Abstract
Deregulation of c-Jun NH2-terminal kinase (JNK) signaling occurs frequently in a variety of human cancers, yet the exact role(s) of JNK deregulation in cancer cell biology remains to be fully elucidated. Our recent demonstration that the activity of JNK is required not only for self-renewal of glioma stem cells but also for their tumor initiation has, however, identified a new role for JNK in the control of the stemness and tumor-initiating capacity of cancer cells. Significantly, transient JNK inhibition was sufficient to cause sustained loss of the tumor-initiating capacity of glioma stem cells, suggesting that the phenotype of “lost tumor-initiating capacity” may be as stable as the differentiated state and that the tumor-initiating capacity might therefore be under the control of JNK through an epigenetic mechanism that also governs stemness and differentiation. Here, in this article, we review the role and mechanism of JNK in the control of this “stemness-associated tumor-initiating capacity” (STATIC), a new hypothetical concept we introduce in this review article. Since the idea of STATIC is essentially applicable to both cancer types that do and do not follow the cancer stem cell hypothesis, we also give consideration to the possible involvement of JNK-mediated control of STATIC in a wide range of human cancers in which JNK is aberrantly activated. Theoretically, successful targeting of STATIC through JNK could contribute to long-term control of cancer. Issues to be considered before clinical application of therapies targeting this JNK-STATIC axis are also discussed.
Introduction
The c-Jun NH2-terminal kinases (JNKs) are a subgroup of mitogen-activated protein kinases that has long been implicated in cancer development. Although accumulating evidence suggests that JNK may contribute to the development of cancer as a signal transducer of cellular proliferation, differentiation, survival, and migration, the exact role(s) of JNK in cancer still remains to be fully delineated.1-4 Only recently, we and others have shown that JNK is required for self-renewal and the tumor-initiating capacity of human glioma stem cells, giving rise to the novel possibility that the maintenance of cancer stem/initiating cells may be one of the critical roles of JNK in cancer development.5,6 Most importantly, we have demonstrated for the first time using preclinical animal tumor models that therapeutic targeting of JNK is a safe and effective measure to selectively eliminate the tumor-initiating stem cell population of glioblastoma cells
Tumor-Initiating Capacity and Self-Renewal: 2 Disparate yet Closely Associated Characteristics of Cancer Stem/Initiating Cells That May Be under the Control of JNK
The cancer stem cell hypothesis posits that tumors are heterogeneous, being composed of a rare subpopulation of tumor cells termed cancer stem cells and the remaining cells accounting for the vast majority of the tumor cells.12-16 The hypothetical cancer stem cells, but not the remaining tumor cells (nonstem cancer cells), possess the capacity to initiate a tumor that reproduces the heterogeneity and characteristics of the original tumor when transplanted
Here, we introduce a novel hypothetical concept of “stemness-associated tumor-initiating capacity” (STATIC), an evolutionary version of the cancer stem cell hypothesis. Significantly, since Pierce’s observations, researchers have consistently observed that self-renewing, undifferentiated tumor cells have a higher capacity to initiate a tumor compared to their differentiated counterparts.15,24 This is, in a sense, quite surprising because the control of self-renewal/differentiation and that of tumor initiation are both intricate cellular functions apparently disparate from each other except that both may be associated with the capacity to undergo limitless cellular replication. The close association between these apparently distinct cellular mechanisms, however, points to the possibility that they share a common regulatory module. Also, the regulatory module most likely involves epigenetic mechanisms since the control of self-renewal and differentiation is known to be epigenetically controlled.25,26 In addition, the mechanism controlling self-renewal/differentiation should originally be subject to control by cell-extrinsic factors, 27 so that cell fate decision may be coordinated to maintain the homeostasis of the whole tissue (e.g., to keep the stem cell population small relative to the entire population). Put together, these ideas are schematically presented in Figure 1A. Figure 1A illustrates that 1) the tumor-initiating capacity is under the control of a common mechanism that maintains the self-renewal capacity/undifferentiated state (= stemness), 2) the mechanism is most likely epigenetic, and 3) the mechanism is subject not only to cell-intrinsic but also to cell-extrinsic factors. According to this hypothetical model (STATIC), the subpopulation of immature tumor-initiating cells may not necessarily be small in cancers in which the extrinsic factor–mediated control mechanism is disrupted (Fig. 1B). In cancers in which the epigenetic control of differentiation is lost (Fig. 1C), tumor-initiating cells may no longer be sorted by markers associated with a cellular stem/differentiation status. It is also possible that self-renewing tumor cells may not necessarily be tumor-initiating cells in some cancers (Fig. 1D). Thus, while being applicable to cancers that do not follow the cancer stem cell hypothesis, on one hand, this hypothetical model, on the other hand, inherits the advantages of the original cancer stem cell hypothesis and has even more:
The identification of genes/molecules controlling tumor initiation is time consuming and laborious because it inevitably involves animal xenograft studies. However, both the cancer stem cell hypothesis and our new one predict that we can greatly facilitate the identification process by focusing on surrogate markers of tumor initiation. For instance, by using stem cells derived from cancers that conform to the cancer stem cell hypothesis as a model, exploration of candidate molecules can be done simply by
According to the hypotheses, transient targeting of the identified genes/molecules is supposed to provide a sustained inhibitory effect on the tumor-initiating capacity of tumor cells because the condition of “lost tumor-initiating capacity” is expected to be as epigenetically stable as the differentiated state. In case the loss of tumor-initiating capacity is “irreversible,” the therapeutic intervention could have a “curative” effect.
Once identified, it would be feasible and relevant to explore the role of the genes/molecules involved in the regulation of STATIC even in cancers that do not conform to the cancer stem cell hypothesis, as may be explained by Figure 1B and 1C.
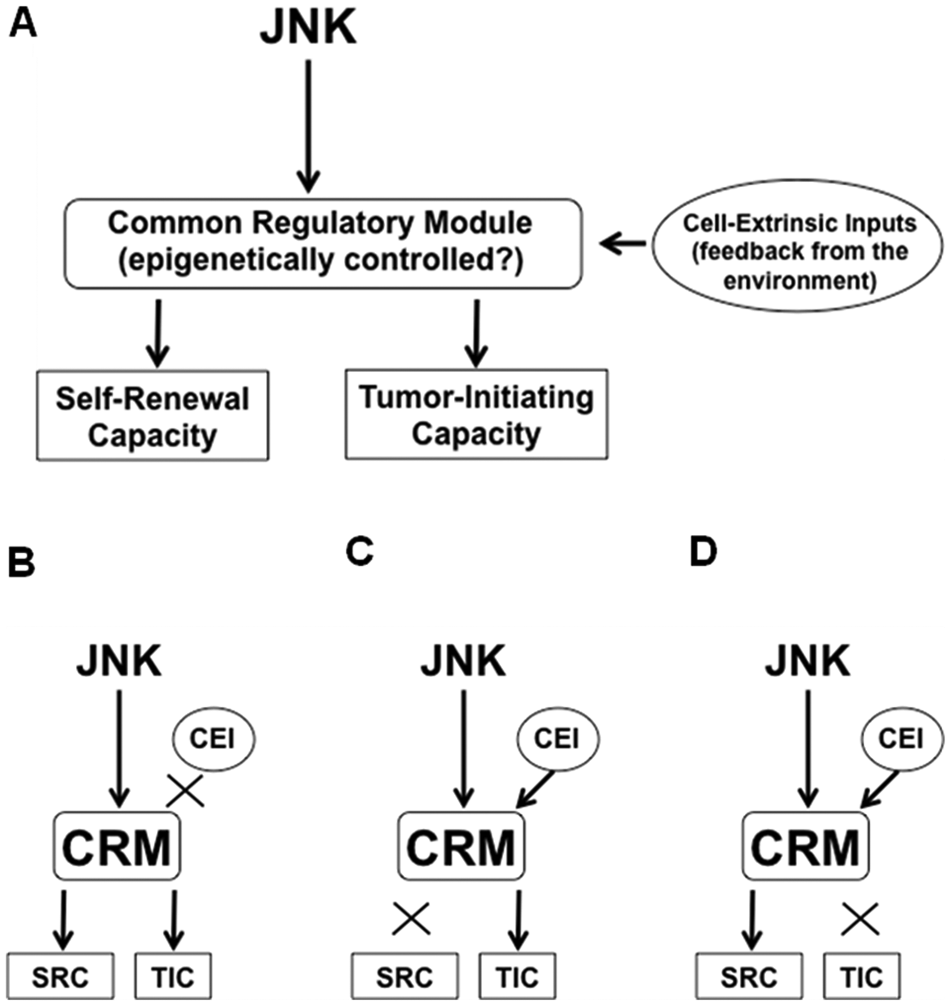
Stemness (= capacity to self-renew as immature, undif- ferentiated cells) and tumor-initiating capacity may be 2 disparate yet closely associated properties of cancer cells that are controlled by a common regulatory module and are also subject to modulation by cell-extrinsic inputs. (
Based on a prototypical idea of this hypothetical STATIC model, we set out to search for molecules involved in the control of the tumor-initiation capacity of glioblastoma cells. Consequently, we discovered that JNK is among the key molecules regulating STATIC of glioblastoma cells. 5
Role of JNK in the Control of STATIC of Glioblastoma Cells
JNK is more activated in glioma stem cells than in their differentiated counterparts
In our recently reported study, 5 we searched for molecules differentially expressed and/or activated in self-renewing glioma stem cells and in those that have undergone serum-induced differentiation, with the intention to identify molecules involved in the control of STATIC of glioblastoma cells. Examination using 6 glioma stem cell lines established directly from patient glioblastoma tissues or from conventional glioblastoma cell lines revealed that the JNK pathway is consistently more activated in self-renewing glioma stem cells than in their differentiated counterparts, suggesting that JNK may be involved in the maintenance of the undifferentiated stem cell state (i.e., stemness) of glioblastoma cells.
Activation of JNK in human glioblastoma
So far, a series of studies examining the expression and activation (= expression of the phosphorylated form) of JNK in human glioblastoma tissues by immunoblot analysis have demonstrated that JNKs are expressed and activated in the majority of glioblastoma cases.28-30 Strong expression of phosphorylated JNK in the majority (>90%) of glioblastoma cases has been confirmed independently by an immunohistochemical study, which also showed that JNK activation is associated with the histological grade of glioma and is virtually nil in the normal brain. 31 A subcutaneous xenograft experiment using serum-cultured U87 glioblastoma cells constitutively expressing a dominant-negative form of JNK showed that the tumor growth of glioblastoma is retarded when JNK activity is inhibited. 30 Together, the reports consistently demonstrated that JNK is activated in glioblastoma tissues and also suggested that the activation of JNK may have a role in promoting the tumor growth of glioblastoma. Nevertheless, the exact role(s) of JNK in glioblastoma biology still remained to be fully elucidated.
JNK is essential for the maintenance of the stemness of glioma stem cells
To determine, therefore, whether the maintenance of stemness is among the critical roles of JNK in glioblastoma cells, we examined, in a recently reported study,
5
the impact of JNK inhibition on the stem cell properties of glioma stem cells. Both pharmacological and genetic inactivation of JNK in glioma stem cells
JNK is essential for the maintenance of the tumor-initiating capacity of glioma stem cells
We then examined whether JNK, which has just been shown to control the stemness of glioma stem cells, is also involved in the maintenance of the tumor-initiating capacity in a manner associated with the stemness of glioma stem cells. Orthotopic xenograft experiments demonstrated that transient JNK inactivation
Therapeutic targeting of JNK to control STATIC of glioma stem cells in vivo
Given that cancer stem cell research is an applied medical science in contrast to normal stem cell research, which is essentially a basic one, it was of utmost importance for us to demonstrate the medical significance of our findings. To test, therefore, the clinical relevance of JNK as a therapeutic target, we examined whether
Mechanism of JNK-Mediated Regulation of the Stemness and Tumor-Initiating Capacity of Glioblastoma Cells
While it is now clear from what has been described so far that JNK plays a pivotal role in the control of the stemness and tumor-initiating capacity of glioblastoma cells, much remains to be shown as to how JNK is activated in glioblastoma cells and how JNK controls the stemness and tumor-initiating capacity of glioblastoma cells. We therefore discuss possible mechanisms in this section (Fig. 2).
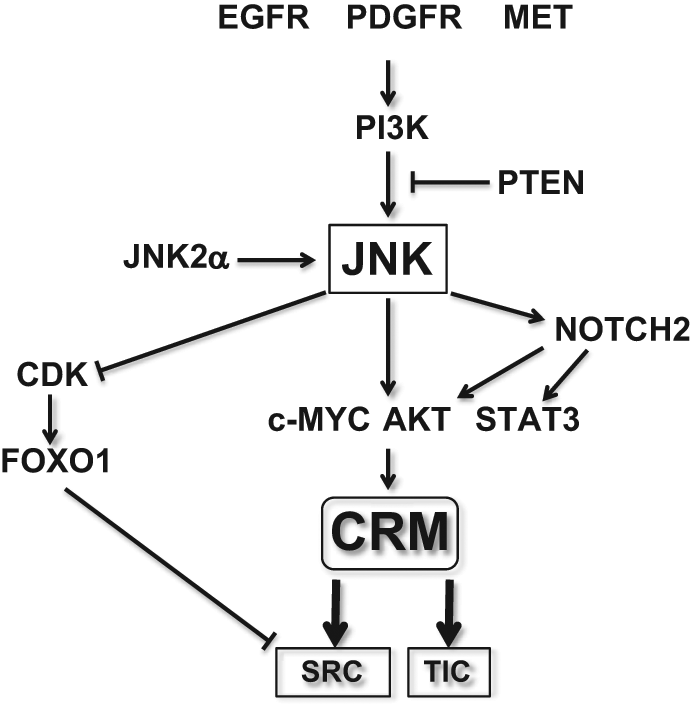
A hypothetical model for JNK-mediated regulation of the stemness and associated tumor-initiating capacity in glioma stem cells. The possible mechanistic components that are assumed to control or to be under the control of JNK in glioma stem cells are schematically depicted. See text for details. Also note that not all the functional interactions depicted in the figure have been demonstrated in glioma stem cells. CRM = common regulatory module; SRC = self-renewing capacity; TIC = tumor-initiating capacity.
How is JNK activated in glioblastoma cells?
Then, how do these receptor tyrosine kinases control JNK and the stemness of glioma stem cells? Recent evidence suggests that PI3K is required for the maintenance of the self-renewal capacity and stem cell marker expression of glioma stem cells45,46 and that the JNK signaling pathway is a downstream target of PTEN/PI3K. 47 Just in line with these findings, PI3K-dependent activation of JNK has been implicated in the maintenance of the stemness of glioma stem cells. 6 Since EGFR, PDGFR, and MET activate intracellular pathways mediated by PI3K, 48 PI3K may possibly play a role as a molecular link between these receptor tyrosine kinases and JNK, although this idea has not yet been experimentally validated. It would also be of interest to speculate that the significant impact of the PTEN status on the stem cell phenotypes of glioma stem cells 49 is mediated through PI3K-dependent activation of JNK.
In addition to the mechanism of JNK activation by upstream signals, an autoactivation mechanism of JNK has been reported.29,30,50 JNK2 isoforms, in particular JNK2α2, autophosphorylate and activate themselves without requiring the participation of upstream kinases.50,51 Significantly, it has been suggested that such autoactivated JNK isoforms are expressed in a great majority of glioblastoma cases.29,30 Apparently, the expression of the autoactivated JNK isoforms is of therapeutic importance since attempts to control JNK by targeting upstream molecules would be futile if such isoforms account for the majority of the JNK activity in glioblastoma cells.
Another important issue that may deserve consideration in this section is whether the JNK activity level serves as a key determinant in the cell fate (i.e., stem or nonstem) decision of glioma stem cells in patient glioblastomas. Although we observed that JNK activity is decreased in association with the serum-induced differentiation of glioma stem cells
How does JNK control the stemness and tumor-initiating capacity of glioblastoma cells?
We and others have shown that JNK maintains the stemness and tumor-initiating capacity of glioma stem cells through its kinase activity.5,6 Among known direct substrates of JNK, AKT and c-MYC have been implicated in the control of the stemness and tumor-initiating capacity of glioma stem cells.52-54 Since JNK phosphorylation of Thr450 of AKT reportedly activates its kinase activity 55 and phosphorylation of Ser62 and Ser71 of c-MYC is required for its biological function, 56 JNK may control the characteristic properties of glioma stem cells through such phosphorylation-mediated regulation of AKT and/or c-MYC. Of note, in addition to their role in the control of the stem cell state of glioma stem cells, AKT and c-MYC may also contribute to the maintenance of glioma stem cells by promoting their survival.52,53 Another JNK substrate of interest is STAT3. Accumulating evidence suggests that dysregulation of STAT3 has a role not only in glioblastoma cells in general but also in their stem cell population, and as such, STAT3 is currently regarded as a potential target of glioblastoma therapy. 57 Although the roles of JNK in STAT3 regulation are not simple and appear to be multifaceted, JNK may possibly maintain the stemness and/or tumor-initiating capacity of glioblastoma cells through phosphorylation-mediated activation of STAT3 since direct phosphorylation of STAT3 by JNK on its serine residue (Ser727) has been shown to promote STAT3-mediated gene regulation.58-60
We found in our study that FOXO1 is activated upon JNK inhibition and that its activation is required for JNK inhibition–induced differentiation of glioma stem cells. 5 Although FOXO1 may not be a direct substrate for JNK and the mechanism of JNK-mediated inhibition of FOXO1 remains largely obscure at this moment, a recent report suggested a role for CDK in the JNK control of FOXO1. 61 Importantly, knockdown of FOXO1 prevented the differentiation but not loss of stem cell marker expression and sphere-forming capacity caused by JNK inhibition, suggesting that FOXO1 function may not be required in the earlier phase of differentiation but is required for full differentiation. 5 Nevertheless, prevention of premature activation of FOXO1 appears to be one of the mechanisms by which JNK maintains glioma stem cells. Meanwhile, NOTCH2 has also been implicated in JNK-mediated control of glioma stem cells, based on the observation that NOTCH2 expression is closely associated with the JNK activity and hence the stem cell state of glioma stem cells. 6 Given the demonstrated role of NOTCH in the control of glioma stem cells that likely involves AKT and STAT3, 62 NOTCH2 may also have a role in the JNK control of glioma stem cells through AKT and STAT3.
Issues to Be Considered before Clinical Application of the JNK-Targeting Therapy to Glioblastoma Treatment
How long does the therapeutic effect of transient JNK inhibition last?
Our study demonstrated that the tumor- initiating capacity of glioma stem cells is closely coupled with their stem cell state and that transient JNK inhibition results in sustained loss of their tumor-initiating capacity. These findings indicate that, as discussed earlier, the tumor-initiating capacity of glioma stem cells is indeed stemness associated and is therefore most likely under an epigenetic control. Thus, one could safely expect that once we successfully deprive glioblastoma cells of their tumor-initiating capacity, the condition (loss of tumor-initiating capacity) would then be as stable as the differentiated state and last for a long time period. However, although epigenetic factors are usually stable, they are not necessarily static and can be modulated by the environment.
25
In this regard, use of mouse xenograft models apparently has its own limitation in examining how stable the condition of “lost tumor-initiating capacity” is since the stability cannot be assessed beyond the life span of nude mice (~2 years). It is therefore expected that future clinical trials will provide valuable information regarding this issue through the demonstration of whether and to what extent targeting of STATIC is of therapeutic benefit. On the other hand, what should we do if the condition of “lost tumor-initiating capacity” turns out to be not so stable as to prevent tumor recurrence during the rest of the patients’ lives? In such cases, it would be of benefit to try to irreversibly eliminate glioma stem cells with cytotoxic therapies while the cells remain in the nonstem condition after the JNK-targeting therapy since the stem cell state has been associated with higher therapeutic resistance than the nonstem cell state and
Which should be spared: neural stem cells or the brain function?
For clinical application of therapeutic agents targeting glioma stem cells, it is essential that they do not cause deleterious adverse events at therapeutic doses. Given that glioma stem cells, and possibly other cancer stem cells as well, may share properties with normal tissue stem cells including neural stem cells, the possible effects of cancer stem cell–targeting therapies on neural stem cells should deserve particular consideration. However, rather surprisingly, there has been no definitive proof that adult neurogenesis has an essential role in learning and memory, with most of the studies investigating the role of adult neurogenesis in learning and memory proving to be negative.64,65 There are a few that report positive results; however, the results are inconsistent even among the positive reports.
65
Furthermore, the reported differences are in general quite subtle despite substantial ablation of neurogenesis, and the possibility of the ablation methods affecting beyond neurogenesis has not been excluded.
65
Most convincingly, the role of adult neurogenesis in learning and memory was called into question by the demonstration that cyclin D2–null mice, in which adult neurogenesis is almost totally lost, performed as well as the wild-type mice in any behavioral tasks known to be hippocampal dependent.
65
Although these findings may not totally exclude the possibility that adult neurogenesis has some role in cognitive functions, in particular when the brain is damaged, it is clear from the findings that glioma/cancer stem cell–targeting agents should be assessed not in terms of whether or not they affect neural stem cells and/or neurogenesis but in terms of whether they do affect the brain function
In this line, we investigated in our study the effect of systemic JNK inhibitor treatment on the brain function of nude mice. 5 Our JNK inhibitor treatment protocol using SP600125 had no discernible effect on mice when they were examined by the Y-maze test, which is currently regarded as a sensitive indicator of brain lesions, including hippocampal lesions, rather than a specific indicator of memory function alone.66-68 Together with the reported neuroprotective activity of SP600125,69,70 the JNK inhibitor may be suitable for clinical application also in view of its effects on the nervous system.
Role for JNK in the Control of STATIC in Human Cancers: Is STATIC a Paradigm Limited to Glioblastoma?
We have demonstrated for the first time that JNK is involved in the control of STATIC, using glioma stem cells as an experimental model. 5 Undoubtedly, it is of interest and importance whether such a role for JNK could be generalized to other human cancer types.
Documented role for JNK in the tumor initiation process of human cancers other than glioblastoma
Relevant literature indicates that JNK is deregulated not only in glioblastomas but also in other human cancers.
4
Furthermore, the essential role of JNK has been documented at least in some of them using animal models corresponding to human cancers. For instance, in an animal model of chemically induced hepatocellular carcinoma, loss of the
JNK control of STATIC may possibly go beyond brain tumors
Collectively, the studies in the literature indicate that JNK has a pivotal role in the initiation of animal models of human cancers in which JNK is deregulated. Of note, in those studies, the mechanism of JNK-dependent tumor initiation has been accounted for mostly by JNK-dependent proliferation of tumor cells, and the possible role of JNK as a regulator of cancer stem cells and/or STATIC has not been discussed. However, as clearly demonstrated by the instance of glioblastoma whereby JNK does have a crucial role in cancer stem cells5,6 in addition to its role in bulk tumor cells, 30 it is quite conceivable that JNK also has a cancer stem cell–associated role also in those human cancers with JNK deregulation. In line with this idea, we have recently found, using human cancer cells of nonbrain tumor origin, that JNK is required for what is most likely to be STATIC of the human cancer cells (manuscript in preparation). Together, these lines of evidence strongly suggest that the paradigm may not be unique to glioblastoma and may be shared at least by some other human cancers.
Concluding Remarks
The JNK signaling pathway is aberrantly activated in various human cancers, and the growing body of evidence suggests that it has a definitive role in, albeit not limited to, tumor initiation.1-4 Combined with the recent demonstration that JNK is involved in the control of STATIC of glioblastoma cells, the evidence is now in support of the idea that a similar mechanism may be operative in other human cancers.5,6,76 Apparently, testing of this idea is warranted in future studies: if the mechanism is shared by many human cancers, JNK targeting would realize the long-term control of such cancers, irrespective of whether they follow the cancer stem cell model or not. Currently, the concept of “STATIC” is only operationally defined. Future elucidation of how JNK controls the molecular mechanism underlying STATIC will not only help redefine the concept in molecular terms but also help identify novel molecular targets for the long-term, hopefully curative, control of human cancers.
Footnotes
Acknowledgements
The authors are indebted to Dr. Kimishige Ishizaka for invaluable advice on writing this review article. They also thank members of their laboratory and their collaborators for productive and stimulating discussion and Dr. Tomoko Kagawa for her continuous support and encouragement.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants-in-Aid for Scientific Research, Challenging Exploratory Research, and Young Scientists from the Ministry of Education, Culture, Sports, Science and Technology of Japan; a Grant-in-Aid from the Global COE Program of the Japan Society for the Promotion of Science; the National Cancer Center Research and Development Fund (23-A-20); and a grant from the Japan Brain Foundation.
