Abstract
Background
Intervertebral degenerative disc (IDD) disease is one of the most common clinical conditions causing low back pain. The main objective of this study was to investigate the repair effect of platelet-rich plasma (PRP) and ferulic acid (FA) hydrogel compound on degenerative discs in rats in combination with bioengineering technology, which may provide a strong theoretical basis for the future treatment of IDD.
Methods
Forty-five male Sprague-Dawley rats were randomly divided into groups A-F; MRI was performed in each group at 0, 4, and 8 weeks after injection; and disc tissues were obtained after executing the animals. The histomorphology, apoptosis, and protein synthesis of intervertebral discs in each group were observed by hematoxylin-eosin, Masson, terminal deoxynucleotidyl transferase dUTP nick end labeling staining, and Western blot.
Results
The release concentration of all groups reached the peak at 12 hours, and the highest concentration was found in the hydrogel/PRP/FA group at the same time. The MTT assay showed that hydrogel/PRP/FA is well-cytocompatible. The results of animal experiments show that hydrogel/PRP/FA has a good effect on degenerative intervertebral disc in rats.
Conclusion
PRP/FA-rich hydrogel compound plays an active role in promoting extracellular matrix synthesis, strengthening and repairing degenerated intervertebral discs in rats.
Keywords
Introduction
Intervertebral degenerative disc (IDD) disease has been a major cause of discogenic low back and leg pain. Epidemiological investigation has shown that 50% to 80% of adults would experience low back and leg pain due to IDD, which has caused a severe burden to patients and society. 1 Currently, there is no recognized effective treatment for IDD. Although conservative treatment in the early stages of the disease can relieve symptoms, it cannot fundamentally stop the continuous development of disc degeneration. Surgery is required for more advanced disease, but it is complex and risky, changing the mechanics of the spine and causing a series of postoperative complications. 2 However, neither therapy addresses intervertebral disc problems such as inflammatory microenvironment, apoptosis, and impaired matrix synthesis well. 3 Recently, various biological therapies were proposed regarding the molecular mechanism of IDD, such as cell transplantation, growth factor injection, and gene therapy.4,5 Among the above-mentioned biological treatment measures, growth factor treatment was carried out earlier and is more widely used due to ease of operation and promising results. It has become an effective treatment strategy for repairing IDD. 6 It is well known that a variety of growth factors promote cell proliferation in intervertebral disc, inhibit cell apoptosis, stimulate extracellular matrix synthesis, and repair IDD. But studies have found that the repair effect of injecting a single growth factor into the intervertebral disc is very limited.6,7 Some researchers have tried to combine various growth factors to observe the efficacy and find that the effect is superior to the single factor treatment, indicating that synergistic effects may exist between different growth factors. Among them, platelet-rich plasma (PRP) and ferulic acid (FA) have anti-inflammatory, antioxidant, cell growth–promoting, and extracellular matrix synthesis effects, which have more promising clinical applications.8,9
Due to its high content of growth factors, PRP promotes the migration and proliferation of different types of cells. Thus, it not only provides a good microenvironment for the regeneration and repair of various soft tissues, but also prevents the spread of diseases and immune rejection.10,11 Many studies have indicated that PRP stimulates the proliferation of nucleus pulposus (NP) cells, promotes metabolism, and increases the content of extracellular matrix. 12 Therefore, PRP injection into the intervertebral disc may achieve satisfactory efficacy in the treatment of intervertebral disc degeneration.
FA is a natural plant phenolic acid. At present, FA has been adopted and widely used in the clinical therapy of cardiovascular diseases and cancer. It has the functions of scavenging free radicals, antioxidation, inhibiting platelet aggregation, anticoagulant, anti-inflammation, spasmolysis, and analgesia.13,14 It also shows significant therapeutic effects in bone and joint injury repair experiments. 15 It has been indicated that FA can inhibit the expression of important inflammatory factors, such as vascular endothelial growth factor, interleukin 6, inducible nitric oxide synthase, and tumor necrosis factor-alpha, consequently inhibiting angiogenesis and acting as a tissue cell protector. 16 Mir-340-5p was highly expressed in degenerative disc tissues. 17 An investigation on bone marrow mesenchymal stem cells shows that FA effectively inhibits the expression of mir-340-5p, implying the potential therapeutic ability of FA in suppressing the expression of degenerative disc genes and downregulating the rate of matrix breakdown. 18
The above study leads us to consider whether these 2 drugs together would be more effective in treating degenerative discs. Intradiscal drug injection therapy is not only an efficient and accurate intervention, but it is also outperformed by conventional therapy in terms of systemic side effects and treatment outcomes. However, there are still drawbacks in intradiscal injections, such as inability to maintain drug concentration, short duration of action, and tissue defects, which will affect the therapeutic effect of drugs. With the development of tissue engineering and new medical biomaterials, hydrogel has attracted more and more attention in medical applications due to its unique physiological characteristics such as water swelling, good drug delivery carriers, and tissue compatibility. 19 Hydrogel is a kind of tissue engineering material with porous microstructure and water swelling characteristics. It acts as a drug delivery vehicle to maintain the effective concentration and duration of the drug in the intervertebral disc. Meanwhile, it has superior histocompatibility and swelling properties, so it could be used as a tissue filler to repair tissue defects caused by intervertebral disc atrophy. To some extent, hydrogel can assist in the mechanical function of intervertebral disc tissue, ultimately leading to the treatment of IDD. 20
Considering these factors, the overall aim of this study was to investigate the physical properties, biosafety, and therapeutic efficacy of PRP/FA hydrogel compound through animal model experiments.
Materials and Methods
Animals
Forty-five male 6-week-old Sprague-Dawley (SD) rats (clean grade, 200 ± 10 g) were purchased from Changzhou Cavens Experimental Animal Co. Ltd. The laboratory animal license number is scxk (Su) 2016-0010. All experimental animals were acclimatized after 1 feeding to start the formal experiments. The animal was randomly divided into 6 groups: A group (sham group, n = 3 rats), B group (model group, n = 6), C group (model + hydrogel group, n = 9), D group (model + hydrogel + PRP, n = 9), E group (model + hydrogel + FA, n = 9), F group (model + hydrogel + PRP + FA, n = 9).
Animal Model
All animals were fasted with water 1 day before operation, weighed, and the amount of anesthetic calculated. General anesthesia was performed by intraperitoneal injection of 7% chloral hydrate at 0.5 ml/kg. After the pain reaction disappeared, the abdominal hair (about 6 cm × 4 cm) was shaved, the skin and subcutaneous tissues were incised sequentially, and the retroperitoneal cavity was opened to expose the L3-4 and L5-6 intervertebral discs and endplates. A 21G microneedle was then used to access these disc segments parallel to the cartilage endplates. The entire annulus fibrosus was inserted and the subcutaneous fascia and skin were sutured. After modeling, MR images were detected before and after the drug intervention at 4 and 8 W to analyze the changes in disc height and tissue hydration.
Cell Culture and Identification
NP tissue from rats was extracted under aseptic conditions and cultured in DMEM/F12 modified medium (containing 10% fetal bovine serum, 100 units/ml penicillin, and 100 μg/ml streptomycin). Cells were cultured at 37 °C in a humidified atmosphere of 5% CO2. The cultured cells were observed under an inverted microscope. NP cells were cultured to the second generation and observed after fluorescent staining. Collagen II showed green fluorescence, aggrecan protein showed red fluorescence, and the nucleus was in blue.
HE Staining
Intervertebral disc tissues were separated from rats. The prepared intervertebral disc tissue slices were dewaxed and hydrated. After washing with water, intervertebral disc tissue slices were stained in hematoxylin solution for 5 min. Next, it was differentiated with 1% hydrochloric alcohol for 15 seconds, and the slices were washed with water. Furthermore, intervertebral disc tissue slices were stained in eosin solution for 1 minute. Finally, intervertebral disc tissue slices were dehydrated, transparentized, sealed with neutral gum, and observed under an optical microscope (Olympus, Tokyo, Japan).
Western Blot
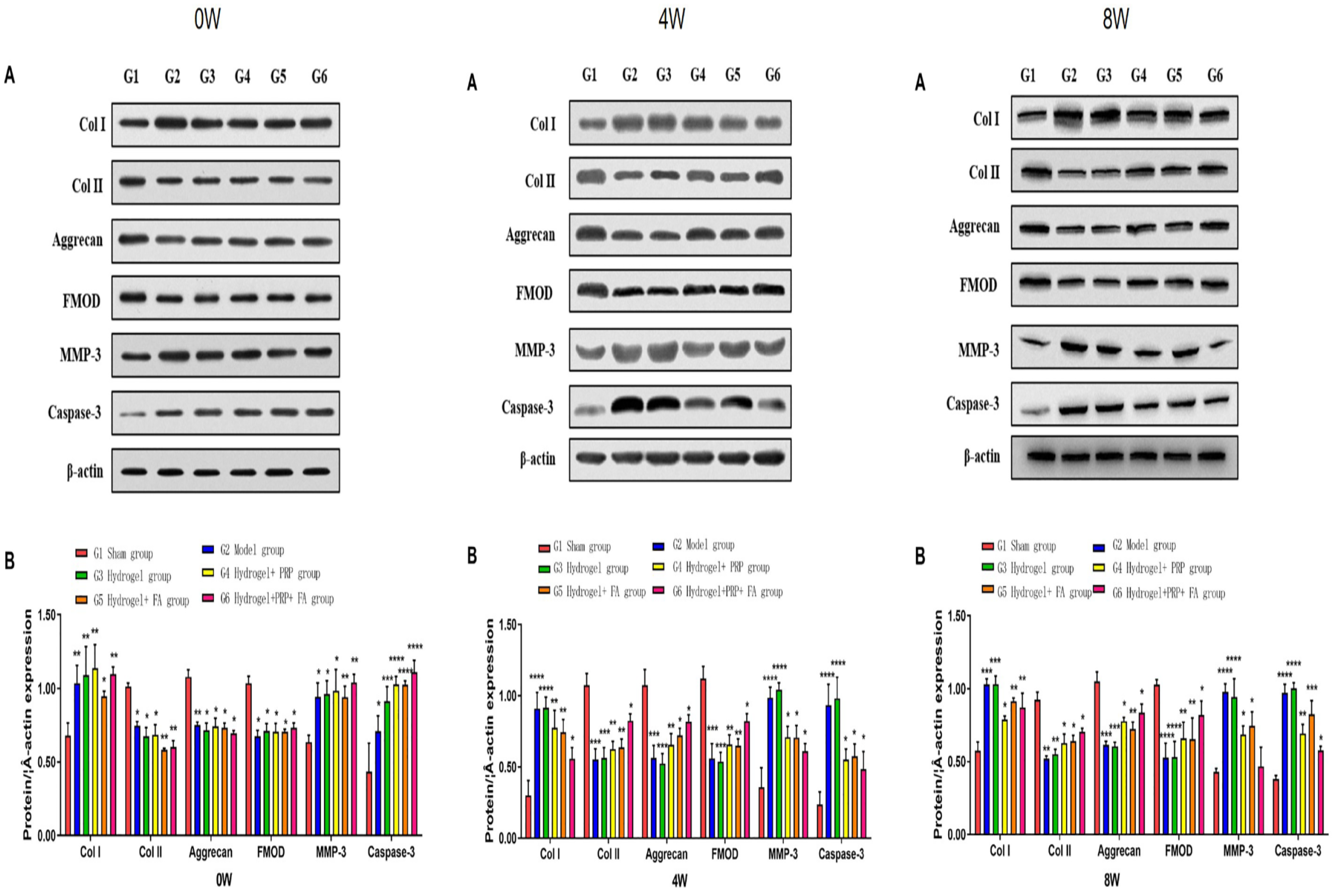
Samples were cracked in radioimmunoprecipitation assay lysis buffer plus phenylmethylsulfonyl fluoride at low temperature, and the Bicinchoninic Acid assay kit (Santa Cruz Biotechnology, Santa Cruz, CA) detected total protein concentration. The prepared protein samples were separated in sodium dodecyl sulphate-polyacrylamide gel electrophoresis and transferred into 0.22 μm polyvinylidene difluoride membranes and incubated with the prepared antibodies. Finally, enhanced chemiluminescence (ThermoFisher, Waltham, MA, USA) visualized this membrane. Antibodies against Col I, Caspase-3, Aggrecan, and β-actin were purchased from Abcam (Cambridge, MA). Antibodies against Col II, Aggrecan, FMOD, and MMP-3 were purchased from Proteintech (Wuhan Proteintech Group, Wuhan, China).
Live/Dead Fluorescence Microscopy Assay
Calcein AM cell staining reagent was used to stain hydrogel-treated cells. Calcein AM could easily penetrate the living cell membrane, and when entering the cytoplasm, esterase would hydrolyze it into Calcein and leave it in the cell, emitting strong green fluorescence. Calcein AM stains only viable cells. As a nuclear staining dye, PI cannot cross the cell membrane of living cells. It passes through the disordered region of the dead cell membrane to reach the nucleus and is embedded in the DNA double helix of the cell to produce red fluorescence (excitation: 488, 545 nm, emission: 617 nm).
MTT
Cells were cultured in 24-well plates for 24 hours and then divided into 2 groups with 3 wells in each group. Hydrogel extract was added to the experimental group, while new DMEM/F12 medium was added to the control group, and the medium was changed every 2 days for 24, 48, 72 and 96 hours, respectively. After cell culture at each time point, the culture plate was taken out and 100 μl of 5 mg/ml pre-prepared MTT solution was added to each well. Then, the culture was left in the cell incubator for 4 hours. The medium was removed, and 600 μl of dimethyl sulfoxide was added to each well, and the liquid was made fully homogeneous by shaking. Then, 200 μl of mixed liquid per well was transferred to a 96-well plate. The absorbance value of cells at 570 nm was measured with a microplate reader. The cell growth curve was drawn with time as abscissa and absorbance value as ordinate.
Preparation of PRP/FA Hydrogel Complex
Collecting the whole blood of rats
After anesthetizing the rats, the hair is cut from their chests. After disinfecting the skin, a syringe with a 4-5G needle is punctured into the area where the heart beats the strongest (third to fourth intercostals space on the left). The blood will automatically enter the syringe after puncturing the heart and the blood is collected into an anticoagulant.
Preparation of PRP
The anticoagulant vacuum blood collection vessel containing 5 ml of arterial blood was placed in a centrifuge and centrifuged at 200g for 10 minutes. Centrifuge the collected whole blood to 3 layers, namely, serum layer, white film layer, and red blood cell layer. The liquid above the erythrocyte layer is aspirated in a sterile environment into a sterile centrifuge tube. After centrifugation, the upper layer is the platelet-poor plasma and the lower layer is the platelet concentrate. Discard three-fourth of the supernatant, and what remains is the PRP.
Preparation of PRP/FA Hydrogel Compound
In all, 1.6% of hyaluronic acid (HA) and 5 BU/ml batroxobin (BTX) were mixed at a ratio of 6:1 to form the HA/BTX mixture. Next, this mixture was added to PRP in a 1:1 ratio to prepare the HA/BTX/PRP mixture. Then, 50 mM of FA was added to the HA/BTX/PRP mixture at a ratio of 1%, and the final concentration of FA was 500 μM to develop the PRP/FA hydrogel compound.
Determination of Properties of Complex
The properties of PRP/FA hydrogel complexes were determined in 3 aspects: temperature, morphology under electron microscope, and drug release in vitro. The compound was placed in a water bath at 20 °C and the temperature of the water was increased by 1 °C every 2 minutes. The flow state of the compound was observed by inverting the centrifuge tube. The highest temperature at which no flow of the compound occurred was considered to be the gel temperature. The structures of PRP/FA hydrogel complexes were observed under scanning electron microscopy. The concentration of the drug was determined by UV-Vis-NIR Spectrophotometer.
Statistical Methods
The experimental data were expressed as mean ± SEM, and all the data were processed and statistically analyzed by GraphPad Prism 6. T test was used to compare the mean values of the 2 groups of samples, and analysis of variance was used to compare the mean values of multiple groups of samples. P < 0.05 indicates that the difference is statistically significant.
Results
Preparation of PRP/FA Hydrogel Compound
To assess the biological function of hydrogel compound, HA/BTX hydrogels were prepared by mixing 1.6% HA with 5 BU/ml BTX at a ratio of 6:1. The HA/BTX blend was then mixed with PRP in a 1:1 ratio. At the same time, 97 mg of FA was dissolved in absolute ethanol to prepare 50 mM FA, and the 50 mM FA was added to the HA/BTX/PRP mixture at a proportion of 1%. Finally, the hydrogel/PRP/FA complex was obtained ( Fig. 1A ).
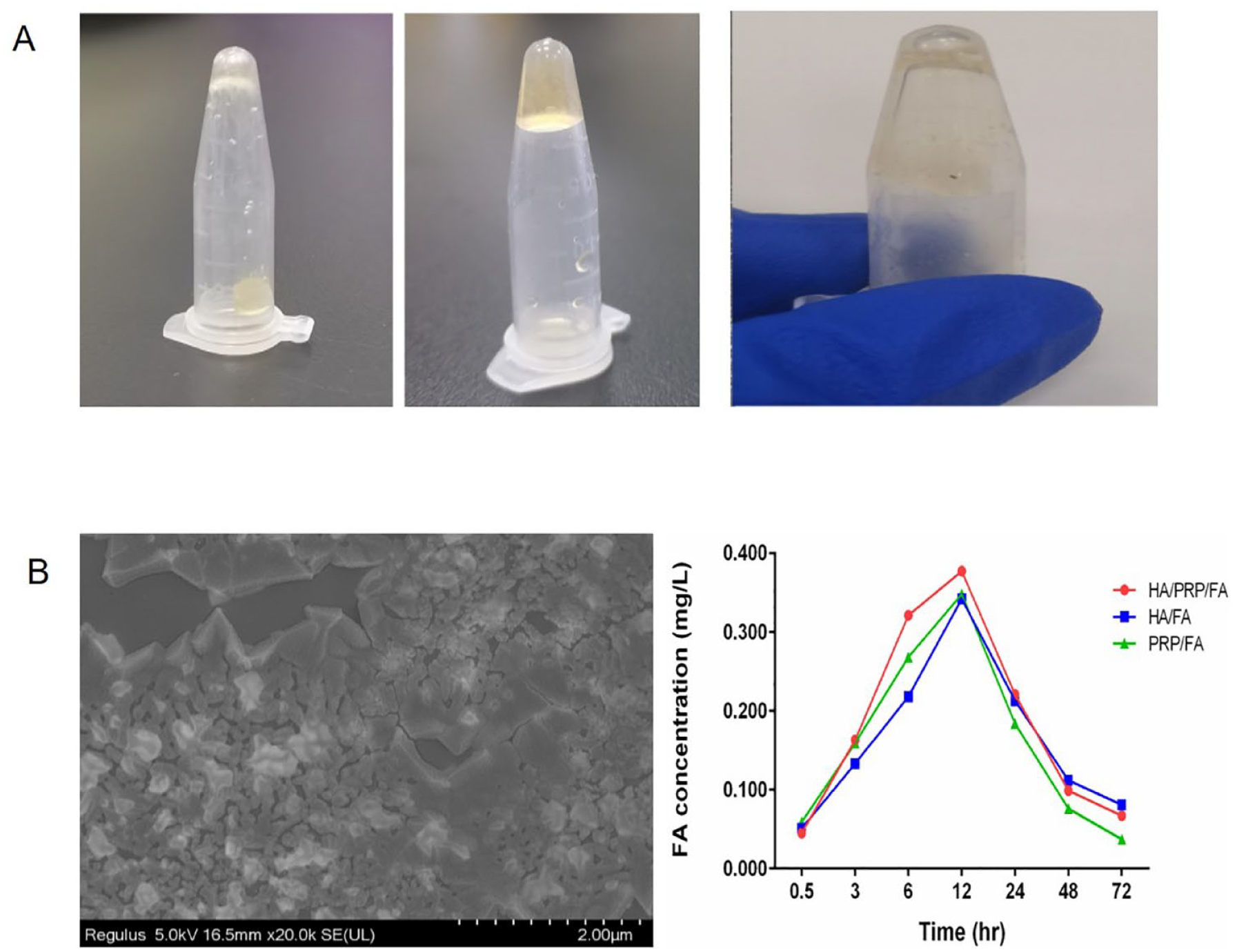
The basic properties of the hydrogel/PRP/FA complex. (
Hydrogel/PRP/FA Compound Is Biocompatible
The safety of hydrogel compound was studied with the MTT assay using NP cells. First, NP cells were incubated in vitro and identified prior to the assay ( Fig. 2A and B ). Then, DMEM-F12 medium was used to co-culture NP cells and hydrogel complexes for 24, 48, 72, and 96 hours, respectively. At the same time, Calcein-AM/PI kit was used for live and dead cell staining to detect the cell survival status. As shown in Figure 3A , compared with the control group, the fluorescence intensity of hydrogel compound group was stronger than that of the control group at each time point. CCK-8 showed similar results, and the proliferation of NP cells in the hydrogel group seemed to be enhanced ( Fig. 3B ). As we all know, NP cells and chondrocytes are similar in mentality and function, so we detected the specific phenotype of COL-2 and Aggrecan expression of chondrocytes by Western blot (WB) assay to reflect the proliferation of NP cells ( Fig. 3C ). As expected, the WB results showed the same trend as the results of the previous 2 experiments. Taking together, through these experiments, we proved that hydrogel has good biocompatibility, and this laid the foundation for our next animal experiment.
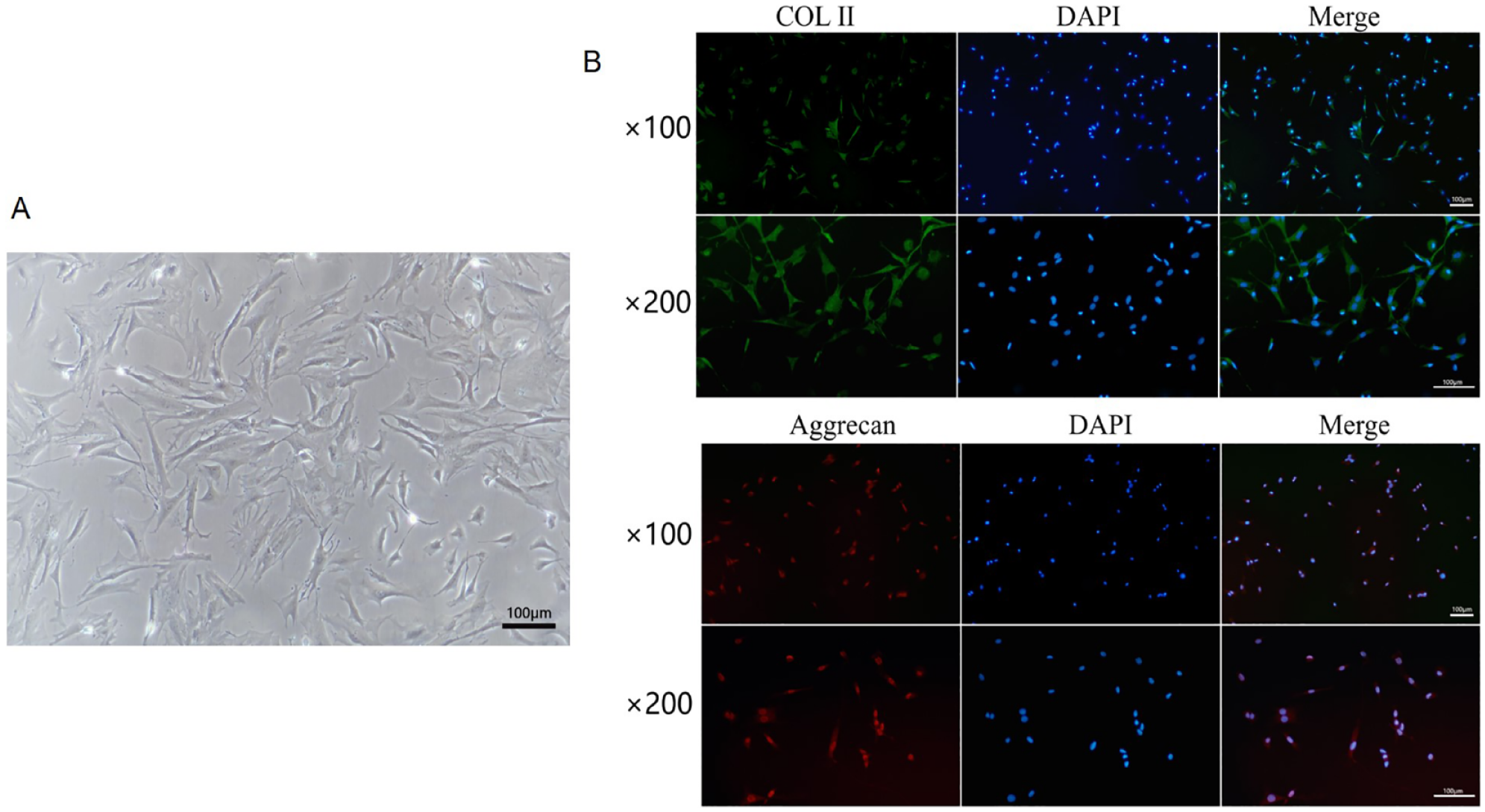
The basic morphology of NP cells used in the experiment. (
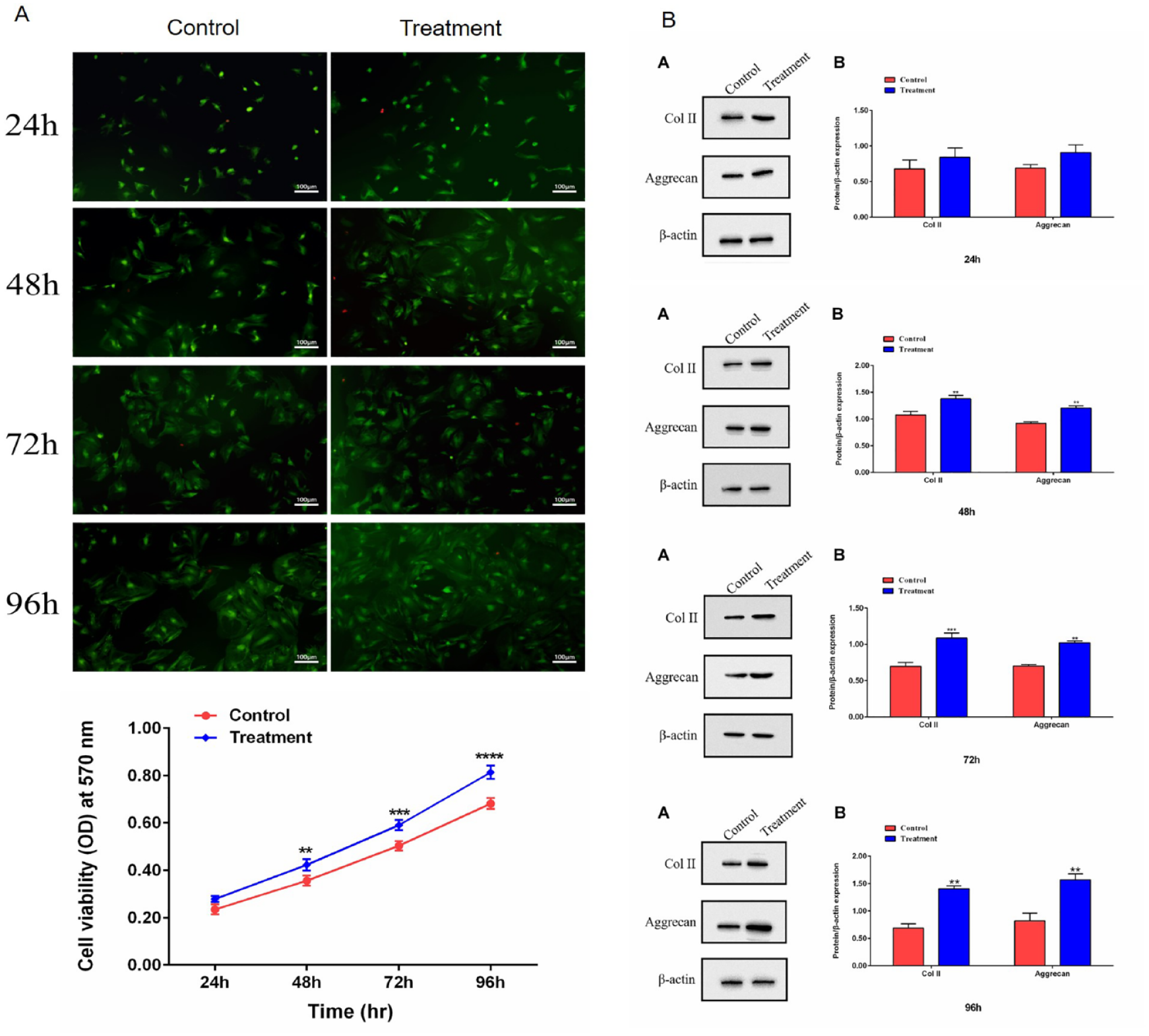
Hydrogel cytotoxicity assay. (
Hydrogel/PRP/FA Compound Can Effectively Treat Disc Degeneration in Rats
As the previous NP cell experiments demonstrated that hydrogel has good biocompatibility and can promote NP cell proliferation to a certain extent, we decided to continue to explore its role in animal model of disc degeneration. First, we observed the general situation of intervertebral disc degeneration in each group by MRI ( Fig. 4A ). Then, hematoxylin-eosin (HE) staining was performed on the samples taken at 3 time points ( Fig. 5A ), and we found that, with the prolongation of disease course, the discs of untreated rats showed a marked aggregation of blue particles, indicating calcification of the discs ( Fig. 5A ). It is well known that fibrous tissue is the major component of the disc, and the fibrous tissue can show the degeneration of intervertebral disc directly. Therefore, Masson staining was used to detect the changes in fibrous tissue of intervertebral dick in each group of rats ( Fig. 5B ). Compared with the control group, in the model group and the sham operation group, the morphology of fiber tissue disappeared, the structure was disordered, and there were obvious calcification spots. The hydrogel-treated rat tissue had less calcification than the model group, although the fibrous tissue structure was damaged but the tissue was orderly. Finally, the WB results of signature fibrin at 3 time points also confirmed our experimental results ( Fig. 6A ). Our experiments demonstrated that the hydrogel complex could effectively inhibit the tissue degeneration caused by disc injury in vivo.
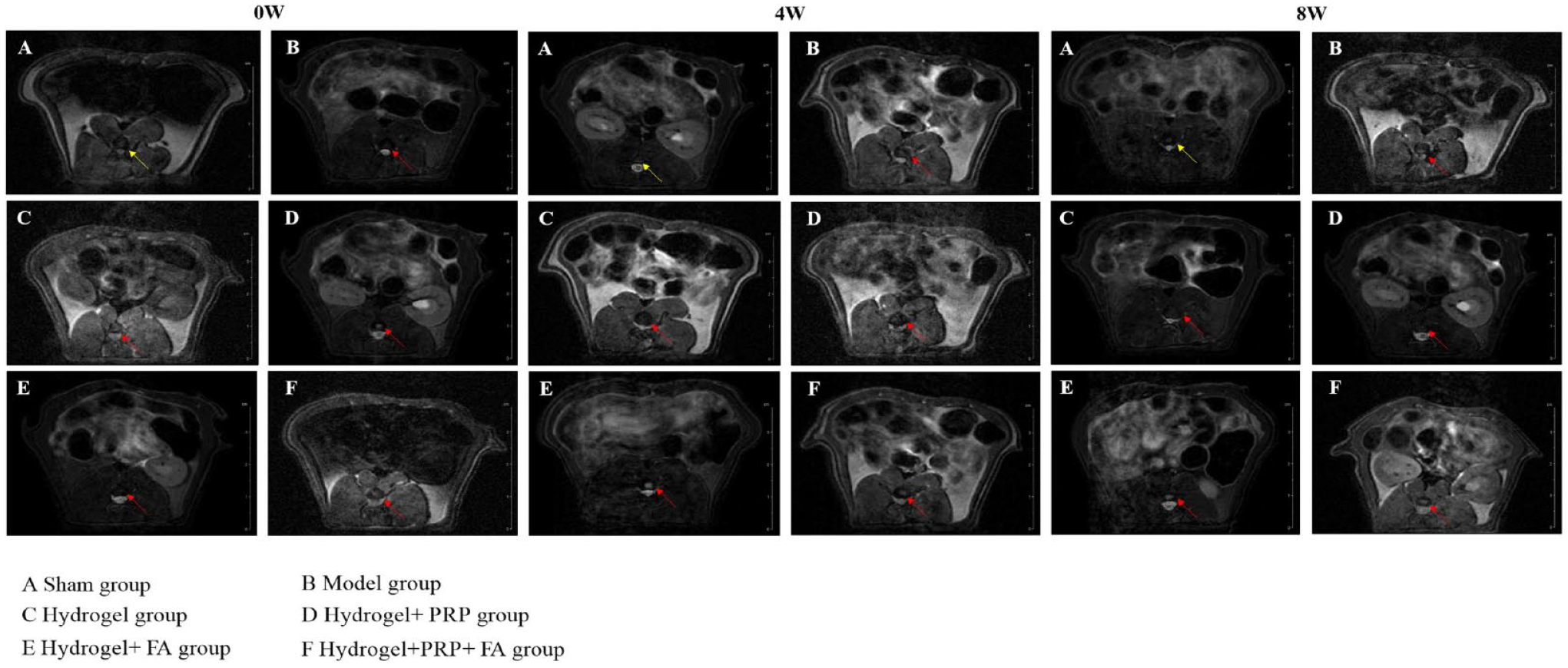
MR images of intervertebral disc in each group.
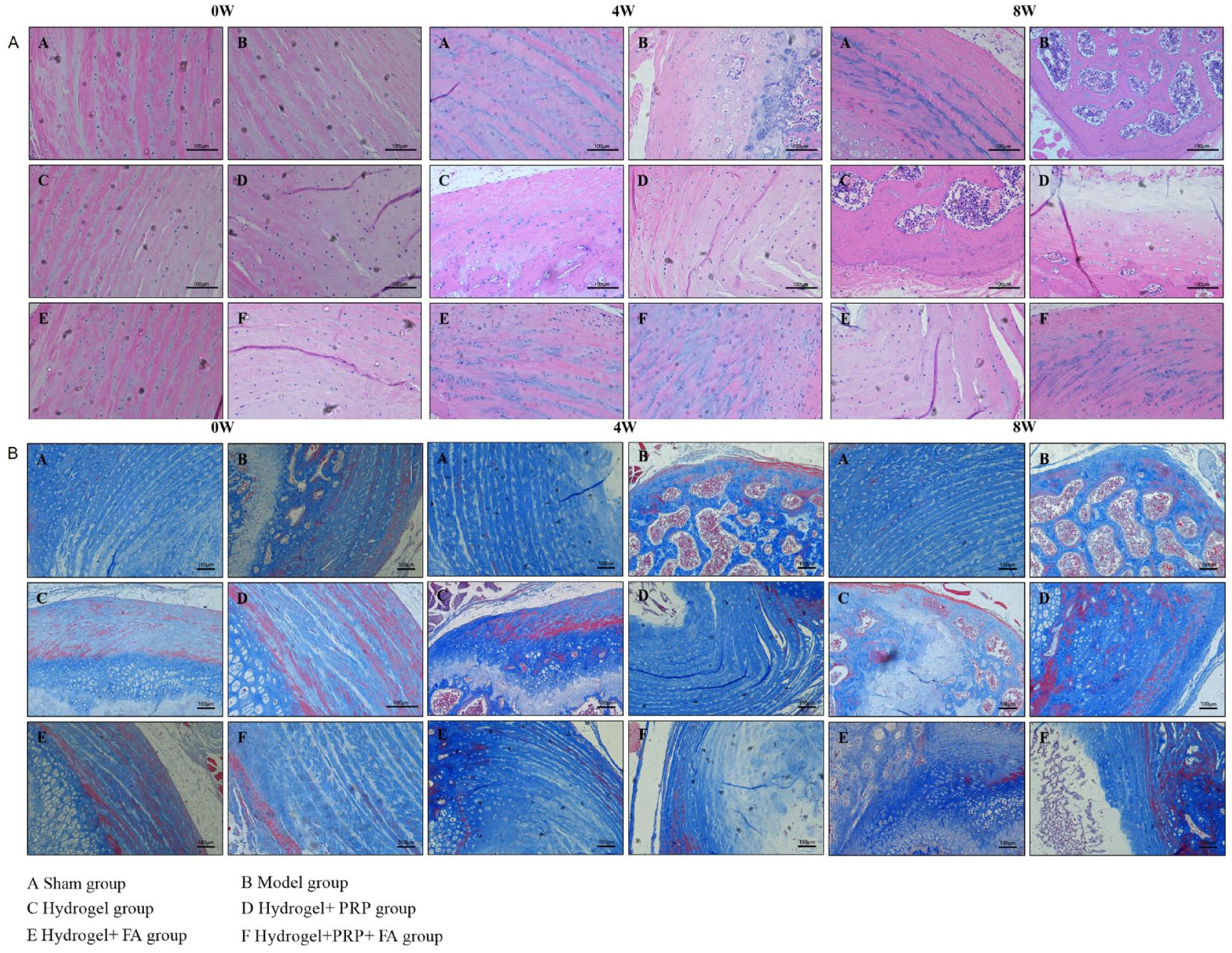
HE and Masson staining of intervertebral disc tissue in each group. (
Therapeutic effect of hydrogel on intervertebral disc fibrosis in each group. (
Discussion
Intradiscal injections are a popular treatment for IDD in recent years. This external intervention therapy through intradiscal injection enhances the ability of the disc to repair itself and thus slows down the degeneration of the disc. At present, hydrogel-based complexes of PRP and FA have shown great potential in the treatment of IDD. Many studies have shown that PRP promotes the proliferation of NP cells, facilitates the anabolic actions and migration of different cell types, and provides a favorable microenvironment for the regeneration and repair of various soft tissues.21,22 The study by Chen et al. 23 cultured human NP cells in PRP and found that PRP could enhance cell proliferation and differentiation through transforming growth factor beta-1, demonstrating for the first time that PRP is a therapeutic option for preventing disc degeneration. In a study of 47 patients with lumbar disc degeneration, Tuakli-wosornu et al. 12 found significant improvements in Functional Rating Index, Numerical Rating Scale of Pain, and North American Spine Society satisfaction scores in participants who received intradiscal PRP compared with those who received contrast alone. Growney et al. 24 first combined PRP with alginate and found that PRP increased the synthesis of extracellular matrix. A study by Nikkhoo et al. 25 revealed that PRP could increase the content of proteoglycans in the disc and restore the biomechanical properties of the intervertebral disc.
FA inhibits matrix breakdown, oxidative stress, and inflammatory responses. 26 As in the study of rheumatoid arthritis, 15 FA suppresses the progression of disease severity and bone erosion by inhibiting interleukin-17, which can be used as an alternative drug to limit interleukin-17 signaling. Also, FA can significantly reduce the expression of matrix metalloproteinases (MMPs) in chondrocytes. The expression of MMPs is highly positively correlated with the degree of IDD. 27 Doss et al. 13 found that FA is related to reducing osteoclast differentiation and bone erosion. Futhermore, FA significantly reduced osteoclast activity by inhibiting RANKL-induced activation of the NF-κB pathway. Based on the above studies, on one hand, PRP upregulates the level of matrix synthesis in the intervertebral disc, and on the other hand, the intervention of FA can inhibit the persistent effect of degenerative factors in the intervertebral disc, so our team carried these 2 drugs in a hydrogel and established a rat disc degeneration model to further confirm the effects of these 3 substances on degenerated discs. Based on these findings, our team designed and conducted this experiment: By injecting FA and PRP hydrogel into the intervertebral disc, it can fill the tissue defect and correct the stress distribution, improve the syngenesis metabolism level in the intervertebral disc, inhibit the tissue catabolic process, and block or even reverse the degenerative process as soon as possible by relying on the biological regulation of FA and PRP.
There are still some limitations to our study. First, in this experiment, the process of injecting hydrogel into the intervertebral disc tissue will inevitably cause damage to the integrity of the fibrous ring tissue and lead to disc degeneration. Therefore, how to safely and accurately deliver the hydrogel biomaterial and minimize the side effects from other manipulations remains an urgent issue to be addressed. In addition, another concern is that although promising results have been achieved in rats, human degenerative disc tissue has a worse microenvironment, such as hypoxia, acidity, and abnormal pressure, which will hinder the clinical translation of experimental results. Therefore, a large number of primates are needed for validation before application in the clinic.
Footnotes
Author Contributions
Qiang Chai and Baining Zhang drafted the manuscript and performed the CCK-8 assay; Yifeng Da performed cell behavior; Yidan Gao and Mingyu Yao performed PCR and Western blot; He Zhu conducted the data analysis; and Yong Zhu and Xuejun Yang designed and supervised this study. All authors read and approved the final manuscript.
Acknowledgments and Funding
This study were supported by the National Natural Science Foundation (82060404) and University Innovation Team Project of the Department of Education of Inner Mongolia Autonomous Region (Fund No.: NMGIRT2229).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal experiments were carried out according to the guidelines for the Care and Use of Laboratory Animals (NIH publication, 8th edition, 2011) and approved by the Institutional Laboratory Animal Care and Use Committee of Inner Mongolia Medical University.
Consent for Publication
Not applicable.
Availability of Data and Materials
All data generated or analyzed during this study are available from corresponding author.
