Abstract
Objective
To recap the historical journey leading to the first cartilage research article using nuclear magnetic resonance (NMR), published in 1955 by 2 Swedish researchers, Erik Odeblad and Gunnar Lindström.
Design
Extensive Internet search utilizing both English and Swedish websites, and reading the dissertations available at the Royal Institute of Technology (Stockholm, Sweden) and via interlibrary loans at Oakland University (Michigan, USA).
Results
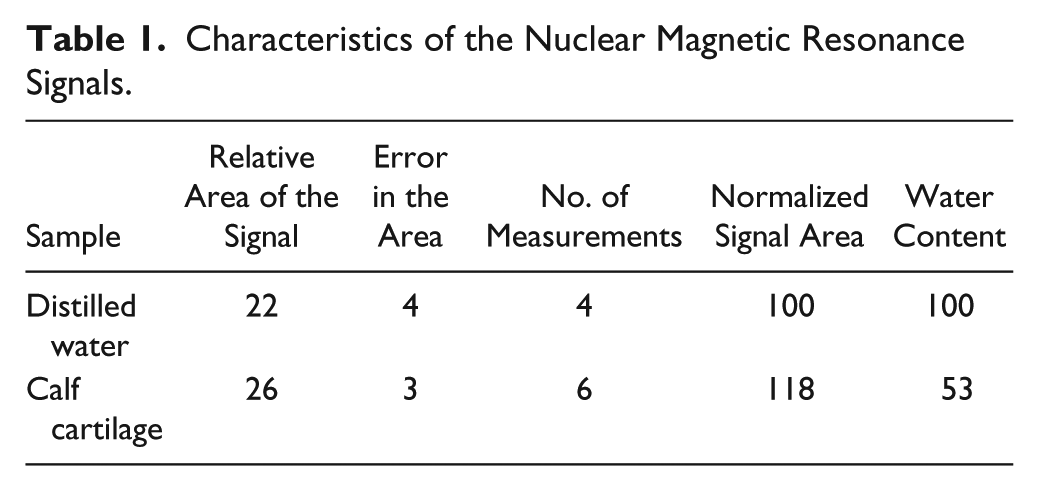
Using a primitive NMR instrument that Lindström built for his graduate research at the Nobel Institute for Physics (Stockholm, Sweden), Odeblad and Lindström studied the characteristics of the NMR signal in calf cartilage. The authors wrote, “In cartilage and fibrous tissue, in which the proton signals probably arise from highly viscous water with short spin-lattice relaxation time, the signals were also larger than would correspond to the water content.” The authors speculated the signal differences between water and biological tissues could be attributed to the absorption and organization of the water molecules to the proteins in the tissue, which was remarkably accurate.
Conclusions
It is quite certain that Odeblad and Lindström published the first biomedical study using NMR in 1955. In this article, cartilage and a number of other biological tissues were examined for the first time using NMR.
Introduction
To find out who actually achieved a first in science and technology is not an easy task; sometimes, the conclusion can be controversial. Even more difficult is, many years after the events, ascertaining the background and journey that led those pioneers to carry out those first studies. The field of study discussed in this historical account is the study of cartilage by magnetic resonance. The technology of magnetic resonance has several acronyms as well as subfields, for example, nuclear magnetic resonance (NMR), which is the original and full name; NMR spectroscopy, which is the spectroscopic version of NMR (without spatial resolution) and commonly used in basic science, in particular, chemistry; NMR imaging, which is the imaging version of NMR and used mainly by nonmedical imaging scientists; magnetic resonance imaging (MRI), which is identical to NMR imaging, mainly used in the medical community; and µMRI or NMR microscopy, which is the high-resolution version of MRI.
The Journey to the First NMR Study of Cartilage
Erik Odeblad and Gunnar Lindström published a paper in 1955 in the journal Acta Radiologica, titled “Some preliminary observations on the proton magnetic resonance in biologic samples,” 1 in which cartilage (and some other biological tissues) was studied using NMR. Their research journey at one time intersected with a group of scientists (Bloch, Hansen, Packard 2 ) who discovered NMR in 1946. Felix Bloch shared the 1952 Nobel Prize in Physics with Edward M. Purcell 3 for their discovery of NMR. Figure 1 shows a photo of Bloch in his laboratory during the 1950s, where the machines, no matter how primitive by today’s standard, were extremely complicated and required lots of skills and experience to properly use.

Felix Bloch in his laboratory in the 1950s.
Erik Odeblad was born in Kristinehamn, Sweden on January 31, 1922. He studied medicine at the Karolinska Institute (Stockholm, Sweden) and received his medical license in 1952. His medical education dissertation was on ovarian phosphate metabolism. In fact, gynecology had been his research interest throughout his entire medical career. Soon after the completion of his medical education, instead of practicing medicine, he became a Rockefeller Foundation Fellow and spent 1953 at the University of California, Berkeley, to learn advanced technologies that he could use in his medical research. One technology he wanted to learn during his fellowship was NMR. Since Sweden is the home for the annual Nobel Prize Award Ceremonies, it is likely that Erik Odeblad had heard about this new physics phenomenon in the previous year during the Nobel Prize Award announcement for NMR.
Stanford University, in California, was where Felix Bloch worked at the time. According to the writings of Dr. Peter A. Rinck
4
(a professor of diagnostic imaging who writes regularly at AuntMinnieEurope.com),
In Stanford, on the other side of San Francisco Bay, he (Erik Odeblad) met Felix Bloch. Odeblad asked him whether he could use his NMR spectrometer to study human samples, but Bloch’s response was negative. He made it clear that NMR was a tool for physicists, not for research into physiology, medicine, or biology.
Given the tools and equipment available for NMR at the time ( Figure 1 ), such refusal was understandable. Without extensive training in experimental physics and electronics, no one would be able to operate such a primitive but complex system. So, Erik Odeblad did not accomplish his first goal during his fellowship.
Now we need to go back further for nearly 20 years, to 1936, when the Swedish Government appointed Dr. Manne Siegbahn (1886-1978) as a professor of experimental physics and the director of a new physics institute. The English name of the new institute was the Nobel Institute for Physics. Dr. Siegbahn received a Nobel Prize in Physics in 1924 for his research on x-ray spectroscopy. In 1937, Dr. Siegbahn and his institute moved into a new building in Stockholm. 5 Much of the research in the institute was in the field of nuclear physics with the use of particle accelerators and cyclotrons. Dr. Siegbahn had a son named Kai Siegbahn (1918-2007), who was also a physicist working at the same institute for some time and who also received a Nobel Prize in Physics in 1981 for his research in high-resolution electron spectroscopy (commonly called x-ray photoelectron spectroscopy in modern literature). The trajectories of both Siegbahns intertwined with these of Erik Odeblad and Gunnar Lindström. Siegbahn senior was the Director of the Nobel Institute for Physics where Gunnar Lindström worked as a physicist; Dr. Manne Siegbahn was also the supervisor for Erik Odeblad’s PhD dissertation on the topic of NMR research in 1966. Siegbahn junior had considerable input into the dissertation work of both Lindström and Odeblad; Dr. Kai Siegbahn was also the Chair and Professor of the Physics Department at University of Uppsala (Sweden) when Odeblad received his PhD in physics in 1966.
Gunnar Lindström (1918-1990) 6 studied at Chalmers University of Technology, where he graduated in 1943. He worked from 1945 to 1954 at the Nobel Institute for Physics. During his time at the institute, he completed a PhD dissertation in physics in 1952, titled “Nuclear Resonance Absorption Applied to Precise Measurements of Nuclear Magnetic Moments and the Establishment of an Absolute Energy Scale in β-Spectroscopy.” Figure 2 shows the NMR machine used in his research. Apparently, the NMR machine that Lindström built by himself, for his PhD dissertation, had an incredibly homogeneous magnet at that time, which even allowed him to measure the proton chemical shifts of mineral oil. (It is commonly accepted that the work of Arnold, Dharmatti, and Packard 7 in 1951 demonstrated the first proton chemical shift in ethanol by NMR.)

The nuclear magnetic resonance (NMR) instrument used in Lindström’s dissertation research.
Having been refused access to the Stanford NMR machine by Bloch, Erik Odeblad returned to Sweden in 1954 without getting any NMR experiments done during his fellowship. However, he soon must have come to know of Gunnar Lindström, with whom he started to study biological tissues using the NMR machine that Lindström had built for his dissertation work. (Note that there was no commercial NMR machine for sale at the time.) In December 16, 1954, Odeblad and Lindström submitted their first NMR research for publication. 1
The Erik Odeblad and Gunnar Lindström 1955 Article
The early 1950s NMR machines had no fast Fourier transform (the modern version of which was invented 10 years later in 1966 by Cooley and Tukey). The experiment in these early machines would run by slowly sweeping the magnetic field to observe the resonance peaks. Two proton signals would appear for each specimen under the sweeping magnetic field, as shown in the article. 1 Studies of a number of biological tissues were reported in this article, including liver, muscle, fat, corpus vitreum, fibrous tissue, tendon, and cartilage. One reached a conclusion by comparing the signal of pure water with the signal of a nonwater sample. A number of differences can be found between the water signal and the tissue signal. For example, the authors wrote that “The proton signals in yeast were lower and somewhat broader than in pure water.”
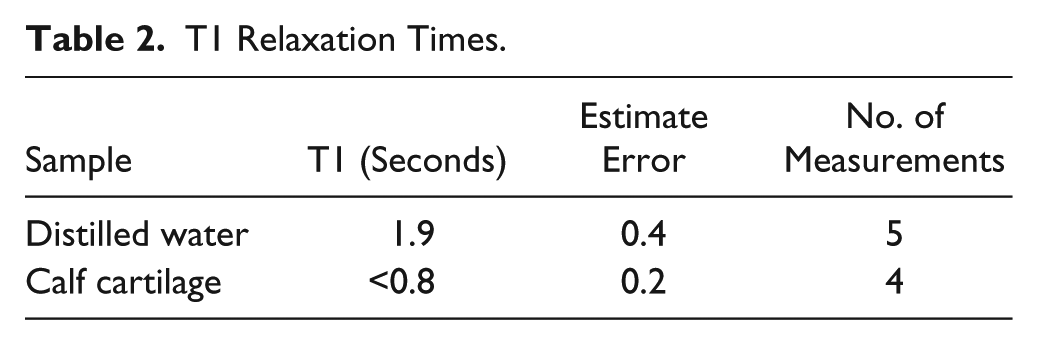
Tables 1 and 2 contain, respectively, the measurement of the proton signals and the T1 relaxation times in cartilage. The authors stated, “In cartilage and fibrous tissue, in which the proton signals probably arise from highly viscous water with short spin-lattice relaxation time, the signals were also larger than would correspond to the water content.” The authors speculated that the signal differences between water and biological tissues could be attributed to the absorption and organization of the water molecules to the proteins in the tissue, which was remarkably accurate.
Characteristics of the Nuclear Magnetic Resonance Signals.
T1 Relaxation Times.
Erik Odeblad’s 1966 PhD Dissertation
Apparently, Gunnar Lindström left academia for industry in 1954, shortly after the completion of these experiments. It is unclear if there were any additional NMR studies of cartilage by Dr. Odeblad. Keep in mind that Dr. Odeblad’s research interest was always in reproductive biology. However, Odeblad must have gained sufficient knowledge in building the NMR machine during his collaboration with Dr. Lindström. In addition to using Gunnar Lindström’s instrument, he started to build his own NMR system in the hospital he worked for as a physician (the Isotope Laboratory, Department of Gynecology, Sabbatsberg Hospital Stockholm, Sweden). In fact, throughout his time, Odeblad built 3 NMR machines, with magnetic fields of 0.4 T, 1.1 T, and 3.9 T. 8
In 1966, Erik Odeblad received his PhD in physics under the supervision of Dr. Manne Siegbahn from the University of Uppsala (Sweden), where Dr. Kai Siegbahn was the Professor and Chair (the same position that his father Dr. Manne Siegbahn had in the past). Erik Odeblad’s PhD dissertation can still be found in the Physics Department of the Royal Institute of Technology (KTH), Sweden. His PhD thesis, titled “Micro-NMR in High Field Permanent Magnetic Fields—Theoretical and Experimental Investigations with an Application to the Secretions from Single Glandular Units in the Human Uterine Cervix.” The dissertation has 188 pages, containing a large portion in NMR instrumentation (chapters 1 to 12, pp. 12-119) and a small portion in biological experiments (chapters 13 to 15, pp 120-154). In the preface, Odeblad acknowledged and thanked the initial contributions of Gunnar Lindström in 1954. Figure 3 shows the magnet of his NMR machine (from his PhD dissertation). Note that his new magnet looked very different from the magnet in Gunnar Lindström’s instrument shown in Figure 2 .

The nuclear magnetic resonance (NMR) magnet in Erik Odeblad’s PhD dissertation.
Final Remarks
Gunnar Lindström left the Nobel Institute for Physics in 1954 to lead a technical division at SAAB, never realizing the historical significance of his work with Erik Odeblad in biomedical NMR and MRI. Lindström had a remarkable career in both academia (becoming a docent of nuclear physics and later a professor of electronic metrology at KTH in Stockholm) and industry (working with electronics and computer design, and nuclear power plant management). In particular, Lindström had a long career at SAAB, retired in 1974 as the Vice President of Saab-Scania, and was counted as the DataSaab’s founder. There is actually a short movie online 9 of Gunnar Lindström describing the development of computers, in Swedish, of course.
Erik Odeblad had an incredible career—some might argue, 2 careers in 1 life. He was a gynecologist and interested in reproductive biology and medicine throughout his entire medical career. At the same time, he was the professor of medical biophysics at the University of Umeå from 1966 to 1988. His passion in research was using NMR to study reproductive biology. One of the last searchable news on the Internet is about Odeblad receiving the 2012 European Magnetic Resonance Award. Figure 4 is a photo of Erik Odeblad during the award ceremony in 2012.

Erik Odeblad during the European Magnetic Resonance Award ceremony in 2012.
In conclusion, it is quite certain that Odeblad and Lindström published the first biomedical study involving the use of NMR. Here, cartilage and a number of other biological tissues were examined for the first time using NMR. This conclusion is further supported by the credit given by Paul Lauterbur in his 2003 Nobel lecture, for his invention of MRI that earned him and Peter Mansfield the Nobel Prize in Physiology or Medicine. Lauterbur said, “In an early predecessor to MRI, …, and actual medical measurements were started when Erich Odeblad, a Swedish M.D., constructed apparatus and devised methods to study very small quantities of human secretions for medical purposes.” 10 Of course, more details could be further discovered in this historical journey, for example, how Erik Odeblad came to be convinced that this seemingly obscure physics phenomenon could be used for his medical research, how he came to know Gunnar Lindström and persuaded him to measure these biological samples using NMR, and what kind of working dynamics was in their first biomedical research using NMR.
Footnotes
Appendix
Acknowledgments and Funding
The authors thank Ms. Carol Searight (Oakland University) and Miss Aimee Xia for editorial comments on the manuscript. Yang Xia has been supported by the National Institutes of Health (USA) for his cartilage research since 1999 (four R01 grants AR045172 (1999-2012), AR052353 (2008-2016), AR069047 (2016-2021) from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS); Principal Investigator: Xia)
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article
