Abstract
The concept of using gene transfer strategies for cartilage repair originates from the idea of transferring genes encoding therapeutic factors into the repair tissue, resulting in a temporarily and spatially defined delivery of therapeutic molecules to sites of cartilage damage. This review focuses on the potential benefits of using gene therapy approaches for the repair of articular cartilage and meniscal fibrocartilage, including articular cartilage defects resulting from acute trauma, osteochondritis dissecans, osteonecrosis, and osteoarthritis. Possible applications for meniscal repair comprise meniscal lesions, meniscal sutures, and meniscal transplantation. Recent studies in both small and large animal models have demonstrated the applicability of gene-based approaches for cartilage repair. Chondrogenic pathways were stimulated in the repair tissue and in osteoarthritic cartilage using genes for polypeptide growth factors and transcription factors. Although encouraging data have been generated, a successful translation of gene therapy for cartilage repair will require an ongoing combined effort of orthopedic surgeons and of basic scientists.
Introduction
Articular cartilage defects and meniscal lesions have a reduced capacity for regeneration. The concept of using gene transfer strategies for cartilage repair originates from the idea of transferring genes encoding therapeutic factors into the repair tissue, resulting in a temporarily and spatially defined delivery of the therapeutic molecule. In this review, we will focus on gene therapy approaches for the repair of articular cartilage and meniscal fibrocartilage, including articular cartilage defects resulting from acute trauma, osteochondritis dissecans, osteonecrosis, and osteoarthritis. Possible applications for meniscal repair will be described for meniscal lesions, meniscal sutures, and meniscal transplantation. As a discussion of cartilage damage resulting from rheumatoid arthritis is beyond the scope of this review, we refer to the many reviews already published on this subject.1-9
Principles of Gene Therapy
Gene transfer is the introduction of foreign genes or gene sequences into different types of cells. Gene therapy is the treatment of diseases using gene transfer techniques. Gene transfer via nonviral vectors is named transfection; gene transfer using viral vectors is termed transduction. The foreign genetic material enters the cell and is next transferred towards the nucleus, where it either integrates into the host genome or remains extrachromosomal as an episome that generally allows only for transient transgene expression. For therapeutic applications, gene transfer into a sufficiently high number of target cells is essential for the secretion of relevant concentrations of the transgene product. Current vectors available for use in gene therapy include nonviral approaches (naked DNA, physical and chemical methods) and various viral (adenoviral, HSV, retroviral, lentiviral, rAAV) vehicles ( Table 1 ).
Nonviral and Viral Gene Vectors Suitable for Gene Transfer to Cartilage Defects

Note: Properties of nonviral and viral gene vectors currently in clinical and experimental use for gene therapy approaches to cartilage defects.
Among the nonviral systems, chemical methods of complexing DNA to various macromolecules include cationic lipids and liposomes,10-12 polymers, 13 polyamines and polyethylenimines,14,15 and nanoparticles, 16 but also calcium phosphate coprecipitates 17 are mainly used. Nonviral systems avoid the risk of acquiring replication competence inherent to viral vectors, can be repeatedly administered, have the capacity to carry large therapeutic genes, are relatively easy to produce on a large scale, and do not elicit a detectable immune response. Nevertheless, their efficacy is often inferior to those of viral vectors. Moreover, the fact that they stay as episomal forms in the target cells often results in short-term transgene expression. To avoid low gene transfer efficacy in vivo, nonviral gene transfer strategies are often based on the transplantation of ex vivo–modified cells to cartilage defects.
Viral vectors utilize natural entry pathways in human cells. Adenoviral vectors have been among the most employed gene vehicles for cartilage repair in the past.18-22 They allow for high transduction efficiencies and transgene expression in a variety of cells, enabling direct approaches in vivo. However, serious concerns about their clinical safety were raised after the death of Jesse Gelsinger, a patient included in a gene therapy trial employing adenoviral vectors. Moreover, transgene expression via adenoviral delivery is limited for about 1 to 2 weeks as the transgenes remain episomal and due to the development of host immune responses against transduction with most of the constructs derived from these viruses.
An advantage of retroviruses is their ability to integrate in the genome of the target, allowing for the replication and maintenance of the transgene over extended periods of time. Yet, this might lead to insertional mutagenesis, with the potential for activating tumor genes. Also, retroviral vectors do not transduce nondividing cells and have a restricted host range. As for nonviral systems, ex vivo approaches with selection of transduced cells are usually required with retroviral vectors23-27 because they are produced only at relatively medium titers and do not exhibit very high efficiencies. Instead, lentiviral vectors, a subclass of retroviruses derived from the human immunodeficiency virus (HIV), can integrate in the genome of nondividing cells. 28 Therefore, such vectors might be good alternatives to the use of retroviruses, as they show also higher levels of transduction in vivo and avoid the need for cell division.29,30 Yet, there are common concerns associated with their application, including the potential for insertional mutagenesis and the psychological problem of introducing genetic material carrying HIV sequences.
Herpes simplex virus (HSV)–derived vectors are large vehicles that can deliver long transgenes to almost all known cell types, including nondividing cells. Although first-generation vectors induced high levels of cytoxicity, recent work has demonstrated that second-generation HSV were less deleterious, in particular for cartilage repair. 31 One problem remains the transient nature of transgene expression mediated by this family of vectors.
In any case, the direct application of viral vectors raises legitimate safety concerns, as potentially infectious agents or sequences (especially lentiviral vectors) might be introduced per se in the body. This is of particular importance for the treatment of cartilage and meniscal lesions that are not life-threatening disorders. In this regard, adeno-associated viral vectors (AAV), which are based on the nonpathogenic, replication-defective human parvovirus AAV, 32 might prove more adequate in direct gene therapy settings. Vectors based on AAV (rAAV) are produced by complete removal of the viral gene coding sequences, making them less immunogenic than adenoviral vectors and less toxic than HSV. Also, the latter vectors generally mediate only short-term expression of the transgenes they carry, whereas rAAV can be transcribed for months to years due to the stabilization of the episomal transgene cassettes by concatemer formation.33-36 Cell division and integration are not required for expression of the foreign material delivered, in marked contrast with retroviral vectors. 37 Redosing of vectors is practicable with rAAV, based on the manipulation of various available serotypes of the virus. For these reasons, rAAV became a preferred gene transfer method for experimental settings in vivo and for clinical applications.35,36,38,39
The greatest obstacle to develop efficient gene transfer protocols targeting sites of articular cartilage and meniscal fibrocartilage damage so far has been the restrained accessibility of the lesions to a treatment. Therefore, the following experimental approaches are currently employed to transfer genes to sites of interest in vivo ( Fig. 1 ):
1. intra-articular injection of the therapeutic formulation, and
2. administration of the therapeutic formulation to the defect via arthrotomy:
2.1. direct application of a gene vector to the repair tissue,
2.2. application of biomaterials carrying a gene vector, and
2.3. matrix-supported application of ex vivo genetically modified cells.
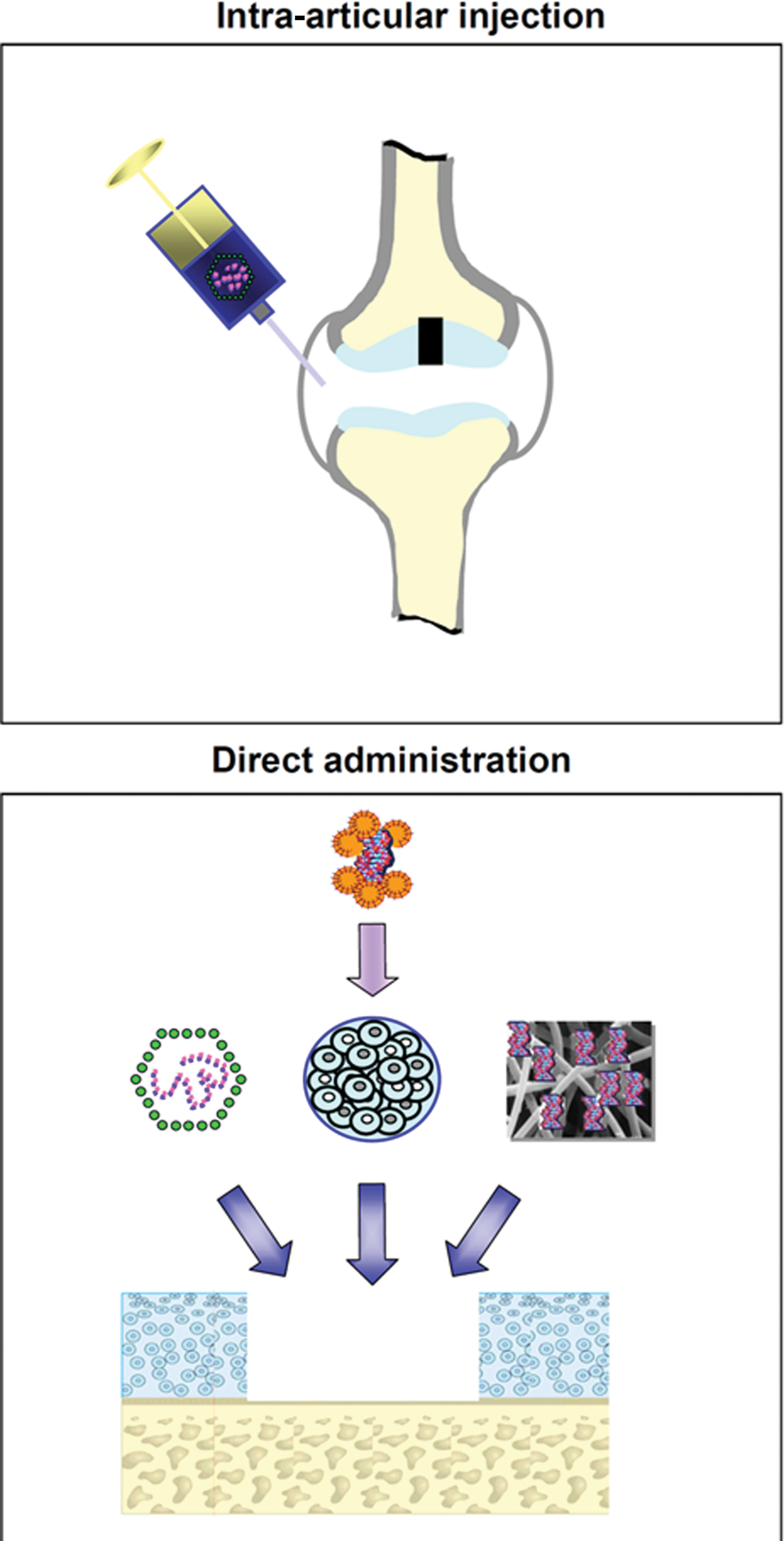
Therapeutic genes may be transferred to sites of articular cartilage damage or to meniscal lesions in vivo via intra-articular injection or by direct application into the lesion. Intra-articular injection (upper panel) of the therapeutic formulation (most often a viral vector) results in a nonselective transduction of many intra-articular tissues. Direct administration of the therapeutic formulation (lower panel) to the target lesion (e.g., an articular cartilage defect) can be achieved by directly applying a gene vector to the repair tissue in the defect (left), by matrix-supported application (e.g., alginate) of target cells (e.g., articular chondrocytes, meniscal fibrochondrocytes, progenitor cells) that were previously genetically modified ex vivo (middle), or by application of a gene vector attached to a biomaterial (right). In vivo, it often includes an arthrotomy.
The target cells in which genes may be transferred include the following:
1. progenitor cells (e.g., resulting from marrow-stimulating techniques or transplanted cells),
2. isolated articular chondrocytes or meniscal fibrochondrocytes that are transplanted into the defect, and
3. cells of the tissues adjacent to the defect:
3.1. articular cartilage: articular chondrocytes from the adjacent cartilage, osteoblasts, and osteocytes from the subchondral bone; and
3.2. meniscal tissue: meniscal fibrochondrocytes, synoviocytes from the synovial lining, and fibroblasts from the joint capsule.
Articular Cartilage
Introduction
Anatomy, Function, and Pathophysiology
Adult hyaline articular cartilage is avascular tand aneural and does not possess a lymphatic drainage. 40 Its major function is to allow for a smooth gliding of the articulating surfaces of a joint and to protect the subchondral bone from mechanical stress. Hyaline articular cartilage is structured in several laminar zones and formed by chondrocytes that are surrounded by an intricate network of extracellular matrix. This cartilaginous matrix is rich in proteoglycans and collagen fibrils composed of type II collagen but also contains types VI, IX, XI, and XIV collagens and a number of additional macromolecules. 41 Normal hyaline articular cartilage contains about 70% to 80% water, which is mainly bound to proteoglycans. Articular chondrocytes synthesize and degrade the extracellular matrix, thereby regulating the structural and functional properties according to the applied loads.
The integrity of articular cartilage can be disrupted as a result of mainly 4 different etiologies. 42 These include focal articular cartilage defects resulting from an acute trauma, osteoarthritis, osteonecrosis, and osteochondritis dissecans. 43 The resulting articular cartilage defect is characterized as being either chondral, involving only the cartilaginous zones, or osteochondral, reaching further into the subchondral bone. 44 Although a chondral defect may be in part repopulated by cells from the synovial membrane,45,46 it usually remains and may expand over time. An osteochondral defect is filled with a blood clot that forms if the bone marrow communicates with the defect.47,48 The pluripotent, undifferentiated mesenchymal cells of the blood clot differentiate into chondrocytes and osteoblasts that later form the cartilaginous repair tissue and the new subchondral bone. However, over time, this repair tissue increasingly exhibits characteristics of fibrocartilage, such as an increased type I and a decreased type II collagen content and may degenerate after several years. 48 If left untreated, secondary osteoarthritis of the joint may result.
Chondrogenic Therapeutic Factors
Strategies for enhancing chondrogenesis in an articular cartilage defect aim at improving the differentiation of mesenchymal cells into chondrocytes for cartilage repair and osteoblasts for the repair of the subchondral bone, the production and maintenance of a new cartilaginous matrix rich in type II collagen and proteoglycans, at increasing the cellularity of the repair tissue to prevent the hypertrophic differentiation of chondrocytes, and at inhibiting articular cartilage degeneration.
Growth and transcription factors are good candidates for these approaches. The therapeutic efficacy of polypeptide growth factors is, however, diminished by their short half-lives.49-51 For example, the fibroblast growth factor-2 polypeptide has a plasma half-life of less than 1 hour and is cleared in some hours after intra-articular administration. 50 To overcome this problem, the idea of applying the gene encoding for a particular therapeutic protein has gained attraction.
Candidate factors to support chondrogenesis include members of the transforming growth factor beta (TGF-β) superfamily such as TGF-β1 and TGF-β2,27,52,53 bone morphogenetic protein 2 (BMP-2),51,53 BMP-7,54,55 members of the fibroblast growth factor family such as the basic fibroblast growth factor (FGF-2), 56 growth/differentiation factor 5 (GDF-5), 57 and the parathyroid hormone–related protein (PTHrP).58,59 Cell proliferation is promoted, among others, by FGF-260,61 and the insulin-like growth factor I (IGF-I). 62 Particularly potent candidates to stimulate matrix synthesis include IGF-I,63,64 BMP-2 and BMP-7, and the cartilage-derived morphogenetic proteins (CDMP).65,66
Transcription factors directly modulate the expression of genes involved in chondrogenesis, such as type II collagen or aggrecan. Experimental models have demonstrated the chondrogenic properties of transcription factors, such as SOX9, 67 Cbfa-1/Runx-2, 68 Cart-1, 69 the Ets family members, 70 and various signaling molecules as well as extracellular matrix glycoproteins themselves.71,72 Another attractive approach is to inhibit degenerative pathways within the repair tissue. Potential targets include cytokines that mediate catabolic events, in particular the members of the interleukin-1 (IL-1), 73 IL-17, 74 and tumor necrosis factor (TNF) 75 families. These strategies are based on the inhibition of the production of matrix-degrading enzymes, 76 proinflammatory mediators, 75 as well as apoptotic mechanisms. 77
Traumatic Articular Cartilage Defects
Intra-articular Injection
Intra-articular injection is a convenient way to target the joint space and has been studied using naked DNA 78 or adenoviral,79,80 retroviral,81,82 HSV, 79 lentivirus, 29 rAAV,81,83,84 and nonviral vectors.79,85 In 1998, Ikeda et al. 80 injected adenoviral vectors encoding for the TGF-β1 gene into the joints of guinea pigs and reported elevated TGF-β1 levels in the synovial fluid for 2 weeks following gene delivery. The effectiveness of a direct intra-articular gene therapy approach in combination with a marrow stimulation technique has been shown by Morisset et al. 86 Full-thickness chondral defects in equine stifle and knee joints were treated by microfracturing, followed by intra-articular application of adenoviral vectors carrying the genes for interleukin-1 receptor antagonist protein (IL-1Ra) and IGF-I. Sixteen weeks postoperatively, articular cartilage defects treated with IL-1Ra and IGF-I showed increased proteoglycan content and type II collagen expression compared with defects treated using a marrow-stimulating technique alone. Yet, articular cartilage defects cannot be specifically targeted with this approach since the transgene is expressed mainly in cells of the synovial membrane and gene transfer into articular cartilage defect is a very rare event. Therefore, many of the gene-based approaches have focused on direct gene vector delivery into a defect exposed by arthrotomy ( Table 2 ).
Therapeutic Gene Transfer Studies to Articular Cartilage Defects In Vivo
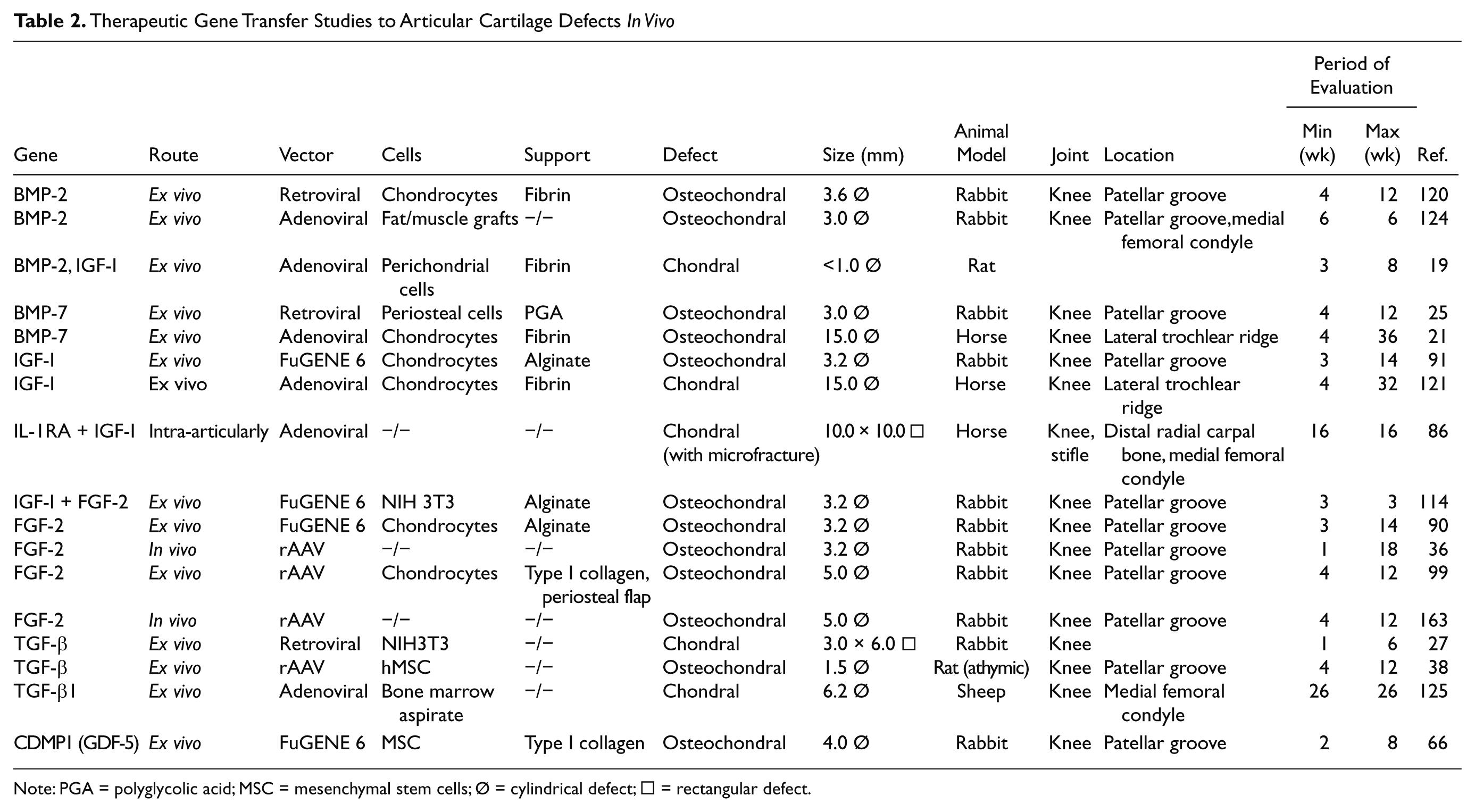
Note: PGA = polyglycolic acid; MSC = mesenchymal stem cells; Ø = cylindrical defect; □ = rectangular defect.
Arthrotomy
Direct application of a gene vector in vivo
The direct delivery of therapeutic genes into cartilage defects in depth has long been arduous due to the reduced capability of nonviral and various viral vectors to penetrate the dense extracellular cartilaginous matrix. Following arthrotomy and gene vector application to cartilage defects, limited transgene expression was observed only in the superficial cartilage layers. 80 With the implementation of rAAV vectors, direct gene transfer to cells within defects and adjacent cartilage has met success. Reporter gene studies demonstrated efficient transgene expression in normal and osteoarthritic human articular chondrocytes within their native matrix in situ to depths relevant for clinical applications. 87 Moreover, transgene expression was also present in chondral and osteochondral articular cartilage defects in vivo for at least 4 months. 87 rAAV vectors have been manipulated recently to deliver therapeutic genes such as FGF-2 directly into osteochondral cartilage defects. 36 Cartilage repair was significantly enhanced 4 months after vector application. 36
Application of biomaterials carrying a gene vector into defects
In order to avoid a dilution of the therapeutic agents, gene vectors or modified cells can be delivered in conjunction with biomaterials such as fibrin, collagen, gelatin, carbohydrate-based polymers (polyactic acid/polyglycolic acid, hyaluronan, agarose, alginate, chitosan), and artificial polymers (dacron, teflon, carbon fibers, polyestherurethane, polybutyric acid, polyethylmethacrylate, hydroxyapatite).45,88 When preparations of adenoviral vectors carrying a marker gene were adsorbed onto type II collagen-glycosaminoglycan matrices and implanted into osteochondral defects, transgene expression was present until day 21. 89
Application of ex vivo genetically modified cells
The direct transplantation of cells genetically modified ex vivo involves their isolation, genetic modification, and reimplantation into articular cartilage defects. These modified cells can be applied without (e.g., as coagulated bone marrow aspirate) or with supportive matrices. Such components include alginate,90-92 agarose,93,94 fibrin or type I collagen gels without95-97 or with a periosteal flap,98,99 and synthetic biodegradable scaffolds.100-102 Kang et al. were the first to transplant genetically modified cells into an articular cartilage defect in vivo. 103 In this study, chondrocytes were transduced with a retroviral vector. Other studies used nonviral,104-106 adenoviral,89,96,107 retroviral,103,108-111 and rAAV vectors 112 to deliver marker genes in defects via ex vivo–modified cells. Although engineered chondrocytes are generally transplanted,21,96,103,104,106,111,113 fibroblasts,27,114 perichondrial, 105 periosteal,108,112 or muscle-derived cells 109 have been also applied. The data from these studies showed that transgenes can be expressed in cartilage defects via ex vivo strategies, remaining active for about 1 month. This is significantly longer compared with the application of recombinant proteins ( Table 2 ). Figure 2 depicts improvements in the repair of osteochondral defects following combined gene transfer of IGF-I and FGF-2 compared with the application of a marker gene (lacZ) to NIH 3T3 fibroblasts. 114
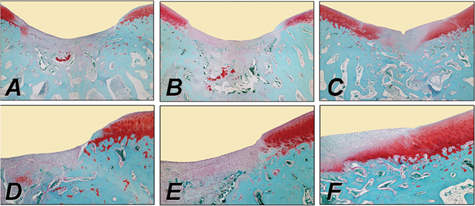
Improvement of cartilage repair in a rabbit osteochondral defect model in the trochlear groove by combined ex vivo gene transfer of human insulin-like growth factor I (hIGF-I) and fibroblast growth factor-2 (hFGF-2) in NIH 3T3 fibroblasts that were then embedded in alginate spheres and transplanted into the defects. Histological appearance of osteochondral defects following treatment with a lacZ implant (left column:
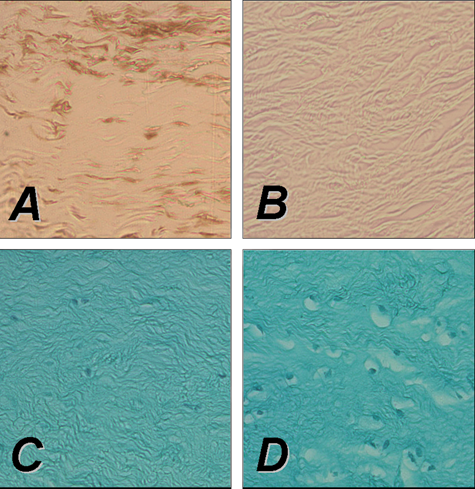
Direct rAAV-mediated gene transfer to rabbit meniscus explants in vitro using an rAAV-lacZ (left panel) or rAAV-hFGF-2 vector (right panel) (50 mL each vector). Persistent transgene expression after 10 days in vitro in meniscal explants following immunohistochemical detection of lacZ (
Periosteal cells transduced by a BMP-7 retroviral vector and attached to a polyglycolic acid scaffold improved cartilage repair at 8 and 12 weeks in vivo. Interestingly, this was the first study in which a growth factor gene was transferred into a focal defect. 25 Since, many reports described the use of a variety of therapeutic genes like BMP-2, BMP-7, IGF-I, FGF-2, and TGF-β.22,90,91,114-120 Significant improvement in articular cartilage repair was noted in these reports ( Table 2 ). Although most of the evaluations were carried out in small animal models, Hidaka et al. 21 and, more recently, Goodrich et al. 121 performed arthroscopic implantation of chondrocytes genetically engineered by adenoviral transduction with the BMP-7 21 or IGF-I 121 gene in horses.
On the basis of such encouraging data, cartilage repair was addressed by matrix-supported implantation of genetically engineered mesenchymal stem cells (MSC). Kuroda et al. 122 implanted BMP-4–transduced MSCs using fibrin glue in full-thickness cartilage defects in the trochlear groove of rabbit femurs. After 24 weeks, histological scoring of the defects revealed significantly better cartilage repair in the BMP-4 treatment group compared with defects receiving lacZ-transduced MSCs. Guo et al. 123 seeded TGF-β1–engineered MSCs onto poly-L-lysine–coated polylactide scaffolds in vitro and allografted them into full-thickness defects in rabbits. This resulted in improved joint repair with regard to extracellular matrix formation, reconstitution of the subchondral bone, and inhibition of inflammatory immune responses. Repair of osteochondral defects was also enhanced by transplantation of MSCs transfected with the CDMP1 gene, applying a lipofection method. 66
A novel method of gene therapy for the repair of osteochondral defects has recently been published by Evans et al. 124 Rather than genetically modifying isolated cells, this technique describes gene transfer to biopsies of muscle and fat. An adenovirus vector carrying cDNA encoding human BMP-2 was used for genetic engineering of tissues. These gene-activated muscle or fad pads were transplanted into osteochondral defects in rabbits. Histological analysis after 6 weeks revealed the formation of a proteoglycan-rich articular surface with subchondral bone beneath and good union with the adjacent cartilage.
Ivkovic et al. 125 used autologous bone marrow, transduced ex vivo, with adenoviral vectors containing the cDNA for TGF-β1. Implantation of the marrow clot improved the histological, biochemical, and biomechanical parameters of partial-thickness chondral defects in sheep at 6 months.
Osteoarthritis
Osteoarthritis (OA) is the leading, most disabling human condition and prevalent form of arthritis (80%), impairing the quality of life of millions of people worldwide. OA is a chronic disorder of diarthrodial joints, mainly characterized by a slow, gradual deterioration of the articular cartilage that remains without effective treatment to date. OA not only affects the cartilage but also the subchondral bone and, to a minor degree, the synovial lining, ligaments, tendons, and muscles. Current options to manage OA, such as pharmacological therapy and reconstructive surgical interventions, do not allow for the restoration of a native cartilage. OA is a complex disorder characterized by an activation of inflammatory cascades at the molecular level, leading ultimately to cartilage breakdown, associated with alterations of the phenotype of chondrocytes and a loss of the major components of the cartilage matrix. Under mechanical or biochemical stress (presence of IL-1 and TNF-α, NO, prostaglandins, matrix degradation products), the chondrocytes undergo pathological changes in their gene expression patterns that lead to an impairment of the overall homeostasis, with diminished production of normal cartilage matrix molecules (proteoglycans, type II collagen), enhanced production of matrix-degrading enzymes (MMPs and adamalysins, including ADAMs and ADAMTs), and decreased responsiveness to reparative stimuli, ultimately leading to the degradation of the matrix and cell senescence and apoptosis (NO, Fas/FasL signaling) by alteration of cell viability.
Gene Transfer In Vitro
Target cells in the joint include cells of the synovial lining, chondrocytes, chondroprogenitor cells, and surrounding tissues (bone, muscle, tendons, ligaments, meniscus). Application of nonviral,12,79,90,91,126-135 adenoviral,79,126,136-154 or retroviral vectors25,79,126,136,140,155-159 has been achieved in these cell types with more or less success. Instead, RAAV vectors are potent alternatives as they can efficiently and durably transduce synoviocytes,160-164 chondrocytes,36,87,99,165-168 MSCs,36,38,169,170 and cells of surrounding tissues.126,171-177
Regeneration of a normal structural and functional cartilage might be achieved by the following:
inhibiting inflammatory and catabolic pathways,
stimulating anabolic pathways to rebuild the matrix,
impeding cell senescence,
avoiding the pathological formation of osteophytes,
prevention of apoptosis, and
influencing several of these processes.
Inhibition of catabolic pathways has been achieved in vitro by expressing inhibitors of matrix-degrading enzymes (tissue inhibitor of metalloproteinases, i.e., TIMP),178,179 inhibitors of proinflammatory cytokines (IL-1Ra, the soluble receptors sIL-1R or Soluble Tumor Necrosis Factor Receptor),107,137,150,155,162 and chondroprotective cytokines (IL-4, IL-10).160,180,181 Activation of anabolic processes in vitro has been noted by single or combined administration of components of the cartilage matrix or of the enzymes that synthesize them,182,183 of growth factors and receptors (IGF-I, FGF-2, BMPs, TGF-β),36,127,131,138,143-145,147,150,151,181,184,185 and of transcription factors (SOX family of DNA-binding proteins, i.e., SOX5, SOX6, SOX9).130,136,149,159,167,168 Restoration of cell vitality and activation of proliferation in vitro have been achieved by application of IGF-I and FGF-2,36,90,127,131,168,185 telomerase (hTERT), 186 of inhibitors of apoptosis (bcl-2), 187 or of HSP70. 132 Interestingly, approaches that influence several of these processes have been also successfully attempted, like combining the transfer of inhibitors of catabolism pathways and of activators of anabolic events (IGF-I/IL-1Ra or IGF-I/IL-4),150,151,181 as well as that of activators of anabolic and proliferative processes (FGF-2/SOX9 or FGF-2/IGF-I). 168
In Vivo Direct Gene Transfer
The key issue in establishing an efficient therapy against OA is the accessibility of the targets to the treatment when they reside in the joint cavity. The following approaches have been developed to deliver a molecular composition:
systemic delivery, and
intra-articular administration (via injection or arthrotomy).
Systemic approaches are better suited to target diseases that are systemic in nature like rheumatoid arthritis (RA).164,188-190 Local administration of components might be preferable in the case of OA that affects only a limited number of joints without major extra-articular or systemic manifestations. The foreign material may be delivered directly (gene vector preparation) or indirectly (genetically modified cells).
Several lines of evidence have demonstrated that intra-articular injection of most vector types leads to a preferential transduction of the synovium,29,79,81,191 being more suited for strategies aiming at inhibiting inflammatory and catabolic pathways and a common approach employed against experimental RA. Successful attempts towards these goals have been reported by direct application of vectors coding for IL-4,35,192 IL-10,193,194 sTNFR alone 162 or combined with IL-10, 195 IL-1Ra alone18,20,34,155,196,197 or combined with sTNFR, 20 antagonists and inhibitors of TGF-β and of the BMPs, 198 HSP70, 132 gene expression silencers, 199 and kallistatin or thrombospondin-1.200,201
Yet, even if cartilage breakdown can be contained, this will not be sufficient to fully compensate for the loss of matrix elements and cells noted during the disease progression. In this regard, increased synthesis of cartilage matrix components has been documented following injection of vectors carrying genes for anabolic factors (IGF-I). 202
Ex Vivo Indirect Gene Transfer
Although more complex, ex vivo gene therapy is considered safer because no free vector particles are introduced in the body. Modified cells can be extensively controlled, tested, and selected while maintained in culture. Administration of cells is also a means to increase the cellularity like needed for severe OA.
Synoviocytes have been predominantly employed to deliver inhibitors of inflammatory and catabolic processes.203-208 Such pathways could be regulated by injecting synoviocytes transduced to overexpress an IL-1Ra alone203-208 or combined with IL-10. 208 Also, dermal fibroblasts have been modified for this purpose to overexpress an IL-1Ra, sTNFR, or a combination of both. 209
Reduced severity of the induced arthritis was associated with a decrease in cartilage breakdown, but complete resurfacing was not achieved. Successful attempts to promote the formation of new cartilage have been made by administrating dermal fibroblasts modified to express BMP-2. 146
Still, preparation of terminally differentiated cells from unaffected sites remains invasive, with a limited supply, and represents an additional burden for the patient. Also, committed cells generally undergo major phenotypic changes upon passaging in culture, especially chondrocytes. Multipotent cells might be more suited for transplantation purposes, possibly leading to the production of a cartilage surface of enhanced quality compared with committed cells that lead to the formation of a poorly differentiated fibrous cartilage. Progenitor cells can be easily isolated from multiple tissues (bone marrow, periosteum, perichondrium, muscle, fat, subdermis, cartilage, bone, synovial membrane, ligaments), even in OA patients, maintaining a multilineage potential with a reliability for differentiation and a capacity for expansion.210,211 Indeed, injection of muscle-derived stem cells modified by combined gene transfer of BMP-4 with sFlt1 (a vascular endothelial growth factor (VEGF) antagonist) allowed for cartilage repair in a rat model of OA.212,213
Osteonecrosis
Osteonecrosis (ON) is primarily a disease of the subchondral bone that secondarily affects the articular cartilage. Initially, a vascular insult is thought to cause an interference of the microcirculation of the subchondral bone, resulting in an edema that leads to an increased intraosseous pressure. This leads to ON of the affected segment of the subchondral bone, which may result in a subchondral insufficiency fracture, destabilizing the overlying articular cartilage and eventually resulting in its collapse and the creation of an osteochondral defect. Treatment options consist of conservative therapy in early stages. Precollapse lesions can be treated with retrograde core decompression, while later-stage lesions presenting with osteochondral defects require osteochondral transplants and/or osteotomies, or ultimately, partial or total knee arthroplasty. 214
Possible experimental gene therapy approaches need to be stage dependent, focusing on early stages (when the articular cartilage is not compromised) at the revascularization of the necrotic bone, while at the stage of osteochondral lesion, only gene-enhanced osteochondral transplants might be useful. Katsube et al. 214 applied gene transfer of VEGF, to accelerate revascularization of the necrotic bone. Using an adenoviral vector encoding for VEGF, endothelial cells of the rabbit saphenous arteries were transduced. These gene-modified arteries were then placed with its venae comitantes into necrotic iliac crest bone in vivo. Angiogenesis in the necrotic bone was quantified by bone blood flow measurement and assessment of vessel density following microangiography. The extent of neoangiogenesis was significantly greater in the VEGF group than the control group, reflected in an increased capillary density, length of newly formed capillaries, and increased bone blood flow at 1 week postoperatively. While this study was restricted to the bone of the iliac crest, it might serve as a paradigm for the treatment of ON in a subchondral location. Such a therapy may allow the healing of avascular necrosis before fracture and subchondral collapse occur, preventing the articular cartilage from damage. More studies with time points longer than the 1-week evaluation are needed, preferentially performed in animal models of subchondral ON, such as the femoral condyles of the knee joint, its second most common location.
Osteochondritis Dissecans
Osteochondritis dissecans (OCD) usually affects children and young adults and occurs mainly in the knee joint, characteristically in the lateral aspect of the medial femoral condyle. Possible etiological factors beside a genetic predisposition include ischemia and epiphyseal abnormalities with subsequent necrosis. For example, disruption of epiphyseal plate vessels may lead to localized avascular necrosis. Its revascularization usually occurs with the formation of a scar tissue, absorption of necrotic fragments, intertrabecular osteoid deposition, and remodeling with new bone formation. When revascularization is delayed, an OCD lesion can occur. Clinical treatment principles focus on stimulation of revascularization or removal of necrotic subchondral bone together with its restoration (e.g., using autologous bone transplants), beside the surgical fixation of an unstable osteochondral fragment. 214
So far, no experimental gene-based treatment has been proposed for the treatment of OCD. In theory, the same principles apply for the revascularization of necrotic subchondral bone, as already outlined for ON with subsequent articular cartilage defects. It may be also possible to enhance the surgical fixation of an osteochondral fragment by applying osteoinductive genes such as the BMPs to the subchondral bone–osteochondral fragment interface to improve integration of the osteochondral fragment. It is unclear whether the integration of a chondral fragment may be achieved, a rare indication currently favored only for surgical refixation of large fragments in juvenile patients. 215 Likewise, gene-modified osteochondral transplants may be applied at later stages of deep osteochondral defects.
Meniscal Fibrocartilage
Anatomy, Function, and Pathophysiology
The menisci are semilunar fibrocartilage structures that transmit weightbearing forces and increase stability, facilitate nutrition and provide lubrication for the articular cartilage, and promote knee proprioception.216,217 As the medial meniscus is less mobile during joint motion,218,219 injuries are much more common compared to the lateral meniscus. 220 Type I collagen is the predominant collagen of the meniscal tissue. 221 It is arranged with a circumferential orientation with interspersed radially oriented fibers. 222 The central parts of the menisci are mainly constituted of fibrochondrocytes, whereas fibroblasts are the predominant cell type in the peripheral regions. 223 Meniscal blood supply is restricted to the peripheral 10% to 25% of the meniscal tissue.224,225 Nourishment in the central area is provided only by diffusion of the synovial fluid, 226 perhaps playing a role in the poor healing capacity of central lesions.225-227 Gene transfer strategies may be applied for the following:
1. meniscal repair, and
2. meniscal reconstruction, using
2.1. meniscal substitutes, and
2.2. meniscal allografts.
Meniscal Repair
Meniscal tears are common228,229 and predispose the affected joint to develop secondary OA. 230 Tears of the meniscus in the vascularized peripheral parts can be repaired by sutures, while tears of the central avascular parts are treated by arthroscopic partial meniscectomy.
Gene Transfer Strategies: In Vitro Studies
Gene transfer strategies for the repair of meniscal tears focus on the delivery of therapeutic agents, for example, growth factors, to the site of the meniscal lesion. This can be performed either via direct application of gene vectors or by transplantation of genetically modified cells overexpressing therapeutic genes. Treatment of meniscal fibrochondrocytes with recombinant growth factor proteins such as the platelet-derived growth factor AB (PDGF-AB),231-234 FGF-2,177,235-237 IGF-I,235,238-240 TGF-β1,152,233,239,241,242 BMP-7, 233 or TGF-β3 235 has been shown to improve the phenotypical and biochemical properties of the cells in vitro. Fibrochondrogenesis of stem cells is enhanced by incubation with growth factors such as TGF-β1 243 or TGF-β3 in combination with BMP-4. 244 The possible application of gene transfer strategies in meniscal repair has first been investigated by Goto et al. 139 The lacZ marker gene was transferred to meniscal cell cultures using retroviral and adenoviral vectors. In a next step, the marker gene was applied to human meniscal fragments and whole lapine menisci using direct adenoviral gene transfer and transplantation of meniscal fibrochondrocytes transduced with a retroviral vector. Transgene expression was detected in meniscal explants following ex vivo gene transfer for at least 20 weeks. Successful transfer of the lacZ marker gene was also achieved by rAAV-mediated transfer into human and lapine fibrochondrocytes in vitro. 173 Encouraged by these findings, in 2000, the group of Chris Evans transferred the gene encoding for TGF-β1 158 to meniscal cells in vitro, resulting in enhanced synthesis of proteoglycans and collagen. Zhang et al. used a lipid-based gene transfer system to deliver the gene encoding for human IGF-I to meniscal fibrochondrocytes, yielding accelerated proliferation and differentiation of the modified cells. 134 Recently, we tested the hypothesis that overexpression of FGF-2 through rAAV vectors leads to detectable metabolic changes in human meniscal fibrochondrocytes and inside defects of human meniscal explants. 177 Application of the rAAV-hFGF-2 vector allowed for enhanced cell proliferation and survival in vitro (Figure 3). The idea of applying gene therapy protocols to deliver fibrochondrogenic agents to meniscal tears was supported by a significant reduction of the amplitude of meniscal tears after FGF-2 treatment in this study. 177
Gene Therapy: In Vivo Studies
Only few reports have evaluated the feasibility of gene therapy strategies to enhance the repair of meniscal tears in vivo. Experimental studies have shown that repair in the central part of the meniscus can be promoted by various chemotactic and mitogenic stimuli delivered by an autologous fibrin clot245,246 or a free graft of synovium247,248 in vivo. In a sheep model, longitudinal tears of the anterior horn of the medial meniscus were sutured using VEGF-coated sutures. Interestingly, meniscal repair was not enhanced in the VEGF treatment group.249,250 In 1999, methods of direct and indirect gene transfer to meniscal lesions were compared. 139 In a lapine model, a suspension of adenoviral vectors carrying the lacZ marker gene was mixed with whole blood, and the clot was inserted into 2-mm-long incisions in the medial meniscus. In the same study, using a canine model, retrovirally transduced allogenic meniscal fibrochondrocytes carrying the lacZ gene were embedded in collagen gels and transferred to partial-thickness circular defects (depth, 3 mm; diameter, 2 mm) in the medial meniscus. Gene expression persisted for at least 3 weeks in the lapine model but for 6 weeks within the transplanted meniscal fibrochondrocytes in the canine model. In another animal study, 173 longitudinal incisions were created in the avascular zone of the medial meniscus of rabbits. When rAAV-lacZ constructs were injected intralesionally, X-Gal staining was present by day 20 postoperatively, the longest time point evaluated.
Meniscal Reconstruction
Meniscal Substitutes
Meniscal substitutes have been proposed as a means to overcome problems associated with meniscal allografts and to promote meniscal repair of segmental defects, for example, resulting from a partial meniscectomy.251,252 Meniscal substitutes already in clinical use are based on porous matrices of type I collagen/glycosaminoglycan (Menaflex, ReGen Biologics, Hackensack, NJ)253,254 or polyurethane (Actifit, Orteq, London, UK).255,256
The feasibility of genetic engineering of meniscal fibrochondrocytes has already been described above. However, in the treatment of circumscribed meniscal defects, direct gene vector administration into injured knee joints may be difficult to achieve because a loss of the bradytrophic meniscal tissue may hardly be restored by local cells, even after administration of mitogenic and anabolic genes. Therefore, gene therapy in the treatment of meniscal defects may need to be used in combination with the transplantation of modified cells or tissues.
Tissue engineering involves the combination of cells, engineered extracellular matrices, and biologically active molecules for tissue regeneration.257,258 Over the last 2 decades, numerous tissue engineering strategies have emerged for the replacement of meniscal tissue.259-261 In general, 2 basic approaches for meniscal replacement can be distinguished:
Several concepts for treating circumscribed meniscal defects concentrate on meniscal replacement by acellular matrices,259,266-269 avoiding possible risks associated with transplantation of human allografts (e.g., failure rate, immunoreaction, 270 disease transmission 271 ). Different types of meniscal substitutes, such as decellularized allogenic and xenogenic grafts,262,263,272,273 collagen grafts,253,274 permanent synthetic scaffolds, 251 and biodegradable scaffolds based on small intestine submucosa,275-278 poly-lactic acid (PLA), or poly-glycolic acid (PGA),279-282 have been used in experimental and clinical studies. However, after transplantation of acellular meniscal constructs into defects, the transplants are populated by synovial fibroblasts, resulting in a scar tissue with poor biomechanical properties.245,283 Therefore, some tissue engineering approaches focus on additional cell-seeding techniques prior to transplantation.251,284 Meniscal cells,282,285 articular chondrocytes,286,287 synovial fibroblasts, 288 and MSC 289 have been proposed as potential cell sources and have been cultivated in vivo and in vitro on various matrices. 267 In addition, different environmental factors such as growth factors have been used to optimize cell proliferation in vitro. 290
Gene therapy may aid to further enhance the fibrochondrogenic potential of tissue-engineered transplants. In 2002, Hidaka et al. 291 applied a gene transfer protocol to enhance the vascularization and blood supply of cell-seeded bioengineered meniscus transplants. Bovine meniscal cells overexpressing hepatocyte growth factor (HGF) were seeded onto PGA scaffolds and transplanted subcutaneously in athymic nude mice for 8 weeks. Ink injection studies showed that HGF-treated meniscal cells formed a tissue that contained significantly more blood vessels than the controls. In another preliminary ex vivo study, Steinert et al. 152 transduced primary meniscus cells and bone marrow–derived MSCs with adenoviral vectors encoding for marker genes or TGF-β1. Modified cells were seeded in type I collagen-glycosaminoglycan (GAG) matrices and transplanted into defects of bovine menisci explants. In vitro, the vectors efficiently transduced meniscal cells and MSCs, and transgene expression remained elevated after incorporation of the cells into matrices. Transfer of TGF-β1 increased the fibrochondrogenic potential of modified cells, and transplantation of the TGF-β1–transduced constructs resulted in satisfactory filling of the lesions ex vivo ( Table 3 ).
Therapeutic Gene Transfer Studies to Meniscal Cells In Vitro or Meniscal Tissue Ex Vivo and In Vivo
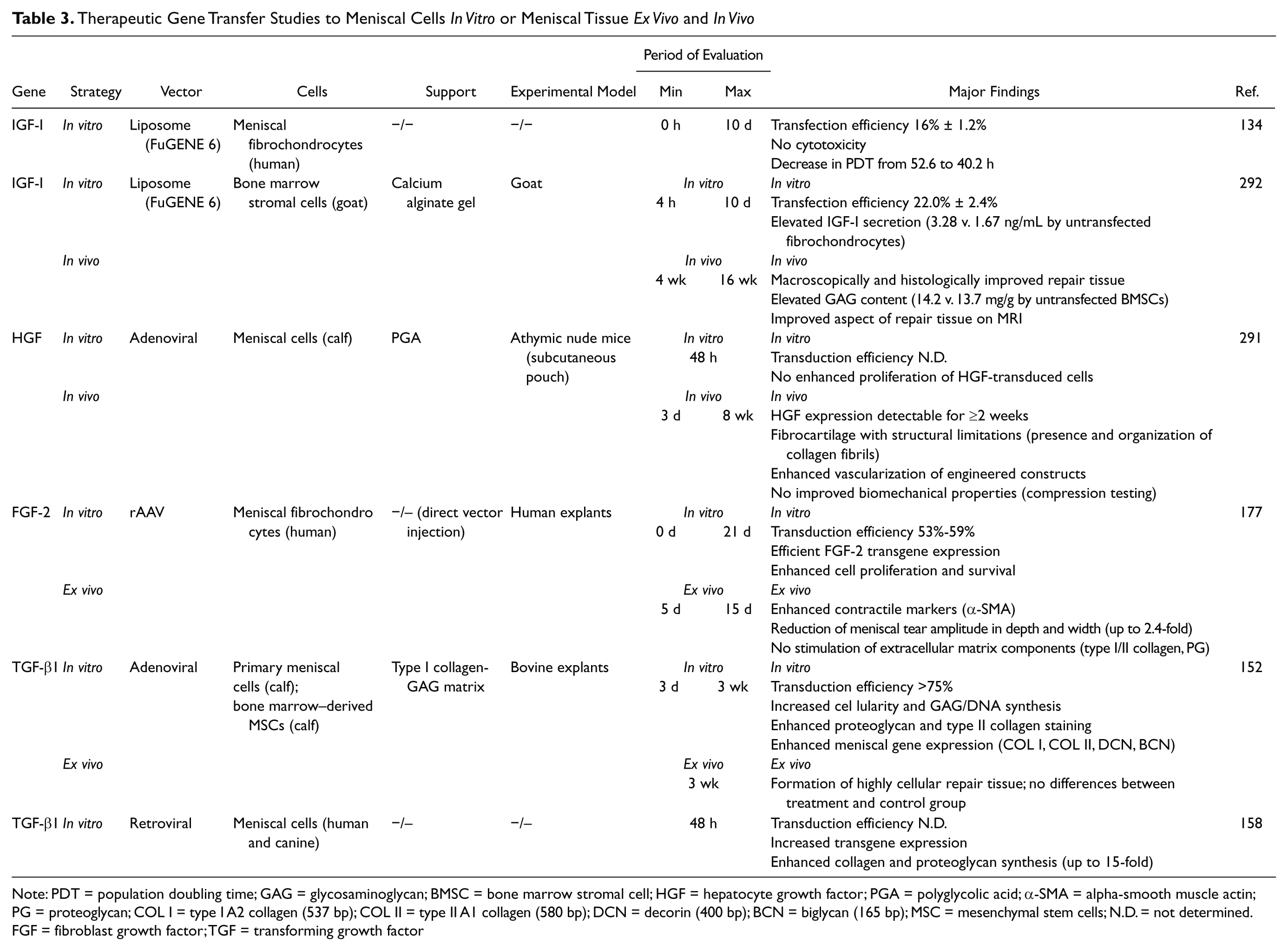
Note: PDT = population doubling time; GAG = glycosaminoglycan; BMSC = bone marrow stromal cell; HGF = hepatocyte growth factor; PGA = polyglycolic acid; α-SMA = alpha-smooth muscle actin; PG = proteoglycan; COL I = type I A2 collagen (537 bp); COL II = type II A1 collagen (580 bp); DCN = decorin (400 bp); BCN = biglycan (165 bp); MSC = mesenchymal stem cells; N.D. = not determined.
FGF = fibroblast growth factor; TGF = transforming growth factor
A recent in vivo work on the use of gene transfer to enhance meniscal repair has been published by Zhang et al. 292 Following an indirect gene therapy approach without tissue engineering features, the authors created full-thickness meniscal defects in the avascular area of the anterior horn of the medial meniscus in a goat model. Bone marrow stromal cells were transfected with the gene encoding for human IGF-I using a nonviral transfection system (FuGENE 6) and suspended in calcium alginate prior to injection into the meniscal defects. After 16 weeks, the resulting repair tissue was improved according to MRI and histological and biochemical evaluation and compared with the controls ( Table 3 ).
Meniscal Allografts
Meniscal reconstitution with allografts293-305 is a therapeutic option especially for young and symptomatic patients with a history of lateral meniscectomy in a normally aligned, stable joint without severe degenerative changes of the articular cartilage. A recent review 306 suggests that meniscal allograft transplantation improves pain and function in the short and intermediate term.
Application of gene-based strategies has been suggested to improve remodeling of meniscal allografts. 307 Martinek et al. 308 studied the feasibility of gene transfer in lapine meniscal allografts ex vivo using a retroviral vector encoding the marker gene lacZ. Subsequently, unilateral meniscal replacements were performed with these engineered allografts. Transduced fibrochondrocytes migrated into the depth of the graft, while transgene expression persisted for up to 8 weeks. This investigation suggests potential promise for growth factor delivery in autografts and allografts prior to implantation.
Clinical Gene Therapy Trials
Preclinical data, as those described above, have encouraged the initiation of human clinical trials originally for arthritis. The first studies were based on the ex vivo retroviral gene transfer of a human IL-1Ra sequence in synoviocytes from patients with end-stage RA followed by reinjection of the modified cells in the metacarpophalangeal joint.23,82,309 The aim of these studies was to evaluate the possibility of transferring genes to human joints and expressing them intra-articularly in a safe fashion acceptable to the patients. The use of these protocols has permitted extensive testing of the cells prior to reimplantation, demonstrating successful expression of the transgene locally vis-à-vis control joints, without adverse events related to the treatment but with clinical improvements in some of the patients, encouraging the implementation of phase II studies (pending).1,5,30,310-315 Another protocol has been initiated for intra-articular plasmid 316 delivery of the HSV thymidine kinase gene to the synovial lining of RA patients followed by administration of ganciclovir to achieve synovial ablation,1,5,309,311-314 but this protocol has been closed because of a failure to recruit. A new phase I trial for RA involved the direct in vivo intra-articular injection of an rAAV vector carrying the sequence for a fusion protein as sTNFR on an immunoglobulin molecule (tgAAC94 protocol). 317 As the study revealed that the treatment was safe and well tolerated in subjects without use of concurrent systemic TNF-α antagonist,1,311,312,314,317 a phase I/II trial was subsequently started 318 with the possibility to include patients who were already taking systemic TNF blockers and the administration of a second injection of tgAAC94. As one of the participants who was simultaneously being treated with systemic TNF antagonist and other immunosuppressive medications died after receiving the second injection, the trial was placed on hold by the U.S. Food and Drug Administration (FDA) to investigate, in parallel with the Recombinant DNA Advisory Committee Recombinant DNA Advisory Committee (RAC), the circumstances of the demise of the patient. The death was apparently due to a disseminated infection with Histoplasma capsulatum, a fungus endemic in the region of origin of the volunteer, and to an immunosuppression.312,319-321 Indeed, known serious complications of the particular TNF antagonist are susceptibility to H. capsulatum. The most probable explanation is that the subject was already infected with the fungus when receiving the second injection of tgAAC94. As the committee felt that the gene therapy protocol was very unlikely to have played any significant role in the event based on a large body of data from the independent investigations and since rAAV has been used safely in 47 previous human gene therapy clinical trials, the evaluation has been reopened with some modifications (exclusion of patients with elevated temperature, localized symptoms, fatigue, or with history of opportunistic infection), requiring additional monitoring (repeated blood counts, serum chemistry, vector DNA and transgene product titration, analysis of T-cell responses to AAV), as a possible role of the gene transfer in this course has not been definitely excluded (presence of neutralizing antibodies to the AAV capsid, occasional detection of vector genomes in the blood at the highest vector dose). Regarding OA, a phase I protocol is currently ongoing, based on an ex vivo approach using the retroviral transfer of TGF-β. 312
Gene Doping
Although the previously discussed gene-based approaches may have potential value for the treatment of articular cartilage defects and meniscal lesions, some of the therapeutic genes used in these studies have been also implicated for gene doping, 322 a term referring to the potential misuse of gene therapy for the purposes of enhancing athletic performance.323-325 Possible genes with such potential include, but are not limited to, growth hormone and IGF-I, 326 erythropoietin (Epo), 327 VEGF, 328 FGF-2, and endorphins. 329
IGF-I, the prime target of growth hormone action, is a potential candidate gene. A number of studies have shown that upregulation of IGF-I stimulates muscle growth and improves muscle function. 326 Interestingly, this increase in muscle volume is not reflected by detectable increases in circulating IGF-I. While favorable responses have been obtained in animal studies, the transfer of such techniques to humans with the goal of a higher performance still presents many technical challenges.
The hormone Epo is produced by the peritubular capillary endothelial cells in the kidney. Under hypoxic conditions, Epo is produced and secreted, increasing the production of red blood cells. Eero Mäntyranta, a Finnish cross-country skier who won 2 gold medals in the 1964 Olympics, was born with a mutation in the Epo receptor gene that allowed his blood to carry significantly more oxygen than an average person. 330 Recombinant Epo has been used already as a performance-enhancing drug. Because of differences in its peptide sequence compared with the endogenous protein, it may be detected in blood. Recently, a viral vector for the release of Epo in response to low oxygen concentrations has been developed under the trade name Repoxygen (Oxford BioMedica, Oxford, UK). The viral vector of undisclosed origin carries the human Epo gene under the control of a hypoxia control element (HRE). At low oxygen concentrations, HRE switches on the expression of the transgene. The vector is designed to be delivered by a simple intramuscular injection, resulting in the synthesis of recombinant Epo by muscle cells, rather than by cells of the liver or kidneys. Initially developed to treat anemia, there have been speculations in the media that it has been already applied for doping purposes. 331
Recently, genetically engineered mice have been created with an alteration in energy metabolism based on overexpression of the gene for phosphoenolpyruvate carboxykinases (PEPCK-C). PEPCK-C is an enzyme of the lyase family that plays a role in the metabolic pathway of gluconeogenesis, converting oxaloacetate into phosphoenolpyruvate and carbon dioxide. These transgenic PEPCK-C mice carry a chimeric gene in which a copy of the cDNA for PEPCK-C is placed under control of the skeletal actin gene promoter, directing overexpression of PEPCK-C exclusively to skeletal muscle. PEPCK-C mice were more active, could run longer and faster, and used fatty acids more efficiently and produced far less lactate than control animals. 332 Whether these data can be corroborated by studies in large animals remains to be determined.
Taken together, there is an emerging body of results from a number of transgenic and somatic gene transfer studies that suggest the principle of gene transfer may find application to enhance athletic performance. Many of the genes are already cloned in functional vectors, and some of them are being evaluated in clinical trials for the treatment of diseases. However, therapeutic gene transfer to humans is still technically challenging, and no clear evidence has been given that athletes have been using gene technology to enhance their performance. For antidoping authorities, the challenge will be to detect these endogenously produced gene products because of the homology between the transferred cDNA, the homology of the endogenously produced protein, and the limited specificity of indirect detection procedures. 333 Further studies in this fíeld are needed since a possible uncontrolled use of these gene vectors imposes potential high risks for both the athlete and the general public.
Outlook
Despite these encouraging data, application of gene transfer approaches in the treatment of articular cartilage and meniscal lesion tears is still in its infancy. Although the use of gene therapy holds great promise, issues that need to be addressed include the duration of transgene expression, further studies in clinically relevant animal models of articular cartilage and meniscal lesions, the benefit of using ex vivo genetically modified cells versus direct gene transfer approaches, and the identification of (an) optimal therapeutic factor(s) for each particular clinical problem. Future studies will also have to shed light on the safety of these approaches regarding the nonlethal nature of these diseases. A successful application of gene therapy for cartilage repair requires the combined effort of orthopedic surgeons continuing to ask clinically relevant questions and of basic scientists further improving the currently available gene transfer systems.
Footnotes
Acknowledgments and Funding
The authors received no financial support for the research and/or authorship of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
