Abstract
Objective
Skeletal tissues such as intervertebral disc and articular cartilage possess limited innate potential to regenerate, in part due to their avascularity and low cell density. Despite recent advances in mesenchymal stem cell (MSC)–based disc and cartilage regeneration, key challenges remain, including the sensitivity of these cells to in vivo microenvironmental stress such as low oxygen and limited nutrition. The objective of this study was to investigate whether preconditioning with hypoxia and/or transforming growth factor–β 3 (TGF-β3) can enhance MSC survival and extracellular matrix production in a low oxygen and nutrient-limited microenvironment.
Design
MSCs from multiple bovine donors were preconditioned in monolayer in normoxia or hypoxia, with or without TGF-β3, and the global effects on gene expression were examined using microarrays. Subsequently, the effects of preconditioning on MSC survival and extracellular matrix production were examined using low oxygen and nutrient-limited pellet culture experiments.
Results
Hypoxic preconditioning resulted in upregulation of genes associated with growth, cell-cell signaling, metabolism, and cell stress response pathways, and significantly enhanced MSC survival for all donors in low oxygen and nutrient-limited pellet culture. In contrast, TGF-β3 preconditioning diminished survival. The nature and magnitude of the effects of preconditioning with either hypoxia or TGF-β3 on glycosaminoglycan production were donor dependent.
Conclusions
These results strongly support the use of hypoxic preconditioning to improve postimplantation MSC survival in avascular tissues such as disc and cartilage.
Keywords
Introduction
Skeletal tissues such as intervertebral disc and articular cartilage possess limited innate potential to regenerate, in part due to their avascularity and low cell density.1,2 Avascularity results in a local tissue microenvironment characterized by low oxygen tension and limited supply of nutrients. This presents challenges for cell survival and function, even in the absence of pathology.3,4 Clinically, back and joint pain from disc and cartilage degeneration, respectively, with age or following trauma, are leading causes of disability in the adult population with associated socioeconomic costs estimated to be in the order of many billions of dollars annually. 5 Current treatment approaches are mostly conservative and involve managing symptoms through physical therapy and administration of pain killers.6,7 In severe cases, patients may undergo surgical procedures such as spinal fusion or joint replacement, which do not maintain or restore native tissue structure or mechanical function. 8 In the case of cartilage damage, surgical repair using techniques such as microfracture or autologous chondrocyte transplantation are potential therapeutic options; however, these approaches often have limited efficacy. 9
To address the limitations of current approaches for treating disc and cartilage damage and degeneration, there has been significant interest in developing biological therapies such as tissue engineered biomimetic scaffolds, targeted drug delivery for inducing endogenous cell activation, and exogenous cell implantation. With regard to the last of these approaches, application of mesenchymal stem cells (MSCs) for tissue repair and regeneration has attracted significant interest. MSCs, which can be derived from many sources including bone marrow, adipose tissue, or synovial fluid, are attractive options for cell-based therapies due to their potential for autologous harvest, expansion, and reimplantation.10-12 Furthermore, MSCs may exhibit immunomodulatory properties and suppress inflammatory damage, making them highly translational with an excellent safety profile.13,14 Importantly, MSCs have the potential to differentiate down multiple lineages, 15 and there have been encouraging studies demonstrating the capacity of these cells to adopt phenotypes similar to those of nucleus pulposus cells16-18 and articular cartilage chondrocytes19,20 under certain in vitro culture conditions. Furthermore, there have been human and animal studies reporting that MSCs are able to regenerate and repair disc and cartilage tissue in vivo with varying degrees of efficacy.11,21
Despite these recent advances in MSC-based disc and cartilage regeneration, key challenges remain, including phenotype instability (dedifferentiation or terminal differentiation and mineralization) in long-term culture and inferior functional extracellular matrix (ECM) production compared to native cell types.22-24 Another key challenge is the sensitivity of MSCs to microenvironmental stress, such as nutrient and oxygen deprivation, to which cells are exposed upon implantation. While native cell types such as chondrocytes and nucleus pulposus cells are adapted to functioning under these conditions,24-27 low oxygen and nutrient availability both negatively impact MSC survival and ECM production capacity.28-30 It has also been shown that MSCs isolated from bone marrow are highly heterogeneous, both within and between donors, exhibiting distinct phenotypic characteristics, including sensitivity to microenvironmental stress in culture.29,31,32
To overcome this challenge, one strategy being explored is the preconditioning of MSCs during monolayer expansion (by exposure to hypoxia or growth factors), in order to enhance their in vivo performance in applications for cartilage and disc regeneration.33-44 The objectives of this study were to build on the results of these previous studies and to (1) investigate the effects of preconditioning through exposure to hypoxia or transforming growth factor–beta (TGF-β), alone or in combination on the global gene expression profile of MSCs and (2) investigate whether these molecular level changes in response to preconditioning can enhance MSC survival and ECM production capacity when subsequently placed under microenvironmental stress, specifically low oxygen and limited nutrition, in 3-dimensional (3D) culture. We hypothesized that preconditioning using hypoxia and TGF-β would enhance subsequent MSC survival and ECM production under microenvironmental stress by inducing requisite metabolic changes.
Methods
Cell Isolation, Expansion, and Preconditioning
The overall study design is presented in Figure 1 . Bone marrow–derived MSCs were isolated from juvenile (age <6 months) bovine femurs and tibiae (Research 87; Boylston, MA) as previously described. 29 MSCs from 3 individual donor animals were maintained as distinct populations (i.e., not pooled) during preconditioning and subsequent microarray analysis or pellet culture, which enabled the analysis of donor-dependent effects on MSC behavior and their response to preconditioning. The 3 MSC donor animals used for the microarray study were not the same as the 3 donor animals used for the pellet culture study (i.e., a total of 6 donor animals across both studies). Following the initial isolation, MSCs were expanded to confluence through a single initial passage in standard tissue culture conditions with normal oxygen tension (atmospheric, 21% O2), 5% CO2, in basal medium composed of high glucose (4.5 g/L) Dulbecco’s modified Eagle medium (DMEM; ThermoFisher Scientific; Waltham, MA), 10% fetal bovine serum (FBS; ThermoFisher Scientific; Waltham, MA), and 1% penicillin, streptomycin, and fungizone (PSF; ThermoFisher Scientific). Cells were then passaged and expanded (preconditioned) in basal medium in 1 of 4 different conditions for 1 week: (1) N−: normoxia (21% O2); (2) N+: normoxia + TGF-β3 (10 ng/mL, R&D Systems; Minneapolis, MN); (3) H−: hypoxia (2% O2); (4) H+: hypoxia + TGF-β3. Hypoxic culture was carried out continuously in an environmental workstation (Hypoxystation 35; Hypoxygen, MD, USA) that enabled media changes without exposure to atmospheric conditions.
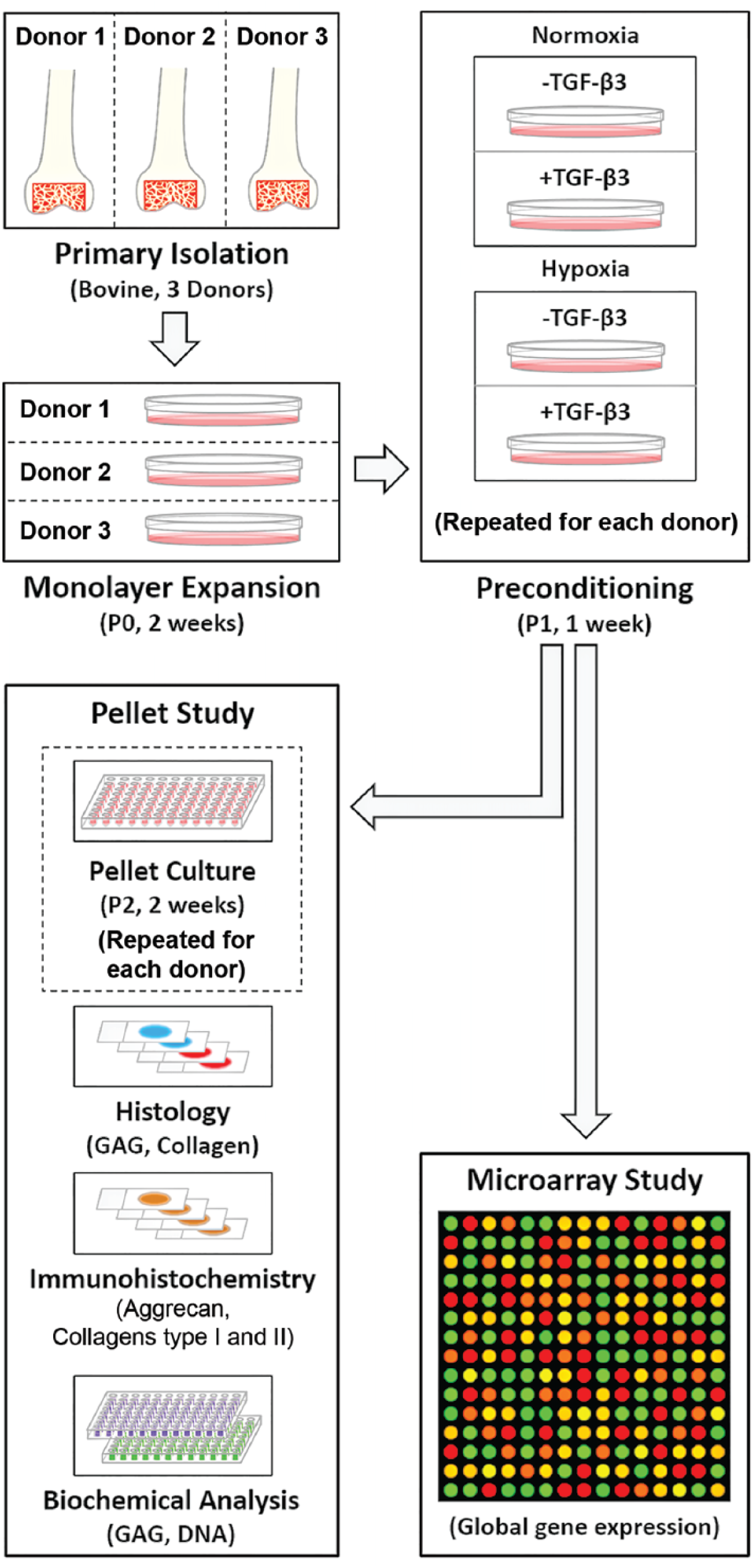
Overall study design. P = passage number.
Microarray Study
MSCs from 3 separate donors were isolated and subjected to preconditioning as described above. MSCs were harvested and RNA was isolated from each sample (n = 3 donors × 4 expansion conditions for a total of 12 distinct samples) using serial TRIzol (Ambion; Austin, TX)-chloroform extractions, in-column treated with RNase-free DNase (Qiagen; Valencia, CA) on miRNeasy columns (Qiagen; Valencia, CA), and eluted following the manufacturer’s protocols. The quality of each RNA sample (RIN > 9) was verified using an Agilent BioAnalyzer and RNA 6000 Pico Kit (Agilent; Santa Clara, CA). Global gene expression was measured using the WTPlus Bovine Gene Chip (Affymetrix; Santa Clara, CA). Gene expression data were normalized using Robust Multi-array Average. 45 Pathway analysis was performed using Ingenuity Pathway Analysis (IPA; Qiagen; Valencia, CA, USA).
Pellet Culture Study
MSCs from 3 separate donors were isolated and subjected to preconditioning as described above. MSCs were then harvested and cultured as pellets (250,000 cells/pellet) in a low oxygen and limited nutrient environment consisting of 2% O2 and serum-free, chemically defined media
26
comprised of low glucose (1g/L) DMEM (ThermoFisher Scientific) supplemented with 1% PSF, 0.1 μM dexamethasone (Sigma-Aldrich; St. Louis, MO, USA), 50 mg/mL ascorbate 2-phosphate (Sigma-Aldrich), 40 mg/ml
Pellet DNA content was quantified using the PicoGreen assay (ThermoFisher Scientific; Waltham, MA) following the manufacturer’s protocol and is presented both as absolute quantity per pellet, and as a percentage of pellet DNA on day 1 of pellet culture (i.e., as an indicator of cell survival after 2 weeks of culture under low oxygen and limited nutrient conditions). GAG content was quantified using the dimethyl methylene blue assay as previously described 46 and is presented both normalized to DNA content (representative of GAG production per cell), and as the absolute quantity per pellet.
Formalin-fixed pellets were processed into paraffin, and 7-μm thick sections were stained with Alcian blue (GAGs) or picrosirius red (collagen) and imaged using bright field light microscopy (Eclipse 90i; Nikon; Tokyo, Japan). For immunohistochemistry, antigen retrieval was carried out on rehydrated sections using a heat-mediated technique in a 95 °C buffered bath of 10 mM sodium citrate with 0.05% Tween 20, pH 6.0 for 20 minutes. Sections were permeabilized in TBS + 0.025% Triton X-100 (TBS-T) and blocked using Background Buster (Accurate Chemical & Scientific Corporation; Westbury, NY) for 30 minutes at room temperature. Sections were then incubated with primary antibodies against aggrecan (12/21/1-C-6, mouse monoclonal, Developmental Studies Hybridoma Bank; University of Iowa, Iowa City, IA), type I collagen (AB749P, rabbit polyclonal, EMD Millipore; Billerica, MA), or type II collagen (II-II6B3, mouse monoclonal, Developmental Studies Hybridoma Bank; University of Iowa, Iowa City, IA), diluted in Background Buster overnight at 4 °C. Slides were rinsed twice, 5 minutes each in TBS-T, then incubated in 0.3% H2O2 in TBS for 15 minutes to suppress endogenous peroxidase activity. Slides were then rinsed 3 times, 2 minutes each with TBS. Antibody staining was visualized using the SuperPicture Polymer Detection kit, diaminobenzidine (DAB), broad spectrum or rabbit (ThermoFisher Scientific; Waltham, MA), according to the manufacturer’s protocols. Slides were rinsed in diH2O and counterstained with hematoxylin QS (Vector Laboratories; Burlingame, CA) for 30 seconds. Slides were then rinsed with tap H2O, dehydrated and cleared, and cover-slipped with Permount Mounting Medium (ThermoFisher Scientific; Waltham, MA). Slides were imaged under bright field light microscopy. As negative controls, sections were treated in parallel with the same protocol, except with only Background Buster (no primary antibodies) during the initial overnight incubation.
Statistical Analysis
For the microarray study, statistical analysis was carried out using the Partek Genomics Suite (v6.6, Partek, Inc., St. Louis, MO). Significant differences between groups were established using a 3-way mixed model analysis of variance (ANOVA), with oxygen tension and TGF-β3 treatment as fixed variables and donor as a random variable, in order to account for intrinsic global gene expression differences present between individual animals. P values were adjusted for false discovery rate.
For the pellet culture study, statistical analyses were performed using Systat (Systat Software Inc.; San Jose, CA). Significant differences between groups were established using three-way ANOVA, with oxygen tension, TGF-β3 treatment, and donor as independent variables, and with Bonferroni post hoc tests used to make pairwise comparisons.
Significance was defined as P < 0.05 and trends defined as P < 0.1. Exact P values were calculated and are presented to 3 decimal places, except in cases where P < 0.001, which are reported as such. All results are expressed as mean ± standard deviation. Significant differences between treatment groups within each donor are presented as solid black lines and trends are presented as dotted black lines.
Results
Microarray Analysis: Effects of Preconditioning on MSC Global Gene Expression
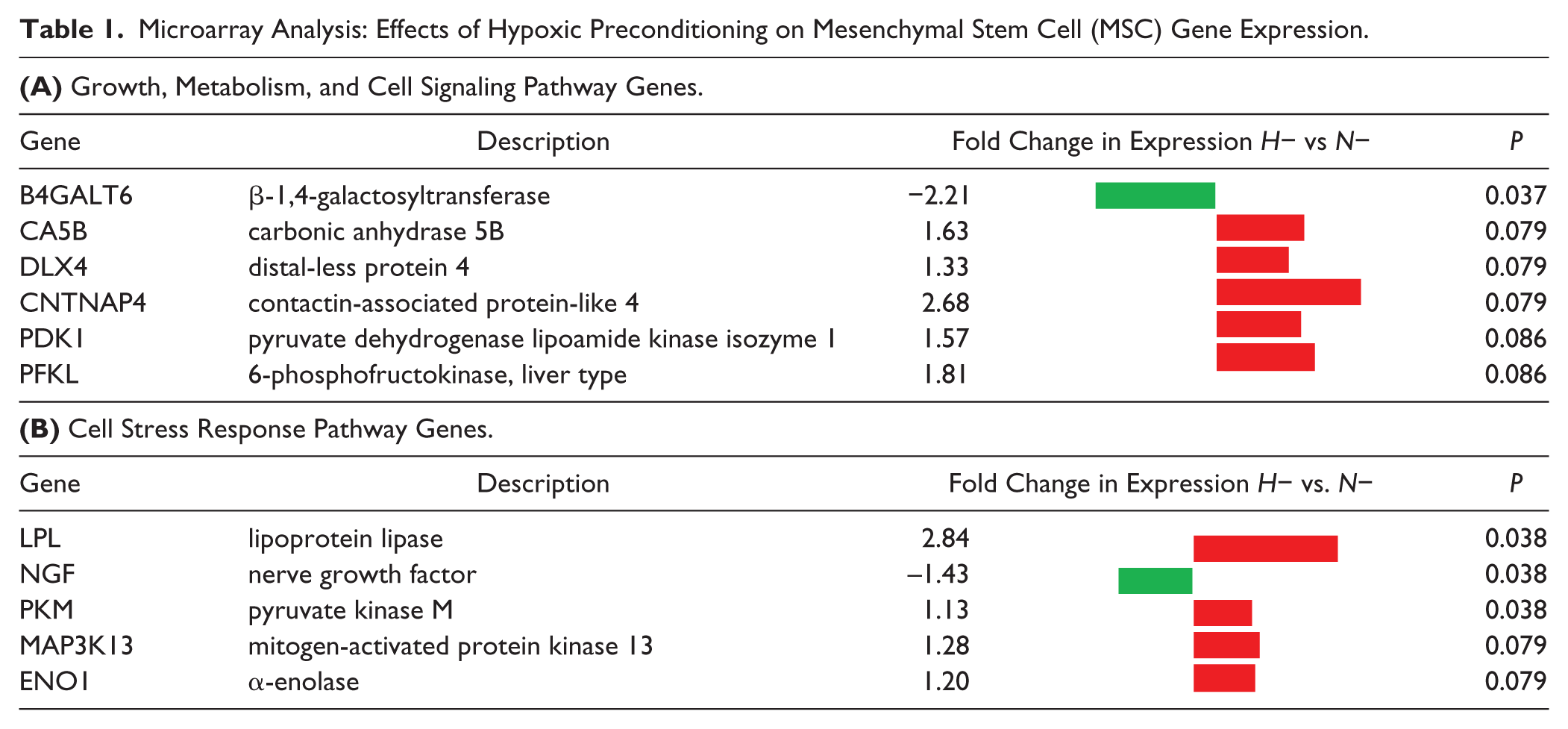
DNA microarrays were used to examine the effects of each monolayer expansion condition and donor animal on the global gene expression profiles of MSCs. Analyses of variance established that TGF-β3 treatment, oxygen tension, and donor animal each exhibited significant effects on gene expression patterns after 1 week of monolayer preconditioning (principal component analysis [PCA] plot, Fig. 2A ). While not affecting the overall statistical findings, retrospective analysis of microarray data revealed that 1 donor lacked genes on the Y chromosome (i.e., it was female). As a result, samples from this donor clustered further from the other 2 donors ( Fig. 2A ). In terms of the total number of genes significantly affected by each variable, TGF-β3 treatment exhibited the greatest effect (3366 genes), followed by donor animal (476 genes) ( Fig. 2B and C ). Oxygen tension had the weakest effect on global gene expression (6 genes). Genes that showed a significant or trending dependence on oxygen tension in the absence of TGF-β3 (H− vs N−) were primarily those involved in growth, cell signaling, or metabolism including β-1,4-galactosyltransferase (B4GALT6), carbonic anhydrase 5B (CA5B), distal-less protein 4 (DLX4), contactin-associated protein-like 4 (CNTNAP4), pyruvate dehydrogenase lipoamide kinase isozyme 1 (PDK1), 6-phosphofructokinase, liver type (PFKL) ( Table 1A ), and cell-stress response pathway genes, including lipoprotein lipase (LPL), nerve growth factor (NGF), pyruvate kinase M (PKM), mitogen-activated protein kinase 13 (MAP3K13), and α-enolase (ENO1) ( Table 1B ). Most of these genes were upregulated in hypoxia, suggesting activation of related pathways. Likely due to the overwhelming effects the addition of TGF-β3 had on gene expression, no genes were significantly differentially expressed as a function of oxygen tension in the presence of TGF-β3 (H+ vs. N+). While only a small number of genes had statistically significant dependence on oxygen tension during monolayer expansion, hypoxic expansion (H−) did result in expected higher (nonsignificant) expression of genes regulated downstream of hypoxia inducible factor–α (HIF-1α) 47 compared with normoxia expansion (N−), including glucose transporters 1 and 3 (SLC2A1 and 3, 1.7, and 1.8-fold higher, respectively), lactate dehydrogenase A (LDHA, 1.3-fold higher) and glycogen synthase 1 (GYS1, 1.3-fold higher). Furthermore, pathway analysis using IPA confirmed HIF-1α as the top predicted upstream regulator of gene expression changes between H− and N− MSCs (activation score of 4.5, P < 0.001).
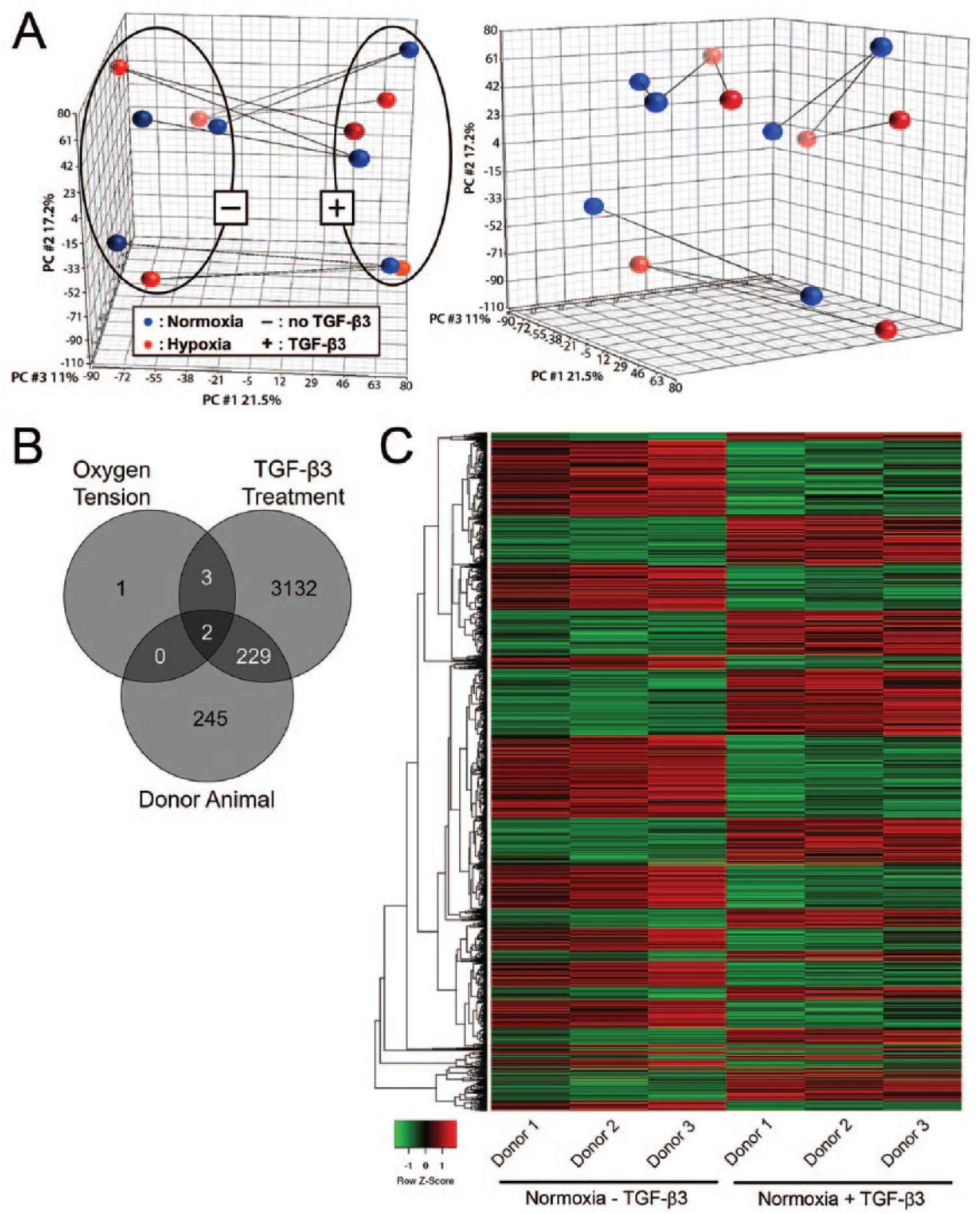
Microarray analysis. (
Microarray Analysis: Effects of Hypoxic Preconditioning on Mesenchymal Stem Cell (MSC) Gene Expression.
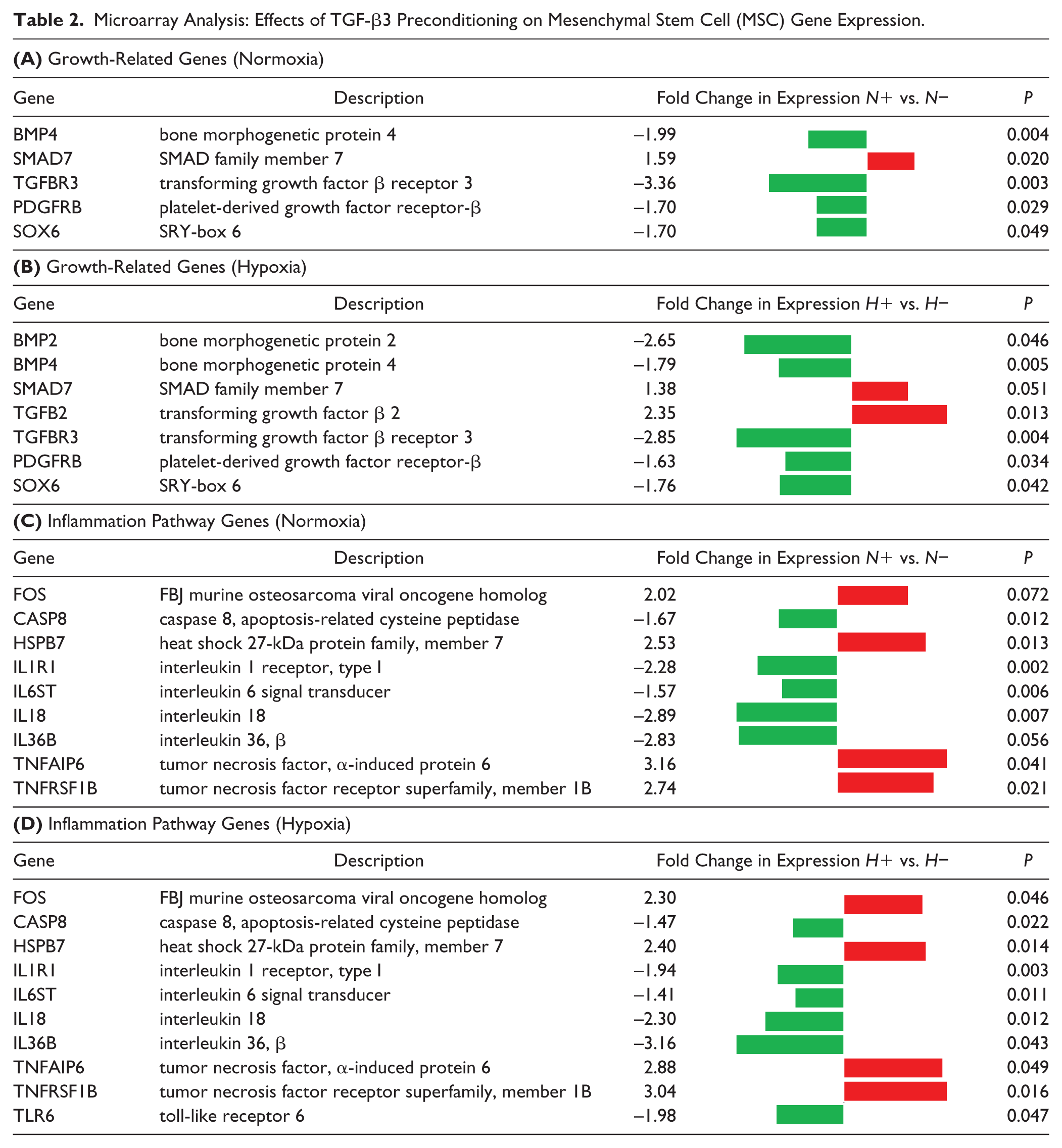
The effects of TGF-β3 on MSC gene expression were independent of oxygen tension, and ANOVA confirmed that there was no significant interaction effect between oxygen tension and TGF-β3 treatment for any genes. TGF-β3 treatment resulted in significant differential expression of genes involved in activating growth pathways both in normoxia and hypoxia (i.e., N+ vs. N− and H+ vs. H−) including many genes in the TGF-β pathway, such as bone morphogenetic proteins 2 and 4 (BMP2, 4), SMAD7, TGFB2, and TGF-β receptor 3 (TGFBR3), along with other growth factor pathway genes such as platelet-derived growth factor receptor–β (PDGFRB) and SOX6 ( Table 2A and B ). TGF-β3 treatment, in both normoxia and hypoxia, also resulted in significant differential expression of genes in inflammatory signaling pathways such as interleukin signaling, tumor necrosis factor (TNF), and caspase pathways ( Table 2C and D ), including heat shock protein family B member 7 (HSPB7), interleukin 1 receptor type 1 (IL1R1), interleukins 18 and 36B (IL18, IL36B), TNF-α inducible protein 6 (TNFAIP6), and TNF receptor super family member 1B (TNFRSF1B). Interestingly, many growth and inflammation-related genes were significantly downregulated in the presence of TGF-β3, suggesting a decrease in cell-signaling within these related pathways in the presence of this growth factor. Global pathway analysis confirmed TGF-β as the top upstream regulator of gene expression changes between N− and N+ groups (activation score of 4.1, P < 0.001).
Microarray Analysis: Effects of TGF-β3 Preconditioning on Mesenchymal Stem Cell (MSC) Gene Expression.
Pellet Study: Effects of Preconditioning on MSC Survival and ECM Production
After establishing the nature of the gene expression changes induced in MSCs by preconditioning with hypoxia and/or TGF-β3 through microarray analyses, we carried out pellet culture studies to assess corresponding effects of preconditioning on cell survival and ECM production in low oxygen and limited nutrient conditions. These experiments were repeated with MSCs from 3 separate donors, and results from all 3 are presented. The results of the ANOVA established that oxygen tension, TGF-β treatment, and donor animal during preconditioning each significantly affected pellet DNA and GAG contents.
DNA content ( Fig. 3 ) is presented both as absolute quantity per pellet and as a percentage of pellet DNA on day 1 of pellet culture (i.e., as an indicator of cell survival after 2 weeks of culture under low oxygen and limited nutrient conditions). Effects of preconditioning on pellet DNA content (cell survival) were broadly consistent across donors, although the magnitude of these effects varied between donors. For all 3 donors, after 2 weeks of culture in low oxygen and nutrient conditions, DNA content for pellets formed from MSCs expanded under normoxia without TGF-β3 (N−, i.e., standard expansion conditions) was approximately 20% of pellet DNA content at day 1 ( Fig. 3 ). Similarly, for all 3 donors, for pellets formed from MSCs expanded in hypoxia without TGF-β3, DNA content was significantly higher than for pellets formed from MSCs expanded in normoxia either with or without TGF-β3 (i.e., H− vs. both N− and N+, all P < 0.001) indicating improved survival. Pellets formed from MSCs expanded in normoxia with TGF-β3 (N+) exhibited the lowest cell survival, although the magnitude was donor dependent and only significant for donor 3 compared with pellets formed from N- expanded MSCs (less than 10% cell survival). For donors 2 and 3, for pellets formed from MSCs expanded in hypoxia, there were significant effects of TGF-β3 on survival (i.e., H− vs. H+). Specifically, H+ resulted in significantly lower pellet DNA content compared with H− (P = 0.027 and 0.049 for donors 2 and 3, respectively). Overall, pellets with MSCs from donor 1 were the least sensitive to different expansion conditions with respect to cell survival and only exhibited significant differences in DNA content for N+ versus H−, and N− versus H−. Pellets formed from MSCs from donor 3 were the most sensitive to expansion condition with respect to cell survival, showing significant differences in DNA content for all possible comparisons between monolayer expansion conditions. DNA content was significantly different between donors 1 and 3 for pellets with N+ MSCs.
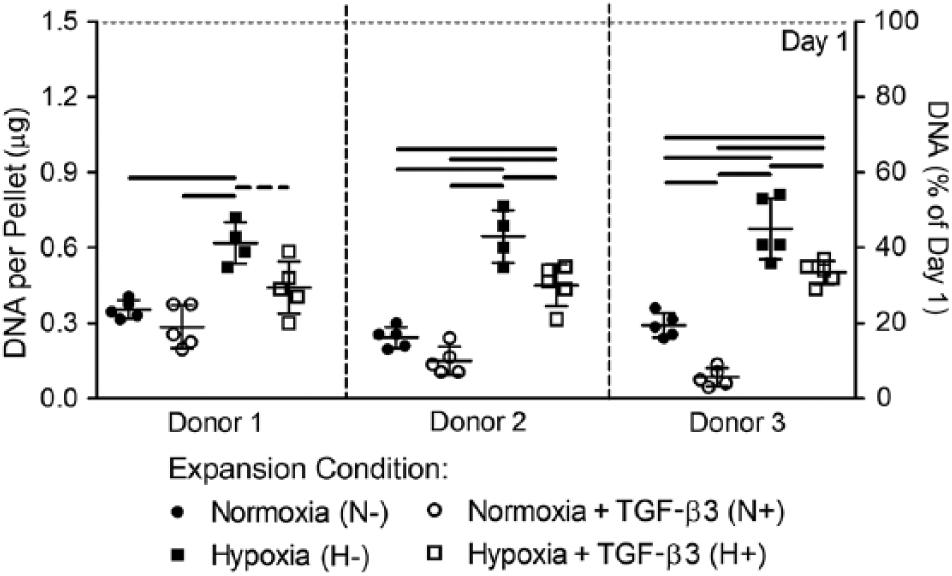
Effects of preconditioning on mesenchymal stem cell (MSC) pellet DNA content after 2 weeks of low oxygen and limited nutrient culture. DNA content is presented both as absolute quantity per pellet and as percentage of day 1 DNA as an indicator of cell survival. N = 5 pellets per donor per condition; solid significance bars represent P < 0.05; dotted significance bars represent trends of P < 0.1.
Pellet GAG content is presented both normalized to DNA content ( Fig. 4A , representative of GAG production per cell) and as the absolute quantity per pellet ( Fig. 4B ). In general, the effects of preconditioning on GAG production exhibited considerable variability between donors. When normalized to DNA, GAG production for pellets formed from MSCs from donor 1 exhibited the greatest sensitivity to expansion condition ( Fig. 4A ). For this donor, GAG per DNA was highest for pellets formed from N+ and H+ MSCs (not significantly different) and was significantly higher for these pellets than those formed from either N− or H− MSCs. Pellets formed from MSCs from donors 2 and 3 showed less sensitivity to expansion condition ( Fig. 4A ). For donor 2, the only significant differences were N+ versus N− and N+ versus H−. Similar to donor 1, donor 2 pellets formed from N+ and H+ MSCs exhibited the highest GAG per DNA overall. For donor 3, GAG per DNA was highest for pellets formed from H− and H+ MSCs, and the only significant difference was between N− and H+. There were also significant donor-dependent effects on pellet GAG per DNA, independent of MSC expansion condition. Specifically, for pellets formed from H− MSCs, GAG per DNA was significantly higher for donor 3 compared with both donors 1 and 2 (P = 0.008 and 0.033, respectively).
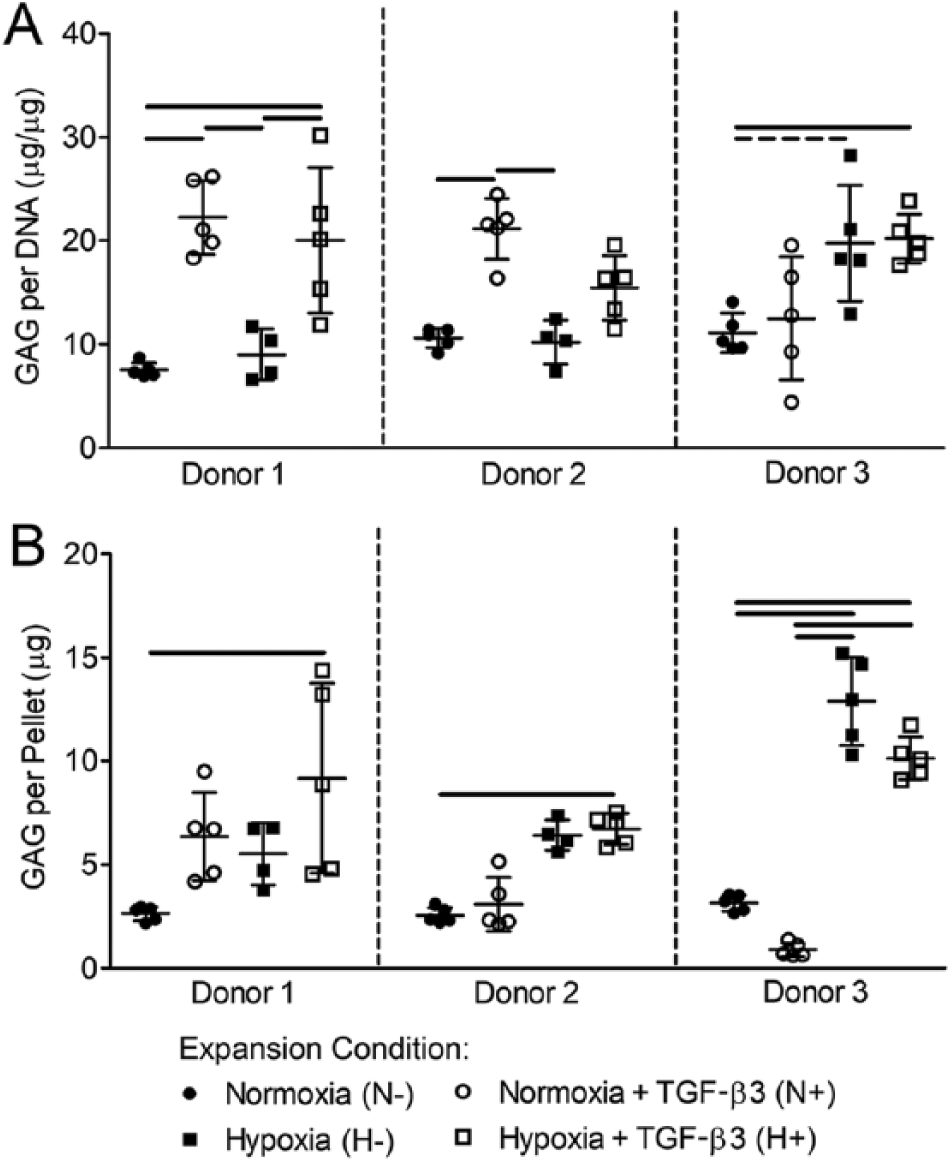
Effects of preconditioning on mesenchymal stem cell (MSC) pellet glycosaminoglycan (GAG) content after 2 weeks of low oxygen and limited nutrient culture. (
For GAG per pellet, donor 3 pellets showed the greatest sensitivity to MSC expansion condition, with GAG per pellet highest for pellets formed from H- and H+ MSCs and significant differences between these 2 conditions and all others ( Fig. 4B ). In contrast, MSCs from donors 1 and 2 were relatively insensitive to expansion condition with the only significant differences in GAG per pellet between N− and H+ MSCs for both. There were also donor-dependent differences in GAG per pellet independent of expansion condition. Specifically, for donor 3, GAG per pellet was significantly greater for pellets formed from H- MSCs than for donors 1 and 2 and significantly lower for N+ MSCs than for Donor 1.
Histological results largely supported findings from the above quantitative assays. Pellets formed from MSCs conditioned in hypoxia (both H− and H+) were larger than those conditioned in normoxia (both N− and N+), again reflecting enhanced cell survival ( Figs. 5 and 6 ). All pellets stained positively for GAG (Alcian blue) and aggrecan with staining intensity largely paralleling quantitative results ( Fig. 5A and B ). All pellets also stained positively for collagen; however, there were no clear observable differences as a function of expansion condition or between different MSC donors ( Fig. 6A ). With regard to immunostaining for collagen subtypes, positive staining was evident for both types I and II collagen, with type II collagen staining, in general, being more intense than type I collagen across all expansion conditions and donors ( Fig. 6B and C ).
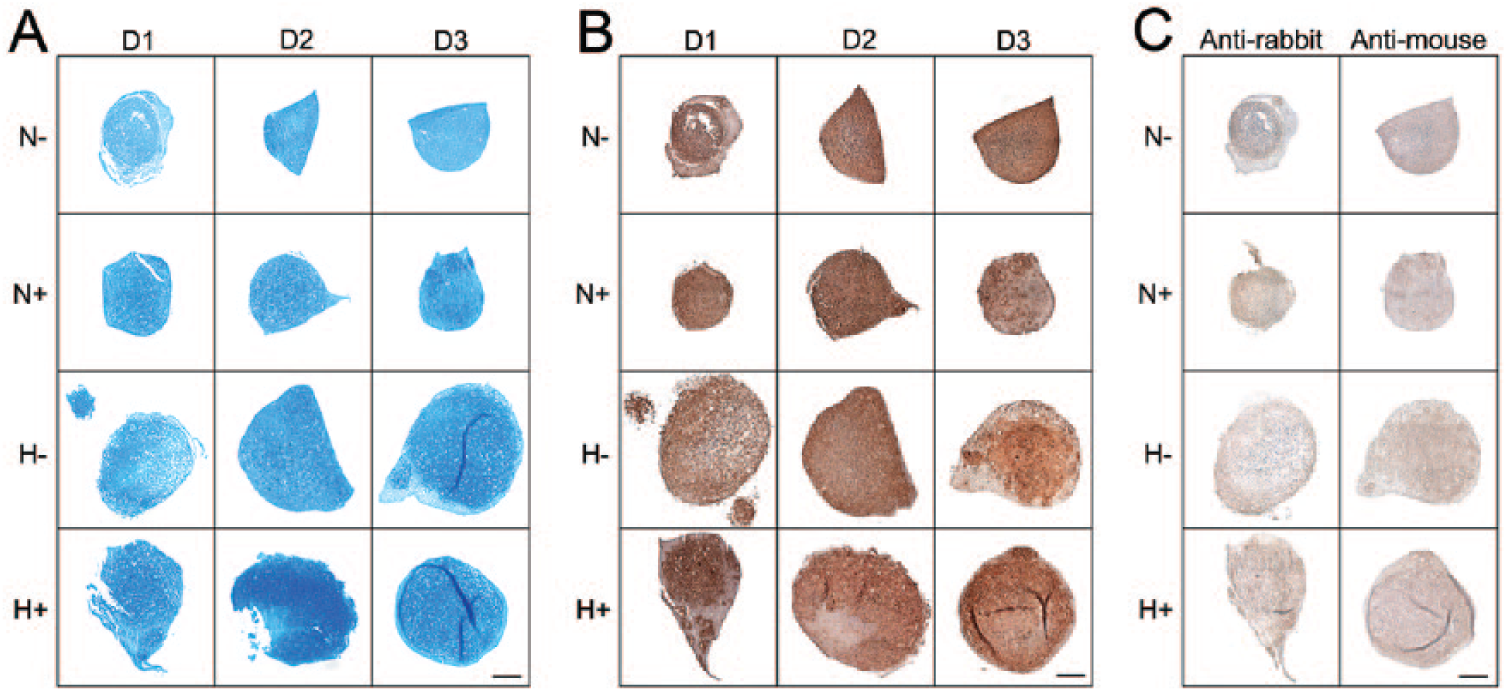
Histological assessment of glycosaminoglycan (GAG) and aggrecan composition for mesenchymal stem cell (MSC) pellets. (
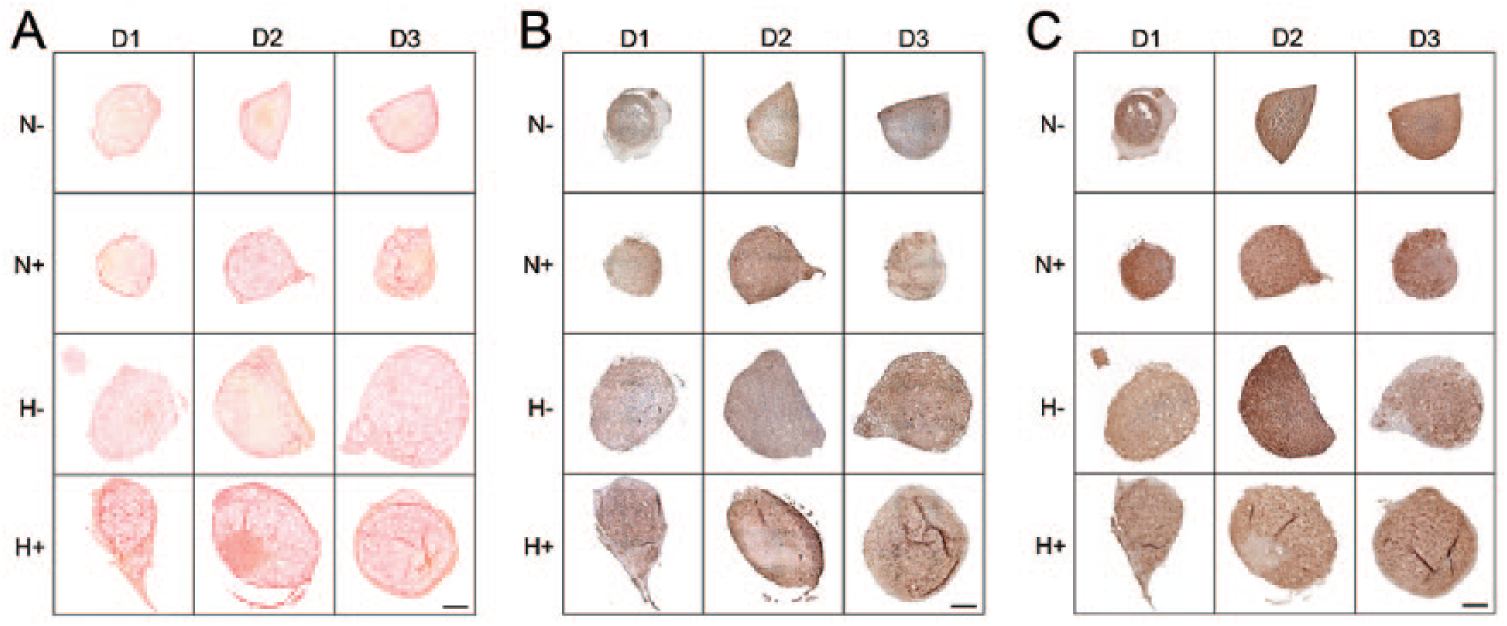
Histological assessment of collagen composition for mesenchymal stem cell (MSC) pellets. (
Discussion
In recent years, MSC-based therapies as clinically viable treatment options for musculoskeletal tissue regeneration and repair have made great progress. 48 While MSCs are multipotent rather than pluripotent, they are putative progenitor cells for many musculoskeletal tissues and have advantages over pluripotent stem cells because of their relative abundance, ease of isolation, biosafety, and immunomodulatory behavior.12,15 Preconditioning during monolayer expansion is a promising technique for improving MSC performance upon in vivo implantation for treatment of tissues such as disc and cartilage, which are characterized by low oxygen and limited nutrient supply. Preconditioning may improve MSC performance by acclimating cells to in vivo microenvironmental conditions by inducing requisite metabolic changes and stimulating lineage-specific differentiation. Previous studies have shown that hypoxic expansion of MSCs can enhance chondrogenesis and suppress osteogenesis35,49-52 and enhance proliferation.33,36,53 Of note, these prior studies reported somewhat disparate findings with respect to the nature of the effects of hypoxic preconditioning, particularly with regard to promoting chondrogenesis and proliferation.38,54 These disparate findings may have arisen from experimental differences (e.g., culture conditions), as well as different MSC donor sources, including age and species, 38 which ultimately may be of significant importance to clinical outcomes. Monolayer preconditioning has the advantage of adding little or no cell preparation time in the clinical setting and therefore, apart from the cost of growth factors and specialized equipment for hypoxic culture, does not detract from the translatability of MSCs. Here, we build on these prior studies and investigate the potential for preconditioning to improve MSC performance specifically for disc and cartilage regeneration applications. We examine whether preconditioning using hypoxia and/or TGF-β3 improves the survival and ECM production capacity of MSCs in a low oxygen and nutrient-limited pellet culture setting designed to partially simulate the microenvironmental stress cells might be exposed to in vivo in these avascular tissues. Furthermore, unlike most prior studies we examine the donor-dependent effects of preconditioning and use transcriptomic analysis to infer a mechanistic understanding of those effects.
As expected, without preconditioning (i.e., under standard expansion conditions), MSCs exhibited poor survival (measured as DNA content) in low oxygen and nutrient-limited pellet culture. Hypoxic preconditioning consistently improved MSC survival for each of the 3 different donors. These positive effects on survival support prior work using a variety of different in vitro culture conditions.41,42 In light of the robust positive effects of hypoxic preconditioning on MSC survival, it was somewhat surprising that the corresponding effects on global gene expression from the microarray analysis were modest. This may suggest that the effects of hypoxic preconditioning are predominantly regulated at the posttranscriptional level. The genes that did exhibit significant changes in expression in response to hypoxia are components of various growth, cell signaling, metabolism, or cell stress response pathways.55-57 The majority of these genes were upregulated in response to hypoxia. These gene expression changes are broadly consistent with the results of recent article by Elabd et al. 58 that examined transcriptome-wide changes for human MSCS cultured in hypoxia. Combined with pathway analysis results that indicate induction of genes downstream of HIF-1α and a switch to anaerobic metabolism, our microarray results suggest that hypoxic preconditioning acclimates MSCs to microenvironmental stress by inducing the metabolic changes necessary to promote survival. For example, it is possible that with hypoxic preconditioning, upregulation of genes such as LPL, PKM, and MAP3K13, which all play important roles in mitigating the cell stress response, prepares the cells for improved survival and function when subjected to microenvironmental stress. Upregulation of these genes during monolayer expansion likely allows cells to dedicate resources and energy to other metabolic processes more quickly once placed into low oxygen and nutrient-limited 3D pellet culture. In future work, we will carry out gene expression analysis of pellets following culture in low oxygen and limited nutrient conditions to assess whether changes to MSC gene expression as identified in the microarray study are maintained, as well as to identify other gene expression changes that may affect MSC survival.
In the absence of hypoxia, preconditioning with TGF-β3 impaired cell survival. TGF-β3 treatment exerted broad effects on MSC gene expression, which included downregulation of genes involved in tissue growth and response to inflammation and had minimal or negative effects on chondrogenic gene expression. This suggests that TGF-β3 treatment during monolayer expansion may induce the opposite effects of hypoxia, rendering cells less prepared to withstand microenvironmental stressors such as oxygen and nutrient deprivation. Previous studies have shown that exposure of MSCs to TGF-β followed by withdrawal can negatively affect sustained anabolic potential. 59 In our study, the overall effects of hypoxia and TGF-β3 on cell survival were inferior to hypoxia alone. Microarray results showed no significant interactive effects of hypoxia and TGF-β3 on global gene expression patterns.
In contrast to the effects on cell survival, the effects of preconditioning (in both nature and magnitude) on ECM (specifically, GAG) production by MSCs were more variable across the 3 donor animals. This variability may be attributed to factors such as genetic diversity, age, and gender, the effects of which should be investigated more comprehensively in the future. In the clinical setting, the potential impact of these factors is likely to be highly significant. Identifying specific genetic factors or molecular signatures that predispose MSC populations from certain donors to enhanced performance, both with and without preconditioning, may lay the foundation for the development of improved, patient-specific MSC treatment strategies. Further elucidating the biological basis of donor-dependent effects of preconditioning on both animal and human MSCs will therefore be a focus of future studies.
Matrix deposition in pellets was fairly homogeneous for all MSC expansion conditions and was composed primarily of aggrecan and type II collagen, with lesser amounts of type I collagen, suggesting chondrogenic rather than pro-fibrotic or osteogenic MSC behavior. Previous studies suggest that long-term 3D culture of MSCs can result in phenotype instability and hypertrophy. 22 Future work will therefore seek to establish whether preconditioning enhances or diminishes MSC phenotype stability in long-term low oxygen and low nutrient 3D culture.
A limitation of this study was that only juvenile bovine MSCs were used, which did not permit an investigation of age-related changes to cellular function, nor inter-species variations. All this work was carried out in vitro, and while our pellet culture simulated low oxygen and limited nutrition, many other characteristics of the in vivo cellular microenvironment, including inflammation, pH, and physical cell-matrix interactions are likely to impact MSC performance. Co-delivery of anti-inflammatory drugs and physical preconditioning on soft substrates may further enhance in vivo MSC performance on implantation to degenerate disc and cartilage.60,61
Conclusions
Through these studies, we provide evidence that preconditioning bone marrow–derived MSCs in hypoxia, but not with TGF-β, significantly enhances their survival upon subsequent culture in a low oxygen and nutrient-limited microenvironment. Preconditioning with hypoxia or TGF-β can enhance ECM production, but the nature and magnitude of these effects are likely donor-dependent. Future studies will seek to further elucidate the factors underlying the varying response of MSCs to preconditioning and progress these findings toward clinical application through evaluation in preclinical in vivo models of disc and cartilage degeneration and regeneration.
Footnotes
Authors’ Note
The microarray datasets generated and analyzed during this study are available in the NCBI Gene Expression Omnibus (GEO) repository, GSE109567.
Acknowledgments and Funding
The authors acknowledge technical support provided by staff members at the Molecular Profiling Facility at the University of Pennsylvania. The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by grants from the Department of Veteran’s Affairs (I01 RX001321 and I01 RX001213), and the National Institutes of Health (R21 AR070959, F32 AR071298). Additional support was received from the Penn Center for Musculoskeletal Disorders (P30 AR069619) and the Catherine D. Sharpe Foundation.
Author Contributions
S.H.P. designed and performed the experiments and drafted the manuscript. J.R.B. performed the experiments. J.W.T. carried out statistical analysis of the microarray data. G.R.D., R.L.M, and N.R.M. provided conceptual input on experimental design. L.J.S. designed the experiments and drafted the manuscript. All authors reviewed and approved the manuscript prior to submission.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Ethical Approval
Ethical approval was not sought for the present study because post mortem bovine tissues were purchased from a third party vendor (Research 87).
