Abstract
To define and therapeutically target mechanisms that mediate nasopharyngeal carcinoma (NPC) metastasis, we have developed a unique orthotopic xenograft mouse model that accurately recapitulates the invasive and metastatic behavior of human disease. Based on clinical and laboratory evidence that the PI3K/Akt/mTOR axis is involved in aggressive NPC tumor behavior, we chose it as a therapeutic target to test the utility of our orthotopic system for evaluating the effectiveness of a targeted treatment for metastatic NPC. Demonstrated herein, we have shown that both the development and growth of metastatic lesions are markedly reduced by the mTOR inhibitor sirolimus. Thus, this orthotopic model provides a platform to study potential therapeutics for advanced NPC and demonstrates that targeting the PI3K/Akt/mTOR pathway is a promising intervention against disseminated disease.
Introduction
Nasopharyngeal carcinoma (NPC) is a serious health problem in many parts of the world. Although rare in the United States and Western Europe, it is the leading form of cancer in men between the ages of 20 and 44 years in parts of Southeast Asia 1 and is prevalent in Indonesia, North Africa, the Middle East, and among Eskimo populations as well. 1 It also represents an important form of juvenile cancer, affecting a population between the ages of 15 and 25 years in North Africa, 2 and represents one of the most frequent epithelial tumors of children in geographically higher risk areas. 3
Current treatment protocols have resulted in good clinical outcomes if the disease is diagnosed and treated at an early stage.4,5 Unfortunately, metastatic NPC is refractory to treatment and carries an extremely poor prognosis. Particularly concerning is, that among head and neck cancers of epithelial origin, it is associated with the highest rate of metastasis. 6 This can be attributed in part to the clinically occult site of primary tumor development, the fossa of Rosenmüller, which often precipitates diagnostic delays that allow the cancer to become advanced before it is detected. This highlights the need for models that are amenable to the study of metastatic NPC and adequate to test new potential therapies.
Much of the information we have about NPC has been derived from studies of well-characterized cell lines
Results
Identification of the PI3K/Akt/mTOR pathway as a potential therapeutic target by comparing types II and III primary biopsy specimens
To delineate pathways relevant to NPC pathophysiology, we conducted gene array analysis of primary human tissue biopsy specimens (
A mouse model of invasive/metastatic NPC
To explore the role of the PI3K axis in NPC tumor development and metastatic progression, we developed an orthotopic xenograft mouse model of NPC. We tagged NPC lines C666-1 and HONE-1 with luciferase and injected them into the nasopharyngeal epithelium of severely immunodeficient NOD.Cg-
Summary of Observed Rates and Sites of Metastasis
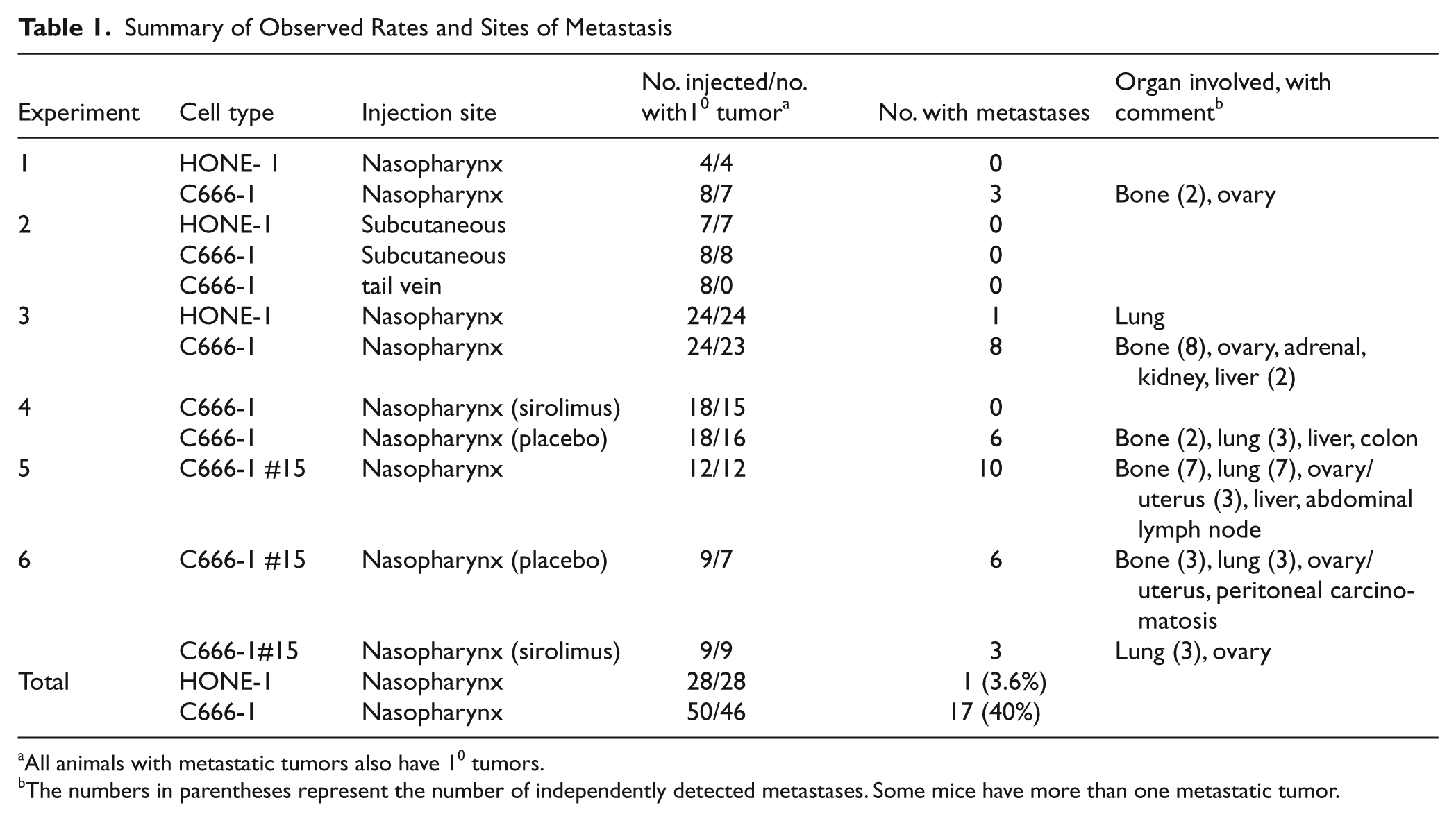
All animals with metastatic tumors also have 10 tumors.
The numbers in parentheses represent the number of independently detected metastases. Some mice have more than one metastatic tumor.
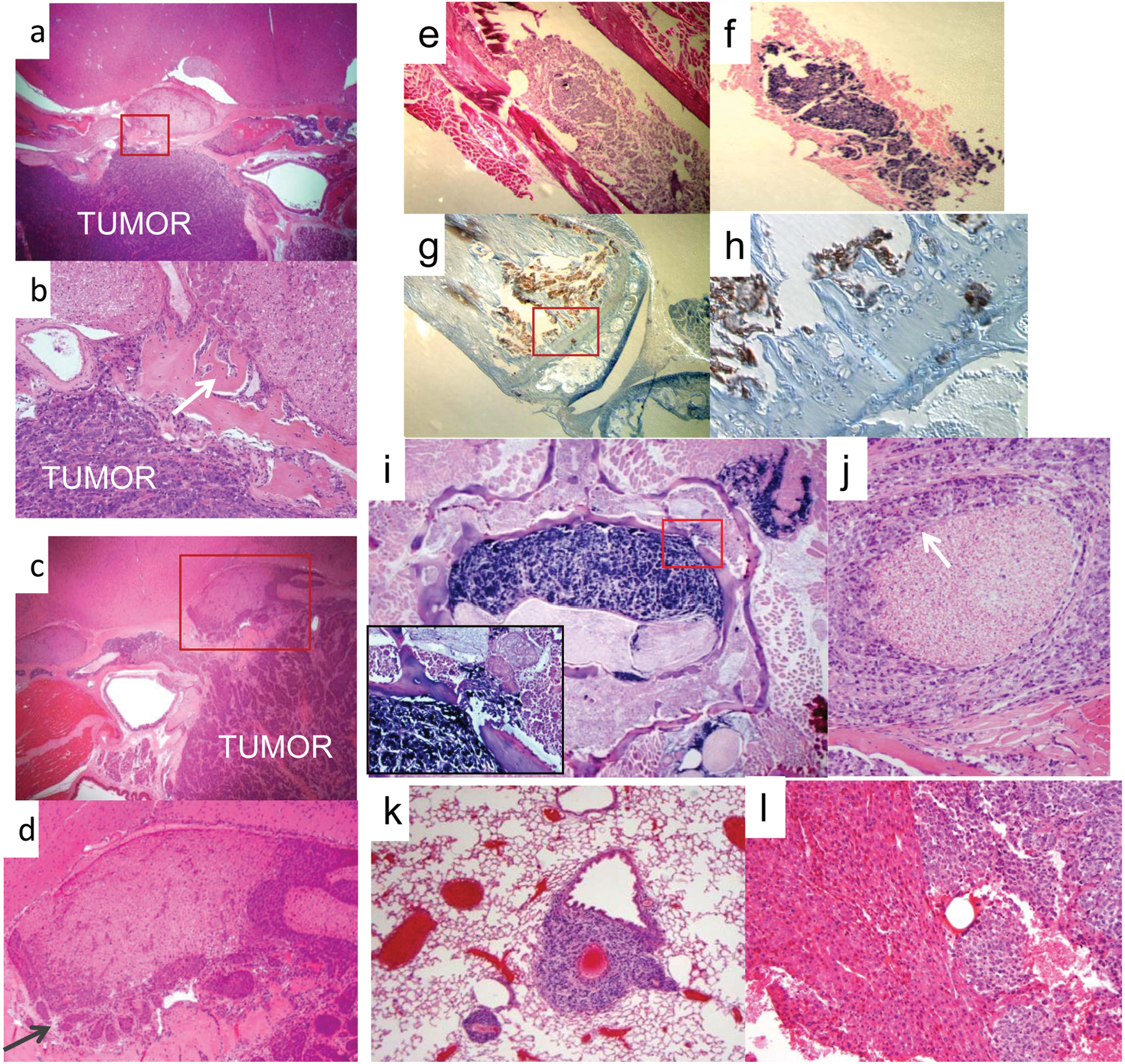
Histology of primary site and metastatic tumors. (
We also observed metastatic dissemination in mice injected with C666-1 cells in a distribution pattern remarkably similar to that seen in human NPC 15 (Table 1), with the majority occurring in the bone, followed by the lung, and the balance distributed in the abdomen including the liver, colon, kidney, ovary, peritoneum, and adrenal gland. Figure 1 shows examples of typical metastases. In Figure 1E to 1H, tumor cells are located in the bone marrow compartment and have also infiltrated into the bony matrix of the distal femur. Figure 1I and 1J demonstrate aggressive features of bony and perineural invasion in a spinal metastasis. Metastatic lesions were also frequently found in the lung and liver, examples of which are shown in Figure 1K and 1L.
To control for the possibility that metastases arise in the C666-1–injected mice by spillover into the bloodstream during tumor cell inoculation, we performed tail vein injections in 8 mice. We intentionally used a much larger number of cells than would be expected to enter the circulation by inadvertent intravascular injections from the nasopharyngeal site. The mice were imaged weekly over 12 weeks with no evidence of tumor growth, nor was any found on dissection (
Sirolimus disrupts metastasis
Having implicated differential regulation of the PI3K/AKT/mTOR axis in type III (undifferentiated) versus type II (differentiated) primary tumor biopsies, we hypothesized that this pathway directs the focally invasive and distant metastatic behavior of NPC
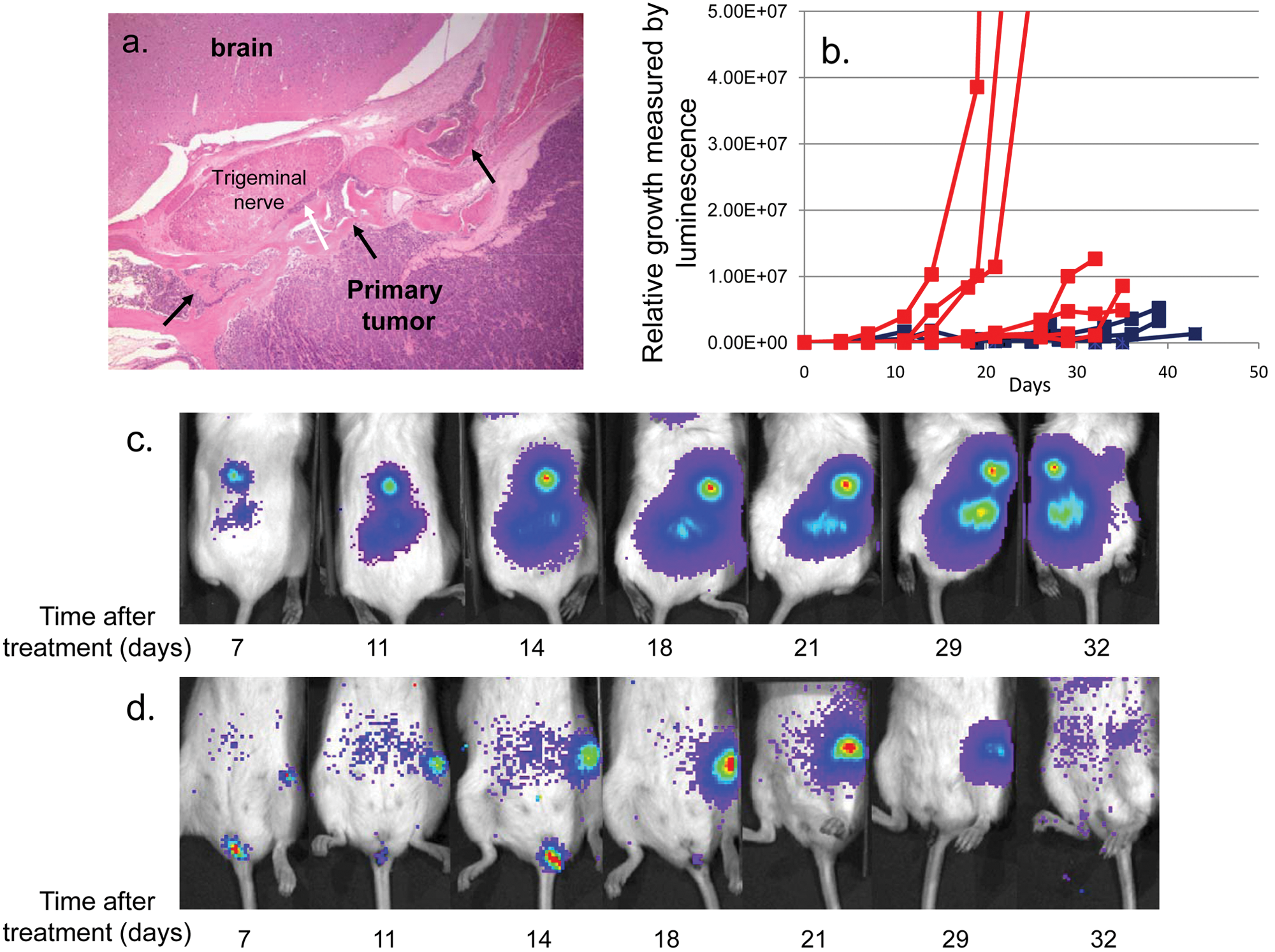
Sirolimus slows the growth of NPC metastasis. (
Strikingly, however, growth of the metastatic lesions in C666-1 tumor-bearing mice was strongly attenuated. Shown graphically (Fig. 2B), metastatic lesions in the placebo-treated mice grew exponentially, while those treated with sirolimus were markedly slowed or eradicated. Specific examples are shown in Figure 2C and 2D. The placebo-treated mouse demonstrated unchecked exponential growth of the spine metastasis (Fig. 2C), whereas the femoral metastasis of the mouse treated with sirolimus dramatically regressed (Fig. 2D). Thus, we have demonstrated that mTOR blockade affects metastatic but not primary-site NPC tumor growth in the xenograft model.
The finding of selective inhibition of metastatic tumor growth led us to speculate that an mTOR-mediated process directs tumor cell dissemination from the primary site and as such should be subject to modulation by sirolimus. To test this, 2 groups of mice were injected orthotopically with C666-1 cells (
Having shown that mTOR activity is necessary for the dissemination and metastatic growth of C666-1 tumors, we hypothesized that the metastatic C666-1 tumor cells have enhanced mTOR activity. To explore this, we removed metastatic C666-1 tumor cells from tumor-bearing mice and expanded them in culture for comparison to the parental line. Two explants are described: one from a bone metastasis, no. 15, and the other from a liver metastasis, no. 7. Both were maintained in culture in a manner identical to that of the parental C666-1 line. As shown in Figure 3A to 3D, Western blot analysis confirms higher baseline mTOR activity by 3 parameters: 1) enhanced phosphorylation of 4E-BP1(Ser65), an indicator of mTOR complex 1 activity 25 ; 2) enhanced phosphorylation of Akt(Ser473), an indicator of mTOR complex 2 activity 26 ; and 3) higher baseline phosphorylation of mTOR(Ser2448), which is phosphorylated by mTOR complex 1 substrate, 70S6K, 27 and participates in both mTOR 1 and 2 complexes. 28
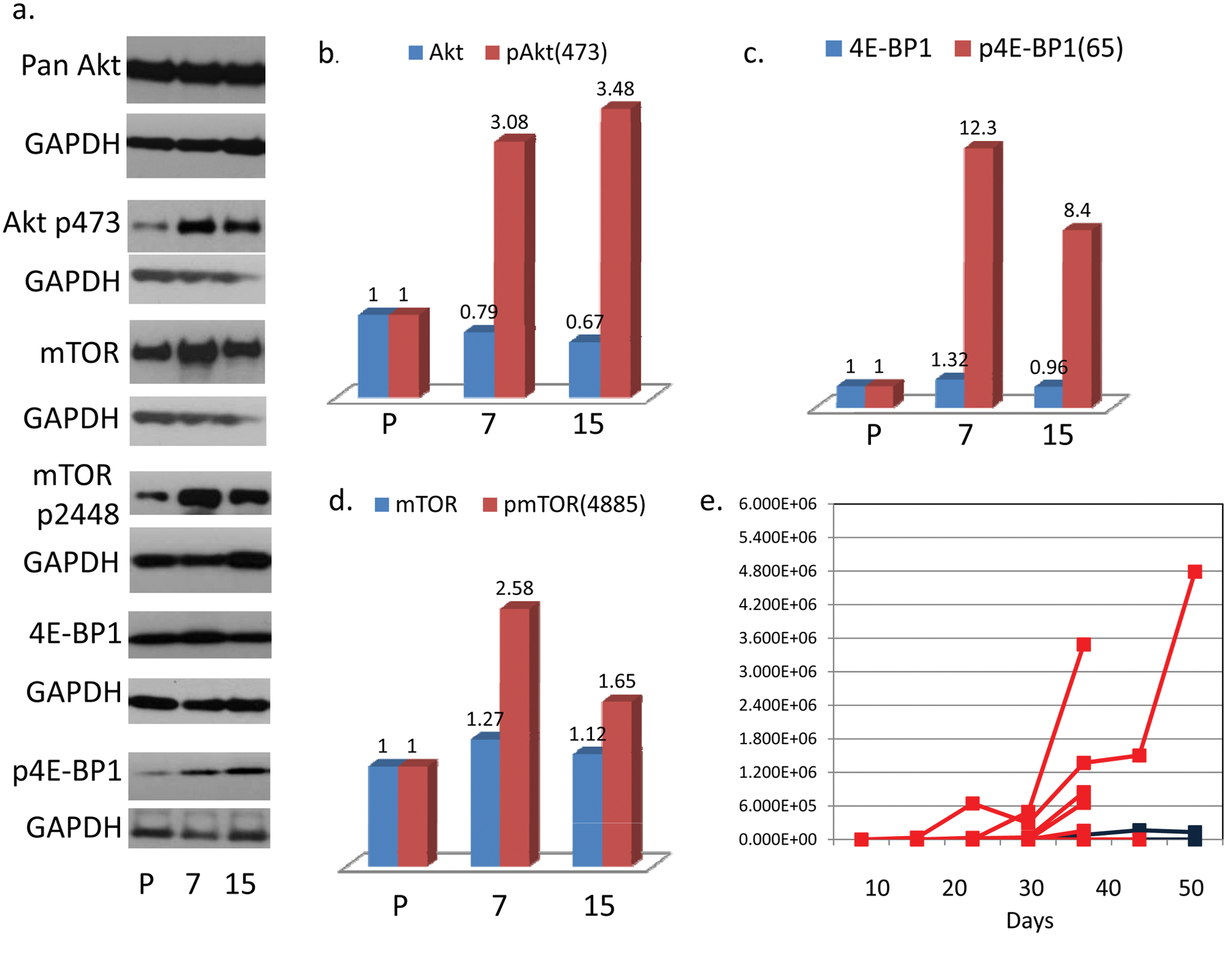
Metastatic explants have a higher baseline activation of mTOR based on phosphorylation levels of pAkt(473), pmTOR(4885), and p4E-BP1. Metastasis of explant no. 15 (from a bone lesion) is inhibited by sirolimus but demonstrates reduced sensitivity to sirolimus compared to the parental line. (
Because the metastatic tumor isolates maintain enhanced activation of mTOR in culture, we speculated that upon reinjection, they would metastasize more readily from the primary tumor site than the parental C666-1 counterpart. We chose subline no. 15 for further orthotopic xenograph experiments and found that 84% of xenograft recipients (compared to 40% of those injected with the parental;
From this observation, we predicted that the more metastatic subline, shown to have higher baseline mTOR activity, would be less sensitive than the parental line to inhibition of metastasis by sirolimus. Following orthotopic placement of the highly metastatic C666-1 explant, no. 15, the mice were treated with sirolimus or placebo starting the day after injection (Table 1, experiment 6). Of the sirolimus treated, 3 of 9 with primary tumors developed metastatic disease compared to 6 of 7 that were treated with placebo (
These studies suggest that differential activation of the mTOR pathway is instrumental in directing NPC tumor dissemination and that treatment with a targeted agent that blocks mTOR is effective for inhibiting NPC dissemination and metastatic growth.
Discussion
By developing a new orthotopic xenograft mouse model of nasopharyngeal carcinoma, we are able to recapitulate patterns of human NPC invasion and metastasis that are useful for the study of tumor behavior and the development of antitumor therapies. We validated the utility of this method by demonstrating specific inhibition of metastatic growth via a targeted approach against mTOR, a key component of the PI3K/Akt/mTOR signaling pathway that is strongly implicated in metastasis across several tumor types.21,22
Fundamental to this work is a detailed study of our orthotopic model to establish that it accurately recapitulates many aspects of advanced disseminated NPC. At the injection site, we saw a highly invasive tumor with skull base erosion, reactive bone formation, and invasion of cranial nerves. Although we do not persistently see cervical lymph node involvement, the model does replicate metastatic disease to multiple distant sites, including the bones, lungs, and liver, which are characteristics of advanced human disease 29 and are highly predictive of poor clinical outcome.30,31 Other reports describing xenograph models of NPC metastasis have not recapitulated the human disease as accurately.11,12 One study in particular described orthotopic injection of tumor cells into nude mice, 11 but we are unable to reproduce these results. In our hands, it was necessary to use highly immunodeficient NSG mice to see reproducible tumor growth and metastasis. The nude mouse study describes the use of a large gauge needle to inject cells directly through the hard palate, but in our experience, such injections caused massive tissue damage and disruption of surrounding tissues, including the base of the skull. Instead, we inject the cells into the soft tissue just distal and lateral to the hard palate with a small gauge needle, which allows us to obtain consistently reproducible focal tumor growth without collateral tissue damage. In the nude mouse study, metastasis was only observed in the brain, cervical lymph nodes, and lungs, a distribution that does not mimic the human disease. Specifically, metastasis to the brain is not a clinical characteristic of NPC, 32 was not detected in our model, and may be accounted for in the nude mouse model by focal trauma caused by passing the needle through the hard palate. Thus, we have developed an orthotopic xenograft model of invasive and metastatic NPC disease that closely mimics human disease well enough to assist in further identifying cellular characteristics that direct aggressively invasive and disseminated growth as well as identifying effective targeted therapies.
The data shown in this particular study indicate that the dissemination and growth of metastatic cells that arise from a primary C666-1 tumor are highly dependent on mTOR activity. Specifically, sirolimus not only blocks C666-1 tumor cell dissemination to distant organs but also selectively inhibits their growth after metastasis has occurred. We further demonstrated that this is a feature of enhanced mTOR activation by showing that cultured metastatic explants maintain a higher baseline level of mTOR activation, which, upon reinjection, renders the cells less sensitive to sirolimus and imparts a higher metastatic potential than the parental C666-1 counterpart. Interestingly, the enhanced mTOR activation of the metastatic explants found by Western blot analysis encompasses both mTOR complex 1 (TORC1) and complex 2 (TORC2) activity. Sirolimus is a rapalog compound that, as is characteristic of this class of drugs, is more active against TORC1 than TORC2. Its primary mechanism of action is understood to be dephosphorylation and consequent activation of eukaryotic translation initiation factor (eIF4E) binding proteins (4E-BP), which function to block eIF4E cap-dependent translation of mRNAs involved in cell growth and survival. 33 Indeed, the metastatic explants have a much higher baseline activation of 4E-BP1, consistent with its involvement in their metastatic behavior and our ability to affect this behavior by TORC1 inhibition. The role of enhanced TORC2 activity in the pathogenesis of C666-1 NPC tumor dissemination, however, is not specifically addressed by these studies. We speculate that more comprehensive mTOR inhibition may further diminish metastatic tumor development beyond what we observe with sirolimus. This can be readily investigated using the mouse model to study agents that inhibit both TORC1 and TORC2.
The results of our studies so far, however, indicate that targeting mTOR in NPC represents an approach of considerable clinical importance because there remains an urgent need for new therapeutic options for disseminated disease. 34 Current treatment protocols have resulted in very good clinical outcomes (currently 70% to 80% 3-year survival) if the cancer is diagnosed and treated while confined to locoregional areas.4,5,34 Metastatic NPC, however, is refractory to treatment and carries an extremely poor prognosis, with a median survival time of less than 12 months.5,35 Particularly concerning is, that among head and neck cancers of epithelial origin, NPC is associated with the highest rate of metastasis. 6 Additionally, approximately half of recurrences are solid organ metastases, 36 which usually arise without nasopharyngeal site recurrence, 14 suggestive of micrometastatic spread that arose prior to and was not ameliorated by treatment of the primary tumor. Thus, novel therapeutics such as mTOR inhibition, which specifically targets mechanisms that mediate dissemination, are essential to comprehensive NPC disease control. Moving forward, we can utilize our orthotopic model to refine mTOR inhibition strategies. As well, we can expand these strategies to include other components of the PI3K axis to develop clinically relevant interventions that will both prevent metastatic dissemination and effectively treat metastatic lesions.
Future studies should include commonly used chemotherapeutic agents for NPC, the prime example being cisplatin. Cisplatin resistance has been shown in a number of cancers, including NPC, to occur through up-regulation of Akt/mTOR,37-39 which we have shown here is linked to NPC metastasis. Furthermore, the EBV latent protein, LMP1, has been shown to confer cisplatin resistance to the C666-1 NPC cell line in association with increased Akt activation. 40 Thus, cisplatin treatment may promote the development of a metastatic phenotype by selecting for resistance related to dysregulated mTOR activity. Our data raise the possibility that targeting mTOR, while treating with cisplatin, may prevent or reduce the development of chemotherapeutically resistant metastasis.
As an outgrowth of these explorations, we have gained some interesting preliminary insights into the pathophysiological differences between invasive and metastatic NPC. Following identification of the PI3K axis as a potential target through microarray analysis of primary biopsy type II and III specimens, we then demonstrated that mTOR activity directs metastatic dissemination in our orthotopic experimental model. Surprisingly, mTOR blockade did not change the local invasive characteristics of either cell line in the xenograft, even though the Connectivity Map analysis of the primary biopsy data indicates a correlation between higher activation of the PI3K/Akt/mTOR pathway and the more invasive features of the type III phenotype. This observation is consistent, however, with clinical studies. For example, PI3K amplification with consequent up-regulation of Akt activity in primary NPC tumors has been shown to correlate with a poor prognosis resulting from higher rates of metastasis, irrespective of classification based on differentiation status. 41 Similarly, enhanced rates of distant NPC metastasis have been shown to correlate with increased activation of mTOR and not the differentiation status of the primary tumors. 42 Thus, we have more precisely defined a signaling pathway alteration that correlates with metastatic NPC behavior and confirmed that histological characteristics of focal invasion do not necessarily predict tumor dissemination to distant organs. Clarity regarding how the functions of focal cell invasion and distant metastatic dissemination are directed through the PI3K network will be gained through directed studies using drugs that specifically inhibit components of the pathway upstream of mTOR. 43 We anticipate that our orthotopic xenograft model, which clearly delineates these features of NPC tumor biology, will be ideal to conduct such studies.
Finally, we anticipate that our model will be useful to more directly interrogate the effects of EBV on invasive NPC pathogenesis. NPC tumors express a restricted pattern of EBV latent genes,
44
several of which have been suggested to play a role in NPC development, invasion, and metastasis.44-47 The orthotopic platform offers a valuable method by which we can more precisely delineate the effects of EBV transcription products on specific aspects of NPC tumor behavior
Materials and Methods
Primary biopsy analysis
Total RNA was isolated from 20 fresh nonkeratinizing nasopharyngeal carcinoma biopsy samples obtained from Taiwan. 48 Biopsy samples were obtained with punch forceps by use of endoscopic guidance. The tumor stage was designated according to the International Union Against Cancer and American Joint Committee on Cancer staging manuals (L.H. Sobin, 5th ed., 1997). NPCs were mostly of the advanced stage (IVA to IVC), and all samples were EBV positive as measured by EBER expression levels. 49
Biopsy material-chip hybridization
Biopsy RNA was isolated by guanidiniumthiocyanate extraction followed by centrifugation in CsCl, and RNA integrity was assessed by gel electrophoresis and optical density. We had sufficient RNA from 16 samples that also met the quality criteria for hybridization to Affymetrix microarray chips (HG-U95A, Santa Clara, CA). All scans were performed on Affymetrix scanners, and the expression value for each gene was calculated using Affymetrix GENECHIP software. A total of 14 samples survived the normalization and scaling criteria. For a detailed protocol and data set, see Pegtel
Computational analysis
The microarray data were normalized as previously described 50 ; details can be found at http://www.broad.mit.edu. We applied conventional marker selection techniques to this data set to identify and rank genes that were up- or down-regulated in type II versus type III NPC, based on fold change. We then used this gene profile to interrogate the Connectivity Map. The Connectivity Map 18 (www.broadinstitute.org/cmap) is a database of gene expression profiles from cultured human cells treated with a large number of bioactive small molecules, many with multiple independent replicates, together with a pattern-matching tool that rank orders each treatment “instance” by its similarity to the query “signature” of interest (in our case, type III v. type II NPC gene array expression patterns).
Cell lines and transfectants
Cell lines were cultured with 10% heat-inactivated fetal bovine serum in either RPMI1640 (C666-1) or DMEM (HONE-1) at 37°C in 5% CO2. C666-1 cells 51 were obtained from Dr. Fei-Fei Liu (Ontario Cancer Institute, Toronto, Canada) and HONE-1 52 from Dr. Ron Glaser, who originated the line (Ohio State University, Columbus, OH). Both lines retain expression of human cytokeratins, and C666-1 harbors the Epstein-Barr virus. Luciferase was expressed in the cells from the pGL4.51 (luc2.CMV/Neo) vector (Promega, Fitchburg, WI) introduced by electroporation with the BTX High Throughput Electroporator (model HT-200, BTX). Alternatively, cells were infected with the pGreenFire1-CMV, TR011VA-1 lentivirus (System Biosciences, Mountain View, CA).
Tumor injection and imaging
The highly immunodeficient mouse strain NOD.Cg-Prkdcscid Il2rgtm1Wjl/SzJ (NSG) was used in all experiments. Mice were maintained and treated according to institutional guidelines of the Tufts University Division of Laboratory Animal Medicine, and the experimental techniques described herein were approved by the Tufts University Institutional Animal Care and Use Committee (protocol #56-08). For nasopharyngeal injections, mice were subject to deep anesthesia with ketamine/xylazine (100 mg/kg and 10 mg/kg) and placed in a prone position with the mouth held open with curved forceps and the tongue pulled aside. Cells (2.5 × 105 cells in 30 uL of PBS) were injected using a 25-gauge needle just distal and lateral to the hard palate. For subcutaneous growth, 5 × 105 tumor cells in 100 uL of PBS were injected into the subcutis of the right flank. For tail vein inoculation, 2 × 105 cells were injected in a total volume of 300 uL of PBS. Tumor appearance was detected and/or growth measured, every 4 to 7 days, by IP administration of luciferin (1 mg in 100 uL of PBS). The mice were immobilized with isoflurane anesthesia and imaged in a Xenogen IVIS 200 Biophotonic Imager, Xenogen. Final numerical representations of tumor size were calculated by determining photon emission/second of a given tumor within a radius encompassing 5% or greater of maximal signal intensity.
Sirolimus administration
Sirolimus (10 mg/mL) (LKT Laboratories, St. Paul, MN) stored at −20°C in 100% ethanol was diluted into PBS containing 5% PEG300 and 5% Tween80, for a final dose of 5 mg/kg in 100 uL. Placebo-treated mice received PBS/PEG300/Tween80 only. Drug was administered IP for 5 of every 7 days.
Histology
Histological analysis was performed by the Clinical Histology laboratory or the Department of Laboratory Animal Medicine at Tufts Medical Center using a Ventata Benchmark XT automated system, Ventana. Tumor tissue and metastases were excised from sacrificed mice, employing luciferase imaging to identify the tissue of interest. For cytokeratin staining, the AE1/AE3/PCK26 anti-pancytokeratin antibody was used (Ventana), and for EBER
Western blot analyses
Western blotting was performed using primary antibodies against Akt (pan) (C67E7, rabbit mAb #4691S, Cell Signaling Technology, Danvers, MA), phospho-Akt (Ser473) (rabbit mAb #4060S, Cell Signaling Technology), mTOR (7C10, rabbit mAb #2983S, Cell Signaling Technology), phospho-mTOR (Ser2448) (rabbit mAb #2971S, Cell Signaling Technology), 4E-BP1 (53H11) (rabbit mAb #9644S, Cell Signaling Technology), phospho-4E-BP1 (Thr37/46) (236B4, rabbit mAb #2855S, Cell Signaling Technology), and GAPDH (14C10, #2118S, Cell Signaling Technology). The secondary antibody was HRP-linked anti-rabbit IgG (#7074, Cell Signaling Technology), and the blots were developed using Super Signal* West Femto Chemiluminescent Substrate (#34095, Thermo Scientific, Waltham, MA).
Footnotes
Acknowledgements
The authors thank Jennifer Bradford for technical assistance with developing the nasopharyngeal tumor injection technique, Justin Lamb for introducing us to the Connectivity Map, Evan Smith for assistance in developing Western blotting protocols, and Lauren Richey for interpretation of pathology slides.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received the following financial support for the research, authorship, and/or publication of this article: This work was supported by Public Health Service grants R01 CA65883, R01 AI18757, and R01 AI062989 to D.A.T.-L.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
