Abstract
Objective. Bone marrow stimulation procedures initiate repair by fracturing or drilling subchondral bone at base of cartilaginous defect. Earlier studies have shown that defect location and animal age affect cartilage repair outcome, suggesting a strong influence of structural and biological characteristics of subchondral bone. Here, we analyzed comprehensive biological characteristics of bone marrow progenitor cells (BMPCs) in subchondral bone of young and old rabbit condyle and trochlea. We tested the hypothesis that in vitro biological properties of BMPCs are influenced by location, age of donor and method of their isolation. Design. In vitro biological properties, including cell yield, colony-forming unit fibroblasts (CFU-f), surface marker expression, and differentiation potential were determined. Comparisons were carried out between trochlea versus condyle and epiphyseal versus metaphyseal bone using old (N = 5) and young animal knees (N = 8) to generate collagenase and explant-derived BMPC cultures. Results. CFU-f, cell yield, expression of stem cell markers, and osteogenic differentiation were significantly superior for younger animals. Trochlear subchondral bone yielded the most progenitors with the highest clonogenic potential and cartilaginous matrix expression. Trochlear collagenase-derived BMPCs had higher clonogenic capacity than explant-derived ones. Epiphyseal cells generated a larger chondrogenic pellet mass than metaphyseal-derived BMPCs. All older pellet cultures and one non-responder young rabbit failed to accumulate glycosaminoglycans (GAGs). Conclusion. Taken together, these results suggest that properties intrinsic to subchondral progenitors could significantly influence cartilage repair potential, and could partly explain variability in cartilage repair outcomes using same cartilage repair approach.
Introduction
Articular cartilage present at the ends of long bones possesses very limited regenerative capacity and is frequently repaired using bone marrow stimulation typically leading to formation of fibrocartilaginous repair tissue. Channels created in subchondral bone, provide access to underlying bone marrow stroma rich in BMPCs that are recruited to cartilage defect site. They proliferate and differentiate into a chondrogenic phenotype leading to formation of a repair tissue with varying amounts of hyaline and fibrous cartilage. 1 In addition to mechanical and structural factors; differences in number, stemness, and differentiation potential of BMPCs are expected to influence cartilage repair. These properties can be characterized in vitro using assays for determination of cell yield, colony-forming unit fibroblast (CFU-f) potential, expression of cell surface markers, and multilineage differentiation potential.
Previous studies have reported an influence of the defect location and age on cartilage repair outcome, suggesting a strong influence of biological characteristics of subchondral BMPCs.2-4 A total of 34% to 58% of chondral injuries are observed in condyles while trochlea accounts for 6% to 8% cases. 2 Whereas human trochlear defects are harder to treat compared with condylar defects—likely due to anatomical and mechanical dissimilarities3-5—animal models present varying scenarios. While trochlea demonstrated superior repair potential in rabbit3,4; quality of repair in a sheep model 2 was better in condyle as shown in earlier studies by our group reflecting important species and site dependencies. The primary goal of this study was to verify influence of location on in vitro properties of rabbit BMPCs that might affect in vivo repair outcome. Earlier, our group also showed that drilling to 6 versus 2 mm improves repair outcome in trochlea, 6 possibly since it provides access to the metaphyseal red marrow rich in BMPCs. Therefore, we compared in vitro biological properties of BMPCs in upper and lower regions of trochlea in order to evaluate the influence of the depth of drilling on the repair response in clinical situations. We also explored influence of age on BMPC properties to improve our understanding of underlying causes of poor repair in older individuals. Finally, BMPCs were isolated from stromal spaces and bone lining regions (collagenase digestion) as well as outgrowths from explants since cells participating in bone marrow stimulated cartilage repair originate from both these sites. Human trabecular cells reacted positive for Stro-1 and CD105 and negative for CD34 and CD45 suggesting presence of multipotential cells in bone. 7 Cells remaining in explants most likely migrate out after collagenase treatment and demonstrate potential to form colonies8-10 and form new bone and cartilage in vivo. 11 We optimized the concentration and duration of enzyme treatment for maximum yield and viability since collagenase digestion is a harsh treatment compared with explant culture with a damaging effect on in vitro properties of BMPCs.12,13 Owing to similar cell surface marker profile and trilineage differentiation potential, we expect that collagenase- and explant-derived BMPCs would demonstrate comparable in vitro biological properties.
Through this study, a comprehensive analysis of biological characteristics of BMPCs from young and old rabbit condyle and trochlea was performed to verify influence of location, age, and isolation method on their in vitro biological properties. The specific hypotheses tested were the following: (1) Cell yield, stemness markers, clonogenic, chondrogenic, and osteogenic potential are superior in trochlea compared with condyles. (2) Cell yield, stemness markers, clonogenic, chondrogenic, and osteogenic potential decrease with age. (3) There is no difference in in vitro biological properties of collagenase- and explant-derived BMPSCs.
Materials and Methods
Necropsy
Animal study protocols were approved by University of Montreal Institutional Committee “Comité de déontologie de l’expérimentation sur les animaux.” Female New Zealand White rabbits, young (4 months; n = 8 knees) and retired breeders (12-36 months; n = 5 knees) were used. Rabbits were anaesthetized with xylazine-ketamine and euthanized by an overdose of sodium pentobarbital. Both femurs were exposed, dissected of all soft tissues, and placed in ice-cold Dulbecco’s modified Eagle medium (DMEM)/F12 medium.
Isolation of BMPCs
Reagents were obtained from Sigma-Adrich (Oakville, Ontario, Canada) unless otherwise specified. Under sterile conditions, bone was rinsed with Hanks’ balanced salt solution (HBSS)–penicillin–streptomycin (P-0781). Condylar and trochlear bone segments were obtained using isomet saw ( Fig. 1a-d ). Trochlear segments were separated into trochlea upper (epiphyseal bone) and trochlea lower (metaphyseal bone) separated by growth plate in case of young animals or epiphyseal line in retired breeders ( Fig. 1e ). Following further fragmentation into 4- to 5-mm chips using flat blades ( Fig. 1f ), chips were weighed in order to determine cell yield/wet mass at end of P0. Bone chips were treated with 3 mg/mL collagenase type-XI (C-9697) for 2 hours/37°C/100 rpm. Postdigestion, cell suspension was centrifuged and cells were used to obtain first population of BMPCs–“collagenase-derived.” Digested bone chips were rinsed twice with HBSS to remove residual collagenase and placed in flasks for 4 days to generate second population of cells–“explant-derived.” Colonies of fibroblast-like-cells were cultured in growth medium composed of DMEM/F12 (Gibco, 12500-062) containing 10% fetal bovine serum (FBS) and 1X penicillin–streptomycin solution. Following first medium change at 4 to 5 days to remove dead and nonadherent cells, medium was changed every 2 to 3 days.
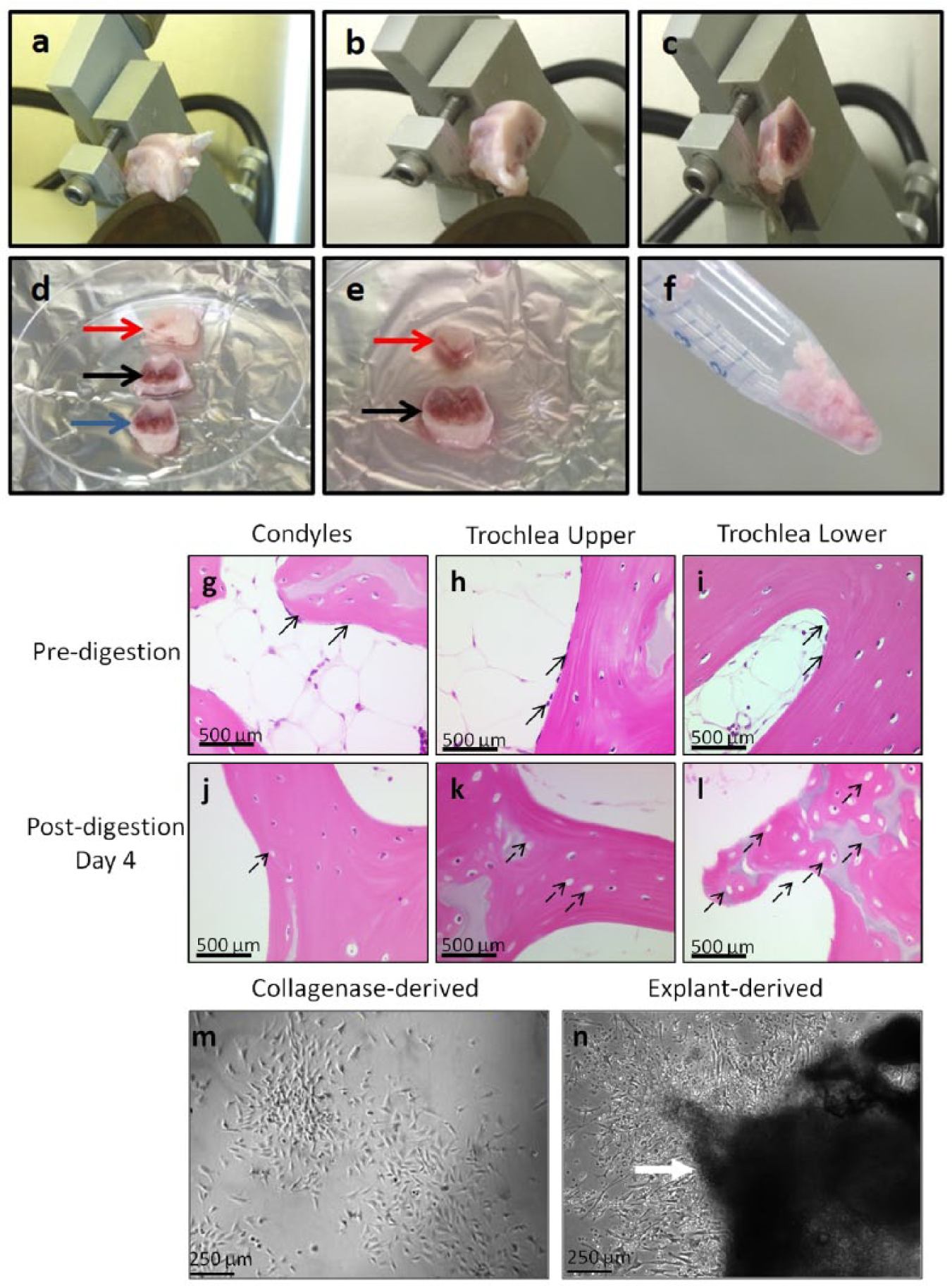
Processing of rabbit femur. Femoral end secured on sample holder of Isomet saw while condylar segment is being sectioned off (
Histology of Bone Chips
Two bone chips were collected at 3 stages—before enzyme treatment (D0, predigested), immediately after enzyme treatment (D0, postdigested), and day 4 of explant culture (D4, postdigested)—for evaluation of collagenase digestion efficiency and migration of cells from explants. They were fixed in 10% normal buffered formalin (Fisher), decalcified in 0.1 N HCl/0.1% glutaraldehyde, embedded in paraffin, sectioned at 5 µm and stained with hematoxylin (Fisher, SH30-500D) and eosin (Surgipath, CI 45380).
Cell Yield
For accurate comparison of initial BMPC population isolated from different locations of bone, all P0 samples were trypsinized at the same time—when at least one of the samples reached 80% to 85% confluency—irrespective of other cultures’ confluency. While sufficient cells were obtained from all 3 locations in all 8 young knees, only 3 of older knees generated substantial cell yield for statistical analysis (n = 8 knees and n = 3 knees for young and old animals, respectively). The cultures were trypsinized at d6-7 (collagenase-derived) and d10-11 (explant-derived) in young and d8-10 (collagenase-derived) and d11-13 (explant-derived) in older animals. Cultures were passaged twice using 0.25% trypsin-EDTA (Invitrogen, 25200-072). Cell viability was determined with Trypan blue exclusion and Countess system (Invitrogen). Cell yield was determined as follows: P0 cell yield (106/g) = viable cell count (106)/ wet weight of bone chips (g).
Flow Cytometry
P1 BMPCs (106/mL) were blocked with 1% solution of BSA-PBS (A-7888) followed by incubation with nonconjugated monoclonal antibodies and goat anti-mouse IgG-FITC. MPC positive antigens used were CD29 (Millipore, MAB1951Z) and CD44 (Cell Marque, 144M-96). Hematopoetic marker CD34 (Cell Marque, 134M-16) and pan-leukocytic marker CD45 (Cell Marque, 145M-97) were used as negative markers. Mouse IgG isotype primary antibody served as negative control. Stained cells were acquired using MoFlo cytometer and data were analyzed using Summit software after appropriate gating to exclude cell debris.
Colony-Forming Unit Fibroblasts
P1 cells were trypsinized, replated as single cells at low density of 100 cells/well, and cultured in complete growth medium for 6 to 7 days. Cultures were stained with 1% (w/v) solution of crystal violet (C-0775) and colonies containing more than 50 cells were counted.
Chondrogenic Differentiation Assay
Chondrogenesis was induced in 3-dimensional high-density pellet cultures of BMPCs using P2 cells. 0.25 × 106 cells (in DMEM/F-12) were centrifuged in 15 mL polypropylene tubes at 300 g/5 minutes to form pellets. Tubes were placed at 37°C/5% CO2 with caps loosened to allow gaseous exchange. Next day, pellets were switched to 0.5 mL chondrogenic medium composed of serum-free high-glucose DMEM (Gibco, 12100-046) supplemented with dexamethasone (100 nM, Cat. No. D-2915), ascorbate (50 µg/mL, A-8960), ITS (1X), sodium pyruvate (100 µg/mL, Gibco), and BSA (1.25 mg/mL, A-7888) for 3 weeks. Ascorbate was added fresh at the time of medium change on alternate days. Control and stimulated pellets were cultured in absence and presence of TGF-βIII (R&D, FB2712091), respectively. Low magnification images of fixed pellets were used to determine their gross structure and diameters (used as an indicator of amount of matrix synthesized) using Northern Eclipse software (Empix Imaging).
Histology of Pellets
Pellets were fixed in 4% paraformaldehyde/0.1 M sodium cacodylate/2.5% w/v cetylpyridinium chloride followed by sucrose infiltration, optimum cutting temperature compound (OCT) embedding, and sectioned at 8 µm using CryoJane tape-transfer cryosectioning system. Sections were stained with Safranin-O (S-2255) and Fast Green (F-7252) to detect glycosaminoglycans (GAGs) as previously described.
14
Collagen type-II (Coll-II) expression was determined by immunostaining. Sections were treated with hyaluronidase (H-3506) and pronase (P-8811) and blocked with goat serum (G-9023) followed by anti-collagen-type II (clone II6B3, DHSB, 1:10) antibody. Following treatment with biotinylated secondary antibody (B-7151) and Vectastain ABC kit (Vector, Cat. No. AK-5000), alkaline phosphate red substrate kit (Vector, SK-5100) was used to develop the stain. Images of Safranin O/Fast Green–stained sections were scored using a 0 to 4 system by 1 blinded observer (AC). A chondrogenesis score of 0 was attributed to pellets that had no Safranin-O staining (panel
Osteogenic Differentiation Assay
P2 cells were trypsinized and replated in 24-well plates and cultured in complete growth medium. Confluent monolayers were stimulated with osteogenic medium comprised of α-MEM (Gibco, 12000-014) supplemented with dexamethasone (100 nM, D-2915), ascorbic acid (
Statistical Analysis
Statistical analysis was performed using SAS Enterprise Guide 5.1 and SAS 9.3. Since several segments were collected from both legs of each rabbit, mixed model was used to account for influence of donor and age (n = 8 young knees and n = 3 or 5 old knees). Fixed effects were location (condyles, upper trochlea, or lower trochlea), age (young or old), isolation method (collagenase- or explants-derived cultures), and treatment (for chondrogenic or osteogenic stimulation only), while donor was a random effect. Data in figures are presented as mean (circle); median (line); box, 25th and 75th percentile; whiskers, box to the most extreme point within 1.5 interquartile range.
Results
BMPCs Can Be Isolated from Different Locations in Rabbit Femur
Bone fragments displayed marrow stroma rich in cells before digestion ( Fig. 1g-i ) following which variable fractions of bone marrow stroma were released into supernatant and marrow spaces became clearer ( Fig. 1j-l ). Bone lining cells seen in predigested samples (arrows in Fig. 1g-i ) were no longer observed in post-digestion samples ( Fig. 1j-l ). Numerous lacunae were observed in bone matrix after 4 days of explant culture (dashed arrows in Fig. 1j-l ). On seeding, BMPCs readily separated from cells of hematopoietic origin owing to their adherence to tissue culture plastic. In both collagenase- and explant-derived cultures, cells adhered as rounded cells, acquired characteristic fibroblastic morphology later and grew as colonies in P0 ( Fig. 1m and n ). No apparent morphological difference was observed in cells from condyles, upper trochlea, and lower trochlea.
Cell Yield, Clonogenic Potential, and Matrix Production Are Highest in Trochlear Segments and Decrease with Age
In young animals, trochlea upper showed higher cell yield versus condyles for both collagenase (P < 0.0001) and explant-derived cultures (P < 0.0001) ( Fig. 2a ). In older animals, cell yield was highest for trochlear upper and lowest for condyles in collagenase-derived cultures (P = 0.0041) ( Fig. 2b ). Cell yield was lower in old versus young animals, significantly for explant-derived cultures (P = 0.028) ( Fig. 2a and b ). Yield for collagenase- and explant-derived BMPCs was found to be comparable in young animals ( Fig. 2a ), while poorer yield was observed for explant-derived cultures in old animals (P = 0.0022) ( Fig. 2b ).
All samples expressed BMPC markers CD29 and CD44 ( Fig. 2c and d ) with no apparent difference between condyles, trochlea upper, and trochlea lower or collagenase- and explant-derived BMPCs. Substantial difference was observed between young and old animals ( Fig. 2c vs. 2d ). Expression of BMPC markers was poor in older animals with a considerable proportion of cells showing no expression. All samples lacked expression of CD34 ( Fig. 2e and f ) and CD45 (data not shown) indicating absence of cells of hematopoietic or leukocytic origin.
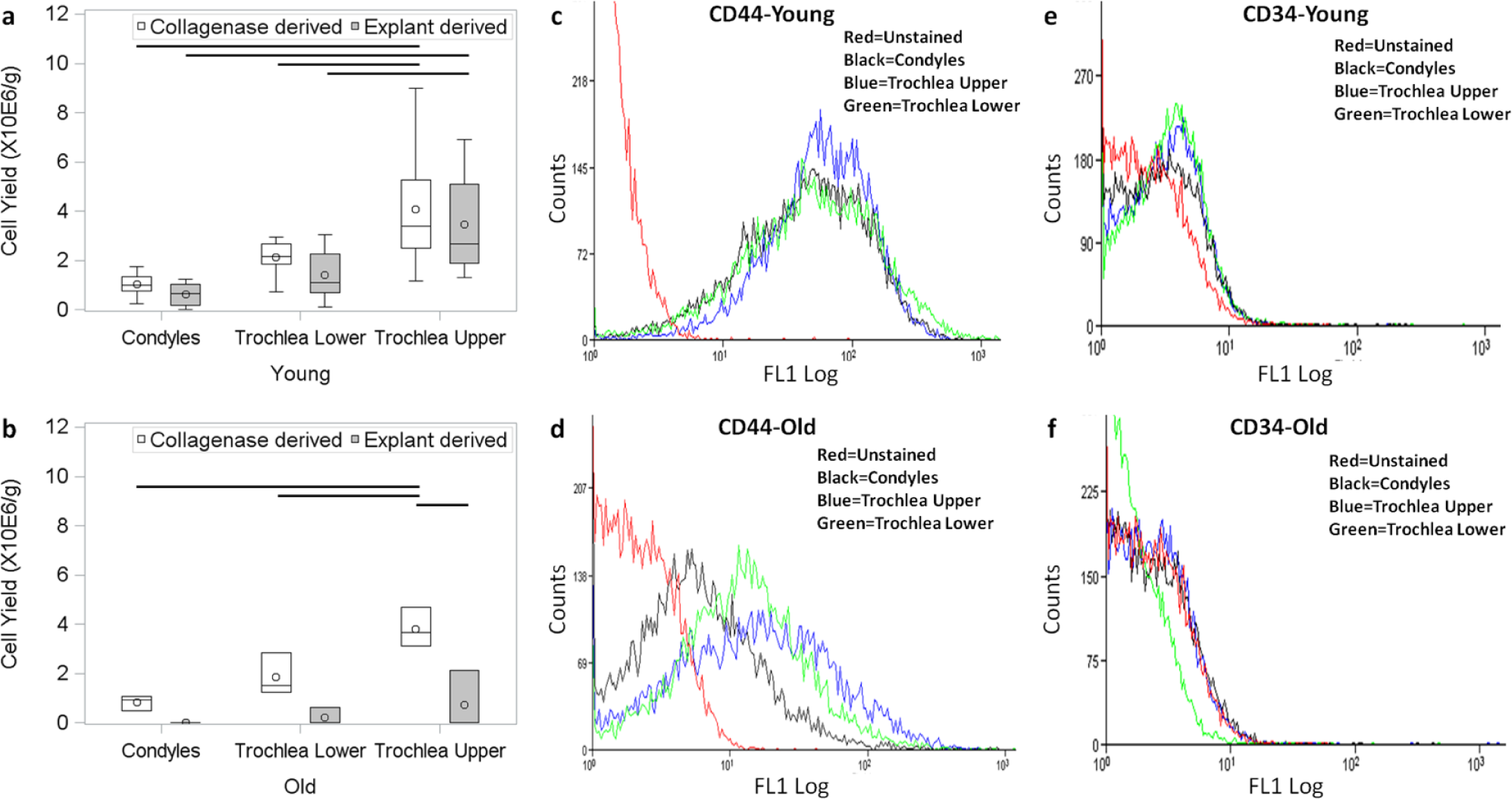
Cell yield observed in young animals (
CFU-f was strongly influenced by location, age and method of isolation ( Fig. 3a-n ). On macroscopic examination, trochlear colonies appeared more numerous and larger ( Fig. 3b and c vs. a and e , f vs. d ). In young animals, both collagenase-derived trochlear segments had higher CFU-f than condylar segments indicating greater clonogenic potential ( Fig. 3m ) (P = 0.02 for trochlea upper and P < 0.0001 for trochlea lower). CFU-f was significantly lower in older animals for both collagenase- (P = 0.02) and explant-derived BMPCs (P = 0.06) ( Fig. 3n ). Finally, explant-derived BMPCs showed inferior clonogenic potential versus collagenase-derived BMPCs in young and old animals indicated by lower CFU-f ( Fig. 3m and n ) (P < 0.0001).
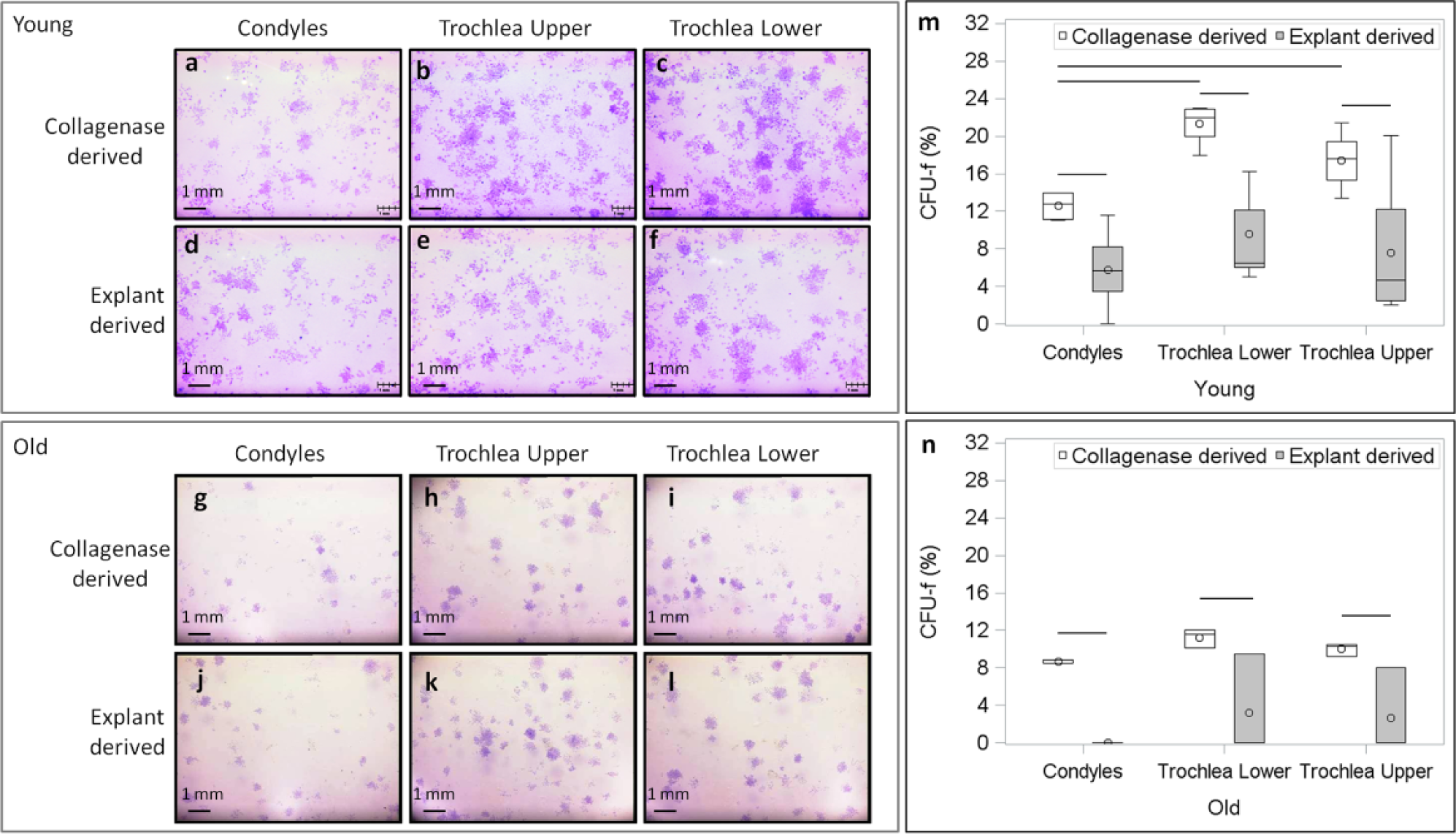
Clonogenic colonies stained with 1% (w/v) crystal violet stain show increased colony formation in the case of young (panels
In young animals, pellets cultured without TGFβ-III were smaller than those cultured in presence of TGFβ-III in both collagenase ( Fig. 4a-c vs. d-f ) and explant-derived cultures (P < 0.0001) (data not shown for explant-derived cultures). Pellets from upper trochlear BMPCs were largest in both collagenase (P = 0.1) and explant-derived cultures (P = 0.03) indicating maximum matrix production ( Fig. 4m and n ). Respective pellets from collagenase- and explant-derived BMPCs from same location were similar in size ( Fig. 4m vs. n ).
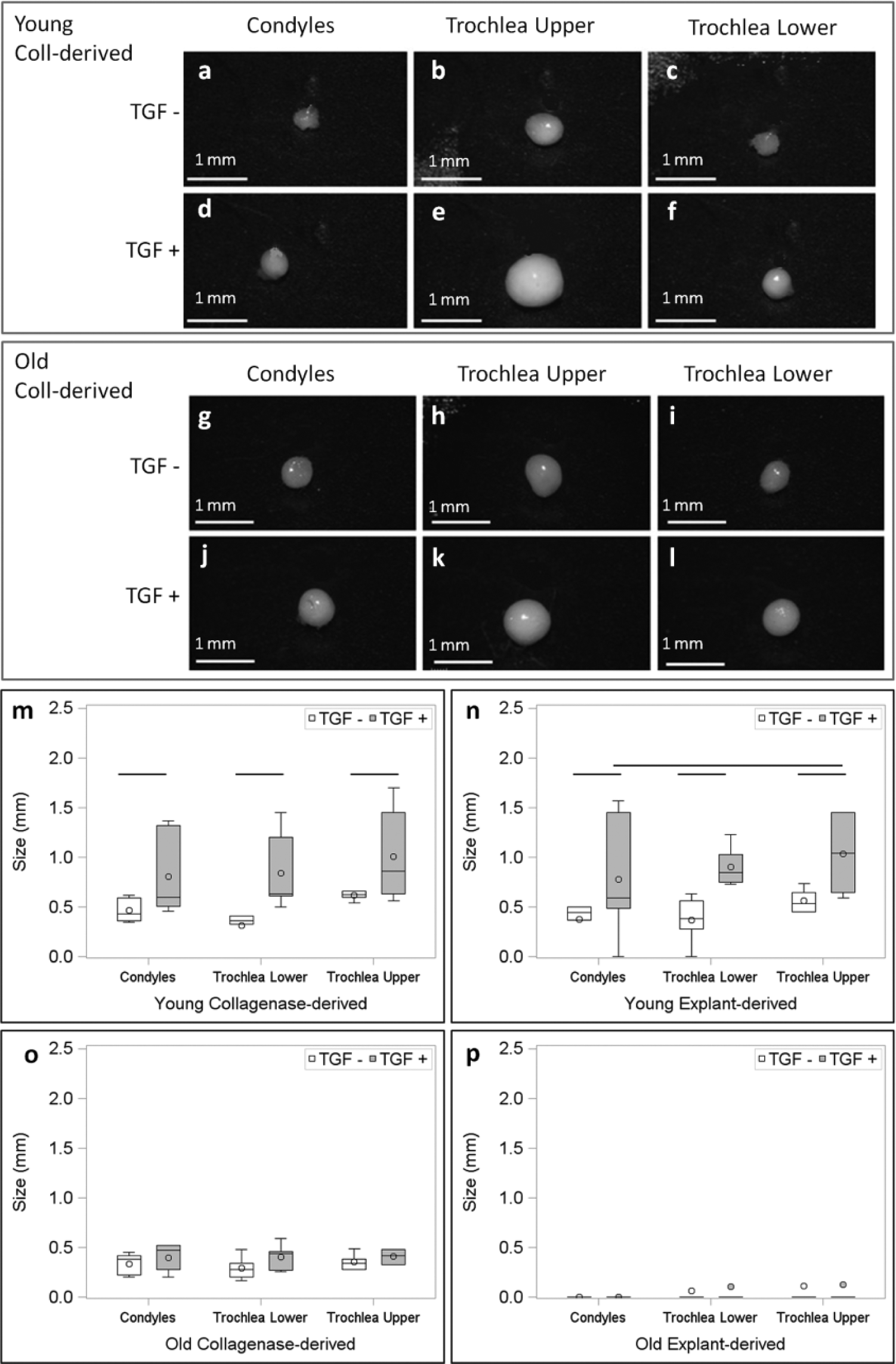
Gross structure of pellets show increase in size on culture in presence versus absence of TGF-βIII in young animals (
Chondrogenic Potential Displays High Interindividual Variability, Is on Average Superior for Trochlear Segments, and Decreases with Age
Pellets cultured without TGF-βIII lacked expression of GAGs ( Fig. 5 ) as well as Coll-II (results not shown). In young animals, enormous variation was observed in quality of matrix produced by pellets cultured in presence of TGF-βIII. In one young animal—a high responder—pellets expressed high levels of GAGs ( Fig. 5a and b ) and Coll-II. Two young animals were labeled medium responders ( Fig. 5c and d ). In another young animal—a low responder—very little chondrogenesis was observed ( Fig. 5e and f ). While chondrogenesis scores reflect this variation and have large spread, on average, both collagenase- and explant-derived BMPCs derived from trochlea upper displayed increased chondrogenic potential ( Fig. 5i and j ). By contrast, outcome in older animals was more consistent ( Fig. 5g and h ). Poor matrix with negligible expression of GAG ( Fig. 5g and h ) and Coll-II and corresponding low chondrogenesis scores ( Fig. 5k and l ) was observed for all samples cultured in presence of TGF-βIII, irrespective of location or method of isolation.
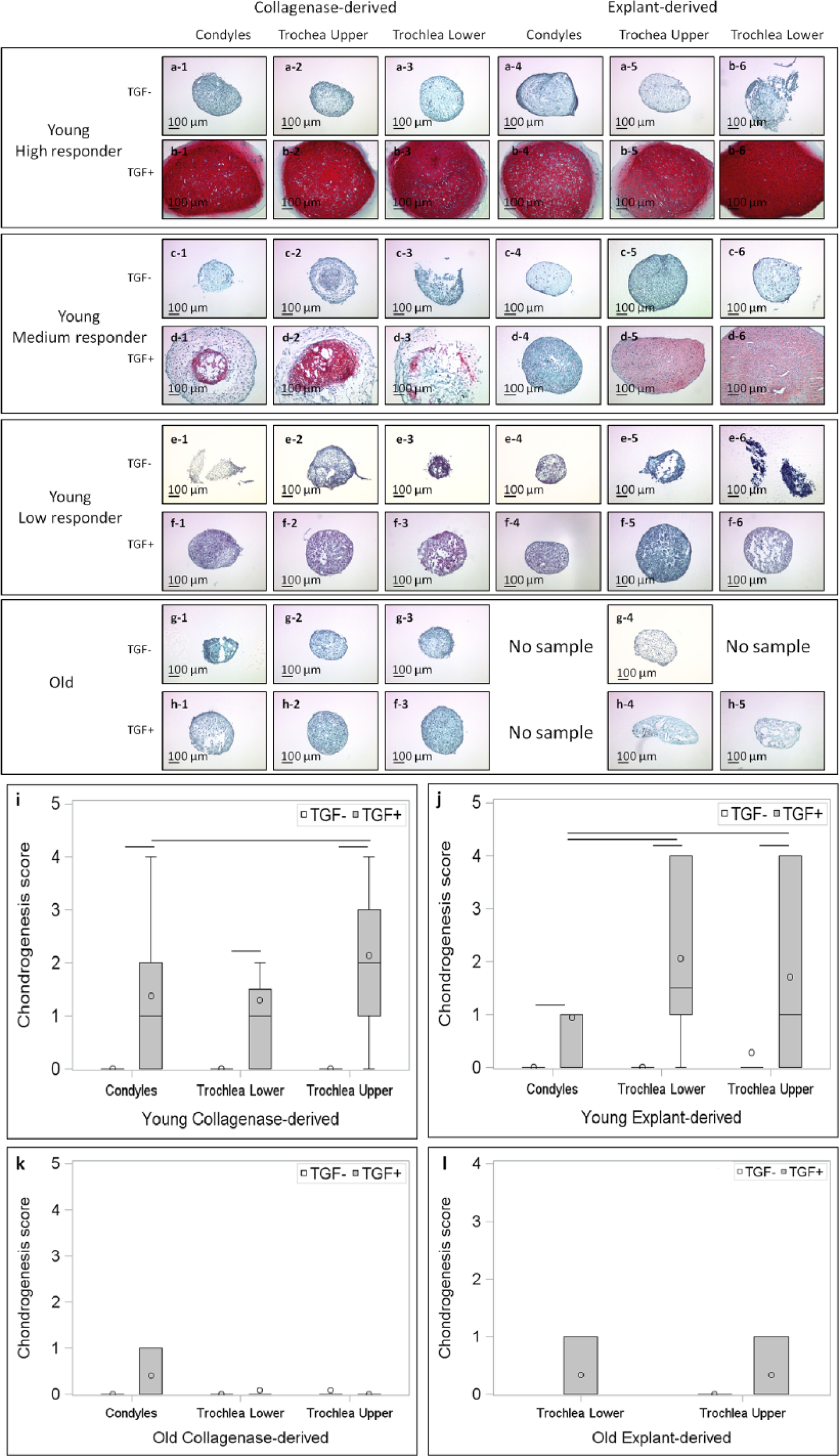
Safranin O/Fast Green staining of pellets derived from collagenase- and explant-derived cultures, showing a high responder (panels
Osteogenic Potential Is Higher in Trochlear Segments and Decreases with Age
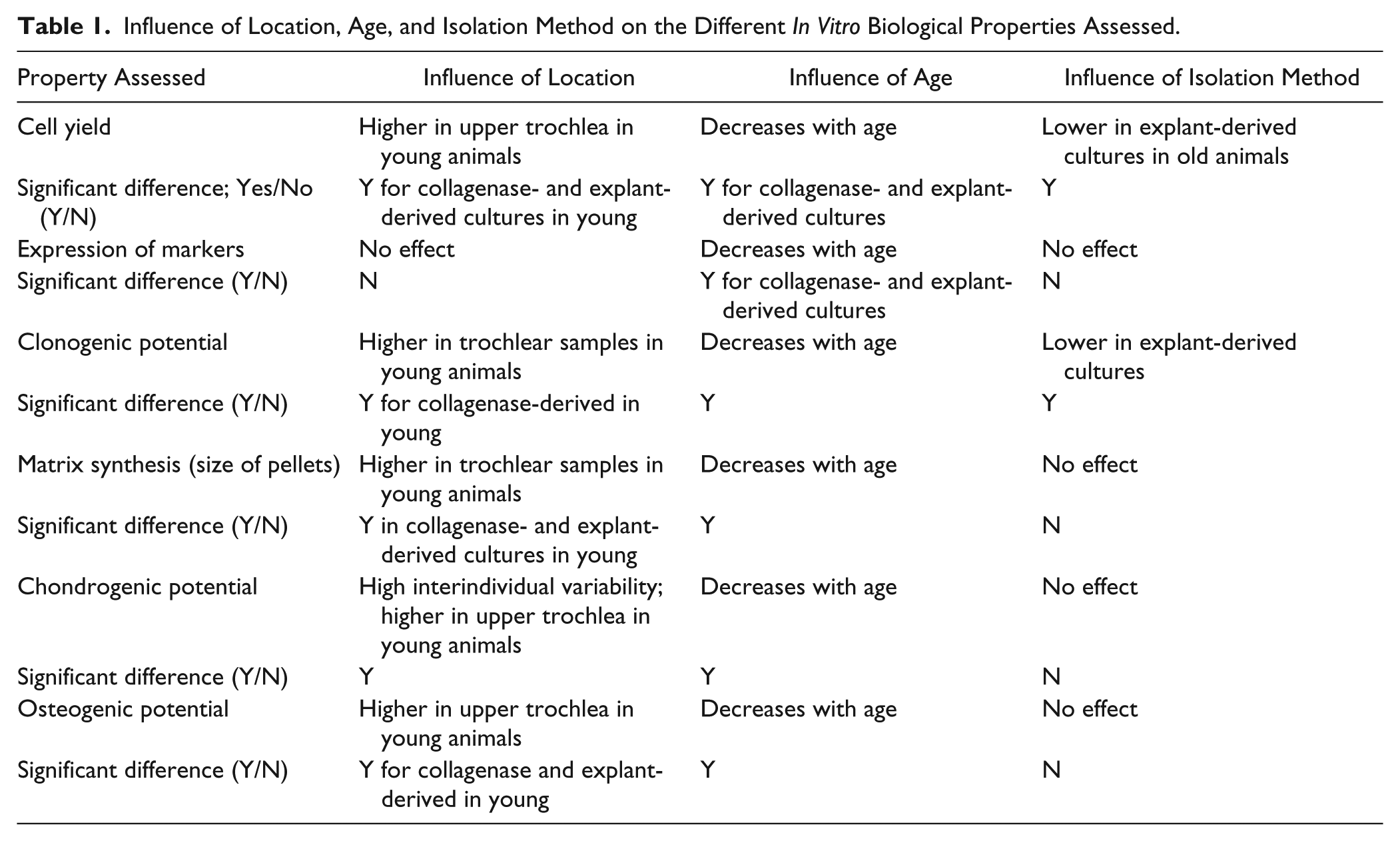
Osteogenic matrix expression was significantly influenced by age, location, and treatment with dexamethasone. In young animals, cultures differentiated into osteogenic phenotype on dexamethasone stimulation and matrix calcification was much stronger in presence of dexamethasone (P < 0.0001) ( Fig. 6a and e vs b-d , and f-h ). Staining observed with AR was superior in trochlea upper for collagenase-derived (P < 0.0001) and explant-derived cultures in young animals (P = 0.0154) ( Fig. 6q and r ), while no apparent difference was observed between collagenase- and explant-derived samples ( Fig. 6q and r ). Osteogenic differentiation was found to be attenuated with increasing age and even when cultured in presence of dexamethasone, a very poor osteogenic matrix was observed in case of old animals ( Fig. 6i-p ). AR extracted from cultures from older animals was much lower in all cases signifying poor osteogenic differentiation and no significant influence of location ( Fig. 6s and t ) or method of isolation ( Fig. 6s vs t ) was observed. Table 1 summarizes the influence of location, age, and isolation method on the different in vitro biological properties.
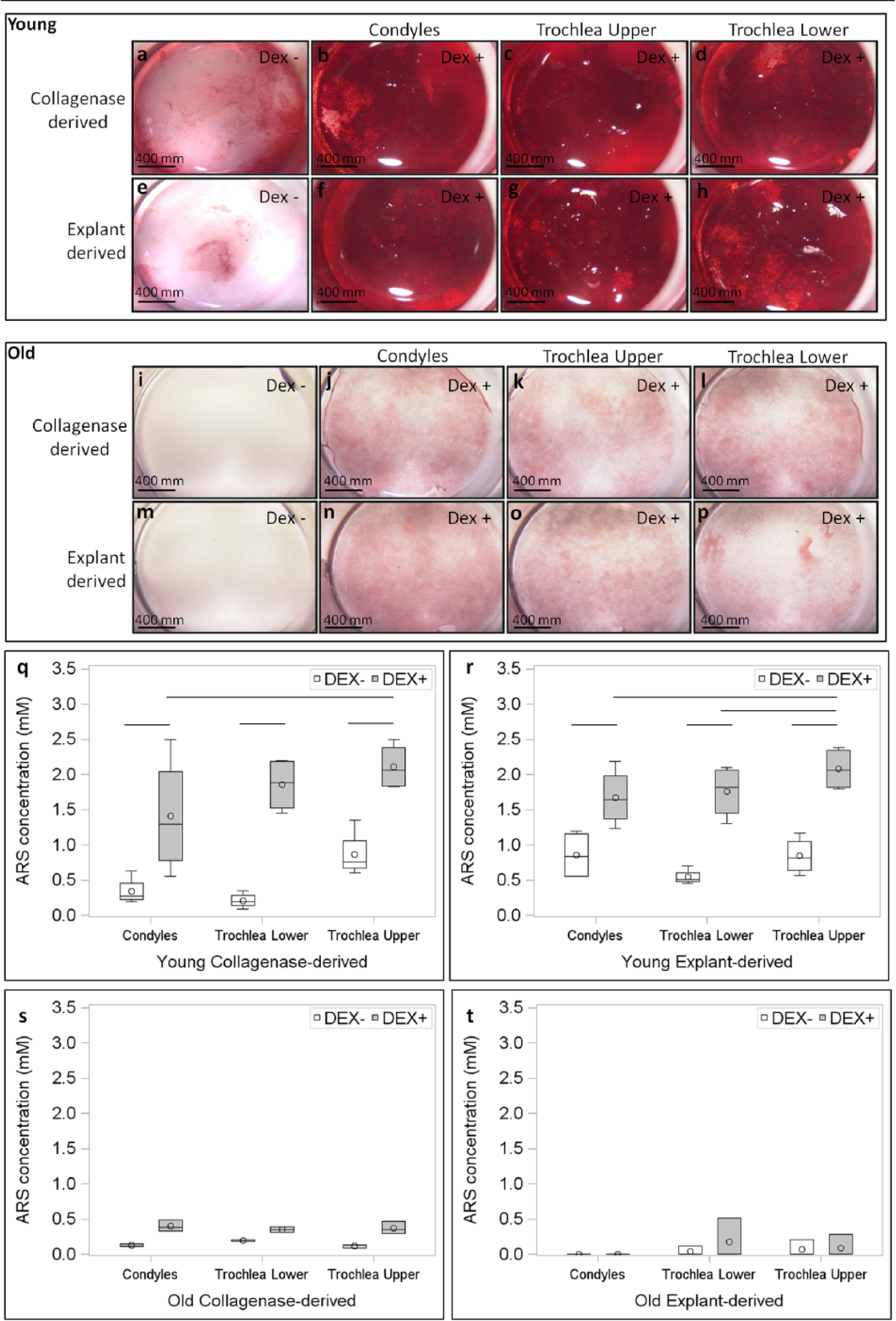
Osteogenic cultures stained with Alizarin Red S (AR) showed increased matrix synthesis in presence (panels
Influence of Location, Age, and Isolation Method on the Different In Vitro Biological Properties Assessed.
Discussion
Clear differences were observed in biological properties of BMPCs from trochlea and condyles, with trochlear cells—more specifically trochlea upper cells—exhibiting increased cell yield, more clonogenic potential and differentiation potency, thus confirming our first hypothesis that location influences chondrogenic potential. Since P0 cell yields reflect in situ difference in initial number of progenitor cells present at these 2 sites, we can conclude that fewer chondrogenic progenitors reside in condyle compartment versus trochlea. Close interaction of densely-packed cells during condensation phase is critical for initiation of chondrogenic differentiation of BMPCs-since high number of cells are needed in initial stages, and a low density BMPC population in condyles could be directly correlated to poor repair outcome. Also, since the number of self-renewing cells possessing clonogenic potential is positively correlated with repair potential, lower CFU-f might reduce in vitro chondrogenic potential of condyles. At molecular level, a difference in the expression of Sox-9 -master regulator of chondrogenic determination of BMPCs 15 or response of BMPCs to TGFβ-III might be yet another factor responsible for poor chondrogenic differentiation with condylar BMPCs. Although MSCs niche is deemed to be dynamic, it is challenging to determine their mobilization and in situ behavior in the absence of methods for their localization in vivo. As is common in in vivo studies, inter-animal variation was also prominent in pellet cultures in young animals. Medium- and low-responders showed significantly improved cartilaginous phenotype in trochlear pellets indicated by higher GAG and collagen type II expression, consistent with our earlier observations of better repair outcome in rabbits.3,4 Alternatively, high-responders consistently generated matrix rich in cartilage components in all samples. However, it is impossible to ascertain if this observation was due to inherent differences or influence of in vitro culture conditions on differentiation potency. For instance, the same number of condylar and trochlear BMPCs were used for pellet culture. In vivo, the number of cells participating in cartilage repair would depend on initial population of BMPCs which are scarcer in condyles, based on our cell yield assay. Moreover, an ideal and identical cocktail of growth factors and other cytokines was provided to BMPCs in vitro, unlike in vivo. Previous studies pointed out that TGF and BMPs released from the underlying subchondral bone influence cell migration and repair tissue formation.16,17 In vivo differences in production, release, and metabolism of these cytokines and responses from condyle vs trochlea would be less pronounced in vitro. MSC populations are known to be heterogeneous mixture of uni-, bi-, and tripotent subpopulations with varying levels of lineage commitment. 18 It is possible that a population isolated from a particular donor is richer in bi- and tripotential progenitors and might have higher chondrogenic potential. This might explain higher chondrogenesis seen in some condylar high-responders since even condylar BMPCs with low yield but relatively higher proportion of chondroprogenitors might have a superior differentiation potential. Finally, a clear influence of location was observed when cells were stimulated for osteogenic differentiation. Although, mineralization appeared comparable on macroscopic examination, quantitative assay indicated a weaker osteogenic phenotype of condylar cells. Because of its role in cartilage repair, better subchondral bone repair in trochlea might positively influence the cartilage repair. It also reemphasizes the superior differential potential of trochlea. Interestingly we did not observe any influence of location on expression of cell surface markers. Although used frequently for characterization, these markers are not specific BMPC markers and do not indicate their levels of self-renewal or differentiation capacity.
In our previous animal studies, cartilage repair outcome was superior when subchondral bone was drilled to 6 mm compared with 2 mm6 motivating us to examine BMPCs isolated from epiphyseal (upper) and metaphyseal (lower) marrow. In this study, we found yield and differentiation potential to be superior in trochlea upper versus lower, leading us to reject our initial assumption that more progenitors occur in the deeper metaphyseal marrow. It is important to consider here that volume of subchondral bone participating in repair in deep drilling is 3 times that of shallow drilling and therefore produces a larger volume of GAG+ callus.6 In young animals, such a large callus could have potential to proliferate towards the articular cavity and produce articular cartilage repair tissue, while in older animals; callus with continued proliferation of senescent cells could lead to an outgrowth of fibroblasts into the articular cavity leading to a fibrous repair. One limitation of this study is that recruitment of progenitor cells in the clinical setting may be different than how cells were isolated here in vitro (collagenase or explant culture).
Increasing age had a profound impact on all the biological properties studied here confirming our second hypothesis that age influences chondrogenic potential was true. Our data supported the earlier findings which report the reduction in differentiation potency of stem cells in older animals. 19 Older animals were characterized by reduced number as well as clonogenic potential in addition to a weaker chondrogenic and osteogenic differentiation potential. CFU-f has been reported to be inversely correlated to age leading to a decrease in differentiation potential in senescent animals. 20 Moreover, frequency of tripotential clones reduces with increasing age-replaced first by bipotential and then unipotential clones—thus reducing differentiation potential. 18 Studies indicate reduction in pellet size with increasing age 21 and poor expression of Col-II accompanied by negligent GAGs deposition in older rats. 22 BMPCs possess very low levels of telomerase-enzyme responsible for maintaining telomere length. 23 In addition, there is a downregulation of genes involved in DNA replication, cell cycle, mitosis, and DNA repair with age. 24 Most importantly, expression of stemness related markers, including Oct-4, Sox-2, and Nanog has also been found to diminish with increasing age. 25 Taken together, the past knowledge combined with the observations in this study may be used in development of approaches to overcome this deleterious effect of age on stem cells and improve the repair outcome in clinical situations.
Our third hypotheses—that biological properties of BMPCs are unaffected by method of isolation—was partially supported, given that explant- and collagenase-derived cultures had similar differentiation potential though there were some striking differences in the clonogenic potential. Age had a more profound impact on the yield of progenitor cells derived from bone since the yield of explant-derived BMPCs was comparable to collagenase-derived cultures in young animal while reduced drastically in older animals. Bone chips from young rabbit still possess growth plate with on-going endochondral ossification. This could influence the type of cells growing out of the explant in young (chondrocytes and osteocytes) versus old (osteocytes only). This heterogeneity might explain the reduced chondrogenic differentiation potential of explant-derived BMPCs in old animals. In addition, decline in progenitor cell population or reduced migration potential of existing progenitors might lead to poor BMPC yield in explants from older animals. Clonogenic potential was found to be particularly depleted in explant-derived cultures which could be a function of both method of isolation and culture conditions. Moreover, since explant-derived BMPCs are believed to originate from dedifferentiation of osteoblast cells, 8 it is possible that population of such cells is very low in the initial passage used for CFU-f assay. The homogeneity of cultures along with an increase in population of undifferentiated MPCs might lead to an improved chondrogenic and osteogenic differentiation potential comparable to collagenase-derived cultures in later passages. Cells recovered from old explants appear to be senescent or to have lost TGF receptor expression or chondrogenic capacity, and may be only capable of generating fibrous tissue. This result could be related to the fact that young bone is undergoing remodeling and therefore probably contains progenitor cells embedded in the collagen type I matrix; inaccessible to collagenase. Moreover, in explant cultures, trace residual collagenase could more readily liberate cells from partly mineralized young bone matrix than heavily mineralized old bone. Along these lines, we speculate that remodeling bone could potentially liberate stem cells into the repair zone. Our findings are also supported by a recent study which reported comparable in vitro biological properties including proliferation and differentiation potential for collagenase- and explant-derived BMPCs isolated from several tissue sources. 26 In summary, despite differences in the initial passages, the origin of cells from explant versus collagenase-derived does not affect the differentiation potential of cultures and therefore is not expected to influence the in vivo cartilage repair outcome.
Conclusion
This study is first of its kind to carry out detailed analysis of biological properties of young and old BMPCs isolated from 2 locations of knee using cells obtained with 2 different isolation methods. Location of BMPCs was found to be an important factor influencing yield, clonogenicity and differentiation potential of BMPCs. A wide variation was observed in chondrogenic potential in younger animals, where some young animals showed the same loss of capacity for proliferation, stemness, and potency as BMPCs isolated from older animals. The mechanisms to explain young nonresponders remain elusive. Finally, biological properties—with the exception of clonogenic potential—were similar for explant- and collagenase-derived cells. Owing to high spontaneous repair potential of young animals and limitations of in vitro models in simulating in vivo conditions, a study that directly correlates the properties of stem cells and repair outcome in an animal model might be the next step in understanding preexisting differences at the 2 locations. Taken together, our data suggest an influence of location as well as age on the in vitro biological performance of BMPCs that could be directly responsible for location and age-dependent repair outcome seen in our previous animal studies. These results enhance our understanding of location and age-dependent cartilage repair in humans and point toward effective approaches to improve repair outcomes, for example, by stimulating cell recruitment from subchondral sites via surgical techniques and biological factors as well as promoting subsequent proliferative and specific differentiation behaviors.
Footnotes
Acknowledgments and Funding
We gratefully acknowledge the excellent technical contributions of Geneviève Picard and Catherine Trudeau. The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Canadian Institutes of Health Research Operating Grant (MOP 115186).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from the University of Montreal Institutional Committee “Comité de déontologie de l’expérimentation sur les animaux” (approval number 12-127).
Animal Welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
