Abstract
The objective of this study was to analyze morphometric anatomy and damages with aging in cadaveric knee specimens specific to the cruciate ligaments, the articular cartilage of the tibial plateau, and the menisci. Morphometric analyses to cadaveric anatomy of the knee were performed using Image-Pro® software on 3 age populations: <70 years old, 70 to 79 years old, and ≥80 years old. An average thickness of the cruciate ligaments was assessed with 5 circumferential measurements per specimen using nylon thread. Percent degeneration of the tibial plateau’s articular cartilage and coverage by menisci was assessed with surface area measurements. The articular cartilage of the medial tibial plateau in ≥80 years old specimens showed a 1.7-fold increase in surface area degeneration (mm2) compared to 70 to 79 years old specimens (P < 0.05). The medial meniscus also experienced degenerative changes with aging, which were expressed as decreases in tibial plateau coverage. The anterior cruciate ligament (ACL) experienced substantial degenerative thinning with aging. The 70 to 79 years old specimens had a 1.2-fold (10.5%) decrease in average ACL circumference (mm) compared to the <70 years old specimens (P < 0.001). The ≥80 years old specimens had a 1.24-fold (19%) decrease in ACL circumference compared to the <70 years old specimens (P < 0.001). ACL thinning during aging may be leading to substantial articular cartilage and menisci degeneration given the cruciate ligaments are a primary restraint that combats shearing forces at the knee joint.
Introduction
Osteoarthritis (OA) is the most common form of joint disease that damages the joint due to abnormal inflammatory signaling and joint tissue remodeling. The articular cartilage, subchondral bone, intracapsular ligaments, menisci, synovium, and joint capsule of the knee joint may all be affected by this disease process, making OA an organ-based disease. 1 The objective of this study was to analyze morphometric anatomy and damages with aging in cadaveric knee specimens specific to the cruciate ligaments, the articular cartilage of the tibial plateau, and the menisci. The etiology of OA is multifactorial; therefore, classifications of OA have been generated to better quantify prevalence of cases where etiology of disease is idiopathic or secondary (i.e., posttraumatic, congenital, developmental, or secondary to other diseases such as calcium deposition disease, endocrine diseases, gout, avascular necrosis, etc.). Simple variation in developed biomechanics may give rise to secondary, localized OA due to obesity, valgus or varus deformities, unequal lower extremity length, scoliosis or hyper- and hypomobility syndromes. 2
The Centers for Disease Control and Prevention reported a 2008 estimate on the prevalence of OA in the United States in 2005. The study states that OA affected 13.9% of Americans 25 years and older, whereas 33.6% of Americans 65 years and older were affected. In 1990, there was estimated to be 21 million 65+ individuals with OA, and by 2005 the estimate grew to 26.9 million, an approximately 22% increase over 15 years. These estimates are believed to be conservative, but if the trend continues in 2020, there will potentially be 32.8 million Americans with OA. 3 The knee joint is one of the most common sites of OA and these knee joint pathologies greatly contribute to the OA-related hospitalizations and deaths.4,5 The female population has been shown to have greater prevalence of OA compared with the male population.5-7 In the United Kingdom, a 14-year longitudinal study on 561 women determined that 47.8% of women 60 years or older had radiographic signs and symptoms of OA. 8 Several of the specimens included in this study specifically state in their clinical history their likely cause of OA symptoms, whether it be idiopathic and directly stated as OA or secondary and stated as a musculoskeletal pathology. The majority of specimens do not indicate a cause of OA in their clinical history, thus presenting a limitation of this study. The study assumes all damages are due to OA given the prevalence of the disease although other previously mentioned pathologies may cause similar degenerative alterations.
Several studies have highlighted the fact that aging relates to the degenerative changes seen in the articular cartilage and the anterior cruciate ligament (ACL).9-16 Little to no cell division or cell death occurs in adult chondrocyte populations; therefore, they can accumulate age-related changes or damages with time.9-12,16 Collagen fiber disorganization, chondroid metaplasias, cystic occurrences, and calcium phosphate crystal infiltration has been shown to increase in the ACL with aging.13,14 Common signs of OA related to chondral lesions are the presence of linear cracks, stellate fractures, articular cartilage erosion, fibrillations, and gross degradation on the articular surfaces. 15
The articular cartilage and the ACL have been proven to change with age; therefore, it is hypothesized that these structures will display the most prominent pathology in the examined population. In this population, all of the specimens (ages of 58 through 94) displayed some extent of articular cartilage degeneration. This excludes a single, male specimen of 26 years old. Given the population findings and the prevalence of OA in the United States, it is hypothesized that OA is occurring in varying degrees of severity in all patients 60 years and older. The cruciate ligaments are a primary restraint that combats shearing forces at the knee joint; therefore, it is hypothesized that with gross thinning of the ACL the articular cartilage will display degeneration with aging. Typical tibiofemoral compression forces are biomechanically highest on the medial tibial plateau; therefore, thinning of the ACL will especially increase degeneration of the articular cartilage on the medial respect of the knee joint. 17
Materials and Methods
Cadavers and Dissection
Twenty-one cadavers, 12 males and 9 females, were utilized for this study. Age and sex for each cadaver are provided in Table 1 . Specimens were divided into 3 age populations: <70 years old (n = 6), 70 to 79 years old (n = 9), and ≥80 years old (n = 6). Knees were approximately 50% right knees and 50% left knees. Knee joints were disarticulated from the cadavers with transfemoral and transtibiofibular cuts after the skin and subcutaneous tissue was dissected away from the deeper structures. Once disarticulated, the quadriceps tendon was transversely cut and the quadriceps were bluntly dissected off of the distal femur. The quadriceps tendon was reflected inferiorly with the patella, and the anterior joint capsule was cut away from its periosteal attachment superior to the articular cartilage lining the anterior femoral condyles and the patellar fossa. The quadriceps tendon was reflected inferiorly to the tibial tuberosity to expose the intracapsular joint space. Posteriorly, the joint capsule was dissected away from the posterior respects of the femoral condyles, menisci, posterior meniscofemoral ligament, and posterior cruciate ligament. This project (Protocol 150430-04) was reviewed by Rocky Mountain University of Health Profession’s Institutional Review Board in Provo, UT, and was determined to be research not involving human subjects.
Table lists all of the cadavers by identification number contributing knee specimens to this study. They are listed in ascending age. Sex is indicated in the central column.
Analysis of the Cruciate Ligaments and the Posterior Meniscofemoral Ligament
Once the joint space was opened, the medial and lateral collateral ligaments were bisected. This left the femur and the tibia attached solely by the intracapsular ligaments. The presence of the posterior meniscofemoral ligament was documented, and prevalence for the population was calculated. The posterior and anterior meniscofemoral ligaments were reflected away from the intercondylar fossa leaving the femur and tibia attached by only the anterior and posterior cruciate ligaments. Black nylon thread was used to measure the average circumference of the anterior and posterior cruciate ligaments. Five measurements were taken at approximately equal intervals between the intercondylar fossa and the intercondylar eminence with the joint in full extension. All measurements on a specimen were taken by a single, blinded investigator. The nylon thread was wrapped around the cruciate ligament tautly and then the intersection of the 2 ends of thread was bisected with a scalpel. After bisection, nylon threads were laminated between a folded adhesive strip and photographed with a calibration ruler. A 12.1 mega pixel Sony® digital camera was utilized for photography. These photographs were all imported into Image-Pro® software, and each image was manually calibrated to millimeters using the calibration ruler. Linear measurements were achieved by tracing each nylon thread.
Analysis of Menisci and Tibial Plateau Articular Cartilage
Once the cruciate ligaments were measured, they were bisected and pruned toward their attachments to avoid interference with photography and measurement of the tibial plateau and menisci. For each specimen, a calibrated photograph was taken of the superior respects of the tibial plateau with the menisci intact. Once imported to Image-Pro® software, the medial and lateral menisci were traced independently to achieve a surface area measurement (mm2). Traces included the most medial attachment sites of the menisci to allow for the most accurate measurements of menisci surface area loss. Once photographed, the menisci were dissected away from the tibia with the transverse meniscal ligament and their synovial and capsular attachments. The joint capsule, collateral ligaments, and the intracapsular tendon of the popliteus muscle were reflected farther inferiorly to avoid interference with photography and measurement of the tibial plateau. A calibrated photograph of the tibial plateau was then taken to acquire a surface area measurement.
Analysis of the Degeneration of the Tibial Articular Cartilage
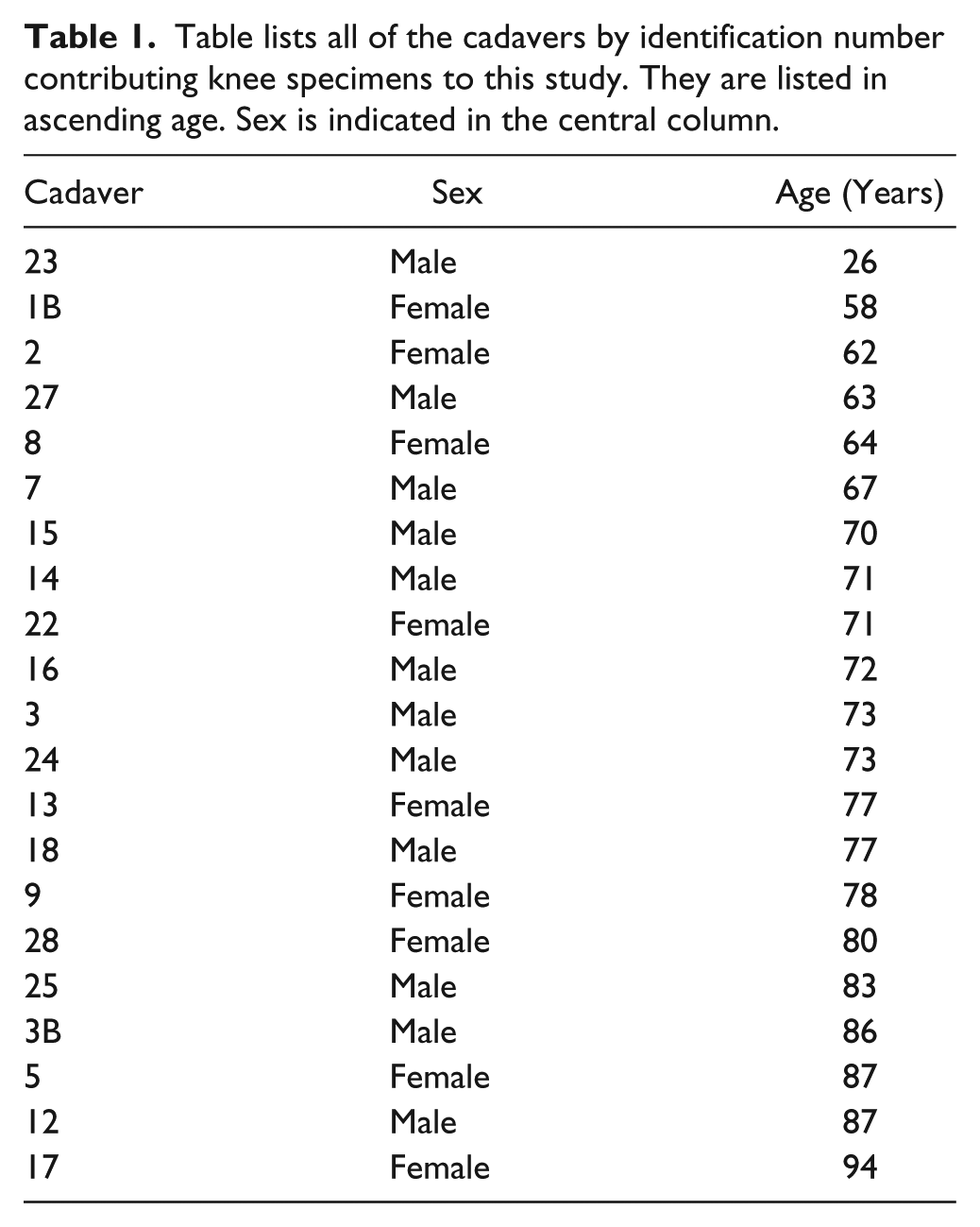
The photography of each specimen’s tibial plateau gave rise to the total surface area of the medial tibial plateau and the lateral tibial plateau independently. Using Image-Pro® software, the 2 regions of the tibial plateau were traced. Tracing occurred at the most peripheral margins of the medial, lateral, and posterior plateau. Tracing did not include the intercondylar eminence or the superior respects of the tibial tuberosity. Once the overall surface area was collected, the sites of degeneration on the medial and lateral tibial plateau were traced within their respective total surface area traces ( Fig. 1B ). Visible craters, fibrillated areas, large stellate cracks, and losses of articular cartilage to the point of cortical bone exposure were included as traceable signs of degeneration. Each region of degeneration was traced independently, and surface areas of all of the sites of degeneration were summed. This summed quantity of degeneration was calculated as a percentage of degeneration for each side of the tibial plateau by using the total surface area of each side of the plateau.
Analysis of the Degeneration of the Menisci
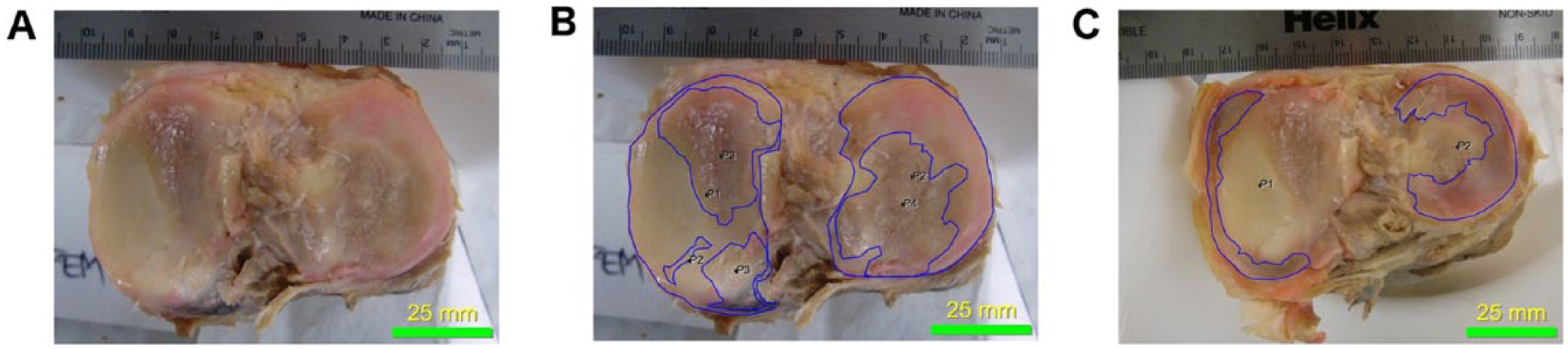
The menisci of the <70 years old specimens served as a control for the typical coverage of the medial and lateral tibial plateau by their respective meniscus. A percentage of tibial plateau surface area coverage was achieved by using the surface area of each meniscus ( Fig. 1C ) and dividing it by the total surface area of the respective side of the tibial plateau. Menisci measurements stayed internal to the opaque rim of the menisci where it fused with the joint capsule to ensure size was not overestimated. Figure 2 shows a magnified image of the medial meniscus for visualization of the fusion of the meniscus with the capsule and the substantial erosion of the meniscus and articular cartilage that occurred in the selected specimen.
Photographs of the tibial plateau of a right cadaveric knee specimen. (
Photographs of the tibial plateau of a right cadaveric knee specimen. (
Statistics
ACL thickness for the 3 age populations were compared using an ANOVA statistical test with a Newman-Keuls post-test for multiple comparisons. Total areas of tibial plateau degeneration and menisci coverage for the 3 age populations were also compared using an ANOVA statistical test with a Newman-Keuls post-test for multiple comparisons.
Results
Cruciate and Meniscofemoral Ligaments
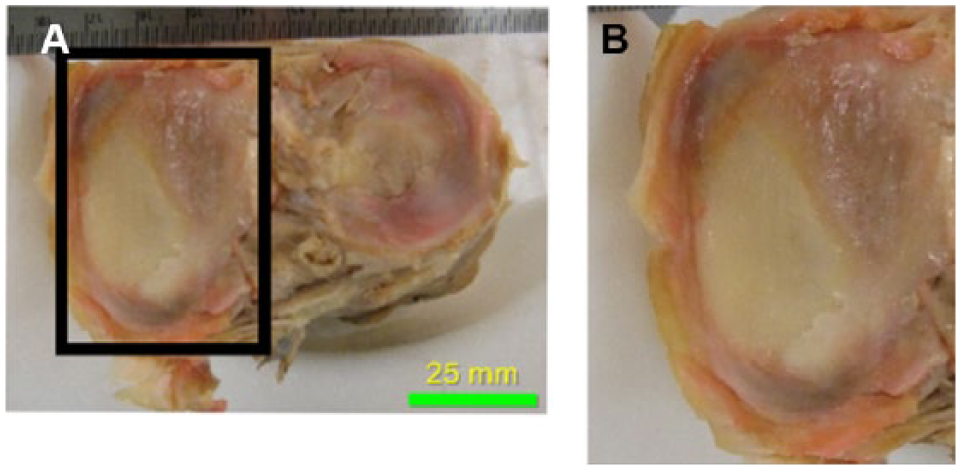
The ACL experienced substantial degenerative thinning with aging; this was demonstrated as a loss of ACL thickness (mm) from the <70 years old age group to the 70 to 79 years old age group and from the 70 to 79 years old age group to the ≥80 years old age group. The average ACL thickness for the <70 years old specimens was 27.49 ± 2.7 mm, whereas the ACL thickness for the 70 to 79 years old specimens and the ≥80 years old specimens was 24.63 ± 3.6 mm and 22.24 ± 3.0 mm, respectively. The 70 to 79 years old specimens had a 1.2-fold decrease in average ACL circumference (mm) compared to the <70 years old specimens (P < 0.001). The ≥80 years old specimens had a 1.24-fold decrease in ACL thickness compared to the <70 years old specimens (P < 0.001). There was also statistically significant thinning of the ACL when comparing the 70 to 79 years old specimens to the ≥80 years old specimens. There was a 1.1-fold decrease in the average circumference of the ACL in the ≥80 years old specimens compared to the 70 to 79 years old specimens (P < 0.01; Fig. 3 ). There was no statistical significance in variation of circumference of the PCL in the 3 age populations. Sixty-seven percent of the specimens analyzed contained a posterior meniscofemoral ligament.
Average thickness (mm) of the anterior cruciate ligament (ACL) in specimens <70 years old (blue), between 70 and 79 years old (green), and 80 years old and above (red). Histogram represents mean ± standard deviation millimeters of ACL circumference. Significantly different from “Below 70 years old” at ***P < 0.001. “70-79 years old” significantly different from “80 years old and above” at ++P < 0.01.
Tibial Plateau Articular Cartilage
The articular cartilage of the medial tibial plateau experienced statistically significant gross degeneration with aging. The <70 years old specimens had an average of 13.33 ± 7.8% surface area (mm2) damage to the medial articular cartilage, whereas the surface area damage for the 70 to 79 years old specimens and the ≥80 years old specimens was 25.11 ± 18.89% and 42.67 ± 15.79%, respectively. The ≥80 years old specimens had a 3.2-fold increase in surface area degeneration on the medial tibial plateau compared to the <70 years old specimens (P < 0.05) and a 1.7-fold increase in degeneration compared to the 70 to 79 years old specimens (P < 0.05; Fig. 4A ). The articular cartilage of the lateral tibial plateau experienced degeneration with aging, although there was no statistical significance. The <70 years old specimens had an average of 15 ± 15.03% surface area (mm2) damage to the lateral articular cartilage, whereas the surface area damage for the 70 to 79 years old specimens and the ≥80 years old specimens was 30 ± 21.26% and 40.67 ± 16.74%, respectively ( Fig. 4B ).
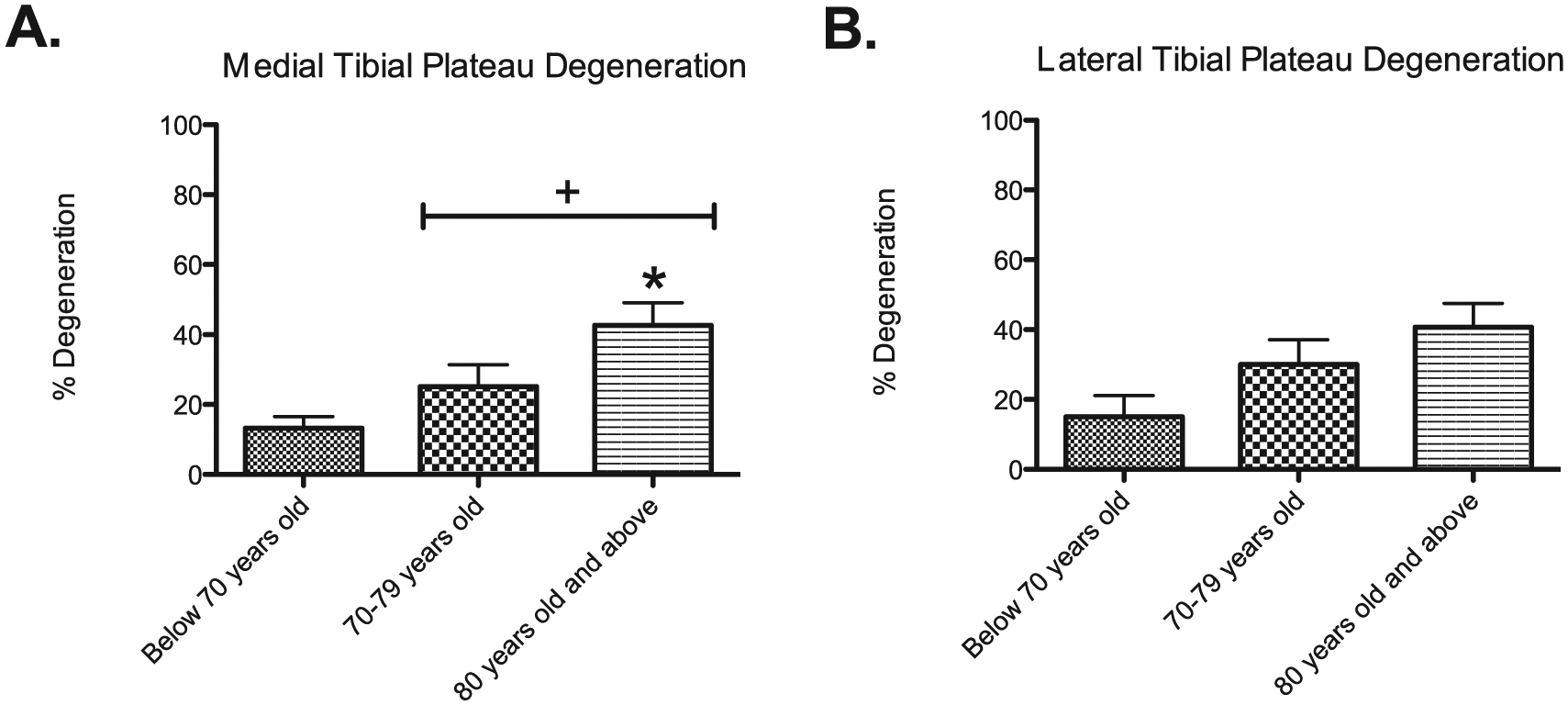
Average percent degeneration (mm2) of the medial (
Menisci Coverage of the Tibial Plateau
The medial meniscus had greater coverage of the medial tibial plateau in the <70 years old specimens compared with the 70 to 79 years old specimens and the ≥80 years old specimens. A total of 49 ± 6.54% of the medial tibial plateau was covered by the medial meniscus in the <70 years old specimens. A total of 38.33 ± 12.7% and 35.83 ± 9.45% of the medial tibial plateau was covered by the medial meniscus in the 70 to 79 years old specimens and the ≥80 years old specimens, respectively ( Fig. 5 ). There was no trend in surface area coverage of the lateral tibial plateau by the lateral meniscus in the 3 age populations. The <70 years old specimens had approximately 52% coverage of the lateral tibial plateau by the lateral meniscus, whereas the 70 to 79 years old specimens and the ≥80 years old specimens had approximately 49% and 52.5% coverage, respectively. One hundred percent of the specimens contained either the anterior meniscofemoral ligament or the posterior meniscofemoral ligament. This additional support to the lateral meniscus could reduce its degeneration as well as the degeneration of underlying lateral articular cartilage.
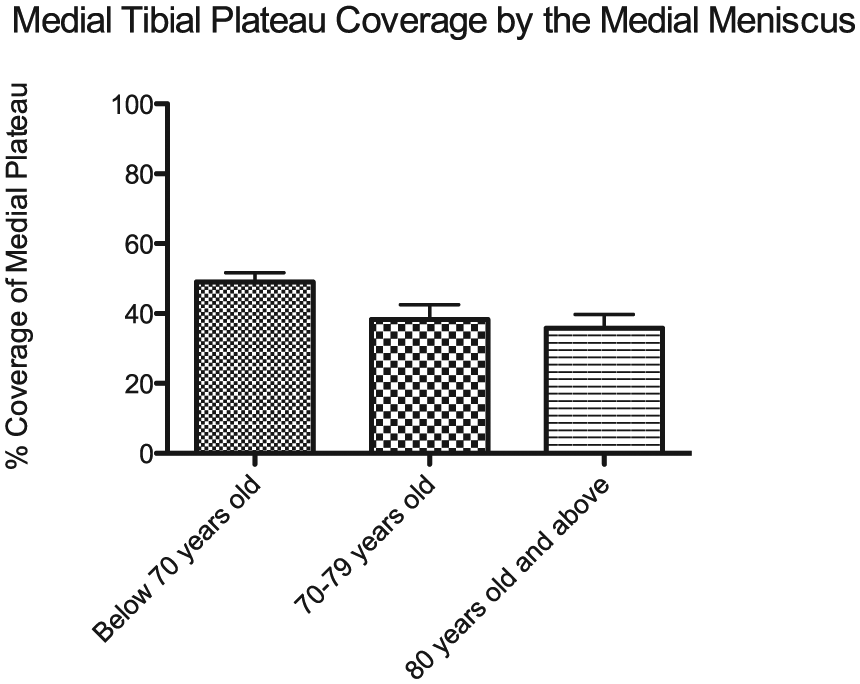
Average percent coverage (mm2) of the medial tibial plateau by the medial meniscus in specimens below 70 years old (blue), between 70 and 79 years old (green), and 80 years old and above (red). Histogram represents mean ± standard deviation mm2 of medial tibial plateau coverage. A total of 49 ± 6.54% of the medial tibial plateau was covered by the medial meniscus in the <70 years old specimens. A total of 38.33 ± 12.7% and 35.83 ± 9.45% of the medial tibial plateau was covered by the medial meniscus in the 70 to 79 years old specimens and the ≥80 years old specimens, respectively.
Discussion
Thinning of the ACL was the most statistically significant sign of degeneration with aging related to the intracapsular anatomy of the knee. There was a loss of 10.5% of the average circumference of the ACL from the <70 years old age group to the 70 to 79 years old age group. Compared with the <70 years old age group, there was a loss of 19% of the circumference of the ACL in the ≥80 years old age group. The current body of literature does not include a quantitative analysis that demonstrates a loss of thickness in the ACL with aging. The loss of circumference of the ACL with aging paralleled the loss of articular cartilage on the medial tibial plateau. There was an approximate loss of 12% articular cartilage surface area from the <70 years old age group to the 70 to 79 years old age group. Compared with the <70 years old age group there was a loss of 30% of the articular cartilage surface area in the ≥80 years old age group. These losses were statistically significant. The current body of literature has yet to depict such precise measurements related to chondral lesions and articular cartilage degradation on the tibial plateau with aging. Precise surface area losses have never been analyzed in a cadaveric model; these methods allow for more direct visualization of the damages and observation of finer cracks and regions of erosion that may exacerbate into more profound disease.
There are several factors that can lead to an increased propensity to degenerate the medial side of the articular cartilage on the tibial plateau compared with the lateral. During normal gait, 60% of the tibiofemoral compression force bears down on the medial tibial plateau. Tibiofemoral compression force equals 1.6 times the subject’s body weight during isometric contraction, whereas tibiofemoral compression force equals 3 times the subject’s body weight during 60° of flexion. 17 Individuals that regularly have the knee joints in a high degree of flexion due to recreational or occupational lifestyle choices may experience greater degeneration on the medial respects of the knee joint compared with the lateral. The principle anatomical structures that prohibit shearing forces are the cruciate ligaments and the musculature that acts on the knee joint; with their deterioration or atrophy medial tibial plateau degeneration may occur. 17 Given the medial tibial plateau and ACL displayed statistically significant and parallel degenerative changes with aging, it appears that the loss of ACL integrity or thickness allowed for greater shearing on the medial tibial plateau where tibiofemoral compressive forces are high.
There was an approximate loss of 15% articular cartilage surface area (mm2) on the lateral tibial plateau from the <70 years old age group to the 70 to 79 years old age group. Compared with the <70 years old age group, there was a loss of 26% of the articular cartilage surface area in the ≥80 years old age group. These losses were not statistically significant yet display a trend. Many individuals experience altered torque of their body weight at the knee joint due to obesity. With obesity, tibiofemoral compression forces shift from medial to lateral due to a lateral shift in weight. This lateral shift increases the degree of valgus orientation of the knee joint and simultaneously increases pressure on the lateral tibiofemoral interface. This could account for the approximately 50% of specimens that had greater degeneration of articular cartilage on the lateral tibial plateau compared with the medial tibial plateau. Currently in the United States, over two thirds of the population is overweight or obese with 1 in 20 individuals having extreme obesity with a body mass index over 40. 18
The average coverage of the medial tibial plateau by the medial meniscus was approximately 49%, 38%, and 36% for the 3 age populations, in ascending order. These values differ from the accepted 66% or two thirds coverage of the tibial plateau by the menisci. 19 There was a male, 26 years old specimen included in this analysis. The coverage of the medial tibial plateau by the medial meniscus in this individual was 59%, closer to the accepted value. Although there was no statistical significance in the loss of surface area coverage of the medial tibial plateau by the medial meniscus, there was a trend that warrants reinvestigation. Compared with the <70 years old specimens, the 70 to 79 years old specimens and the ≥80 years old specimens had a loss of 12% and 27% medial tibial plateau surface area coverage, respectively. The <70 years old specimens that had a 3% or greater loss of menisci coverage (n = 3) of the medial tibial plateau compared with the age group average for coverage displayed medial tibial plateau degeneration that was at least 3% greater than the group average. The 70 to 79 years old specimens that had a 3% or greater loss of menisci coverage (n = 3) of the medial tibial plateau compared with the age group average for coverage displayed medial tibial plateau degeneration that was at least 7% greater than the group average. The ≥80 years old age group had 2 specimens that had a loss of menisci coverage of the medial tibial plateau that was 4% or greater than the group average for coverage. One of these specimens had medial tibial plateau degeneration that was 25% greater than the group average. The second specimen did not fit the trend where low meniscal coverage of the tibial plateau led to high tibial plateau degeneration. This specimen had 8% less tibial plateau degeneration than the average degeneration for the ≥80 years old age group. This individual was a male philosophy professor that lived to the age of 87 and had no major clinical history that would increase the potential for a sedentary lifestyle. Due to the relative health of the donor, the lack of tibial plateau degeneration may imply that OA of the knee commenced later in life for this individual and the menisci were very functional in their protective role of the articular cartilage and underlying cortical bone.
This study will be repeated to confirm current findings and additional measurements will be collected to assess the intracapsular ligaments. Prevalence of the posterior meniscofemoral ligaments will be reassessed as well as the average circumferences for the PCL. Prevalence of the anterior meniscofemoral ligament will be assessed in the future. Epitenon and endotenon fat associated with the PCL in this population was a limitation for the assessment. It is likely that several anterior meniscofemoral ligaments were missed in dissection due to the substantial infiltration. Although the anterior meniscofemoral ligament typically does not greatly contribute to the circumference of the PCL and large anterior meniscofemoral ligaments were noted and reflected, those measurements should be reassessed to validate the circumference findings related to the PCL. There was an additional limitation related to the photography of the tibial plateau. A degree of error can be attributed to the fact that the tibial plateau is not perfectly flat; therefore, measurements for the total tibial plateau surface areas and the surface areas of degeneration are likely underestimated. As previously mentioned, the fact that every donor did not provide a complete clinical history to the body donation program presents a limitation of the model. In some cases, undeclared disease, unknown disease, and medication regimens may have led to the increased degeneration in the knee joint oppose to attributing all analyzed damages to OA. The sample size and age distribution of the population may contribute to experimental error, although follow-up studies will analyze degenerative changes in a population of twice the size.
This initial study validated the measurement methods for degenerative changes seen on the tibial plateau. Future studies on cadaver knee joints will aim to more specifically isolate discrete regions of the tibial plateau and femoral condyles experiencing articular cartilage loss. With the increasing use of pluripotent stem cell injections into arthritic joints, this study may determine discrete sites for stem cell administration based on age and age of symptom appearance. In the study at hand, there were no sex comparisons based on the population size but the follow-up study aims to increase population size so that potential sexual dimorphisms can be noted. This study and its following associated assessments are significant as they isolate specific age-related degenerative changes in the knee in a more precise method than previously implemented. Arthroscopic and radiographic analyses of age-based patient populations have yet to achieve ACL thickness or cartilage surface area losses associated with the age groups. Isolating trends in degeneration of the intracapsular anatomy of the knee with aging will allow clinicians to advise lifestyle changes in specific at-risk populations and may potentially lead to preventative surgical innovations that can correct deficits before they exacerbate into more serious degenerative alterations. For example, now having the understanding that the ACL greatly thins between the eighth and ninth decades of life, physical therapists may recommend hamstring and quadriceps strengthening to support a knee joint now prone to shearing in patients turning 70 years old. Seventy-year-old patients with difficulties gaining muscle mass may be recommended to undergo preventative complete or partial ACL reconstructions to prohibit cartilage and cortical bone degeneration, avoiding a more invasive joint replacement surgery. Precise age-based histological changes in the ACL have yet to be thoroughly studied nor have mechanistic events leading to connective tissue wasting; authors aim to address these unexplained aspects of OA in future studies. Gross cross-sectional photographs of the ACL at average thickness will also be included in future analyses for better visual representation of the thickness alterations with age. An ongoing catalog of degenerative changes in the knee based on occupation, sex, age, and clinical history is in its infancy and will be assessed in a future longitudinal study that follows cadaver populations by year of donation.
Footnotes
Acknowledgment and Funding
Authors would like to acknowledge the aid of Kerry Peterson and Wayne Woodward of the University of Utah’s Body Donation Program in preparing and distributing the specimens. This study was supported by an internal grant from Rocky Mountain University of Health Professions for Protocol No. 150430-04.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was waived by Rocky Mountain University of Health Profession’s Institutional Review Board in Provo, UT because it was determined to be research not involving human subjects.
