Abstract
The aims of this review article are (a) to describe the principles of morphologic and compositional magnetic resonance imaging (MRI) techniques relevant for the imaging of knee cartilage repair surgery and their application to longitudinal studies and (b) to illustrate the clinical relevance of pre- and postsurgical MRI with correlation to intraoperative images. First, MRI sequences that can be applied for imaging of cartilage repair tissue in the knee are described, focusing on comparison of 2D and 3D fast spin echo and gradient recalled echo sequences. Imaging features of cartilage repair tissue are then discussed, including conventional (morphologic) MRI and compositional MRI techniques. More specifically, imaging techniques for specific cartilage repair surgery techniques as described above, as well as MRI-based semiquantitative scoring systems for the knee cartilage repair tissue—MR Observation of Cartilage Repair Tissue and Cartilage Repair OA Knee Score—are explained. Then, currently available surgical techniques are reviewed, including marrow stimulation, osteochondral autograft, osteochondral allograft, particulate cartilage allograft, autologous chondrocyte implantation, and others. Finally, ongoing research efforts and future direction of cartilage repair tissue imaging are discussed.
Introduction
The first autologous chondrocyte transplantation surgery was described by Brittberg and colleagues in 1994. 1 Since then, knee cartilage repair or restoration surgery has evolved rapidly thanks to a number of factors including improvement of presurgical assessment, imaging techniques, increased availability of matrix products including both fresh and frozen allografts, and focused research on the clinical outcomes including all cartilage surgical techniques. 2 Cartilage repair or restoration surgery is aimed to alleviate patient symptoms, to promote cartilage healing, and to prevent or delay the onset of osteoarthritis. McAdams and colleagues published a review article on articular cartilage injuries in athletes and the available surgical repair technique in 2010. 3 There are still a variety of barriers (including cost, regulatory, insurance, and logistical issues) between new cartilage repair products/techniques and their routine clinical applications. 4 However over the past 7 years there have been significant advances in our scientific knowledge with regard to cartilage repair or restoration techniques and imaging methods for evaluating both preoperative defects and postoperative repair status. Our review article will describe the principles of morphologic and compositional magnetic resonance imaging (MRI) techniques for the imaging of both cartilage repair and restoration surgery. We will then illustrate their application to longitudinal studies, and to illustrate the clinical relevance of pre- and postsurgical MRI with correlation to intraoperative images of various surgical techniques.
Imaging Features of Repair Tissue
Morphological Evaluation
Morphological MRI Techniques
MRI systems with high static magnetic fields can increase signal to noise ratio (SNR) and produce greater spatial resolution images than those with lower magnet strength, an important feature for cartilage imaging. Therefore, 1.5-Tesla (T) and 3-T magnet systems with extremity coils are recommended. In particular, the higher SNR allows isotropic 3D images to be acquired in a clinically relevant acquisition time. Advances in coil technology and parallel imaging techniques have also contributed to reduced scan times. The same acquisition techniques are used for repair tissue and native cartilage. High spatial resolution is essential to obtain images of diagnostic quality. In-plane resolution of 0.3 mm has been recommended to resolve fibrillation of the native cartilage surface using fat suppressed 3D gradient-recalled echo imaging. 5 Similarly, fissures and incomplete cartilage repair tissue integration require high-resolution imaging. In this section, we describe the pulse sequences most commonly used for morphological assessment of cartilage and cartilage repair tissue.
Two-Dimensional (2D) Fast Spin Echo (FSE)
Two-dimensional FSE or turbo spin echo imaging sequences provide excellent SNR, contrast between tissues, and faster acquisition times. 6 2D-FSE is the core imaging technique for the musculoskeletal system, in general, and the acquisition type most commonly used for clinical assessment of cartilage lesions; it is part of the cartilage imaging protocol recommended by International Cartilage Repair Society. 7 The most common MRI acquisitions for morphological assessment of cartilage include proton density-weighted (PDW), intermediate-weighted (IW), and T2-weighted (T2W) sequences with or without fat suppression. 8 On the other hand, T1W images are not considered to be cartilage-sensitive because they only allow poor differentiation between joint effusion and cartilage unless used in conjunction with MR arthrography. By comparison, fluid-sensitive images provide good evaluation of cartilage, ligaments, menisci, and edema-like marrow signal. T2W images provide greater contrast between cartilage surface and effusion and can detect subtle changes such as fibrillation, although the signal intensity (SI) in the cartilage near the osteochondral junction becomes low due to decreased signal intensity in the cartilage with longer TE times, which makes differentiation of the deeper layers of cartilage within the radial zone and at the tidemark challenging. PDW images (TE < 30 ms) provide good cartilage surface contrast and have shown high sensitivity for internal cartilage signal changes. IW imaging with longer echo times (TEs) (30-60 ms) combines the advantages of T2W and PDW imaging and is less prone to magic angle artifacts. 9 Care should be taken to adjust the TE on each magnet system to optimize visualization of the cartilage-fluid and cartilage-bone interfaces.
Three-Dimensional (3D) FSE
Two-dimensional FSE imaging often suffers from the need for thicker slices with resultant partial volume averaging effects that may be greater than in 3D acquisitions. 8 To optimally view the cartilage and joint space, 2D-FSE also requires imaging acquisition in multiple planes, which is time consuming. Isotropic, or near isotropic, 3D sequences can produce higher spatial resolution and high-quality reformatted images in any plane. All image planes are potentially provided from a single sequence obviating the multiple acquisitions required to image all planes with standard 2D sequences. 3D-FSE acquisitions use an additional phase-encoding gradient for spatial encoding along the slice-select direction and generate a set of contiguous slices. 10 The clinically available 3D-FSE sequences rely on flip-angle modulation to reduce blurring and parallel imaging to reduce scan time. 8
Three-dimensional FSE sequences have a diagnostic accuracy similar or slightly inferior to 2D-FSE for evaluation of focal cartilage defects and other joint structures. It has been shown that 3D-FSE can be applied for multitissue assessment of the whole joint.11 -13 Additionally, IW 3D-FSE sequences with fat saturation allow for evaluation of subchondral bone, although image contrast is better with water sensitive 2D-FSE images.
Spoiled Gradient Recoiled Echo (SPGR)
Fat suppression or water excitation 3D-SPGR, or similar fast low-angle shot (FLASH), imaging produces a contrast in which the SI of cartilage is greater than the surrounding tissues and joint fluid. 8 However, unreliable image contrast between the cartilage and fluid or joint tissues may result in poor detection of subtle cartilage defects. 14 An additional limitation is susceptibility artifacts, which can be prominent at some cartilage repair sites. Another problem with gradient recalled echo based (GRE) sequences, in general, is insensitivity to bone marrow edema and other joint pathology.15,16 While GRE sequences have been used for focal cartilage defect assessment,17,18 several recent studies have shown that they are less sensitive for focal defect detection than other techniques.16,19,20 These sequences are, however, excellent for cartilage segmentation and quantification of cartilage volume and thickness due to the good image contrast between cartilage and subchondral bone.21,22
Dual-Echo Steady-State (DESS)
DESS is another GRE imaging technique for cartilage that was introduced in the 1990s, 23 but did not become widely used until 3D-DESS could be performed with a short imaging time. Flip angles may be adjusted to optimize cartilage-fluid contrast to improve cartilage lesion detection. 24 Fat saturation or water excitation is typically used with DESS. The diagnostic performance of 3D-DESS for cartilage lesions is commensurate with 3D-GRE sequences.
Balanced Steady-State Free Precession (bSSFP)
Three-dimensional bSSFP sequences provide excellent contrast between cartilage and adjacent synovial fluid with bright fluid SI compared to cartilage. 25 Magnetic field inhomogeneities may result in off-resonance artifacts worsened by long TRs. Short TRs can improve the images but result in longer acquisition times. The diagnostic accuracy of bSSFP for morphologic assessment of cartilage is similar to that of standard 2D- and 3D-GRE sequences.25,26 Fat suppression, water excitation, or a Dixon technique such as IDEAL are commonly used with bSSPF. 27 The use of combined IDEAL and bSSFP may result in higher SNR and cartilage-fluid contrast to noise ratio. 28
Semiquantitative Scoring
MRI Observation of Cartilage Repair Tissue (MOCART)
MOCART is a reproducible semiquantitative scoring system for assessment of morphological cartilage repair. MOCART originally defined 9 structural variables but was modified to assess 11 variables, taking advantage of higher resolution 2D and isotropic 3D MR images, as summarized in Table 1 . 29 This “3D-MOCART” improves evaluation by localizing the features within the repair site and the relation of the repair site to the weight-bearing regions of the joint, evaluating the repair tissue-cartilage interfaces or “border zones,” in every plane, and providing a detailed subchondral bone assessment. 29 Since all of the original MOCART features are assessed by 3D-MOCART, for the sake of simplicity we will refer to both types as “MOCART.” 29 The usefulness of MOCART in a randomized controlled clinical trial of autologous cartilage tissue implants has been demonstrated in a study by Anderson and colleagues through its 5-year follow-up period. 30
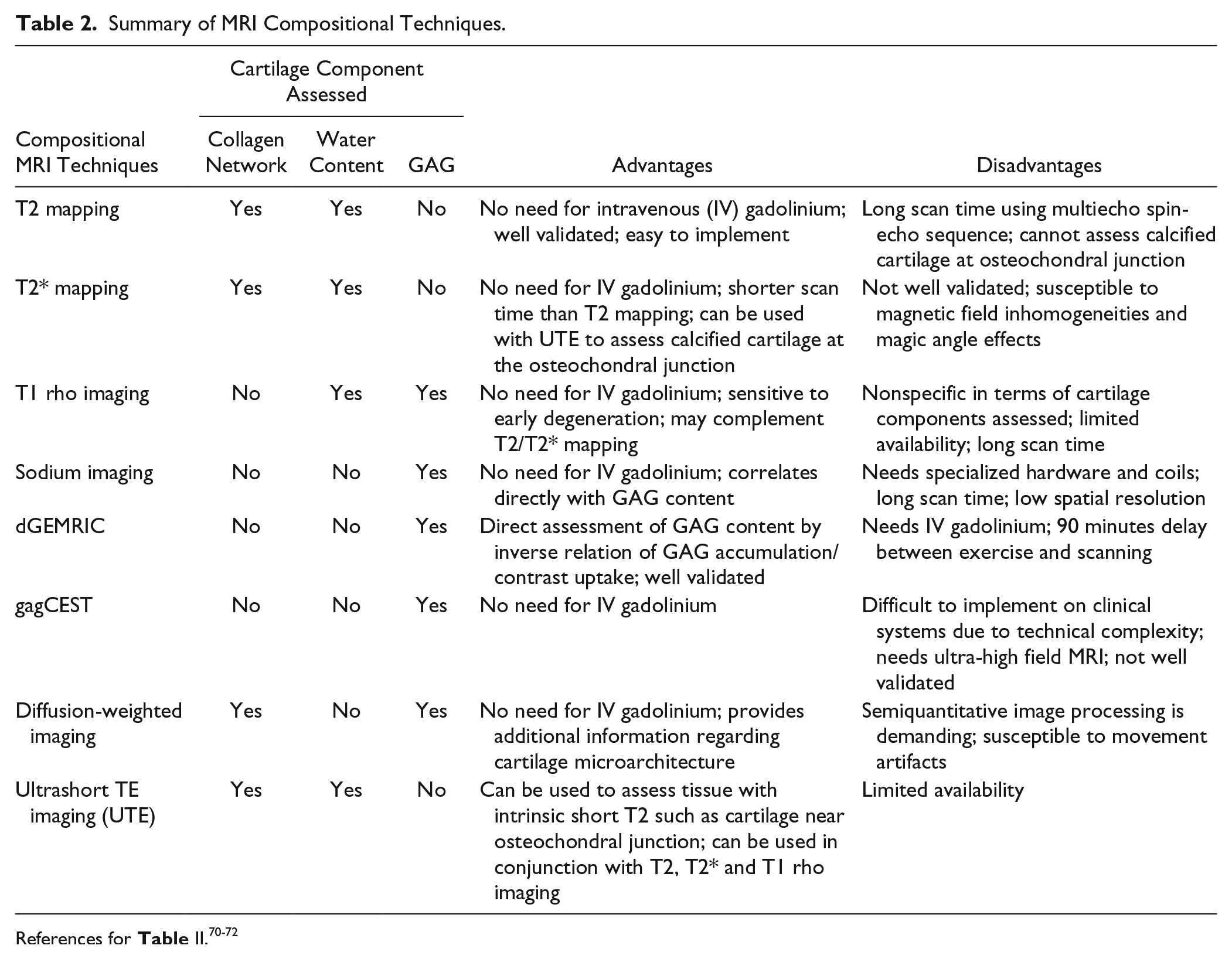
Summary of MOCART Scoring System for Cartilage Repair Tissue.
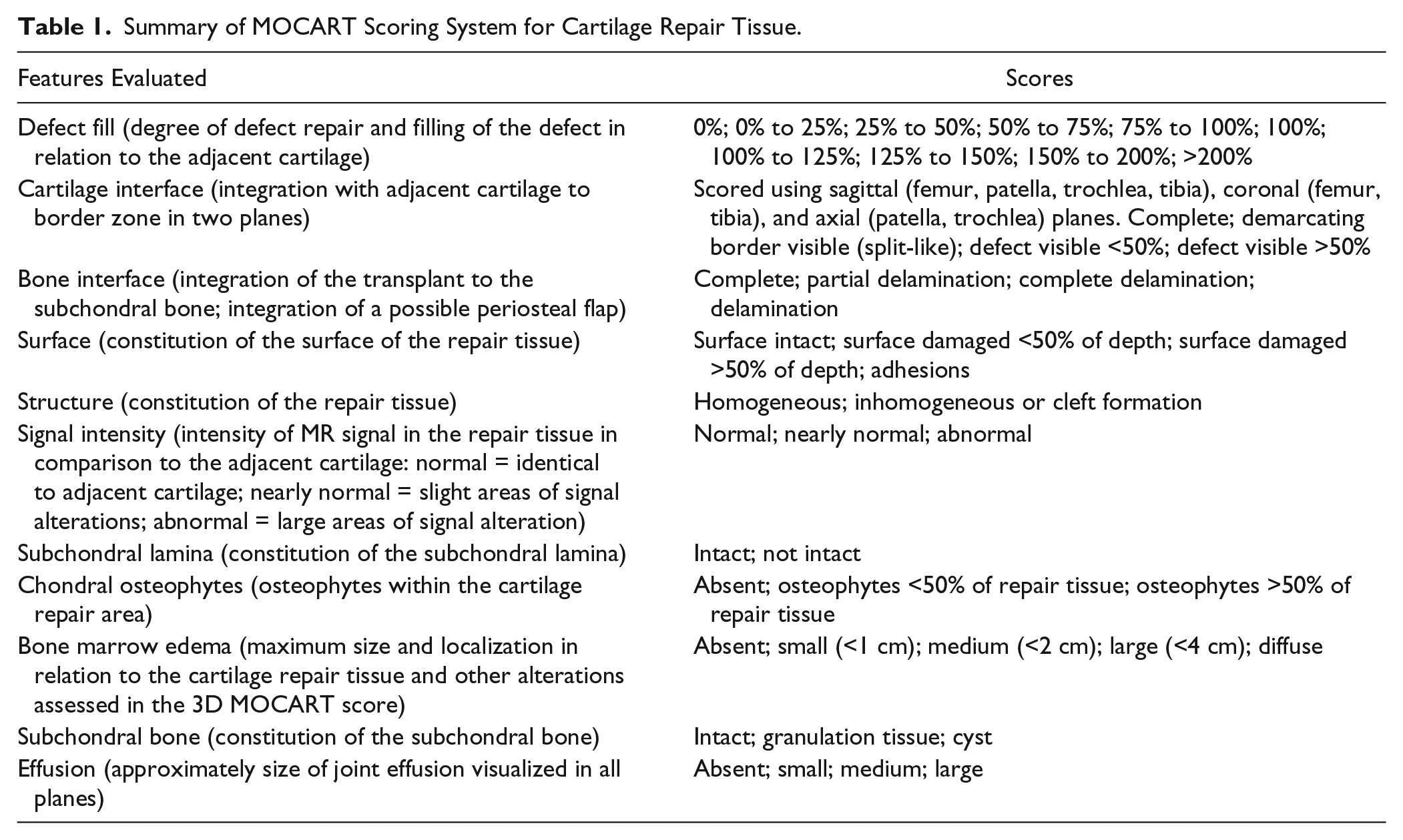
When the repair tissue is as thick as the adjacent cartilage, defect fill is scored as 100%. Hypertrophy (>100%) and incomplete repair (<100%) are recorded in 25% increments. Kreuz
The cartilage-interface variable scores the integration of the repair to adjacent cartilage at the border zone. Absence of a gap between the repair tissue and native cartilage is considered “complete.” Incomplete integration is either a fissure-like border zone or a measurable defect and is scored as less than or more than 50% of the length of the defect in coronal, axial, and sagittal directions. Of note, the conspicuity of fissures at the area of integration with the host cartilage is a function of both the in-plane and through-plane resolutions.
The bone interface grades integration of the repair tissue to the subchondral bone. It is described as complete integration, partial delamination, or complete delamination. For periosteal ACI, partial or complete delamination of the periosteal flap is noted.
The repair tissue surface is considered “intact” when it is even and smooth, without evidence of damage or irregularity. When damage such as fibrillation or ulceration is present, it is scored as involving less than or more than 50% of the tissue thickness. Although rare, synovial adhesions attached to the repair tissue are recorded under the “surface” variable.
The repair tissue structure is classified as “homogenous” when a normal, layered cartilage appearance is seen over the entire repair site. If the structure is lost, it is classified as inhomogeneous. Additionally, if a cleft is present in the structure, it is mentioned specifically.
Based on the visual inspection of MR images, the SI of the repair tissue is considered normal if it is identical to the adjacent native cartilage. If the SI is hyper- or hypointense, it should be classified as “nearly normal” or “abnormal,” depending on the extent and/or degree of altered SI. This SI variable is based on any cartilage-dedicated MR sequence; to simplify scoring, no specific acquisition type is specified.
The subchondral lamina, or bone plate, between the repair tissue and the bone marrow is classified as either intact or “not intact” if irregular.
Chondral osteophytes, that is, ossifications within the repair site above the level of the adjacent subchondral bone plate, are scored as absent or less than or more than 50% of the repair tissue thickness. Of note, chondral osteophytes are not uncommonly seen following the microfracture.
Bone marrow edema subjacent to the repair site is classified according to diameter as small (<1 cm), medium (<2 cm), large (<4 cm), or diffuse. The size of bone marrow edema is calculated in cm2. If the edema is related to other pathology by MOCART scoring, this is noted.
MOCART also scores the subchondral bone marrow adjacent (beneath) to the repair site as intact if there is no abnormality, or note is made of granulation tissue, subchondral sclerosis, and cysts. Joint effusions are estimated by visual inspection and graded as absent, small, moderate, or large.
Cartilage Repair Osteoarthritis Knee Score (CROAKS)
MOCART is comprehensive for assessment of the repair site itself; however, assessment of the other structures of the joint is paramount to assess longitudinal outcomes and development of osteoarthritis. The CROAKS combines features of MOCART and the MRI osteoarthritis knee score, 32 which is an established semiquantitative scoring system for whole organ assessment of the knee, to provide a comprehensive, reproducible tool for longitudinal postoperative assessment after surgical cartilage. 33
Table 2 shows the specific features outside of the repair site, assessed for multiple joint subregions by CROAKS. These include bone marrow lesions beyond the repair site, subchondral cysts, nonoperated cartilage status, osteophytes, synovitis, effusion, menisci, and anterior and posterior cruciate ligaments. Other periarticular features evaluated include the pes anserine bursa, illiotibial band, prepatellar bursa, infrapatellar bursa, and presence of popliteal cysts, ganglion cysts, and/or loose bodies.
Compositional MRI
At the molecular level, cartilage consists of approximately 70% to 80% fluid and 20% to 30% solid extracellular matrix (ECM). 34 The ECM is composed of a network of collagen fibrils and proteoglycan molecules; mostly aggrecans that consist of a protein core with covalently attached glycosaminoglycans (GAGs).34,35 GAGs are negatively charged and make up the majority of the “fixed charge density” of the ECM. Electroneutrality is maintained by the partitioning of cations, mainly sodium (Na+), and anions, mainly chloride (Cl−), between the cartilage and joint fluid. The biomechanical properties of cartilage are largely due to water flow within the ECM and the cartilage-joint fluid distribution of water that is, in part, dictated by the fixed charge density.
Compositional MRI acquisitions provide a way to detect biochemical and microstructural changes in the cartilage ECM even before gross morphological changes occur. Although not in routine clinical use, these techniques have been used extensively in cartilage research. Compositional MRI can supplement morphologic imaging, by potentially defining the biomechanical quality of cartilage repair tissue. The basic concepts of the compositional MRI techniques currently available to evaluate cartilage repair tissue are summarized in
Table 2
. Of note, the stratification of T2 relaxation times across the articular cartilage reflects changes in the orientation of collagen fibers in the extracellular matrix.
36
The disorganized tissue found in cartilage repair sites generally has greater water mobility and therefore more prolonged T2 relaxation times. Laminar analysis of normal articular hyaline cartilage T2 maps, that is, separate measurement of regions of interest of the superficial and deep halves of the cartilage (
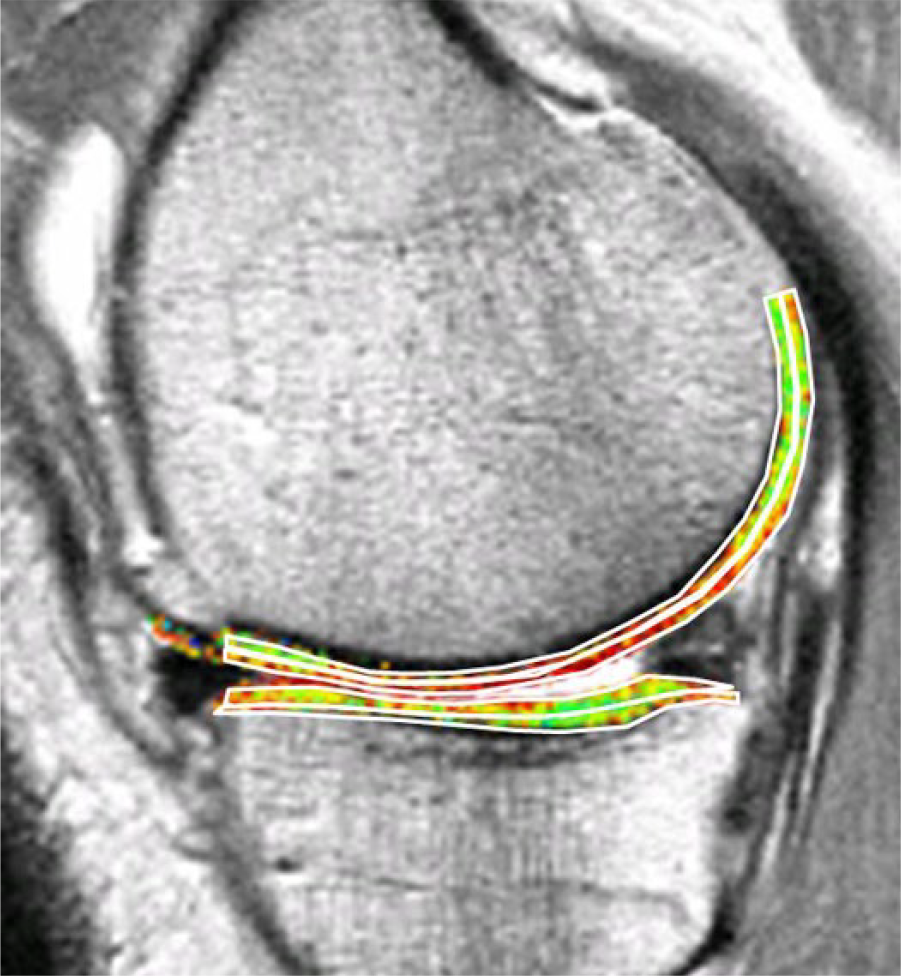
Bilaminar T2 relaxometry. Example of manual segmentation of the cartilage of the medial tibia and weight-bearing femoral condyle on a sagittal multi-echo spin echo (MESE) sequence. The tibial cartilage is segmented from its anterior to posterior end, and the femoral cartilage throughout a weight-bearing region of interest. As cartilage T2 is known to display spatial variation with tissue depth, the segmented cartilage plates subdivided into the top (superficial) and bottom (deep) 50%, based on the local distance between the segmented cartilage surface and bone interface. Color coding displays intrachondral variations in regional T2.
Currently Available Surgical Techniques
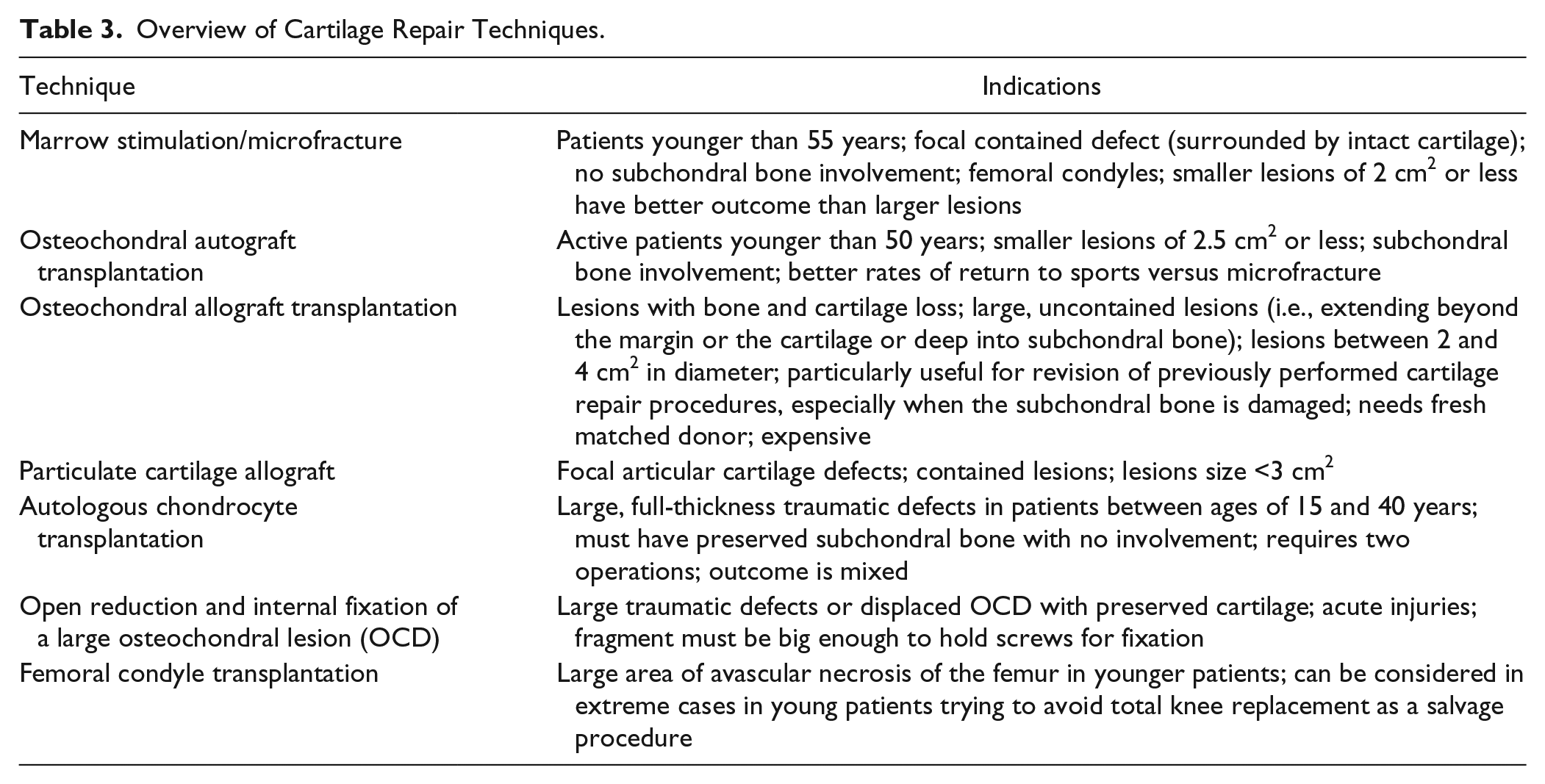
In this review, we will describe the following cartilage repair techniques: microfracure/marrow stimulation, osteochondral autograft/allograft (OATS) transplantation, particulate cartilage allograft, ACI, open reduction and internal fixation of a large osteochondral lesion, and femoral condyle transplantation. Table 3 shows the summary of these techniques. Of note, a recent systematic review of randomized controlled trials of knee cartilage repair surgery concluded no single treatment provides the single “best” clinical outcome among various available techniques. 40
Overview of Cartilage Repair Techniques.
Microfracture/Marrow Stimulation
The microfracture technique is the most commonly used procedure for the repair of focal cartilage defects.
41
This procedure is aimed at inducing marrow stimulation, and it begins by the arthroscopic micropenetration of the cartilage defect surface to the subchondral bone until visible bleeding is achieved. Removal of calcified layer of cartilage is also performed. This allows mesenchymal stem cells from the subchondral bone marrow to be introduced into the debrided cartilage defect, which heal the defect by forming fibrocartilage tissue.
42
This procedure is optimally indicated in patients aged less than 40 years, with a focal contained cartilage defect that is surrounded by intact cartilage or with femoral condyle lesions and small defects <2 cm2 (
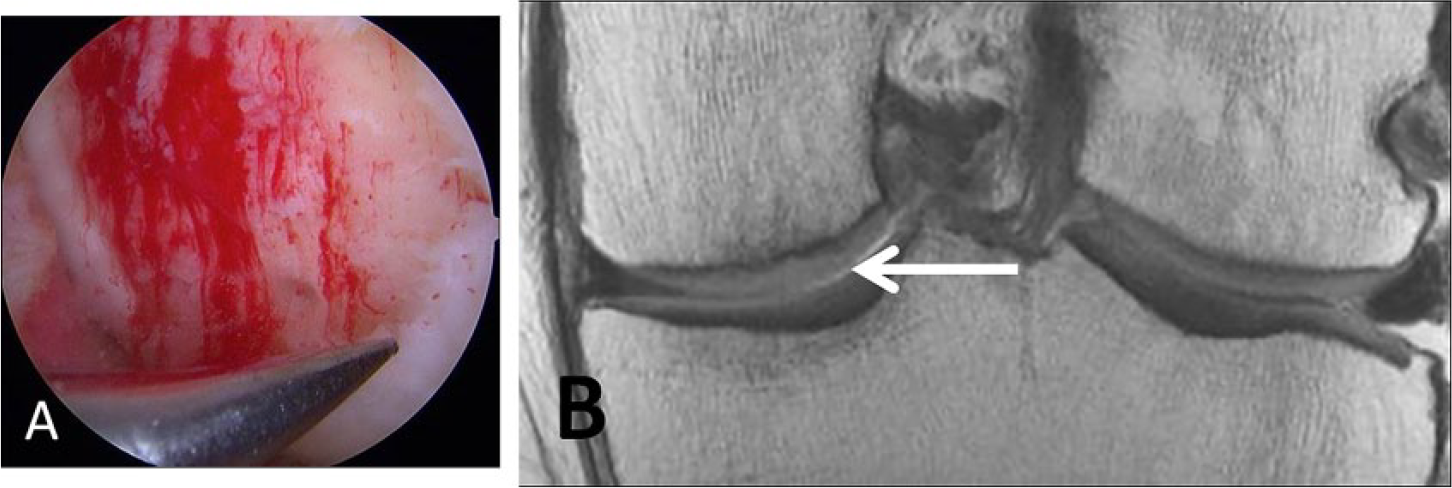
A 46-year-old male laborer who had surgical repair of a high-grade cartilage defect by a microfracture technique. (
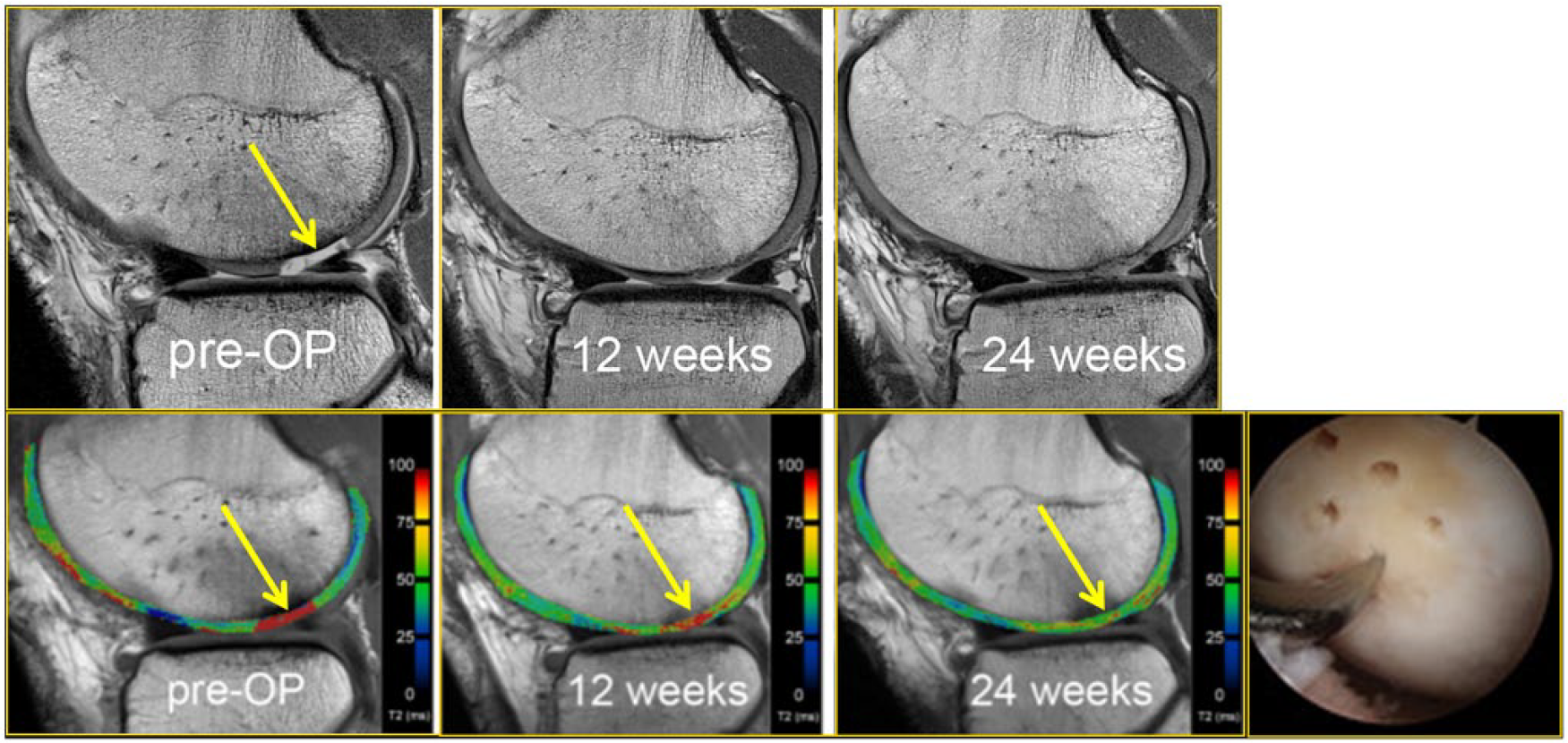
A 25-year-old male soccer player who received microfracture surgery of a cartilage lesion (yellow arrow, preoperative image). Arthroscopic image shows an arthroscopic awl at the site of lesion microfracture. Pre- and postoperative T2-mapping images show how T2 values change longitudinally, demonstrating the maturation of the repaired cartilage (yellow arrows, 6 weeks and 24 weeks postoperatively) with decreasing free water content and thus decreasing T2 values during the longitudinal follow-up. The patient returned to play 8 months after surgery.
Osteochondral Autograft Transplantation (OAT)
The OAT procedure begins by debriding damaged cartilage to create stable margins or cartilage borders around the defect. Donor osteochondral cylinders are harvested from non–weight-bearing margins of the trochlea either on the medial or lateral borders, near the intercondylar notch, or from other joints. Osteochondral grafts are then implanted into matching cylindrical recipient sites created at the cartilage defect (
A 21-year-old mixed martial arts fighter who sustained a full-thickness cartilage defect, anterior cruciate ligament tear, and meniscal tears and who had repair of an osteochondral lesion of the medial femoral chondyle by osteochondral autograft transfer system (OATS). (
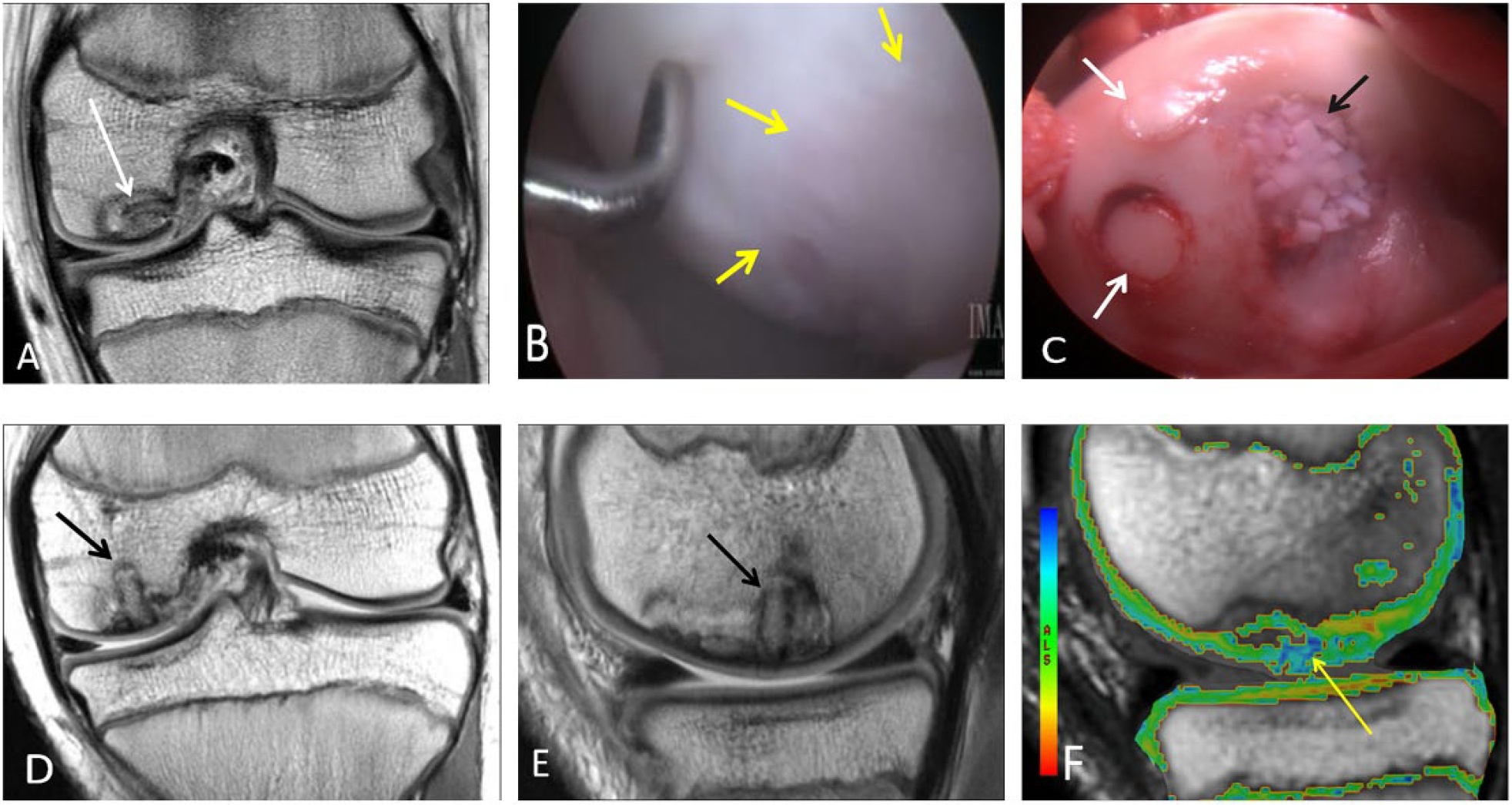
An 11-year-old male who had repair of an osteochondral lesion by osteochondral autograft transfer system (OATS) and de novo juvenile cartilage transplantation technique. Coronal (
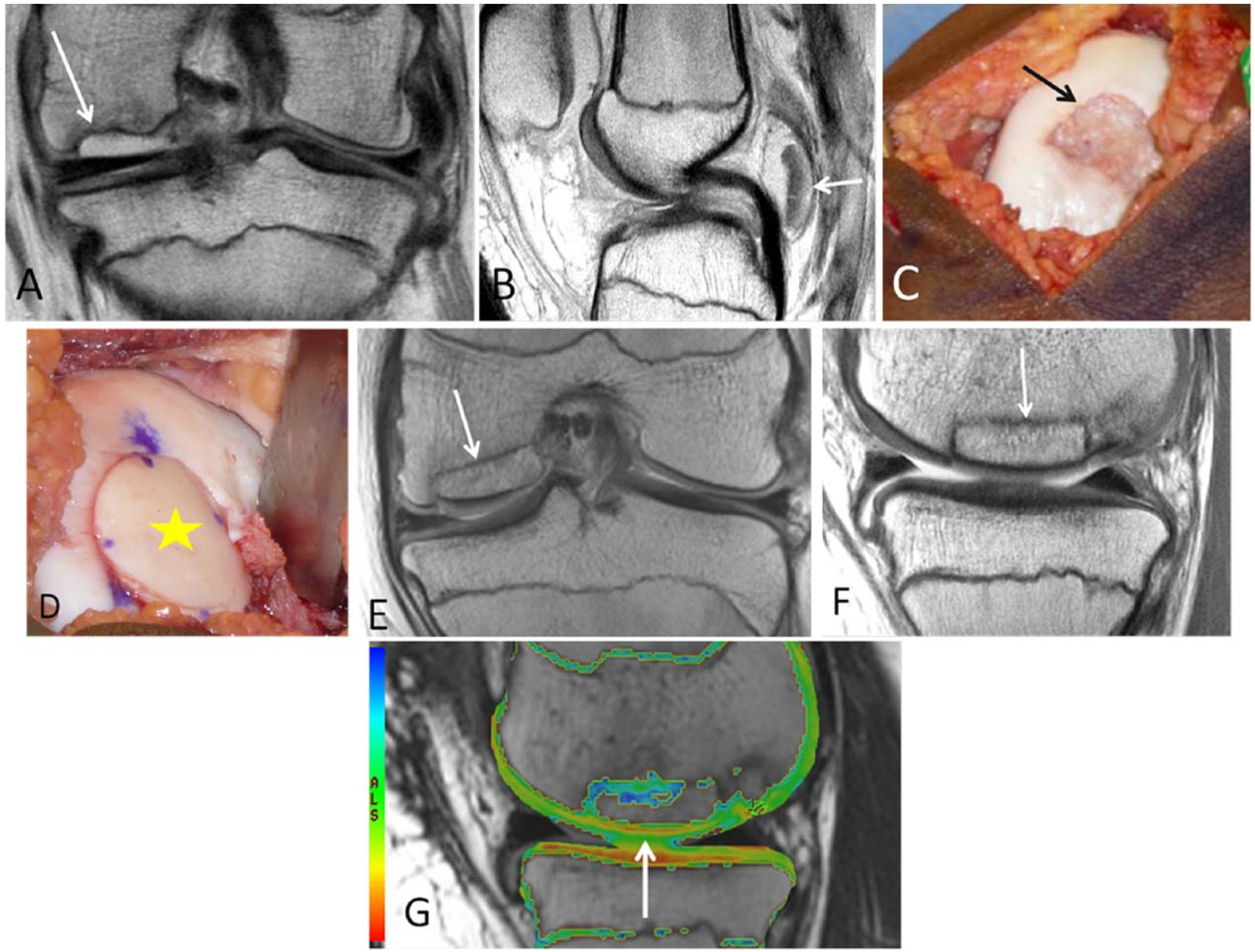
A 15-year-old male who had cartilage repair with osteochondral allograft transplantation and osteochondral autograft transfer system (OATS) for an osteochondral lesion. Coronal (
Application of a quantitative evaluation of articular surface curvature and 3D digital template for cartilage repair has been described and was shown to have a potential to optimize postsurgical restoration of joint articulation. 54 On early postoperative images, the graft should completely fill the defect with uniform signal intensity cartilage, without gaps between plugs and adjacent cartilage or adjacent bone. As bone incorporation progresses, the edema in the plugs and surrounding bone resolves and the plugs become indiscernible from the native tissue. Persistent edema-like marrow signal intensity within subchondral bone beyond 18 months and subchondral cyst formation may indicate poor tissue integration. Osteonecrosis is a rare complication of OATS. 55
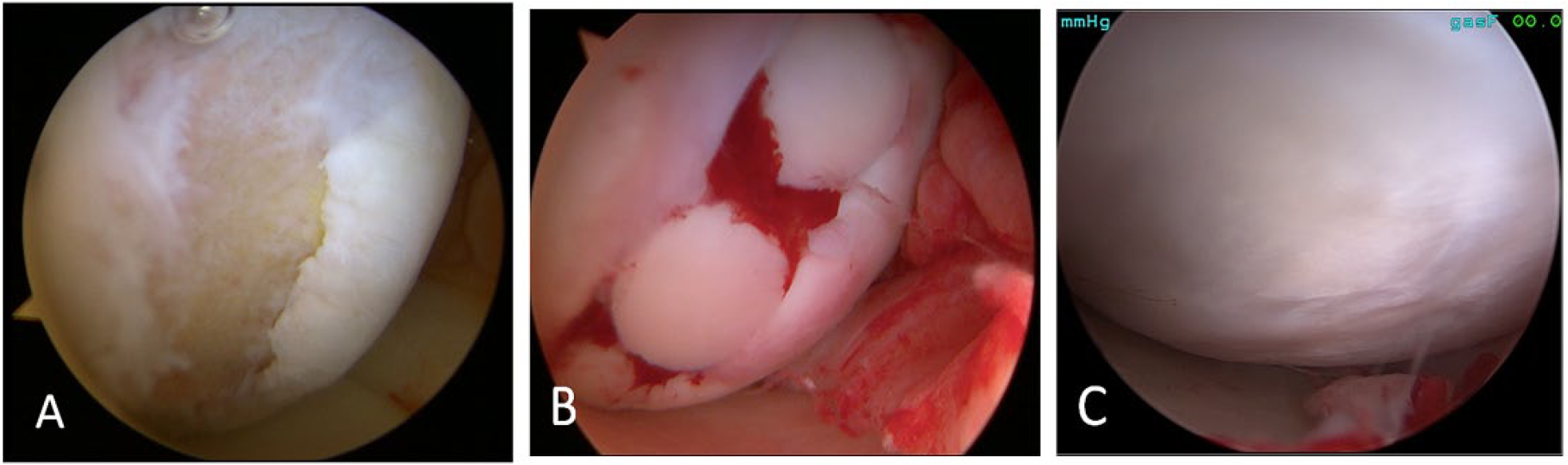
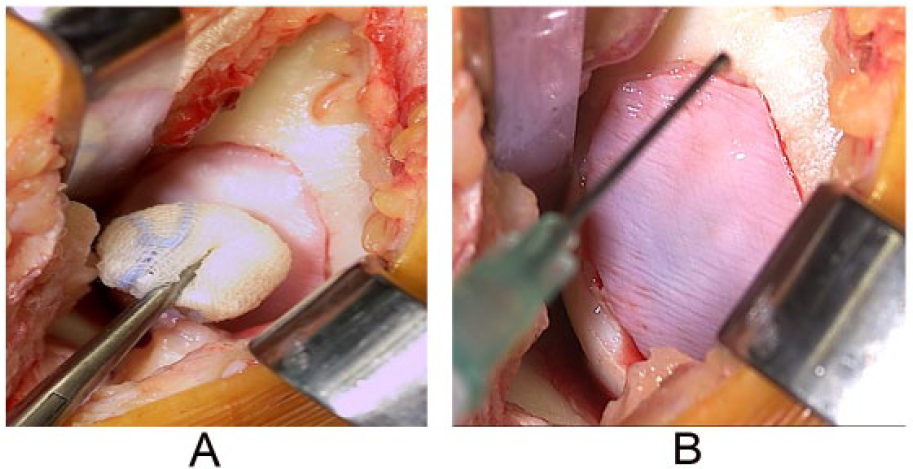
Particulate Cartilage Allograft
Particulate cartilage allograft is a newer surgical technique for focal cartilage defects and comprises small approximately 1-mm cubes of particulated cartilage from a juvenile allograft donor (age <13 years) (
Osteochondral Allograft Transplantation
The osteochondral allograft technique involves harvesting cartilage and bone from a fresh cadaveric donor. Allografts are not limited by the amount of donor tissue, and thus cartilage defects >3 cm2 in diameter can be repaired (
MRI assessment of osteochondral allografts includes evaluation of graft signal intensity, defect fill, cartilage edge integration at host-graft junction, articular surface congruity, subchondral bone plate congruity and bone marrow signal, osseous integration, and presence of cystic changes of graft. 63 Initial defect fill is expected with a smooth articular contour. Diffuse bone marrow edema is expected during the first 3 months postoperatively. With progressive bone incorporation, an advancing high-signal-intensity front develops. Graft marrow signal intensity eventually normalizes. Edema-like marrow signal intensity persisting for more than 12 months or articular surface collapse may be indicative of eventual failure of treatment. MRI features suggestive of delayed or poor allograft incorporation include the presence of persistent fluid signal intensity within the graft, discernible fissure (cartilage edge integration) or cleft (osseous integration) at host-graft junction, disrupted subchondral bone plate integrity, and presence of cystic changes of graft at host-graft junction.63,64Additionally, MRI may help detect potential host-immune response to the allograft tissue, which is demonstrated on MRI as abnormal host marrow edema, thick host-graft interface, and abnormal graft marrow signal intensity. 65
Autologous Chondrocyte Implantation
This two-part procedure begins by arthroscopic harvesting of chondrocytes, usually from the cartilage on the tibial spines.
1
They are subsequently cultured and multiplied ex vivo for 6 to 8 weeks, and then implanted into the chondral defect together with fibrin glue to fix the edges (
Example of autologous chondrocyte implantation procedure for a femoral cartilage defect >2 cm, showing the second step of the procedure, that is, implantation of chondrocytes (
Postoperatively, complete integration of the graft is demonstrated on MRI by the presence of complete filling of the chondral defect with the repair tissue to the expected level of adjacent cartilage. 49 Underfilling of the repair site signifies incomplete defect fill. Incomplete integration of repair tissue after ACI is termed “delamination,” which can be identified by displacement of all or a portion of the graft from the repair site or linear fluid intensity between the repair tissue and underlying bone. 49 A common complication after ACI is periosteal cover hypertrophy, which is depicted on MRI as a graft being thicker than the native cartilage with resultant distortion of articular contour. Adhesions are another postoperative complication commonly seen in the peripatellar regions. Poor integration of the ACI with the subchondral bone is indicated by persistent or increasing edema-like signal in the marrow beneath an ACI site. 49
Other Techniques
Open reduction and internal fixation (ORIF) is indicated for large traumatic defects or displaced osteochondral lesions (
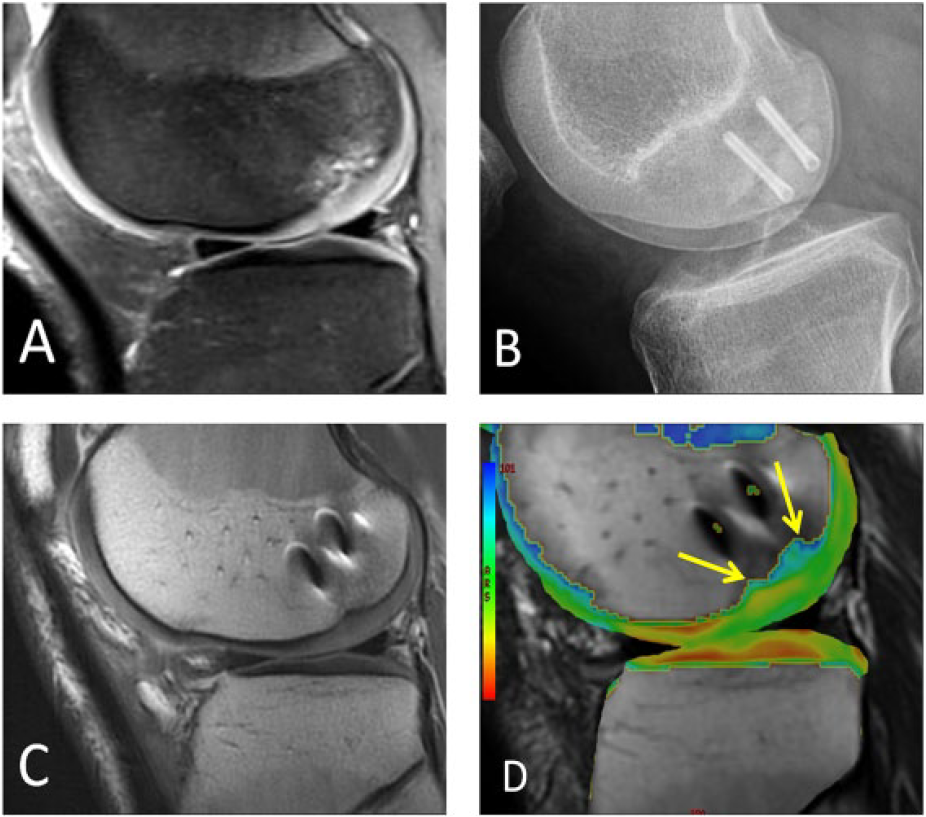
A different male patient who underwent open reduction and internal fixation of a large osteochondral lesion. (
Femoral condyle transplant can be performed for large areas of avascular necrosis with associated osteochondral lesions of the femur in young patients who are not candidates of a unilateral knee replacement (
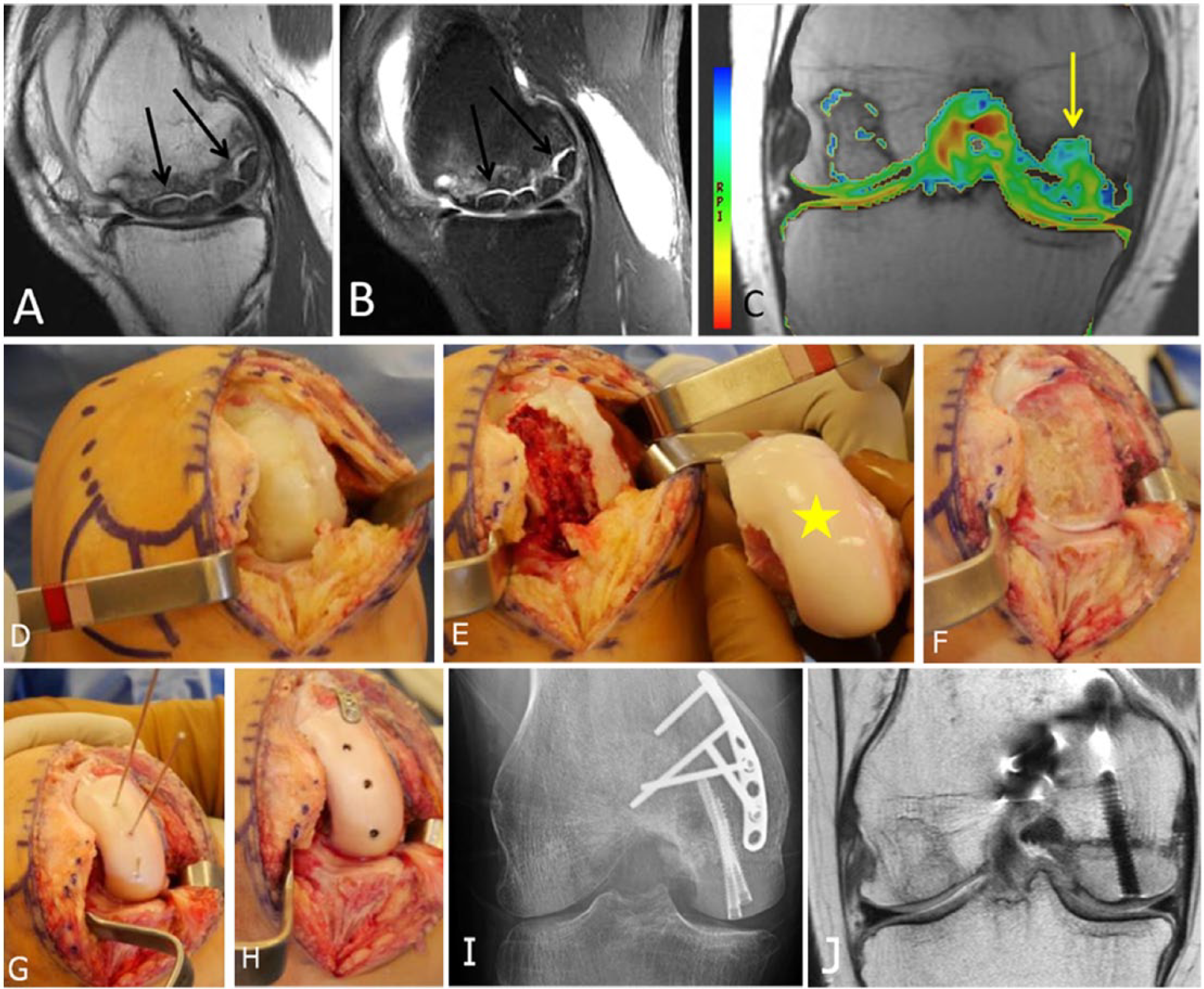
A 26-year-old female who had medial femoral condyle transplantation with matched fresh allograft. (
Conclusion
All cartilage repair techniques have the same primary goal: to decrease pain symptoms, to improve mobility and function, and to prevent the progression of osteoarthritis. The various cartilage repair surgery techniques discussed have shown to improve functional outcomes; however, assessing the efficacy within the orthopedic literature is limited by heterogeneity in surgical technique, lesion type, patient characteristics, and reporting of nonstandardized outcome measures. There is urgent need to define outcomes clinically and by MRI measurements including local assessment and with regard to long-term osteoarthritis development/progression. In clinical practice, the MRI assessment of repair tissue relies heavily on morphologic imaging. The main role of MRI during the presurgery assessment is to give a detailed description of the defect size, depth of the lesion, and associated subchondral bony changes in order to help determining the best treatment choice. Compositional MRI provides the opportunity to measure the biochemical and microstructural time-dependent processes of maturation occurring within the repair tissue. Compositional MRI techniques are mostly used in research and for clinical trials, but they hold great promise for the clinical determination of surgical success. Before they can become clinically useful, however, compositional MRI techniques must be standardized and validated for cartilage repair tissue assessment and made time efficient. The combination of MRI-based morphologic and compositional imaging plays an integral role in the assessment of cartilage repair tissue and its integration to native tissues after cartilage repair surgery.
Footnotes
Acknowledgment and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ali Guermazi is the President of Boston Imaging Core Lab (BICL), LLC, and a consultant for Merck Serono, Genzyme, GE Healthcare, Pfizer, TissueGene, Ortho Trophix, and Astra Zeneca. Frank W. Roemer is a shareholder of BICL. Xinning Li is a Consultant for Tornier Shoulder Arthroplasty and Depuy Mitek Sports Medicine. The other authors have no conflicts of interest to disclose.