ours, since their chondrocytes were extracted from adult individuals who used ethanol, and ethanol was not added to the culture medium of articular chondrocytes extracted from neonates, as in the present study. 25
We conclude that ethanol acts directly on in vitro cultured articular chondrocytes of neonates, altering chondrocyte phenotype and synthesis activity, with reduction of alkaline phosphatase activity, differentiated matrix synthesis, PAS+ cartilaginous matrix, and aggrecan expression, and that these effects are dose dependent.
Footnotes
Acknowledgments and Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo a Pesquisa de Minas Gerais (Fapemig), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethic Approval
All procedures were approved by the Ethics Committee on Animal Use of the Universidade Federal de Minas Gerais, under protocol number 119/2016.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
