Abstract
It has been previously shown that loss of heterozygosity (LOH) at the cytosolic glutathione peroxidase (GPx-1) locus is a common event in the development of several cancer types, including colorectal cancer. GPx-1 is an antioxidant selenium-containing protein, and polymorphisms within this gene have been shown to be associated with the increased risk of cancer. In order to assess whether this genetic change was an early or late event in colon cancer development, we investigated whether LOH at this site was occurring in colorectal adenomas, a premalignant lesion. Twenty-four pairs of DNA samples, obtained from both whole-blood and adenoma tissue from the same individuals, were genotyped at 2 positions in the GPx-1 gene: a codon 198 variation resulting in either a leucine or proline at the corresponding position in the peptide, or a variable number of alanine repeat codons corresponding to the amino terminus of the GPx-1 protein. No evidence of GPx-1 LOH was observed in the examined sample sets. These data indicate that the genetic loss at the GPx-1 locus may be a late event in colon carcinogenesis.
Keywords
Introduction
Colon cancer is a common malignancy, representing approximately 10% of all new cancer cases and cancer-related deaths in the United States. 1 As described in 1988 by Vogelstein et al., discernable morphological stages of colon cancer progression can be associated with the accumulation of genetic alterations that include both the activation of dominantly acting oncogenes and the loss of protective tumor suppressor genes. 2 The coupling of genetic alterations to discrete steps in tumor progression presents the potential to learn more about the biology of carcinogenesis and to possibly develop improved agents to both prevent colon cancer and slow its development.
Glutathione peroxidase (GPx-1) is a selenium-containing antioxidant protein that detoxifies hydrogen peroxide to water, using reducing equivalents from glutathione. Overexpression or underexpression of GPx-1 in vitro results in corresponding changes in the protection against DNA damage.3,4 A role for GPx-1 in human cancer etiology is supported by genetic studies, indicating likely roles for this protein in both cancer risk and progression. GPx-1 is polymorphic at 2 positions, a proline/leucine variation at codon 198 and a variable number of alanine repeats at the amino terminus of the protein. These variations have been shown to be functional and have been associated with the risk of several cancer types. 5 GPx-1 is located at position 3p21; loss of heterozygosity (LOH) at the GPx-1 locus was originally shown to occur at high frequency in lung tumors and lung tumor–derived cell lines. 6 These studies were expanded upon by showing that GPx-1 LOH also occurred at high frequency in cancers of the breast, colon, and kidney as well as those of the head and neck.7-10 In the case of head and neck cancer, GPx-1 LOH occurred both in the tumor DNA and the DNA obtained from histopathologically normal tissue adjacent to the tumor in 2 of 3 sample pairs examined. 8 These results indicate that GPx-1 LOH might be an early event in tumor progression, occurring in the field of cells from which the tumor evolved.
LOH at the GPx-1 locus in colon cancer was previously investigated using laser capture microdissection to obtain both tumor and histopathologically normal adjacent tissue from the same individuals. 9 In this study, 26 of 53 sample sets exhibited germline heterozygosity for either the codon 198 polymorphism or the alanine repeat variation, and of these informative sets, 15 showed GPx-1 LOH. The LOH frequency observed was similar to that seen in several other tumor types. Here, we report the analysis of samples representing colonic adenomas, a benign tumor thought to be the precursor to colorectal adenocarcinoma, for GPx-1 LOH.
Results
In a previous study analyzing samples obtained from patients in Chicago, Illinois, it was shown that there was a high percentage of LOH (>40%) at the GPx-1 locus in DNA obtained from adenocarcinomas of the colon. 9 In order to investigate whether LOH at that chromosomal location occurred in colonic polyps, a total of 24 pairs of DNA samples collected from resected adenoma tissue and the corresponding whole blood obtained from patients in Tucson, Arizona, were analyzed by PCR amplification followed by direct sequence analysis to determine the codon 198 polymorphism and the number of encoded alanine repeats. All of the participants were male, aged 50 to 84 years, and their characteristics are summarized in Table 1.
Patient Characteristics

Three adenomas were obtained from a single patient, 2 located in the proximal colon and 1 in the distal colon.
Recurrence of distal cancer.
As seen in Table 2, the germline genotypes for 10 individuals were heterozygous for both the codon 198 and alanine repeat variations, while another 6 were heterozygous for only the alanine repeats, and thus, samples from 16 individuals were informative for the determination of LOH. Genotyping the DNA from the corresponding adenoma tissue of all 16 of these individuals failed to yield a single case of GPx-1 LOH, as would have been indicated by heterozygosity in blood DNA and apparent homozygosity in the DNA obtained from the tissue. These data include one set for which there were 3 separate polyps obtained from a single individual (#13), none of which exhibited LOH.
GPx-1 and MnSOD Genotypes
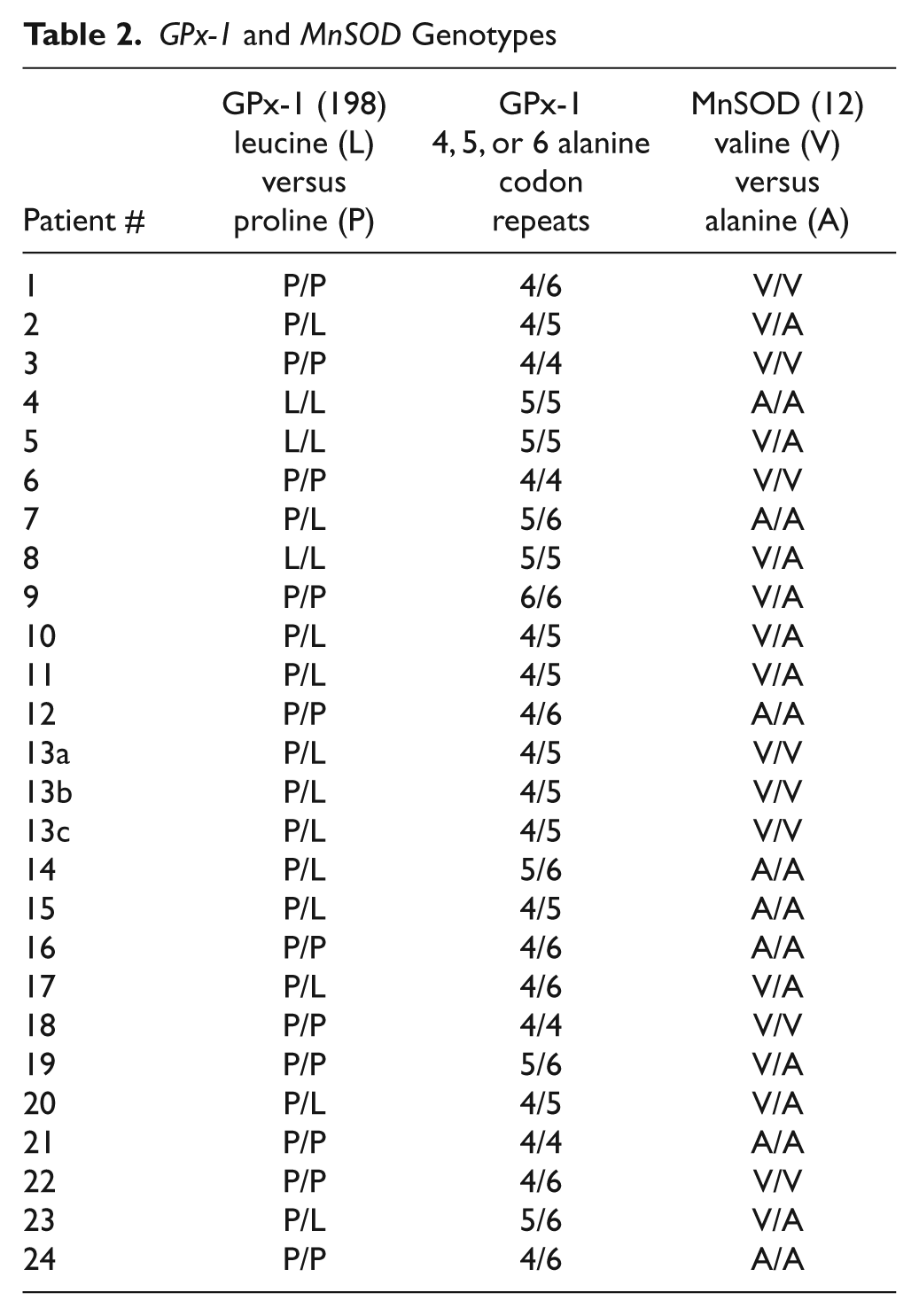
MnSOD is a major protective mitochondrial enzyme that detoxifies superoxide radicals produced during electron transport to the less toxic hydrogen peroxide. A variant MnSOD allele containing an alanine (A) rather than a valine (V) at codon 16 has been described and has been associated with an elevated cancer risk in human epidemiological studies.11-14 Li et al. reported an impressive 10-fold swing in the risk of aggressive prostate cancer among men who expressed the AA genotype between the lowest quartile of total antioxidant consumption and the highest, with those consuming the lowest levels of dietary antioxidants being at greatest risk. 12 When individual dietary components were considered, there was an approximately 5-fold difference in the risk of aggressive prostate cancer in AA men between the lowest quartile selenium consumption and the highest, with those consuming the lowest levels of selenium being at greatest risk. 12 Cox et al. reported a significant risk for breast cancer associated with MnSOD genotype among participants of the Nurse’s Health Study, but only when considered in association with GPx-1 genotype. 15 Further support for the interaction between MnSOD and GPx-1 in determining cancer risk came from human data, indicating that polymorphisms in the gene for the selenium transport protein selenoprotein P (SEPP1) that result in less SEPP1 in the plasma and reduced levels of GPx-1 are associated with a significant risk of prostate cancer only in men also expressing the ala16 MnSOD allele. 16 We therefore genotyped the DNAs obtained from the above-described population for MnSOD, and these results are also presented in Table 2. Eleven of the 24 patients were heterozygous for this polymorphism, and no examples of LOH were observed among these. It is noted that the distribution of MnSOD genotypes among individuals with adenomas (AA:AV:VV) was 9:9:6, which may be different than the 1:2:1 ratio reported among 764 men who were noncancer controls from the Physician’s Health Study. 12 The significance of this observation is uncertain, given the small sample size.
Discussion
The study described above was initiated to investigate whether LOH at the GPx-1 locus, an event shown to occur frequently in colon cancer as well as cancers of other types, occurred early in the development of colon cancer. Previously, we described LOH for GPx-1 at a frequency of 42% when DNA from tumor and adjacent histopathologically normal tissue was analyzed at both the codon 198 and alanine repeat variations. 9 In contrast, no evidence of LOH was observed when DNA obtained from adenomas, considered a colon cancer precursor, was evaluated. Chromosomal alterations occur in all discernable stages of colon cancer development, including adenomas,2,17-19 even being reported in what is likely to be one of the earliest preneoplastic lesions of this disease, the aberrant crypt focus. 20 Consistent with the notion of GPx-1 LOH being a late event in colon carcinogenesis, GPx-1 alleles that are likely to have reduced enzyme activity 22 and have been associated with increased risk of several cancer types 5 have not been reported to be associated with increased risk of colorectal adenoma.22,23 The situation may be less clear, as Hansen et al. reported that although GPx-1 polymorphisms were unrelated to colorectal cancer risk, stratified analyses showed that smoking and higher alcohol intake were associated with increased risks of colorectal cancer, but only among individuals who carried the at-risk variant Leu/Leu genotype. 24 Alternatively, our inability to detect GPx-1 LOH in adenoma samples may be due, at least in part, to the relatively small sample size or differences in the study population from which tumor and blood were obtained in our previous work.
In summary, the inability to detect LOH at the GPx-1 locus in adenoma tissue, while a common occurrence in colon cancers, may indicate that this is a genetic event associated with tumor cell survival or progression as opposed to factors that determine overall risk. Additional studies will be required to delineate the role that reduced levels of this antioxidant selenoprotein play in colon cancer development.
Materials and Methods
Colon samples
The study was approved by the Institutional Review Board of the University of Arizona, and all subjects provided written informed consent. Most patients at the Southern Arizona Veterans Affairs Health Care System (SAVAHCS), Tucson, Arizona, from 2000 to 2005 were asked to donate their tissues to a tissue bank and had known cancers, but many also had large polyps. Tissue samples were collected in the operating room at the time of resection of colorectal tumors. A whole-blood sample was drawn from each participant and immediately placed on ice. Upon resection, approximately 0.5- to 1-cm tissue biopsies were obtained from the adenoma, along with normal mucosa 1 cm and 10 cm adjacent to the tumor. Specimens were immediately placed on ice and transferred into liquid nitrogen.
DNA isolation
Frozen tissue samples were homogenized and DNA extracted following the QIAamp DNA mini kit protocol (Qiagen, Valencia, CA). DNA was isolated from whole-blood samples following the automated EZ1 DNA blood protocol (Qiagen). DNA was quantified by PicoGreen (Invitrogen, Carlsbad, CA).
Genotyping
Genotyping at the GPx-1 and MnSOD loci was accomplished by amplification of the desired region by PCR and sequence analysis across the polymorphic region. To determine the number of alanine repeat codons in the 5′ region of the GPx-1, amplification with forward primer ALAREPF (5′-CCTGCACTGCCGGTAACAT-3′) and reverse primer ALAREPR (5′-GCGCCGAGAAGGC ATACA-3′) resulted in a 509-bp amplicon that was sequenced using the sequencing primer ALAREPSEQ2 (5′-GCACTCTCCAGCCTTTTCC-3′). To determine the identity of the GPx-1 codon 198 polymorphism, resulting in either a leucine or proline at that position, amplification was achieved with forward primer SNPPROLEUF (5′-CGC CACCGCGCTTATG-3′) and reverse primer SNPPROLEUR (5′-GAAAACCCCCCCGAGAC-3′), resulting in a 256-bp fragment, and sequencing with the sequencing primer PRO- LEUSEQ (5′-GACCCCAAGCTCATCACCTG-3′). To determine the identity of the MnSOD codon 16 polymorphism, resulting in either an alanine or valine at that position, amplification was achieved with forward primer RESOD2F (5′-GTAGCACCAGCACTAGCAGCAT-3′) and reverse primer RESOD2R (5′-GCGTTGATGTGAGGTTC CAG-3′), resulting in a 438-bp fragment, and sequencing with the sequencing primer RESOD2SEQ (5′-CGCGGC GCTGACTGA-3′). The anticipated sizes of PCR products were verified by electrophoresis in a 1% agarose gel and bands visualized with ethidium bromide, prior to sequencing by the DNA Service Facility at the University of Illinois at Chicago. Single-nucleotide heterozygosity was apparent as 2 peaks at the same position on the chromatogram that corresponded to the polymorphism of interest. The trinucleotide variation corresponding to the different numbers of alanines at the amino terminus of the GPx-1 protein was readily apparent as the emergence of predicable overlapping patterns of peaks on the sequencing chromatogram at the expected position. In all cases, overlapping peaks differed by no more than 75% peak height, although the possibility that LOH could have been missed due to there being a relatively small number of tumor cells that had undergone LOH could not be completely discounted. All sets of PCR reactions included a negative control that omitted a DNA template and a positive control of a DNA template of a previously determined genotype.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This work was supported the
