Abstract
Background
The management of complex cartilage pathology in young, otherwise healthy patients can be difficult.
Purpose
To determine the nature of the design, endpoints chosen, and rate at which the endpoints were met in published studies and ongoing clinical trials that investigate cartilage repair and restoration procedures.
Study Design
Systematic review.
Methods
A systematic review of the publicly available level I/II literature and of the publicly listed clinical trials regarding cartilage repair and restoration procedures for the knee was conducted adhering to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.
Results
Seventeen published studies and 52 clinical trials were included. Within the 17 published studies, the most common procedure studied was microfracture (MFX) + augmentation (N = 5; 29.4%) and the most common comparison/control group was MFX (N = 10; 58.8%). In total, 13 different cartilage procedure groups were evaluated. For published studies, the most common patient-reported outcome (PRO) measures assessed is the Knee Injury and Osteoarthritis Outcome Score (KOOS) and Visual Analog Scale–Pain (VAS) (N = 10 studies, 58.8% each, respectively). Overall, there are 10 different PROs used among the included studies. Ten studies demonstrate superiority, 5 demonstrate noninferiority, and 2 demonstrate inferiority to the comparison or control groups. For the clinical trials included, the most common procedure studied is MFX + augmentation (N = 16; 30.8%). The most common PRO assessed is KOOS (N = 36 trials; 69.2%), and overall there are 24 different PROs used among the included studies.
Conclusions
Recently published studies and clinical trials evaluate a variety of cartilage repair and restoration strategies for the knee, most commonly MFX + augmentation, at various time points of outcome evaluation, with KOOS and VAS scores being used most commonly. MFX remains the most common comparison group for these therapeutic investigations. Most studies demonstrate superiority versus comparison or control groups. Understanding the nature of published and ongoing clinical trials will be helpful in the investigation of emerging technologies required to navigate the regulatory process while studying a relatively narrow population of patients.
Keywords
Introduction
The management of complex cartilage pathology the young, otherwise healthy patient can be difficult. While patients can experience pain relief and improved function with nonoperative treatment, including activity modification, anti-inflammatories, and injection-based treatments, many patients require surgery to improve symptoms and restore joint biomechanics and function. Certainly, partial or total knee arthroplasty can be a definitive solution for patients with severe and/or diffuse chondral pathology. Joint replacement, however, is less than ideal for younger and/or active patients, especially those with moderate- to high-demand activity levels and more limited articular cartilage disease. 1 Notably, younger patient age has been shown to be a negative prognostic factor for clinical outcomes and reoperations following knee arthroplasty.2-4 Thus, improved strategies toward preserving rather than replacing the joint are critical for the management of this patient population.
Surgical procedures for the management of articular cartilage lesions of the knee are often classified into palliative procedures including debridement and chondroplasty; reparative procedures such as marrow stimulation, including microfracture (MFX); restorative procedures, including autologous chondrocyte implantation (ACI) and surface allograft treatments; and reconstructive procedures, including osteochondral autograft (OAT) and allograft transplantation (OCA).5-18 Most recently, novel reparative techniques, including enhanced/augmented MFX, matrix-associated autologous chondrocyte implantation (MACT/MACI), and an increased offering of off-the-shelf osteochondral and chondral allograft products have become available.19-36 As a result, there is a large volume of prospective clinical trials comparing such surgical treatments for the management of chondral defects of the knee. Such trial-based data are helpful in that treatments are often compared with another or with a gold-standard control, but this data is also a source of confusion given the variability of methodology making it difficult to compare outcomes.
Therefore, the purpose of this study was to perform a systematic review of all published and ongoing clinical trials evaluating cartilage repair and restoration of the knee, and to determine similarities and/or differences in methodology especially as it pertains to trials that demonstrated superiority for the therapies of interest. The authors hypothesized that trials evaluating the effectiveness of ACI would be most prevalent, patient-reported outcome (PRO) measures assessed across trials would be highly variable and nonstandardized, and follow-up would be relatively short-term among the included studies. In addition, obtaining information as to which study designs were most frequently associated with meeting the primary endpoint of the study would be helpful for the planning of future studies.
Methods
Search Strategy for Publicly Available Literature and Clinical Trials
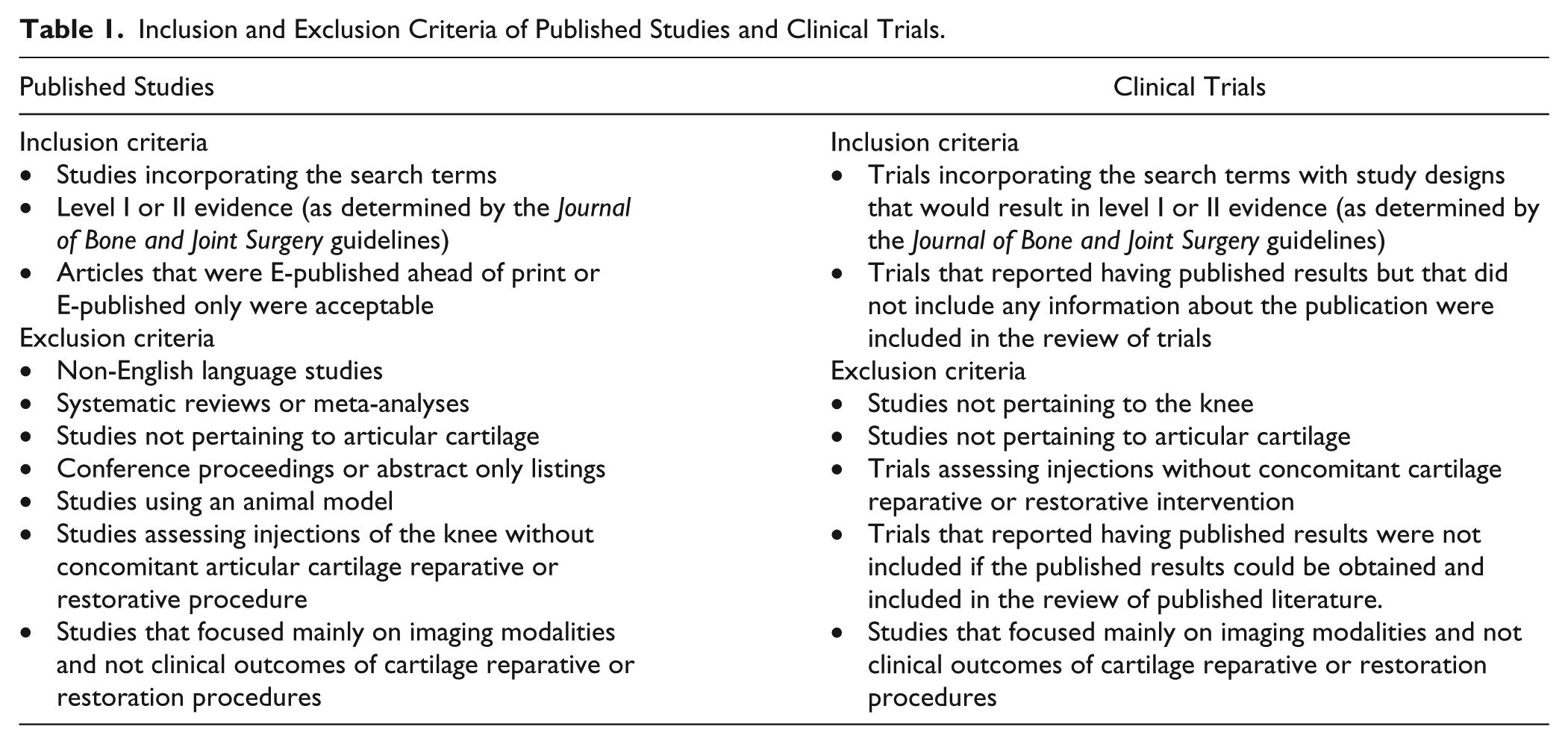
A systematic review of the publicly available literature and of the publicly listed clinical trials regarding cartilage repair and restoration of the knee was conducted adhering to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The search of publicly available literature was conducted on April 7, 2017 using the following databases: Medline (PubMed); Cochrane Central Register of Controlled Trials; and Ovid MEDLINE. The PubMed search strategy was as follows: Search 1, “knee” [All Fields] AND “cartilage” [All Fields] OR “chondral” [All Fields] AND “defect” [All Fields] OR “injury” [All Fields]; Filters: 1/1/2013–12/31/2017; Filters: human. This search strategy was repeated in the Cochrane and Ovid databases. The complete list of studies was obtained and initially screened by title. The remaining studies were then screened by abstract and when necessary by full manuscript. All stages of review (screening, eligibility, and inclusion/exclusion) of both published studies and current clinical trials were performed by 2 independent reviewers. Table 1 reports the inclusion/exclusion criteria. The data collected from the published studies are presented in Table 2 .
Inclusion and Exclusion Criteria of Published Studies and Clinical Trials.
Datapoints Extracted From Published Studies and Clinical Trials.

The search of publicly listed clinical trials was performed on clinicaltrials.gov on April 7, 2017. The search terms were “knee” AND (“cartilage” OR “chondral”) AND (“defect” OR “injury”). The results of this search were filtered to include only studies that have been updated after January 1, 2013. Trials were initially screened by title, and then by review of the study information page on clinicaltrials.gov. Table 1 reports the inclusion/exclusion criteria. Of note, studies used imaging modalities as a primary outcome measure were excluded as were studies that evaluated only injection therapies. The data collected from clinical trials are presented in Table 2.
Data Analysis
As this study is largely observational, advanced statistics were not necessary for data synthesis and presentation. Descriptive statistics were calculated for each study and parameters or variables analyzed. Categorical data were reported as frequencies with percentages. Continuous variable data were reported as either weighted means or mean ± standard deviation.
Results
The initial search terms identified 640 unique published articles from the publicly available literature search, and 338 unique clinical trials from the clinical trials search prior to screening. After the application of eligibility screening and filtering via exclusion criteria, 17 published studies and 52 clinical trials were included in the final analysis from the database and www.clinicaltrials.gov searches, respectively ( Figs. 1 and 2 ).
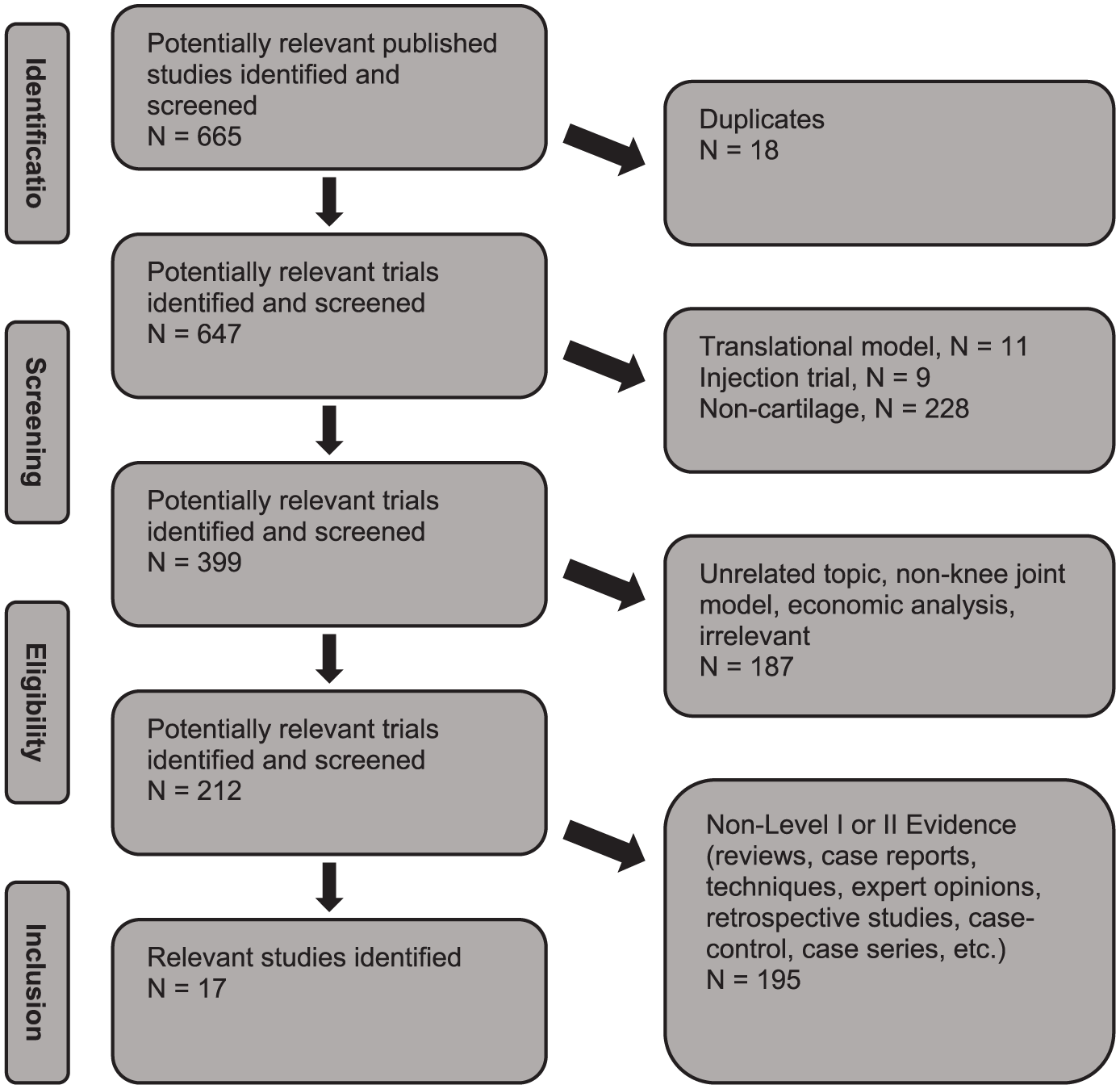
Search algorithm used to identify and screen studies to be included in the review of published literature.
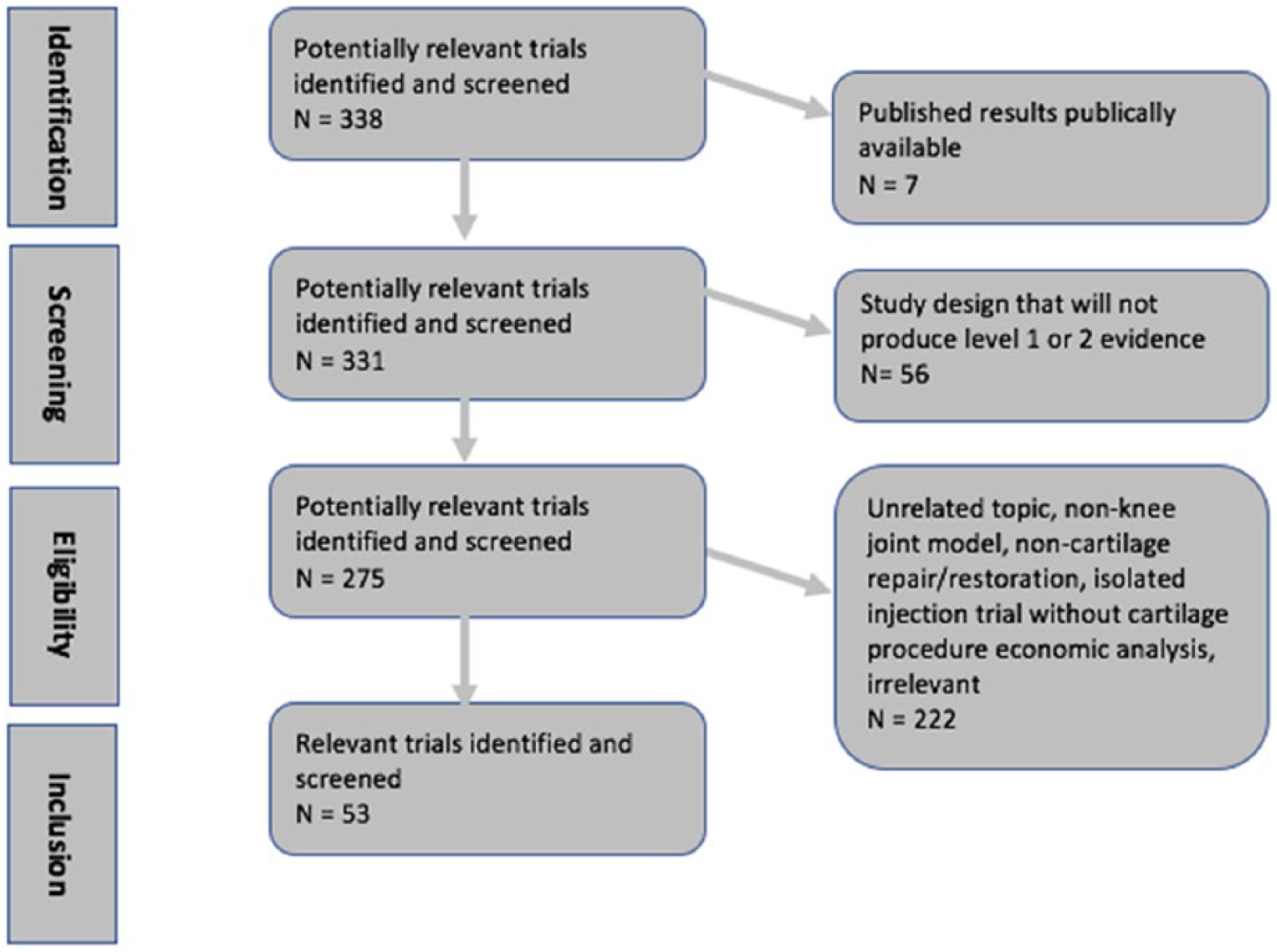
Search algorithm used to identify and screen trials to be included in the review of publicly listed clinical trials.
Published Clinical Trials in Cartilage Repair
Appendix Table A1 details the synthesized information from the publicly available literature. There were 8 journals in which the publicly available articles have been published. The studies were performed in 10 countries (some being performed in >1 country), with Norway (N = 4; 23.5%) as the most common. The majority of studies over the included time period were published in 2016 (N = 6; 35.3%). Most studies were multicenter (N = 12; 70.6%), randomized controlled trials (N = 12; 70.6%), with level I evidence (N = 10; 58.8%), and no financial conflicts of interest reported (N = 11; 64.7%). The duration of study for those of the included articles that reported this datapoint was a mean 3.77 ± 3.88 years (range, 0.17-16.17 years). At final follow-up, the overall mean follow-up rate was 85.6% (range, 55%-100%). The most common cartilage repair/restoration treatment studied was MFX + augmentation (N = 5; 29.4%), followed by ACI (N = 3; 17.6%). In total, 13 different cartilage procedure groups were used in the 17 studies. The most common control/comparison group utilized was MFX (N = 10; 58.8%).
The majority of studies included patients aged 18 to 50 years (N = 5; 29.4%). Most studies specified a radiographic Kellgren-Lawrence grade 0 (N = 6; 35.3%) in the inclusion criteria ( Appendix Table A1 ). Notably, however, all studies were performed in an effort to study the treatment of a single localized cartilage defect. The majority of studies targeted grade 3 or 4 lesions on International Cartilage Repair Society (ICRS) score (N = 11; 64.7%) in the inclusion criteria; 2 studies (11.8%) included only ICRS grade 3 lesions, and the remaining 4 studies (23.5%) targeted Outerbridge grade 3 or 4 lesions for inclusion ( Appendix Table A1 ). The overall weighted mean age for treated patients among all published studies was 35.6 years (SD, 6.2; mean age ranges across studies, 29.2-55.4 years), with overall weighted mean BMI for treated patients among reporting studies at 26.2 kg/m2 (SD, 1.4; mean BMI range 24.1-27.9 kg/m2). The most common time points for reporting of patient follow-up posttreatment was 12 months (N = 11 studies; 64.7%), and ranged from 12 weeks (N = 3; 17.6%) to 15 years (N = 1; 5.9%) with 9 total time points used ( Fig. 3 ). Published studies most frequently reported outcomes at 1 time point (N = 7, 41.2%) or 3 (N = 7; 41.2%), but overall, studies ranged from 1 to 4 time points of patient outcome reporting ( Fig. 4 ). The most common PROs assessed in the included studies were Knee Injury and Osteoarthritis Outcome Score (KOOS) (N = 10; 58.8%) and VAS (N = 10; 58.8%) with KOOS (N = 7; 41.2%) and International Knee Documentation Committee Score (IKDC) (N = 7; 41.2%) being the most common primary endpoints. Overall, there were 10 different PROs used among the included published studies ( Fig. 5 ). Eleven studies (64.7%) obtained imaging. Nine studies (52.9%) used magnetic resonance imaging (MRI) as a primary or secondary endpoint and used scoring systems that included the magnetic resonance observation of cartilage repair tissue (MOCART) score (N = 4; 23.5%), volume of cartilage repair (N = 4; 23.5%), or subchondral bone edema (N = 3; 17.6%). Three studies (17.6%) obtained radiographs as a secondary endpoint. Six studies (35.3%) obtained histologic analysis of cartilage repair/restoration tissue in follow-up, most commonly to determine the ICRS II score (N = 5; 29.4%) ( Appendix Table A1 ).
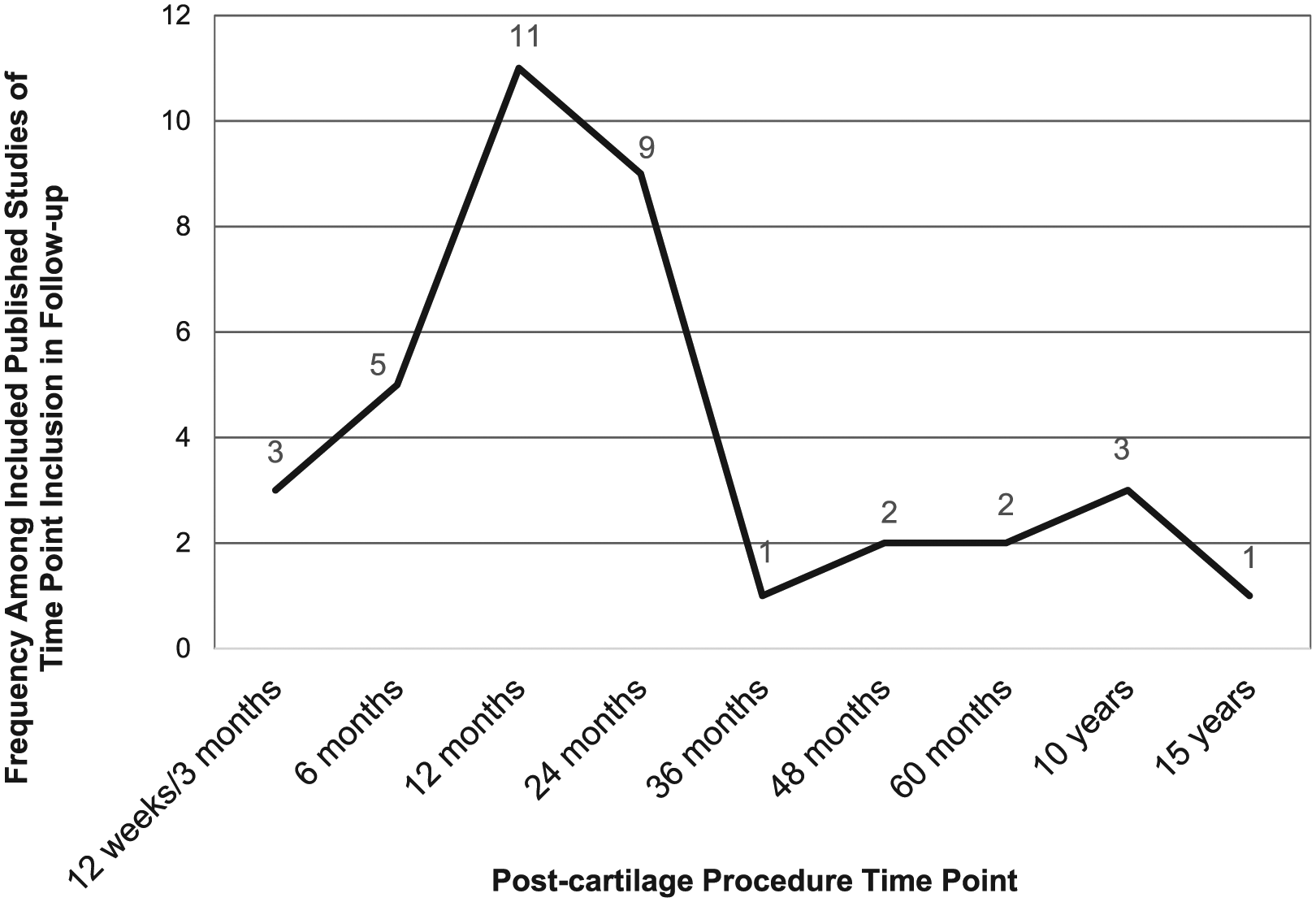
Frequency among included studies of time point inclusion in follow-up.
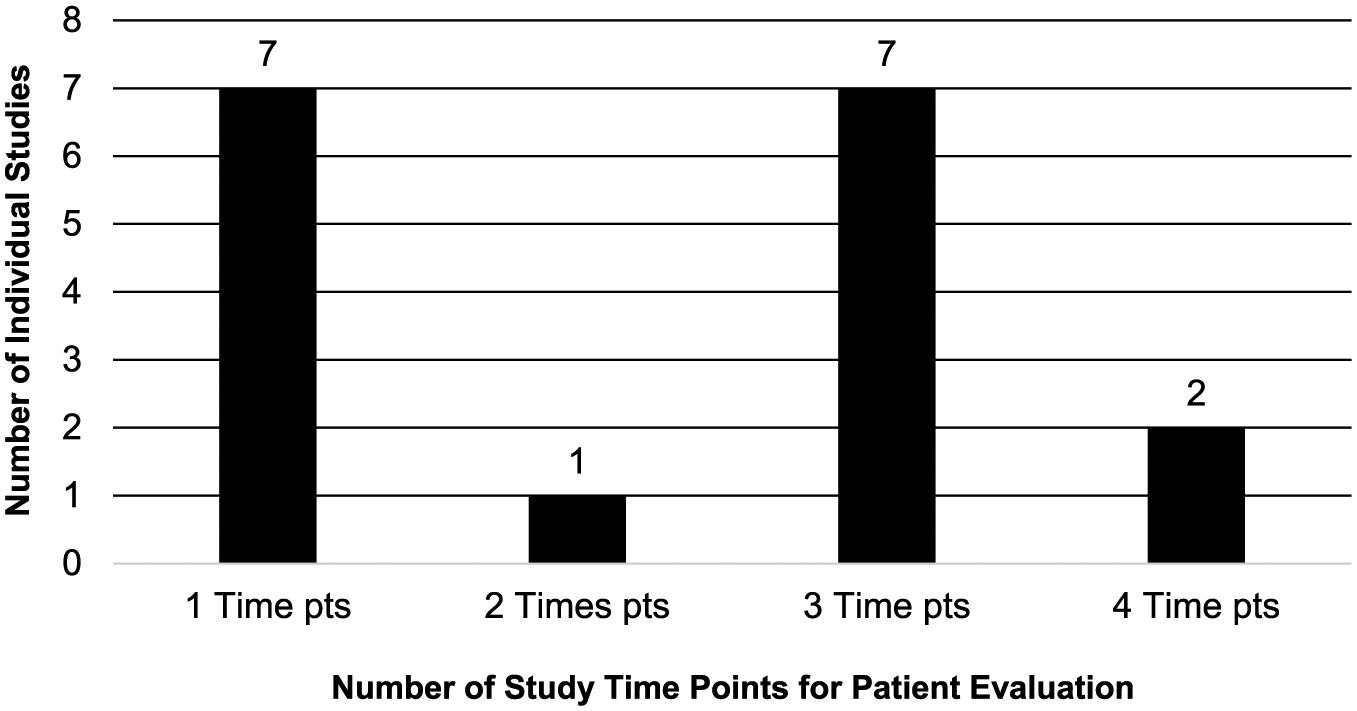
Number of time points for patient evaluation in each individual study.
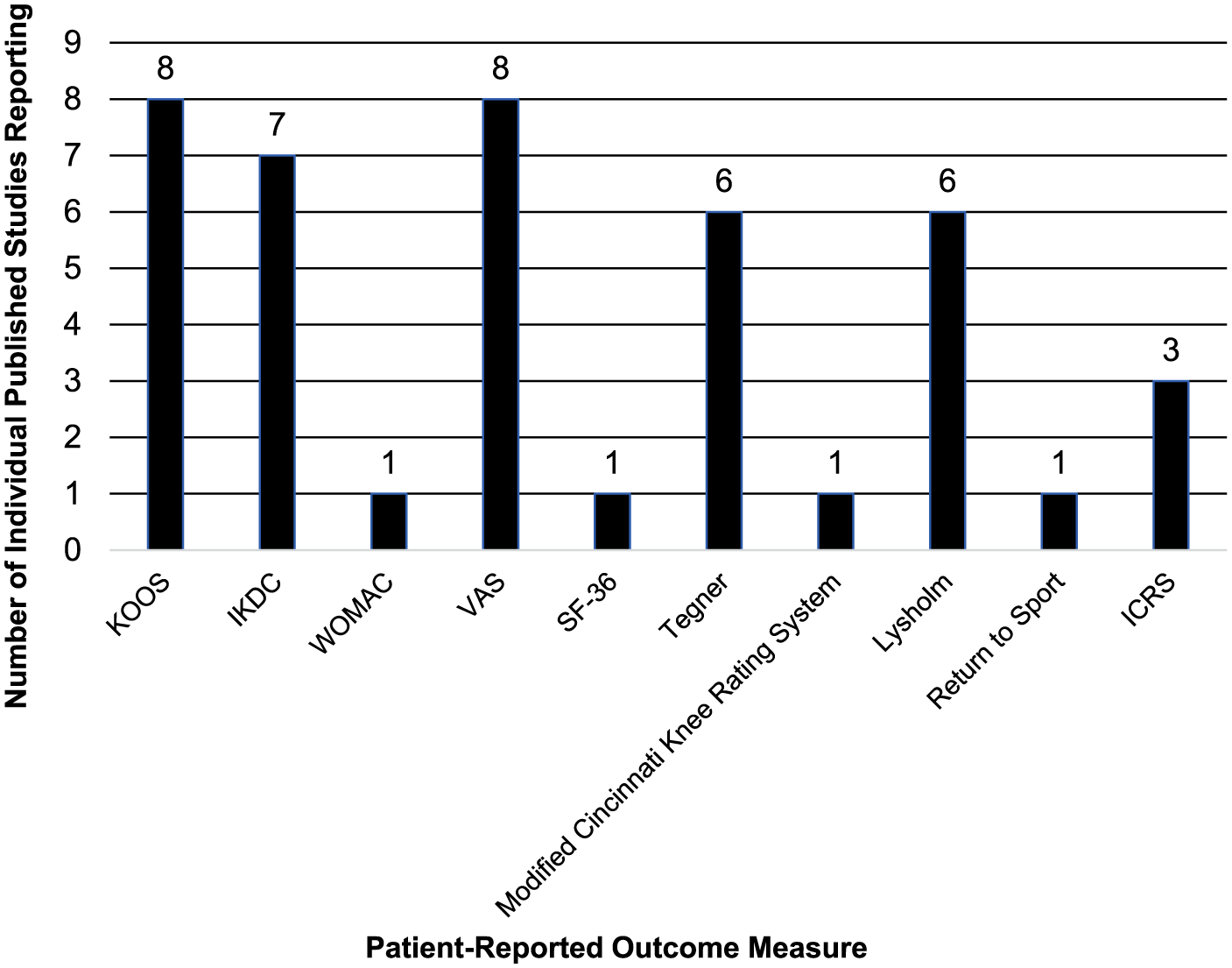
Patient-reported outcome meaures reported by each included study.
Regarding the 17 published studies, superiority with the cartilage procedure treatment group of interest versus the control/comparison group (
Table 3
) was found in the majority of included studies (N = 10; 58.8%).37-46 Several studies (N = 5; 29.4%, ACI vs MFX, MFX vs OAT, biomaterial scaffold + MFX vs MFX, MFX + biomembrane vs MFX, hydrogel implant vs OAT) demonstrated no difference (noninferiority) between the cartilage treatment of interest and the control/comparison group (
Table 4
).47-51 Few studies (N = 2; 11.8%) demonstrated inferiority of the treatment group to the control/comparison group (
Table 5
).52,53 In the published studies that demonstrated superiority of the treatment group, the most common time point for patient evaluation posttreatment was 12 months (N = 9; 90%), the most common primary outcome measure was KOOS (N = 4; 40.0%), and the most common control/comparison group was MFX alone (N = 6; 60.0%). In the published studies that demonstrated no difference (noninferiority) of the treatment group, the most common time point for patient evaluation after the procedure was 12 months (N = 2; 40.0%) and 24 months (N = 2; 40.0%), the most common outcome measures were IKDC and Lysholm (N = 3; 60.0%), and the most common control/comparison group was MFX alone (N = 3; 60.0%) (
Summary of Methodology of All Trials in Which the Treatment Group of Interest Was Shown to be Superior to Comparison or Control Groups. a

ACI = autologous chondrocyte implantation; MFX = microfracture; m-AMI = matrix-induced autologous mesenchymal stem cell implantation; ADSC = adipose-derived mesenchymal stem cells; MACI = matrix-assisted autologous chondrocyte implantation; OAT = osteochondral autograft transplantation; RF = radiofrequency; VAS = visual analog scale; KOOS = Knee Injury and Osteoarthritis Outcome Score; MRI = magnetic resonance imaging; IKDC = International Knee Documentation Committee score; ICRS = International Cartilage Repair Society.
Studies were deemed to have treatment of interest superior to comparison or control group if (1) there was an identified treatment of interest included and (2) the primary outcome demonstrated superiority of the treatment of interest. Given the significant heterogeneity of studies, numbers were used to demonstrate which outcome time points correlate with which outcome measures and comparison/control groups.
Summary of Methodology of All Trials in Which the Treatment Group of Interest Was Shown to be Noninferior to Comparison or Control Groups. a
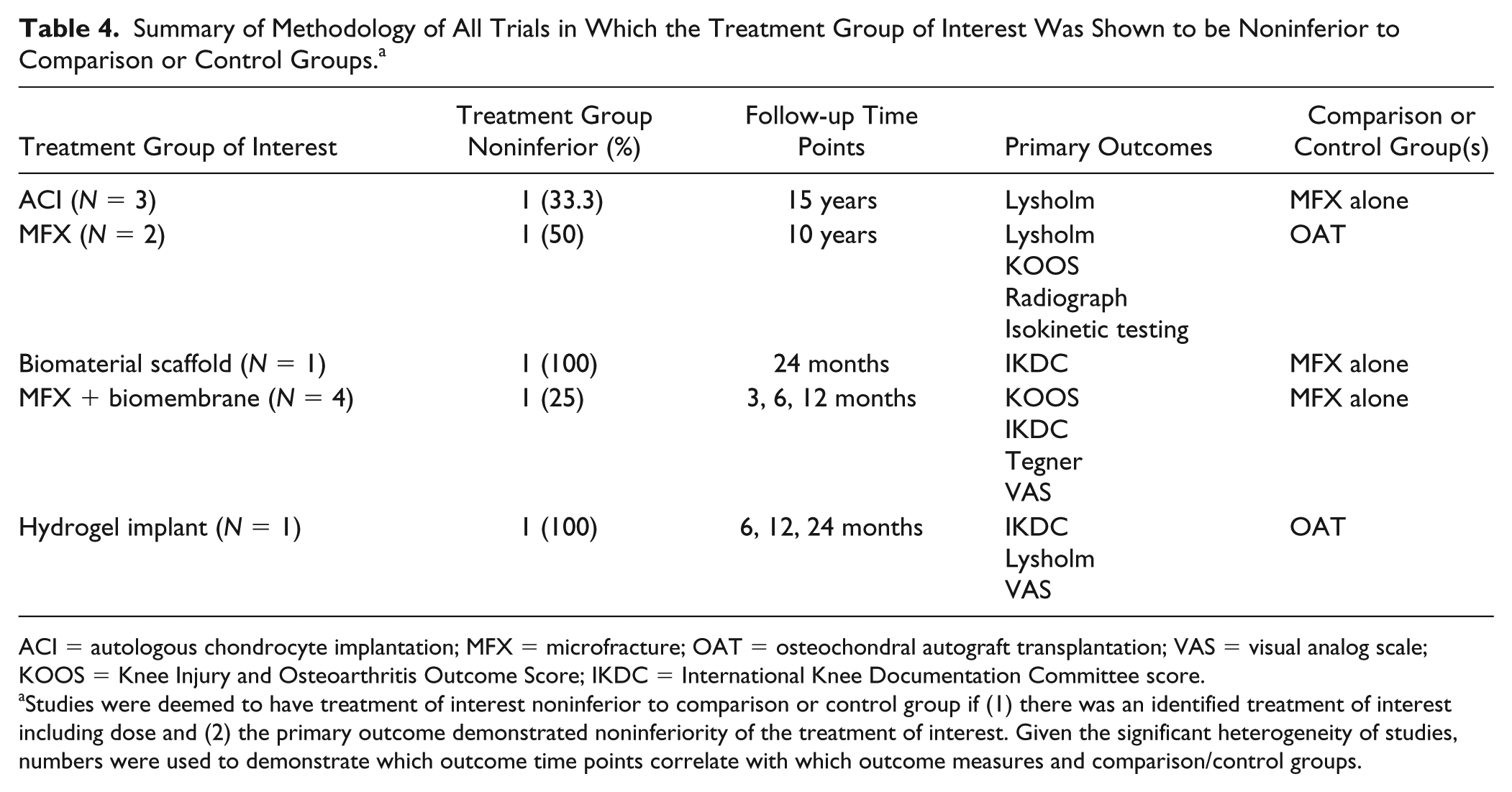
ACI = autologous chondrocyte implantation; MFX = microfracture; OAT = osteochondral autograft transplantation; VAS = visual analog scale; KOOS = Knee Injury and Osteoarthritis Outcome Score; IKDC = International Knee Documentation Committee score.
Studies were deemed to have treatment of interest noninferior to comparison or control group if (1) there was an identified treatment of interest including dose and (2) the primary outcome demonstrated noninferiority of the treatment of interest. Given the significant heterogeneity of studies, numbers were used to demonstrate which outcome time points correlate with which outcome measures and comparison/control groups.
Summary of Methodology of All Trials in Which the Treatment Group of Interest Was Shown to be Inferior to Comparison or Control Groups or There was No One Identified Treatment of Interest. a
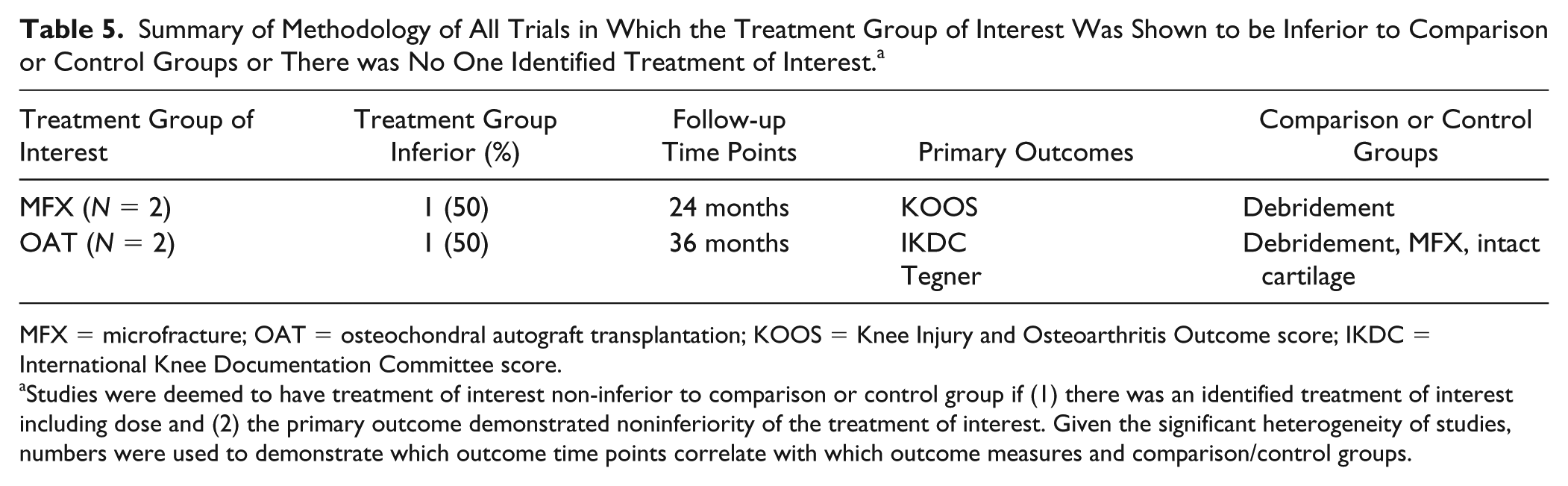
MFX = microfracture; OAT = osteochondral autograft transplantation; KOOS = Knee Injury and Osteoarthritis Outcome score; IKDC = International Knee Documentation Committee score.
Studies were deemed to have treatment of interest non-inferior to comparison or control group if (1) there was an identified treatment of interest including dose and (2) the primary outcome demonstrated noninferiority of the treatment of interest. Given the significant heterogeneity of studies, numbers were used to demonstrate which outcome time points correlate with which outcome measures and comparison/control groups.
All included published studies collected PROs similarly as one-time surveys at target time points. Of the 17 published studies, 15 studies (88.2%) used the primary or co-primary patient-reported outcome to investigate if the changes from baseline to final follow-up differ systematically between cohorts.37-45,48-53 Five (29.4%) published studies compared crude mean PROs between treatment groups at final follow-up.37,40,41,47,52 When using KOOS as a primary endpoint, 3 studies compared crude mean total KOOS (3 of 7; 42.9%),37,40,52 and 4 investigations employed delta KOOS from baseline to final follow-up (4 of 7; 57.1%).42,44,48,50 Two published studies reported mean total KOOS as well as the individual KOOS subscales (2 of 7; 28.6%).37,50 Four studies reported only individual mean KOOS subscales as the primary endpoints of choice (4 of 7; 57.1%).40,42,44,48 Finally, 1 study used mean total KOOS as the primary endpoint without providing a breakdown of the KOOS subscales (1 of 7; 14.3%). 52
Four of the published studies were designed to detect a clinically significant difference by using a previously described minimal clinically important difference (MCID) score (KOOS N = 2; IKDC N = 2).40,42,50,51 A change in KOOS score of 10 was used as the MCID in 2 studies. Two published studies were designed to detect the minimal detectable change (MDC) of the chosen primary outcome (IKDC N = 10; Lysholm N = 8.9).45,53
Unpublished Clinical Trials
Appendix Table A2 details the synthesized information from the clinical trials. Most trials (N = 16 clinical trials; 30.8%) of the trials are completed but not yet published, and 14 clinical trials (26.9%) are actively recruiting participants. The greatest number of trials (N = 17; 32.7%) are performed in the United States, with 29 countries accounted for in total, and currently are in phase III of study (N = 16 clinical trials; 30.8%). The most common cartilage repair/restoration treatment being studied is MFX + augmentation (N = 16; 30.8%), followed by ACI (N = 9; 17.3%) and MACT/MACI (N = 8; 15.4%). In total, 31 different cartilage repair/restoration procedures types/groups are investigated in the 52 clinical trials. The most common control/comparison group utilized is MFX (N = 33; 63.5%).
The most common age ranges for participant inclusion are 18 to 50 years (N = 8; 15.4%) and 18 to 55 years (N = 8; 15.4%). Most trials (N = 27; 51.9%) did not specify Kellgren-Lawrence grade as an inclusion criterion ( Appendix Table A2 ). The most common time points for patient evaluation postinjection are 12 months (N = 31; 59.6%) and 24 months (N = 26; 50.0%) and range from 10 days (N = 1; 1.9%) to 5 years (N = 9; 17.3%) with 15 total time points used ( Fig. 6 ). Most trials have 1 time point postprocedure for patient evaluation (N = 20; 38.5%), and trials range from 1 to 8 time points (N = 3; 5.8%) of postprocedure evaluation ( Fig. 7 ). The most common PROs assessed in the included clinical trials is KOOS (N = 36 trials; 69.2%), IKDC (N = 28; 53.8%), and VAS pain (N = 21; 40.4%) with KOOS (N = 19; 36.5%) and IKDC (N = 14; 26.9%) being the most common primary endpoints. All included trials collect PROs as one-time surveys at target time points. Of the unpublished trial, 19 of 52 trials use PROs to investigate the change in outcomes from baseline. Eight of 52 unpublished trials compare crude PRO means at final follow-up. Three trials are designed to evaluate percent change from baseline. Only 1 trial aims to find a clinically relevant responder rate. Twenty-one unpublished trials do not specify how the PROs are being used. Overall there are 24 different PROs used among the included studies ( Fig. 8 ). Postprocedure imaging is performed in 39 trials (75.0%). Thirty-eight of the 39 trials obtain postprocedure MRI, most commonly, in evaluation of MOCART score (N = 16; 30.8%) ( Appendix Table A2) . Notably, there are 5 clinical trials (9.6%) that include arthroscopic evaluation in the outcome reporting ( Appendix Table A2 ).
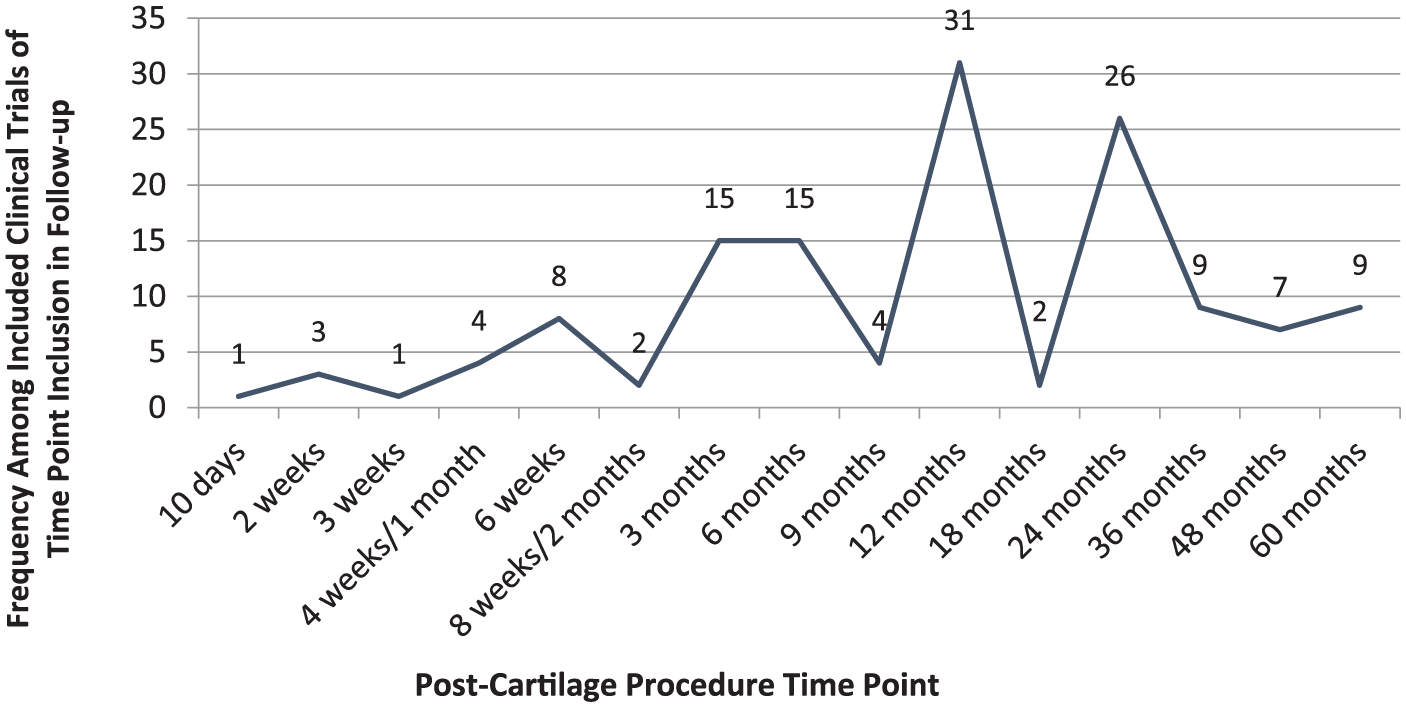
Frequency among included clinical trials of time point inclusion in follow-up.
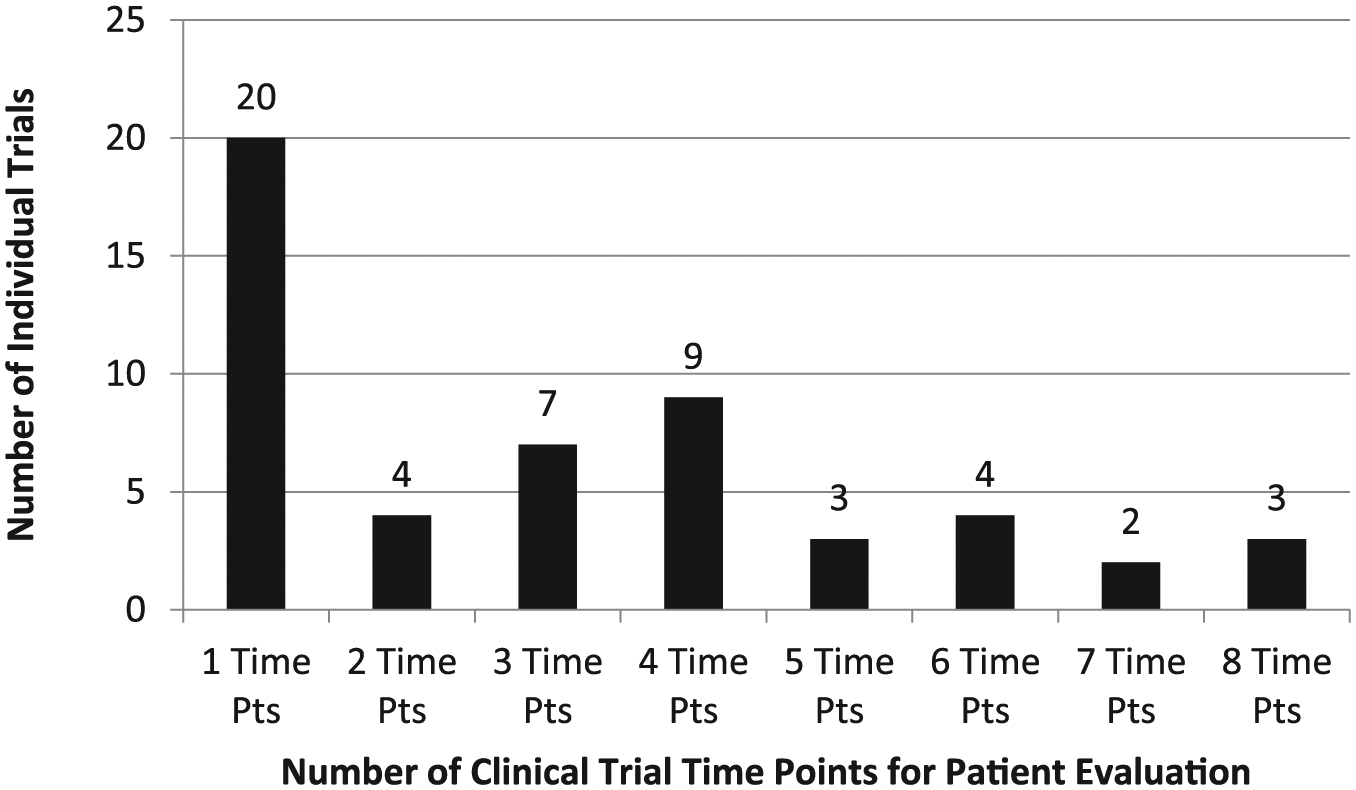
Number of time points for patient evaluation in each individual clinical trial. Pts = points.
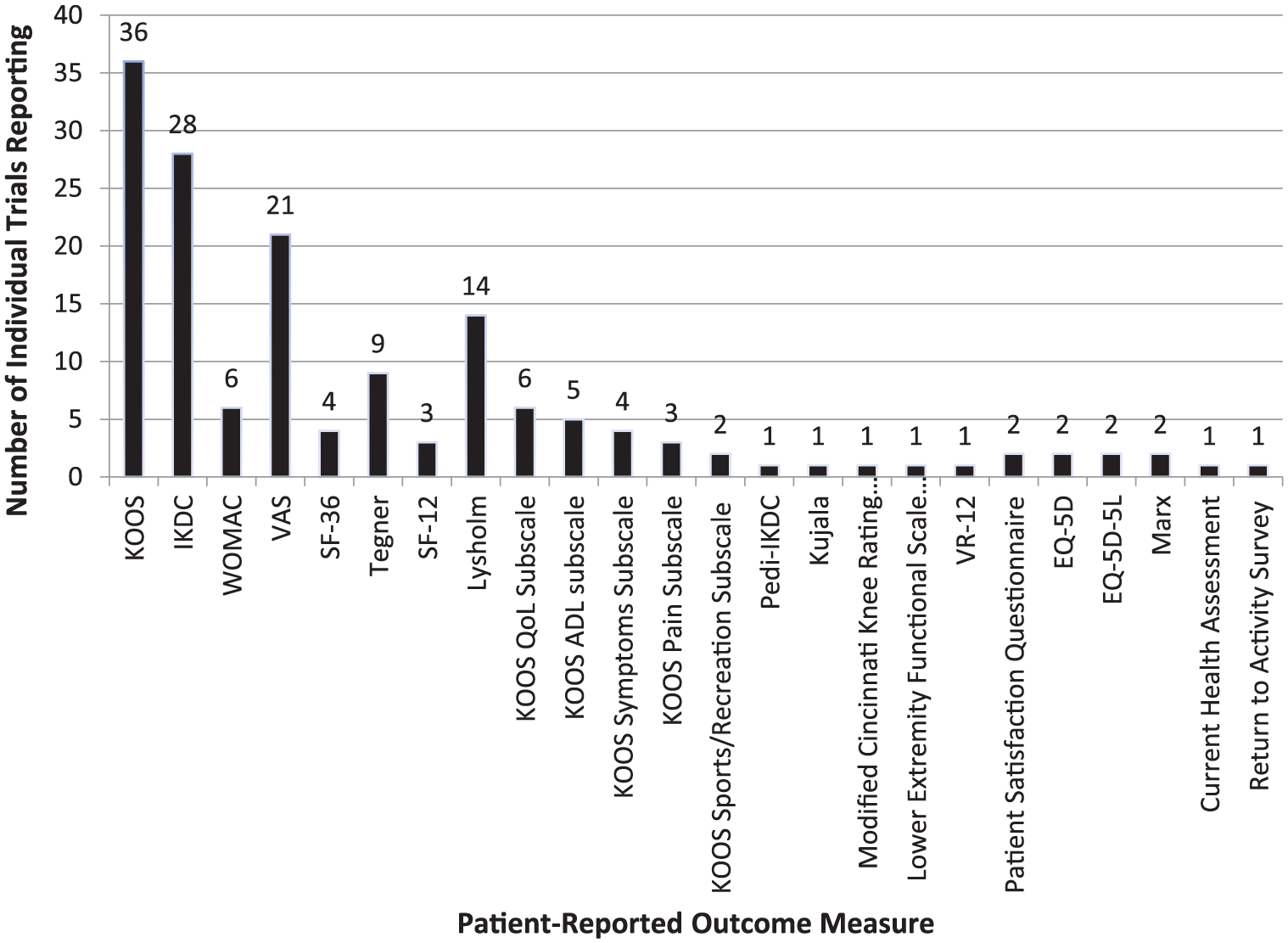
Patient-reported outcome measures reported by each included clinical trial.
Discussion
The findings from our study of only high level (level I or II evidence and ongoing clinical trials) demonstrate that the majority (N = 10; 58.8%) of published studies reported superiority with the cartilage procedure treatment group of interest versus the control/comparison group. In these studies, the most common time point for patient evaluation posttreatment was 12 months (N = 9; 90%), the most common primary outcome measure was KOOS (N = 4; 40.0%), and the most common control/comparison group was MFX alone (N = 6; 60.0%). Overall, there was significant variation in methodology regarding the primary outcomes of interest, secondary outcomes, use of imaging postoperatively, control groups used for comparison, and time points of outcomes. These data should serve to demonstrate the significant heterogeneity in design of high-level studies evaluating cartilage procedures in the knee and inform future investigators on study design.
Current clinical trials, however, are most commonly occurring in the United States and are evaluating MFX + augmentation strategies, but 31 different cartilage repair/restoration procedures are being evaluated in the 52 clinical trials. KOOS, again, is most commonly used in follow-up outcome scoring, but 24 different outcome measures are used among the included trials.
The search for the successful, reproducible cartilage repair/restoration procedures continues to grow, as is evident with the 13 different cartilage procedures being evaluated in the 17 recently published studies, and the 31 different procedures being evaluated in the 52 current clinical trials. It is clear that no intervention has emerged as superior to the others as a single treatment, and in part this is certainly due to the varying patient demographic and disease-specific indications for each of the different procedures available. MFX remains the most common comparison group by which the investigative treatments are still measured in both the published and current clinical trials, likely because of its long track record over several decades, significant body of literature, ease of performance, and low cost.54,55
This systematic review of the published studies and clinical trials is also helpful in demonstrating the patient demographic of interest for researchers looking to find benefit from cartilage repair/restoration efforts in the knee. Young to middle-aged adults, between 18 and 50 years of age, with normal or near-normal range BMI were the most common patient populations in these studies. This finding reflects how the results of these studies are not generalizable to the more challenging population of older patients with moderate to advanced osteoarthritis.
With 10 and 24 different PROs used in the 17 published studies and 52 clinical trials, respectively, it is clear that standardization of outcome measures is lacking among knee cartilage repair/restoration procedure studies. Even with the KOOS score being the most common PRO, it was used in less than 50% of published studies, and only utilized as the primary endpoint in 41.2% and 36.5% of the published studies and clinical trials, respectively. The use of KOOS as a primary endpoint may further add heterogeneity due to the various methods of reporting the outcome and the corresponding subscales. Similarly, wide variations in the frequency and patient follow-up evaluations were identified, with few clinical trials and even fewer published studies evaluating patients more than 2 years after surgery, leading to a paucity of data that includes long-term durability assessment. The difficulty in maintaining high patient follow-up (>70%) in these prospective level I and II studies beyond 2 years must be acknowledged; however, the importance of achieving this cannot be understated and is often what is required should their role in disease modification remain of interest. Moreover, a vast minority of published studies and clinical trials included histologic (18.8% of published studies; 25.0% of clinical trials) or second-look arthroscopic (5.6% of published studies; 9.6% of clinical trials) analysis of cartilage repair/restoration tissue as a datapoint. Given that these procedures are meant to improve on the biologic integrity of the articular cartilage, we propose that an actual evaluation of the chondral surface remains of interest to fully understand the biomechanical function of these treatment modalities. Overall, it would be prudent with further investigative research efforts to regulate and homogenize the timing of patient outcome assessment and means by which outcomes are measured and reported.
As the “pay for performance” model of health care trends toward replacing the “fee-for-service,” accurate investigative clinical research of current and emergent techniques will be even more critical in the near future. 56 Despite the growing emphasis in clinically significant rather than statistically significant outcomes, only 6 of the 17 published trials used a threshold, such as MCID or MDC, to describe clinical impact. Additionally, which thresholds authors used was variable, as MCID (KOOS N = 2; IKDC N = 2) and MDC (Lysholm N = 1; IKDC N = 1) were implemented in 4 and 2 studies, respectively. This represents a wide variation in how current thought leaders employ PROs to establish treatment superiority. For the 2 most common primary endpoints found, MCID values of 11.5 for IKDC and 8 to 10 for KOOS have been suggested in association with general “knee injury.”57-59 While these thresholds are helpful, clinically significant outcomes such as MCID are both diagnoses and intervention specific. In the future, investigations to determine more specific thresholds of clinical importance may be helpful in limiting variation and heterogeneity when reporting outcomes.
Finally, it is interesting to note that the majority of published studies (58.8%) demonstrated superiority with the cartilage procedure treatment group of interest. For these studies demonstrating superiority with the treatment group, the most common time point for patient evaluation was 12 months using KOOS outcome measures, as compared with 1 and 2 years using IKDC, respectively, for those studies (29.4%) demonstrating no difference (noninferiority). Only 2 studies (11.8%) demonstrated inferiority of the treatment group to the control/comparison group. Although speculative, this might suggest that superiority is more visible in the shorter term, and that the additional categories of “Pain” and “Quality of Life” that are evaluated in more detail with the KOOS outcome measure (as compared with the IKDC) are domains where these cartilage treatment options have the potential for significant benefit.
Overall, there was significant variation in methodology and design of high level studies aimed at evaluating cartilage restoration/repair techniques, making it difficult to inform future investigators on study design. The authors suggest more consistency regarding the primary outcomes of interest, secondary outcomes, and time points of outcomes will add greatly in establishing successful, reproducible articular cartilage procedures and treatment algorithms. A potential method for reducing the current heterogeneity may be more widespread implementation of clinically significant thresholds to power or analyze outcomes. These thresholds are specific to treatments and respective outcome measures, and more investigations using these thresholds may help standardize “significant improvement” and treatment impact. Additionally, long-term clinical evaluation is recommended to assess durability, especially due to the younger patient population cartilage restoration/repair procedures target. Finally, while no PRO evidently predominates in the area of research discussed in this review, the authors recommend that future investigations focus on the ability of specific PROs and PRO subscales, such as KOOS “Pain” and “Quality of Life,” to increase the accuracy and replicability of cartilage treatment assessments.
Limitations
There are inherent limitations in this study. First, while our application of a 5-year span for search strategy was used with the intent of reporting the most recent research efforts of this topic, it does limit a review of trends in knee cartilage repair/restoration treatments from prior to the included study dates. Since our study was a comprehensive review and summary of heterogeneous data, we were unable to provide any specific statistical analyses beyond the presentation of data through means and medians.
Conclusion
Recently published studies and clinical trials continue to evaluate a variety of different cartilage repair and restoration strategies for the knee—most commonly ACI in recent published studies, and MFX + augmentation in ongoing or current clinical trials—at various time points of outcome evaluation and using a variety of PRO measures, with the KOOS score being used most commonly as a primary endpoint. MFX remains the most common comparison group for these therapeutic studies/trials. Most demonstrate superiority versus comparison or control groups, and KOOS was the most common outcome scale used in studies demonstrating superiority to comparison/control groups. Understanding the nuances of published and ongoing clinical trials will be helpful to the investigation of emerging technologies required to navigate the regulatory process while studying a relatively narrow population of patients that are otherwise historically difficult to enroll.
Footnotes
Appendix
Information Regarding the Clinical Trials.

| Data | Result |
|---|---|
| Total number of studies (N) | 52 |
| Study status, n (%) | |
| Completed | 16 (30.8) |
| Recruiting | 14 (26.9) |
| Not yet recruiting | 3 (5.8) |
| Unknown status | 6 (11.5) |
| Active, not recruiting | 6 (11.5) |
| Terminated | 6 (11.5) |
| Withdrawn | 1 (1.9) |
| Country of study a , n (%) | |
| United Kingdom | 8 (15.4) |
| United States | 17 (32.7) |
| Canada | 7 (13.5) |
| Belgium | 5 (9.6) |
| Croatia | 2 (3.8) |
| Netherlands | 5 (9.6) |
| Germany | 9 (17.3) |
| Korea | 6 (11.5) |
| Austria | 4 (7.7) |
| Hungary | 1 (1.9) |
| Italy | 3 (5.8) |
| Switzerland | 4 (7.7) |
| Czech Republic | 2 (3.8) |
| France | 4 (7.7) |
| Poland | 4 (7.7) |
| China | 1 (1.9) |
| Israel | 1 (1.9) |
| Denmark | 2 (3.8) |
| Ireland | 1 (1.9) |
| Norway | 6 (11.5) |
| Sweden | 3 (5.8) |
| Mexico | 1 (1.9) |
| Spain | 3 (5.8) |
| Australia | 1 (1.9) |
| South Africa | 1 (1.9) |
| Finland | 1 (1.9) |
| Brazil | 1 (1.9) |
| Singapore | 1 (1.9) |
| Taiwan | 1 (1.9) |
| Study phase, n (%) | |
| Phase II | 5 (9.6) |
| Phase III | 16 (30.8) |
| Phase IV | 7 (13.5) |
| Phase II-III | 1 (1.9) |
| NR | 23 (44.2) |
| Treatment group a , n (%) | |
| Microfracture | 4 (7.7) |
| Microfracture + Cartifill | 2 (3.8) |
| Microfracture + ChonDux | 1 (1.9) |
| Microfracture + HTO | 1 (1.9) |
| Microfracture + Biocartilage | 1 (1.9) |
| Microfracture + Cartistem | 2 (3.8) |
| Microfracture + Chondrosphere | 1 (1.9) |
| Microfracture + Chondro-Gide | 1 (1.9) |
| Microfracture + BST-CarGel | 3 (5.8) |
| Microfracture + AMIC | 1 (1.9) |
| Microfracture + MaioRegen | 1 (1.9) |
| Microfracture + ADSC | 1 (1.9) |
| Microfracture + BiCri | 1 (1.9) |
| Debridement | 3 (5.8) |
| ACI | 9 (17.3) |
| Osteochondral allograft | 1 (1.9) |
| BMAC + cartilage procedure | 4 (7.7) |
| DBX Demineralized Bone Matrix Putty | 1 (1.9) |
| Autologous adipose cell therapy | 1 (1.9) |
| Autologous collagen-induced chondrogenesis | 1 (1.9) |
| Retro articular drilling for OCD | 1 (1.9) |
| Cartilage Autograft Implantation System | 1 (1.9) |
| Ultrasound therapy | 1 (1.9) |
| TissueGene-C injection | 1 (1.9) |
| TruFit catilage bone transplant | 1 (1.9) |
| HTO with Calcibon | 1 (1.9) |
| Pulsed electromagnetic therapy | 1 (1.9) |
| Synovium brushing | 1 (1.9) |
| Debridement with adipose-derived stem cells | 1 (1.9) |
| CR-Plug allograft | 1 (1.9) |
| MACT/MACI | 8 (15.4) |
| Control group, n (%) | |
| No treatment | 3 (5.8) |
| Microfracture | 34 (65.4) |
| Debridement | 3 (5.8) |
| ACI | 1 (1.9) |
| Osteochondral autograft | 3 (5.8) |
| Transarticular drilling for OCD | 1 (1.9) |
| Saline injection | 1 (1.9) |
| HTO | 1 (1.9) |
| Included age range in years, n (%) | |
| 8-18 | 1 (1.9) |
| <55 | 1 (1.9) |
| 14-50 | 1 (1.9) |
| 15-40 | 1 (1.9) |
| 15-65 | 1 (1.9) |
| 15+ | 1 (1.9) |
| 16-55 | 1 (1.9) |
| 16-60 | 2 (3.8) |
| 18-45 | 1 (1.9) |
| 18-49 | 1 (1.9) |
| 18-50 | 8 (15.4) |
| 18-55 | 8 (15.4) |
| 18-59 | 1 (1.9) |
| 18-60 | 5 (9.6) |
| 18-65 | 7 (13.5) |
| 18-68 | 1 (1.9) |
| 18-70 | 1 (1.9) |
| 18+ | 5 (9.6) |
| 19-65 | 1 (1.9) |
| 20-50 | 1 (1.9) |
| 21-50 | 1 (1.9) |
| 30+ | 1 (1.9) |
| 45+ | 1 (1.9) |
| Included Kellgren-Lawrence grade, n (%) | |
| 0 | 12 (23.1) |
| 0-I | 3 (5.8) |
| 0-II | 7 (13.5) |
| 1-2 | 2 (3.8) |
| 3 | 1 (1.9) |
| NR | 27 (51.9) |
| Additional outcome variables a , n (%) | |
| Adverse events | 9 (17.3) |
| Imaging | 39 (75.0) |
| MRI: Unspecified measure | 9 (17.3) |
| MRI: MOCART | 16 (30.8) |
| MRI: WORMS | 1 (1.9) |
| MRI: Degree/volume of filling | 5 (9.6) |
| MRI: ICRS | 1 (1.9) |
| MRI: T2 relaxation time | 1 (1.9) |
| MRI: dGEMRIC graft remodeling | 3 (5.8) |
| MRI: T1 rho glycosaminoglycan content | 1 (1.9) |
| MRI: T2 loss of type II collagen | 1 (1.9) |
| MRI: T2 collagen fiber orientation | 2 (3.8) |
| MRI: T2 Hydration of cartilage | 1 (1.9) |
| MRI: Cartilage lesion score | 1 (1.9) |
| MRI: Graft incorporation | 1 (1.9) |
| MRI: MOCART II | 1 (1.9) |
| Radiograph: Kellgren-Lawrence grade | 1 (1.9) |
| Radiograph: Unspecified measure | 1 (1.9) |
| Histology: Unspecified measure | 3 (17.3) |
| Histology: ICRS II | 2 (3.8) |
| Histology: Histomorphometry safrarin-O + anti-collagen II antibody staining | 1 (1.9) |
| Histology: ICRS | 3 (5.8) |
| Histology: Grade of cartilage regeneration | 1 (1.9) |
| Histology: Bern score | 1 (1.9) |
| Histology: Osseous integration evaluation | 1 (1.9) |
| Histology: Mean change in MSCs present | 1 (1.9) |
| Arthroscopy | 5 (9.6) |
| ICRS grade decline at week 48 | 1 (1.9) |
| Safety and efficacy | 1 (1.9) |
| Change in pain/function | 4 (7.7) |
| Radiostereometric analysis | 1 (1.9) |
| Lower extremity functional scale | 1 (1.9) |
| Number of injection site reactions | 1 (1.9) |
| 6-minute walk test | 1 (1.9) |
| Pain at end of 6-minute walk test | 1 (1.9) |
| Semiquantitative scoring of knee joint | 1 (1.9) |
| Treatment failure rate | 1 (1.9) |
| Change in VAS | 1 (1.9) |
| Cut-to-suture time | 1 (1.9) |
| Incision length | 1 (1.9) |
| Time to failure | 2 (3.8) |
| ICRS | 3 (5.8) |
| Return to sport | 1 (1.9) |
| Return to work | 1 (1.9) |
| Physical examination (vital signs, blood tests) | 1 (1.9) |
| Effusion/tenderness | 3 (5.8) |
| Range of motion | 3 (5.8) |
| Hop test | 2 (3.8) |
| Clinical evaluation | 2 (3.8) |
| Economic evaluation | 2 (3.8) |
| Localized mechanical symptoms | 1 (1.9) |
| Quality of life | 1 (1.9%) |
| Hip-knee-ankle axis | 1 (1.9) |
| Time to healing | 1 (1.9) |
NR = not reported; RCT = randomized controlled trial; ACI = autologous chondrocyte implantation; HTO = high tibial osteotomy; OCD = osteochondritis dissecans; MSCs = mesenchymal stem cells; MRI = magnetic resonance imaging; dGEMRIC = delayed gadolinium-enhanced MRI of cartilage; WORMS = whole-organ magnetic resonance imaging score; MOCART = magnetic resonance observation of cartilage repair tissue; ICRS = International Cartilage Repair Society; VAS, visual analog scale; AMIC = autologous matrix-induced chondrogenesis.
Signifies that the sum total of the category results may be >52 (total number of studies) as more than 1 variable in the category may be true for an individual study (i.e., in the case of “Country of Study,” some studies were performed across >1 country).
Author’s Note
Rachel M.Frank is now affiliated with University of Corolado School of Medicine, Department of Orthopaedic Surgery, Aurora, CO, USA.
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
