Abstract
Objective
Cam-type femoroacetabular impingement (FAI) syndrome is one of the most frequent reasons for cartilage damage in the hip. Autologous chondrocyte transplantation has proven high success rates in the treatment of focal chondral defects; however, harvesting of chondrocytes in the hip has been reported but not specifically from the region of femoral cam lesions. Therefore, the goal of this study was to analyze the growth and redifferentiation potential of cartilage samples harvested from the cam deformities in patients with FAI.
Design
Cartilage samples were gained from 15 patients with cam-type FAI undergoing arthroscopic femoral cam resection. Healthy (hyaline cartilage of the hip and knee joint, n = 12) and arthritic control groups (degenerative changes in cartilage of the hip joint, n = 8) were also analyzed. Chondrocytes were initially cultured under monolayer, and subsequently under pellet conditions. A comparative representation of the groups was performed by Mankin score classification, immunohistochemistry (IHC) (Col1, Col2, aggrecan), and quantitative reverse transcription–polymerase chain reaction (qRT-PCR) (Col1, Col2, Col10, Sox9, RunX2).
Results
Mankin score of FAI-samples (4.1±3.1, Range 0-10) showed a wide variation but was significant lower (P = 0.0244) when compared with the arthritic control (7.5 ± 2.7, range 4-12). IHC showed an increased deposition of Col2 (P = 0.0002) and aggrecan (P = 0.0261) after pellet culture compared with deposition after monolayer culture in all groups. In qRT-PCR, FAI samples showed after pellet culture increased Col2 (P = 0.0050) and Col10 expression (P = 0.0006) and also Mankin score correlated increasing gene-expression of Col10 (r = 0.8108, P = 0.0341) and RunX2 (r = 0.8829, P = 0.123).
Conclusions
Cartilage samples of patients with cam-type FAI showed sufficient but heterogeneous composition relating to histological quality and chondrogenic potential. However, harvesting of chondrocytes from the cam lesion might be a valid option especially if a cartilage lesion is noted in a diagnostic arthroscopy and individual preexisting stage of cartilage degeneration and appropriate pellet-culturing conditions are considered.
Introduction
Cartilage defects of the hip pose a common therapeutic problem and progression toward osteoarthritis is often induced. 1 Recent studies reported a high rate of patients developing mechanically induced osteoarthritis secondary to subtle deformities, and especially femoroacetabular impingement (FAI) syndrome, has attracted major attention. 2 Inherently, 2 different types of FAI can be distinguished. The pathomechanism is based on an abnormality in the shape of the acetabulum or femoral head-neck junction. Pincer type impingement occurs through repeated contact between prominent, over covered acetabular rim and normal configurated head-neck junction.3,4 Cam lesions arise by a nonspherical portion of the femoral head hitting against the acetabular rim in terminal flexion and internal rotation of the hip. This causes repeated contusion and finally delamination of the acetabular cartilage at the anterolateral acetabular rim, often in combination with labral defects. 5 Because of the mechanical conflict, treatment is usually surgically with resection of the nonspherical portion and restoration of the natural shape of femoral head-neck junction. This leads to increased range of hip motion, reduction of clinical symptoms and should prospectively limit mechanical damage of the acetabular cartilage.6,7 Results after surgery mainly depend on the grade and extend of the chondral damage; thus, measures for cartilage restoration are urgently warranted. Currently, besides conventional options like debridement and microfracture, treatment of hip cartilage lesions can also be performed with autologous chondrocyte transplantation (ACT),8,9 mirroring the established treatment options of the knee. 10 Certainly, cell harvesting for femoroacetabular ACT is still a major problem. A knee arthroscopy is indeed a fast and viable option with low morbidity to harvest chondrocytes for ACT but means an additional surgery to an unaffected joint. Harvesting of chondrocytes at the hip for ACT is currently not well established and both harvesting from the area surrounding the pulvinar at the fossa acatabuli and from the peripheral femoral head have been suggested.11,12 Bretschneider et al. 12 reported that chondrocytes derived from damaged acetabular and femoral site are qualified for ACT. However, chondrocyte extraction from the central acetabulum affects the weight bearing acetabular surface. Therefore, culture of chondrocytes harvested from the peripheral femoral head seems most attractive since cam resection usually extends proximally into the chondral area of the femoral head. However, histological and molecular alterations in tissue samples from nonspherical portion of the femoral head-neck junction of patients with FAI has been shown and examined samples revealed significant signs of degeneration; nonetheless, hyaline cartilage was detected in all specimens.12,13
Thus, the goal of our study was the qualitative analysis of cartilage samples from patients with solely cam-type FAI undergoing arthroscopic resection of the femoral head-neck junction. We hypothesize that the harvested chondrocytes have the potential for ex vivo growth and redifferentiation to be used as a potential substrate in the field of ACT. Tissue samples of the impingement group were further compared with degenerative cartilage samples of patients with osteoarthritic changes as an osteoarthritic control and unaffected cartilage samples from patients undergoing joint replacement because of tumor disease as a healthy cartilage control group.
Methods
Patients and Cartilage Samples
The present prospective, controlled, single-center study was approved by the institutional review board of the Technische Universität München (Project No.: 2906/10). Informed consent was obtained from all patients. We analyzed articular cartilage samples from 15 patients with cam-type femoroacetabular impingement, 8 patients with osteoarthritis of the hip, and 12 patients undergoing tumor surgery of the knee (n = 3) and of the hip (n = 9) with healthy cartilage. Diagnosis of FAI was established by clinical examination, X-ray images (pelvic X-ray and frog-leg lateral) and magnetic resonance imaging (MRI) and confirmed during hip arthroscopy. Exclusion criteria for the FAI group included narrowing of the joint gap <3 mm on native X-rays and global cartilage damage. 14 The cartilage was harvested during arthroscopic surgical resection of the nonspherical portion of the anterolateral part of the femoral head-neck junction ( Figs. 1 and 2 ). The healthy control group included patients undergoing hip- or knee replacement because of tumor disease with intact articular surface. Degeneration was excluded by preoperative X-ray, by inspection during surgery and by Mankin classification during histopathological analysis. The healthy control samples were obtained from a macroscopic intact part of the tibial plateau or femoral head cartilage. The arthritic control group consisted of patients undergoing hip replacement for osteoarthritis grade Tönnis 1 and 2. Exclusion criteria for this group included narrowing of the joint gap <3 mm on native X-rays and global cartilage damage, which means advanced osteoarthritis (Tönnis 3).
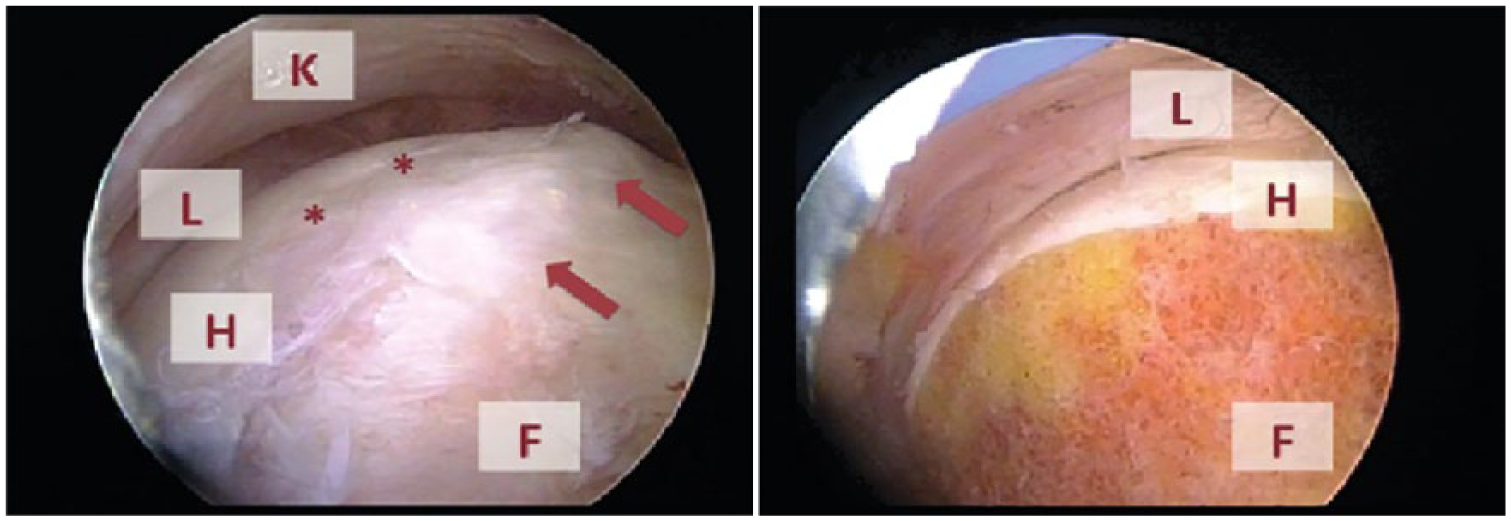
Arthroscopic osteochondroplasty of the femoral head: intraoperative view before (left) and after (right) arthroscopic of cam-deformity resection (arrows). K = hip capsule; L = acetabular labrum; H = femoral head; F = femoral neck. **Indicates border of hyaline cartilage.
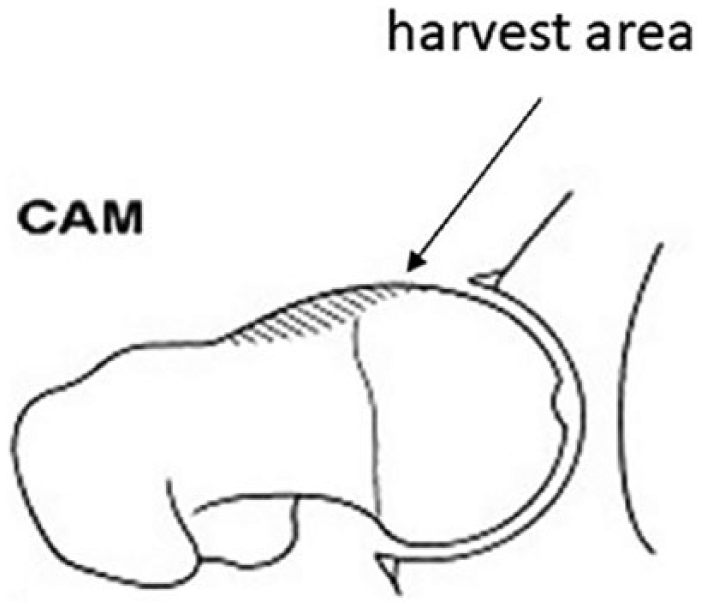
Area of chondrocyte harvesting at the hip joint.
Study Population
Cartilage samples from patients with FAI were obtained during arthroscopic resection of the nonspherical portion of femoral head-neck junction from 15 patients (3 women, 12 men) with a mean age of 30.9 ± 9.3 years (range 17-46 years). As control, healthy hyaline cartilage samples from 12 patients (6 women, 6 men) with a mean age of 40.7 ± 20.0 years (range 11-70 years) were obtained. Nine patients were undergoing hip replacement and 3 were undergoing knee replacement as described above.
In addition, samples were compared with osteoarthritic cartilage of 8 patients (6 women, 2 men) with a mean age of 53.3 ± 9.7 years (range 45-74 years) undergoing total hip replacement. FAI patients were significant younger than patients of the osteoarthritic control group (P = 0.0003).
Macroscopic Cartilage Classification
Cartilage degeneration was graded according to the criteria of Mankin by a pathologist specialized in musculoskeletal pathology (JS-H). According to previous studies, Mankin grade 0-3 was rated as healthy, nonarthritic cartilage, Mankin grade 4-14 was rated as cartilage degeneration. Furthermore, acetabular cartilage was analyzed during arthroscopy and evaluated by Beck score, 15 Outerbridge score, 14 and Sampson score. 16
Cell Culture and Histopathology
After harvesting, cartilage samples were manually cut into pieces of 1 mm × 1 mm, washed with phosphate buffered saline (PBS; Biochrom, Berlin, Germany) and digested in a collagenase solution containing 15 mg collagenase A (Roche, Mannheim, Germany) in 10 mL Dulbecco’s modified Eagle medium (DMEM; Biochrom) for 18 hours under standard cell culture conditions (37°C, 5% CO2, 95% air). After digestion, cell suspension was centrifuged (120 × g, 3 minutes), supernatant was removed, the pellet resuspended in 5 mL DMEM and placed into culture flasks at a cell density of 5 × 104/cm2. Growth medium for monolayer culture was DMEM High Glucose (Biochrom) supplemented with 15% fetal bovine serum (FBS superior; Biochrom), 1% glutamine (Biochrom), 2% HEPES (Biochrom), 1% MEM-vitamine (Biochrom), and 1% penicillin/streptomycin (Biochrom). At 80% confluence, chondrocytes were subcultured by releasing from flasks with trypsin/EDTA solution (0.05% trypsin, 0.02% EDTA, Biochrom). Chondrocytes were incubated in a humified incubator (Heraeus Instruments GmbH, Hanau, Germany) under standard cell culture conditions (37°C, 5% CO2, 95% air).
After chondrocyte expansion in monolayer to a cell count of 10 × 106, pellet culture was performed as earlier described by Johnstone.
17
In brief, each pellet consisted of 1.5 × 106 chondrocytes. Cells were spun down at 250 × g in 15 mL polypropylene conical tubes. Pellets were cultured in DMEM supplemented with 0.01% Media Supplement ITS + 1 (Sigma, Steinheim, Germany), 100 nM dexamethason (Sigma), 1% glutamine (Biochrom), 2% HEPES (Biochrom), 60 µg/mL
Immunohistochemistry
After monolayer culture chondrocytes were cultivated on chamber slides (70,000 cells/cm2) and fixed after 96-hour culture period with paraformaldehyde (PFA; 4%). Pellets were embedded in “Jung-Gel” (Jung Einbettmedium; Leica, Nussloch, Germany), cut into 7-µm sections using a cryotome (Leica Microsystems, Wetzlar, Germany), mounted on SuperFrost glass slides, air dried, and also fixed with PFA (4%). For immunohistochemical (IHC) analysis, the Vectastain Elite avidin-biotin complex kit (Vector Laboratories, Burlingame, CA) was used. Staining was performed with primary antibodies against human collagen type 1 (Collagen Type I Polyclonal Antibody, Rabbit, Quartett, Berlin, Germany), type 2 (Collagen Type II Polyclonal Antibody, Rabbit, Quartett, Berlin), and aggrecan (Anti-Aggrecan Monoclonal Antibody (BC-3), Mouse/IgG1, kappa, Thermo Scientific, Rockford, IL) according to manufacturer’s protocol. Negative controls were probed with PBS and antibody-specific isotype controls. Results of IHC were assessed by semiquantitative analysis by light microscope with an adapted immunoreactive score 18 taking only staining intensity into account. Points given for staining intensity ranged from 0 to 4 (0, no staining; 1, moderate staining; 2, intermediate staining intensity; 3, strong intensity; 4, very strong intensity).
Isolation of RNA and Quantitative RT-PCR
Two different time-points were defined to analyze FAI, healthy and osteoarthritic chondrocytes by real-time reverse transcription–polymerase chain reaction (RT-qPCR). First, after completed monolayer culture, FAI cartilage samples (n = 15) and a healthy pool of chondrocytes (cDNA of healthy cartilage samples, Mankin score ≤3, n = 8, in equal shares) were investigated for comparison. In a second step, after pellet culture, FAI samples (n = 8), healthy (n = 4) and osteoarthritic probes (n = 4) were also analyzed.
After in-house evaluation and according to previ-ous results, glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was chosen as reference housekeeping gene for all gene expression analysis. 19 All mentioned genes were subject to gene expression analysis by RT-qPCR (according to MIQE guidelines) 20 as stated in the following. RNA was extracted after monolayer and after pellet culture. Chondrocytes of monolayer culture were seeded in 6-well plates until confluent growth. For homogenization, pellets were treated with ultrasound waves for 30 seconds. Cells in each well or homogenized cell pellets were suspended in 1 mL of TRIzol reagent (Invitrogen, Darmstadt, Germany). Isolation of total RNA was done using the TRIzol protocol according to the manufacturer’s instructions. Finally, the gained RNA pellet was air dried and solved in 30 µl of RNA-free water. To determine RNA purity and concentration, NanoDrop spectrophotometer (NanoDrop Techn-ologies, Wilmington, DE) was used. To prevent contamination, only probes with A260/280-ratio ≥1.8 were used. All RNA preparations were carried out under RNAse-free conditions. To synthesize first strand complementary DNA (cDNA), total RNA was reverse-transcribed with QuantiTect Reverse Qiagen Kit (Qiagen, Milden, Germany). Oligonucleotide primers and TaqMan probes (for all genes: COL1A1, COL2A1, COL10A1, Sox9, RunX2; Applied Biosystems) were designed to exclude amplification of genomic deoxyribonucleic acid (DNA), the probe or one of the primers was selected to overlap an exon-exon junction. PCR was performed under standard conditions with TaqMan Universal PCR master mix (Applied Biosystems), 900 nM primers (forward and reverse), and 250 nM TaqMan probe. All primers were evaluated for comparable efficiency before the course of experiment. Relative quantification was performed using the comparative delta-delta-CT method.
Statistical Analysis
Statistical analysis of the data was performed with GraphPad Prism (GraphPad Software, Inc., La Jolla, CA, Version 5.02). Data are expressed as single value together with mean or median and standard deviation unless otherwise indicated. The nonparametric Mann-Whitney U test was used for comparisons of normally distributed data, whenever applicable. Correlation of 2 groups was determined by Spearman’s rank correlation coefficient. Differences of values were considered significant at a P -value of <0.05.
Results
Mankin Score
According to Mankin criteria, grading of cartilage from FAI patients showed a mean Mankin grade of 4.1 ± 3.1 (range 0-10). The osteoarthritic cartilage samples from patients undergoing hip replacement resulted in Mankin grade of 7.5 ± 2.7 (range 4-12). Samples of healthy control group showed a Mankin grade of 1.8 ± 1.3 (range 0-3). Mankin grade of healthy control group was significantly lower compared with FAI (P = 0.0283) and osteoarthritic group (P = 0.0002). The difference in Mankin grade between FAI and osteoarthritic group was also significant (P = 0.0244). There was no significant correlation between Mankin score and age of patients with FAI (P = 0.3483). The grading of the acetabular cartilage defect, classified by Beck score (r = 0.0428, P = 0.069), Outerbridge score (r = 0.411, P = 0.128), and Sampson score (r = −0.056, P = 0.844) showed no significant correlation to the Mankin score of the femoral cartilage harvested and analyzed during arthroscopic cam resection.
Cell Culture
During monolayer cultivation, chondrocytes of all groups showed extensive morphological changes. Subsequent to isolation, the chondrocytes showed phenotypical spherical cell morphology. During the course of cultivation, cells presented a more and more fibroblast-like phenotype. After monolayer culture for a mean of 42.5 ± 8.6 days (range 13-55 days), chondrocytes were transferred into pellet culture for another 21 days, where a stable cell pellet with morphology of a flattened spherical aggregate was built.
Immunohistochemical Analysis
IHC staining was done for collagen type 1 (Col1), collagen type 2 (Col2), and aggrecan (Agg) ( Fig. 3 ). After monolayer culture, Col1 staining showed strong intensity for all groups. After pellet culture, no differences in staining intensity were found in FAI (P = 0.98), healthy (P = 0.87), and osteoarthritic control (P = 0.70).
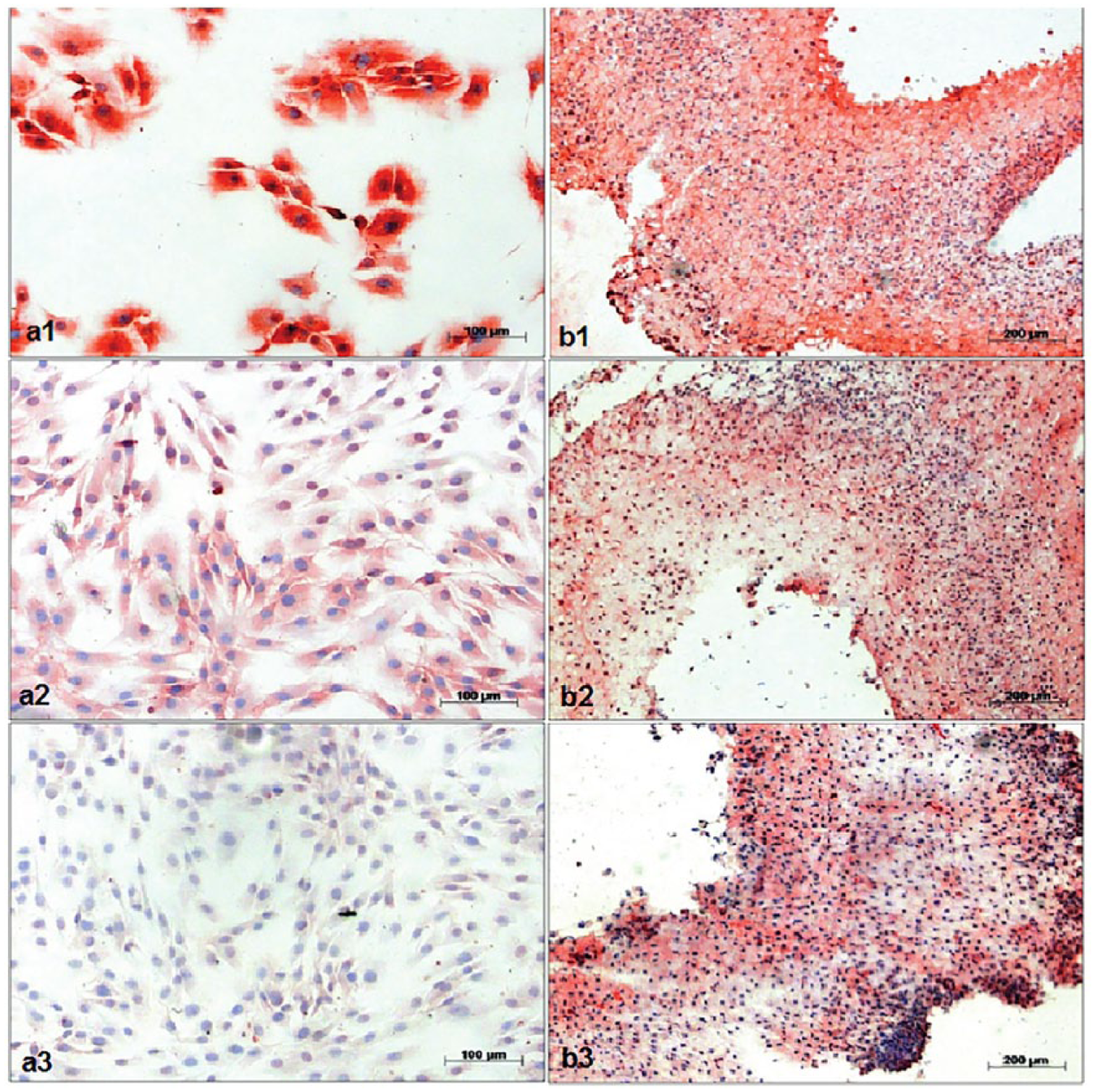
Immunhistochemistry of femoroacetabular impingement (FAI) chondrocytes after monolayer culture (
In contrast, FAI (P = 0.002) and healthy (P = 0.015) control group showed a significant stronger staining of Col2 after pellet culture compared to monolayer culture. There was no significant upregulation in Col2 staining for osteoarthritic control ( Fig. 4 ). Aggrecan deposition increased significantly in FAI (P = 0.0261), healthy (P = 0.0102), and osteoarthritic control group (P = 0.0325) after pellet culture compared with monolayer culture.
Col2 expression in chondrocytes after monolayer and pellet culture: immunohistochemical staining of (
Gene Expression
Gene expression in FAI samples was compared with gene expression in control samples of healthy cartilage as well as in osteoarthritic samples. Analysis was done after monolayer and after pellet culture to compare different stages of gene expression.
After monolayer culture FAI chondrocytes from donors with Mankin score >3 showed a significant higher expression level of Col1 (P = 0.0007) and RunX2 (P = 0.0080) than FAI chondrocytes with Mankin score ≤3. The expression of Col2 was significantly decreased (P = 0.0002) in FAI samples compared with healthy controls. Col10 (P = 0.2331) and Sox9 (P = 0.8552) expression levels were not significantly affected by donor type.
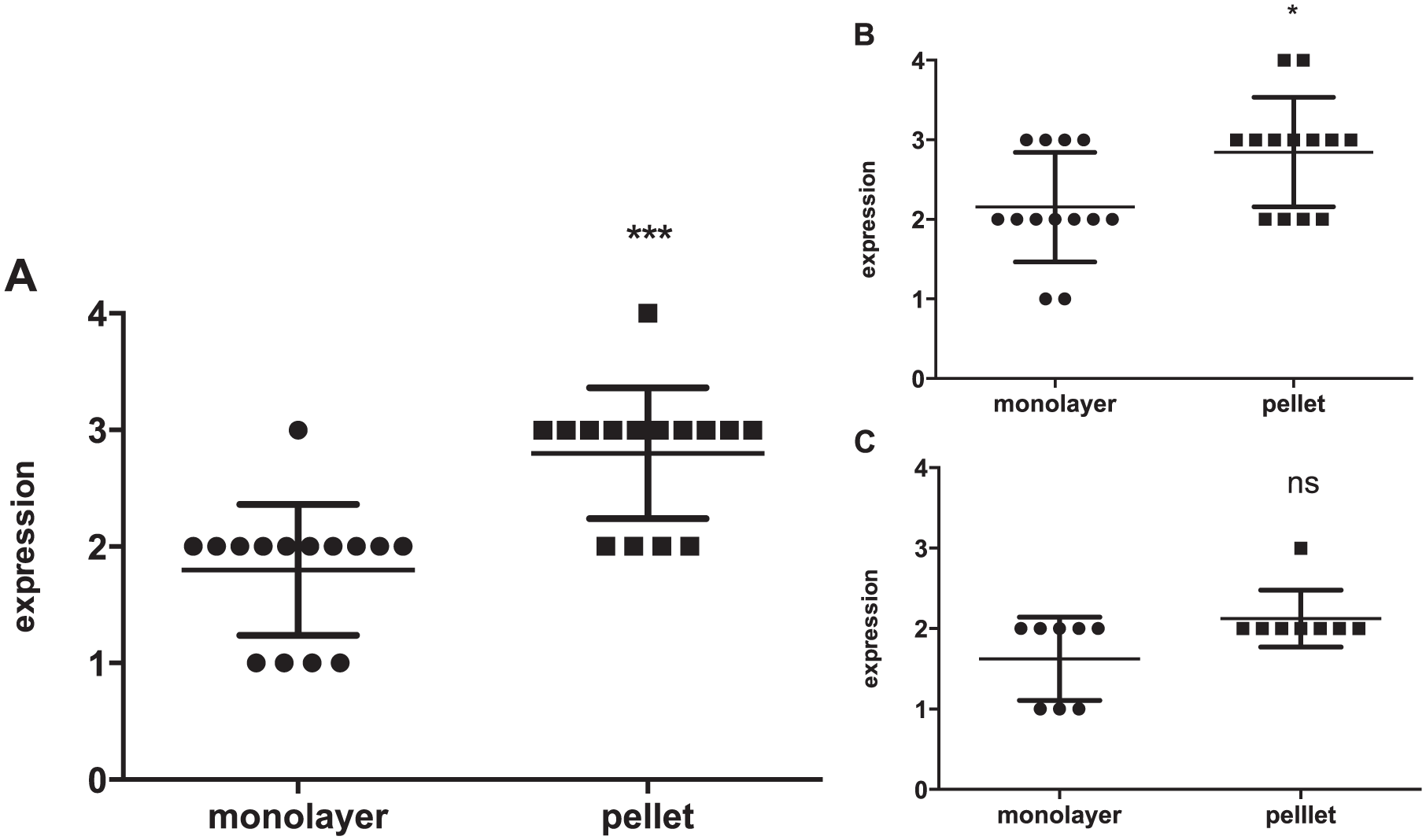
After pellet culture, healthy controls showed significantly decreased (P = 0.0050), osteoarthritic controls significantly increased expression (0.0050) of Col 1. For FAI group no effect of pellet culture on Col1 expression levels was observed. Col2 expression levels increased significantly in FAI group (P = 0.0050) as well as in healthy control (P = 0.0038) after pellet culture compared with healthy control after monolayer. For osteoarthritic control, no effect of pellet culture on Col2 expression levels was observed. Collagen 10 expression levels increased strongly after pellet culture in all analyzed groups (P = 0.0006). Increased Mankin score correlated significantly with Col10 (r = 0.8108, P = 0.0341) and RunX2 (r = 0.8829, P = 0.123) expression levels in the FAI group. Sox9 and RUNX2 gene expression levels were not significantly affected by donor tissue origin or cell culture conditions ( Fig. 5 ).
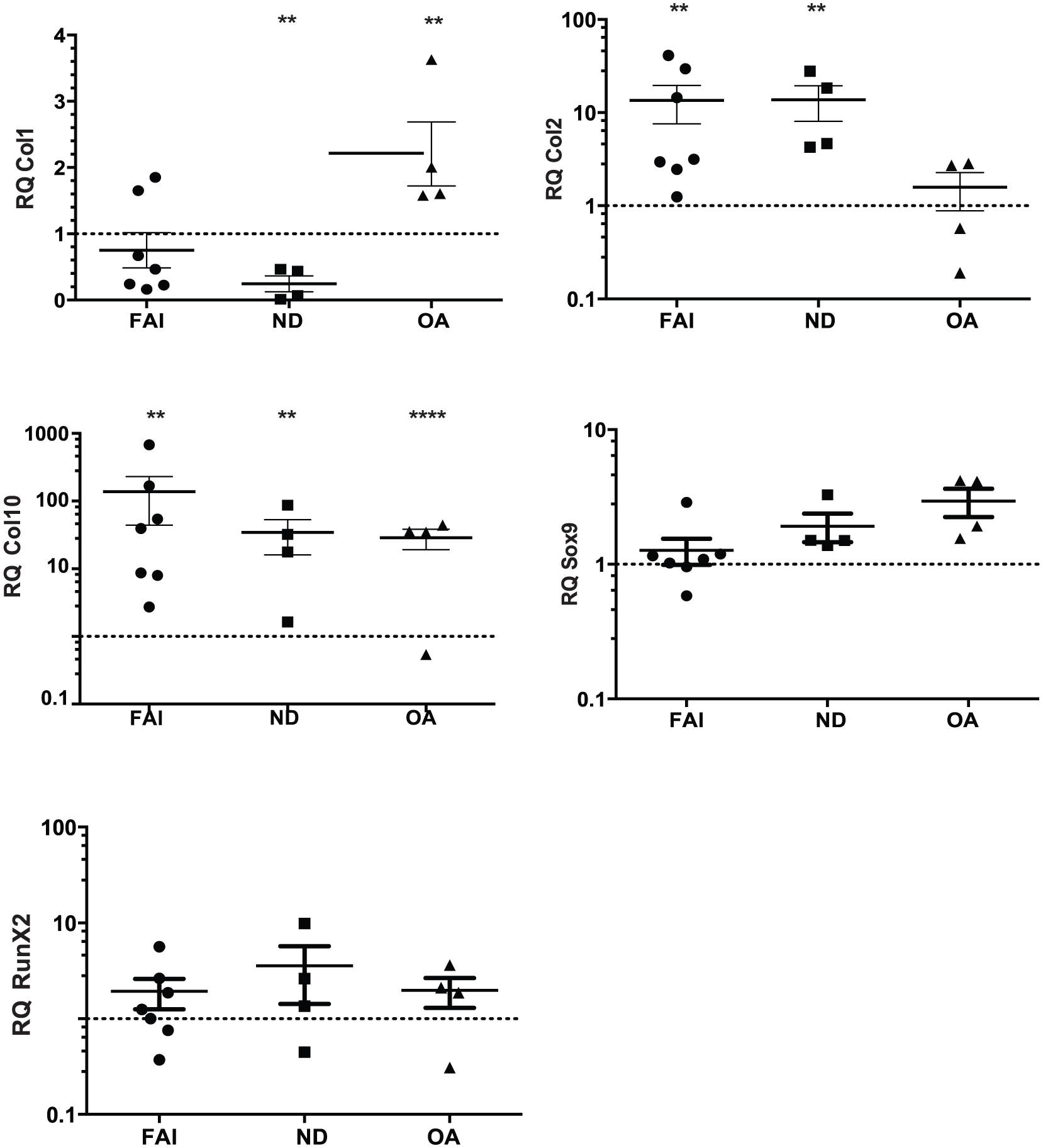
Relative gene expression after pellet culture for collagen 1, collagen 2, collagen 10, Sox9, and RunX2; normalized to relative gene expression of normal donors (ND) after monolayer culture (qRT-PCR; GAPDH as endogenous control) for femoroacetabular impingement (FAI; n = 7), normal donors (ND; n = 4), and osteoarthritic control (OA; n = 4). The data are expressed as single values and median ± standard deviation; gene expression of ND after monolayer culture as reference line (y = 1). Statistically significant differences are indicated by *P < 0.05, **P < 0.01, and ***P < 0.001.
Discussion
In the present study, cartilage samples of patients with cam-type FAI showed sufficient but heterogeneous composition relating to histological quality and chondrogenic potential with distinct differences compared with healthy cartilage. Therefore, cell harvesting within a low morbidity procedure like a knee arthroscopy should be still preferred, especially if the cartilage lesion is securely verified in the MRI. However, harvesting of chondrocytes from the cam lesion might be a valid option especially if a cartilage lesion is noted in a diagnostic arthroscopy and individual preexisting stage of cartilage degeneration and appropriate pellet-culturing conditions are considered. The concomitant cam surgery should preferably be done at the same time as ACT surgery to avoid 2 major surgeries.
Cam-type FAI is a common cause of hip pain and one of the most frequent reasons for cartilage defects at the hip. According to cartilage defects of the knee joint, different non-arthroplasty treatment options to regain functional cartilage at the hip are under investigation. In this field, ACT is a recent, promising method with good to excellent outcome in the knee joint, 7 though limited studies of ACT at the hip are available. Fontana et al. 11 demonstrated that patients who underwent ACT at the hip joint had a significantly better clinical outcome compared with patients who underwent debridement alone. In contrast to ACT at the knee joint, no standard biopsy location is defined.8,9 Cartilage for ACT of the hip can be harvested from unaffected joints, like the knee joint, with low morbidity and excellent cell quality.7,8,11 However, this procedure means an additional surgery to an unaffected joint with inherent complications. Therefore, femoroacetabular cell harvesting was suggested and showed satisfying results with adequate tissue quality in recent studies.12,13 Nonetheless, hip arthroscopy is a procedure with an increased morbidity and chondrocyte extraction from the central acetabulum affects the weightbearing acetabular surface and is still controversial. Since resection of the cam deformity commonly extends into the cartilage area covering the periphery of the femoral head, it might be a beneficial cartilage biopsy location for ACT of the hip beyond the weightbearing joint surface. Present study encouraged this possibility and showed acceptable histologic quality and chondrogenic potential of cartilage samples from the cam impingement area. All harvested samples were analyzed using Mankin score classification. This score is a valid method to classify cartilage according to degenerative, osteoarthritic changes. Cartilage samples harvested from patients with FAI demonstrated wide Mankin score spreading, and both healthy cartilage as well as degenerated cartilage could be observed in the FAI group. No correlation of Mankin score and patient age was detected. Furthermore, there was no correlation of arthroscopically determined acetabular cartilage damage (Beck score, Outerbridge score, Sampson score) with Mankin score of the cartilage covering the cam deformity of the femur. Thus, in case of harvesting FAI chondrocytes for ACT, quality of the used cartilage has to be analyzed individually. Neither extent of acetabular cartilage damage nor patient age seems to be a reliable predictor for quality of harvested cartilage.
In vitro monolayer expansion of chondrocytes is necessary to obtain high cell numbers for ACT, though it leads to dedifferentiation of chondrocytes. 21 It is an economical procedure modified by use of different culture media, growth factors, and substrates. 22 In order to be able to use FAI chondrocytes for ACT, it is important to analyze whether FAI chondrocytes have the potential to redifferentiate toward hyaline cartilage after in vitro expansion.
Expansion of chondrocytes in monolayer culture results in a decreased capacity to produce hyaline-like matrix. 23 To verify the chondrogenic potential of the harvested chondrocytes, Col1/Col2 ratio is still a classical marker. 24 After expansion in monolayer, FAI chondrocytes showed downregulation of Col2 expression compared with healthy control group. No further significant differences in expression of Col1, Col10, Sox9, and RunX2 between FAI and healthy chondrocytes were detected. Regarded separately, FAI chondrocytes with Mankin score >3 showed increased expression of Col1 and RunX2 compared with healthy chondrocytes and compared with FAI chondrocytes with Mankin score ≤3. This may give a hint on reduced chondrogenic potential of FAI chondrocytes with degenerative alterations. Col1 and RunX2 are merely expressed in small quantities in healthy cartilage. Increased expression is associated with dedifferentiation and loss of chondrogenic potential. 25 This fact complies with data shown by Yang et al., 26 who reported that chondrocytes with degenerative alteration show stronger decrease in Col2 expression during expansion compared with healthy chondrocytes. In conclusion, FAI chondrocytes with Mankin score ≤3 behave toward given monolayer culture conditions almost like healthy chondrocytes, whereas FAI chondrocytes with Mankin score >3 resemble arthritic cells. It had to be clarified if incipient dedifferentiation after monolayer culture affects redifferentiation potential of FAI chondrocytes in pellet culture. Different previous studies showed conflicting results concerning use of degenerated chondrocytes in ACT. Merely limited ability of proliferation and expression of chondrocyte-specific parts of extracellular matrix 27 is attested for osteoarthritic chondrocytes. Other studies attested osteoarthritic chondrocytes high chondrogenic potential and the ability to redifferentiate after loss of chondrocyte-specific phenotype. 28 Clinical outcome following knee ACT is, among others, significantly related to expression levels of Col2 at the time of transplantation. 29 Col2 is the most abundant and important of the extracellular matrix molecules in cartilage. 30 Both FAI and healthy chondrocytes were associated with significantly increased expression of Col2 after pellet culture, whereas osteoarthritic chondrocytes showed no significant increase of Col2 expression after pellet culture compared with monolayer cultivation. Thus, it must be assumed that there is an osteoarthritic threshold, particularly with regard to chondrogenic redifferentiation potential of chondrocytes isolated of osteoarthritic cartilage. Different expression levels of Col2 after monolayer culture between FAI and healthy chondrocytes were equalized during pellet culture.
Despite significant increase of Col2 expression, a near-constant Col1 expression could be detected in FAI chondrocytes after pellet culture compared with expression in monolayer. Incomplete redifferentiation is to trace back to the fact that elongated culture in monolayer took place before transfer to more physiological pellet culture. 31 In contrast to nearly similar results for Col1/Col2 ratio for FAI and healthy chondrocytes, osteoarthritic controls showed further dedifferentiation. Decreased Col2 expression linked with high expression of Col1 demonstrated that osteoarthritic chondrocytes seem not to be able to withstand nonphysiological culture conditions like monolayer culture. In contrast, healthy chondrocytes demonstrated the ability of chondrogenic redifferentiation as well as FAI chondrocytes. Deposition of Col2 and aggrecan after monolayer and pellet culture in IHC confirmed the results of gene expression analysis. The cartilage specific markers increased significantly after pellet culture, especially in FAI and healthy chondrocytes.
Other markers of de- and redifferentation of chondrocytes were additionally analyzed. Despite several efforts to enhance cell expansion, fibrocartilage formation and hypertrophy still represent frequent undesired occurrences during cell preparation for ACI. 32 Osteoarthritic chondrocytes are attributed to high gene levels of hypertrophic markers like Col10 and RunX2. 33 After pellet culture, all analyzed groups showed significantly increased gene expression of Col10. Upregulation of Col10 is ascribed to usage of chondrogenic growth factor TGF-β1. 34 Instead of TGF-β1, different growth factors may be used for culture of FAI chondrocytes in further studies to improve redifferentiation potential. This should minimize the risk of hypertrophy of FAI chondrocytes during culture process. Furthermore, our results demonstrated no significant difference in gene expression levels of Col10 and RunX2 in FAI chondrocytes (Mankin score ≤3) and healthy chondrocytes after monolayer culture, whereas again FAI chondrocytes with progressed osteoarthritic degeneration (Mankin score >3) showed increased expression of Col10 and RunX2. This observation further emphasizes the wide variety of degenerative changes that can be observed in chondrocytes of patients with FAI, and each sample should be evaluated individually.
No additional gain of information was obtained from Sox9 gene expression analysis. Transcription factor Sox9 has a crucial function concerning chondrocyte differentiation in vivo 35 ; furthermore, it is presumed that there is a connection between activity of transcription factor and increased Col2 expression. 36 Contrary to these results, Aigner et al. 37 could not detect a connection between these genes in adult chondrocytes. This fact is corroborated by in vitro study; Sox9 gene expression was not significantly altered in FAI chondrocytes and control groups.
An important aspect of this study was the availability of healthy control cartilage. On the basis of healthy controls, tissue from hips with FAI could be analyzed and classified. We conclude that hyaline cartilage samples with Mankin score ≤3 harvested during arthroscopic osteochondroplasty of FAI patients and healthy hyaline cartilage controls showed nearly similar behavior after expansion in monolayer and final redifferentiation in pellet culture.
Conclusion
Cartilage samples of patients with cam-type FAI showed sufficient but heterogeneous composition relating to histological quality and chondrogenic potential with distinct differences compared with healthy cartilage. However, harvesting of chondrocytes from the cam lesion might be a valid option especially if a cartilage lesion is noted in a diagnostic arthroscopy and individual preexisting stage of cartilage degeneration and appropriate pellet-culturing conditions are considered.
Footnotes
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The present prospective, controlled, single-center study was approved by the institutional review board of the Technische Universität München (Project No.: 2906/10).
Informed Consent
Written informed consent was obtained from all patients before the study.
Trial Registration
Not applicable.
