Abstract
Two or more drugs that individually produce overtly similar effects will sometimes display greatly enhanced effects when given in combination. When the combined effect is greater than that predicted by their individual potencies, the combination is said to be synergistic. A synergistic interaction allows the use of lower doses of the combination constituents, a situation that may reduce adverse reactions. Drug combinations are quite common in the treatment of cancers, infections, pain, and many other diseases and situations. The determination of synergism is a quantitative pursuit that involves a rigorous demonstration that the combination effect is greater than that which is expected from the individual drug’s potencies. The basis of that demonstration is the concept of dose equivalence, which is discussed here and applied to an experimental design and data analysis known as isobolographic analysis. That method, and a related method of analysis that also uses dose equivalence, are presented in this brief review, which provides the mathematical basis for assessing synergy and an optimization strategy for determining the dose combination.
Keywords
Introduction
Drugs that produce overtly similar effects are often given in combination. These include drugs for treating many conditions such as hypertension, pain relief, and cough suppression. Certainly almost all cancer chemotherapy and many antimicrobial regimens use multiple drugs given simultaneously. There is also a growing interest in drug combinations that might prevent cancer, as summarized in a recent
Some drug effects are desirable whereas others are undesirable. The adverse effects of chemotherapeutic drugs are well documented, and some of these such as neuropathic pain present a real challenge that has been somewhat addressed in some of the author’s collaborative preclinical studies.
2
The ideal situation is one in which the drug combination synergizes the desired effect but exhibits subadditivity for the undesirable effects. A quantitative approach to that ideal situation is discussed in a later section of this communication. Much experience has shown that synergism (or subadditivity) is not merely a property of the 2 drugs. It also depends on the doses of each in the combination.3-7 In this discussion we present the most common quantitative methods that guide the experimental design and the data analysis that are used in quantitative drug combination studies. These constitute a generalization and expansion of the method of isoboles. This method is a nonmechanistic approach, that is, a procedure that uses the dose-effect data of the individual drugs. The potency and efficacy information derived from the individual dose-effect curves allow a determination of the expected combination effect or dose combination that produces a specified effect. This expected effect (termed
Drug Combination Analysis: Isoboles
The most common method for the quantitative assessment of unusual interaction between agonist drugs is the method of isoboles. This is a graphical procedure, introduced and developed by Loewe,8-10 that uses the dose-effect relation of each drug (alone) in order to derive the set of dose combinations that are expected to give a specified effect level. Most often the selected effect level is 50% of the maximum effect, and the doses of each full agonist drug that individually give this effect are therefore their ED50 doses. In its simplest form this procedure uses the ED50 doses of the individual drugs and uses these as intercept values on a Cartesian coordinate system in which doses are represented on the x- and y-axes. The straight line connecting these intercepts represents the set of points (dose pairs) that give the specified effect (50% of Emax) when there is no interaction between the drugs. This line, called an

where
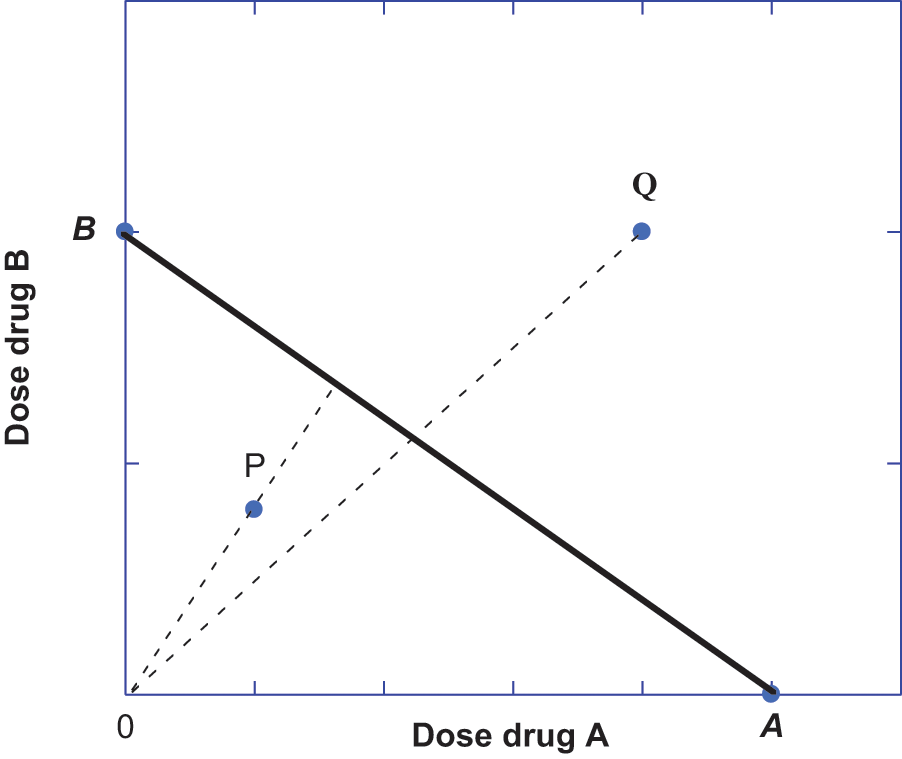
Dose pairs (
Dose Equivalence
The analysis leading to the isobole is derived from dose-effect data of each compound using some well-defined effect metric such as cell antiproliferation or induction of apoptosis as the effect. The dose-effect relations of the individual drugs are first determined, and, in many cases, it is found or assumed that these have a constant potency ratio, which means that at any effect level, the equally effective doses (
The linear isobole is easy to construct and was described in detail by Loewe, 9 but that discussion did not include an explicit mathematical derivation. Also, a rather cumbersome notation used by Loewe seems to have prevented a clear understanding of its basis and that fact may account for its initial limited application. The method of isoboles ultimately gained popularity when Gessner and Cabana 12 used it to assess toxic and hypnotic interactions between chloral hydrate and ethanol. A further review by Gessner 13 amplified details on the use of the isobologram. A number of other investigators applied this method: Adams et al 14 studied combinations of opioid mu and delta agonists in a rat analgesic assay; Fairbanks and Wilcox 15 examined antinociceptive combinations of morphine and clonidine in tolerant mice; Hammond et al 16 studied antinociceptive combinations of delta 1 and delta 2 opioid receptors in mice; Kimmel et al 17 tested combinations of buprenorphine and cocaine on locomotion in the rat. Antinociceptive combinations of morphine and clonidine were also tested,18,19 and Wilcox et al 20 examined this same combination using motor and sensory responses in the rat. Porreca et al 21 studied antinociceptive combinations of combinations of morphine and[Leu5]-enkephalin, and Raffa et al 22 examined combinations of the enantiomers of tramadol. More recent studies include work by Woolverton et al,23,24 in which various combinations including cocaine and remifentanil were examined in protocols involving self-administration in monkeys. Tanda et al 25 examined combinations of cocaine and other dopamine uptake inhibitors in mice.
The works cited above represent only a partial list of studies in which the linear isobole was used to distinguish between additive and nonadditive drug interactions. In all of these studies the potency ratio was found to be constant or could reasonably be assumed to be constant so that the simple linear isobole of additivity applied in the assessment of superadditive and subadditive combinations. A special case is that in which 1 of the 2 drugs lacks efficacy in producing the effect. In that case the linear isobole is a horizontal line whose intercept is the ED50 of the active agent. This situation was found in our study in which glucosamine, which lacks antinociceptive efficacy, was paired with ibuprofen in the mouse abdominal constriction test. 26 This study demonstrated synergism, and that finding formed the basis of a U.S. patent for this drug combination.
Curved Isoboles
In this section is a summary of our work showing that the isobole of additivity is not necessarily linear
27
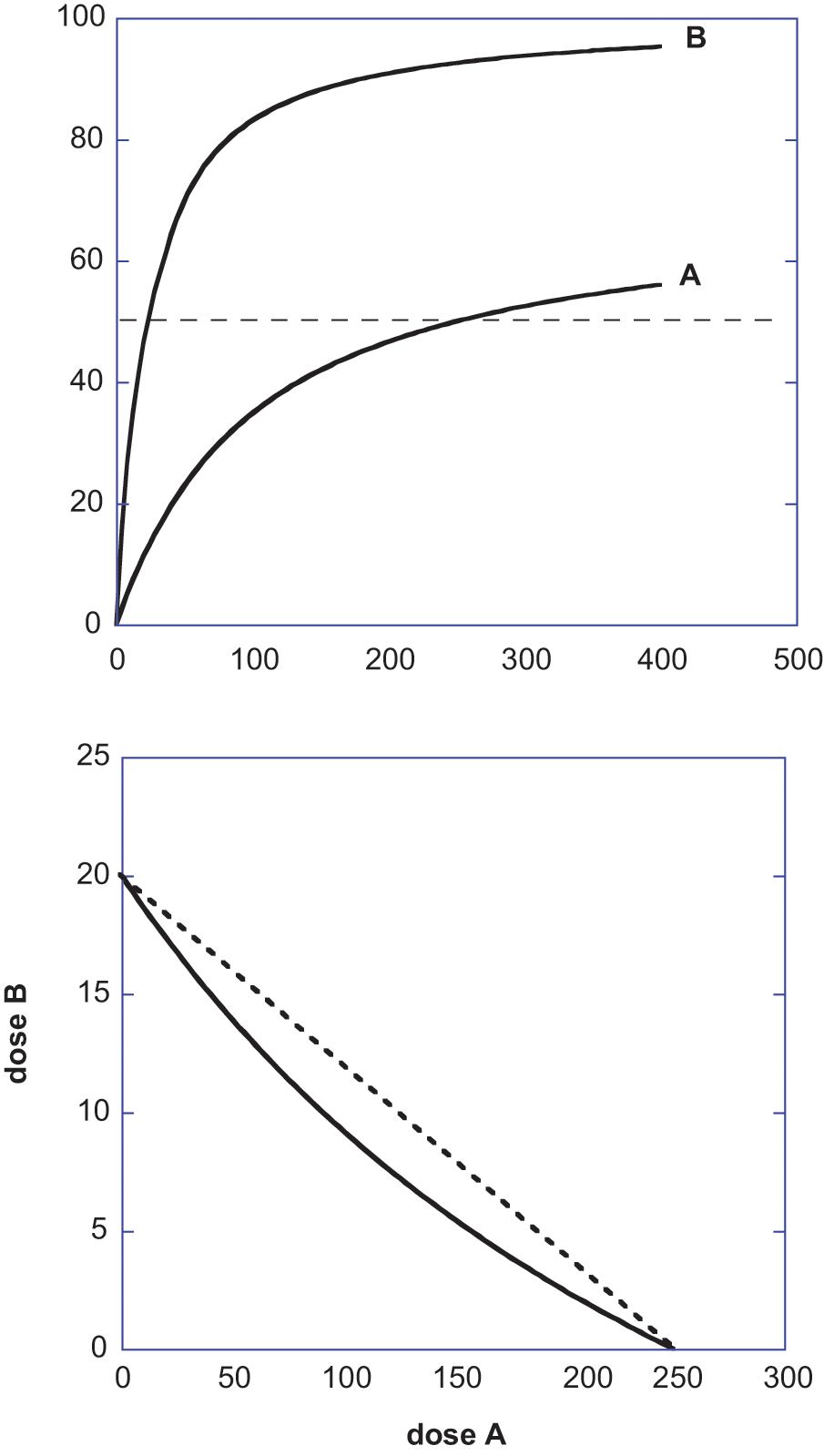
and, therefore, that the use of Equation 1 as the basis for defining nonadditive drug interactions is sometimes incorrect. Toward this end we consider 2 agonist drugs whose maximum effects differ such as those shown as Drug A and Drug B in Fig. 2. To derive the isobole for this case we proceed as before by using the common dose-effect model equation with maximum effects denoted
(
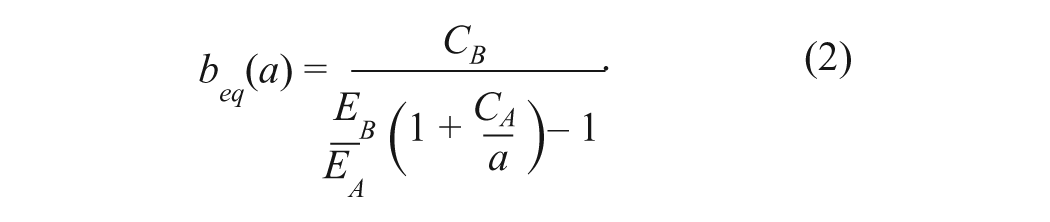
When beq(
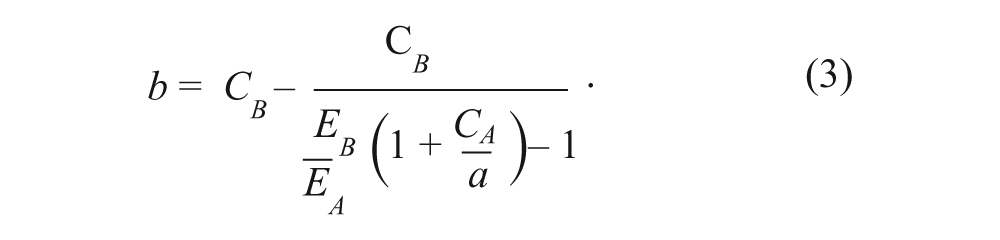
It is seen that Equation 3 is nonlinear. This form, derived by Grabovsky and Tallarida,
27
is even further generalized in that publication to apply to parent dose-effect equations that contain Hill coefficients (exponents
A View from the Effect Scale
The isobole provides the view of the additive dose combinations (points on the line) that give a specific effect level such as 50% of the maximum and, thus, this curve becomes the basis for detecting departures from additivity such as synergism or subadditivity. An alternate approach views the dose combination on the effect scale. This approach, however, does not mean the direct addition of effects. (For example, if Dose a gives a 70% Emax effect and Dose b gives 75% Emax effect, the addition of these percentage effects is without meaning.) A visual that illustrates the correct way of determining combination effects is illustrated below. In this example, explained in detail below, we show how 2 agonists with different efficacies and potencies yield the expected (additive) effect when the drugs are simultaneously administered. The dose-effect parameters of each agonist are shown in the legend of Figure 3, and these values allow calculation of the Drug B–equivalent of each Dose
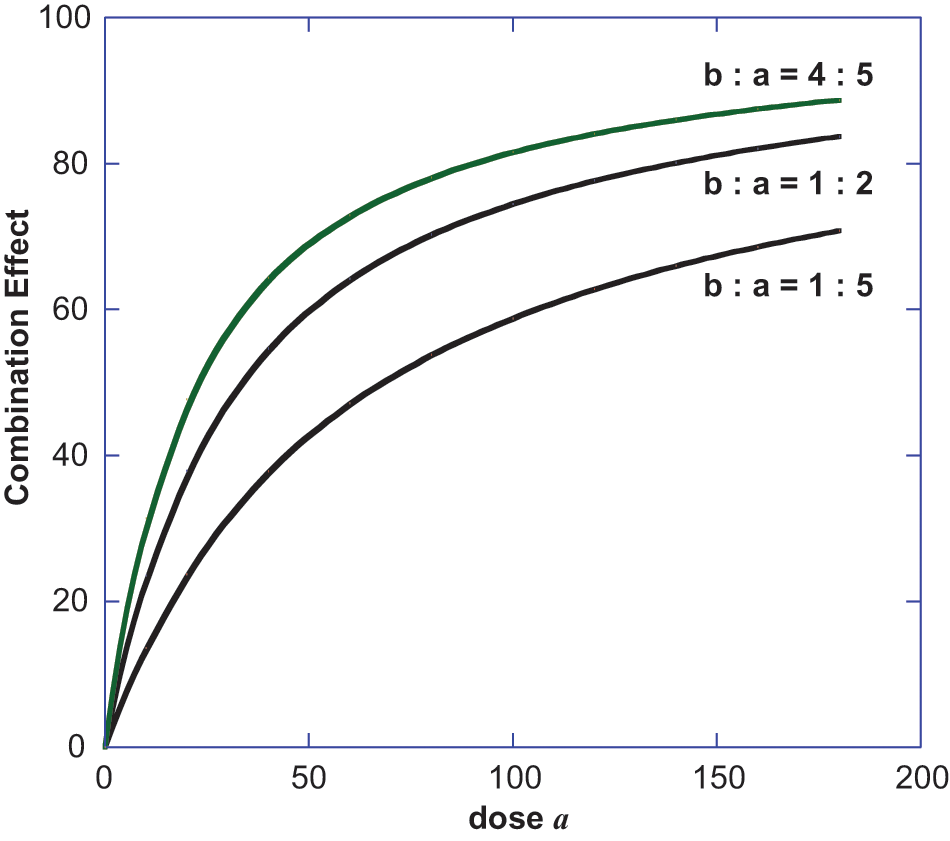
Illustration of how a fixed dose ratio combination of 2 agonist drugs are used in a calculation of the expected (additive effect) of a combination dose. The graphs are shown for 3 different fixed ratio dose combinations and are expressed and plotted in terms of the dose
To demonstrate how these curves were constructed, we illustrate with a value from the lower curve of Figure 3 where the ratio of Dose
Optimizing the Drug Combination Dose Ratio
It is well known that synergism (or subadditivity) for a drug pair is not only dependent on the agonist drug pair; it also depends on the ratio of the doses. A good example is afforded by our analgesic studies using the combination tramadol and acetaminophen, where it was found that several dose ratios were synergistic, whereas others were simply additive. 28 This concept is useful in the development of an optimization strategy. By this we mean finding the range of dose ratios that are synergistic for the desired effect but subadditive for the toxic effects. In this regard the isobologram provides a useful view of the strategy and this is illustrated in Fig. 4. A fixed ratio dose combination is defined by each radial line. In other words the isobole plot, whose axes are doses of the respective drugs, shows the dose combination as the radial line for the particular dose ratio being tested. The intersection of that radial line with the isobole defines the additive dose combination and thereby allows a view of either superadditive (synergistic) or subadditive for the point (dose combination) that is observed to give the specified effect. As previously mentioned, a point below indicates synergism whereas a point above indicates subadditivity. Most often there is a range of dose combinations that are synergistic and other ranges that are either additive or subadditive. Using the range that is synergistic we obtain a sector on the isobologram. That sector (pie-shaped region) therefore gives all dose ratios that are synergistic in producing the effect. If a similar procedure is applied to an adverse effect, we can identify the dose combination range (sector in the plot) that is subadditive. The optimal choice of dose ratio is therefore the intersection of these 2 sectors. That intersection shows synergy for the desired effect and subadditivity for the adverse effect. This view is illustrated in Fig 4. In that illustration it is assumed that that synergism for the desired effect has been found for dose ratios contained within radial lines (C) and (D), whereas the toxic effect is subadditive within the dose ratios indicated between radials (A) and (B). The intersection of these sectors is therefore a set of dose ratios that maximizes the desired effect and minimizes the toxic effect.
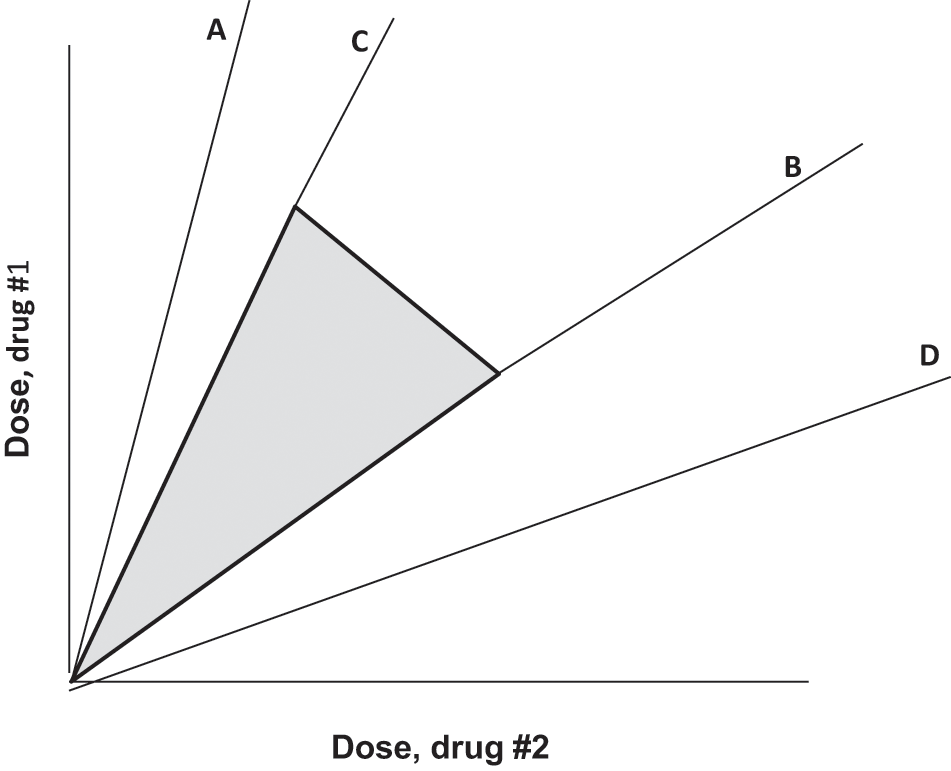
Illustration in which synergy for the desired effect is found to exist for dose combinations in the region between radial lines C and D, whereas the toxic effect is subadditive only between radial lines A and B. The intersection is the region between C and B, and therefore dose combinations in this intersection, with practical upper limits, are a region (shown shaded) that is optimal for selecting dose ratios.
Summary and Discussion
The assessment of drug combinations for determining synergy is a quantitative pursuit. This determination requires a quantitative approach that begins with the individual dose-effect curves from which the combined additive effect is calculated. If the combined effect observed is significantly greater than the expected (additive) effect, there is synergism. This analysis follows from the individual drug’s dose-effect relations and the concept of dose equivalence. Additivity is most often viewed as an isobole, which is a plot of individual doses that give a specified effect. Thus, additivity may be viewed on the dose scale as an isobole or on the effect scale. When synergism is detected, it is almost always dependent on the dose ratio of the combination that is tested and, thus, this information allows a determination of dose combinations that are optimal. Other approaches are available for exploring synergistic interactions. One that has been extensively quoted is based on a mechanistic model arising from mass action, Michaelis-Menten kinetics, Henderson-Hasselbalch equation, and the Hill equation. The interested reader is referred to this more theoretical approach 29 for examining multiple drug-effect systems.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Supported in part by 2P30 DAO13429, NIH/NIDA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
