Abstract
Objective
Moderate mechanical stress is necessary for preserving the cartilage. The clinician empirically understands that prescribing only exercise will progress osteoarthritis (OA) for knee OA patients with abnormal joint movement. When prescribing exercise for OA, we hypothesized that degeneration of articular cartilage could be further prevented by combining interventions with the viewpoint of normalizing joint movement.
Design
Twelve-week-old ICR mice underwent anterior cruciate ligament transection (ACL-T) surgery in their right knee and divided into 4 groups: ACL-T, controlled abnormal joint movement (CAJM), ACL-T with exercise (ACL-T/Ex), CAJM with exercise (CAJM/Ex). Animals in the walking group were subjected to treadmill exercise 6 weeks after surgery, which included walking for 18 m/min, 30 min/d, 3 d/wk for 4 weeks. Joint instability was measured by anterior drawer test, and safranin-O staining and immunohistochemical staining were performed.
Results
OARSI (Osteoarthritis Research Society International) score of ACL-T/Ex group showed highest among 4 groups (P < 0.001). And CAJM/Ex group was lower than ACL-T/Ex group. Positive cell ratio of IL-1β and MMP-13 in CAJM/Ex group was lower than ACL-T/Ex group (P < 0.05).
Conclusions
We found that the state of the intra-articular environment can greatly influence the effect of exercise on cartilage degeneration, even if exercise is performed under the same conditions. In the CAJM/Ex group where joint movement was normalized, abnormal mechanical stress such as shear force and compression force accompanying ACL cutting was alleviated. These findings may highlight the need to consider an intervention to correct abnormal joint movement before prescribing physical exercise in the treatment of OA.
Introduction
Knee osteoarthritis (OA) is caused by articular cartilage degeneration and its progression results in arthralgia, motor dysfunction, and impairment of activities of daily living. As societies are aging worldwide, it is imperative to develop strategies for extending the healthy life expectancy and minimizing medical expenses associated with aging-specific disorders such as OA. In this context, substantial effort has been expended to elucidate the pathology of OA and to develop therapeutic methods. However, to date, only symptomatic treatment is available for OA.
It is currently accepted that moderate mechanical stress is necessary for preserving the cartilage. In rats with healthy cartilage, treadmill exercise was associated with increased expression of factors involved in extracellular matrix synthesis, as well as with suppression of inflammatory factors. 1 Therefore, exercise is required for cartilage preservation. Furthermore, recent evidence from animal models of surgically induced OA indicates that the progression of cartilage degeneration can be delayed by moderate exercise.2-7
On the other hand, joint instability induces excessive mechanical stress and contributes to OA onset and progression. 8 In addition, joint instability resulting from knee joint ligament injury, meniscus damage, or malalignment leads to inflammation and cartilage degeneration.9,10 Many patients with knee OA have joint instability, and the characteristic gait disturbance in such patients is involved in OA progression.11-13 The association between joint instability and knee OA is well established.
Studies using animal models of OA aim to elucidate the mechanisms underlying the onset and progression mechanisms of OA. The anterior cruciate ligament transection (ACL-T) model, which is a commonly used animal model of surgically induced OA, involves cutting the ACL in order to induce joint instability and progression to OA. However, studies based on ACL-T cannot clarify the influence of biological factors such as inflammation accompanying ligament rupture or secondary joint instability. In contrast, the controlled abnormal joint movement (CAJM) model, proposed by Murata et al.14,15 and Onitsuka et al. 16 involves ACL-T followed by another surgical procedure to re-stabilize the joint and reduce joint instability. In the CAJM model, the expression of inflammatory factors and cartilage matrix degrading factors is suppressed, joint instability is reduced, and progression to OA is slower.
Previous studies often used models involving ACL-T or destabilization of the medial meniscus to examine the effect of exercise intervention. However, in all such studies, exercise intervention was performed in the presence of joint instability caused by ligament rupture. Therefore, the impact of joint instability on the effectiveness of exercise intervention is unknown.
We hypothesized that exercise intervention is more effective at preventing the degeneration of articular cartilage in knee OA if joint instability is controlled. In the present study, we used mouse models of surgically induced OA to test this hypothesis.
Methods
Animals and Experimental Design
This study was approved by the Animal Research Committee of Saitama Prefectural University (approval number: 29-12), and the animals were handled in accordance with the relevant legislation and institutional guidelines for humane animal treatment. This study used 28 adults (12-week-old) ICR (Institute for Cancer Research) male mice. The mice were divided into 4 experimental groups: ACL-T without exercise intervention, CAJM without exercise intervention, ACL-T with exercise intervention (ACL-T/Ex), and CAJM with exercise intervention (CAJM/Ex) ( Fig. 1 ). All mice were housed 2 to a plastic cage maintained at a temperature of 23°C ± 1°C with a 12-hour light/dark cycle.
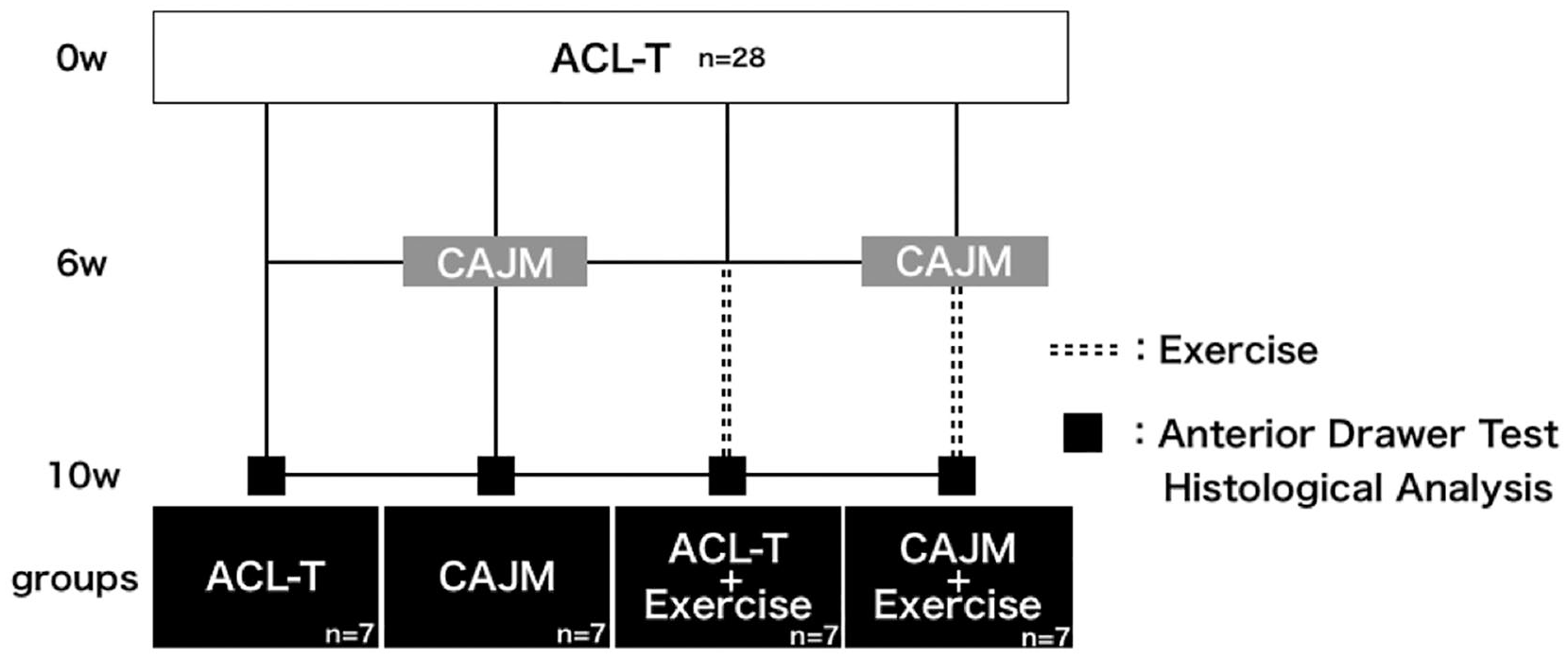
Experimental design. anterior drawer test, histological evaluation, and immunohistochemical evaluation were performed. These analyses involved the ACL-T group without exercise intervention, CAJM group without exercise intervention, ACL-T with exercise intervention (ACL-T/Ex) group, and CAJM with exercise intervention (CAJM/Ex) group (for each group, n = 7). ACL-T, anterior cruciate ligament transection; CAJM, controlled abnormal joint movement.
Surgical Procedures
The ACL-T and CAJM procedures were performed with the mice under a combination anesthetic (medetomidine, 0.375 mg/kg; midazolam, 2.0 mg/kg; and butorphanol, 2.5 mg/kg). To induce knee OA in the right hind limb, the medial capsule was exposed, and the ACL was transected using scissors. For mice assigned to ACL-T groups, the joint was loosely stitched using thread. For mice assigned to CAJM groups, a 25-gauge needle was used to create bone tunnels in the anterior portion of the proximal tibia and in the posterior portion of the femoral condyle in order to decrease the anterior translation of the tibia onto the femur. Subsequently, 4-0 nylon thread was passed through the bone tunnels to compensate for ACL function and help control the anterior translation of the tibia. The results of verifying the validity of the model were added to the supplemental material.
Anterior Drawer Test
To assess knee joint instability, the anterior drawer test was performed using a constant force spring (0.05 kgf; Sanko Spring Co., Ltd., Fukuoka, Japan) and a soft X-ray device (M-60; Softex Co., Ltd., Kanagawa, Japan). The right hind limb was collected for this experiment. The half of the quadriceps proximal to the femur and the hamstrings were cut with scissors, and then the femur was fixed with a clamp. The test pulled the proximal tibia forward with 4-0 nylon thread. The radiographic parameters were as follows: tube potential, 28 kV; tube current, 1.5 mA; exposure time, 1 second. The image was digitized using a NAOMI digital image sensor (RF Co., Ltd., Nagano, Japan). Based on the soft X-ray image, anterior displacement was quantified using dedicated image analysis software (Image J; National Institutes of Health, Bethesda, MD, USA).
Treadmill Exercise Intervention
Mice allocated to the exercise groups (ACL-T/Ex and CAJM/Ex) were exercised on a rodent treadmill. After the mice in the CAJM groups underwent the second surgery, all mice were allowed to become familiarized with the treadmill environment for 3 days. Afterward, the mice allocated to exercise groups were exercised on the treadmill at a constant speed of 18 m/min for 30 min/d, 3 d/wk, for 4 weeks.
Histological Analysis
At the end of the study period, the mice were sacrificed, and the knee joint was collected and fixed in 4% paraformaldehyde/phosphate-buffered saline for 24 hours, followed by decalcification in 10% ethylenediaminetetraacetic acid for 21 days, dehydration in 70% and 100% ethanol and xylene, and embedding in paraffin blocks. Thin sections (7 μm) were cut in the sagittal plane using a microtome (ROM-360; Yamato Kohki Industrial Co., Ltd., Saitama, Japan), stained with safranin-O/fast green, and subjected to histology evaluation to estimate the degree of cartilage damage. The Osteoarthritis Research Society International (OARSI) histopathology grading system was used to assess cartilage damage reflected as structural changes and fibrillation lesions. 17 Two independent observers (T Kano and KO) performed OARSI scoring on a scale of 8 stages (0, 0.5, 1-6), and the average value was retained. Furthermore, cartilage roughness and thickness were evaluated as characteristics of degenerative OA. Cartilage thickness was measured in the tibia anterior and tibia posterior regions. For each region, the roughness of the cartilage surface was calculated as the ratio between the actual length along the articular surface and the length of an approximately straight line connecting 2 points in the region of interest.
Definition of Regions
Two regions of interest were used in this study, namely the tibia anterior and tibia posterior. The tibia anterior was defined as the region beyond the outer edges of the anterior meniscal horn, while the tibia posterior was defined as the region beyond the outer edges of the posterior meniscal horn.
Immunohistochemical Analysis
In this study, we examined interleukin-1β (IL-1β) as a marker of inflammation and matrix metalloproteinase-13 (MMP-13) as a marker of tissue catabolism. To evaluate the expression of IL-1β and MMP-13, we performed immunohistochemical staining using the avidin-biotinylated enzyme complex method and the VECTASTAIN Elite ABC Rabbit IgG Kit (Vector Laboratories, Burlingame, CA, USA). The tissue sections were deparaffinized with xylene and ethanol, and antigen activation was carried out using proteinase K (Worthington Biochemical Co., Lakewood, NJ, USA) for 30 minutes. Endogenous peroxidase was inactivated with 0.3% H2O2/ethanol (FUJIFILM Wako Pure Chemical Co., Osaka, Japan) for 30 minutes. Nonspecific binding of the primary antibody was blocked using normal goat serum for 20 minutes, and then the sections were incubated with anti-IL-1β and anti-MMP-13 primary antibodies overnight at 4°C. Afterward, the sections were incubated with biotinylated secondary antibody anti-rabbit IgG and stained with aniline. For analysis, we calculated the ratio between the number of IL-1β- or MMP-13-positive cells and the number of chondrocytes in an articular cartilage area of 10000 μm2 (100 μm × 100 μm).
Statistical Analysis
All analyses were performed using R version 3.5.1 (http://www.R-project.org/). First, the Shapiro-Wilk test was used to check for normality of the distribution for each data set. To clarify the impact of joint instability on the effectiveness of exercise intervention in knee OA, the following data were compared among the 4 experimental groups (ACL-T, ACL-T/Ex, CAJM, CAJM/Ex) using analysis of variance with Tukey’s post hoc test: tibia anterior displacement, OARSI score, cartilage thickness, and cartilage roughness. The percentages of IL-1β- and MMP-13-positive cells were compared among groups using the Kruskal-Wallis test with Steel-Dwass post hoc analysis. Parametric data are shown as the mean with 95% confidence interval, whereas nonparametric data are shown as the median with interquartile range. All significance thresholds were set at 5%.
Results
X-Ray Radiography
Joint instability was quantified using X-ray radiography with the anterior drawer test ( Fig. 2A ).
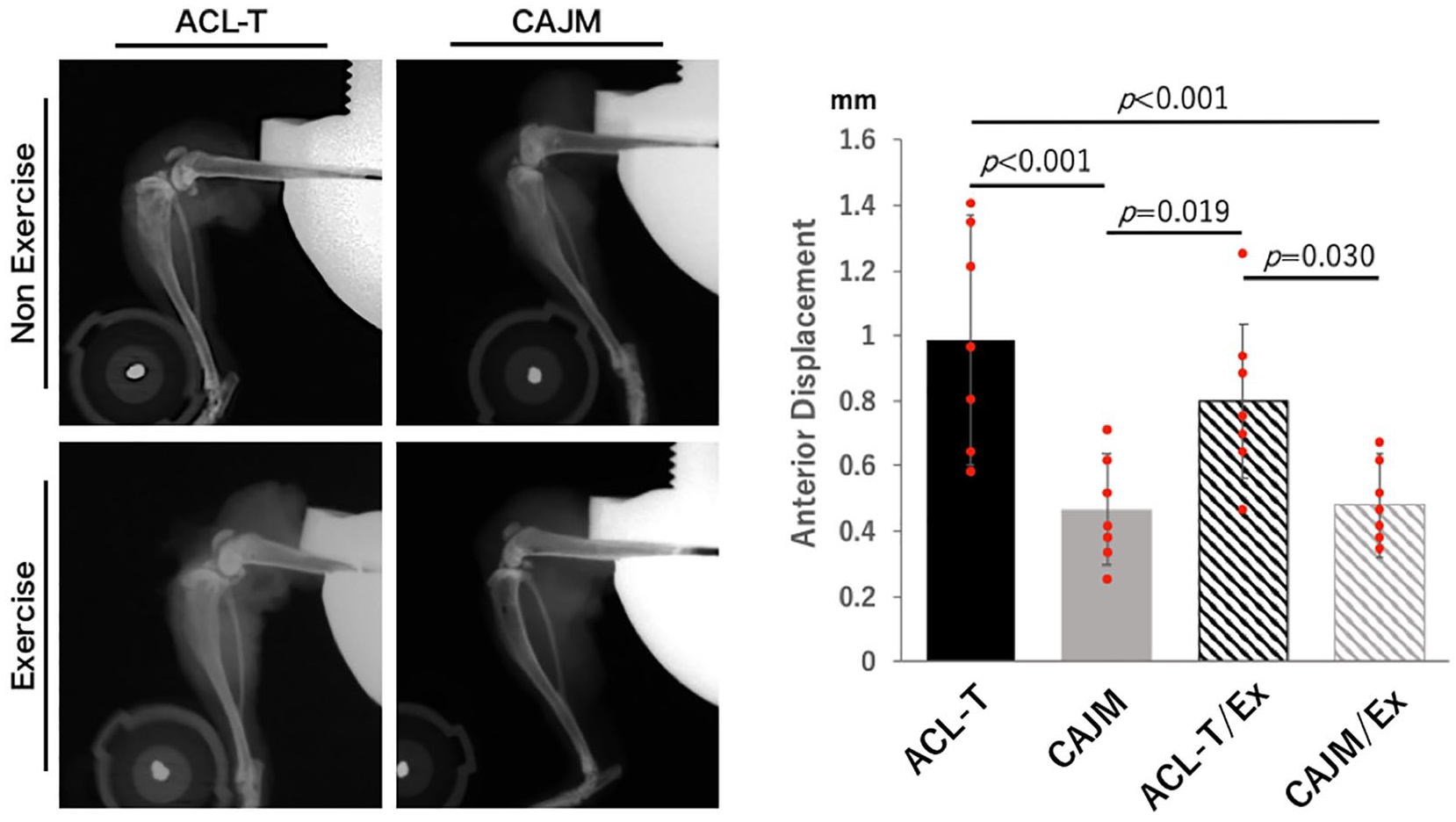
Knee joint instability with and without exercise intervention in mice stratified according to the joint environment following surgical induction of osteoarthritis (OA). (
Compared with the ACL-T and ACL-T/Ex groups, the CAJM and CAJM/Ex groups had significantly lower anterior displacement of the tibia (CAJM vs ACL-T, P < 0.001; CAJM vs ACL-T/Ex, P = 0.019; CAJM/Ex vs ACL-T, P < 0.001; CAJM/Ex vs ACL-T/Ex, P = 0.030) (ACL-T, 0.99 [0.60-1.37] mm; CAJM, 0.47 [0.30-0.64] mm; ACL-T/Ex, 0.80 [0.56-1.04] mm; CAJM/Ex, 0.48 [0.32-0.64] mm) ( Fig. 2B ).
Histological Analysis
Chondrocyte hypertrophy in the anterior part of the articular cartilage was confirmed for both models (ACL-T and CAJM) but no significant OA progression was noted in any group ( Fig. 3 ). Exercise groups had significantly lower OARSI score than that noted for the nonexercise groups: ACL-T, 0.8 [0.3-1.2]; CAJM, 0.6 [0.2-1.0]; ACL-T/Ex, 0.6 [0.3-0.9]; CAJM/Ex, 0.6 [0.2-0.3] (P = 0.754). In the ACL-T/Ex group, marked surface fibrillation and reduction in cartilage thickness were noted in the posterior part of the articular cartilage, together with a significantly higher OARSI score than that noted for the other 3 groups (ACL-T/EX vs. ACL-T, CAJM, or CAJM/Ex, P < 0.001) (ACL-T, 1.6 [0.8-2.3]; CAJM, 1.0 [0.4-1.6]; ACL-T/Ex, 3.6 [2.4-4.9]; CAJM/Ex, 1.1 [0.7-1.5]).
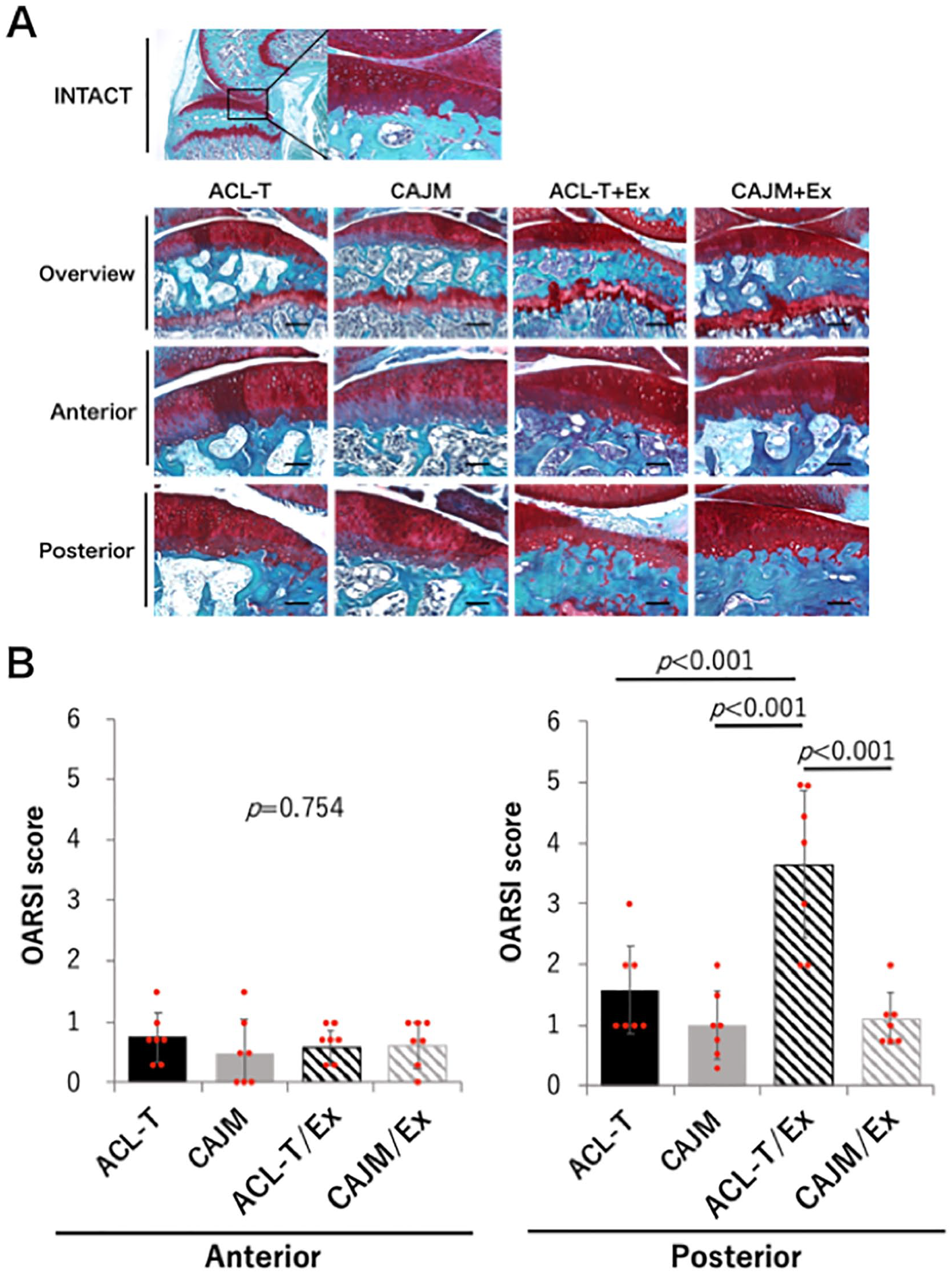
Knee cartilage degeneration with and without exercise intervention in mice stratified according to the joint environment following surgical induction of osteoarthritis. Mice in the CAJM groups underwent joint restabilization after ACL-T. (
There was no significant difference among the 4 groups with regard to cartilage thickness in the anterior part (ACL-T, 175.6 [167.5-183.7] μm; CAJM, 174.2 [166.9-181.5] μm; ACL-T/Ex, 178.8 [171.9-185.6] μm; CAJM/Ex, 169.7 [157.0-182.5] μm; P = 0.401) ( Fig. 4A ). However, in the posterior part, cartilage thickness was significantly higher for the ACL-T/Ex group (ACL-T/EX vs. ACL-T, CAJM, or CAJM/Ex, p<0.001) (ACL-T, 140.0 [127.4–152.6] μm; CAJM, 152.3 [139.0–165.6] μm; ACL-T/Ex, 94.9 [86.8–103.0] μm; CAJM/Ex, 152.0 [144.5–159.5] μm).
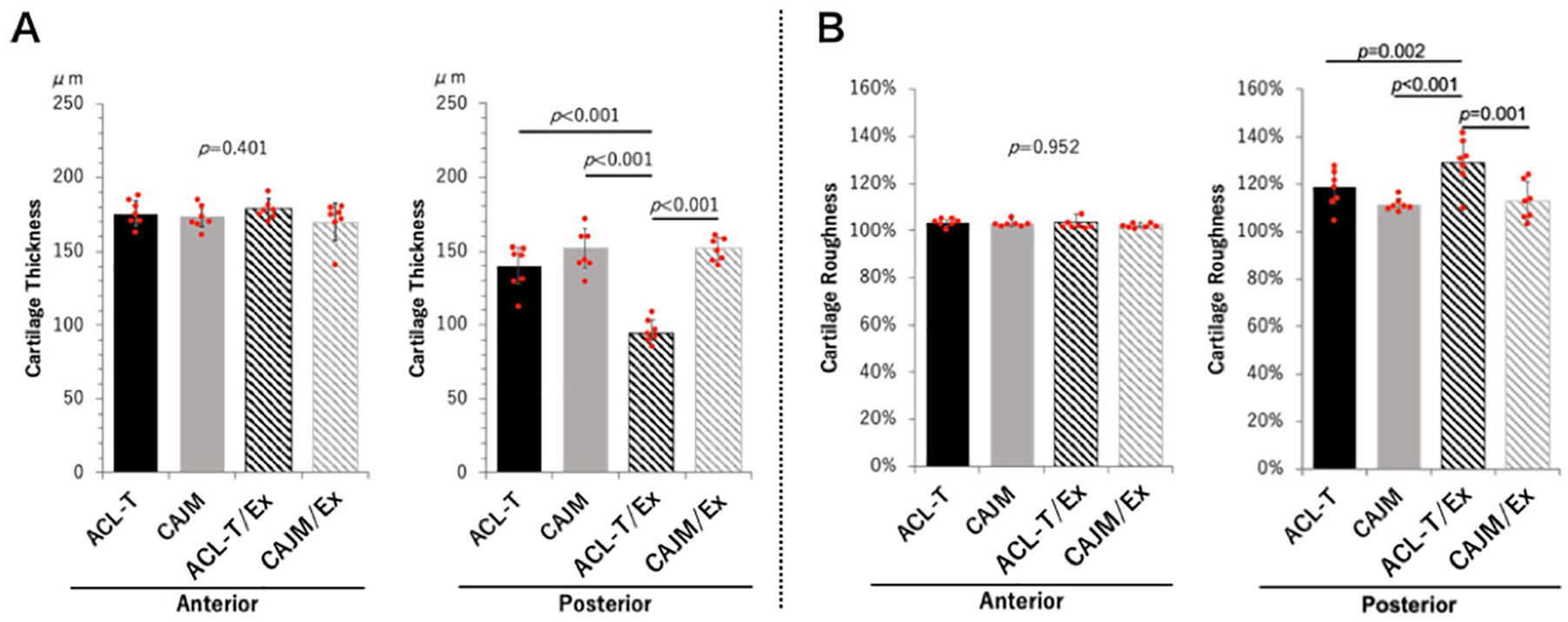
Knee cartilage thickness and roughness with and without exercise intervention in mice stratified according to the joint environment following surgical induction of osteoarthritis. Mice in the CAJM groups underwent joint restabilization after ACL-T. (
With regard to the roughness of the cartilage surface in the anterior part, there was no significant difference among the 4 groups (ACL-T, 103.5% [102.3%-104.7%]; CAJM, 103.1% [101.5%-104.8%]; ACL-T/Ex, 103.7% [100.3%-107.2%]; CAJM/Ex, 102.6 [101.7%-103.6%]; P = 0.952) ( Fig. 4B ). However, the cartilage roughness in the posterior part was significantly higher for the ACL-T/Ex group (ACL-T/Ex vs. ACL-T, P = 0.002; ACL-T/Ex vs. CAJM, P < 0.001; ACL-T/Ex vs. CAJM/Ex, P = 0.001) (ACL-T, 118.9% [111.8%-125.9%], CAJM, 111.2% [108.5%-114.0%]; ACL-T/Ex, 129.0% [119.2%-138.8%]; CAJM/Ex, 112.9% [104.4%-121.3%]).
With regard to inflammation, IL-1β-positive cells were confirmed from the cartilage surface layer to the deep layer in the ACL-T, CAJM, and ACL-T/Ex groups but not in the CAJM/Ex group ( Fig. 5A ). The percentage of IL-1β-positive cells in the anterior part of the cartilage was significantly higher in the ACL-T and ACL-T/Ex groups than in the CAJM/Ex group (ACL-T vs. CAJM/Ex, P = 0.031; ACL-T/Ex vs. CAJM/Ex, P = 0.013) (ACL-T, 32.2% [27.4%-42.1%]; CAJM, 24.6% [21.0%-34.5%]; ACL-T/Ex, 28.3% [27.5%-36.7%]; CAJM/Ex, 15.5% [13.4%-21.5%]) ( Fig. 5B ). A similar trend was noted for the posterior part of the cartilage, where the percentage of IL-1β-positive cells was significantly higher for the ACL-T and ACL-T/Ex groups than for the CAJM/Ex group (ACL-T vs. CAJM/Ex and ACL-T/Ex vs. CAJM/Ex, P = 0.048) (ACL-T, 40.6% [34.3%-51.0%]; CAJM, 24.1% [17.2%-32.5%]; ACL-T/Ex, 35.8% [31.1%-38.0%]; CAJM/Ex, 20.0% [14.5%-24.9%]).
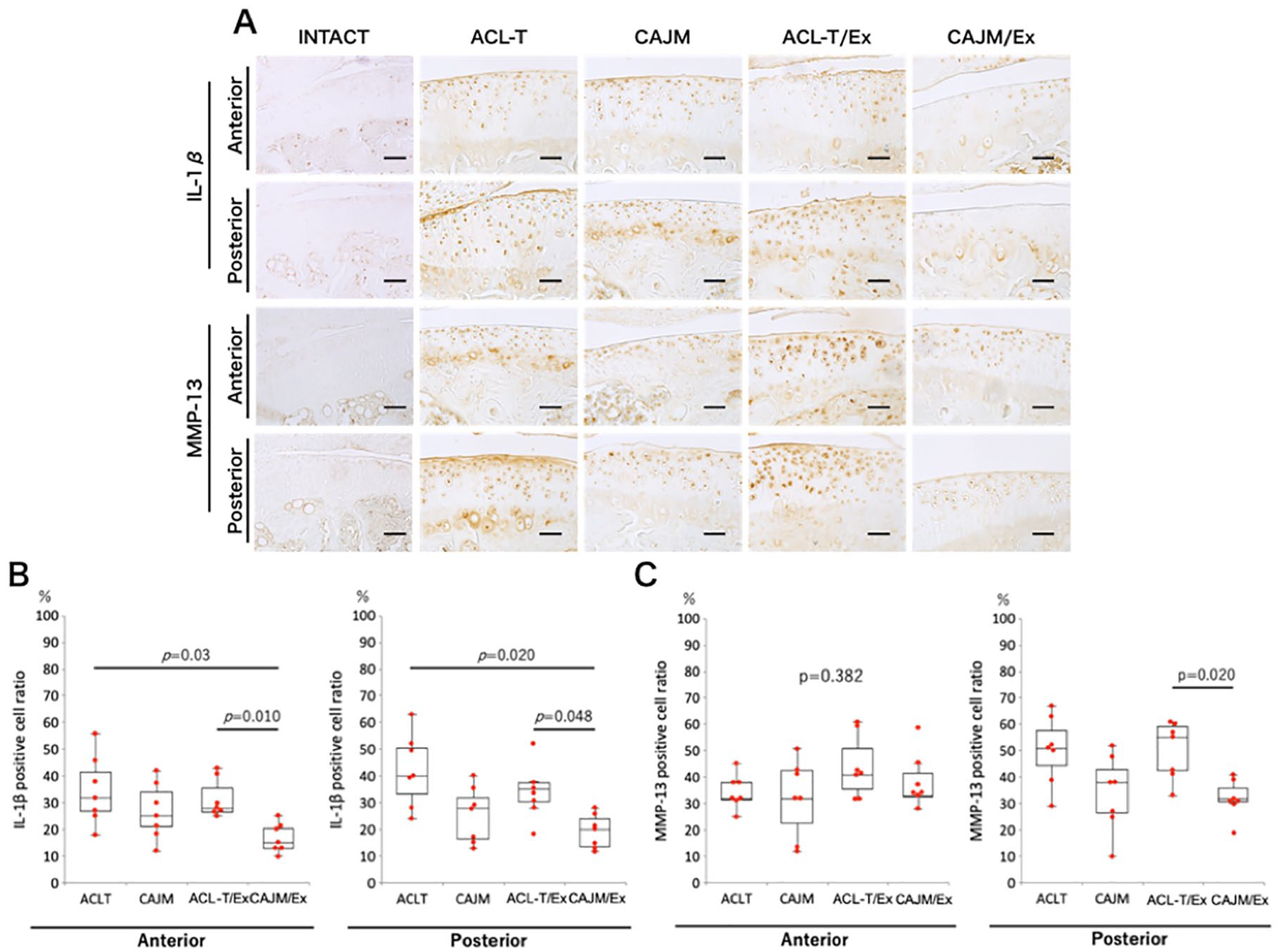
Knee inflammation and tissue catabolism with and without exercise intervention in mice stratified according to the joint environment following surgical induction of osteoarthritis. Mice in the CAJM groups underwent joint restabilization after ACL-T. (
Similarly, MMP-13-positive cells were confirmed in the ACL-T and ACL-T/Ex groups, compared with very weak MMP-13 expression in the CAJM and CAJM/Ex groups ( Fig. 5A ). However, while no significant difference among the four groups was noted in terms of the percentage of MMP-13-positive cells in the anterior part of the cartilage (ACL-T, 32.2% [32.0%-38.1%]; CAJM, 32.6% [23.3%-43.5%]; ACL-T/Ex, 41.1% [36.0%-51.8%]; CAJM/Ex, 33.8% [30.0%-42.4%]; P = 0.382), the percentage of MMP-13-positive cells in the posterior part of the cartilage was significantly higher for the ACL-T/Ex group than for the CAJM/Ex group (ACL-T/Ex vs. CAJM/Ex, P = 0.020) (ACL-T, 51.5% [45.3%-58.2%]; CAJM, 33.4% [27.6%-43.1%]; ACL-T/Ex, 55.2% [43.5%-59.1%]; CAJM/Ex, 32.9% [31.0%-36.5%]) ( Fig. 5C ).
Discussion
Previous studies reported that exercise alone could delay the progress of OA. However, our present results indicate that the internal environment of the joint strongly modulates the effect of exercise, sometimes completely canceling the benefit. In other words, when prescribing exercise, we need to consider the internal environment of the knee joint. This is the first study to assess and confirm the impact of controlling abnormal joint movement on the effectiveness of exercise therapy in knee OA.
Certain aspects of our results warrant further discussion. First, histological analysis revealed no difference between the ACL-T and CAJM groups regarding OARSI score, IL-1β-positive cell percentage, or MMP-13-positive cell percentage. Murata et al. 14 and Onitsuka et al., 16 who used the same CAJM model, also found no difference in OARSI score at 4 weeks after intervention but did report that differences could be seen after 8 or 12 weeks. These findings suggest that a follow-up of 4 w after joint intervention might be insufficient to reveal the influence of the internal joint environment on cartilage degeneration by OARSI score. On the other hand, Murata et al., 14 reported a difference in tumor necrosis factor (TNF)-α levels even if the absence of a difference in OARSI scores. In other words, whereas only IL-1β and MMP-13 were examined in this study, ACL-T and CAJM may differ in terms of other factors related to cartilage degeneration.
Interestingly, we found that the OARSI score was significantly higher for the ACL-T/Ex group than for the other three groups. Moreover, in the ACL-T/Ex group, the cartilage thickness was lower, and the roughness of the cartilage surface layer was substantially higher than for the other groups. High OARSI score, decreased cartilage thickness,14,18,19 and increased surface roughness20,21 are pathological findings characteristic of OA. In the ACL-T model, forward dislocation of the tibia is clearly visible on X-ray radiography. In addition, osteophytes start to develop on the posterior part of the tibia as soon as at 2 weeks after ACL-T, and the osteophyte volume increases with time.22,23 Onur et al. 24 reported that degeneration of the articular cartilage progresses after ACL-T but not only due to compressive force. In other words, the ACL-T model exhibits forward instability of the tibia and results in OA onset and progression. In the present study, we found worse OARSI score after ACL-T with exercise alone (ACL-T/Ex), which was different from the previous study. Previous reports indicate that moderate-intensity exercise helps preserve the articular cartilage while high-intensity exercise accelerates OA.25-28 Our present study is the first to include treadmill exercise in a mouse OA model, whereas previous studies focused mainly on rats. Mice are small compared with rats and, when exercising under the same conditions, would need to perform a higher number of steps. In other words, in our study targeting mice, the treadmill exercise was conducted at high intensity, which might have accelerated OA progression in the ACL-T/Ex group. On the other hand, mice in the CAJM/Ex group were subjected to the same exercise regime and did not exhibit OA progression. In the CAJM/Ex group, abnormal joint movement during exercise was controlled, which reduced abnormal shear and compression forces and thus might have prevented OA progression. Taken together, our results indicate not only that cartilage degeneration can be prevented by improving the intraarticular environment, thus improving the quality of joint motion during exercise, but that this protective effect holds even at high-intensity exercise, which would otherwise promote OA.
Compared with the CAJM/Ex group, the ACL-T/Ex group exhibited a higher percentage of IL-1β-positive cells throughout the entire articular cartilage, and a higher percentage of MMP-13-positive cells in the posterior portion of the cartilage. IL-1β and MMP-13 promote cartilage catabolism and inflammation, serving as representative biomarkers of OA, though it should be highlighted that OA is a complex condition.29,30 Previous reports showed that abnormal mechanical stress causes an increase in inflammatory factors and cartilage matrix degrading factors, 31 and that IL-1β and MMP-13 levels increase with the intensity of the stress on the articular cartilage, supporting the role of these proteins as cartilage degeneration factors produced in OA. On the other hand, IL-1β levels were increased throughout the cartilage and not only in the posterior part of the tibia, suggesting that this may simply be the result of direct mechanical stress. Biomechanical changes in knee joint following ACL-T are characterized by decreased aggrecan and collagen gene expression in chondrocytes and synovial cells. 32 In addition, abnormal knee mechanics lead to increased IL-1β, TNF-α, and MMP expression in the entire knee joint, including the synovium and synovial fluid.33,34 Thus, it is possible that ACL-T-induced changes in the synovial fluid affect the entire environment of the joint enclosed in the synovial membrane. In this study, we confirmed that not only the cartilage lesion but also the insult to the tendon (ACL-T) causes an inflammatory response throughout the joint, with increased IL-1β expression both on the anterior and posterior part of the tibia, and that this response is due to the mechanical abnormality.
Cartilage matrix-degrading proteinases, evaluated here in terms of MMP-13, are expressed in the articular cartilage not only due to the increase in inflammatory factors such as IL-1β but also due to chondrocyte hypertrophy, which occurs in response to abnormal mechanical stress.35,36 In terms of MMP-13 expression, there was no difference between the ACL-T group and the CAJM group for either side of the articular cartilage. However, MMP-13 expression on the posterior part of the tibia was increased in the ACL-T/Ex group and decreased in the CAJM/Ex group, suggesting that the abnormal joint movement, which was not controlled in the ACL-T/Ex group, resulted in concentration of stress on the posterior tibia, with corresponding expression of matrix-degrading proteinases in addition to the inflammatory reaction. Meanwhile, such concentration of stress did not occur in the CAJM/Ex group, suggesting that it is possible to avoid local accumulation of stress in the joint by imposing exercise therapy after controlling abnormal joint movement, thus suppressing the expression of MMP-13 through the anti-inflammatory action of exercise.
The exact influence of physical therapy intervention on cartilage degeneration remains unclear. Although correcting abnormal joint movement is generally the most important focus of physical therapy in patients with knee OA, especially in the clinical setting, the relationship with cartilage degeneration has not been elucidated. In the present study, we found that the state of the intra-articular environment can greatly influence the effect of exercise on cartilage degeneration, even if exercise is performed under the same conditions. In the clinical setting, these findings may translate in the need to consider an intervention to correct abnormal joint movement before prescribing physical exercise in the treatment of OA ( Fig. 6 ).
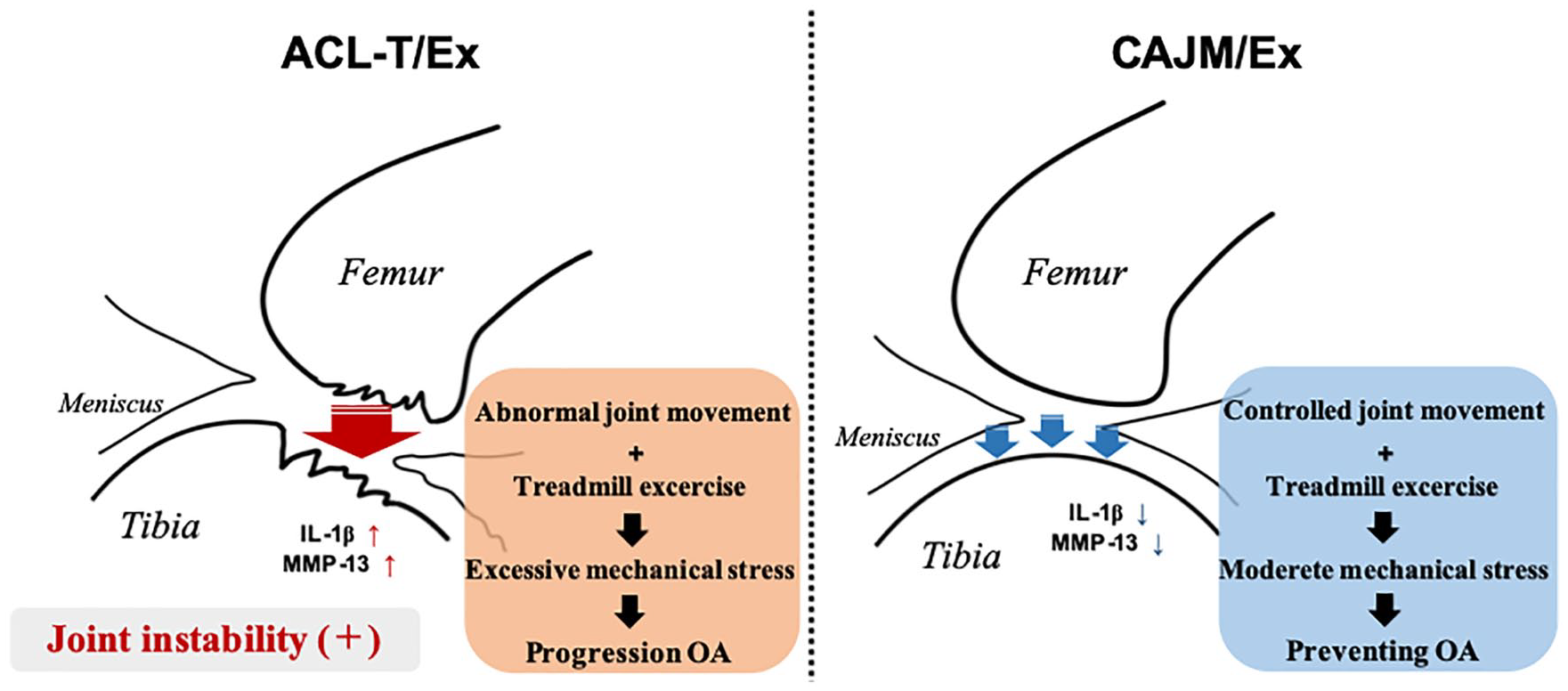
It is necessary to consider surgical intervention to correct abnormal joint movement before prescribing physical exercise in the treatment of knee osteoarthritis.
There are some points to consider when interpreting the results of this research. First, there was no difference between the CAJM group and CAJM/Ex group. Our expectation was that the CAJM/Ex group would have less damage, but there was no difference. Considering the increased OARSI score in the ACL-T/Ex group, having normal joint movement during walking appears to be more important to than exercising, with a treadmill for example, to prevent cartilage degeneration.
On the other hand, we think it is likely that the exercise intensity utilized in the present study was strong. Moderate exercise has been reported to have a protective effect on articular cartilage, such as reducing inflammatory factor expression and increasing the expression of cartilage matrix synthesis factors. In a rat model of OA, moderate exercise with beneficial effect is defined as treadmill running at a speed of 12 to 18 m/min, for 30 to 60 min/d, 3 to 7 d/wk. However, it is known that high-intensity exercise adversely affects cartilage. Therefore, setting the exercise intensity within an adequate range is important to clarify the relationship between exercise and OA. Studies in mice typically do not involve the use of a treadmill and instead use a rotating cylinder. While the exact protocol depends on the lineage, mice typically perform active exercise in the rotating cylinder at a rate of 15 to 25 m/min per day. Therefore, with reference to the rat model, we set the treadmill speed to 18 m/min, the exercise duration to 30 min/d, and the exercise frequency to 3 d/wk, expecting that this protocol would be within the comfortable range for mice. Therefore, by implementing various exercise protocols in the future, it may be possible to prevent cartilage degeneration by exercise alone. If the appropriate conditions are found, then the CAJM/Ex paradigm may be more effective than CAJM alone.
Second, the therapeutic intervention period was short (4 weeks), and only 1 time point was analyzed. According to the results of immunostaining, we believe that mice in the CAJM/Ex group might also be protected from future articular degeneration, though this is only speculation, as we did not conduct analyses at a later time. However, it is worth noting that Assis et al., 19 who conducted similar investigations in a rat ACL-T model, reported no change in OARSI scores later than 4 weeks. Further research is warranted, where each treatment effect is analyzed in detail.
Third, among the many peripheral factors involved in OA-related cartilage degeneration, our study focused only on IL-1β and MMP-13. Other inflammation and degradation factors including TNF-α, IL-6, MMP-3, and ADAMTS-5 might account for some of the conclusions drawn in this study. Tissue inhibitors of metalloproteinases inhibit MMPs (i.e., substrate degrading factors) and contribute to maintaining cartilage homeostasis. Further investigation of such factors is warranted. Furthermore, although immunostaining is the optimal method for confirming cellular localization, this technique cannot be used to quantify the expression of the factors of interest. Further studies should conduct quantitative and kinetic analysis of the gene expression of inflammatory factors and cartilage matrix degrading factors
Supplemental Material
Supplementary_data_1_1 – Supplemental material for Impact of Controlling Abnormal Joint Movement on the Effectiveness of Subsequent Exercise Intervention in Mouse Models of Early Knee Osteoarthritis
Supplemental material, Supplementary_data_1_1 for Impact of Controlling Abnormal Joint Movement on the Effectiveness of Subsequent Exercise Intervention in Mouse Models of Early Knee Osteoarthritis by Yuichiro Oka, Kenji Murata, Takuma Kano, Kaichi Ozone, Kohei Arakawa, Takanori Kokubun and Naohiko Kanemura in CARTILAGE
Footnotes
Supplemental Material
Author Contributions
All authors approved the final version to be published.
Study design: YO, KM, T Kokubun, and NK.
Data collection, Histological analysis: YO, T Kano, KO, and KA.
Manuscript composition: YO, KM, T Kokubun, and NK.
Acknowledgments and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Animal Research Committee of Saitama Prefectural University (approval number: 29-12).
Animal Welfare
The animals were handled in accordance with the relevant legislation and institutional guidelines for humane animal treatment.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
