Abstract
Objective
The current study aims to investigate the factors that could predict response to intra-articular corticosteroid injection (IACI) in patients with knee osteoarthritis (KOA).
Methods
Data of participants were retrieved from the Osteoarthritis Initiative database. Participants with at least one IACI treatment on single or bilateral knees within the first 5 years of follow-up were retrospectively included. Demographic data, clinical and radiographic variables were collected at both baseline and the first follow-up after IACI treatment. Positive response to IACI treatment was defined as >20% reduction of Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain score from V0 to V1. All the variables with P < 0.2 after the comparison between the response and non-response groups were included in a multivariable logistic regression model to identify independent response predictive patient-specific valuables. Receiver operating characteristic curves were performed to establish the cutoff values of independent predictors.
Results
The current study included a total of 385 participants (473 knees), with 155 and 318 knees classified into the response group and non-response group, respectively. Those with satisfied responses to IACI treatment had significantly higher WOMAC pain score (P < 0.001), disability score (P = 0.002), and stiffness score (P = 0.015) at the baseline. Baseline WOMAC pain score showed significant association with positive response to IACI treatment in multivariate logistic analysis and the best cutoff value was 5 points. The rate of analgesics utilization was lower (P = 0.014) in the response group than the non-response group after the IACI treatment.
Conclusion
KOA patients with a baseline WOMAC pain score ≥5 are more likely to benefit from IACI treatment.
Introduction
Due to its well-proven anti-inflammatory effect on knee synovitis,1,2 intra-articular corticosteroid injection (IACI) is traditionally regarded as an effective treatment for short- to mid-term pain control in patients with early-stage knee osteoarthritis (KOA).3,4 However, not all patients with symptomatic osteoarthritis (OA) could respond to IACI effectively. 5 The pervious study indicated that 73.4% of participants responded to IACI within 2 weeks and only 29% of participants maintained improvement within 6 months. 6 Furthermore, the concern about the safety of IACI was recently raised as the latest research indicated that there was a 3- to 4-fold increased risk of OA radiographic progression and future arthroplasty in KOA patients who underwent IACI treatment.7,8 Given the relatively low response rate and potential risk of long-term complications, it would be more cost-effective to only provide IACI to patients who are most likely to benefit from the therapy in modern-day evidence-based medicine. 9
It was reported that the efficiency of IACI varied between different patient populations, which was largely related to individual differences. 6 Consequently, knowledge of the patient-specific predictors related to better response and lower risk of complications could aid in selecting patients who would benefit more from the IACI therapy and reduce complications associated with IACI. 9 However, there are few published data on this issue, and those studies with conflict results were subject to small sample size and the heterogenization of study design.6,10,11 A systematic review also claimed that it was not possible to pool the results about the predictors of positive response to IACI due to the different predictors studied, variable outcome measures, different criteria for symptom change, and missing data. 9 Thus, the current study aimed to further investigate this issue by using a public database (Osteoarthritis Initiative [OAI]) based on a multi-center, longitudinal observational study. Furthermore, we also investigated whether a positive response to IACI treatment is associated with delayed radiographic progression of KOA patients.
Methods and Materials
Study Design
Data of participants were retrieved from the OAI database, which is available at https://nda.nih.gov/oai. OAI is a multi-center, longitudinal observational study proposed to identify the onset and progression of knee OA, which includes 4,796 participants (9,592 knees). 12 The OAI study was conducted following the ethical principles of the Helsinki declaration. Written informed consent was obtained from all participants, and the ethics review was waived by the local ethics committees.
Participants fulfilling the following criteria at baseline were retrospectively included in the present study: (1) with at least 1 IACI treatment on single or bilateral knees within the first 5 years of follow-up; (2) with intact Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score of the injected knee during the follow-up. The exclusion criteria of the OAI study were (1) rheumatoid arthritis, gouty arthritis, or other inflammatory arthritis; (2) an inability to walk without aids; (3) participants with bilateral end-stage knee osteoarthritis, and the exclusion criteria of the current study were the presence of a WOMAC pain score = 0 in the injected knees at the last follow-up before IACI treatment.
Those with a “Yes” response to the query “Have you had a treatment with injections of corticosteroid in either of your knees (right, left, both) for your arthritis during the past 6 months?” at any of the OAI visits was defined as patients undergoing IACI treatment. Unfortunately, further details of IACI treatment including the concrete type of glucocorticoid, dosage, and the treatment frequency are unavailable due to the lack of origin record in the OAI database. For those who received more than 1 course of IACI treatments during their follow-up in the OAI, we selected the first IACI treatment for further analysis. All participants were followed yearly after being registered in the OAI database. In the present study, baseline visit (V0) was defined as the latest follow-up before the IACI treatment, while the end point visit (V1) was defined as the first follow-up after IACI treatment.
Baseline (V0) demographic data including age, gender, body mass index (BMI), Charlson comorbidity index (CCI), family arthroplasty history, knee injury history, maximum flexion and extension force, physical summary scale (PSS), and mental summary scale (MSS) that obtained for the Medical Outcome Study 12-item short-form health survey, as well as the physical activity scale for the elderly (PASE) were retrieved from the OAI database as we previously described. 10 Pain, stiffness, and disability that quantified by WOMAC score (0-20), Kellgren-Lawrence grade (KLG), medial and lateral joint space narrowing (MJSN and LJSN) that evaluated by Osteoarthritis Research Society International (OARSI) grades, and concomitant medication that might alleviate arthritis symptoms at both V0 and V1 were also recorded.
Patients’ response to IACI treatment was defined as positive when there was a >20% reduction of WOMAC pain score from V0 to V1, according to the Osteoarthritis Research Society International set of responder criteria for osteoarthritis clinical trials revisited. 13 Subsequently, all the involved knees were divided into response and non-response groups before further analysis. The progression of KLG, MJSN, or LJSN from V0 to V1 was defined as positive when there was an upgrade of these scores. The time between IACI therapy and structural progression assessment varied from 1 to 6 months.
Statistical Analysis
Category data were summarized as frequency, and chi-square or Fisher’s exact tests were utilized to compare the category data between response and non-response group. Continuous data with normal distribution were expressed as mean ± standard deviation, while continuous data with obviously skewed distribution were expressed as median with interquartile. Student’s t tests or Mann–Whitney’s U tests were applied for comparisons for continuous data according to the distribution. Subsequently, all the variables with P < 0.2 after the comparison between the response and non-response groups were included in a multivariable logistic regression model to identify independent response predictive patient-specific valuables. 10 Receiver operating characteristic (ROC) curves were performed to establish the cutoff values of independent response predictors. The area under the curve (AUC) was also applied to evaluate the predictive performance of the selected variables. All tests were two-sided and P < 0.05 was set as statistically significant. All analyses were performed using R 4.0.4 (The R Foundation for Statistical Computing) with the packages “CBCgrps” and “rms.” 14
Results
Demographic and Clinical Data
In total, 385 participants (473 knees) were included in the current study after screening ( Fig. 1 ). Demographic and clinical data of all the participants are shown in Table 1 , with 155 and 318 knees classified into the response group and non-response group, respectively. As shown in Table 1 , those with satisfied responses to IACI treatment were prone to be older (P = 0.121) and thinner (P = 0.151), as well as with significantly higher WOMAC pain score (P < 0.001), disability score (P = 0.002), and stiffness score (P = 0.015) at the baseline. Although the concomitant arthritis medication showed no difference at the baseline of our study, the rate of analgesics utilization was significantly lower (P = 0.014) in the response group than the non-response group after the IACI treatment ( Table 2 ).
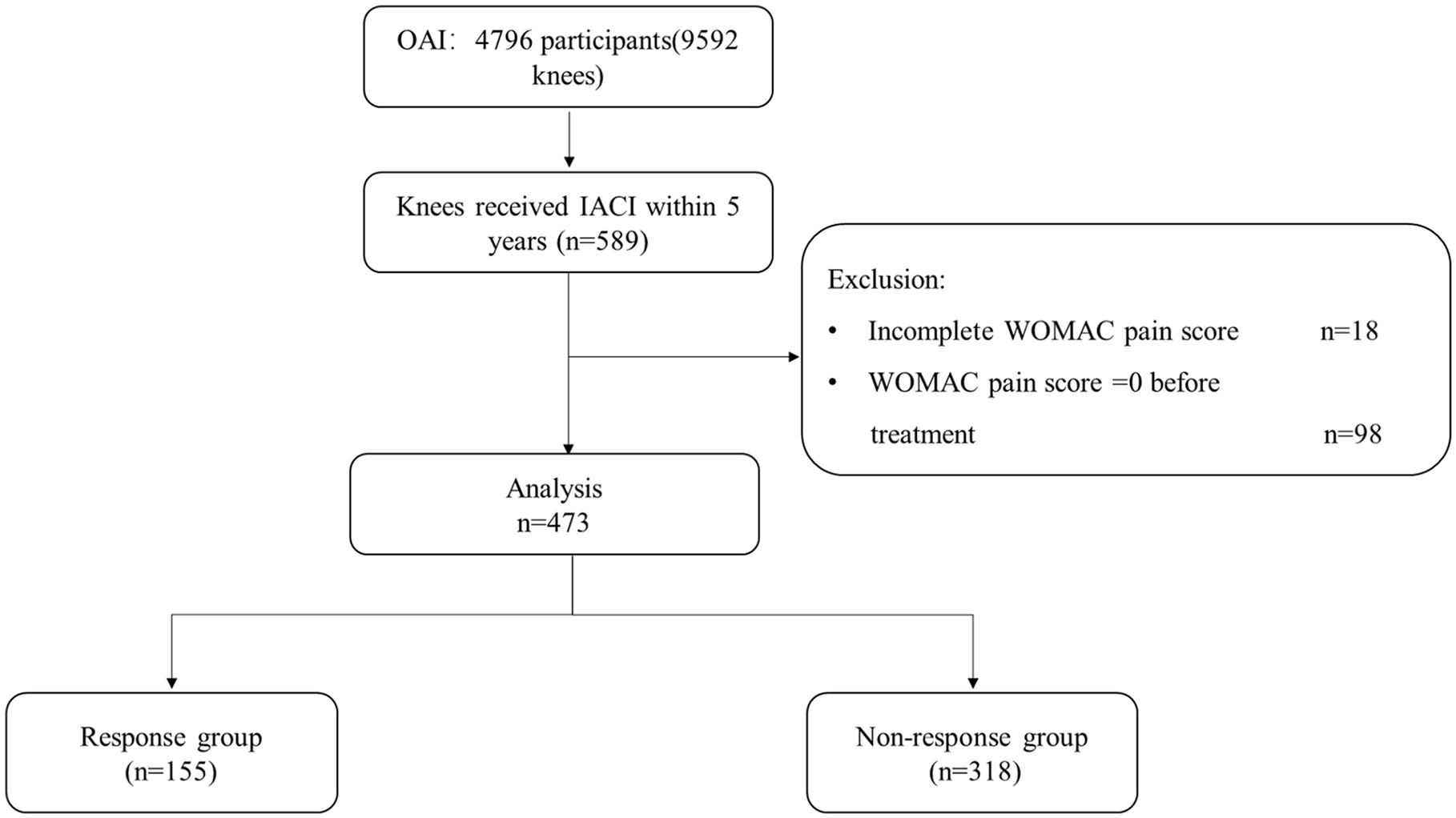
Flowchart of the current study. OAI = Osteoarthritis Initiative; IACI = intra-articular corticosteroid injection; WOMAC = Western Ontario and McMaster Universities Osteoarthritis Index.
Baseline Demographic and Clinical Data.
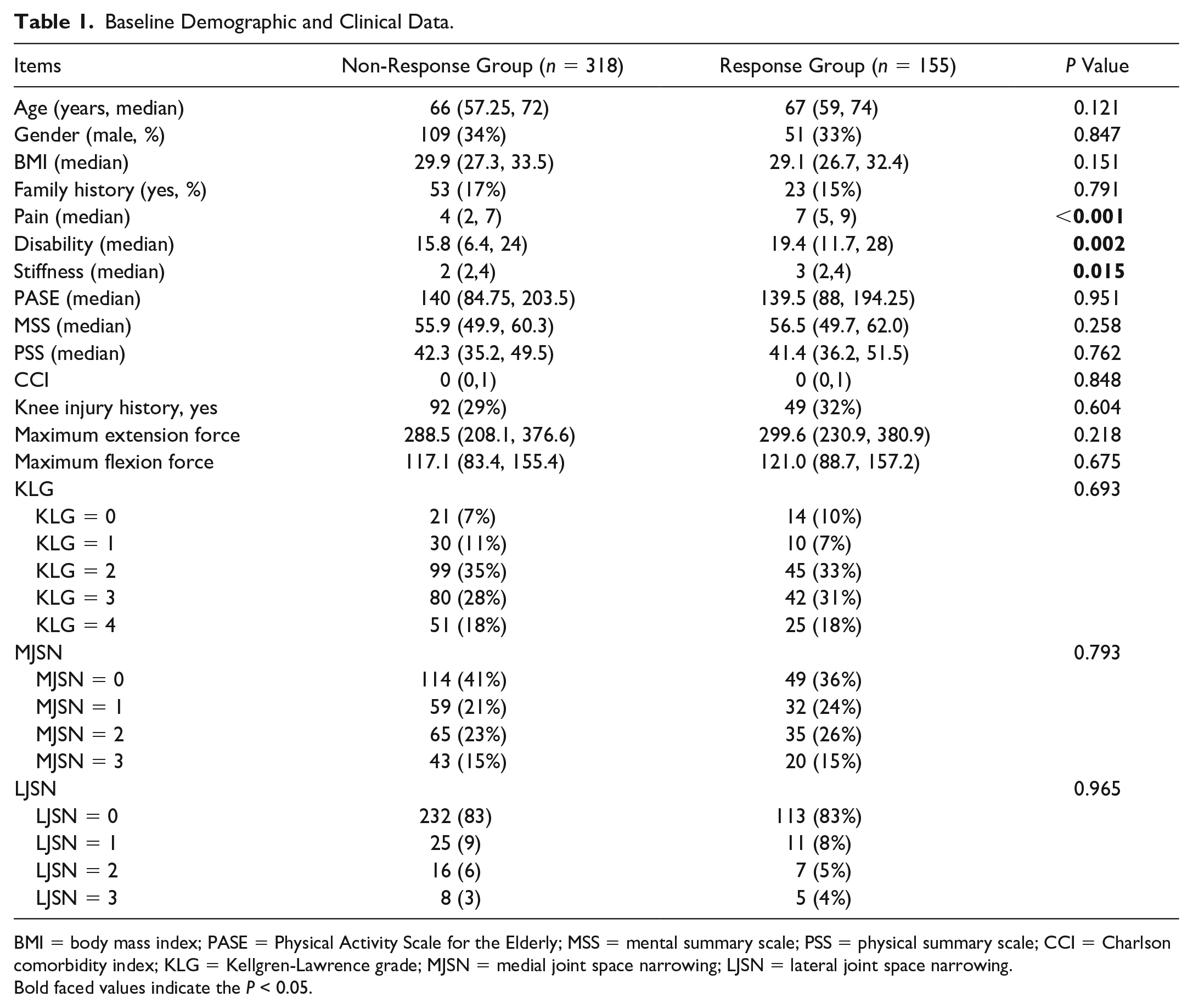
BMI = body mass index; PASE = Physical Activity Scale for the Elderly; MSS = mental summary scale; PSS = physical summary scale; CCI = Charlson comorbidity index; KLG = Kellgren-Lawrence grade; MJSN = medial joint space narrowing; LJSN = lateral joint space narrowing.
Bold faced values indicate the P < 0.05.
Concomitant Arthritis Medication before and after Intra-Articular Corticosteroid Injections Treatment.
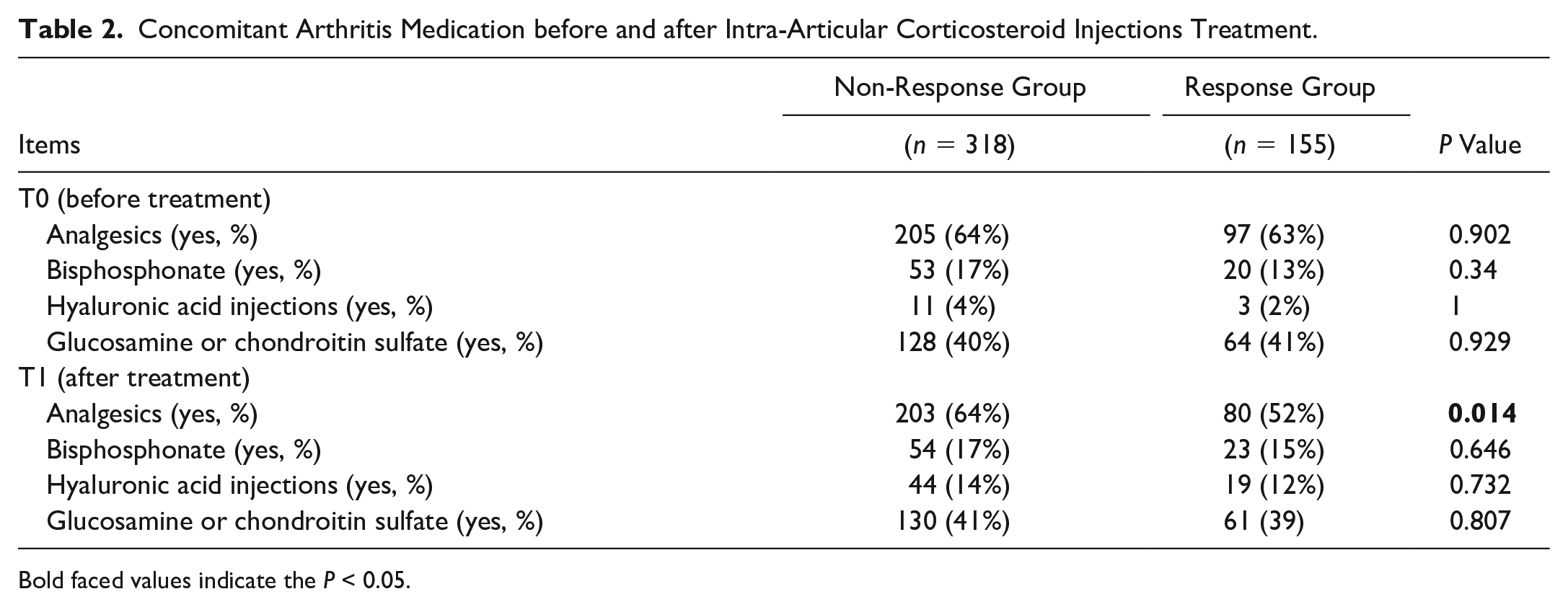
Bold faced values indicate the P < 0.05.
Predictive Performance of WOMAC Pain Score
Six variables including age, BMI, WOMAC pain, stiffness, disability score, and the utilization of analgesics at V1 were selected for the multivariable logistic regression analysis. Baseline WOMAC pain score and the utilization of analgesics after IACI treatment showed significant association with positive response to IACI treatment according to the results of multivariate logistic analysis ( Table 3 ).
Multivariable Logistic Regression for Variables with P < 0.2 in Univariable Analysis.
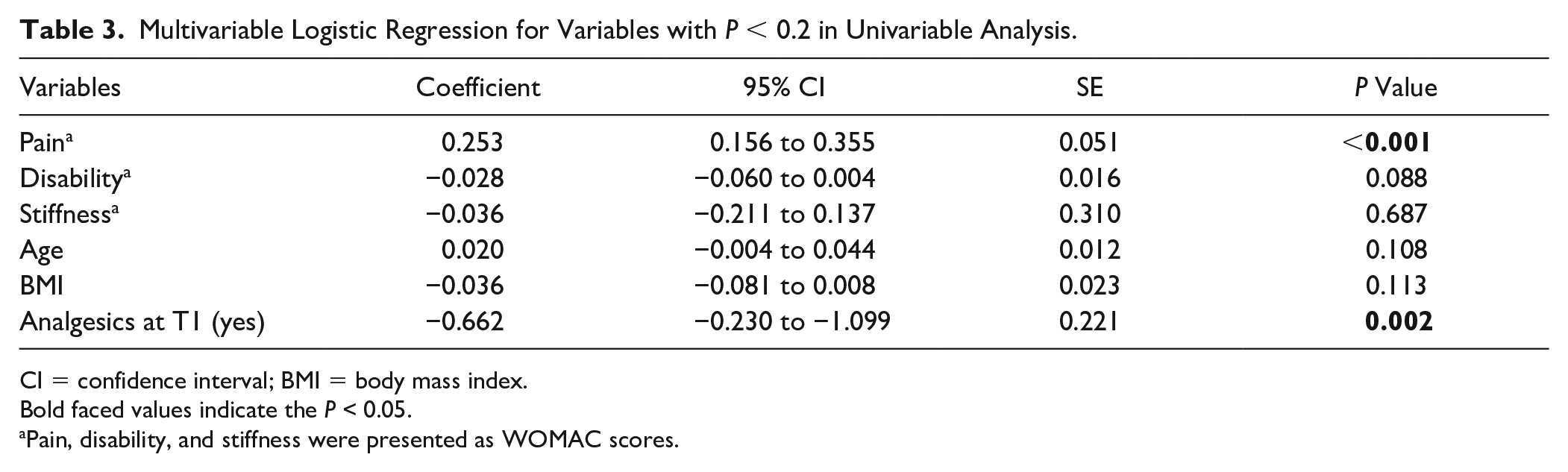
CI = confidence interval; BMI = body mass index.
Bold faced values indicate the P < 0.05.
Pain, disability, and stiffness were presented as WOMAC scores.
As shown in Figure 2 , the AUC for WOMAC pain score was 0.67. The best cutoff value of baseline WOMAC pain score was 5, with the specificity and the sensitivity being 54.4 and 76.1%, respectively. Among the 473 knees with symptomatic OA in the current study, 210 knees (44.3%) had the baseline WOMAC pain score < 5 points. And patients with a WOMAC pain score ≥ 5 points showed a 2.54 times higher positive response rate to IACI (44.8% vs 17.6%, P < 0.001, odds ratio: 3.81, 95% confidence interval: 2.48-5.85).
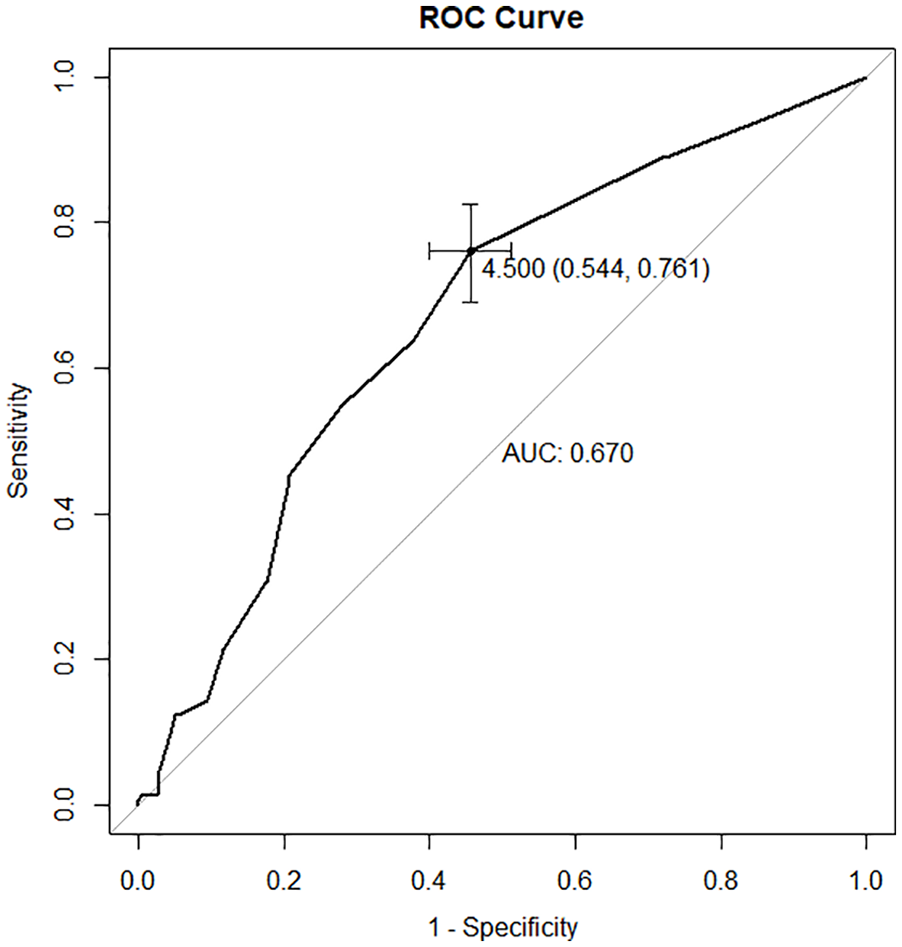
The receiver operating characteristic curve (ROC) showing the area under the curve (AUC), specificity and sensitivity for utilizing Western Ontario and McMaster Universities Osteoarthritis Index pain score to predict response of intra-articular steroid injection in knee osteoarthritis.
The Association between a Positive Response to IACI Treatment and Structure Progression of KOA
As shown in Table 4 , there was no significant difference in the structural progression of KOA between response and non-response groups from V0 to V1.
Structure Progression of Patients Received IACI from V0 to V1.

IACI = intra-articular corticosteroid injections; KLG = Kellgren-Lawrence grade; MJSN = medial joint space narrowing; LJSN = lateral joint space narrowing.
Discussion
IACI is recommended and commonly practiced to relieve symptoms in patients with knee OA; however, response to steroid injections is heterogeneous as some patients respond very well and some have a poor response.11,15 So far, the studies concerning the factors determining response to IACI are scarce and few predictors were identified, make it hard to target patients who are more likely to respond to IACI therapy. 9 As far as we know, our study represented the first time to show that baseline WOMAC pain score was predictive for a long-term positive response to IACI treatment, with the best cutoff being 5 points. Meanwhile, nearly 33% (155/473) of the participants maintained their improvement of pain relief in the latest 6 months after IACI treatment, which is comparable to a previous study (29%). 16
Although several studies previously reported the association between baseline knee pain and the response to IACI treatment in KOA patients, their results remain controversial, and the level of evidence of most studies was restricted to the limited sample size. Pendleton et al. 17 reported that a higher baseline pain predicted a better response to IACI within the first 6 weeks via a prospective clinical trial involving 86 patients, which was consistent with our finding. An individual patient data meta-analysis also found that patients with severe baseline pain were found to benefit more from a steroid injection at short-term follow-up (<4 weeks) than those with less severe pain. 18 But other studies with the sample size between 84 and 199 showed opposite results.19,20 As the current study was conducted on basis of a relatively larger sample size (473 knees) and a more comprehensive list of clinical predictor factors from a public database (OAI) with 4793 participants, we suggested that our study provided relatively strong evidence.
Furthermore, we also considered the potential confounding effect of concomitant arthritis medication on patients’ response to IACI treatment, which has not yet been considered in the former-mentioned studies. Based on our results, the participants who had positive response to IACI treatment were more likely to withdraw analgesics treatment. In other words, some participants who actually responded to IACI might probably withdraw analgesics treatment due to a totally or partial pain relief, which would subsequently lead to an underestimation of the efficiency of IACI treatment. Moreover, that might be one of the possible causes of the conflicting results among previous studies.
Inflammation plays an essential role in the development of knee pain in OA as it triggers cytokines and chemokines release to participate in pain generation, 21 which has long been considered as a therapeutic target of KOA. 22 Besides, the nociceptive system develops a state of hyperexcitability and might be activated by normally innocuous stimuli in inflammatory conditions. 23 Steroids act as effective local anti-inflammatory medications and are thought to counteract the inflammatory processes in KOA by altering T-cell and B-cell immune function. 24 Interestingly, it was reported that the synovial inflammation was closely related with the pain score in KOA patients, indicating that the knee pain score might partially represent the severity of inflammation. 25 That might be a reasonable explanation for why the typical anti-inflammation treatment—IACI showed better effect on patients with higher baseline knee pain in our study.
To keep the balance between benefit and potential harm, the appropriate pain threshold for IACI treatment was urgently needed in OA management. Nevertheless, no study or guideline has ever proposed a specific pain criterion for IACI treatment. The indication of IACI treatment was mainly based on the experience of physicians and the requirement of patients. To the best of our knowledge, our study represented the first time to proposed a reasonable cutoff value of baseline knee pain score to select candidates who would be more likely to benefit from IACI treatment, which we believed to be more practical when making clinical decisions.
We also found that 98 of 589 IACI treatment knees had a zero WOMAC pain score at baseline in the OAI database ( Fig. 2 ), which we supposed that IACI might be applied to reduce the inflammation and prevent OA progression in those patients. However, the previous study reported patients receiving IACI therapy did not show delayed structure progression in OA. 26 In the current study, we further compared responders with non-responders in structure progression to determine whether positively responding to IACI could modify the OA progression. The patients shared similar baseline OA grades and factors affecting the structural progression of OA (age, BMI, etc.). Furthermore, it was the first time the involved participants received IACI, which meant that no previous injection would potentially affect the progression of knee OA. The results showed that there was no difference in structure progression from V0 to V1 between the response and non-response groups, which suggested that IACI could not modify the progression of OA even in patients positively responding to the therapy. The local inflammation in OA was more likely to be a simple epiphenomenon that was elicited by joint destruction rather than the factor directly contributing to the disease progression. 22 Thus, we suggested the IACI treatment, which was only effective in alleviating symptoms but was suspected to accelerate cartilage loss and even OA progression, should not be over practiced in the clinic.
Our study subject to several limitations. First, the type of injected corticosteroid, the operator, and the dose varied among participants in the OAI study. It has been reported that triamcinolone could be more effective when compared with other injectable steroids. 27 Thus, the heterogeneity of corticosteroid dosage and the operators in the present study might affect the outcome of IACI treatment. Second, as we mentioned before, the evaluation of IACI injections was based on the question: “Have you had a treatment with injections of corticosteroid in either of your knees (right, left, both) for your arthritis during the past 6 months.” Thus, the exact time from IACI treatment to the last follow-up was unknown. Moreover, the number of IACI was also not queried in the OAI, which would lead to potential bias as some participants might receive more than 1 dosage of IACI during the annual visit. Third, the examination of synovial fluids was not included in the OAI study, and the relative data were unavailable in our study. The examination of synovial fluids should be performed in future studies, which aim to identify predictors for positive response of IACI in KOA patients. Finally, some variables such as a positive belief to IACI therapy, knee effusion, and synovitis that were reported as the candidate predictive factors were unavailable in the current study due to the lack or incompleteness of original data in OAI database.17,19 A larger-scale prospective cohort or randomized control trial was required to explore and validate more predictors of IACI treatment response to build a predictive model with higher accuracy.
Conclusion
Baseline knee pain is the independent predictor for the response of intra-articular steroid injection in KOA patients after adjusting for age, BMI, stiffness, disability, and concomitant analgesics. KOA patients with a baseline WOMAC pain score ≥5 are more likely to benefit from IACI treatment. Responders to IACI treatment shared a similar risk of knee structure progression with the non-responders.
Footnotes
Acknowledgments and Funding
The OAI is a public-private partnership comprising 5 contracts (N01-AR-2-2258; N01-AR-2-2259; N01-AR-2-2260; N01-AR-2-2261; N01-AR-2-2262) funded by the National Institutes of Health, a branch of the Department of Health and Human Services, and conducted by the OAI Study Investigators. Private funding partners include Merck Research Laboratories; Novartis Pharmaceuticals Corporation, GlaxoSmithKline; and Pfizer, Inc. Private sector funding for the OAI is managed by the Foundation for the National Institutes of Health. This article was prepared using an OAI public use data set and does not necessarily reflect the opinions or views of the OAI investigators, the NIH, or the private funding partners. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Guangdong Basic and Applied Basic Research Foundation (2021A1515110458, 2022A1515011103), NSFC Incubation Program of GDPH (KY012021163), Natural Science Foundation of Guangdong Province (2021A1515011008), and the Program of Science and Technology of Guangzhou (201904010424).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The OAI study was conducted following the ethical principles of the Helsinki declaration. Written informed consent was obtained from all participants and the ethics review was waived by the Guangdong Provincial Peoples’ Hospital.
