Abstract
Objective
To investigate the presence of WNT antagonists Dickkopf-related protein 1 (DKK1), Frizzled-related protein (FRZB) and BMP antagonist Gremlin 1 (GREM1) in synovial fluid (SF) and serum, respectively, from end-stage knee osteoarthritis (OA) patients, and correlate their expression with other markers of OA.
Design
In a cross-sectional study, SF and serum were collected from OA patients (n = 132). The concentrations of DKK1, FRZB and GREM1 in SF and serum were determined using immunoassays. Correlation measurements were performed between groups and previously assessed disease markers, such as synovium nitric oxide (NO), inerleukin-1β (IL1β), tumor necrosis factor–α (TNFα), and prostaglandin E2 (PGE2).
Results
The OA patients with the celecoxib treatment till surgery have higher median SF FRZB values compared with the control (no treatment); the celecoxib 3-days before surgery stopped treatment group has higher median serum FRZB values than the control and the naproxen treatment group. The combinational analysis of SF DKK1 and SF FRZB negatively correlated with macroscopic cartilage scores and histological synovium scores in OA patients. The expression of DKK1 and FRZB in SF showed the same expression trend as their expression in serum. Furthermore, the SF concentration of DKK1 was positively correlated with FRZB in both SF and serum. In contrast, it was negatively correlated with synovium NO and IL1β. SF FRZB was negatively correlated with synovium NO, IL1β, cartilage PGE2, and age.
Conclusions
Our findings suggest DKK1 and FRZB were negatively correlated with OA severity and multiple pro-inflammatory cytokines. Our data indicate that DKK1 and FRZB can be joint disease–specific biomarkers.
Keywords
Introduction
Osteoarthritis (OA) is the most common chronic joint disorder, characterized by progressive degradation of joint cartilage, formation of bony tissue at the joint margins (osteophytes), subchondral bone changes (sclerosis), inflammation of the synovial membrane, and impairment of the periarticular soft tissues. 1 Although large research effort has been devoted in recent years to explore the molecular mechanism underlying this disease, the pathogenesis of OA is still largely unknown and no effective treatment is available.
Multiple signal pathways have been implicated in the pathogenesis of OA, such as the alteration of secreted wingless-type MMTV integration site (WNT) and the bone morphogenetic protein (BMP) signal pathways.2,3 In articular cartilage, moderate activity of WNT is essential for chondrocyte proliferation and cell phenotype, 4 while overexpression and dysregulation can lead to cartilage disease.5,6 BMPs are widely known as potent inducers of bone. Increased expression of BMP2 has been reported in OA chondrocytes and cartilage. 7
WNT and BMP activators and inhibitors could therefore play an important role in OA pathogenesis. Multiple whole genome studies have shown that the WNT antagonist Frizzled-related protein (FRZB) is a candidate gene associated with hip OA in females. 8 This association of FRZB with susceptibility for OA was further confirmed in other cohorts.9,10 Dickkopf-related protein 1 (DKK1), another WNT antagonist, was associated with reduced progression of radiographic hip osteoarthritis in elderly women. 11 This is in line with our previous study showing that DKK1 and FRZB have protective roles in cartilage by preventing hypertrophic differentiation 12 and the loss of their expression is observed in OA. 13 Considering the role of the BMP pathway, it has been shown that upregulation of GREM1, which is a BMPs antagonist, is observed in human OA chondrocytes 14 and its expression increases with OA progression in dog OA. 15 This indicates there might be an involvement of GREM1 in OA pathophysiology.
Even though OA is not widely seen as an inflammatory disease like rheumatic arthritis, serological and histological evidence of synovitis is commonly found in OA. 16 In fact, synovial inflammation can be involved in the early onset of OA as well as in advanced stages of OA.17,18 Interleukin-1β (IL1β) and tumor necrosis factor–α (TNFα) play a key role in the destructive process. These proinflammatory cytokines stimulate synthesis and release of nitric oxide (NO) and prostaglandin E2 (PGE2), which are produced in elevated amounts during the degenerative process, contributing to joint pathology.17,19
In this study, a comprehensive cross-sectional analysis was performed in synovial fluid (SF) and serum samples collected from OA patients. We measured the concentration of DKK1, FRZB, and GREM1 in patients with or without pharmaceutical treatment in SF and serum and correlated their expression levels with multiple OA features (macroscopically, histologically, and biochemically) and inflammatory factors. We hypothesize that the concentration of Wnt natural antagonists DKK1, FRZB, and BMP antagonist Gremlin 1 in SF and serum could reflect joint inflammation and the disease severity of OA patient to some extent.
Materials and Methods
Subjects
Material of 132 patients with severe knee osteoarthritis, who were eligible for total knee replacement surgery, could be obtained. These samples have been used in previous investigations. 20 The study was conducted according to the Declaration of Helsinki and received ethical approval. Each patient gave written informed consent before participating in the study.
Originally patients were randomized to 1 of 4 treatment groups 4 weeks prior to total knee replacement surgery. Patients were randomized to celecoxib 2dd200mg (until surgery), celecoxib 2dd200mg until 3 days before surgery (to control for the obligated stop of naproxen treatment 3 days before surgery), and naproxen 3dd250mg until 3 days before surgery, or no treatment (controls). Because of its platelet-inhibiting effect, the use of naproxen had to be stopped 3 days prior to surgery. All 4 groups included 43 patients originally. Because of the increased risk for gastrointestinal adverse effects with the use of naproxen, all patients also received omeprazol 20 mg once a day. SF and serum samples in combination with complete data as collected in the original collected data was available for 132 patients. Specific characteristics are given in Table 1 .
Characteristics of the Study Subjects.

Macroscopy and Histochemistry
Macroscopic cartilage damage was evaluated on digital high-resolution photographs of femur surface parts. Severity of cartilage damage was graded from 0 to 3 (0 = fibrillation or focal degeneration, 1 = degeneration at multiple locations, 2 = degeneration at multiple locations with focal lesions, and 3 = degeneration throughout the tissue with severe focal lesions and focally full cartilage abrasion). Synovial tissue inflammation was evaluated by histochemistry. Synovial tissue inflammation was graded using modified Goldenberg and Cohen scores. 21 Two observers blinded to the source of the samples, scored all samples and the averages of observers were taken as representative score of each donor. In the limited case, when observers scored >1-point difference, consensus was sought. The average of all multiple tissue samples (n = 4 for cartilage histology, n = 3 for synovial tissue histology, and n = 3 for cytokine production) of each donor was taken as representative value for each donor. Data are subsequently expressed as mean (±SD) of donor.
Analysis of DKK1, FRZB, and GREM1 in Synovial Fluid and in Serum
SF was collected during knee replacement surgery, aliquoted and stored at −80°C. Blood samples were obtained shortly before the surgery, serum was separated, and aliquots stored at −80°C. SF and serum concentrations of DKK1, FRZB, and GREM1 were determined using enzyme-linked immunosorbent assay (ELISA) according to the manufacturer guidelines (DKK1, Catalog Number: DY1906, R&D system; FRZB, Catalog Number: DY192; GREM1, Bio-connect diagnostics, Catalog Number: E01G0253).
All samples were diluted 1:2 for DKK1 and FRZB detection, and 1:24 for GREM1 detection according to the sample specificity and manufacture protocols. Randomly selected knee SF and serum samples were used to assess dilution linearity (n = 3), effects of freeze-thawing (n = 5) and spiking recovery (n = 3). Details on the technical performance of the DKK1, FRZB, and GREM1 ELISA in SF and serum in terms of dilution linearity and spiking recovery, intra- and interassay coefficients of variation, and the influence of thawing and freezing of samples is referred to Huang et al. 22 (for SF) and presented in Supplementary Data Table S1 (for serum).
Analysis of Pro-Inflammatory Cytokines, PGE2, and NO in Cartilage and Synovial Tissue Culture Medium
To assess the IL1β and TNFα production by the OA cartilage and synovial tissue, samples were taken (n = 8 randomly taken from femoral cartilage per donor; n = 2 (medial and lateral) per donor for infrapatellar synovial tissue) and cultured individually ex vivo for 3 days, and subsequently IL1β and TNFα release in the culture supernatants were determined by ELISA (Biosource) according to manufacturer’s instructions and expressed as pg/mL per mg (wet weight) tissue. PGE2 was determined by Enzyme Immuno Assay (EIA, Caymann Chemical) and expressed as pg/mL per mg tissue. NO levels were determined using the standard Griess reaction and expressed as μM per mg (wet weight) of tissue.
Clinical Outcome
Patients were asked to fill out a Western Ontario and McMaster Universities Arthritis Index (WOMAC) questionnaire to evaluate pain, stiffness, and function, before and after medical treatment. 23
Statistics
Nonparametric analyses were conducted in this study. Between-group comparisons were made using Mann-Whitney U tests, and for correlation analysis, Spearman’s rank correlation (R) analysis was used. Pearson chi-square test was used for comparison of genders between subject groups, and for comparison of age, Student t test was used. All tests were 2-tailed, P < 0.05 was considered statistically significant, P values <0.1 were considered borderline significant. All statistical analyses were performed using SPSS (standard version 15.0, SPSS, Chicago, IL, USA).
Results
OA Patients with Celecoxib Treatment Have Higher SF FRZB Median Value Compared with the Control (No Treatment) Group
In order to detect changes of the antagonist’s expression in relation to the medicine treatment for OA patients, we measured the concentrations of FRZB, DKK1, and GREM1 in SF and in serum by ELISA. Analysis of SF FRZB revealed that OA patients with celecoxib treatment have a 2 times higher SF FRZB median value (2207.55 pg/mL) compared with the control (no treatment) group (1086.45 pg/mL; P = 0.049). The group treated with celecoxib until 3 days before surgery had 1.97 times higher serum FRZB median value (533.1 pg/mL) than the control (270.2 pg/mL; P = 0.003) and 2.14 times higher than the naproxen treatment group (248.6 pg/mL; P = 0.034). However, there is no significant difference for expression levels of DKK1 and GREM1 among different treatments ( Fig. 1 ).
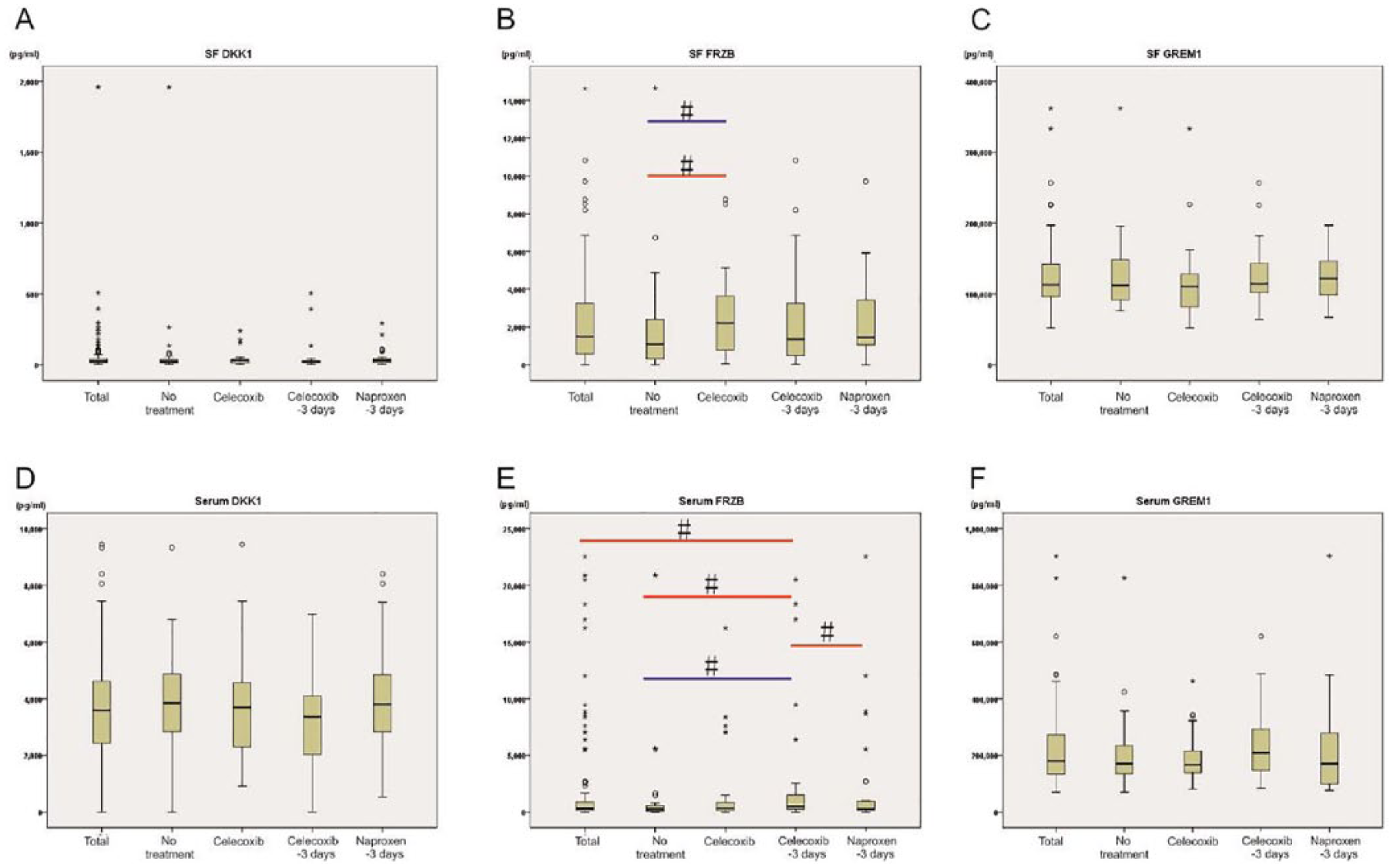
Concentrations of DKK1, FRZB, and GREM1 in synovial fluid (SF) and serum in no treatment group, celecoxib-full group (Celecoxib), celecoxib–3 days group, and naproxen–3 days group. Samples were grouped according to Table 1 . The results are expressed as median and the 25th and 75th percentiles with whiskers (10th and 90th percentiles). Circles and asterisks stand for discrete values and extreme values, respectively. The red line means median difference and blue line means distribution difference. FRZB = frizzled-related protein; GREM1 = Gremlin 1; DKK1 = Dickkopf-related protein.
DKK1 and FRZB Are Expressed in Serum and Have the Same Expression Trend as Their Expression in Synovial Fluid from OA Patients
Next, we investigated the relation between expression in SF and the expression in serum for the antagonists. The DKK1 concentration median (range) is 26.9 (3.9-1958.9) pg/mL in SF and 3589.7 (3.9-9452.5) pg/mL in serum. For FRZB, this is reversed where the concentration median (range) is 1486.7 (3.6-14635.8) pg/mL in SF and 332.7 (3.6-22539.8) pg/mL in serum. The GREM1 concentration median (range) seems more similar for both compartments: 112.9 (51.9-362.1) ng/mL in SF and 179.89 (70.0-903.4) ng/mL in serum. After statistical comparison, we found that both DKK1 and FRZB have a similar expression trend in SF and serum (R = 0.305, P < 0.001; R = 0.300, P = 0.001). However, for GREM1, there was no correlation found ( Fig. 2 , Table 2 ).
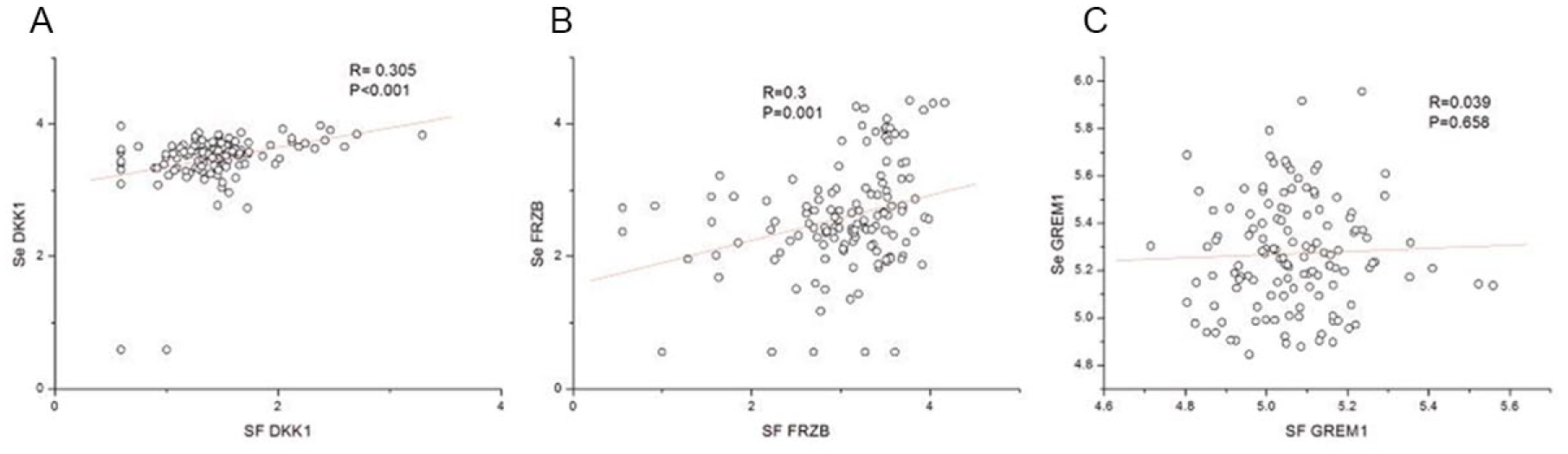
Correlation of concentration of DKK1 and FRZB between synovial fluid (SF) and serum. A bivariate scatter plot of DKK1 and FRZB concentrations (showed in denary logarithm) in synovial fluid with the corresponding concentrations in serum (n = 132). Spearman coefficient (R), P values and the first-order regression lines are indicated. FRZB = frizzled-related protein; DKK1 = Dickkopf-related protein
Correlation between DKK1, FRZB, GREM1, in Synovial Fluid or Serum. a
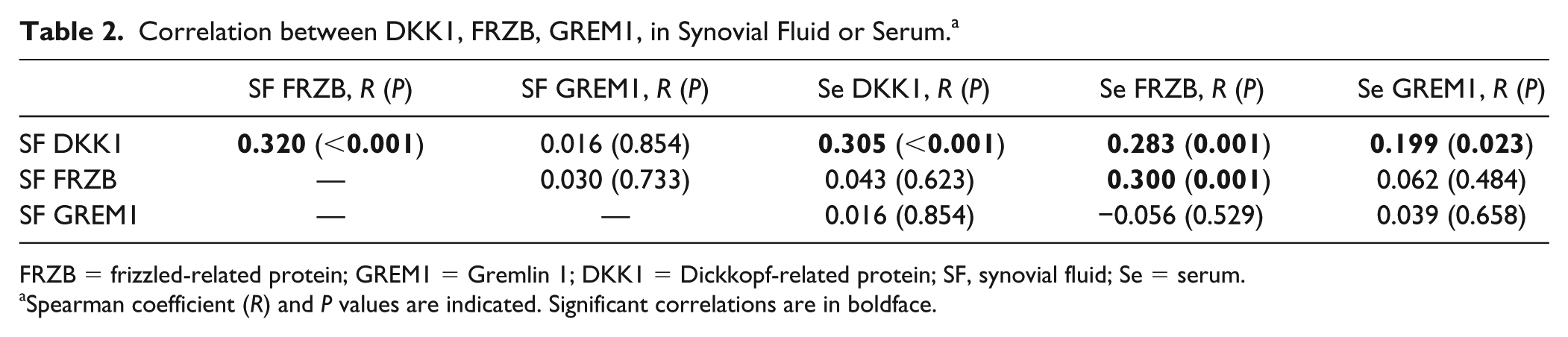
FRZB = frizzled-related protein; GREM1 = Gremlin 1; DKK1 = Dickkopf-related protein; SF, synovial fluid; Se = serum.
Spearman coefficient (R) and P values are indicated. Significant correlations are in boldface.
SF DKK1 and SF FRZB Are Negatively Correlated with OA Severity
In order to assess the severity of joint damage of the OA patients, directly after surgery macroscopic analysis and histochemistry were performed for the collected tissues. We found that both SF DKK1 and SF FRZB have a minor negative correlation with the macroscopic cartilage score (R = −0.169, P = 0.056; R = −0.163, P = 0.065). Moreover, if SF DKK1 and SF FRZB log values were combined together, there was a stronger significant negative correlation with the microscopic score (R = −0.217, P = 0.014) ( Fig. 3a ). Moreover, we assessed the correlation with the histological synovial inflammatory scores. It was found that DKK1 in SF significantly negatively correlated with the histologic synovium score (R = −0.255, P = 0.006) ( Fig. 3b , Supplementary Fig. S1). Similarly, if SF DKK1 and SF FRZB log values were combined together, there was a significant negative correlation with the histologic synovium score as well (R = −0.202, P = 0.031) ( Fig. 3c ). When the serum compartment was studied only GREM1 demonstrated a mild negatively correlation with the score (R = −0.155, P = 0.099) ( Table 3 ).
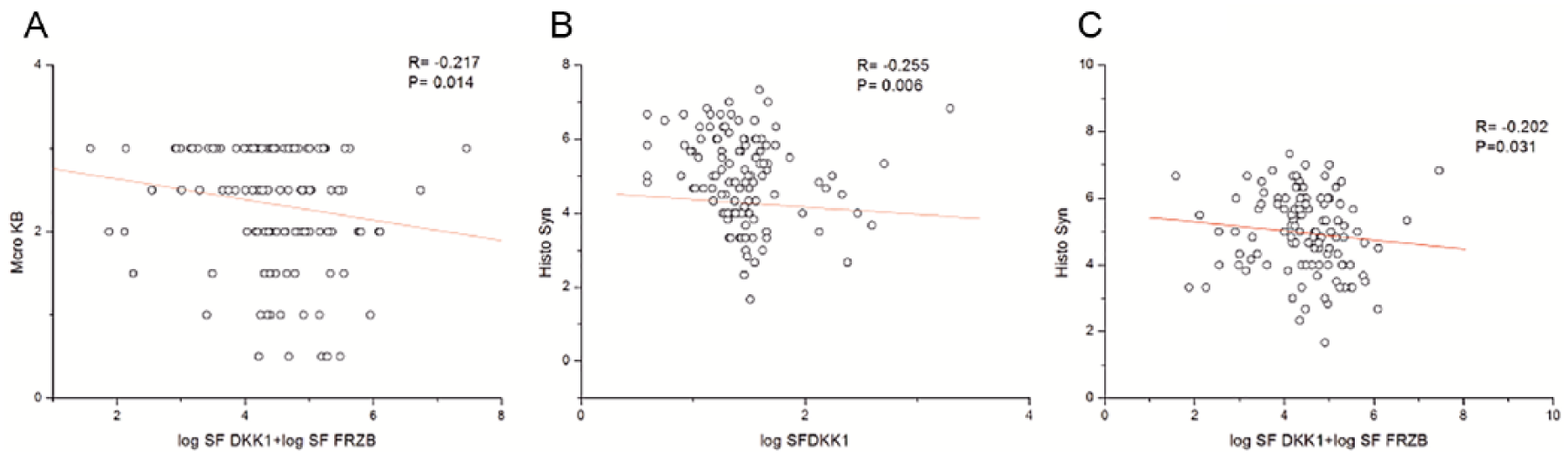
Correlation between DKK1, FRZB, and GREM1 in synovial fluid (SF) and severity of osteoarthritis (OA). A bivariate scatter plot of DKK1, FRZB, and GREM1 concentrations in SF (n = 132) with the scores of cartilage damage (Marco KB) and synovium inflammation (Histo syn). Spearman coefficient (R), P values, and the first-order regression lines are indicated. FRZB = frizzled-related protein; GREM1 = Gremlin 1; DKK1 = Dickkopf-related protein.
Correlation between Synovial Fluid or Serum DKK1, FRZB, GREM1, and Macro KB, Histo Syn. a
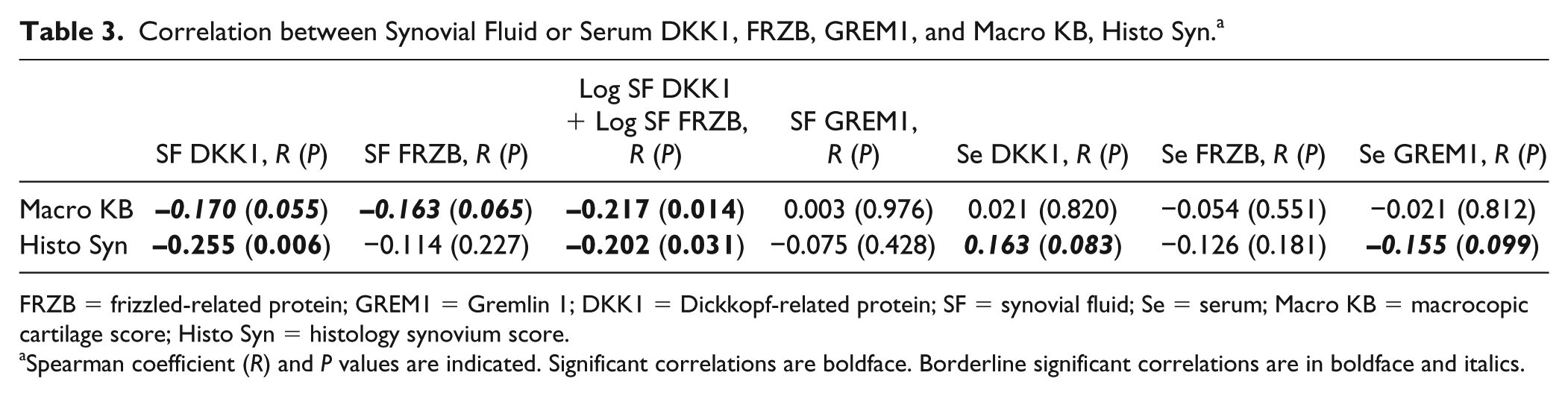
FRZB = frizzled-related protein; GREM1 = Gremlin 1; DKK1 = Dickkopf-related protein; SF = synovial fluid; Se = serum; Macro KB = macrocopic cartilage score; Histo Syn = histology synovium score.
Spearman coefficient (R) and P values are indicated. Significant correlations are boldface. Borderline significant correlations are in boldface and italics.
The Expression of DKK1 in SF Shows Positive Correlation with FRZB in Both SF and Serum, and with GREM1 in Serum
Since all 3 factors (DKK1, FRZB, and GREM1) are involved in the joint homeostasis, we assessed the correlation between these factors in SF and serum as a next step. The results showed that SF DKK1 positively correlated with FRZB in both SF (R =0.32, P < 0.001) ( Fig. 4a ) and serum (R = 0.283, P = 0.001) ( Fig. 4b ). Interestingly, the SF DKK1 concentration was also positively correlated with GREM1 in serum (R = 0.199, P = 0.023) ( Fig. 4c , Table 2 ).
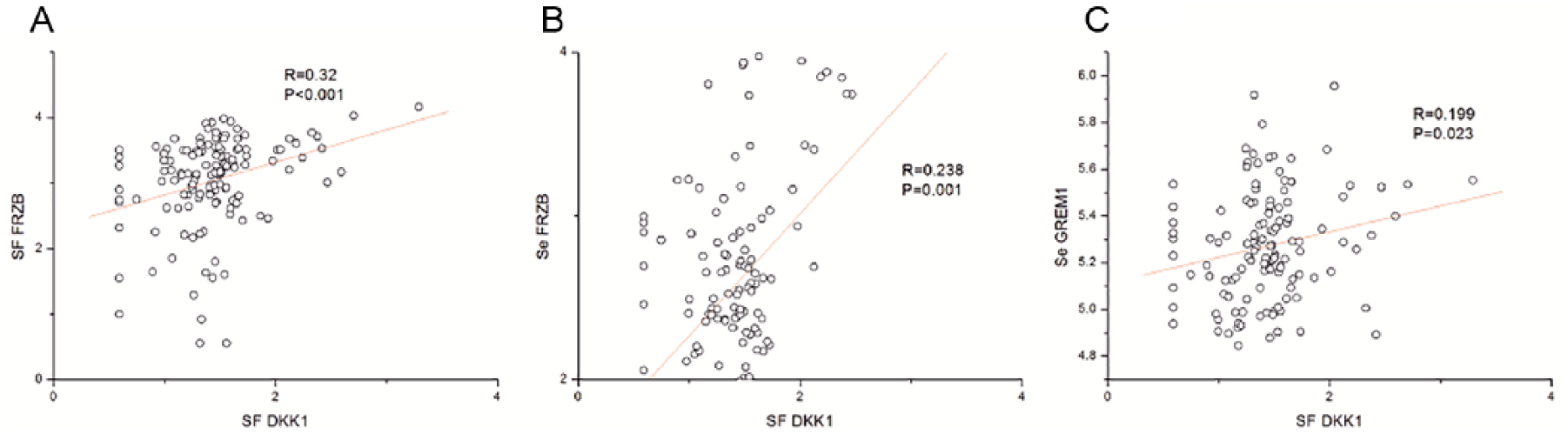
Correlation between concentration of DKK1, FRZB, and GREM1 in synovial fluid (SF) and serum. A bivariate scatter plot of DKK1 concentrations in synovial fluid with the FRZB concentration both in SF and serum or GREM1 concentration in serum (n = 132). Spearman coefficient (R), P values and the first-order regression lines are indicated. FRZB = frizzled-related protein; GREM1 = Gremlin 1; DKK1 = Dickkopf-related protein.
The Expression Levels of DKK1 and FRZB, but Not of GREM1, Are Negatively Correlated to Pro-Inflammatory Factors
In order to assess the correlation between the concentration of SF DKK1, FRZB, GREM1 and local inflammation, the production of pro-inflammatory factors IL-1β and TNFα by the corresponding OA cartilage and synovial tissue was studied. 20 To gain more information, PGE2 and NO was included as well. Age, weight, and body mass index were also taken into account. Correlation analysis showed that SF DKK1 negatively correlated with NO (µM)/mg synovium (R = −0.327, P = 0.001) and IL1β (µM)/mg synovium (R = −0.186, P = 0.059). Similarly, SF FRZB negatively correlated with PGE2 (µM)/mg cartilage (R = −0.184, P = 0.041), IL1β (µM)/mg synovium (R = −0.164, P = 0.096), PGE2 (µM)/mg synovium (R = −0.175, P = 0.064), NO (µM)/mg synovium (R = −0.189, P = 0.047), and age (R = −0.220, P = 0.011). However, SF GREM1 positively correlated with IL1β (µM)/mg synovium (R = 0.213, P = 0.03) and TNFα (µM)/mg synovium (R = 0.206, P = 0.041). Moreover, serum GREM1 positively correlated with PGE2 (µM)/mg cartilage (R = 0.315,P < 0.001), TNFα (µM)/mg synovium (R = 0.274, P = 0.006), weight (R = 0.2, P = 0.023), and body mass index (R = 0.183, P = 0.038) while negatively correlated with NO (µM)/mg cartilage (R = −0.306, P = 0.003) ( Table 4 ). WOMAC questionnaire (0-100 scale) was used to compare clinical effects on pain, stiffness, and functional ability. Lower score means more pain, stiffness, and functional limitation. Interestingly, Both SF DKK1 (R = 0.194, P = 0.045) and serum DKK1(R = 0.206, P = 0.034) positively correlated with WOMAC total score ( Table 4 ).
Correlation between DKK1, FRZB, and GREM1 with Pro-Inflammatory Factors and Painfulness. a
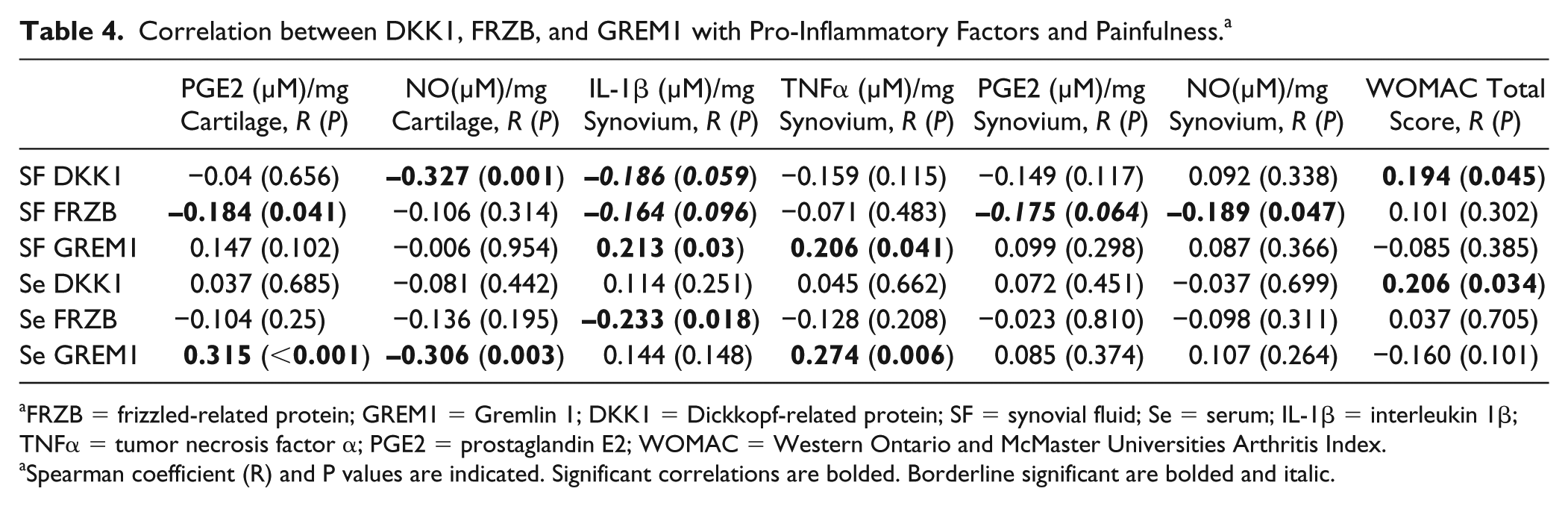
FRZB = frizzled-related protein; GREM1 = Gremlin 1; DKK1 = Dickkopf-related protein; SF = synovial fluid; Se = serum; IL-1β = interleukin 1β; TNFα = tumor necrosis factor α; PGE2 = prostaglandin E2; WOMAC = Western Ontario and McMaster Universities Arthritis Index.
Spearman coefficient (R) and P values are indicated. Significant correlations are bolded. Borderline significant are bolded and italic.
Discussion
The patients in this study had severe knee OA and received in vivo pharmaceutical treatment shortly before joint replacement surgery. We found that celecoxib treatment results in higher FRZB expression, which can be explained as beneficial. It has been reported that celecoxib, a selective cyclooxygenase-2 inhibitor has positive effects on human OA cartilage. 24 Ample evidence confirms that genetic variation in the FRZB gene is associated with either hip OA or OA at multiple joint sites,9,25 indicating that FRZB is important for joint homeostasis. The induction effect of celecoxib on FRZB may suggest that the chondro-protective role of celecoxib is partially mediated by inducing anabolic factors like FRZB.
The correlation between the expression level of 3 factors (DKK1, FRZB, and GREM1) and actual cartilage damage indicates that the DKK1 concentration in SF negatively correlated with the severity of OA. This is in accordance with the evidence that DKK1 is associated with OA development and that high levels of DKK1 may play a protective role against cartilage degeneration. 26 Similarly, FRZB in SF correlated negatively with OA severity scores as well. Moreover, FRZB in synovial fluid has a comparable expression trend as in serum. Previous studies showing that loss of FRZB contributes to cartilage damage by increasing matric metalloproteinase expression and enhancing their activity 27 supports this finding. The expression level of DKK1 and FRZB in synovial fluid is negative correlated with the cartilage damage degree, which is in accordance with our other study, in which the median expression values of both DKK1 and FRZB are lower in OA patient than in healthy people. 22
It has been shown that DKK1 is anti-inflammatory by inhibiting multiple signaling pathways in pericytes and myofibroblasts 28 and expression of DKK1 and FRZB is downregulated by IL1β in human chondrocytes. 13 In our study, DKK1 in SF was negatively correlated with NO (µM)/mg cartilage and IL1β (µM)/mg synovium; SF FRZB negatively correlated with PGE2 (µM)/mg cartilage, IL1β (µM)/mg synovium, PGE2 (µM)/mg synovium, and NO (µM)/mg synovium ( Fig. 5 ). This suggests that the high level of inflammatory factors during OA might induce the downregulation of WNT antagonists DKK1 and FRZB, which may consequently contribute to OA development.
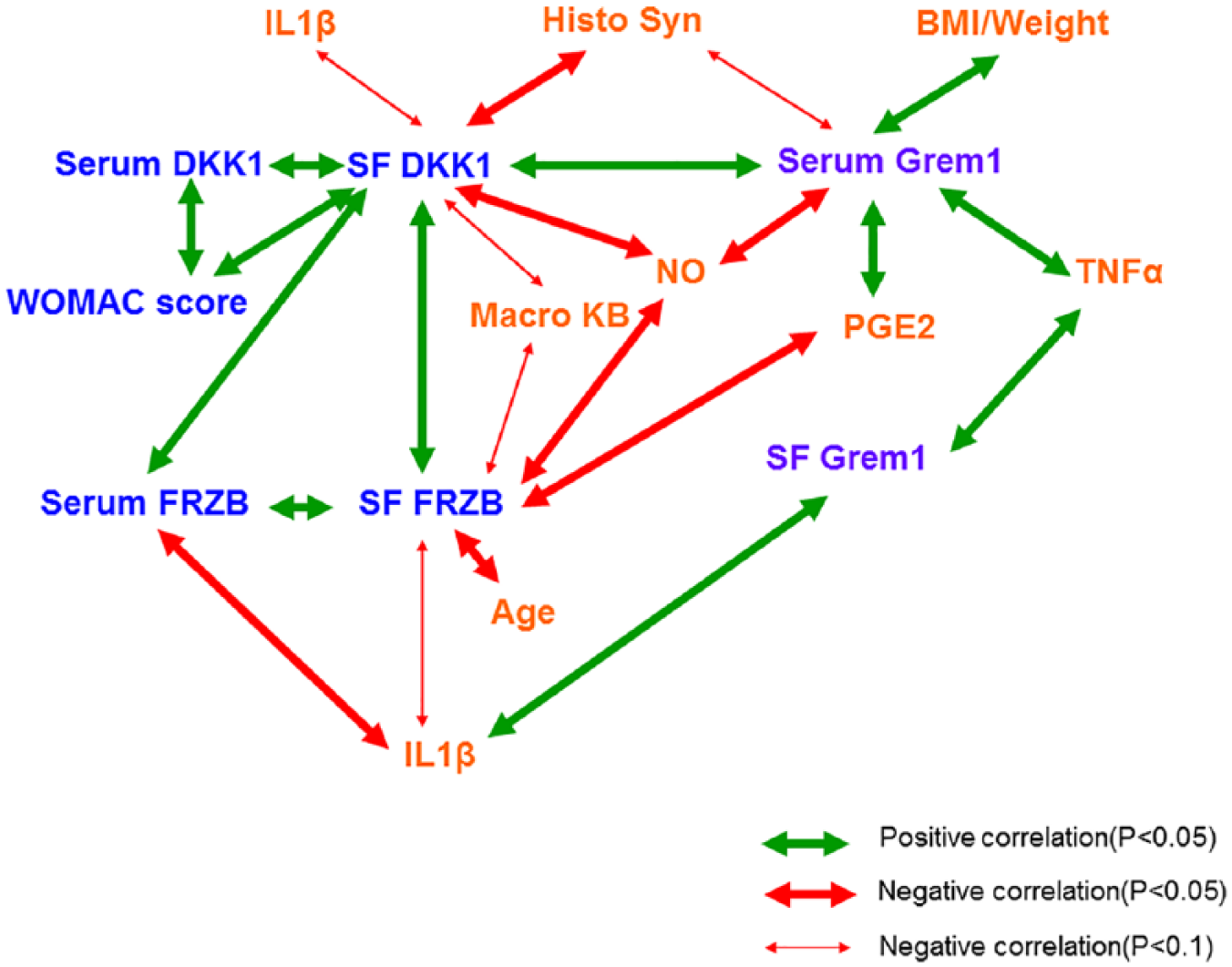
Correlation between DKK1, FRZB, GREM1, and pro-inflammatory factors. Thick red arrows show positive correlation (P < 0.05), thick blue arrows show negative correlation (P < 0.05), light blue arrows show negative correlation (P < 0.1). FRZB = frizzled-related protein; GREM1 = Gremlin 1; DKK1 = Dickkopf-related protein; IL = interleukin; TNFα = tumor necrosis factor α; PGE2 = prostaglandin E2; WOMAC = Western Ontario and McMaster Universities Arthritis Index; Macro KB; Histo Syn; BMI. Data from Table 4 .
In this study, we observed that DKK1 from synovial fluid is negatively correlated with both synovium inflammation and cartilage degeneration. FRZB from synovial fluid only negatively correlated with cartilage degeneration but not synovium inflammation. However, if SF DKK1 and SF FRZB log values were combined together, there was a strong significant negative correlation with both cartilage degeneration and synovium inflammation. Moreover, we found the concentration of FRZB in both SF and serum significantly correlated with DKK1 in SF. This indicates that DKK1 and FRZB might work synergistically to alleviate the inflammation symptom by blocking high activity of WNT signaling in OA.
Pain and loss of function is a frequent symptom in patients with OA. Studies have shown that the presence of local joint inflammation and the local release of pro-inflammatory mediators such as prostaglandins and cytokines accompanied by the destruction of tissue, may contribute to the pain. 29 Interestingly, our data show that the expression of DKK1 in SF and serum positively correlated with WOMAC total score, suggesting that DKK1 somehow benefits the patient with OA, which might relate to the anti-inflammatory interaction of DKK1.
GREM1 has been shown to be highly expressed in fibroblast-like synoviocytes in rheumatoid arthritis (RA), and microarray technology has shown that GREM1 is significantly upregulated in OA chondrocytes. 14 In our study, we found that synovial fluid GREM1 positively correlated with IL1β (µM)/mg synovium, TNFα (µM)/mg synovium. Moreover, serum GREM1 positively correlated with PGE2 (µM)/mg cartilage, TNFα (µM)/mg synovium, weight, and body mass index. These results indicate that the inflammatory environment in OA might contribute to high GREM1 expression and further lead to synovial hyperplasia.
Conclusions
Taken together, our data provide further evidence for the important involvement of DKK1, FRZB, and GREM1 in OA and shows that their expression is associated with inflammation factors. This suggests that DKK1 and FRZB can be potential biomarkers for joint disease. To our knowledge, this is the first investigation of expression levels of DKK1, FRZB, and GREM1 from SF and paired serum simultaneously together with an evaluation of their relationship with pro-inflammatory cytokines and actual cartilage damage degree as evaluated by macroscopy and histology.
Supplemental Material
Supplemental_materials-10292018 – Supplemental material for The Expressions of Dickkopf-Related Protein 1 and Frizzled-Related Protein Are Negatively Correlated to Local Inflammation and Osteoarthritis Severity
Supplemental material, Supplemental_materials-10292018 for The Expressions of Dickkopf-Related Protein 1 and Frizzled-Related Protein Are Negatively Correlated to Local Inflammation and Osteoarthritis Severity by Xiaobin Huang, Leilei Zhong, Eefje van Helvoort, Floris Lafeber, Simon Mastbergen, Jan Hendriks, Janine N. Post and Marcel Karperien in CARTILAGE
Footnotes
Acknowledgments and Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
X.H. and L.Z. finished the experiments and analysis, and all authors wrote the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted according to the Declaration of Helsinki and received ethical approval study protocol K06-002, January 2 2006.
Informed Consent
All patients gave written informed consent before participating in the study.
Trial Registration
Not applicable.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
