Abstract
Background:
In focal repair of joint cartilage and meniscus, initial stiffness and strength of repairs are generally much less than surrounding tissue. This increases early failure potential. Secure primary fixation of the repair material is also a problem. Acrylamide polymer double-network (DN) hydrogels are candidate-improved repair materials. DN gels have exceptional strength and toughness compared to ordinary gels. This stems from the double-network structure in which there is a high molar ratio of the second network to the first network, with the first network highly crosslinked and the second loosely crosslinked. Previous studies of acrylic PAMPS/PDMAAm and PAMPS/PAAm DN gels demonstrated physicochemical stability and tissue compatibility as well as the ability to foster cartilage formation.
Methods:
Mechanical properties related to surgical use were tested in 2 types of DN gels.
Results:
Remarkably, these >90%-water DN gels exhibited dynamic impact stiffness (E*) values (~1.1 and ~1.5 MPa) approaching swine meniscus (~2.9 MPa). Dynamic impact energy-absorbing capability was much lower (median loss angles of ~2°) than swine meniscus (>10°), but it is intriguing that >90%-water materials can efficiently store energy. Also, fine 4/0 suture tear-out strength approached cartilage (~2.1 and ~7.1 N v. ~13.5 N). Initial strength of attachment of DN gels to cartilage with acrylic tissue adhesive was also high (~0.20 and ~0.15 N/mm2).
Conclusions:
DN gel strength and toughness properties stem from optimized entanglement of the 2 network components. DN gels thus have obvious structural parallels with cartilaginous tissues, and their surgical handling properties make them ideal candidates for clinical use.
Introduction
Repair of Cartilage Lesions
Cartilage and meniscal lesions have limited potential for spontaneous repair.1,2 Specifically, joint surface lesions with surface areas larger than 4 cm2 are now believed to inevitably lead to degenerative arthritis. 3 This is especially the case in the knee, with serious consequences (debilitating pain and markedly restricted mobility), often leading to the need for major surgery, that is, total joint arthroplasty. As a consequence, in recent years, much effort has been devoted to developing methods for repairing such lesions.
The pioneering work of Peterson et al.4-6 showed that a suspension of the patient’s own previously harvested and expanded chondrocytes, injected behind a periosteal flap, is able to build up hyaline-like cartilage tissue. However, this method has its disadvantages, mostly the time required for the tissue to form and a consequent long rehabilitation period, up to 1 year, until pain-free full weightbearing is possible and joint homeostasis is re-established. As a result, alternative methods have been proposed and pursued. The main approach has been to place harvested and expanded chondrocytes in a scaffold material and stimulate them in vitro to begin cartilage formation in the scaffold prior to implantation. The resultant “construct” is then implanted. 7 Using this approach, the rehabilitation period can be shortened considerably.
However, such tissue-engineered constructs still do not have mechanical properties (e.g., stiffness, strength) at the time of implantation that are even remotely similar to natural articular cartilage. As a result, the rehabilitation period must still be on the order of several months in order to establish repaired tissue capable of bearing cyclic impact loads in the knee of the magnitude and frequency associated with normal daily activity.1,3,8,9
In order to further shorten the rehabilitation period needed after a cell-based cartilage repair, a tissue-engineered scaffold with cartilage-like initial mechanical properties (and of course the ability to foster cartilage formation) would be an attractive solution. Alternatively, for small repairs, one could also consider using plugs of a completely artificial solid material with cartilage-like mechanical properties rather than a scaffold. In this case, the plugs must also be extremely durable (lasting years) in order to be of clinical use. In either case (scaffold or plug), it is also necessary to have a means for securing the implant to the osteochondral bone and the surrounding intact cartilage, which can be particularly challenging in defects that are not well contained. After initial surgical wound healing, scaffolds or plugs with cartilage-like mechanical properties would be able to immediately distribute gait-related biomechanical impacts nondisruptively to the surrounding natural cartilage and also protect the sensitive subchondral bone.
Double-Network Hydrogels
Double-network hydrogels (DN gels) are a new family of candidate materials for potential use in the repair of skeletal system soft tissues. They have been developed for these and other purposes by Gong et al. at Hokkaido University and reported in the literature starting in 2003. 10 Ordinary single-network hydrogels containing 85% to 95% water do not have cartilage-like compressive strength. For example, articular cartilage is reported to have a compressive fracture strength of approximately 36 MPa. 11 In contrast, an example of a single-network 92%-water gel, based on an acrylamide polymer, poly(2-acrylamido-2-methylpropanesulfonic acid), that is, PAMPS, which is highly crosslinked (~4 mol %), has a compressive fracture stress of only 0.4 MPa. However, when a large molar ratio of a second acrylamide polymer, poly(acrylamide), that is, PAAm, is added to PAMPS and controlled to be lightly crosslinked (e.g., 0.1 mol %), the result is a 90%-water PAMPS/PAAm DN gel with a markedly higher compressive fracture stress, 17.2 MPa, which is 43 times higher than the PAMPS gel. 10 In addition, a PAMPS/PAAm hydrogel with a >90%-water content does not fail until compressive strain is over 90%. For comparison, a commercially available PVA hydrogel is reported to have a compressive fracture stress in the range of only 1.4 to 2.0 MPa and fails at 47% to 62% compressive strain. 12 The tough 90%-water PAMPS/PAAm DN gel studied here thus approaches the compressive strength of articular cartilage. It should be noted that cartilage and other skeletal system tissues are also high water-content materials and employ crosslinking and a double-network strategy (e.g., highly crosslinked collagen plus proteoglycan gel) to achieve their mechanical properties.
Gong et al. have performed and reported a variety of preliminary, promising biomechanical and biological studies of DN gels over the past few years.10,13-17 Recently, a study of the repair of induced osteochondral defects in rabbit knees with a DN gel composed of a PAMPS first network and a PDMAAm, that is, poly(N,N′-dimethylacrylamide), second network was performed. 18 The PAMPS/PDMAAm DN gel used had been shown previously in a rabbit model to exhibit no decline in stiffness, strength, or strain at failure at 6 weeks 13 and to elicit little inflammatory response. 16
For the repair study, 18 the defects created in the patellofemoral groove of the femoral condyle were 4.3 mm in diameter and 15 mm deep, thus extending approximately 12 mm or more into osteochondral bone. The bony part of the defect was partially filled with a cylindrical plug of the same diameter made from the PAMPS/PDMAAm DN gel, leaving the last 1.5 to 2.5 mm of depth (relative to the original cartilage surface) empty. After 4 weeks, the empty space (above the DN gel plugs) had become completely filled with white, opaque tissue. It appears that hyaline-like cartilage was formed on top of the PAMPS/PDMAAm cylinders in the osteochondral defects.
Tissue adhesives are increasingly being evaluated and used as an alternative to sutures for small-scale repairs. They offer the potential advantage of distributing the load over a much larger interfacial area than is possible with sutures and thus markedly reducing the focal stresses created by sutures. They also offer speed and simplicity compared to sutures, and the repairs have been found to be sufficiently durable to allow subsequent healing in many applications. The inflammatory response to clinically approved adhesives is acceptably low. A recent orthopedic surgery-related in vitro study compared a clinically approved tissue adhesive, Histoacryl (primary active ingredient of N-butyl-2-cyanoacrylate; B. Braun Melsungen AG, Melsungen, Germany), with sutures in the repair of knee meniscal tears. 19 They found Histoacryl (B. Braun Melsungen AG) significantly increased the force required to produce a 2-mm gap in the repairs. Because of the technically easier handling for the surgeon, it might be a good idea to develop and use cartilage-like repair materials that can be safely glued into the defect, thus avoiding the need for sutures.
Because the DN gels studied here are based on acrylamide polymers, it seemed likely that an acrylic tissue adhesive might work well to bond them to other surfaces, which is why we chose to investigate whether this was the case. As a result of reviewing the intriguing work of Gong and her group, we were fortunate to establish a collaboration with her to study further the properties of DN gels that might be of interest before their clinical use.
The aims of this study of 2 types of acrylic polymer DN gels were the following:
to measure dynamic stiffness and the ability to dissipate energy using function-related mechanical techniques previously established by the authors20,21;
to devise and use methods to measure the surgical attachment strength that can be achieved with 1) sutures and 2) a surgical tissue adhesive; and
to compare the results for the 2 DN gels and compare the properties with those of natural cartilage where applicable.
Materials and Methods
Materials
For this study, 2 different acrylamide polymer DN gels, known as PAMPS/PDMAAm and PAMPS/PAAm, were provided. The preparing ratios and water content of the 2 DN gels are given in Table 1 . While the second-network component was different for the 2 gels, (DMAAm v. AAm), the only difference in preparing ratios for the 2 gels was for the ultraviolet initiator for the second-network component (0.03 v. 0.01 mol %). As shown, both DN gels contained more than 90% water. However, the difference in water content (94.0% v. 90.9%) means that the PAMPS/PDMAAm gel contained only approximately 66% as much polymer as the PAMPS/PAAm. The methods for producing the DN gel structures from the polymeric components are described elsewhere.10,22 The DN gels were then placed in normal saline and shipped to Basel by ordinary post, where they were kept in saline at 4 °C to 6 °C before testing at room temperature. Specimen dimensions for each test mode are described later.
Preparing Ratios and Water Content of the 2 Double-Network (DN) Polymer Hydrogels
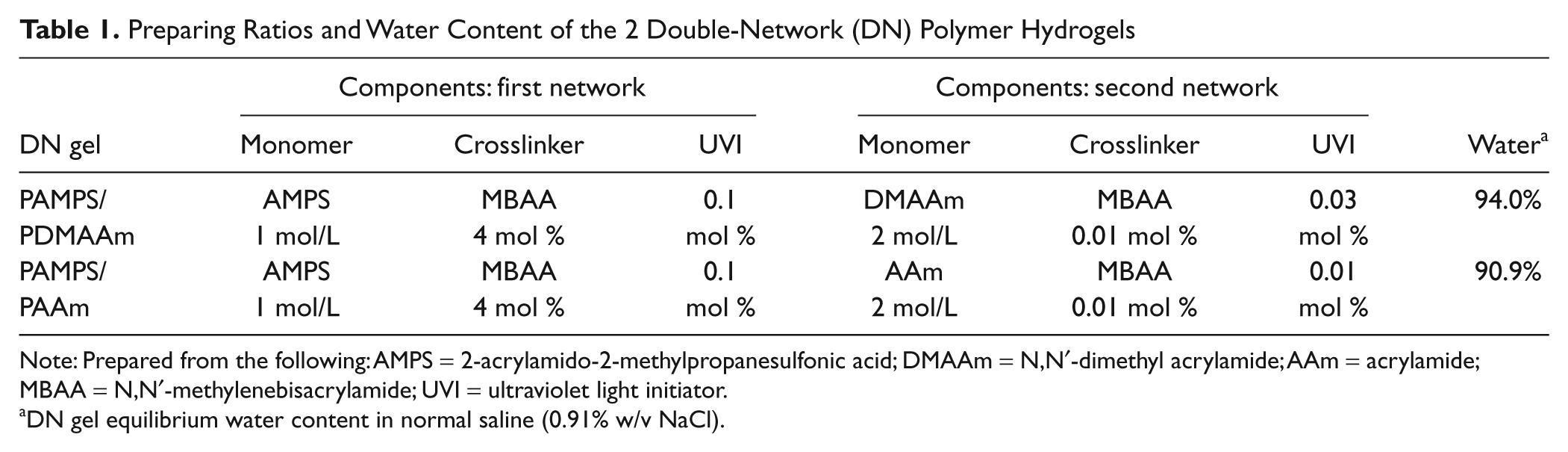
Note: Prepared from the following: AMPS = 2-acrylamido-2-methylpropanesulfonic acid; DMAAm = N,N′-dimethyl acrylamide; AAm = acrylamide; MBAA = N,N′-methylenebisacrylamide; UVI = ultraviolet light initiator.
DN gel equilibrium water content in normal saline (0.91% w/v NaCl).
Methods
Dynamic stiffness by millimeter-scale microindentation
The Basel authors have previously developed and used microindentation methods for determining the dynamic stiffness parameters (aggregate modulus [E*] and loss angle [δ]) of cartilage and meniscus.20,21 The methods were employed here on the DN gel specimens. Briefly, the methods are designed to recognize 2 things: 1) the values of E* and δ for poroviscoelastic materials are extremely dependent on deformation rate; and 2) articular cartilage must function in 2 extremely different loading regimes: the sudden transient deformations that occur during gait, and the slow quasicyclic deformations that cause fluid to move in and out of cartilage and thus provide a means for nutrition. Thus, both a “gait mode” and a “nutrition mode” testing procedure have been developed. In both tests, described briefly below, it is possible to measure the loss angle directly ( Fig. 1 ).
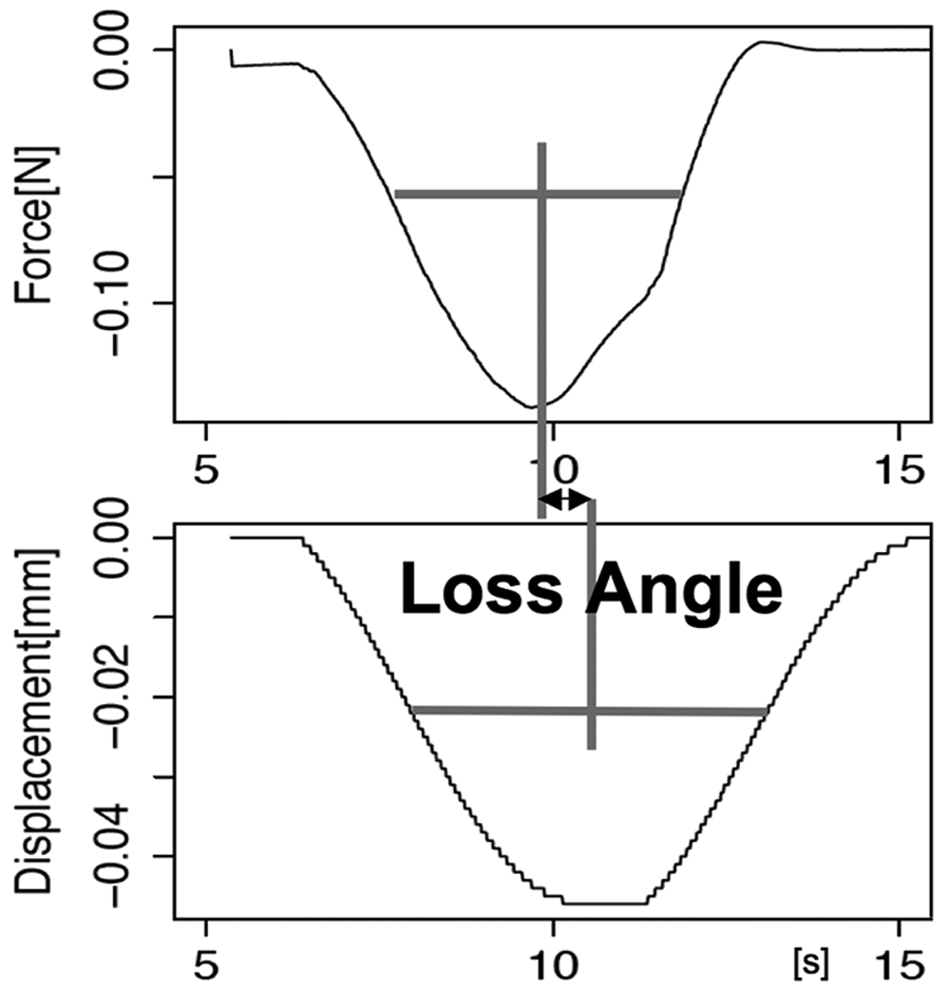
Example of the force and displacement data for an articular cartilage specimen tested in nutrition (N)–related slow sinusoidal microindentation (SSMI) to a depth resulting in cycles at 0.1 Hz with a maximum speed of 0.015 m/s. The loss angle was calculated from the time length of the phase shift between displacement and load curves in both SSMI mode and gait (G)–related fast impact microindentation (FIMI) mode.
In our gait mode, evaluation of dynamic stiffness is accomplished by fast impact microindentation (FIMI), using a modified version ( Fig. 2 ) of an instrument developed at the Minsk Institute of Physics. 23 FIMI does not precisely duplicate the complex impact loading/unloading patterns seen in gait. However, the indenter velocity at impact is in the gait range: approximately 0.3 m/s. 23 Briefly, the dynamic motion (distance v. time) of a falling microindenter (steel, 1.0-mm-diameter spherical tip; 1.9-g mass of indenter) is captured electromagnetically. The velocity at impact is among the parameters captured by the electromagnetic coil through which the indenter moves. In these tests, the mass- and gravity-produced acceleration of the indenter results in nondestructive indentations having depths of 0.1 to 0.2 mm. From the dynamic motion data and indenter mass and geometry, it is possible to calculate the same parameters as in cyclic loading tests: E* and δ. Ten DN gel specimens were tested; they were 3 mm thick and about 10 × 20 mm in lateral dimensions. They were kept moist with saline during testing at room temperature. For a given specimen, 10 replicate impact tests were performed at the same spot at intervals of approximately 20 seconds, and the resultant E* and δ were averaged to produce E* and δ values for a given specimen.

Gait-related (G) mode modulus and loss angle measurement device, mounted on a stable loading frame, equipped with a load cell and laser positioning system.
In our nutrition mode, evaluation of dynamic stiffness is accomplished by slow sinusoidal microindentation (SSMI). The SSMI tests are performed with a MTS Synergie 100 mechanical testing instrument (MTS Systems Corporation, Eden Prairie, MN), programmed to perform a series of single sinusoidal cycles at 0.1 Hz. The microindenter (steel, ~3.2-mm-diameter spherical tip) moves under sinusoidal displacement control with a maximum speed of 0.015 m/s to a depth of approximately 0.1 mm ( Fig. 3 ). The same 10 DN gel specimens were tested as in gait mode. They were again kept moist with saline during testing at room temperature. For a given specimen, 10 replicate slow sinusoidal tests were performed at the same spot at intervals of about 20 seconds, shown to be sufficient to allow dimensional recovery. The resultant E* and δ were averaged to produce E* and δ values for a given specimen.

Spherical steel indenter, 3.2 mm in diameter, mounted on a material testing system (MTS Synergie 100, MTS Systems Corporation, Eden Prairie, MN), indenting a 3-mm-thick PAMPS/PDMAAm hydrogel specimen. This configuration was used for the nutrition (N)–related slow sinusoidal microindentation (SSMI) tests.
Suture tear out
There are clinical situations in which cartilage defects are not perfectly contained. In such cases, inserting a simple unsecured plug of repair material is not an adequate repair technique. It is thus an advantage if a repair material can be secured with sutures. DN gels are known to be highly resistant to propagation of a preinduced slit in a standardized test of tear resistance. 24 Therefore, high suture tear-out forces could be expected. To test this hypothesis, again, the MTS Synergie 100 test instrument (MTS Systems Corporation) was used. DN gel specimen dimensions were 3 × 10 × 20 mm; 3 specimens of both types of DN gel were tested. One end of a DN gel specimen was fixed using acrylic adhesive in an aluminum fixture matching the thickness and exceeding the width of the DN gel specimens. A small-diameter (4/0 = 0.15 mm) surgical suture was passed through the other end of the DN gel specimen, laterally centered and approximately 3 mm from the end of the specimen, using the needle integrated with the suture by the manufacturer. The suture was Vicryl 4/0 (Ethicon, Johnson & Johnson Medical GmbH, Neuss, Germany), a type used in fine-scale soft tissue approximation. It is composed of a braided bioabsorbable copolymer (Polyglactin 910 = glycolide-L-lactide random copolymer) coated with another bioabsorbable copolymer (Polyglactin 370 = 65/35 mole ratio lactide-glycolide copolymer). The suture is further coated with calcium stearate to promote easy passage through tissues, precise knotting, and so on. After passage through the gel, the suture was then tied to itself to form a loop. The loop was slipped through a hook fixture attached to the test machine platen. The aluminum fixture was attached to a 100-N load cell attached to the crosshead of the test machine. Specimens were kept moist with saline during testing at room temperature. The crosshead was moved upward at a speed of 1 mm/s, while recording load cell force versus crosshead motion, until the suture completely tore through the DN gel specimen.
The MTS Synergie 100 test instrument (MTS Systems Corporation) was also used for the tissue adhesive tests. The adhesive used was Histoacryl (B. Braun Melsungen AG) (see above). Three specimens of both types of DN gel were tested; the dimensions were 3 × 10 × 20 mm. One end of the specimen was secured with acrylic glue in a slot opening in a small aluminum fixture with slot dimensions matching the thickness and exceeding the width of the DN gel specimens. The fixture was attached to the load cell/crosshead of the test machine. A drop of Histoacryl (B. Braun Melsungen AG) was applied to the other end of the DN gel specimen, which was then lowered to contact a test surface secured to the test machine’s fixed platen. Test surfaces were either ordinary plate glass (precleaned with ethanol) or articular cartilage in the form of osteochondral plugs 7.6 mm in diameter, taken from the knees of 9-month-old swine, obtained from a retail meat vendor. These specimens were fresh frozen in 0.9% saline solution and thawed before testing. The cartilage surface was used without any cleaning except removal of surface moisture with a soft paper tissue. After allowing a minimum 60 seconds for adhesion to become secure, the crosshead of the test machine was raised at a speed of 1 mm/s while recording force and crosshead displacement. The results were normalized to the apparent contact area between DN gels and material surface.
Statistical Analysis
Wilcoxon rank–sum and signed–rank tests were performed on stiffness data (P < 0.05). Statistical analysis was performed and created using R (http://www.R-project.org).
Results
Stiffness
For aggregate modulus E* and loss angle δ, the 2 DN gels were measured in 2 modes: gait-related (G) FIMI and nutrition-related (N) SSMI ( Tables 2 and 3 ; Fig. 4 ). In general, it is expected for viscoelastic materials to have a higher E* and a lower δ at more rapid deformation rates. The E* for both DN gels was significantly higher at the more rapid (G mode) deformation rate. However, δ did not change significantly between the 2 deformation rates employed.
Aggregate Modulus E* of Double-Network Hydrogels in Gait (G) and Nutrition (N) Mode Tests
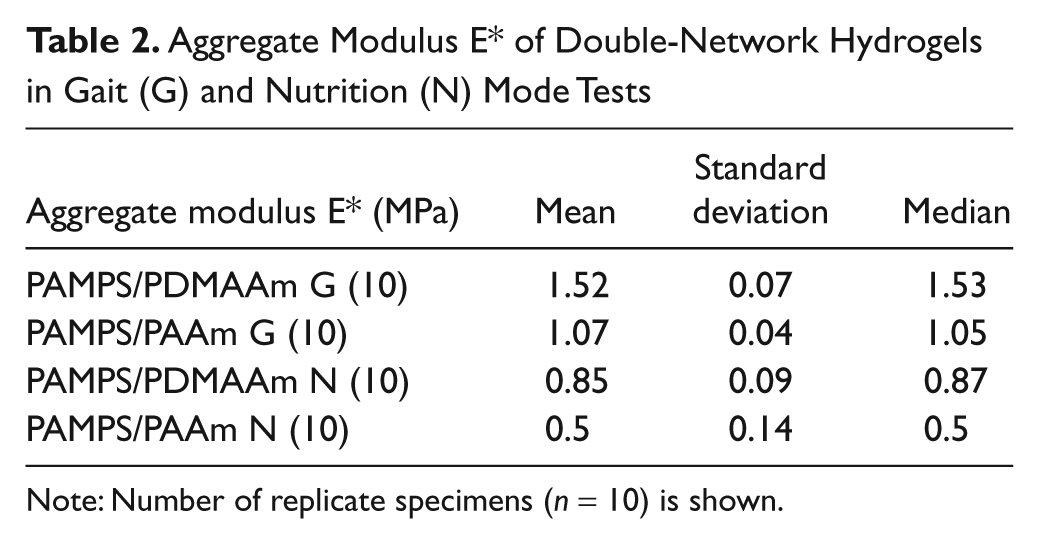
Note: Number of replicate specimens (n = 10) is shown.
Loss Angle δ of Double-Network Hydrogels in Gait (G) and Nutrition (N) Mode Tests
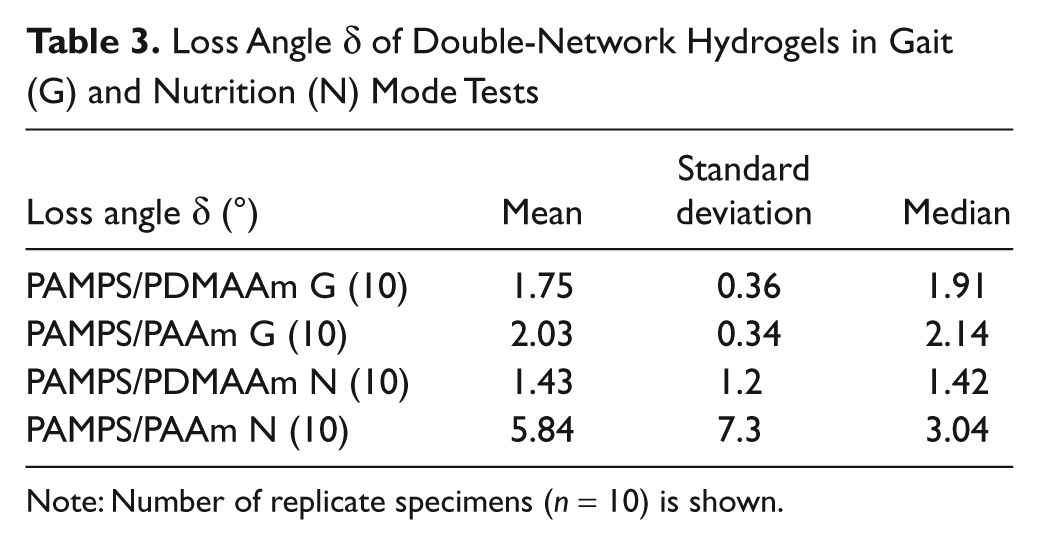
Note: Number of replicate specimens (n = 10) is shown.
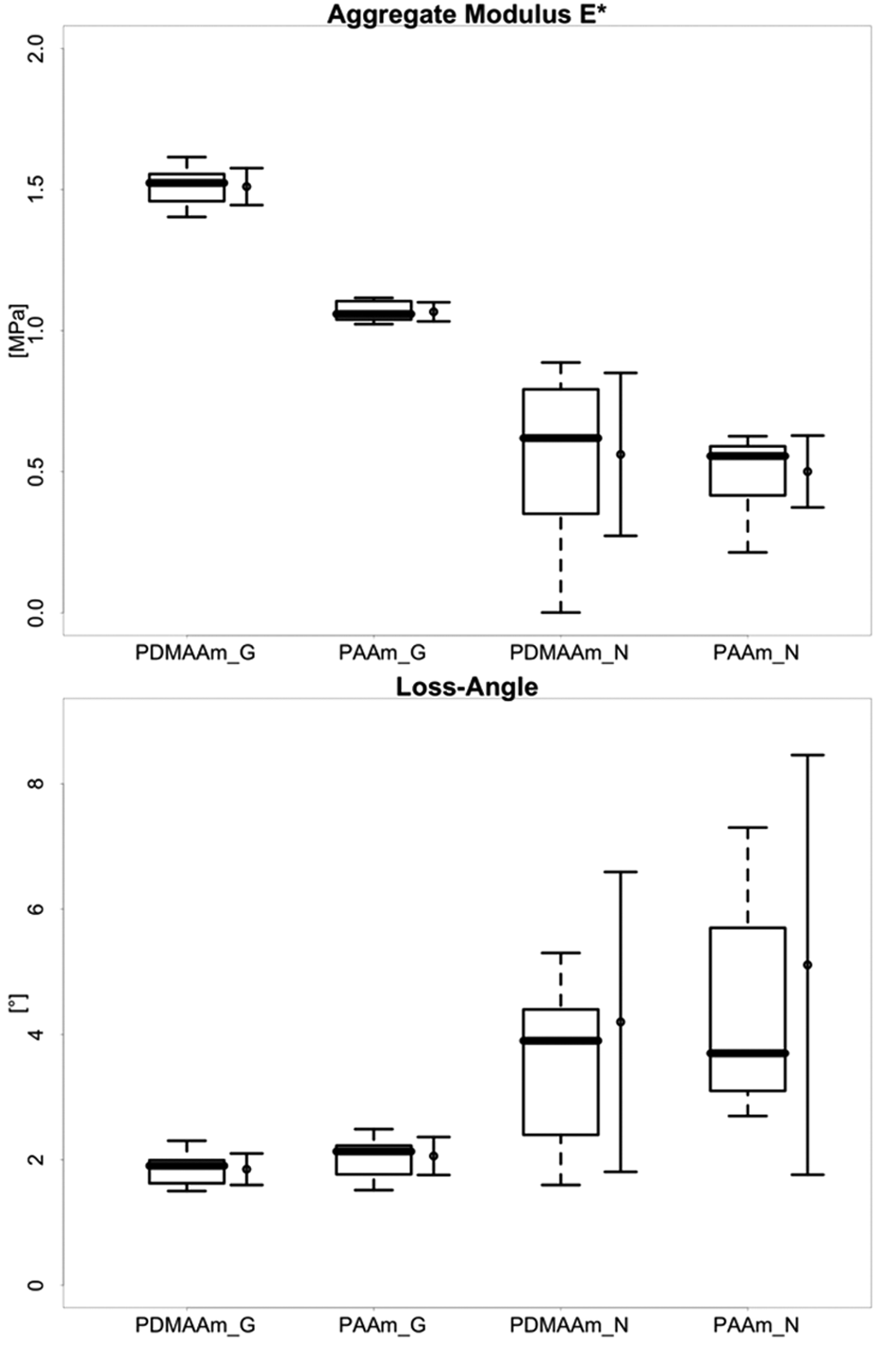
Comparison of stiffness properties of PAMPS double-network hydrogels (DN gels) with different second-network components (PDMAAm and PAAm). Aggregate modulus E* and loss angle δ measured in both gait-mode (G) fast impact microindentation (FIMI) and nutrition-mode (N) slow sinusoidal microindentation (SSMI). See text for complete descriptions of test modes. For each DN gel and mode, the left bar shows box and whisker plot showing median and quartiles, and the right bar shows mean and standard deviation.
A paired Wilcoxon rank–sum test revealed a significant difference for each DN gel between G- and N-mode values for E* but not for δ. The differences between the 2 gels were significant for E* but not for δ.
Suture Tear Out
Recordings of resultant force versus test instrument crosshead position are shown in Figure 5 . In these exploratory experiments, only 3 replicate tests for each DN gel were possible, so statistical comparison of results for the 2 gels was not feasible. The median maximum tear-out force for 94%-water PAMPS/PDMAAm was approximately 2.1 N, and the median maximum for 90.9%-water PAMPS/PAAm was approximately 7.1 N.
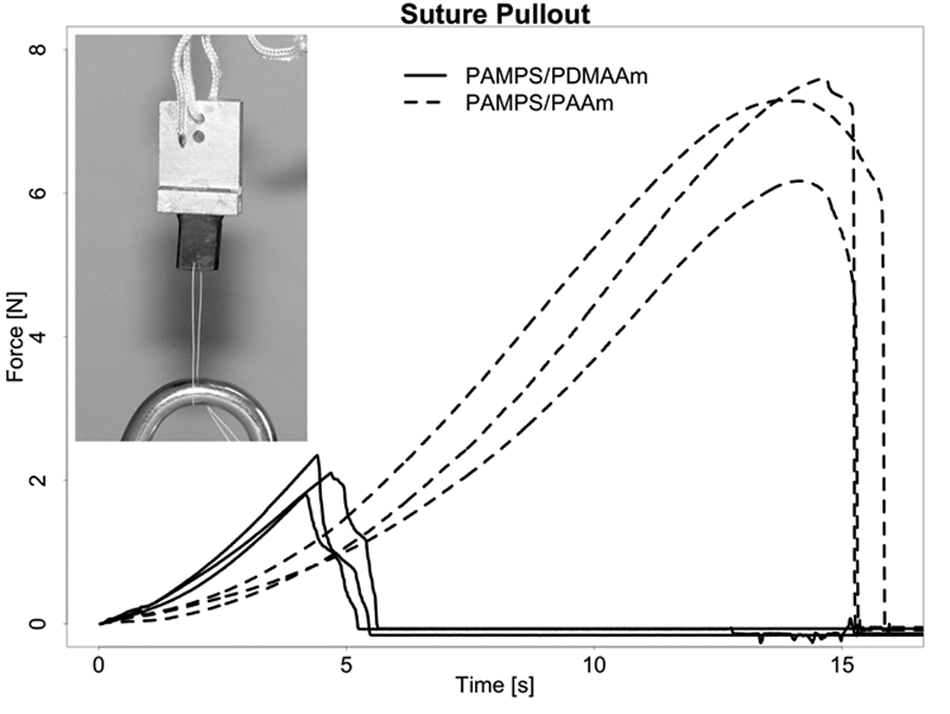
Tear-out forces for 4/0 (0.15-mm diameter) braided suture versus crosshead displacement. Three replicate tests for each 3-mm-thick gel specimen. Median maximum force of PAMPS/PDMAAm was 2.1 N, and median maximum force of PAMPS/PAAm was 7.4 N.
Attachment to Surfaces Using a Tissue Adhesive
Figure 6 shows the force versus crosshead position for the adhesion of DN gels to a glass plate using Histoacryl (B. Braun Melsungen AG) tissue adhesive. Also in these exploratory experiments, only 3 replicate tests for each DN gel were possible, so statistical comparison of results for the 2 gels was not feasible. The median maximum pull-off force of PAMPS/PDMAAm was 0.23 N/mm2 (range, 0.21-0.59 N/mm2), and median maximum pull-off force of PAMPS/PAAm was 0.18 N/mm2 (range, 0.09-0.23 N/mm2). To the naked eye, pull off always occurred at the interface between the gel and the glass surface rather than in the gel substance.
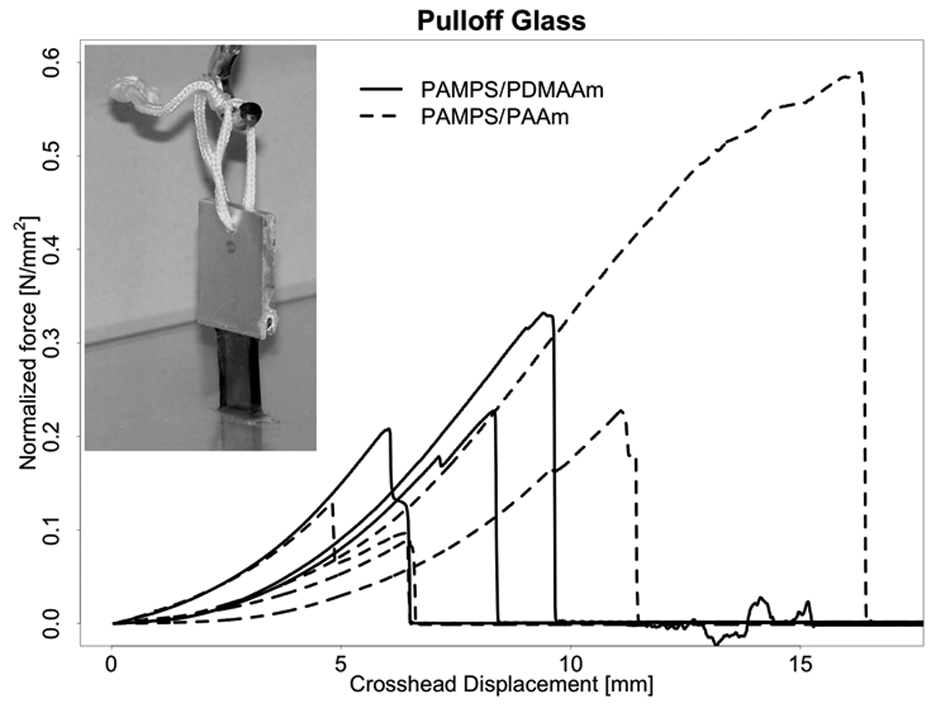
Pull-off force normalized by contact area versus crosshead displacement for double-network (DN) specimens attached to a glass plate using an acrylic tissue adhesive. Nominal area of contact = 3 × 10 mm. Three replicate tests for each DN hydrogel. Median maximum pull-off force of PAMPS/PDMAAm was 0.23 N/mm2, and median maximum pull-off force of PAMPS/PAAm was 0.18 N/mm2.
Similarly, Figure 7 shows force versus crosshead position for the adhesion of DN gels to porcine articular cartilage using Histoacryl (B. Braun Melsungen AG) tissue adhesive. Again, only 3 replicate tests for each DN gel were possible. The median maximum pull-off force of PAMPS/PDMAAm was 0.20 N/mm2 (range, 0.15-0.23 N/mm2), and median maximum pull-off force of PAMPS/PAAm was 0.15 N/mm2 (range, 0.14-0.24 N/mm2). To the naked eye, pull off always occurred at the interface between cartilage surface and the DN gels rather than in either the cartilage or gel substance. It is interesting to note that the forces required to produce adhesion failures approached the forces required to produce single-suture tear out.
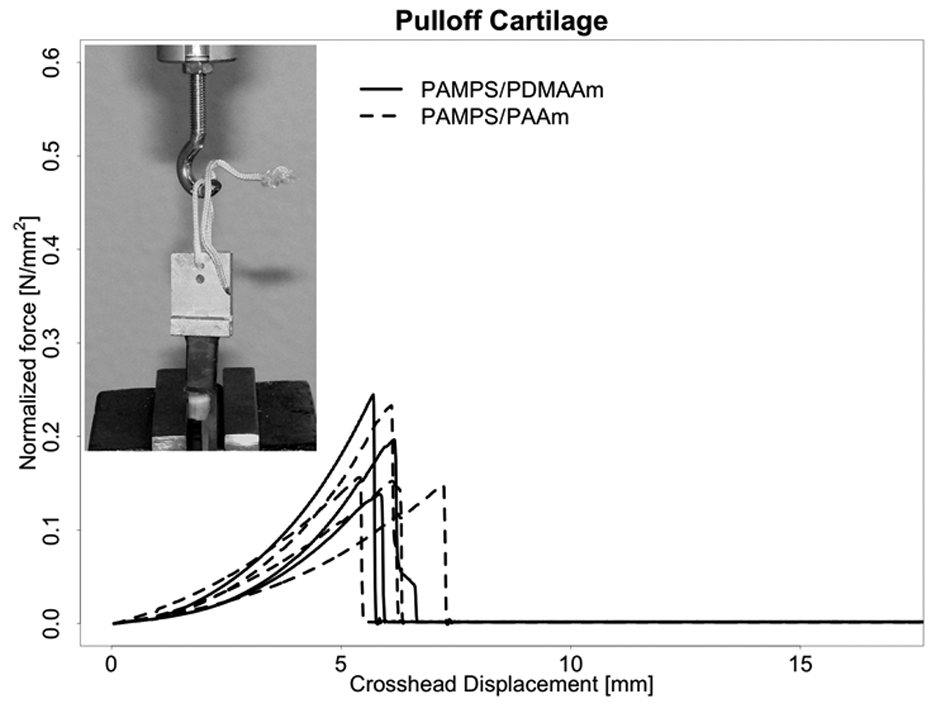
Pull-off force normalized by contact area versus crosshead displacement for double-network hydrogel (DN gel) specimens attached to porcine articular cartilage using an acrylic tissue adhesive. Three replicate tests for each DN gel. Median maximum pull-off force of PAMPS/PDMAAm was 0.20 N/mm2, and median maximum pull-off force of PAMPS/PAAm was 0.15 N/mm2.
Discussion
The DN gels showed very promising results, being almost as stiff as normal cartilage and allowing for a safe fixation to the surrounding tissue, either by suturing or gluing. The results of a given test of fixation (e.g., suture tear out or attachment of specimens to surfaces with tissue adhesive) showed a wide spread. This was due to the fact that it was intended to simulate the variable situation in the operation room. For example, for the attachment with cyanoacrylate glue to cartilage, we dried the surface with a paper towel, added a drop of glue, and placed the gels by hand on the cartilage surface.
Stiffness Parameters
The PAMPS/PDMAAm gel was significantly (~41%) stiffer (higher E*) than the PAMPS/PAAm gel under fast (gait mode) deformation. PAMPS/PDMAAm was also stiffer (~12%) in slow deformation (nutrition mode), but the difference was not significant. In a separate study,16,18 we have evaluated the stiffness of swine knee articular cartilage and meniscus in the same 2 test modes. The DN gel E* values were only about 10% of cartilage values in either G mode or N mode. However, the PAMPS/PDMAAm gel was approximately 50% as stiff as swine meniscus in both G mode and N mode. It seems likely that PAMPS/PDMAAm DN gels with lower water content, closer to cartilage (e.g., 65%-80% rather than >90%), may have E* values more closely approaching that of the articular cartilage surface.
The 2 DN gels exhibit some characteristics of a viscoelastic material because their moduli decline when the deformation rate is decreased (i.e., from G mode to N mode). However, one measure of viscoelasticity, that is, the loss angle, of the 2 gels did not differ significantly, and both DN gels were relatively “rubbery,” that is, more energy storing, in that the loss angle δ was low even at 0.1 Hz in the slow, cyclic N-mode tests. In both modes, median values were below 4° for both gels. In general terms, the low loss angles of these DN gels in slow (0.1 Hz) cyclic deformation can be attributed to the fact that their structures are chemically crosslinked. Therefore, deformation does not entail much sliding between polymer chains, and there is thus little of the attendant frictional dissipation of energy that is known to result from such sliding. This less viscoelastic, more energy-storing character of these acrylamide DN gels is particularly evident in comparison to swine articular cartilage and meniscus. 18 The G-mode loss angle for both these tissues is approximately 12°, and the N-mode loss angle is approximately 37° for cartilage and approximately 26° for meniscus. 18 Thus, the tissues, in spite of being stiffer, can be viewed as better in absorbing and dissipating energy than the 2 DN gels. What difference this might make in implant use and whether it would be possible to produce a DN gel structure comparable to either of these tissues in both dynamic stiffness and viscoelasticity have not been explored yet.
It is interesting to note that the PAMPS/PDMAAm gel was stiffer than the PAMPS/PAAm gel in spite of having a higher water content (94% v. 90.9%) and correspondingly containing only approximately 66% as much polymer. This suggests that the gel stiffness parameters are more strongly influenced by differences in structure related to the use of different second-network components (PDMAAm v. PAAm) than by gel water content.
Surgical Fixation Stability
As mentioned in the Introduction, there are clinical situations in which cartilage defects are not perfectly contained. It is thus an advantage if a repair material can be secured with sutures. The suture tear-out strength experiments showed that both of these DN gels were tear resistant enough to be secured with fine surgical sutures. The suture tear out of PAMPS/PDMAAm was comparable to nasal cartilage pull out of sutures. Farhadi et al. 25 showed for nasal cartilage a suture pull-out force of 4.5 N/mm normalized to the thickness of the specimen. The value for PAMPS/PDMAAm normalized to the thickness is 3.5 N/mm. Perhaps in the suture tear-out tests reported here, the somewhat lower water content (higher polymer content) of the PAMPS/PAAm gel also contributed to increased toughness compared to the PAMPS/PDMAAm gel. The tear-out strength for this DN gel should be seen as truly remarkable considering its 90.9%-water content. The high suture tear-out strength stems from the exceptional fracture energy of acrylamide DN gels, as high as 103 J/m2. In studies of PAMPS/PAAm, it has been shown that achieving such fracture energies depends critically on several factors, and it is believed that the molecular weight of the second-network component is the most important one. If it is above a certain value, the increase in chain entanglement greatly increases the work to fracture. 22
The tissue adhesive pull-off strength tests also showed promising high results for both of the 2 DN gels to either inorganic (silicate glass) or tissue (cartilage) surfaces. The results thus suggest that pull-off strength is more closely related to bonding phenomena between the primary DN gel component (PAMPS in both cases) and the glass or cartilage surface than to any effects related to the second component. Overall, bonding strength of the gels to glass was somewhat higher than bonding to cartilage (0.23 and 0.18 N/mm2 v. 0.20 and 0.15 N/mm2). The values were variable but of the same order of magnitude. The variability of the results may be explained by the relatively big impact of small changes of the contact areas between the DN gels and the surface they were glued to. Also, a small amount of not perfect perpendicularity between glass or cartilage and the DN gel might have influenced the forces measured.
These preliminary results for the adhesion of DN gels to cartilage with tissue adhesive are certainly promising. The force-to-failure values are of the same order of magnitude as for single-suture tear out. On the one hand, multiple sutures might provide even greater tear-out strength, although practically speaking, each suture has the disadvantage of damaging the normal surrounding cartilage. 26 On the other hand, no effort was made here to optimize the adhesion of gel to cartilage; for example, the actual area of cartilage-gel apposition may have been less than the apparent area, and no effort was made to prepare either surface in any special way. It should also be noted that attachment was to a mechanically cut gel surface. Thus, one can conclude that this pull-off strength in this test was due to bonding with the internal gel bulk structure, not just to a bond with as-prepared surface structures. However, the as-prepared surface of DN gels is known to be covered with the second-network component, and thus, adhesion to this surface might well be different. However, it should be noted that retention of water in these DN gels does not depend on a special surface structure. The gels are not observed to leak water either when they are cut or subjected to substantial mechanical deformation for short times. It is certainly a weakness of our study that no more than 3 measurements per DN gel type were recorded, but we felt that this was sufficient data to give a good impression that DN gels can either be glued or sutured to surrounding cartilage and bone.
Concerning the future of PAMPS/PAAm hydrogels for clinical cartilage repair, it may be possible to alter their structure to render them anisotropic and thus further mimic the structure and properties of articular cartilage. Also, as yet unpublished research suggests that cell infiltration is possible. In addition, PAMPS/PAAm hydrogels can be produced in desired shapes and/or trimmed with surgical instruments in the operating room. Finally, sterilization can be accomplished by conventional methods without altering structure and properties. These statements are based on works in progress, and the details of methods and results are reserved for future reports.
Conclusions
The previous work of Gong and her colleagues has already shown that acrylamide-based DN gels have intriguing and promising potential for use in the repair of skeletal system soft tissues. This study was performed to further investigate several mechanical properties related to clinical implant use. The results further support the potential of acrylamide-based DN gels for such use. In spite of their very high water content, >90%, the gels studied exhibited stiffness (E*) values approaching that of swine meniscal tissue.
Suture tear-out strength values approached those for natural cartilage, again in spite of the extremely high water content. Equally intriguing was the finding that the strength of attachment of a cut gel surface to natural cartilage with an acrylic tissue adhesive approached single-suture tear-out strength.
Finally, the double-network structure has obvious parallels to the double-network strategies employed by the body in creating cartilage and other load-bearing soft tissues. Further laboratory mechanical property studies are underway with acrylamide DN gels of much lower water content, similar to articular cartilage.
Footnotes
Acknowledgments and Funding
Funding was provided by
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
