Abstract
Objective:
Our previous work showed that epithelial membrane protein 1 (EMP1) is highly expressed in nucleus pulposus of the human degenerative intervertebral disc. The present study was designed to investigate the role of EMP1 in nucleus pulposus cells in intervertebral disc degeneration (IDD).
Design:
Human nucleus pulposus cells derived from degenerative intervertebral discs were cultured. EMP1 expression was knocked down by lentivirus-mediated specific interfering RNA. Cell morphology was observed, and cell proliferation, apoptosis, and cycle were evaluated.
Results:
Knockdown of EMP1 inhibited cell proliferation, caused cells to shrink, and accelerated the apoptosis induced by serum deprivation or addition of cycloheximide but did not evoke apoptosis in normal culture conditions.
Conclusions:
These findings suggest that EMP1 promoted chondrocyte proliferation, survival, and morphological change of cells during IDD, implying that EMP1 may be a target for biological therapy for IDD.
Keywords
Introduction
It has been demonstrated that intervertebral disc degeneration (IDD) is a causative factor of neck or low back pain and disc herniation, the incidence of which is on the rise. 1 Ample evidence has shown that extensive histomorphological changes occur in IDD, including fibrosis of the nucleus pulposus, lamellae disorganization of the annulus fibrosus, and thinning and calcification of the cartilaginous end plates. These tissue remodelings are mediated by disc cells. 2 With degeneration, disc cells in the nucleus pulposus undergo alteration in cell phenotype. Cell density increases, but the number of viable cells decreases because cell death, proliferation, and senescence are increased.3,4 However, the molecular mechanism of the nucleus pulposus cell (NPC) biological changes during IDD remains unclear.
Epithelial membrane protein 1 (EMP1) is a member of the peripheral myelin protein (PMP)−22 family and has been documented to regulate cell proliferation, 5 apoptosis, 6 membrane trafficking, and cell adhesion 7 depending upon the cell type. The results of gene scanning analysis in our previous study showed that the EMP1 gene was upregulated significantly in nucleus pulposus of human degenerative intervertebral discs. 8 We therefore hypothesized that EMP1 might participate in the pathogenesis of IDD. In the present study, we cultured nucleus pulposus cells from three patients who underwent discectomy for IDD and knocked down EMP1 expression by lentivirus-mediated RNA interference to examine the proliferative capability, morphological change, apoptosis, and cell cycle of the degenerated nucleus pulposus cells in an attempt to clarify the biological function of the EMP1 gene in the process of IDD.
Materials and Methods
Lentivirus Vectors for Human EMP1 Small Hairpin RNA
Small hairpin RNA (shRNA) of the human EMP1 lentivirus gene transfer vector encoding enhanced green fluorescent protein (EGFP) was constructed by Genechem Co. Ltd. (Shanghai, China). The targeting sequence of shRNA was GGACTTAGAAGTAGTATGT and was confirmed by sequencing. The recombinant lentivirus of small interference RNA targeting EMP1 (EMP1-RNAi-lentivirus) and control lentivirus (EGFP-lentivirus, containing scrambled nucleotide and EGFP gene), were prepared and titered to 108 TU/mL (transfection unit).
Culture of Human Nucleus Pulposus Cells Derived from Degenerative Intervertebral Disc
This study was approved by the Ethical Board of the Second Military Medical University (Shanghai, China). The nucleus pulposus at L5 to S1 of the degenerative intervertebral disc from 3 patients (64 ± 5 years) who underwent discectomy for IDD were collected. The degeneration level was grade IV according to the Pfirrmann grading system. 9 Tissue was sheared into 1-mm3 chips, digested in 0.2% collagenase (Roche, Meylan, France), and cultured in DMEM/F12 medium (HyClone, Logan, UT) supplemented with 2 mM L-glutamine and 10% fetal bovine serum (FBS) (HyClone) at a density of 2 × 105/T25. Passage 2 cells were used for viral transfection. Confluent nucleus pulposus cells from the second to the fourth passage were used for all experiments.
Lentivirus siRNA Gene Transfection
Cell transfection was achieved by incubating cells in a culture flask at a concentration of 3 × 105/T25. After 48 hours of culture, virus stock solution was added at a Multiplicity of Infection (MOI) (viral genomes for every cell) of 15 in the presence of polybrene at a final concentration of 8 µg/mL. The medium was changed 10 hours later.
Semiquantitative Reverse Transcription Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted from cells that had been transfected and cultured for 1 week using a TRIzol kit (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions. The reverse transcription reaction was performed using 1.5 µg total RNA and oligo (dT) primer. EMP1 (sense primer: 5′-ATCTTTGTGGTCCACATCGCT-3′, antisense primer: 5′-CTTCTCCATGGTGAAGAGCT-3′; designed to produce a 252-bp product) and GAPDH (sense primer: 5′-TTCAGCTCAGGGATGACCTT-3′, antisense primer: 5′-GGCATGGACTGTGGTCATGAG-3′; designed to produce a 220-bp product) genes were amplified. The PCR reaction was performed under the following conditions: 1 cycle at 94 °C denaturation for 5 minutes, 28 cycles at 94 °C denaturation for 20 seconds, 54 °C annealing for 30 seconds, and 72 °C extension for 40 seconds, then 1 cycle at 72 °C with a final extension for 10 minutes. The PCR products were separated electrophoretically in a 1.2% agarose DNA gel stained with ethidium bromide. Semiquantitative analysis was performed using a computing densitometer and AlphaEase software (Alpha Innotech, Santa Clara, CA).
Western Blot Analysis
One week after lentivirus-mediated siRNA transfection, total proteins were extracted using RIPA lysis buffer. There were 20 µg of proteins separated on 12% SDS-PAGE gels. Separated proteins were transferred to 0.2 µm polyvinylidene difluoride membranes. The membranes were blocked in 5% skim milk for 3 hours before incubation with mouse antihuman EMP1 antibody (Abnova, Taipei City, Taiwan) and mouse antihuman GAPDH antibody (Biosynthesis Biotechnology Co. Ltd., Beijing, China) overnight. The secondary antibodies labeled with HRP were incubated for 1 hour at room temperature. Membranes were washed twice in TBST and once in TBS and finally soaked in ECL reagent. Protein bands were visualized by using a Western blotting system (Amersham, Amersham, UK). Data obtained from the Western blot experiments were analyzed by Bio-Rad Quantity One 1-D Analysis software (Bio-Rad Laboratories, Hercules, CA).
Cell Proliferation and Viability Assay
Seven days after transfection, proliferation and viability of cultured human degenerative disc cells treated with EGFP-lentivirus or EMP1-RNAi-lentivirus were measured by Cell Counting Kit-8(CCK-8) assay at a 24-hour interval for 6 consecutive days. Briefly, the human degenerative disc cells were seeded into a 96-well plate on day 0 at an initial density of 3 × 103/well. Before detection, 10 µL CCK-8 was added to each well containing 200 µL of culture medium and incubated for 3 hours at 37 °C. Viable cells were counted by absorbance measurements at 450 nm using an automicroplate reader.
Analysis of Apoptosis and Cell Cycle
Experiments were performed 1 week after transfection. The same amount of cells (5 × 105) were seeded and cultured for 24 hours. Medium was changed to DMEM/F12 with no serum for 24 hours, or cycloheximide was added at a final concentration of 40 µM for 16 hours. Then, cells (including suspension cells in the medium) were collected, washed with PBS, fixed with 75% ethanol, stained with PI, and detected for the apoptotic peak and cell cycles by flow cytometry. Cells were also collected, treated with 1 µL Red-DEVD-FMK (BioVision, Mountain View, CA) that could bind active caspase 3, cultured for an additional 30 minutes, washed with wash buffer (BioVision) 3 times, and finally detected by flow cytometry.
Statistical Analysis
All data were presented as mean ± standard deviation (SD). Differences between groups were analyzed using the rank-sum test. P < 0.05 was considered to be statistically significant.
Results
EMP1 Is Expressed in Cultured Degenerative Nucleus Pulposus Cells and Can Be Knocked Down Using Specific Interfering RNA
After transfection with small interfering RNA targeting EMP1 mediated by lentivirus, more than 70% cultured NPCs derived from the degenerative intervertebral disc exhibited green fluorescence, and relative EMP1 expression (compared to GAPDH) in mRNA and protein declined from 0.87 ± 0.09 to 0.19 ± 0.01 ( Fig. 1A ) and from 0.45 ± 0.01 to 0.3 ± 0.01 ( Fig. 1B ), respectively.
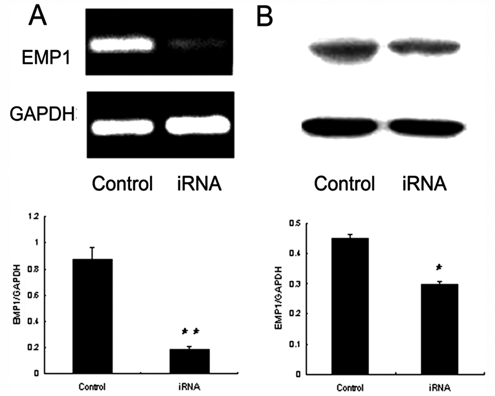
EMP1 was knocked down by special RNA interference. EMP1 expression was detected with semiquantitative RT-PCR (n = 3) (
RNA Interference of EMP1 Inhibits Proliferation of Degenerative Nucleus Pulposus Cells Ex Vivo
The NPCs transfected with EGFP-lentivirus began increasing markedly from day 3 on, and the absorbance of NPCs at day 6 was 2-fold as high as that at day 1. Contrarily, no significant increase was detected in the absorbance of cells treated with EMP1-RNAi-lentivirus throughout the experimental course ( Fig. 2A ). Additionally, trypan blue exclusion confirmed that there was no significant difference in the number of dead cells between the 2 groups (data not shown).

Proliferation and viability of cells slowed down after downregulation of EMP1. (
In addition, change in cell cycle was examined by using flow cytometry. It was found that the percentage of G0/G1 phase population was 58.1% ± 4.51% in the group treated with EGFP-lentivirus and 69.8% ± 5.37% in the group treated with EMP1-RNAi-lentivirus when the cells were cultured in media containing 10% FBS ( Fig. 2B ).
RNA Interference of EMP1 Causes Shrinkage of Degenerative Nucleus Pulposus Cells Ex Vivo
Degenerative nucleus pulposus cells cultured in medium supplemented with 10% FBS looked stellated with multiple cytoplasmic processes extending into the surrounding matrix. Two days after transfection with lentivirus mediating RNA interference targeting EMP1, degenerative NPCs began shrinking, and many of them became elongated or rounded ( Fig. 3 ). In addition, no morphological change was observed in the degenerative NPCs transfected with EGFP-lentivirus when the transfection unit was increased (MOI = 20) purposely, while morphological shrinkage was still observed in NPCs transfected with EMP-RNAi-lentivirus when the transfection unit was decreased (MOI = 5).
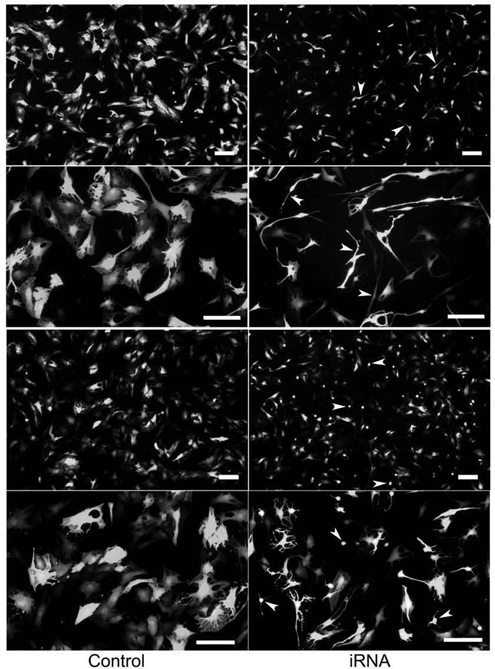
Morphological changes of the nucleus pulposus cells after knockdown of EMP1. A representative photograph of the cultured nucleus pulposus cells was taken 3 days after transfection under fluorescence microscope. Arrows indicate shrinking cells. Bar, 20 µm.
RNA Interference of EMP1 Does Not Influence Apoptosis of Cultured Nucleus Pulposus Cells Unless Serum Is Removed or Cycloheximide Is Added
In the presence of 10% FBS in the media, few apoptotic cells (no more than 1.01%) were detected either in NPCs treated with EGFP-lentivirus or with EMP1-RNAi-lentivirus. However, apoptosis was evoked in NPCs cultured in serum-free medium or in medium containing additional protein synthesis inhibitor cycloheximide. EMP1 knockdown increased the percentage of sub-G1 population from 11.44% ± 1.13% to 15.02% ± 1.20% and the percentage of active caspase 3–positive cell population from 13.77% to 22.91% after 24-hour serum deprivation. Similarly, EMP1 knockdown increased the percentage of sub-G1 population from 11.44% ± 1.13% to 15.02% ± 1.20% and the percentage of active caspase 3–positive cells from 32.32% to 60.21% after addition of a final concentration of 40 µM cycloheximide to the medium for 16 hours ( Fig. 4 ).
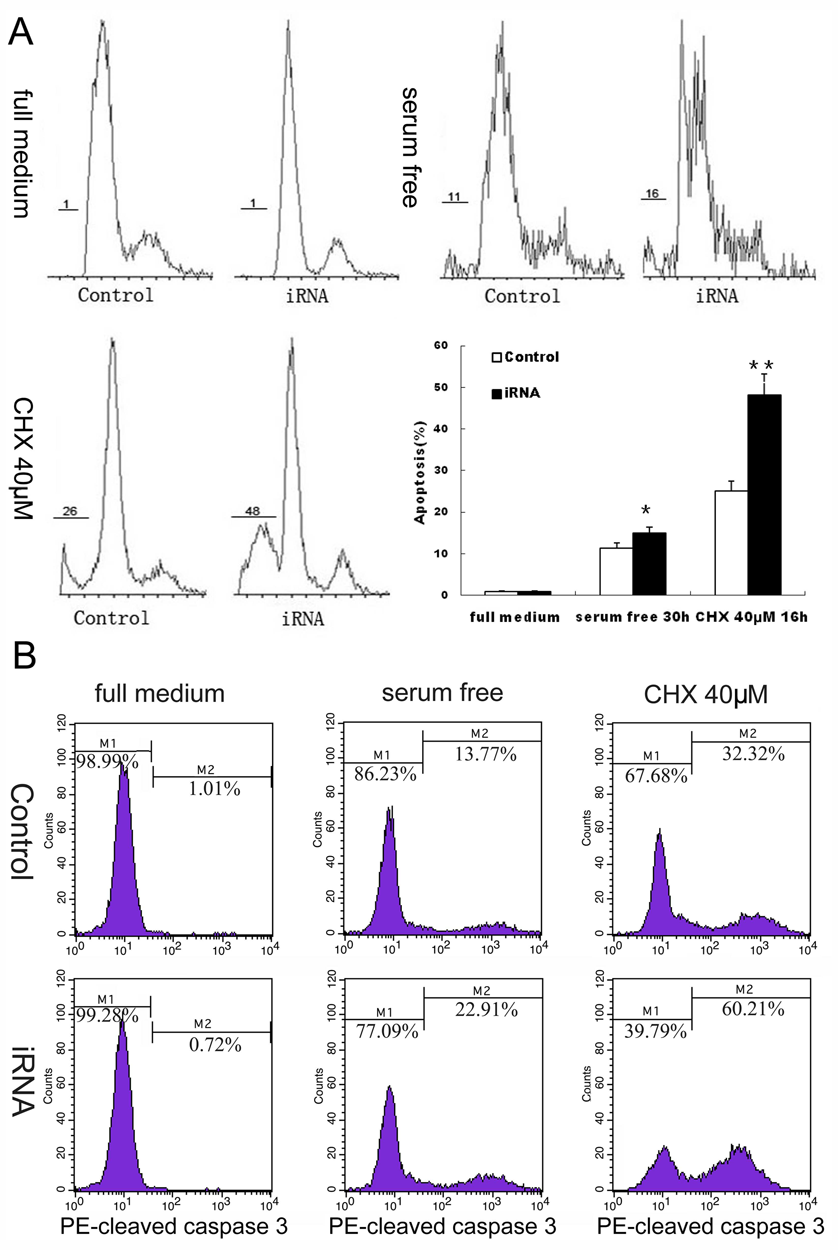
Cell apoptosis was increased under the induction conditions after knockdown of EMP1. Sub-G1 population (
Discussion
EMP1 was first identified in squamous cells in 1995 10 and named as the tumor-associated membrane protein (TMP) in 1996. 11 Since then, EMP1 transcript has been detected successively in many tissues.12-14 In 2004, we discovered that EMP1 expressed more highly in degenerative intervertebral discs than it did in normal ones (ratio = 3.75). We therefore hypothesized that EMP1 might participate in the pathogenesis of IDD. But what role did EMP1 exactly play in the process of IDD? What cell process did it participate in: cell apoptosis, proliferation, senescence, change of morphology, or phenotype? These questions remain unanswered, for EMP1 exhibited different functions in different cell types. The results of the present study seem to suggest that EMP1 participated in cell proliferation and cell morphological change but not in apoptosis during IDD.
EMP1 May Be Associated with the Formation of Cell Clusters and Cell Proliferation in IDD
Healthy human intervertebral discs contain relatively few sparsely distributed cells. A characteristic feature of IDD is the appearance of cell clusters, particularly in damaged areas. They are formed by cell proliferation, as demonstrated by cell proliferation–associated markers Ki67 and PCNA. 15 Cell proliferation is also observed in cervical discs in mouse models of cervical spondylosis. 16 These observations suggest that disc cell proliferation is associated with disc degeneration. But the mechanism of cell proliferation during IDD remains unknown. It is found in this study that knocking down EMP1 reduced S/G2/M population, with an increase of G0/G1 population, implying that cell proliferation was reduced at a certain time point, which is consistent with the result of the CCK-8 assay. The cell growth curve also showed that knockdown of EMP1 slowed down cell growth, suggesting that silencing EMP1 inhibited proliferation of degenerative NPCs, as was observed in fetal NPCs, indicating that EMP1 participated in regulating proliferation of nucleus pulposus cells in degenerative intervertebral discs and could be associated with the formation of cell clusters.
EMP1 Is Associated with Cell Morphological Change in IDD
Normal nucleus pulposus cells in the healthy nucleus pulposus are more rounded or oval, but diseased nucleus pulposus cells looked stellated or dendritic, having multiple branching cytoplasmic processes extending into their surrounding matrix.17,18 The degenerative disc cells cultured in the present study also had a stellated appearance with cytoplasmic processes extending into the surrounding matrix, similar to degenerative nucleus pulposus cells in vivo. But knocking down EMP1 caused cells to shrink, reduced cytoplasmic processes, and even helped cells restore the round appearance, without inducing apoptosis. We also found that fetal nucleus pulposus cells formed multiple, tiny cytoplasmic processes, which appeared to flatten after overexpression of EMP1 in our previous work. These results implied that EMP1 participated in the formation of cytoplasmic processes during IDD. Downregulated PERP gene, another member of the peripheral myelin protein (PMP)–22 family, also caused notochordal cells of zebrafish to exhibit a round appearance, 19 which is similar to our finding.
EMP1 Is Not Associated with Apoptosis in Degenerative Intervertebral Discs; Rather, It Improved Cell Survival
To determine whether EMP1 participated in apoptosis during IDD, apoptosis of EMP1 knockdown NPCs cultured in full medium (containing 10% fetal calf serum), serum-free medium, and full medium with CHX was examined by detection of sub-G1 population and caspase 3 activity. Few apoptotic cells were observed either in control cells or in EMP1-RNAi-lentivirus–treated cells cultured in full medium. After serum deprivation for 24 hours or addition of 40 µM CHX for 16 hours, apoptosis was observed both in experimental and control groups, but the apoptosis rate in the experimental group was significantly higher than that in the control group, suggesting that silencing EMP1 accelerated the apoptosis markedly. These findings indicate that knocking down EMP1 did not evoke cell apoptosis in full medium but promoted the apoptosis induced by serum deprivation or addition of CHX and that upregulation of EMP1 expression in degenerative intervertebral discs could improve survival of nucleus pulposus cells.
In conclusion, this study shows that EMP1 silence can inhibit the proliferation and change the morphology of cultured human degenerative disc cells but not increase apoptosis. These results suggest that EMP1 may regulate the function of nucleus pulposus cells and participate in the degeneration of intervertebral discs.
Footnotes
Acknowledgments and Funding
This study was supported by the National Science Fund for Distinguished Young Scholars of China (no. 30400453).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
