Abstract
Sirtuins are a class of histone deacetylases that have a wide range of regulatory roles in the cell. Three sirtuins, SIRT3 to SIRT5, localize to and function within the mitochondria. Mitochondrial dysfunction is thought to be the underlying mechanism of several age-related diseases, such as metabolic syndrome, cancer, and neurodegeneration. This review examines current evidence that mitochondrial sirtuins are involved in regulating mitochondrial function and pathogenesis.
Introduction
Over the last decade, many research efforts have been dedicated to understanding the changes in metabolism that contribute to the pathology of cancer, neurodegeneration, and other age-related conditions. An emerging key player in the field is a class of histone deacetylases (HDACs) called sirtuins. Sirtuins, which stands for silent information regulator proteins, are class III HDACs that require nicotinamide adenine dinucleotide (NAD+) for their function. 1 Sirtuins first became molecules of interest when they were discovered to prolong the life span in the yeast Saccharomyces cerevisiae. 2 Seven mammalian homologs (SIRT1-SIRT7) have since been identified, 3 and they were found to have a wide range of roles in mammalian aging processes. 4 In this review, we will focus on the 3 mitochondrial sirtuins, SIRT3 to SIRT5, and their recently discovered, emerging metabolic roles associated with cancer, neurodegeneration, and other age-related diseases.
Mitochondrial Sirtuins
Mitochondria are the main organelles in the cell that are responsible for energy balance and metabolism. They have been implicated in several human diseases, and an important hypothesis has emerged that mitochondrial dysfunction is associated with the onset of age-related disorders and cancer. 5 Supporting this hypothesis are findings that mitochondrial function is impaired in diseases that occur with age, such as cancer,6,7 neurodegenerative diseases, 8 and diabetes. 9 Therefore, mitochondrial sirtuins, which affect mitochondrial function by affecting metabolic processes, will likely have roles in these diseases and conditions.
There are 3 mitochondrial sirtuins that localize to and function within the mitochondria. SIRT3 has strong deacetylase activity, 10 while SIRT4 has adenosine diphosphate (ADP)–ribosylation activity only, 11 and SIRT5 has desuccinylase and demalonylase activity in addition to weak deacetylation activity.12,13 A number of metabolic substrates have been identified for SIRT3, whereas only a handful of substrates have been identified for SIRT4 and SIRT5 (Table 1). These substrates function as part of the basic metabolic machinery of the cell, in tasks such as fatty acid oxidation, electron transport chain function, and urea cycle metabolism 14 (Fig. 1). The mitochondrial sirtuins therefore have a chance to affect basal and pathological metabolic processes in the cell via their interactions with these metabolic substrates. Below, we will discuss these interactions and highlight their possible contributions to pathologies such as metabolic syndrome, cancer, and neurodegeneration.
Key Metabolic Substrates of Mitochondrial Sirtuins
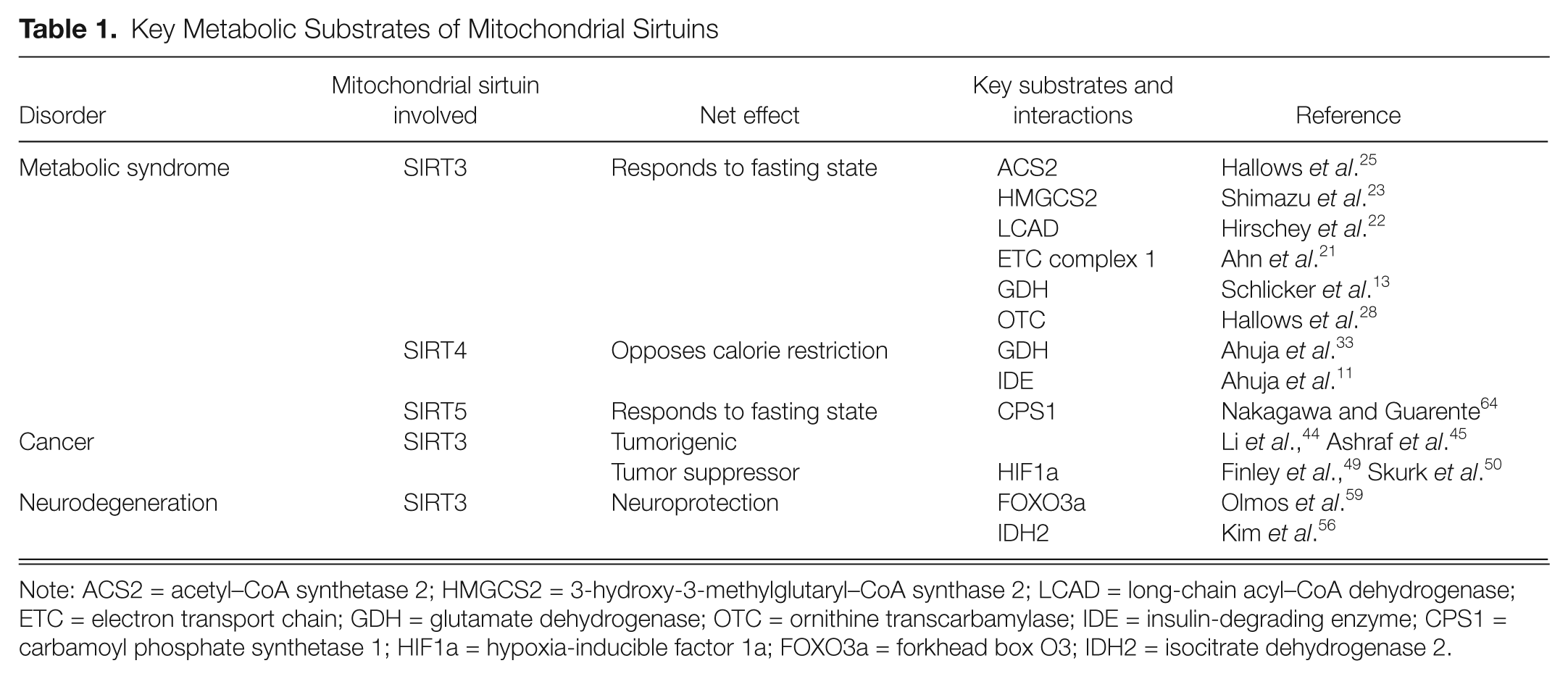
Note: ACS2 = acetyl–CoA synthetase 2; HMGCS2 = 3-hydroxy-3-methylglutaryl–CoA synthase 2; LCAD = long-chain acyl–CoA dehydrogenase; ETC = electron transport chain; GDH = glutamate dehydrogenase; OTC = ornithine transcarbamylase; IDE = insulin-degrading enzyme; CPS1 = carbamoyl phosphate synthetase 1; HIF1a = hypoxia-inducible factor 1a; FOXO3a = forkhead box O3; IDH2 = isocitrate dehydrogenase 2.
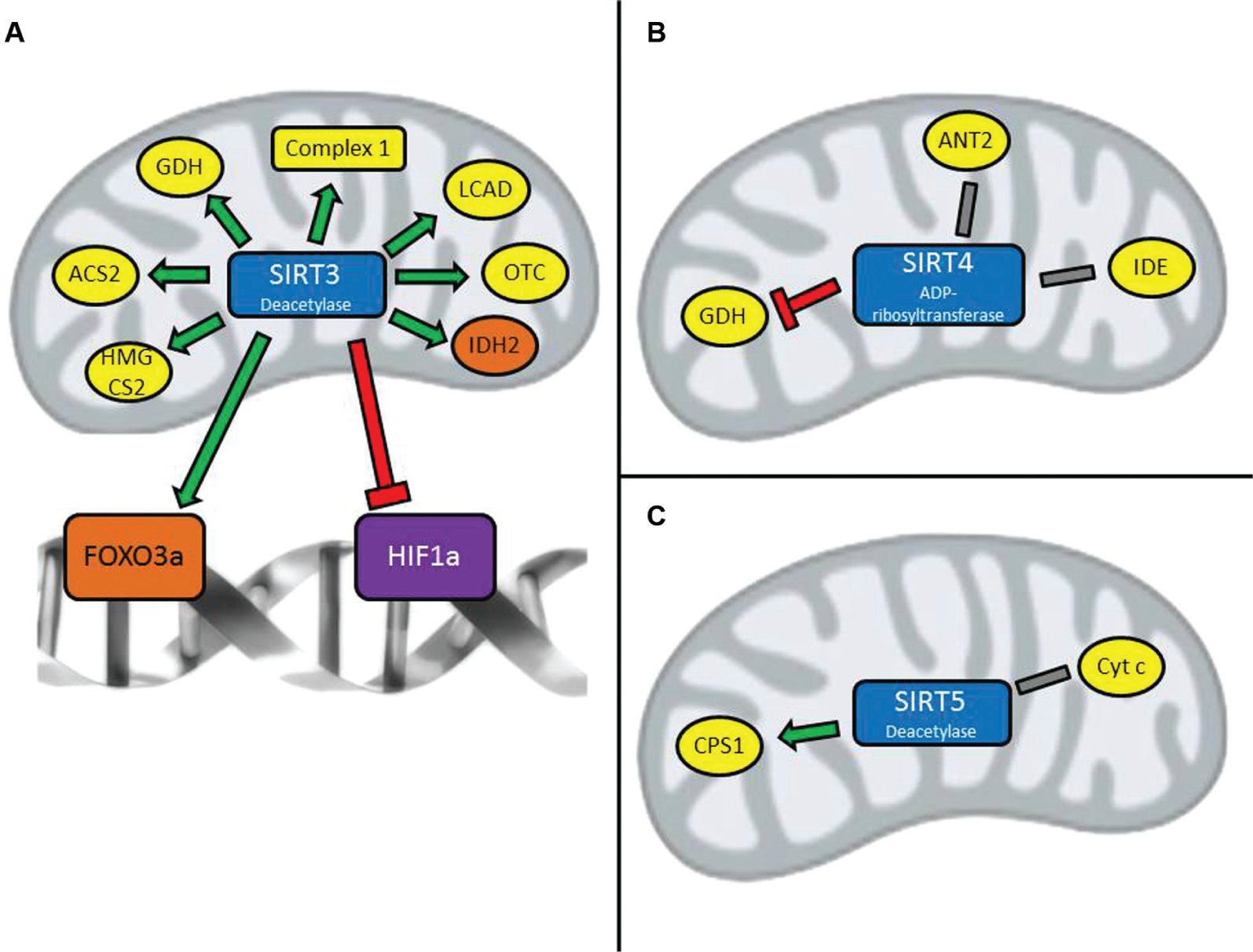
Key actions of mitochondrial sirtuins. SIRT3 (
Metabolic Syndrome
Metabolic syndrome is generally characterized by obesity, hypertension, insulin resistance, and dyslipidemia. The prevalence of metabolic syndrome today is largely due to a Western diet and physical inactivity but is also associated with aging and other genetic factors. 15 The functions of sirtuins in the mitochondria are heavily implicated in metabolic syndrome since mitochondria contain the key cellular metabolism components that are affected in metabolic syndrome. Importantly, sirtuins mediate the beneficial effects of calorie restriction, 16 which oppose the effects of metabolic syndrome. Through several studies on sirtuins and calorie restriction, it was shown that sirtuin function is dependent on the energy status of an organism, and sirtuins are able to regulate metabolic pathways accordingly.17,18 Additionally, it has also been shown that metabolic enzymes are regulated by acetylation,19,20 and therefore, the deacetylase capabilities of sirtuins will be crucial for metabolic regulation.
Role of SIRT3
SIRT3 regulates a number of metabolic enzymes in the cell by deacetylation. It is known to respond to the energy level in a cell and, in turn, act on various components of the tricarboxylic acid (TCA) cycle, fatty acid oxidation, and oxidative phosphorylation. 14 In general, SIRT3 is activated when nutrient levels in the cell are low and NAD+ levels are increased, promoting metabolic processes that are characteristic of a fasting state: amino acid catabolism, 13 basal levels of oxidative phosphorylation, 21 fatty acid oxidation, 22 and ketone body synthesis. 23 SIRT3 also promotes adaptive thermogenesis and promotes activity within brown fat. 24
SIRT3 was first discovered to deacetylate and activate acetyl–CoA synthetase 2 (ACS2). 25 ACS2 converts free acetate to acetyl-CoA, which is the most abundant intermediate metabolism molecule in the cell. Acetyl-CoA can be oxidized in the citric acid cycle (TCA cycle) to produce energy, and impaired acetate metabolism has been demonstrated in diabetic patients. 26 SIRT3 contributes to the fasting state of the cell by promoting fat metabolism and amino acid metabolism. It has been shown to deacetylate and activate long-chain acyl–CoA dehydrogenase (LCAD), promoting fatty acid oxidation. 22 Under starvation conditions, when the cell depends on fatty acid catabolism for energy, SIRT3 deacetylates and activates 3-hydroxy-3-methylglutaryl–CoA synthase 2 (HMGCS2), promoting the formation of ketone body by-products from acetyl-CoA. 23 During starvation, cells also rely on amino acid catabolism, and SIRT3 has been shown to deacetylate and activate glutamate dehydrogenase (GDH), which forms α-ketoglutarate from the amino acid glutamate. 13 SIRT3 also deacetylates and activates ornithine transcarbamylase, a key component of the urea cycle, which is important to prevent toxic levels of ammonia build-up from amino acid catabolism.27,28
The loss of SIRT3 in vivo leads to hyperacetylation of these enzymes, with implications for altered metabolic function. 10 Several studies were then conducted to assess if and how the loss of SIRT3 and hyperacetylation of metabolic enzymes contribute to metabolic syndrome. SIRT3 knockout mice, high-fat diet administration, and mouse diabetes models were used in several studies. One study showed that a high fat diet is associated with a decrease in SIRT3 protein expression. 29 The same study also showed that the loss of SIRT3 led to decreased mitochondrial oxidation and increased reactive oxygen species (ROS) production, which then induces a signaling cascade that ultimately decreases insulin release and contributes to metabolic syndrome characteristics. Another study showed that the SIRT3 knockout mice have hyperacetylated mitochondrial proteins, which ultimately increase their susceptibility to and progression of metabolic diseases. 30 This study identified a polymorphism in the human SIRT3 gene that decreases SIRT3 function and is associated with the development of metabolic syndrome, indicating a genetic basis for the disorder.
Role of SIRT4
SIRT4 was first discovered to ADP-ribosylate and inhibit GDH, an action that inhibits insulin release by the pancreas. 31 Insulin is a central metabolic regulator that responds to energy levels in the body and is released in response to increased levels of glucose in the bloodstream or amino acids such as glutamate. 32 Insulin signals the cell to use carbohydrate metabolism for energy. Therefore, by negatively regulating insulin secretion in response to glucose and amino acids, SIRT4 opposes carbohydrate metabolism as seen in calorie restriction. 33 This finding indicates that SIRT4 might play a part in metabolic syndrome via GDH by adding another layer of regulation to the metabolic processes that are known to be involved. Indeed, SIRT4 has also been shown to interact with insulin-degrading enzyme (IDE) and the mitochondrial adenosine-5′-triphosphate (ATP)/ADP translocase, ANT2/3, ultimately leading to a decrease in insulin secretion. 34 One function of IDE is to degrade insulin and control insulin levels in the bloodstream; loss of function mutations of IDE can lead to diabetes in experimental mouse models. 35 Although it is not known what the significance of SIRT4 interaction with IDE is, one can speculate that SIRT4 might also play a role in the onset of metabolic syndrome by affecting IDE function. Similarly, ANT2/3 has roles in regulating the cytosolic levels of ATP. Changing levels of ATP in the cell might trigger responses in many other metabolic pathways, which provides another possible mechanism by which SIRT4 can affect metabolic function in the cell.
Role of SIRT5
SIRT5 was first identified to regulate entry into the urea cycle and promote urea cycle function by deacetylating and activating carbamoyl phosphate synthetase 1 (CPS1). 36 SIRT5 might also regulate CPS1 via desuccinylation. 12 The urea cycle is important for metabolizing and detoxifying ammonia, which can accumulate during amino acid catabolism. Therefore, it is logical that SIRT5 activates CPS1 and the urea cycle during fasting states or with a high protein diet, when higher levels of amino acid breakdown are required. These situations indicate that there is less fat and carbohydrate available for energy consumption. Since SIRT5 regulates these metabolic processes during specific energy states, the loss of SIRT5 function might have a role in metabolic diseases, either contributing to or resulting from abnormal energy profiles in these patients.
Cancer
Cancer is a widespread, heterogeneous disease that is the leading cause of death worldwide. It is characterized by oxidative damage, which then leads to metabolic imbalance within the cell, abnormal macromolecule production, and uncontrolled tumor growth. Uncontrolled tumor growth is attributed to a phenomenon called “aerobic glycolysis,” where cells have high rates of glycolysis and lactic acid production in the presence of oxidative phosphorylation.37,38 Normally, cells either function anaerobically and produce lactic acid or consume oxygen and proceed with oxidative phosphorylation; the upregulation of glycolysis in the presence of oxygen does not occur normally and indicates malfunction. Since glycolysis and oxidative phosphorylation are both mitochondrial processes, mitochondrial dysfunction is implicated in cancer progression. It has been shown that oncogenes, such as Ras, Akt kinase, and Myc, promote metabolic processes such as glycolysis and glucose uptake.39-41 Now, as mitochondrial sirtuins have emerged as metabolic regulators, they are being studied intensely for their possible roles in cancer progression.
Role of SIRT3
Since SIRT3 is a key mitochondrial deacetylase that affects the function of metabolic enzymes and oxidative phosphorylation components, and since abnormal metabolic profiles in cells are a hallmark of cancer pathology, it seems likely that SIRT3 would play some role in cancer progression. The precise role, however, is unclear, as there is evidence for both tumorigenic and tumor-suppressing functions of SIRT3.
On one hand, SIRT3 has been shown to be prosurvival, preventing cell death in response to stress and starvation. 42 SIRT3 has also been shown to protect from cell death in response to hypoxia or the apoptotic inducer staurosporine. 43 These studies support the general role of sirtuins in promoting longevity, but it also might indicate a role in tumor growth. Indeed, SIRT3 has been shown to promote cell proliferation in oral cancer cell lines 44 and to block the growth arrest function of the transcription factor p53 in bladder cancer cells. 45 SIRT3 gene expression levels are also increased in malignant human breast biopsies. 46
On the other hand, SIRT3 has been shown to be proapoptotic under certain pathway conditions. 47 SIRT3 has also been identified as a tumor suppressor in several studies, which directly link SIRT3 to metabolic processes. The loss of SIRT3 in mouse embryonic fibroblasts (MEFs) was first shown to induce tumor growth in response to oncogene expression. 48 SIRT3 expression in MEFs was also shown to destabilize HIF1a, a transcription factor that promotes glycolytic metabolism as dictated by the “Warburg effect.”37,49,50 The same study also showed that SIRT3 overexpression in the breast cancer cell lines MCF7, T47D, and CAMA1 reverses the characteristic metabolic profile indicated by the Warburg effect. SIRT3 has also been shown to interact with the FOXO3a transcription factor, which is known to upregulate apoptotic signaling cascades. 51 This is one mechanism by which SIRT3 might act as a tumor suppressor. SIRT3 might also prevent cancer by deacetylating and activating mitochondrial superoxide dismutase (MnSOD), which is thought to control the levels of ROS and prevent oxidative damage that triggers cancer pathology.52,53
These conflicting results show that there are dual hypothetical roles of SIRT3 in cancer and tumor progression. Since SIRT3 is involved in many aspects of cellular metabolism, including fatty acid oxidation, amino acid catabolism, and oxidative phosphorylation, its role in cancer is likely to be complex and multidimensional. Different stimuli and environmental effects seem to trigger different responses from SIRT3, and it will be interesting to determine which external stimuli will trigger specific regulatory roles of SIRT3.
Role of SIRT4 and SIRT5
SIRT4 and SIRT5 have not been studied in the context of cancer. However, given their roles in regulating metabolism, it would be very interesting to study whether they have tumor suppressor or tumorigenic roles. The metabolic functions of SIRT4 and SIRT5 that have been discovered so far, such as SIRT4 inhibition of insulin release, can possibly mirror or antagonize the established effects of oncogenes on metabolism, and further studies are necessary to place SIRT4 and SIRT5 actions in the context of cancer.
Neurodegeneration
Neurodegeneration is the process by which neurons are lost due to genetic and environmental stressors. There are many types of neurodegenerative diseases, which affect different populations of neurons and cause a range of untreatable symptoms; the most common types are Alzheimer disease, Parkinson disease, amyotrophic lateral sclerosis (ALS), and Huntington disease. The central risk factor for neurodegeneration is advanced age. Sirtuins have been implicated in neurodegenerative diseases due to their reputation as aging, stress response genes 42 and due to the fact that they are highly expressed in the brain and central nervous system. 54 Furthermore, mitochondria are also implicated in aging processes as their function declines.5,8 Therefore, mitochondrial sirtuins present promising targets to study in deducing the pathology of neurodegeneration.
Role of SIRT3
SIRT3 is known to deacetylate many substrates in the mitochondria that are important to metabolic processes, but this has mainly been shown in other tissues besides the brain. In general, SIRT3 displays protective effects against ROS production by upregulating SOD function.24,52,53,55,56 Since ROS have been shown to induce neurodegenerative processes, 8 SIRT3 might be protective in neurons against ROS-related damage. SIRT3 was first shown to be protective against excitotoxic injury in primary neuron cultures. 57 In this study, SIRT3 was increased following massive ROS production induced by the addition of N-methyl-D-aspartate (NMDA), indicating the role of SIRT3 as a downstream effector of ROS injury. Recently, SIRT3 was shown to be neuroprotective in response to Alzheimer disease–induced cellular stress in addition to ROS-induced injury. 58 Possible mechanisms for SIRT3 upregulation in response to neuronal stress include interaction with the FOXO3 transcription factors. SIRT3 has been shown to interact with FOXO3a, but the physiological implications, especially in the context of neurodegeneration, are not clear. 59 FOXO3 transcription factors are thought to offer protection from oxidative stress by activating detoxifying genes such as catalase and MnSOD. 60 In addition, while FOXO3 may function as a tumor suppressor in other tissues, its role in apoptosis has implications for sirtuin-mediated neurodegeneration. 61 More studies are needed to determine whether SIRT3 can be neuroprotective through its interaction with FOXO3a or if there is another mechanism involved.
Role of SIRT4 and SIRT5
The role of SIRT4 and SIRT5 in neurodegeneration is not clear. While recent discoveries of SIRT4 and SIRT5 substrates in the cell and other metabolic tissues may provide some clues, little is known about SIRT4 and SIRT5 function in brain tissues. The fact that SIRT4 interacts with IDE is one example 11 since IDE has been shown to degrade pathological β-amyloid plaques. 62 Another example is SIRT5 interaction with cytochrome c 13 since cytochrome c is released from the mitochondria during apoptosis. 63 Additional studies have to be conducted to understand the functions of mitochondrial sirtuins in the brain.
Concluding Remarks
This review highlights the major roles of mitochondrial sirtuins in regulating metabolism and how these roles then affect the progression of metabolic syndrome, cancer, and neurodegeneration. In many cases, the roles of mitochondrial sirtuins seem to have opposing effects in different tissue type and disease settings. For example, the apoptotic functions of mitochondrial sirtuins might be beneficial in preventing tumor growth but might also facilitate neurodegeneration. This review has speculated on several dual regulatory roles of mitochondrial sirtuins, but much more work is needed to untangle the complex metabolic and regulatory pathways these sirtuins are involved in. Understanding mitochondrial sirtuin function will undoubtedly lead to improved therapeutic strategies for age-, metabolism-, and stress-related diseases, benefiting a large proportion of our current population.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The research in Donmez lab is supported by a start-up fund from the Department of Neuroscience, Tufts Center for Neuroscience Research Pilot Award, Charlton Research Award, and Ellison Medical Foundation New Scholar in Aging Award (to G. D.). J.S. is a graduate student in Neuroscience Program at Sackler School of Graduate Biomedical Sciences, Tufts University.
