Abstract
Objective
Summarize the biologic effects of Supartz FX for knee osteoarthritis (OA), the first worldwide clinically approved intra-articular (IA) hyaluronic acid (HA) product.
Design
To determine the mechanism of action from preclinical and clinical studies, a literature search was conducted of Supartz FX using academic databases from 1987 to 2016. Articles on Supartz FX that deal with its mechanisms of action were extracted, categorized, and reviewed.
Results
Supartz FX has 2 potential mechanisms of action: (1) biomechanical: IA Supartz FX directly improves the viscoelasticity and lubrication of synovial fluid; (2) physiologic: IA Supartz FX penetrates synovium and cartilage tissues to reach HA receptors on the surface of synoviocytes and chondrocytes. In synovium, suppression of gene expression in inflammatory mediators results in improved endogenous HA production, improved properties of synovial fluid, and reduction in pain. In cartilage, suppression of gene expression of collagenases and aggrecanases suppresses cartilage degeneration.
Conclusion
The net results of basic and clinical studies is that IA Supartz FX provides a more favorable biomechanical and functional environment in the knee joint. Hence, it is not only a lubricant but is also physiologically active. These actions may help explain both short- and long-term improvement in pain and function often achieved from IA Supartz FX in knee OA.
Introduction
Hyaluronic acid (hyaluronan [HA]) is one of the few constitutively produced molecules uniquely preserved across animal species from bacteria to humans. It is ubiquitous throughout the human body but is particularly prominent and important in the joint. Its presence on the joint surface provides pain-free and smooth motion of the articulating surfaces. Among its roles in the diarthrodial joint, HA is a basic structural component of articular cartilage creating the backbone of the large negatively charged proteoglycan aggrecan matrix. This property of the aggrecan allows for the compressibility and durability of articular cartilage.
Based on the general presence and basic functional characteristics, HA has been used in many different applications from dermal treatments to intra-articular (IA) injections. The therapeutic use of IA HA for osteoarthritis (OA) of the knee has steadily increased each year in recent years. Although there is controversy over its benefits, many studies on IA HA for knee OA have demonstrated significant improvement in clinical parameters such as pain and function. Because of the biomechanical and lubricating properties of HA, the US Food and Drug Administration as well as several other national regulatory agencies have approved IA HA of the knee as a medical device. However, there are suggestions that IA HA has additional actions that may not be biomechanical that may be relevant in the joint with OA.
There are several known product differences in IA HAs, for example, molecular weight, viscosity, origin (source). Since it is unknown if they all have the same unique functions, we have elected to summarize the knowledge base of the biologic effects of one of the IA HA products, the first worldwide approved IA HA product Supartz FX (Artz, Artzdispo, Artzal, Supartz, Visco-3; Seikagaku Corporation, Tokyo, Japan) as there is robust basic science research available. We have integrated preclinical and clinical findings in order to improve the understanding of the actions of this specific IA HA, in order to better understand the potential long-term clinical impact of this therapy in patients with mild to moderate knee OA. Similarities and differences of other HA products can be compared by defining the actions of Supartz FX, the first HA product available.
Methods
We conducted a literature search of Supartz FX using databases covering academic articles in English (MEDLINE, EMBASE, BIOSIS, PubMed) and in Japanese (PhaDoMs, JDreamII, Ichusi-Web) with the search terms “Supartz,” “Artz,” “Artzdispo,” “Artzal,” “Osteoartz,” “hyaluronic acid,” “hyaluronate,” and “hyaluronan” published from January 1987 to April 2016. There were 460 articles screened. All articles were abstracted, and Supartz FX data were compiled into a single database.
Then the articles on Supartz FX that deal with these mechanisms of action were extracted from all articles of it. There were 39 articles extracted. Moreover, 5 articles that cover the same biochemical effects or are the expansion of the same experiments of IA Supartz FX were excluded from these 39 articles. Ultimately, 34 articles were reviewed.
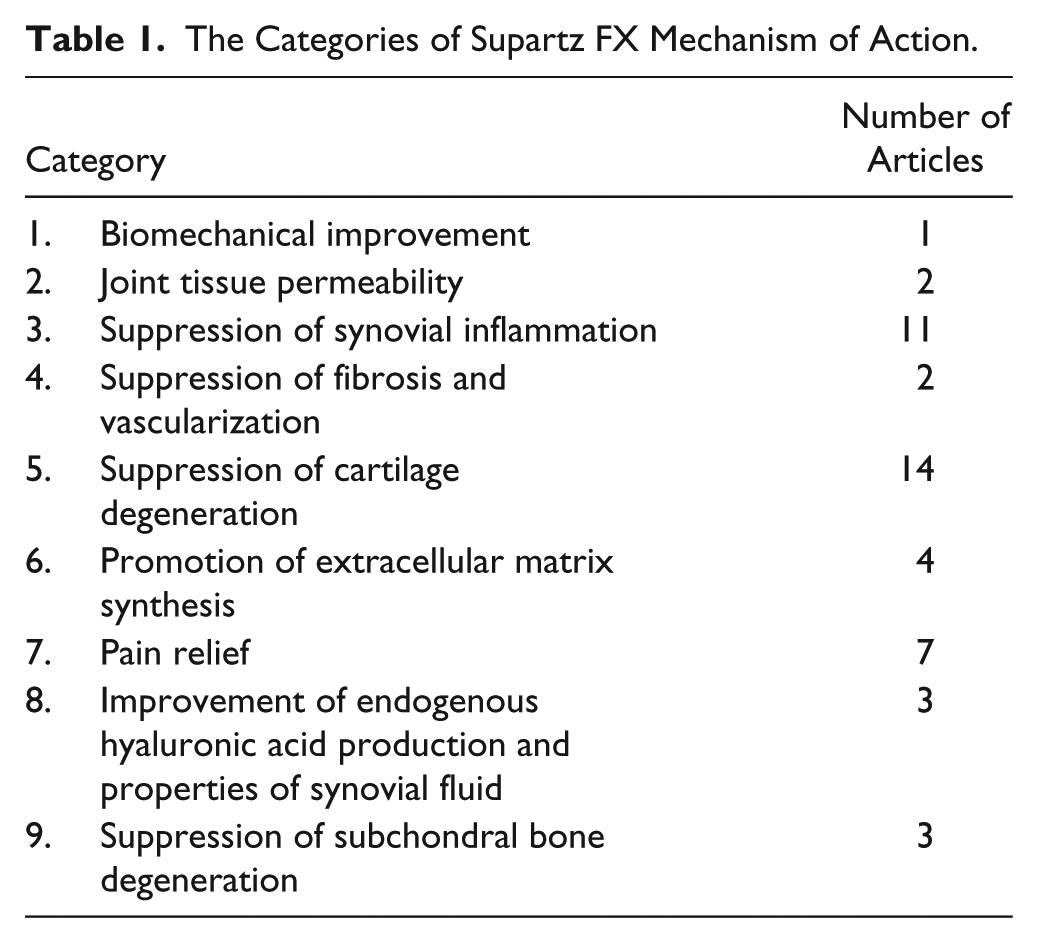
The articles reviewed suggested 9 categories of mechanisms of action. The categories and the numbers of articles that were reviewed are shown in Table 1 . Moreover, the effects of IA Supartz FX on these articles were subdivided into (1) in vivo, (2) in vitro, and (3) clinical (human synovial fluid), and are listed in Supplementary Tables S1 to S9.
The Categories of Supartz FX Mechanism of Action.
Results
Supartz FX
Overview
HA is a nonsulfated glycosaminoglycan (GAG) composed of repeating subunits of D-glucuronic acid and N-acetyl-D-glucosamine ( Fig. 1 ). Supartz FX is a sterile, viscoelastic, nonpyrogenic solution of highly purified, high molecular weight (620-1,170 kDa, average 900 kDa) HA. In 1987, Supartz FX was the world’s first clinically approved IA HA product for the treatment of pain in knee OA. After extraction and purification from chicken combs and dissolved in physiologic saline, each milliliter of Supartz FX contains 10 mg of HA, and its volume is 2.5 mL per syringe. Supartz FX is administered by weekly IA injections for a total of 5 injections. Some patients may experience benefit with 3 weekly injections.
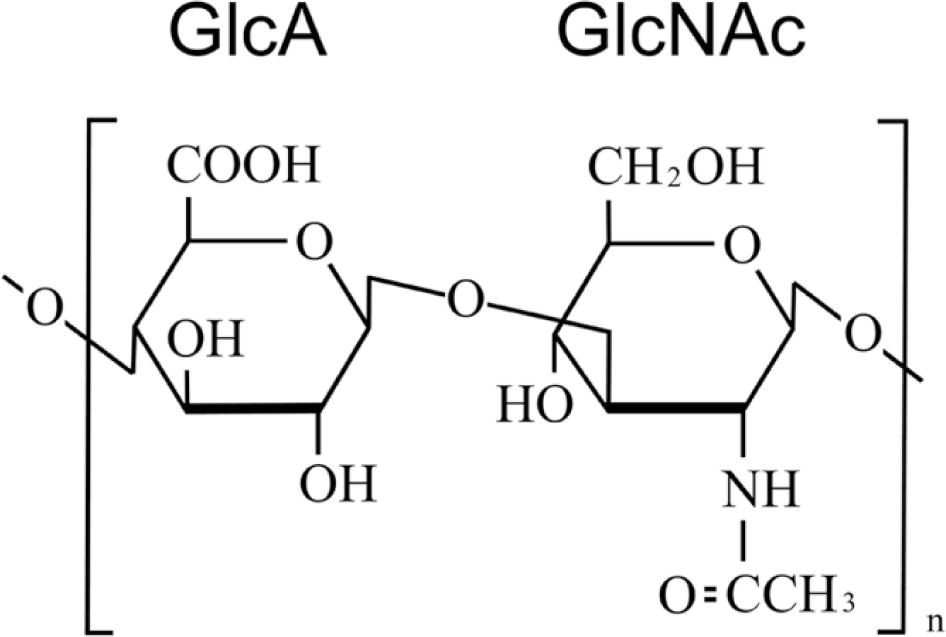
Structure of hyaluronic acid. Hyaluronic acid is a nonsulfated glycosaminoglycan composed of repeating subunits of D-glucuronic acid and N-acetyl-D-glucosamine.
Biomechanics
Preclinical
Equine synovial fluid from acutely injured joints, with and without the addition of Supartz FX, were assayed by a microshear cartilage testing system. The test examined friction of cartilage between human cadaver femoral condyle and tibial plateaus. The addition of Supartz FX to the equine synovial fluid immediately reduced the values of shear strain and friction coefficient 1 (Suppl. Table S1). In a rat strenuous-running knee OA model, IA Supartz FX once a week for 5 weeks maintained the smoothness of the surface of articular cartilage and suppressed degeneration at 6 weeks 17 despite clearance of most Supartz FX from the synovial space within approximately 3 days through normal HA metabolism. 45
The biomechanical effect of Supartz FX appears limited by the relatively short duration of time the majority of Supartz FX is present in the joint.
Permeability
Preclinical
In a guinea pig spontaneous model of knee OA, IA Supartz FX labeled with fluorescein penetrated deeply into synovium. Its distribution area was deeper and wider than that of HA with greater molecular weight (2,300 kDa). 2 Supartz FX was also shown to penetrate deeply into bovine cartilage in culture. 3 Permeability of the fluorescein-labeled HA in these models was also inversely correlated with molecular weight (Suppl. Table S2).
Supartz FX has the ability to permeate synovial and cartilage tissues to reach targeted cells such as synoviocytes and chondrocytes.
Synovial Inflammation
Preclinical
In a dog anterior cruciate ligament (ACL) transection model of knee OA, IA Supartz FX reduced the volume of synovial fluid and decreased synovitis caused by inflammatory cell infiltration typically seen in this model. 4 Studies on synoviocytes in vitro showed that Supartz FX suppressed the gene expression of several pro-inflammatory cytokines (interleukin [IL]-1, IL-6, IL-8, tumor necrosis factor [TNF]-α), inducible nitric oxide synthase (iNOS), cyclooxygenase (COX)-2, collagenases (matrix metalloproteinase [MMP]-1, MMP-3), and aggrecanase (a disintegrin and metalloprotease with thrombospondin motifs [ADAMTS]5).5-8 These effects were blocked by anti-cluster of differentiation (CD)44 and anti-intercellular adhesion molecule (ICAM)-1 antibodies. CD44 and ICAM-1 are HA receptors that are localized on cell surface, and these antibodies interrupt the binding between HA and these HA receptors.5,6,8 In synoviocytes stimulated by IL-1, Supartz FX suppressed the phosphorylation of second messengers c-Jun N-terminal kinase (JNK), p38 mitogen-activated protein (MAP) kinases, and inactivated nuclear factor (NF)-κB7,8 (Suppl. Table S3).
Clinical
Several clinical studies have observed that IA Supartz FX reduced inflammation in patients with knee OA, as indicated by decreased pro-inflammatory cytokines, reactive oxygen species (ROS), and signaling molecules in synovial fluid (IL-6, IL-8, O2−, H2O2, NO, prostaglandin [PG]E2) and reduced synovial fluid volume9-13 (Suppl. Table S3).
Pro-inflammatory cytokines, IL-1, and TNF-α are believed to induce the expression of inflammatory genes in synoviocytes through activation of MAP kinases and NF-κB transcription factor ( Fig. 2 ). In the presence of Supartz FX, however, this signaling cascade is interrupted. The biochemical explanation for Supartz FX’s suppression of synovial inflammation appears to be its binding to CD44 and ICAM-1, thereby suppressing activation of second messengers JNK, p38 MAP kinases, and NF-κB transcription factor, and reducing expression of inflammatory genes ( Fig. 2 ).
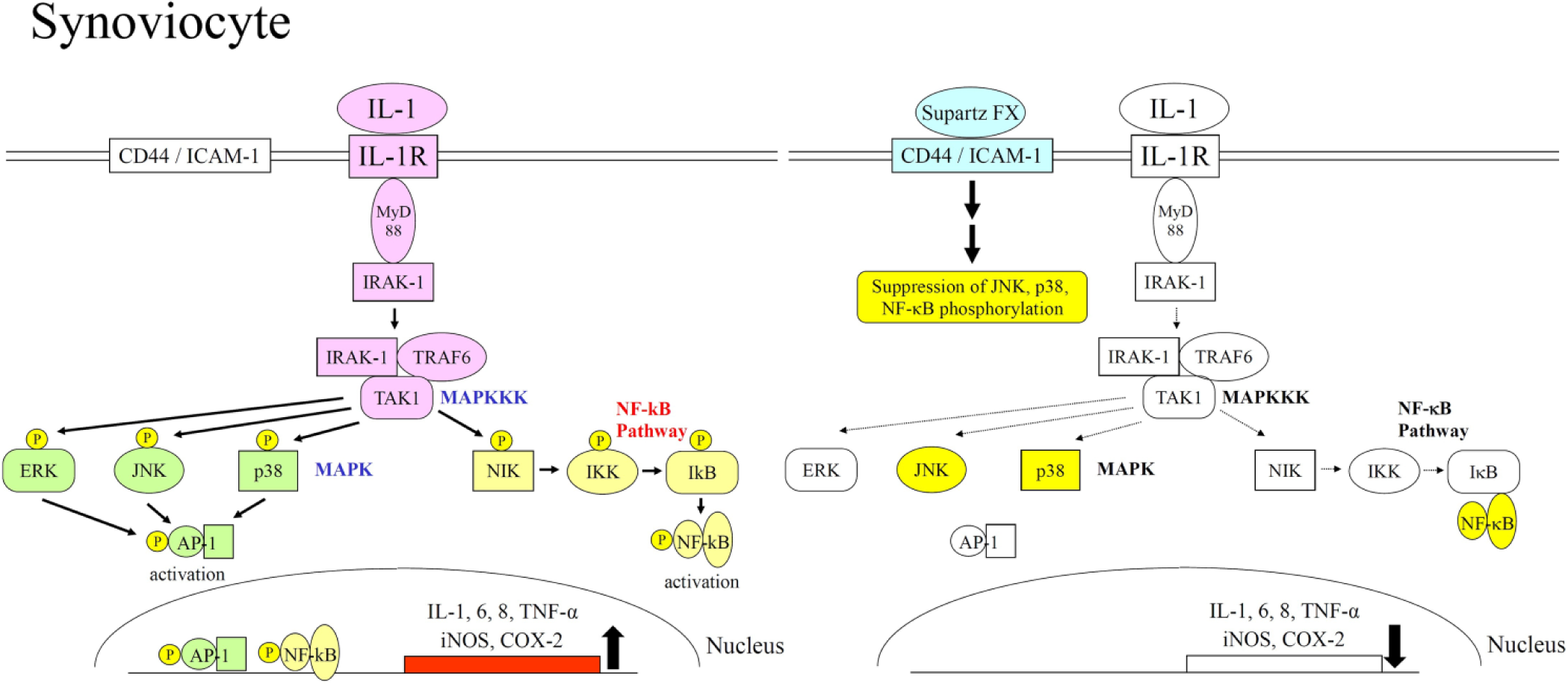
Upregulation of inflammatory gene expressions by IL-1 and the molecular mechanism of its downregulation by Supartz FX in synoviocytes. The binding of IL-1 to its receptor on cell surface activates MAP kinase and NF-κB signal transductions in synoviocytes. These signal transductions induce activation of AP-1 and NF-κB transcription factors, and inflammatory gene expressions are upregulated by these transcription factors. The binding of Supartz FX to CD44 or ICAM-1 suppresses phosphorylation of JNK, p38, and NF-κB. These molecular changes inactivate AP-1 and NF-κB, and inflammatory gene expressions are downregulated.
Fibrosis and Vascularization in Synovium and Infrapatellar Fat Pad
Preclinical
In the mouse transforming growth factor (TGF)-β1 injection plus treadmill running (TTR) model of knee OA, IA Supartz FX suppressed the gene expression of type I, III, and V collagens and prevented fibrosis and vascularization in the synovium. 15 Also, in the rat strenuous-running knee OA model, administration of Supartz FX prevented fibrosis of the infrapatellar fat pad and decreased the number of cells positive for α-smooth muscle actin 16 (Suppl. Table S4).
The mechanism for Supartz FX’s suppression of fibrosis is believed to suppress directly type I, III, and V collagens’ gene expressions and be the result of reduced inflammation.
Cartilage Degeneration
Preclinical
In several animal knee OA models, IA Supartz FX suppressed collagenases (MMP-1, -3, and -13) and aggrecanase (ADAMTS5) gene expressions, prevented chondrocyte apoptosis, decreased the number of Caspase-3 and Fas positive cells, and suppressed the decrease of pyridinoline (a protective, naturally occurring collagen cross-linker) in cartilage.15,17-21 Similar results from in vitro experiments have indicated that Supartz FX suppressed the expression of collagenases (MMP-1, -3, and -13), aggrecanases (ADAMTS4 and 5), and COX-2, and promoted the expression of antioxidant enzymes (heme oxygenase [HO]-1, NAD(P)H:quinone oxidoreductase [NQO]-1, glutathione peroxidase [GPX]-1, catalase, and superoxide dismutase [SOD]1).22-27 Consistent with the findings from synoviocytes, anti-CD44, ICAM-1 antibodies, or siRNA of CD44 blocked these effects in chondrocytes22,24-27 (Suppl. Table S5).
Furuta et al. have shown that Supartz FX induced the gene expression of dual-specificity phosphatase (DUSP)10/mitogen-activated protein kinase phosphatase (MKP)5 MAP kinase phosphatase that dephosphorylates JNK and p38, and inactivated activated protein (AP)-1 transcription factor. Induction of MAP kinase phosphatase by Supartz FX is believed to be the molecular mechanism of MAP kinases inactivation 24 ( Fig. 4 ). Also, Onodera et al. have shown that Supartz FX induced the gene expression of nuclear factor erythroid 2–related factor 2 (Nrf2) transcription factor that induces the gene expression of various antioxidant enzymes (HO-1, NQO-1, GPX-1, catalase, and SOD1). Nrf2 induced by Supartz FX is the key molecule of protective mechanism against ROS in chondrocytes. 26 Also, Chang et al. have shown that Supartz FX induced the gene expression of peroxisome proliferator-activated receptor (PPAR)γ nuclear receptor that blocks the NF-κB pathway and its attendant inflammatory responses. It may be one of molecular mechanisms of NF-κB inactivation.23,37,38
Clinical
Clinical studies have reported that IA Supartz FX reduced the level of MMP-9 and biomarkers associated with cartilage degeneration, namely, keratan sulfate and chondroitin 6-sulfate in synovial fluid14,28 (Suppl. Table S5). As keratan sulfate and chondroitin 6-sulfate are components of the aggrecan molecule, free keratan sulfate and chondroitin 6-sulfate are considered indicators of aggrecan breakdown.35,36
As in synoviocytes, pro-inflammatory cytokines, IL-1, and TNF-α in chondrocytes induce MMP and ADAMTS transcriptions via MAP kinases and NF-κB ( Figs. 3 and 4 ). In IL-1 or TNF-α-stimulated chondrocytes, Supartz FX was shown to attenuate this signaling cascade by suppressing phosphorylation of JNK, p38, and extracellular signal-regulated kinase (ERK) MAP kinases.23-27 The binding of Supartz FX to CD44 and ICAM-1 directly induces MAP kinase phosphatase gene expression, suppresses MAP kinase phosphorylation, inactivates AP-1, and reduces the expression of MMP and ADAMTS in chondrocytes ( Figs. 3 and 4 ). Indirectly, reduction of the expression of MMP and ADAMTS is also due to the result of lower pro-inflammatory cytokine levels from Supartz FX-treated synovium.
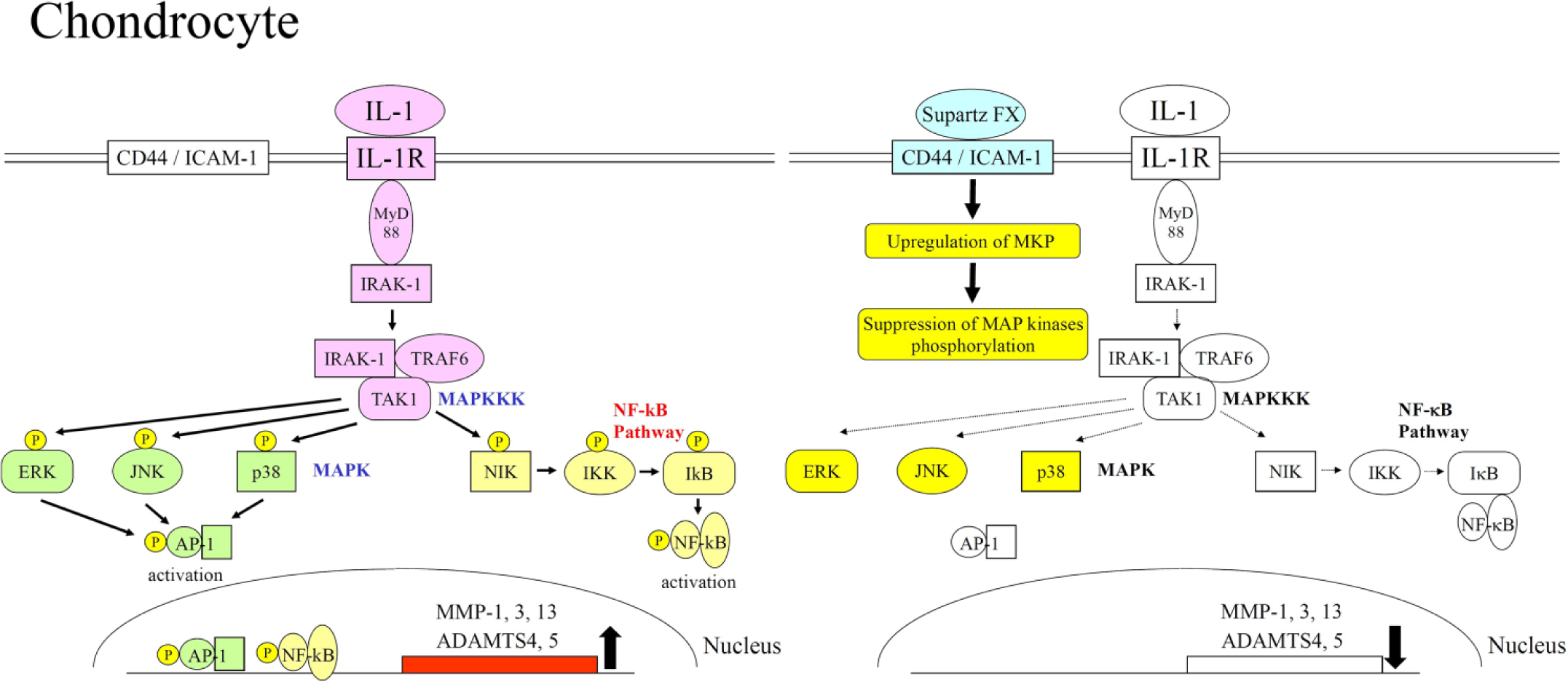
Upregulation of MMP and ADAMTS gene expressions by IL-1 and the molecular mechanism of its downregulation by Supartz FX in chondrocytes. As in synoviocytes, the binding of IL-1 to its receptor activates MAP kinase and NF-κB signal transductions in chondrocytes. These signal transductions induce activation of AP-1 and NF-κB transcription factors, and MMP and ADAMTS gene expressions are upregulated by these transcription factors. The binding of Supartz FX to CD44 or ICAM-1 suppresses phosphorylation of MAP kinases. These molecular changes inactivate AP-1, and MMP and ADAMTS gene expressions are downregulated.
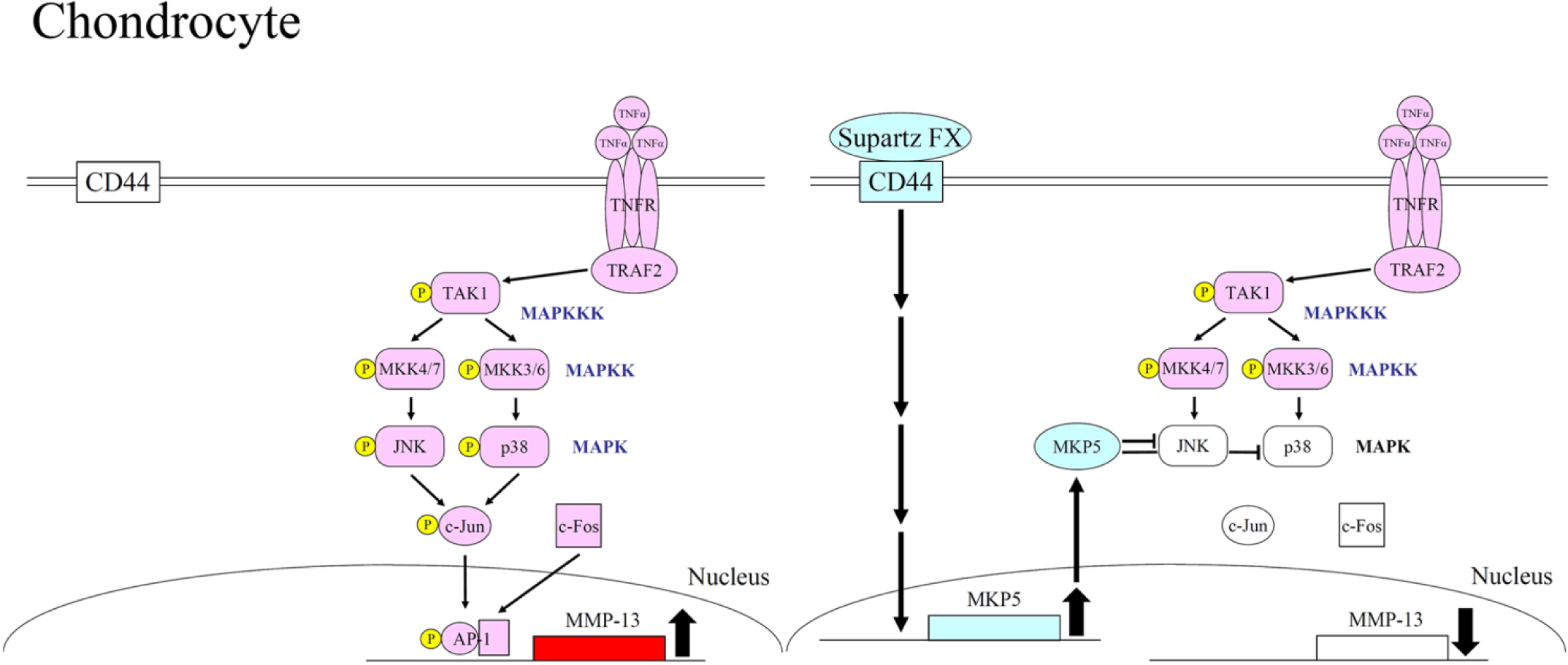
Upregulation of MMP-13 gene expression by TNF-α and the molecular mechanism of its downregulation by Supartz FX in chondrocytes. The binding of TNF-α to its receptor on cell surface activates phosphorylation of JNK and p38 MAP kinases. Activated JNK and p38 MAP kinases phosphorylate c-Jun that is a subunit of AP-1 transcription factor. Phosphorylated c-Jun and c-Fos bind together to form AP-1, and the gene expression of MMP-13 is upregulated by AP-1. The binding of Supartz FX to CD44 induces upregulation of MKP5 MAP kinase phosphatase gene expression. MKP5 dephosphorylates JNK and p38 MAP kinases and inactivates AP-1; therefore, the gene expression of MMP-13 is downregulated.
Extracellular Matrix Synthesis
Preclinical
In the mouse TTR knee OA model, IA Supartz FX promoted the gene expression of type II collagen and aggrecan in cartilage. 15 Supartz FX was also shown to increase proteoglycan synthesis in bovine cartilage ex vivo. 3 Similar result from in vitro experiment have indicated that Supartz FX promoted the gene expression of type II collagen in chondrocytes 26 (Suppl. Table S6).
Clinical
IA Supartz FX increased the level of pCOL II-C in synovial fluid of patients with knee OA 13 (Suppl. Table S6). pCOL II-C is the C-terminal propeptide of type II collagen, which is known to be removed during maturation of the type II collagen molecule. Therefore, it is thought that pCOL II-C is a marker for type II collagen synthesis. 39
The mechanism for Supartz FX’s promotion of extracellular matrix synthesis is believed to directly promote the gene expression of type II collagen and aggrecan in cartilage.
Pain
Preclinical
In a dog ACL transection model of knee OA, IA Supartz FX reduced the level of PGE2 in synovial fluid. 4 PGE2, the product of the COX-2 enzyme, is not only an inflammatory factor but also an enhancer of pain reactions. In a rat ACL transection model of knee OA, IA Supartz FX was shown to reduce synovial-fluid levels of glutamate and aspartate, 29 2 neurotransmitters that stimulate nociceptors. In an IA mono-iodoacetate (MIA) model of knee OA in rats, IA Supartz FX decreased pain-related behavior as well as the number of pain-transmitting cells in the dorsal root ganglion, as determined by marker for calcitonin gene-related peptide (CGRP). 30 Further in vitro experiments indicated that Supartz FX decreased COX-2 mRNA levels in synoviocytes and chondrocytes5,23 (Suppl. Table S7).
Clinical
IA Supartz FX was associated with lower levels of PGE2 and adenosine triphosphate (ATP) in synovial fluid in clinical trials13,31 (Suppl. Table S7). As mentioned above, PGE2 is an enhancer of pain reactions. Extracellular ATP mediates pain signals by stimulating purinergic receptors P2X ion channels and P2Y G-protein coupled receptors on nociceptive nerve fibers.
The reductions of PGE2 and ATP in synovial fluid are likely to have direct effects on patient-reported pain.
Endogenous HA Production and Properties of Synovial Fluid
Preclinical
In the dog ACL transection model of knee OA, IA Supartz FX enhanced production of endogenous HA in synovium 4 (Suppl. Table S8).
Clinical
Some clinical studies have shown that IA Supartz FX increased HA concentration in synovial fluid and improved viscosity of synovial fluid9,14 (Suppl. Table S8).
Knee OA is associated with a decrease in the concentration and molecular weight of endogenous HA in synovial fluid. 40 Reduced HA concentration can be explained by an increase in synovial fluid volume, the result of inflammatory effusion from leaky blood vessels. Lower molecular weight is due to degradation of HA by reactive oxygen species (ROS) and hyaluronidase, and an upregulation of the gene that encodes HA synthase (HAS)3. Unlike the HAS1 and HAS2 isoforms that normally produce long-chain (approximately 4-6 million Da) HA, HAS3 produces only smaller chains. 41 Production of ROS, hyaluronidase, and HAS3 are induced by synovial inflammation.42-44 It is thought that the suppression of synovial inflammation by Supartz FX leads to improvement of endogenous HA production in synovium and improvement of synovial fluid properties.
Subchondral Bone
Preclinical
In a rabbit ACL transection model of knee OA, IA fluorescein-labeled Supartz FX was observed to reach subchondral bone. 32 This result suggests that mechanistic effects of Supartz FX in this deep tissue are possible, if not yet demonstrated. In support of this hypothesis, in vitro experiments have indicated that IA Supartz FX has specific and possibly relevant effects in osteoblasts, including the suppression of gene expression from MMP-13, COX-2, membrane-bound PGE synthase (mPGES)-1 and receptor activator of nuclear factor kappa-B ligand (RANKL),32,33 and in osteoprogenitor cells, including the suppression of gene expression from alkaline phosphatase and runt-related transcription factor (Runx)2 through Ras homolog gene family member (Rho)A activation and Smad1/5/8 inactivation 34 (Suppl. Table S9).
It is unknown whether IA Supartz FX affects subchondral bone clinically. But in vitro experiments suggest a possible role in suppressing the degeneration of subchondral bone.
Discussion
There are several biomechanical and physiological activities that may explain the clinical benefit of Supartz FX in OA of the knee. IA Supartz FX directly improves the viscoelasticity and lubrication of synovial fluid in the short term, as nearly 90% of Supartz FX is cleared from the synovial space within approximately 3 days that is extrapolated from studies on metabolism of IA 14 C-labelled Supartz FX in rabbits. 45 In order to explain the long-lasting impact of Supartz FX beyond 3 days, a biological impact must be present to potentiate its effect beyond the initial peri-injection time period. IA Supartz FX penetrates into synovium and cartilage tissues to reach CD44 and ICAM-1 HA receptors on the surface of synoviocytes and chondrocytes. In the synovium, Supartz FX suppresses the expression of inflammatory factors, including pro-inflammatory cytokines. In cartilage, Supartz FX suppresses the expression of MMP and ADAMTS. These transcriptional changes have cascading effects, reducing pro-inflammatory cytokines levels mitigate cartilage degeneration, and reduced COX-2 production reduces the sensation of pain. In addition, decreased synovial inflammation results in an improvement in endogenous HA (approximately 4-6 million Da) production by synoviocytes; hence, the functional properties of synovial fluid, in terms of HA concentration and molecular weight, are improved. Importantly, despite the relatively short IA half-life of Supartz FX, repeated doses appear to reprogram cellular activity ( Fig. 5 ).
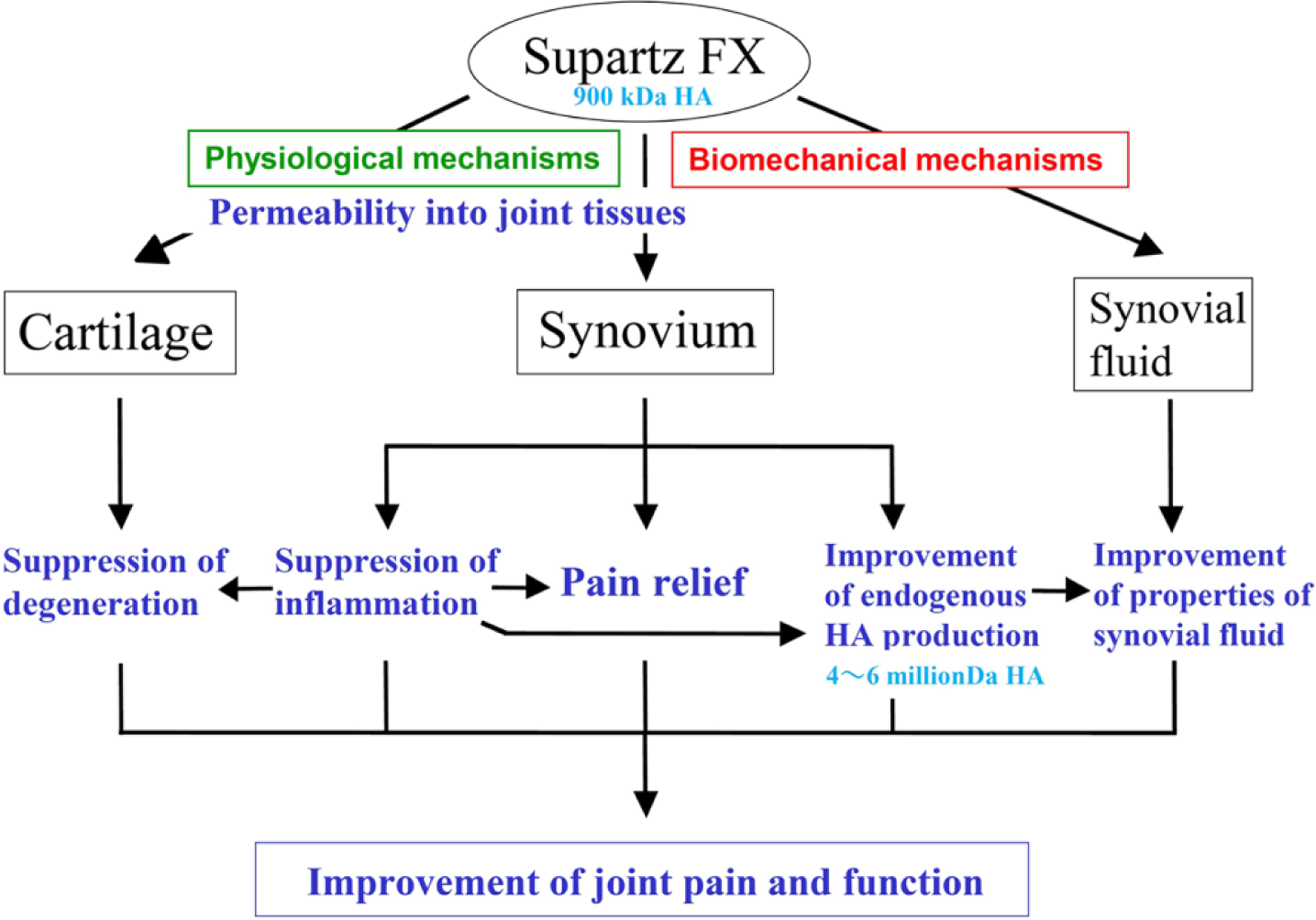
The mechanism of action for Supartz FX in knee OA. Supartz FX potentially has 2 mechanisms of action: (1) biomechanical mechanisms, by which IA Supartz FX directly improves the viscoelasticity and lubrication of synovial fluid; (2) physiological mechanisms, by which Supartz FX penetrates into synovium and cartilage tissues to reach CD44 and ICAM-1 HA receptors on the surface of synoviocytes and chondrocytes. In synovium, Supartz FX suppresses the gene expression of inflammatory factors. The decrease of inflammatory factors leads to pain relief. Moreover, the suppression of synovitis induces improvement of endogenous HA production. It leads to improvement in properties of synovial fluid. In cartilage, Supartz FX suppresses the gene expression of collagenases and aggrecanases, and it leads to suppression of cartilage degeneration. The net result is a more favorable biomechanical environment and better function of the knee joint. This cascade of events represents a virtuous cycle by which IA Supartz FX improves pain and function in knee OA.
However, these mechanisms of action still have many unknown parts. For example, the molecular mechanism of the downstream signaling from HA/CD44 or ICAM-1 in synoviocytes and chondrocytes, and except for PGE2, the intracellular molecular mechanism that result in reduction of pain are to be elucidated. Therefore, further studies are needed to better understand the mechanism of action for Supartz FX in knee OA.
The clinical efficacy of Supartz FX had been shown in an integrated analysis of 5 double-blind, randomized controlled trials (Australia, 50 France, Germany, 51 Sweden, 52 and the United Kingdom). 49 In the integrated analysis of 1,155 patients with knee OA (619 received IA Supartz FX injections, 536 received placebo saline injections), the treatment of Supartz FX significantly improved total Lequésne Algofunctional Index score compared with placebo. This integrated analysis data were used for application to the US Food and Drug Administration. After those 5 randomized controlled trials, 2 randomized, double-blind, noninferiority trials with Supartz FX were conducted.53,54 In both these 2 trials, the clinical efficacy of Supartz FX has been also shown.
In clinical trials of Supartz FX for knee OA, it was often observed that there were nonresponder patient populations the cause of which is unclear. From the mechanism of action perspective, Supartz FX may be not effective in knee OA patients with extreme obesity, severe cartilage degeneration, meniscus injuries, or bone marrow lesions. Further work is needed to determine responders versus nonresponders from IA HA.
Moreover, recent clinical studies have suggested that IA HA may significantly delay major interventions such as arthroplasty. 46 This raises the question as to the clinical relevance of these molecular and cellular findings on pain reduction as well as improving chondrocyte viability. As the impact of these treatments continue to be evaluated, the basic science supports the use of IA HA and should give the clinician confidence in their use in the management of mild and moderate knee OA.
In this review, we described the mechanism of action for IA Supartz FX. For other IA HA products for knee OA, similar effects have generally been shown. Moreland reviewed physiological effects of HA products in 2003. 47 In the review, the effects of HA on the extracellular matrix, on inflammatory mediators, on immune cells, and on cartilage were described. 47 In 2015, Altman et al. also reviewed biomechanical and physiological effects of HA products. 48 In the review, the HA’s effects of chondroprotection, proteoglycan and glycosaminoglycan synthesis, anti-inflammatory, mechanical, subchondral bone, and analgesic were described. 48 The mechanism of action for Supartz FX in knee OA are consistent with similar scientific evidences for other IA HA products’ physiological effects reported in these reviews.
The molecular weight of Supartz FX is 620 to 1,170 kDa (average 900 kDa). From a physiological activity perspective, HA >500 kDa creates inflammatory suppression along with reduction of MMP gene expression. 55 HA oligosaccharides (800-5,000 Da) and low molecular weight HA (24-50 kDa) do not exert such activities and induce inflammation. 55 Therefore, in terms of physiological activity, HA of more than 500 kDa may be considered high molecular weight. 55
Clinically, IA HA products can be classified into relatively low molecular weight, high molecular weight, and cross-linked HA product groups. Supartz FX is categorized into low molecular weight HA product group in the United States.
In low molecular weight HA products, the ratio of physiological mechanisms to biomechanical mechanisms is high. Most of the IA Supartz FX molecules are cleared from the synovial space within approximately 3 days 45 but its permeability into knee joint tissues is higher.2,3 The molecular weight allows for penetration of a larger number of molecules of Supartz FX to reach HA receptors on synoviocytes and chondrocytes.
In high molecular weight HA products, the ratio of biomechanical mechanisms to physiological mechanisms is high. High molecular weight HA products exert physiological mechanisms, but there is limited data permeability into knee joint tissues therefore the impact of size in this group is unclear.
In cross-linked HA products, the ratio of biomechanical mechanisms to physiological mechanisms is the highest, but there are limited data of the change of molecules during metabolic clearance of it; therefore, the impact of cross-linking in this group is also unclear.
There are probably patient populations that may respond to a particular HA product group over others; however, large long-term clinical trials will be needed to clarify and understand this issue.
A strength of this review lies in coordinating the English and translated non-English literature on the mechanism of action of a single HA, Supartz FX. Another strength is in explaining the mechanism of action as a cascade of physiological effects. Namely, Supartz FX (average 900 kDa) penetrates into synovium, reaches HA receptor on the surface of synoviocytes, and suppresses the gene expression of inflammatory factors. The decrease of inflammatory factors leads to pain relief. Moreover, the suppression of synovitis induces improvement of endogenous HA (approximately 4-6 million Da) production, resulting in improvement of properties and functions of synovial fluid. This cascade of events represents a virtuous cycle by which IA Supartz FX reduces pain and improves function in knee OA ( Fig. 5 ). Additionally, and perhaps more important, with penetration into cartilage, it reaches the HA receptor on the surface of chondrocytes, reprogramming their activity and potentially resulting in a decrease in cartilage degeneration ( Fig. 5 ).
This review has limitations. The relative ratio of contribution for clinical improvement between biomechanical mechanisms and physiological mechanisms of IA Supartz FX is unclear. There is no data about the effect of IA Supartz FX for meniscus and meniscal cells in knee OA. In addition, in comparison to clinical improvement of synovial inflammation, there are only a small number of articles that have shown evidence for clinical suppression of cartilage degeneration by treatment with IA Supartz FX. In the future, it is expected that the progress in methodology such as magnetic resonance imaging technology may lead to discovery of scientific data that show clinical suppression of cartilage degeneration is relevant with treatment with IA Supartz FX.
Conclusion
The studies we summarized in this review suggest that IA HA, Supartz FX, is not only a lubricant but also has physiological activity. The physiological mechanisms of action for IA HA, Supartz FX, may help explain the long-lasting effects such as reduction in pain and improvement in function many patients with OA of the knee experience after treatment with IA HA, Supartz FX.
Footnotes
Acknowledgments and Funding
We thank Dr. Christopher Brodie of Bioventus LLC and Mr. Sooyeol Lim of Seikagaku Corporation for reviewing and editing the article. The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RDA is a consultant for Cytori, Dupuy, Ferring, Flexion, Iroko, McNeil, Novartis (GSK), Olatec, Pfizer, Q-Med, Rotta (MEDA), Strategic Science & Technologies and Teva. VD is a paid consultant of Bioventus LLC. JT is an employee of Seikagaku Corporation.
Supplemental Material
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
