Abstract
Objective
The purpose of this study was first to externally validate the Oswestry Risk of Knee Arthroplasty index (ORKA-1) by applying it to an autologous chondrocyte implantation (ACI) patient cohort in the United States with a broader definition of failure than only arthroplasty, and second, to determine predictive factors for the risk of ACI failure as defined by the senior author.
Design
A total of 171 patients that underwent ACI were included to validate the ORKA-1 as all factors needed for calculation and outcomes were recorded. For Cox regression analysis, 154 patients were included as they completed preoperative Knee Injury and Osteoarthritis Outcome Score (KOOS), Tegner, Lysholm, International Knee Documentation Committee (IKDC), and 12-item Shor Form (SF-12) scores. Patient- and lesion-associated parameters were recorded for each patient.
Results
At final follow-up (maximum of 10 years post-ACI), a total of 27 patients (15.8%) were considered a failure by senior author’s definition. With ACI failure as endpoint, the mean survival was 7.96 years in risk group 1 and 5.4 years in risk group 5. Cox regression analysis identified preoperative KOOS Sport/Recreation as the only significant predictive factor for ACI failure (P = 0.007).
Conclusion
The ORKA-1 is a helpful tool for surgeons to estimate an individual patient’s likelihood of ACI survival. Further studies with larger patient cohorts as well as a consensus definition of failure are needed to further refine predictors of ACI failure.
Introduction
Articular hyaline cartilage with its type II collagen and glycosaminoglycan network provides exceptionally low-friction motion between joint surfaces but shows limited capacity of spontaneous healing. Successful treatment of articular cartilage lesions remains a challenge for the treating surgeon. In 1994, Brittberg and colleagues first reported outcomes after autologous chondrocyte implantation (ACI) for the treatment of symptomatic focal cartilage lesions in the knee. 1 In recent literature, ACI has been shown to result in successful outcomes in 82% of patients after a mean follow-up of 11.4 years and a survival rate of 63% after 20 years.2,3 However, numerous factors such as age, female gender, lower preoperative patient-reported scores, Worker’s Compensation (WC) status, longer duration of symptoms, defect number, location and size, and prior cartilage repair procedures have all been associated with less favorable results.4-7 A definite answer to which patients would benefit most from ACI for cartilage lesions is yet to be determined. In an attempt to predict survival of the ACI graft, Dugard et al. recently developed the Oswestry Risk of Knee Arthroplasty index (ORKA-1) for patients undergoing ACI for focal cartilage lesions of the knee. 4 This tool is designed to predict the risk of arthroplasty after ACI by assigning patients to a risk group from 1 to 5 based on individual patient and lesion characteristics. The risk of conversion to arthroplasty at 10 years after ACI ranges from a high of 100% in groups 4 and 5, to 70% in group 3, 11% in group 2, and 1% in group 1.
ACI is typically indicated in a younger population in which conversion to arthroplasty is generally a last resort. Therefore, the decision for arthroplasty is likely influenced more by patient age and availability of other treatment options than “just” objective graft failure, which is only inconsistently defined.8,9 Additionally, osteochondral allograft transplantation (OCA) has emerged as a salvage procedure for cartilage repair in the United States, 10 but availability is limited outside the United States, leading to discrepancies in treatment algorithms for failed cartilage repair between the United Kingdom and the United States.
We therefore sought to externally validate the ORKA-1 by investigating its applicability on overall failure (as defined in this study), rather than only conversion to arthroplasty, as an outcomes endpoint. The second objective was to identify predictors of failure, as defined in this study, and developing a score estimating the risk of failure after ACI for focal cartilage lesions in patients treated in the United States. We hypothesized that the ORKA-1 will have limited predictive value in patients undergoing ACI in the United States when used to predict overall failure.
Materials and Method
Our institution prospectively collects data for all cartilage repair patients. Patients indicated for cartilage repair with ACI for focal cartilage defects in the knee by a single surgeon between March 2007 and August 2017 were enrolled for this study. Our institutional review board approved the study prior to initiation. Surgical treatment was indicated in patients after failed nonoperative treatment, including physical therapy and injectable therapies. Comorbidities including inflammatory joint disease, unresolved or recent septic arthritis, metabolic or crystal disorders, or deficient soft-tissue coverage were considered contraindications for ACI. Patients with patellar maltracking, lateral and/or distally located patellar chondral lesions, and/or tibiofemoral malalignment >3° from the neutral mechanical axis into the involved compartment were treated with concomitant osteotomy. Patients with concomitant ligament tears, or meniscal deficiencies were addressed with adequate ligamentous reconstruction or meniscal allograft transplantation (MAT). Patients with concomitant procedures at the time of ACI were not excluded from this study.
The first objective of this study was to externally validate the ORKA-1 by applying it on overall failure, rather than only conversion to arthroplasty, as an outcomes endpoint. Based on their age, sex, number of cartilage lesions, occurrence of a patellar defect, whether one had multiple previous operations, and the preoperative Lysholm score, the ORKA-1 designates patients to risk groups from 1 to 5, with group 1 having the lowest and group 5 the highest risk of arthroplasty after ACI. The risk for each patient requiring arthroplasty in a period of 15 years after ACI can then be estimated by a Kaplan-Meier survival analysis provided by the authors. 4 Therefore, patients with incomplete preoperative Lysholm, incomplete patient demographic data, unreported defect morphology, or unreported outcome were excluded from this part of the study. While the ORKA-1 was designed to predict solely the risk of arthroplasty, our study defined overall failure after ACI as a new endpoint. Hence, all patients who underwent revision ACI, subsequent osteochondral autograft transfer (OAT), OCA, arthroplasty, or failure of more than 25% of the ACI graft as judged on postoperative imaging or second-look arthroscopy were considered as failure.
The second objective was to determine potential predictive factors for ACI failure as described above. Each patient enrolled in this analysis completed the 12-item Shor Form (SF-12), Knee Injury and Osteoarthritis Outcome Score (KOOS), Lysholm, Tegner, and International Knee Documentation Committee (IKDC) evaluation forms. Patient’s age at the time of surgery, body mass index (BMI), sex, smoking status, WC status, and whether the patient had concomitant surgeries such as osteotomy or MAT, and/or multiple previous surgeries on the index knee were recorded. Cartilage defect morphology, including the size, number, subchondral bone involvement, and the occurrence of a patella lesion were collected from surgical notes.
Statistical Analysis
For the first objective, the ORKA-1 score was calculated for each patient. A Kaplan-Meier survival analysis was utilized to estimate the cumulative risk of failure for patients based on their designated risk group. Patient outcomes were compared between risk groups using the Mantel-Cox log-rank test. Generated Kaplan-Meier curves were compared to the original Kaplan-Meier survival analysis provided by the ORKA-1 publication to validate if designated risk groups demonstrated predicted outcome. 4
For the second objective, descriptive statistics were calculated to determine the sociodemographic and clinical characteristics of patients. Categorical variables were coded as dummy variables for univariable and multivariable Cox proportional hazard models (i.e., for sex, 0 represented male and 1 represented female). Models included patient age, sex, BMI, osteotomy, MAT, multiple previous surgeries, WC status, smoking status, SF-12, KOOS subgroups, Lysholm, Tegner, and IKDC scores, defect number, defect size, and occurrence of a patella lesion. Each potentially predictive factor was assessed by using univariable Cox regression. Associations displaying significance at a level of P < 0.15 were included in a multivariable Cox proportional hazard model utilizing stepwise regression.
All statistical analyses were performed in SPSS for Mac (Version 23.0. SPSS Inc., Chicago, IL) and Stata (Version 13, Statacorp LP, College Station, TX). Significance was set at P < 0.05.
Results
The senior author treated a total of 287 patients with ACI for focal symptomatic cartilage lesions within the knee joint during the study period. Of these patients, 119 patients were excluded for the validation of the ORKA-1, as 72 patients had no preoperative Lysholm score and the outcome for 44 patients could not be determined as they neither returned to our clinic within the last 12 months nor provided up-to-date contact information. Thus, 171 patients (59.6%) were evaluated for the first objective of this study. The Kaplan-Meier survival analysis is presented in Figure 1 .
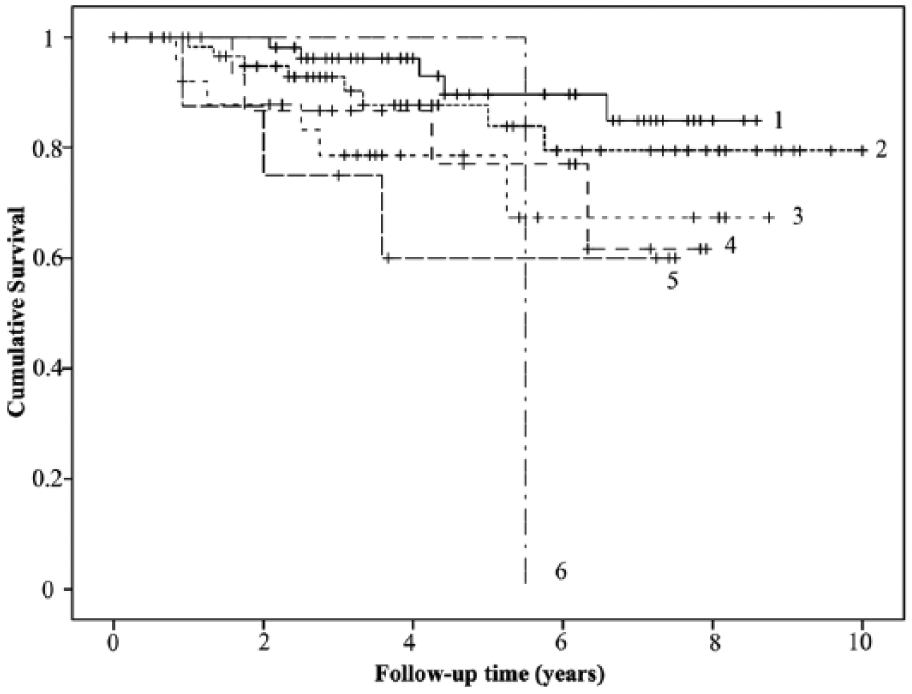
Survival of ACI (autologous chondrocyte implantation) with the senior author’s definition of failure as the endpoint for patients belonging to the 6 designated risk groups based on the ORKA-1 (Oswestry Risk of Knee Arthroplasty index).
Of the included patients, 6 patients had a rounded risk index value of 7, and 1 patient presented with a risk index value of 9. As the ORKA-1 is not clearly defined for patients with risk index values above 5, patients with a risk index value of 7 were designated to risk group 5, whereas the patient with a risk index value of 9 was designated to a new risk group 6. Hence, 57 (33.3%) patients were designated to risk group 1, 64 (37.4%) to group 2, 26 (15.2%) to group 3, 15 (8.77%) to group 4, 8 (4.68%) to group 5, and 1 patient to group 6.
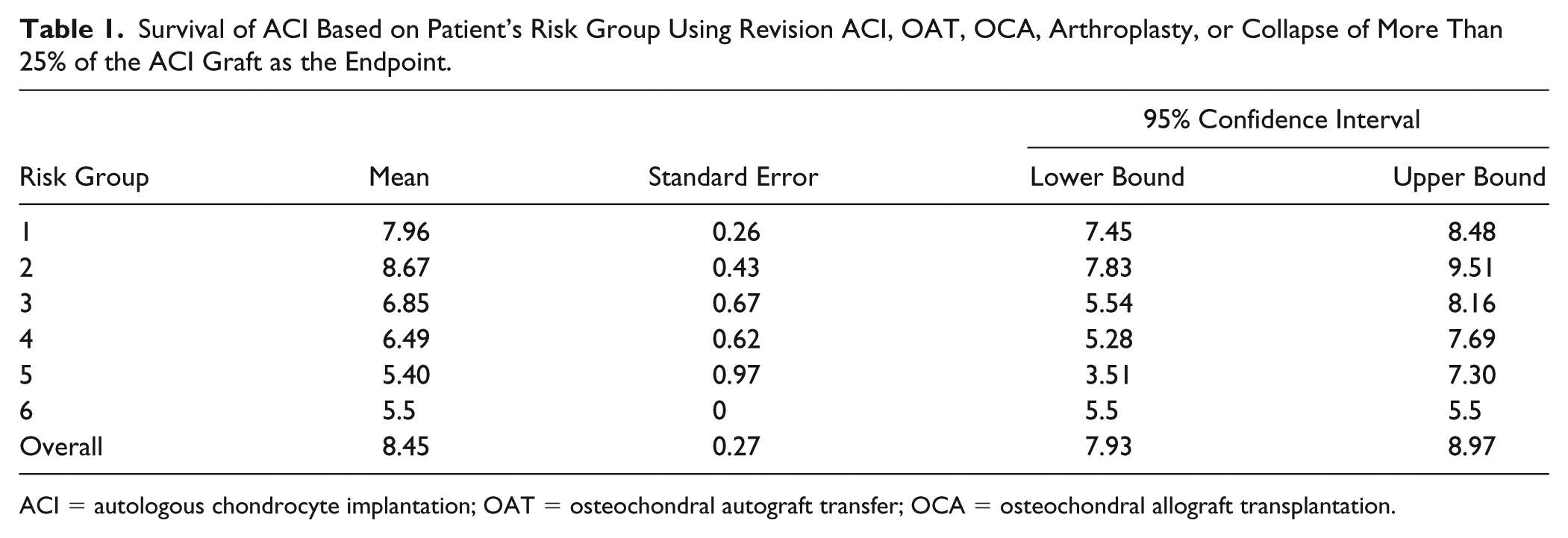
With the endpoint set as this study’s definition of failure, 27 patients (15.8%) underwent revision ACI, OAT, knee arthroplasty, OCA or were diagnosed with ACI graft failure of more than 25% at a mean of 8.45 (95% confidence interval = 7.93-8.97) years after ACI ( Table 1 ). At 7 years, the cumulative risk of ACI failure was 14% in risk group 1, 20% in risk group 2, 30% in risk group 3, 38% in risk group 4, and 40% in risk group 5. The patient in risk group 6 underwent OCA after 5.5 years ( Fig. 1 ). The Mantel-Cox log-rank test revealed no significant difference in hazard rates between risk group 1 and groups 2 and 4 (P = 0.32 and P = 0.079, respectively). Setting group 1 as baseline, risk groups 3, 5, and 6 showed significantly increased hazard ratios (P = 0.03; P = 0.017; P = 0.025).
Survival of ACI Based on Patient’s Risk Group Using Revision ACI, OAT, OCA, Arthroplasty, or Collapse of More Than 25% of the ACI Graft as the Endpoint.
ACI = autologous chondrocyte implantation; OAT = osteochondral autograft transfer; OCA = osteochondral allograft transplantation.
For the second objective of the study, 154 (53.1%) patients were included in the Cox proportional hazard model to define potential predicators for failure as 72 patients had no preoperative Lysholm score, an additional 8 patients had also incomplete preoperative KOOS scores, 10 patients had no documented preoperative BMI, and the outcome for 44 patients could not be determined as they neither returned to our clinic within the last 12 months nor provided up-to-date contact information. Of the included patients, 84 (54.9%) were female, 11 (6.2%) were active smokers, 10 (6.5%) were WC, 59 (38.6%) had multiple previous operations on their index knee, 106 (68.8%) underwent concomitant osteotomy, 13 (8.4%) underwent concomitant MAT, 16 (10.4%) had an osteochondral lesion, and 99 (64.3%) had at least one cartilage lesion on the patella. Concomitant osteotomies included 19 high tibial osteotomies, 89 tibial tubercle osteotomies, and 8 distal femoral osteotomies. The number of lesions ranged from 1 to 5 defects, with 49.7% of patients presenting with 1 defect, 30.7% with 2 defects, 14.4% with 3 defects, 3.9% with 4 defects, and 1.3% with 5 defects across the knee joint. The mean age was 31.8 (SD, 9; range, 15-56) years with an average BMI of 26.5 (SD, 3.98; range, 18-38.2) kg/m2. The combined size of all assessed cartilage defects in a patient averaged 760 (SD, 528.41; range, 56-2753) mm2.
Age, defect number and size, WC, all KOOS subscales, Lysholm, IKDC, and SF-12 Physical Component Summary (PCS) showed significant association with ACI failure in the univariable Cox regression models (with P < 0.15) ( Table 2 ). When including all significant factors in a stepwise multivariable Cox proportional hazard model, KOOS Sport/Recreation subscale was the only factor associated with an increased risk of ACI failure (hazard ratio = 0.97; 95% confidence interval = 0.95-0.99; P = 0.007).
Univariable Analysis of Predictive Value from Potential Predictors on the Survival of ACI Using Senior Author’s Definition of Failure as Endpoint.
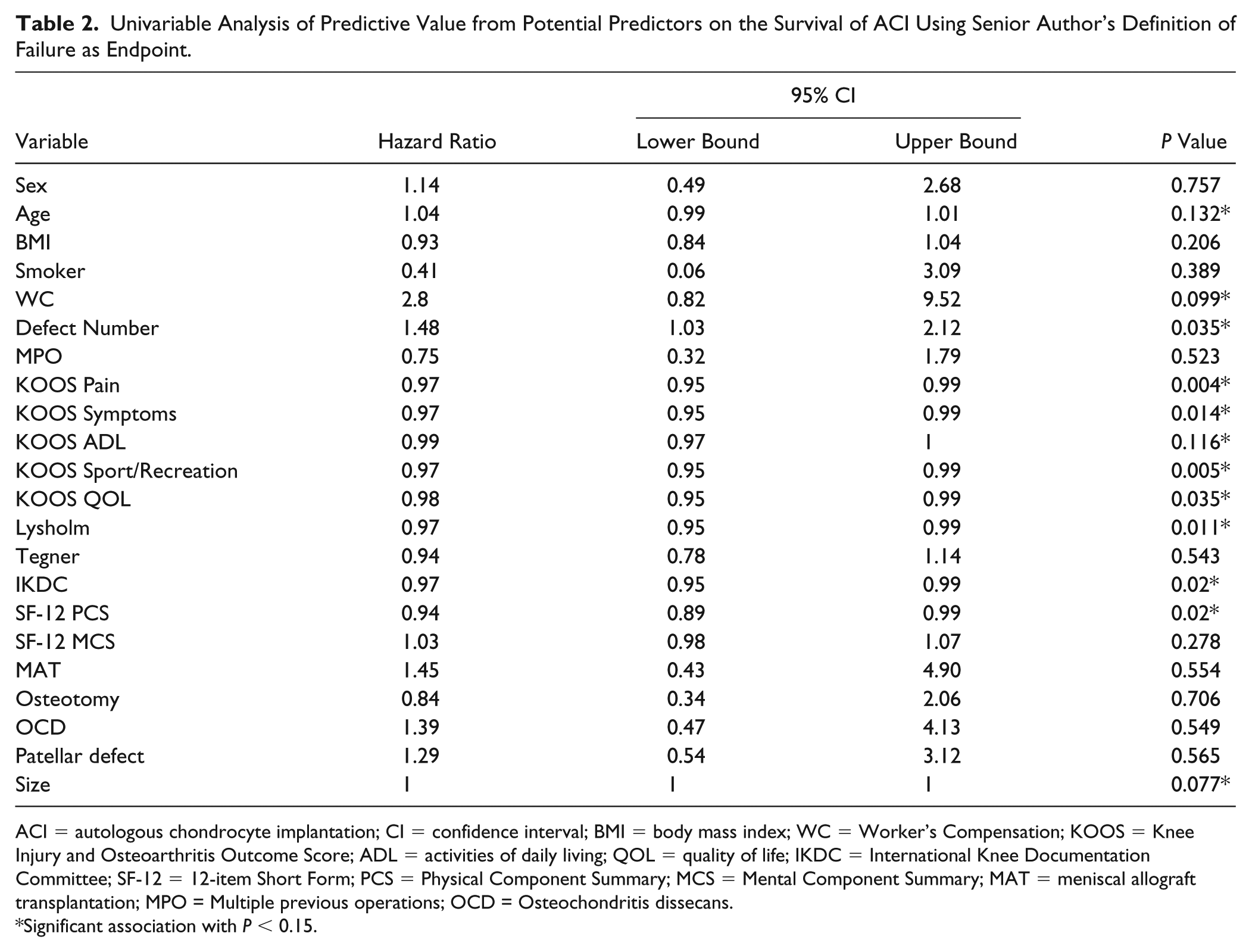
ACI = autologous chondrocyte implantation; CI = confidence interval; BMI = body mass index; WC = Worker’s Compensation; KOOS = Knee Injury and Osteoarthritis Outcome Score; ADL = activities of daily living; QOL = quality of life; IKDC = International Knee Documentation Committee; SF-12 = 12-item Short Form; PCS = Physical Component Summary; MCS = Mental Component Summary; MAT = meniscal allograft transplantation; MPO = Multiple previous operations; OCD = Osteochondritis dissecans.
Significant association with P < 0.15.
Discussion
Since being introduced in 1994, ACI has become an established therapeutic option for full-thickness cartilage defects of the knee. 1 Having been in use for over 20 years, multiple studies have reported on both its safety and efficacy.11-20
Even though techniques for ACI have continued to develop and improve, failure still occurs. In one recent systematic review, the failure rate was found to be 14.9% among 4294 patients after an average follow-up between 5 and 12.3 years for both ACI and matrix-assisted autologous chondrocyte transplantation. 9 Different authors have created their own definition of what constitutes ACI failure, yet there is no universal consensus on a standard definition. 21 In the same systematic review mentioned above, high variability was noted in authors’ definitions of failure. In analyzing 58 articles, 12 studies did not state the definition of failure in their study while the remaining 46 studies varied widely in their definition. Thirty-eight studies used a surgical definition, 2 studies a radiologic definition, and 1 study a clinical definition. Five other studies used a combination of the above. 9 This discrepancy in the definition of failure of ACI between authors may lead to inaccurate representation of facts when discussing goals of care with patients, or when providers are attempting to estimate the success of a procedure.
Recently, the ORKA-1 index was developed to estimate the long-term hazard of arthroplasty after ACI. 4 The potential value of this index is that it may be used in the preoperative setting to better inform patients and provide surgeons with data to estimate ACI survival rates for patients with specific risk factors. Even though it was described as useful by the center where it was developed with their specific patient population, any new score benefits from validation in other clinical settings with potentially different patient populations and revision options. This article focuses on the external validity of the ORKA-1 index for settings with expanded treatment options and more accurately represent a broader patient population, and we expanded the failure definition for the ORKA-1 index beyond revision with arthroplasty.
This definition expanded the definition of failure to include patients who underwent revision ACI, subsequent OAT, OCA, arthroplasty, or a failure of more than 25% of the ACI graft, as evaluated on postoperative imaging or second-look arthroscopy. The threshold of revising failed cartilage repair with OCA is likely lower than when arthroplasty is the only option, especially for younger patients. While OCA is not readily available in many parts of the world, it is an important treatment option for revision of failed cartilage repair, and we postulated that many of the patients in the original patient group treated with arthroplasty might have been offered OCA. Therefore, we felt that broadening the failure definition would more accurately reflect the reality where some patients who objectively have failed might decline, or not be offered, revision with arthroplasty, choose to live with pain, and therefore not be counted as a failure under the more narrow definition. Thus, if treatment options such as OCA were more readily available in other parts of the world, ACI revision rates would likely increase, while still allowing patients to retain their biological knee. Conversely, indication for ACI in the United States differs from other countries since treatment alternatives such as OCA exist. Hence, bilayer collagen membrane ACI with bone graft (sandwich ACI) for osteochondral lesions is less likely performed in the United States compared with Europe, which might also contribute to varying outcomes seen after ACI.
With the expanded definition of failure in this study as the endpoint, the mean survival was 7.96 years in risk group 1 and 5.4 years in risk group 5. This demonstrates mean survival times that are representative of the risk assumed for each group: higher mean survival time for low-risk groups and lower mean survival times for higher risk groups ( Table 1 ). By expanding the definition of failure to include post-ACI treatments more commonly seen in a younger patient population, the ORKA-1 index demonstrates predictive character. Calculations that, at the very least, can be extrapolated to a larger patient demographic, particularly in terms of patient age and revision options that might be determined by geographic location of the patient.
The second objective of this study was to determine predictive factors for risk of ACI failure using this study’s definition of failure. For this portion of the study, 154 patients were included as they had completed preoperative KOOS, Tegner, Lysholm, IKDC, and SF-12 scores. Age, sex, smoker status, BMI, WC, previous surgeries, concomitant surgeries, osteochondral lesions, number of defects, lesion location in the patella, and total defect size were recorded for each patient. Univariable regression analysis demonstrated that age, defect number and size, WC, all KOOS subscales, Lysholm, IKDC, and SF-12 PCS showed significant association with ACI failure. In combining all significant factors, however, a stepwise multivariable Cox proportional hazard model determined KOOS Sport/Recreation subscale as only factor associated with an increased risk of ACI failure. Though being statistically significant, this association can be considered as mild since patients with lower scores in the KOOS Sport/Recreation subscale experienced just slightly shorter ACI survival times than did patients with higher scores (hazard ratio = 0.97; 95% confidence interval = 0.95-0.99; P = 0.007).
With over 70% of patients expecting pain-free participation in sports post ACI, 22 tempering patient expectations for success through improved knowledge of preoperative risk factors is paramount. It is important to remember that a patient’s postoperative outcome/satisfaction after surgery has shown to correlate with their initial expectations for the procedure.23,24 Aside from discussing expectations, cost of the procedure should also be taken into account. With ACI being an expensive procedure with long rehabilitation, cost-benefit ratios are becoming increasingly important for patients and the social system, and should be discussed together with the risk for failure. If a patient falls into a high-risk category, he or she may not wish to incur financial liability or care for dealing with the rigors of postoperative physical therapy knowing he or she has a relatively high chance of failure. A better understanding of preoperative risk factors allows the patient and physician to work together on deciding the best treatment strategy to address cartilage lesions.
Regarding limitations, one potential shortcoming in our study is the size of our patient cohort. In order to determine further risk factors for ACI failure, more studies with larger patient cohorts are needed. To encourage uniformity across studies, future studies should include a consensus definition of failure.
In summary, caution must be exercised when using indices established in other countries with different treatment algorithms. These differing treatment protocols may lead to inaccurate estimates of ACI survival due to different thresholds for recommending revision with subsequent cartilage repair procedures versus arthroplasty. However, the ORKA-1 does show predictive character for estimating patient’s risk of ACI failure as defined by this study. Nonetheless, the identification of risk factors for ACI failure and communication of these results to patients is of great importance and helps set patient expectations. In addition, a comprehensive and specific definition of failure for ACI, preferably one that takes into account the patient’s perception of outcome, may be of help for better patient selection and counseling.
Footnotes
Authors’ Note
The study was performed at the Cartilage Repair Center and Center for Regenerative Medicine, Brigham and Women’s Hospital, Harvard Medical School, Harvard University, Boston, MA, USA.
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The senior author (AGH) is a consultant for Vericel Corporation.
Ethical Approval
Ethical approval for this study was obtained from Partners Human Research Commitee (2017P002003/PHS).
Informed Consent
Waiver of informed consent was received from IRB.
