Abstract
In this perspectives article, we review scientific literature regarding de novo formation of vascular networks within tissues undergoing a significant degree of motion. Next, we contrast dynamic pattern formation in embryos to the vascularization of relatively static tissues, such as the retina. We argue that formation of primary polygonal vascular networks is an emergent process, which is regulated by biophysical mechanisms. Dynamic empirical data, derived from quail embryos, show that vascular beds readily form within a moving extracellular matrix (ECM) microenvironment—which we analogize to the de novo vascularization of small rapidly growing tumors. Our perspective is that the biophysical rules, which govern cell motion during vasculogenesis, may hold important clues to understanding how the first vessels form in certain malignancies.
Morphological Complexity in Amniotes
As a first approximation, it is reasonable to assume that morphological complexity in the animal kingdom would correlate with genetic complexity. However, this assumption is untenable based on comparative genomic studies. It is now clear that the genomes of primitive animals, which do not form organs, “include many of the genes responsible for guiding development of other animals’ complex shapes and organs.” 1 Thus, developmentally simple animals possess similar molecular genetic circuits that amniotes use to make complex body parts; indeed, in some cases, the size of complex animal genomes is surprisingly similar to that of primitive animal genomes. This unexpected realization prompts the obvious question: if genomic complexity is not the underpinning of vertebrate organogenesis, what is? This question can be rephrased as follows: why do the embryos of warm-blooded animals employ a markedly different morphogenetic strategy compared to more ancient animals? A teleological answer is because the evolution of an amniotic cavity and maternal care allowed new selective strategies to be realized. For example, frog and fish have larval intermediate forms that are free-living and that develop quickly. In contrast, amniote embryos enjoy a safe, food-rich environment (amniotic cavity) in which to grow and develop. Moreover, even after hatching or parturition, the parent/mother provides food and protection. The point being that the amniote embryo/fetus is not on a morphogenetic fast track and does not have to fend for itself—leaving time to employ unique strategies for the construction of complex tissues and organs.
Our view is that amniote tissues and organs emerge from processes that require more time and rely on overt biophysical and biomechanical processes. The most striking example of an emergent biophysical process that distinguishes amniotes and nonamniotes during early embryogenesis may be heart morphogenesis. In amniotes, the midline tubular heart results from huge (i.e., tissue-level) physical displacements of mechanically distinct bilateral heart fields—by forces unknown (Aleksandrova et al., manuscript under review). Perhaps the most curious characteristic of amniote heart morphogenesis is that the process is secondary to the formation of a foregut. Simple naked-eye inspection demonstrates that formation and caudal regression of the chicken foregut (anterior intestinal portal) require millimeter-sized tissue deformations in an embryo that is, itself, millimeter sized. Thus, deformations required to make the future esophagus are the underpinnings of tissue folding and vast mechanical displacements resulting in a midline heart tube. There is no comparable morphogenetic counterpart for such tissue-level deformations during the formation of nonamniote frog or fish hearts.
Another good example of biophysical inputs to organ formation is amniote vasculogenesis—a process that requires formation of a pattern within a constantly deforming and rapidly expanding tissue mass. Conventional explanations of vasculogenesis often assume that endothelial cells migrate to predetermined positions following extracellular guidance cues or chemoattractants.2-4 We argue that the networks forming during vasculogenesis are self-organized in the sense that we do not expect a gene expression prepattern or other external cues within the microenvironment to specify the position of individual segments within the primary vascular network. This can be contrasted with the angiogenesis process within the retina, another widely studied system for blood vessel assembly. In the retina, the intricate structure of glial cell processes and the associated extracellular matrix (ECM) were shown to guide endothelial cells and organize the vasculature into a characteristic pattern.5,6 Thus, in the retina—a largely static environment compared to the early embryo—the patterning process is likely to be dominated by a prepattern, extrinsic to the endothelial cells. Clearly, there are poorly understood deterministic aspects of vasculogenesis as well, such as the determination of major vessels in the later stages of the developing vascular network. In fish, the major vessels assemble directly, without forming an intermediate vascular plexus, and specific vascular malformations are correlated with genetic defects. 7
In recent years, researchers have employed computational time-lapse imaging to study the forces that shape the embryo. In particular, vascular morphogenesis occurs at times and places in avian embryos that are readily observed and manipulated in vivo. 8 Due to recent improvements in digital microscopy, it is now possible to address directly how new blood vessels form de novo.9,10 This technology allows the visualization of both cellular displacements and tissue motion patterns during early embryogenesis.11-13 In particular, motion patterns between disparate tissues and their respective extracellular matrix scaffolds can be compared. These data can be further analyzed using quantitative engineering and statistical approaches, which allow computation of tissue displacements, ECM fiber motion, and total cellular displacements. For example, using ECM filaments as passive markers of the micro- environment, the difference between ECM displacements and total cellular displacements is a measure of active cellular motility. The result is a more precise representation of active cell locomotion during developmental processes.
In this article, we describe vascularization within a highly dynamic tissue milieu and discuss various proposed patterning mechanisms. Such comparisons highlight a critical need for understanding purported cellular guidance mechanisms and chemotactic gradients said to drive vascular morphogenesis and pattern formation (e.g., gradients of bioactive vascular endothelial growth factor [VEGF] in situ).
Vasculogenesis in Avian Embryos
Vasculogenesis is the formation of a primary vascular pattern from mesodermal-derived precursors (angioblasts). The optically transparent amniote embryo, which can be cultured and imaged ex ovo, is an excellent model in which to study the dynamics of a forming vascular system in a warm-blooded vertebrate. Amniote vasculogenesis begins at the same time as axis elongation and neurulation, both complex tissue-level events that entail huge physical deformations, unlike the more stable platform found in the embryos of cold-blooded animals. Vasculogenic processes are also relevant in certain pathophysiologies. Recent data provided evidence that vascular endothelial cell progenitors exist in the adult and may become blood borne, enter extravascular tissues, and promote de novo vessel formation.14,15 For that reason, endothelial progenitors, mobilized in situ or transplanted, are a major target of therapeutic vascularization approaches to prevent ischemic disease and control endothelial injury. Moreover, endothelial progenitors represent potential targets for strategies to block tumor growth and requisite components for construction of engineered tissues. 16
In bird embryos, well before the onset of circulation, hundreds of essentially identical vascular endothelial cells create a polygonal network within a relatively uncomplicated, sheet-like anatomical environment. 18 Each link in the polygonal network is a cord consisting of 3 to 10 endothelial cells. 19 Endothelial cells differentiate from solitary primordial cells within the avian mesoderm (area pellucida).20,21 Committed angioblasts display a random spatial distribution within the mesoderm, without an observable preexisting pattern at length scales comparable to that of the future primary vascular polygons. 19 Whole-mount embryo immunolabeling at vasculogenic stages for VEGF shows a widespread distribution that does not exhibit a discernible pattern (Figure 1). Thus, based on available static imagery, there is no evidence for a prepattern guiding the formation of the primary vascular plexus.
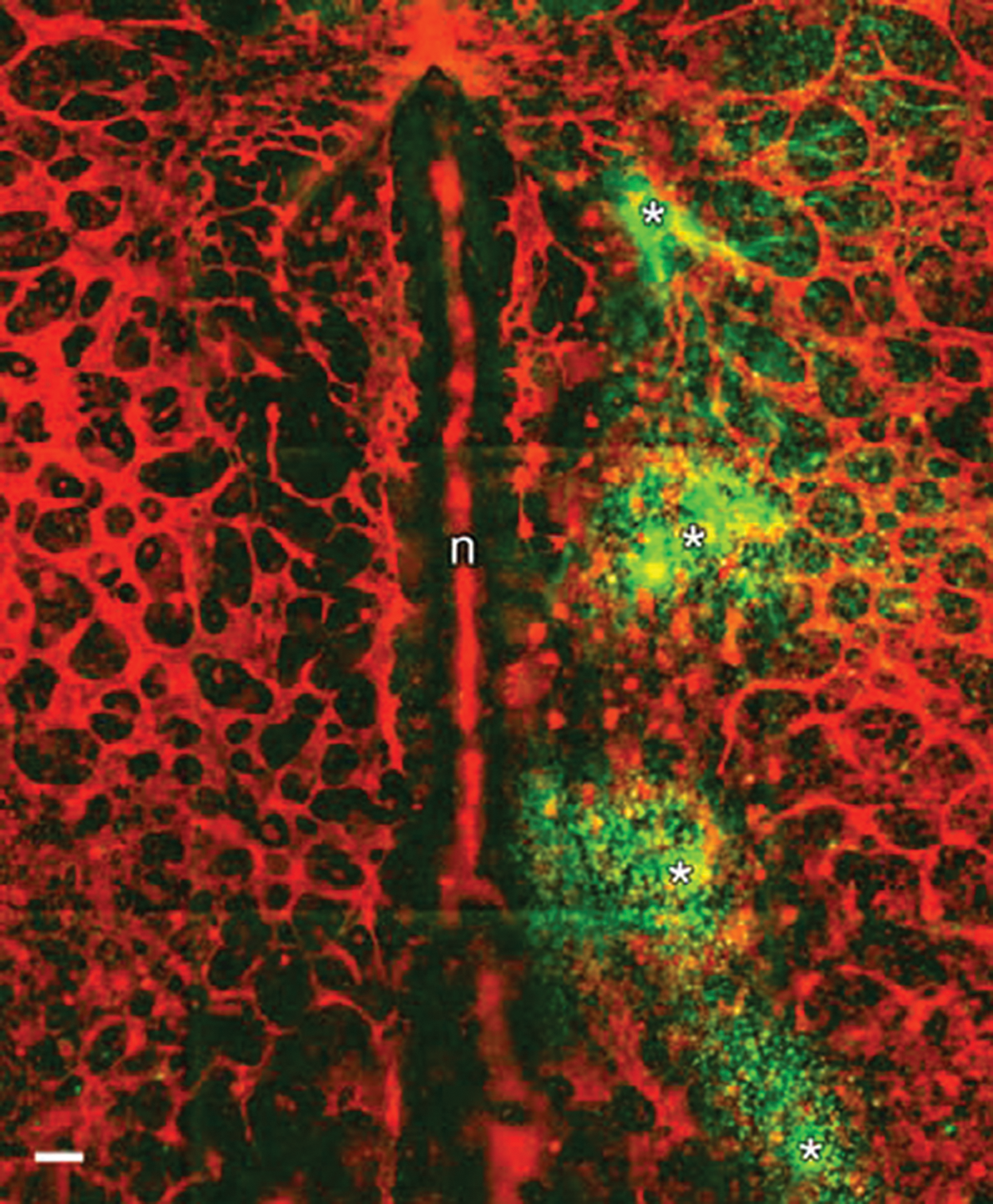
Tagging of endogenous vascular endothelial growth factor (VEGF) with fluorescent probes. Live quail embryos (Hamburger Hamilton [HH] 7) were injected, at 4 positions (asterisks), with 20-nL boluses of recombinant human VEGFR2 coupled to human IgG Fc (rVEGFR2), approximately 1 ng/nL. After 45 minutes, the embryo was fixed, made permeable, and incubated with secondary antibodies to human Fc (green) and QH1 antibodies directly conjugated with Alexa 555 (red). The red signal thus depicts quail endothelial cells. The image shows that upon injection, the rVEGFR2 binds to dense punctate foci of ligand near the injection sites. The diffusion of the rVEGFR2 is spatially limited by the high concentration of reactive VEGF ligand. No network-like pattern is discernible when viewing the green signal. Note that endothelial cells near the injection sites are not configured in a normal polygonal-like pattern when compared to the contralateral side. That rVEGFR2 can perturb vascular patterns, in vivo, is a well-established observation. 17 This is direct evidence that in a live amniote embryo, VEGF, which is physically accessible, is not distributed in a polygonal pattern, nor is VEGF distributed in a manner that suggests a higher order chemotactic prepattern. n = notochord; scale bar: 100 µm.
Endothelial Cell Movements
Endothelial cells can be tracked by use of microinjected and fluorochrome-conjugated QH1 antibodies, specific for quail vascular endothelium,22,23 or using the recently developed transgenic quail line in which endothelial nuclei contain yellow fluorescent protein (YFP). 24 In the Tg(tie1:H2B-eYFP) quail, a fusion between histone H2B and enhanced YFP is driven by the promoter of the TIE1 gene. Thus, transgenic embryos express H2B-eYFP in the nuclei of endothelial and endocardial precursor cells. TIE1+ nuclei are already detectable at the earliest stages of vasculogenesis, approximately at Hamburger Hamilton 25 (HH) stage 7 (5 somites)—the same stage when QH1 appears. 24
To record the vasculogenic process, we use a wide-field scanning optical microscopy approach. 9 At each time point, images are taken in multiple optical modes: a fluorescence channel to visualize endothelial cells and another for visualizing the ECM environment using an immunolabeled ECM component. A third image, bright-field or differential interference contrast (DIC), is also acquired to provide an anatomical frame of reference. The resulting images are aligned such that certain anatomical reference points (e.g., the intersomitic clefts) remain stationary. Furthermore, due to the availability of images from multiple focal planes, no object is lost or rendered out of focus.
Active versus Passive Movement of Endothelial Structures
As Figure 2 and Supplementary Video 1 demonstrate, at stage HH7, there is a substantial medial movement of the forming vascular plexus, a phenomenon predicted by Coffin and Poole. 26 This process, vascular drift, was shown to occur across the entire nascent vasculature. 23 Early vasculogenesis, however, occurs during a time of vigorous rearrangements in the embryo, with movements occurring at various length scales that range from the migration of individual cells to global morphogenic events, such as gastrulation, neurulation, and formation of the foregut. Tissue movements, which can be quantified using ECM fibrils as passive tracers12,13,27 or using the “optical flow” established from DIC images (Figure 3 and Supplementary Video 2), profoundly influence vascular pattern formation. In fact, the drift motion of vascular segments is largely coincident with the morphogenetic tissue movements,24,27 as would be required if vascular structures are embedded in a mechanical continuum.
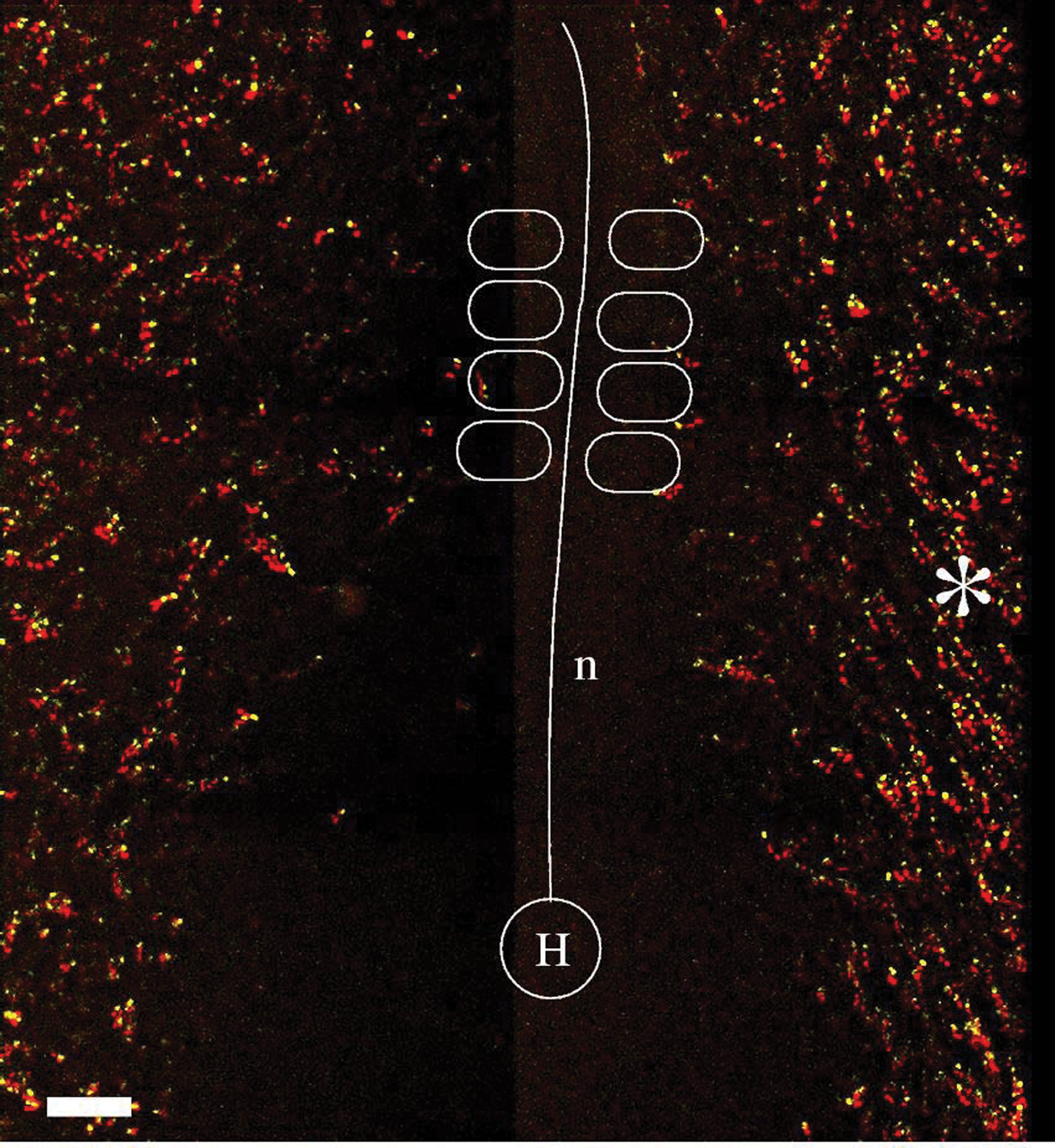
Endothelial cell movements at the onset of vasculogenesis in a Hamburger Hamilton (HH) stage 7 (5 somite) transgenic quail embryo. Motion of TIE1+ nuclei is shown in a somite-attached reference system by projecting 4 consecutive frames, the first 3 in red and the most recent time point in yellow. The asterisk marks an area where mediocranial motion is especially apparent. H = Hensen’s node; n = notochord. Scale bar: 100 µm.
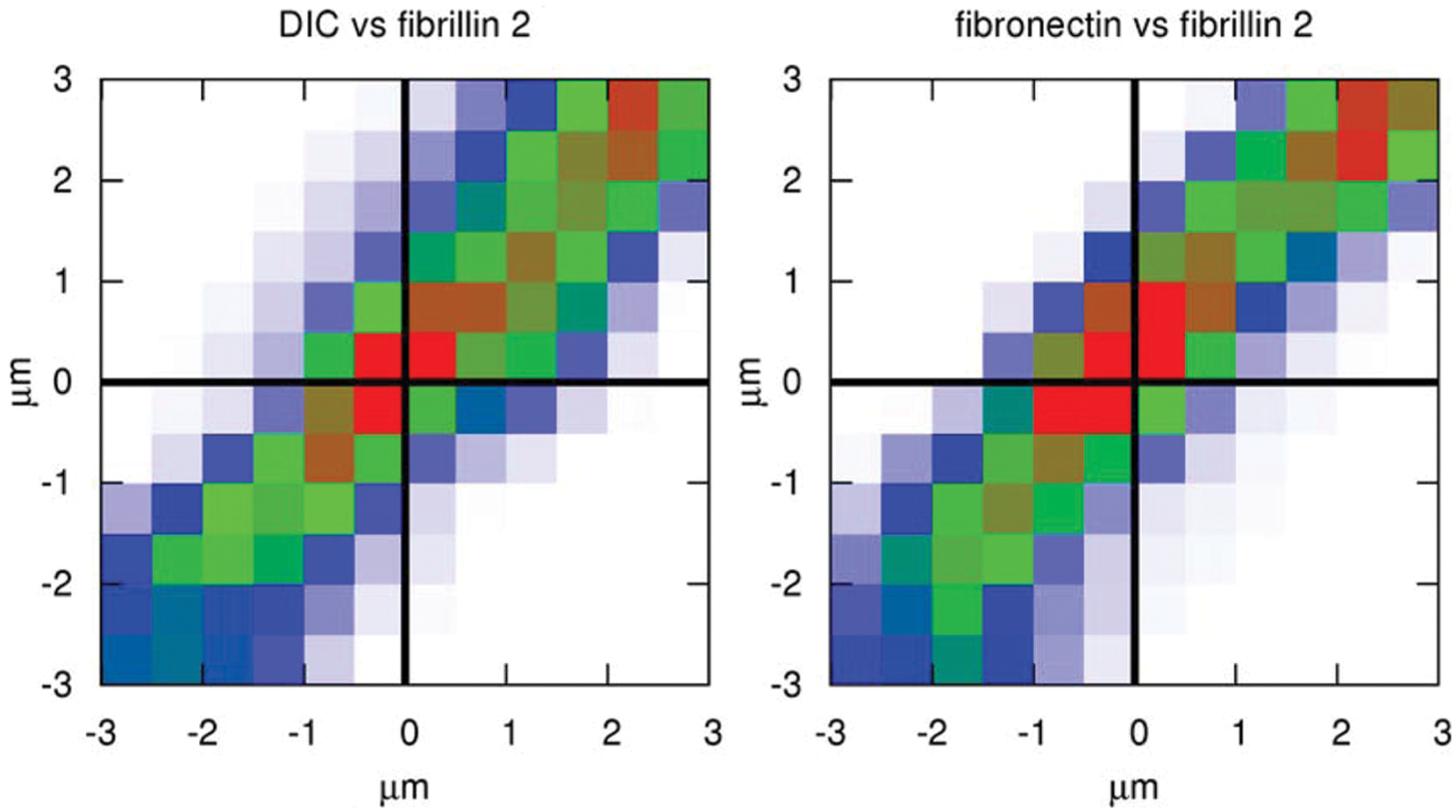
Tissue displacement vector components can be estimated from various microscopy modes, including differential interference contrast (DIC) and extracellular matrix (ECM) immunofluorescence. Embryonic development was recorded using multiple optical modes: DIC and two epifluorescent channels, visualizing fibronectin and fibrillin-2, two distinct ECM components. Particle image velocimetry analysis was performed on all 3 image sequences, yielding 3 estimates for tissue movement—one from each imaging mode. The correlation plot compares corresponding components of tissue displacement vectors derived either from DIC and fibrillin-2 immunofluorescence (A) or fibronectin and fibrillin-2 immunofluorescence (B). Blue, green, and red indicate progressively higher data densities in the correlation plot. High densities along the diagonal of the correlation plots indicate that analysis of all 3 optical modes yields consistent estimates.
To remove tissue movements computationally from image sequences, we use a two-stage approach. First, tissue displacements are predicted using particle image velocimetry (PIV) analysis. 11 Then, using the resulting sequence of displacement maps for each image, the pixels are moved against the displacement field in a compensatory manner (see Supplementary Video 1). This procedure results in an image sequence that depicts changes as if tissue movements did not occur. The remaining movements (i.e., the difference between the total cell displacements and the tissue drift) are autonomous cell movements. As Figure 4 demonstrates, the active endothelial cell movements at the onset of vasculogenesis are largely random despite the apparent, medial-directed vascular invasion seen in Figure 2. Thus, although primordial endothelial cells are actively motile, the majority of their gross medial displacement at these stages is a consequence of tissue flow.
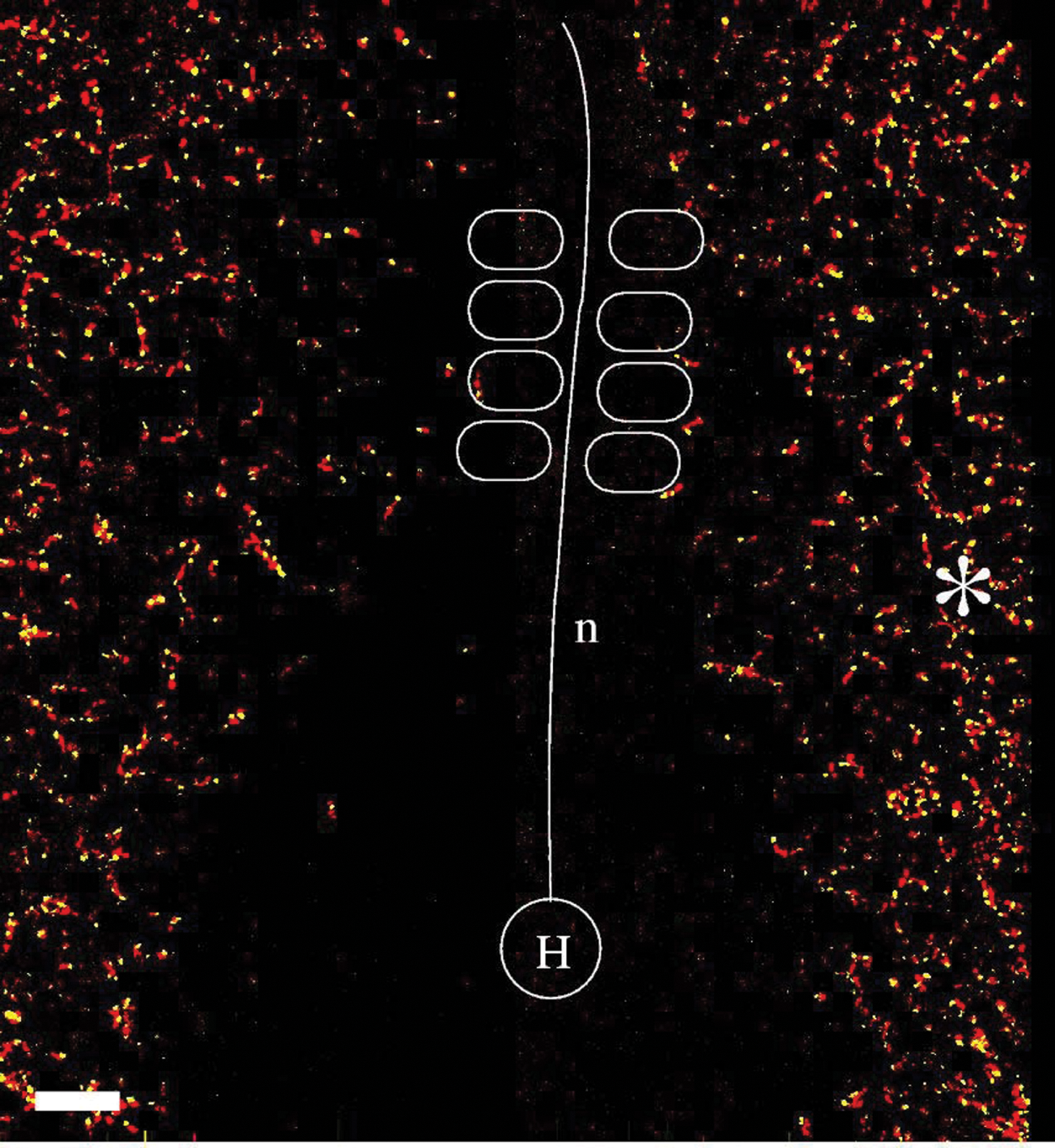
Active movements of endothelial cells. Data presented in Figure 2 were digitally transformed to remove the effect of tissue movements. The difference between the apparent cell motion and local tissue motion is the active cell movement, which is more random. Marked locations correspond to those in Figure 2.
Multicellular Sprouts and Motility along Vessel Segments
By stage HH8 (7 somites), TIE1+ cells assemble into a primary network lateral to the somites. Most endothelial cells, however, continue to exhibit vigorous autonomous motility. The intensity of the autonomous motion can be used to pseudo-color the images, as seen in Figure 5. These images reveal that important sites of active endothelial cell movements are the expanding vascular branches, or sprouts. The early vascular plexus is characterized by disconnected endothelial clusters. To establish a network, endothelial cells next send out extensions across the avascular ECM. This type of protrusive behavior is reminiscent of angiogenic sprouting, a process thought to be a characteristic of later vascular development.
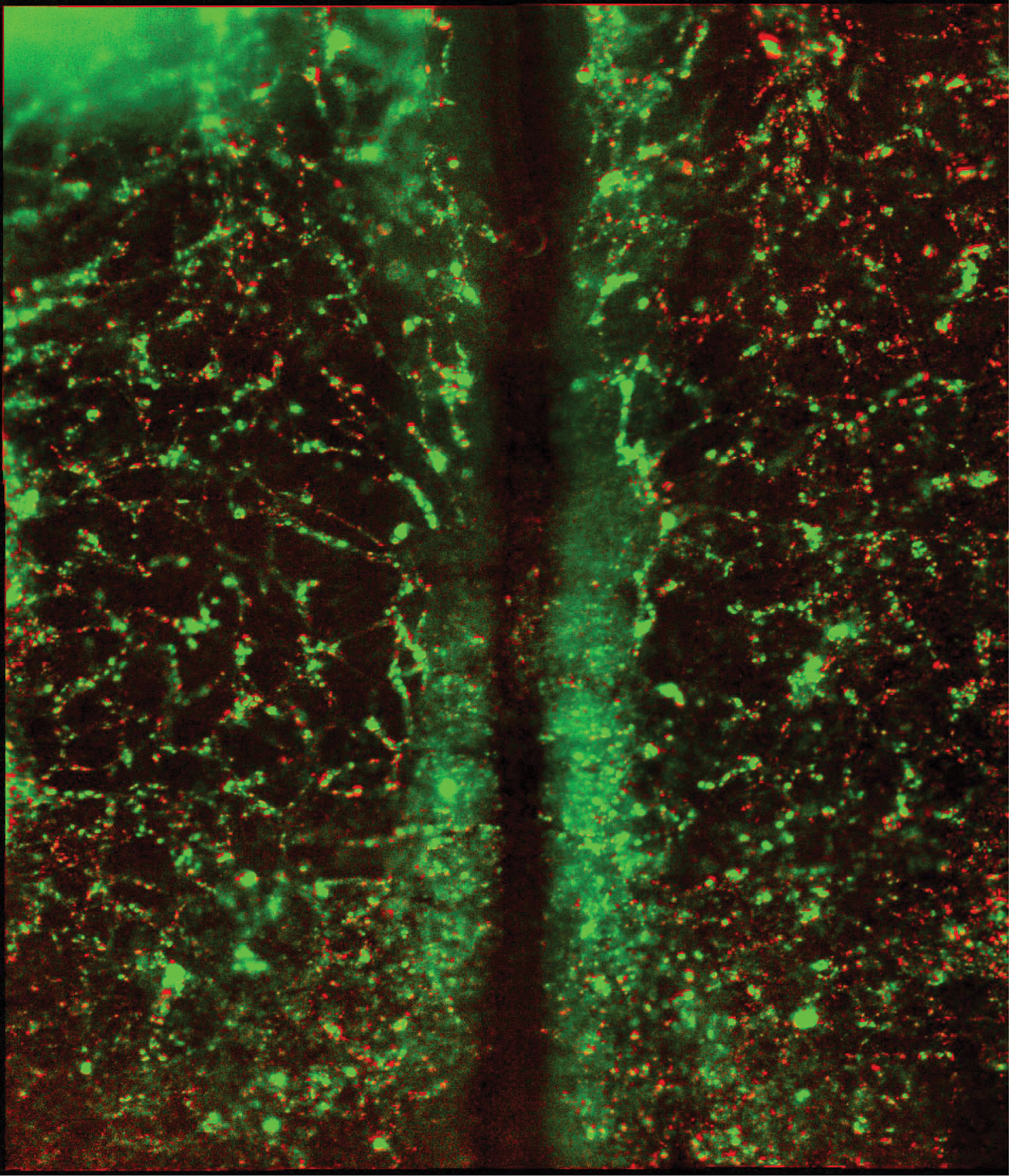
Active cell movements in the nascent vascular plexus of HH8 (6 somite) embryos. Red and green colors were assigned depending on the magnitude of active cell movements to a QH1-immunolabeled image. Fluorescence sources that move with the tissue environment are colored green. In contrast, QH1 foci that move relative to the surrounding tissue (i.e., display active motility) are colored red. Vascular sprouts are locations of intense cellular motility.
As analyzed in detail by Rupp et al., 23 vasculogenic sprouts can contact neighboring extensions and thus eventually establish endothelial cords. A stabilized protrusion may later be reinforced by subsequent addition of cells—making thereby new vertices and segments in the primary vascular pattern. As sprouts can extend hundreds of micrometers, they are multicellular structures, and thus sprout extension involves the coordinated activity of several (3-10) endothelial cells. Conversely, existing connections are also observed to retract, albeit with much lower frequency. There is an overall tendency for a lateral-to-medial autonomous motion, but individual cell movement directions scatter widely. In particular, cell chains moving in opposite directions within the same vascular segment are frequently observed as well as cells (nuclei) switching movement directionality 24 (Figure 6 and Supplementary Video 3). Therefore, as cellular motility is multidirectional, active cell movements are unlikely to be determined by relatively stable, long-range concentration gradients—such guidance may, however, explain the overall lateral-to-medial drift of the vascular network.
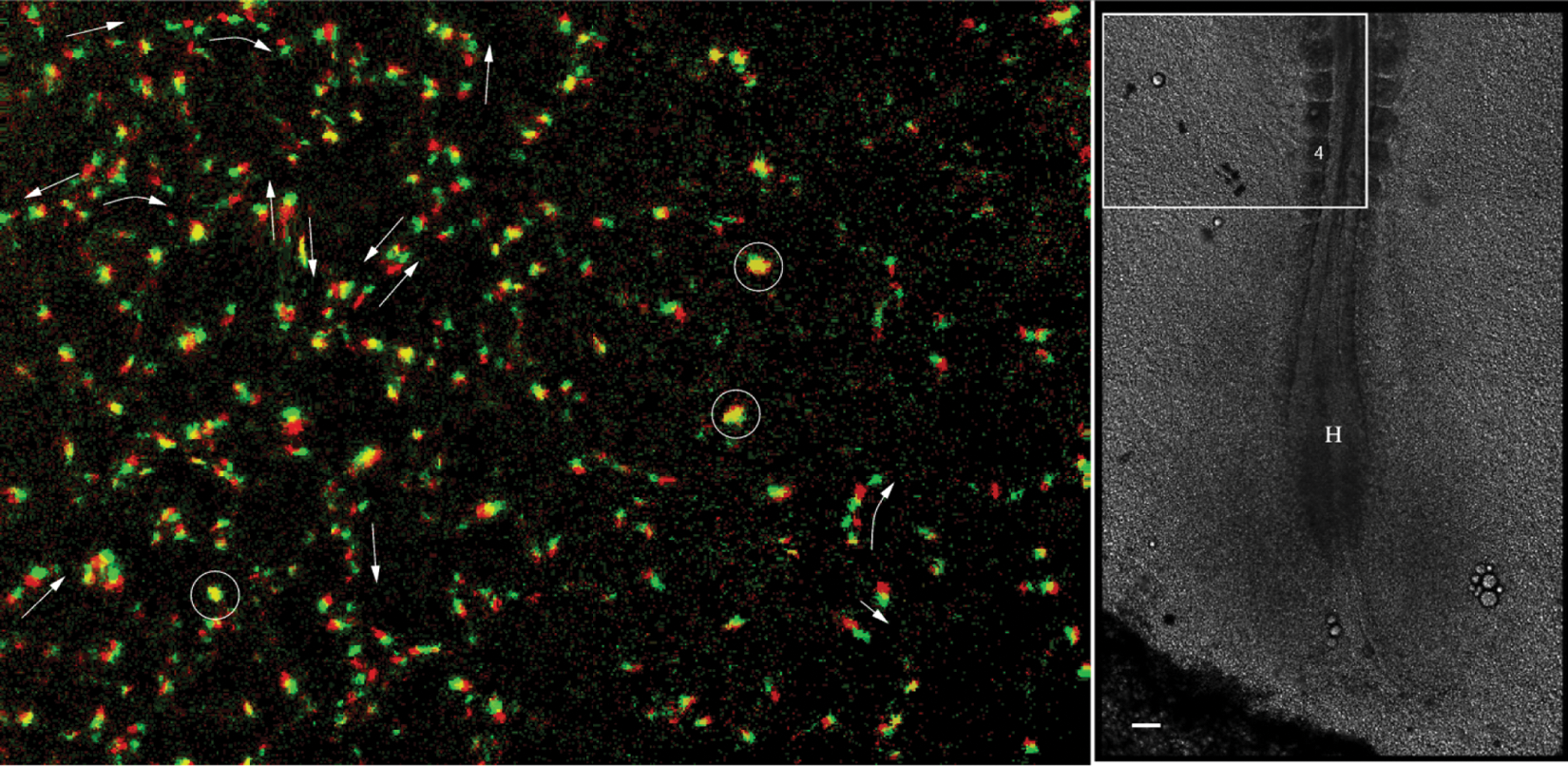
Active movement of TIE1+ nuclei, obtained after digitally correcting for the deformations associated with tissue motion (left). Two consecutive frames, separated by 8 minutes, are shown—the first as red, the second as green. Autonomous cell movement is inhomogeneous: some nuclei do not move (appear as yellow; some are marked by circles), whereas most cells move in a chain-migration fashion (indicated by arrows). At this stage of development, movement directions are highly variable: even in the same vascular segment, groups/chains are seen moving in opposite directions. The right panel marks the location of the area shown in the embryo. The fourth somite and Hensen’s node is marked. Scale bar: 100 µm. After Sato et al. 24 ; see also Supplementary Video 3.
The empirical motility data thus reveal that protrusive activity or sprouting is the mechanism used to generate new vascular cords resulting in the polygonal vascular pattern. In multicellular sprouts, protrusive activity is integrin dependent and requires an active engagement of the ECM. 23 Endothelial cell motility along existing vascular structures appears to rely more on vascular endothelial (VE)–cadherin-mediated cell-cell interaction. 28 Simultaneous monitoring of endothelial sprouts and changes in the surrounding ECM configuration revealed no evidence for sprout guidance by local ECM deformations.
Newly available dynamic imaging in amniotes shows that the motility and tissue displacements of precirculation stage endothelial cells are complex and bridge a large time and length scale.23,24 Based on hundreds of empirical time-lapse recordings, no simple set of displacement/motility rules specifies the formation of a primary vascular network within a large class of amniotes (aves), and although there are no corresponding wide-field time-lapse recordings of mammalian vasculogenesis, there is no evidence to suggest that mammals form their primary vascular beds in a different manner. The simplest conclusion, based on the time-lapse recordings, is that amniote vascular patterns, prior to circulation, are not predetermined by a dedicated network of patterning genes and cell signaling pathways (i.e., the process is not hardwired but is emergent).
Theoretical Models of Vascular Self-Assembly
The capacity of endothelial cells to form a polygonal pattern is preserved in various in vitro systems, where the presence of a genetic (or environmental) prepattern is not possible. The mouse allantois, when explanted, forms a vascular network very similar to the primary vascular pattern of the avian embryo—instead of a pair of umbilical vessels. 29 Similarly, a vascular network emerges when endothelial cells are placed in 3-dimensional collagen gels.30,31 Thus, endothelial cells are clearly capable of self-assembling a network, and we argue that this procedure occurs during early vasculogenesis in amniotes. During the past 20 years, a number of hypotheses have been proposed to explain the self-organizing aspect of vasculogenesis.
Contact Guidance
The ability to reorganize the ECM is well documented for tissue explants or cell aggregates embedded within an ECM gel. As revealed by the early experiments of Stoplak and Harris 32 and studied in more detail recently, 33 cell traction creates aligned ECM bundles radiating from a cell aggregate. Even individual cells can reorganize and align collagen fibers. 34 The developing oriented ECM structure, in turn, can guide cell migration35,36 in a manner similar to collagen gels oriented by magnetic fields.34,37-39 Combining these observations, an early model of vasculogenesis proposed that angioblasts first segregate into compact clusters and engage the surrounding ECM fibers. 35 As a result of traction forces, ECM bundles develop, which in turn later route the trajectory of motile primordial endothelial cells between clusters.19,35,40 Mathematical formulation of the mechanics of cell-ECM assemblies41-43 revealed patterning mechanisms in which a random initial inhomogeneity in the cell densities results in a coarsening (i.e., cell clustering) process by which increasingly large cell-free areas develop similar to the dynamics of foams. 40 This theory is a reasonable explanation of pattern formation on Matrigel cultures, a popular in vitro model of vascular assembly. Pattern formation on Matrigel surfaces indeed requires subconfluent cell seeding densities (thus, a confluent monolayer will not form a network), and the main patterning mechanism involves progressive elimination of small cell-free areas. As the theory suggests, this type of pattern formation is expected to occur with any type of cell that exerts traction forces and responds to ECM alignment (contact guidance). Indeed, fibroblasts, smooth muscle cells, and cells of the murine Leydig cell line TM3 formed networks on basement membrane matrix in much the same fashion. 44 When compared with the in vivo observations, the lack of sprouting and any obvious ECM bundles make this patterning mechanism unlikely for primary vasculogenesis.
Autocrine Chemotaxis
A recent body of research has focused on pattern emergence guided by autocrine chemotactic signaling.45,46 The proposed mechanism relies on the secretion of a diffusing chemotactic morphogen by the cells. This morphogen is assumed to possess chemoattractant properties, and in general, VEGF (or a particular VEGF isoform) is assumed to be a likely candidate. Although an autocrine chemoattractant is expected to result in cell aggregation, 47 further assumptions can steer the system toward branching patterns. Of particular importance is the finite compressibility of the cells: cells are assumed to resist being compressed into an arbitrary small area; instead, cells behave as if an effective pressure is developed within the aggregate. If the diffusion length (mean distance a secreted morphogen molecule moves before degradation or immobilization) is small enough, then the strongest gradients develop at the surface of the aggregate. Thus, the aggregate surface may become unstable: if random fluctuations (for example) move a cell away from the aggregate, it will sense a bit weaker gradient, and hence it will have a lower tendency to return to the aggregate. Moreover, the pressure of the compressed cells continues to push the cell outwards. By exploring this system with computer simulations, it has been shown that finite cell size, elongated cells, and increased chemotactic sensitivity at free cell surfaces all facilitate the sprouting process.48,49
The suggested chemoattractant, VEGF165, however, is unlikely to fit the model assumptions during embryonic vasculogenesis. VEGF165 is expressed throughout the embryo except in angioblasts or early endothelial cells.50,51 Thus, even if early endothelial cells secrete some small amount of VEGF, the concentration of the autocrine VEGF molecules is likely to be too small compared to the amount already present in the ECM microenvironment. Similar objections can be raised when this explanation is applied to the in vitro 3D collagen invasion assays. In such experiments, endothelial sprouts readily elongate even in the presence of relatively large concentrations of exogenous VEGF in the culture medium.52,53
Despite these objections, a mathematically equivalent patterning process does result from a related and more likely hypothetical mechanism. If a secreted proteolytic agent is assumed to increase the bioavailability of the ECM-bound VEGF, then a local gradient (of the bioavailable VEGF) may be produced in the microenvironment of an endothelial cell aggregate. Unfortunately, it is difficult to visualize morphogen gradients in vitro and more so in vivo. Thus, experimental validation of the autocrine signaling mechanism remains an interesting challenge.
Tip Cells and Stalk Cells
Recent experimental evidence suggests that during angiogenic sprouting, the leading tip cells have a different intracellular signaling activity than the remaining (often termed stalk) cells. 5 Tip cells are thought to be more motile, invasive, and responsive to chemotactic signals. Invoking a lateral inhibition mechanism, tip cells prevent adjacent cells from becoming tip cells as well. 54 This selection process is likely to be a dynamic one: tip cells assume this phenotype only for a few hours. 55
Although stalk cells are supposedly less active than tip cells in angiogenesis assays, they are quite motile during vasculogenesis in avian embryos (see Figures 4-6 and Supplementary Videos 1 and 3). Time-lapse recordings of allantois explants, an in vitro model of primary vasculogenesis, also indicate vigorous cell displacements, on a scale comparable to the sprout length. 28 In particular, expanding sprouts recruit cells from the aggregate. The newly recruited cells move along and may overtake (“leapfrog”) the cells comprising the sprout.
Theoretical considerations also suggest that stalk cells cannot be passively dragged by a motile tip cell: arguably, this process is inconsistent with widely accepted models of cell-cell adhesion. In particular, cadherin-mediated cell-cell adhesion has been repeatedly shown to be analogous to surface tension of immiscible liquid droplets56-60 and has been modeled accordingly in theoretical studies.61-63 Surface tension–stabilized structures are, however, prone to the Plateau-Rayleigh instability: a liquid jet with a circular cross section should break up into drops if its length exceeds its circumference.64,65 Due to this instability, a sprout pulled by a leader cell and held together by surface tension–like cell-cell adhesion should also break up. Therefore, the presence of leader cells and cell-cell adhesion alone cannot fully account for multicellular sprouting activity. 66
Preferential Attraction to Sprout Cells
If stalk cells within an expanding sprout move actively—as we argued is the case during vasculogenesis—then a guidance mechanism is needed to recruit such cells into the expanding sprouts. That is, cells must “prefer” to be adjacent to other stalk cells rather than remain in a cell aggregate.
The assumption of a “preferential adhesion mechanism” with persistently moving tip cells is sufficient to obtain expanding sprouts in computer simulations (Figure 7 and Supplementary Video 4). 66 Furthermore, multicellular sprouting behavior is exhibited not only by endothelial but also by glioma, muscle, or liver epithelial cells, even when cultured on a rigid, 2-dimensional surface.67-69 Thus, a rather generic mechanism was suggested that relies on cell-cell guidance—the preferential attraction to elongated cells. 68 Such a mechanism is sufficient to stabilize sprouts: as a sprout elongates, the constituent cells become increasingly attractive migration targets. The influx of additional cells (if available) helps to restore normal cell shape and stabilize the sprout.
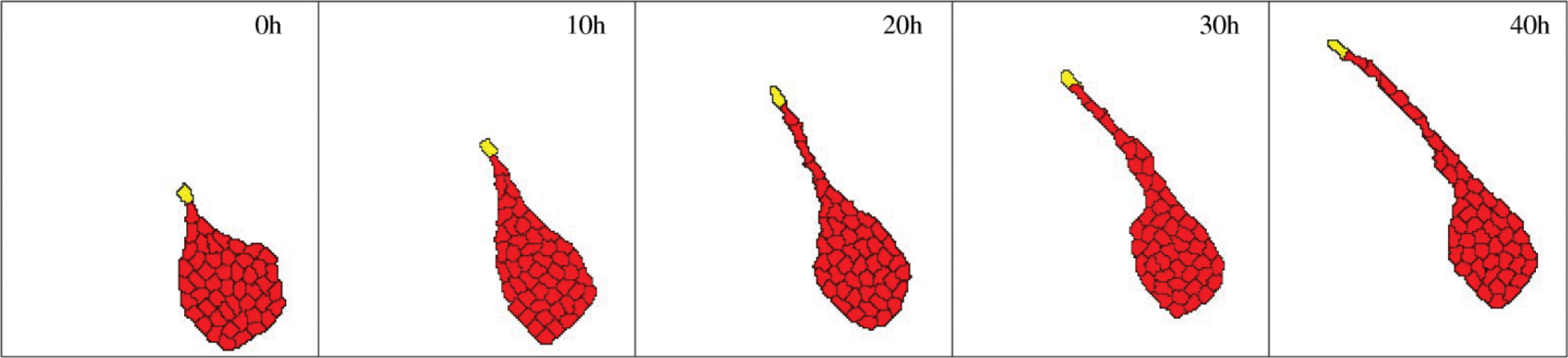
Computational model of multicellular sprout elongation. A leader cell (yellow) is assumed to move randomly with a persistent polarity; remaining cells (red) are assumed to prefer adhesion to elongated cells instead of to well-spread cells. This preference helps cells to leave the initial aggregate and enter the sprout. After Szabo and Czirok 66 ; see also Supplementary Video 4.
As speculated, the molecular mechanism for sensing elongated cells may involve micromechanical differences in the cytoskeleton of elongated and well-spread cells or even alterations in the available contact surface between the cells if elongated cells are “thicker.”
Relations to Tumor Vasculature
Arguably, the least understood and one of the most important questions facing vascular developmental biologists and tissue engineers today is, What are the general principles guiding morphogenesis of an endothelial tube network? The capacity of less than fully differentiated cells to assemble into vascular-like tubes is also manifested in various tumors. Best characterized are highly malignant melanomas in which tumor cells are assembling into tubes to secure a blood supply. 70 Thus, understanding how cells self-organize into interconnected cords is expected to be directly relevant for tumor biology.
As endothelial cells also invade and vascularize tumors, studies on avian vasculogenesis may provide important hints regarding the invasion process as well. Recent dynamic studies of blood vessel formation in a host of model systems suggest an interesting hypothesis: multiple strategies have evolved to accommodate vascular morphogenesis depending on the degree of relative motion in the target tissue. With respect to amniotes, the list of potentially unique strategies includes vascularization of wounds, nascent organs, tumors, various pathologies, retinas, embryonic brain tissue, and newly gastrulated embryos. In this article, we contrasted avian primary vasculogenesis with formation of mammalian retinal vessels, which are different not only because of the presence of a prepattern but also due to their physically distinct motion characteristics—the cardinal difference being that in the case of the gastrulating and elongating bird embryo, there is a large degree of expansion and reorganization on the part of the target tissue, whereas in the case of the mammalian retina, there is negligible tissue growth and motion. In our opinion, the physical dynamics required for vascular assembly in rapidly expanding tumors may be more similar to conditions in the embryo than to the retina.
We have shown that during amniote vasculogenesis, the extracellular environment is in motion and that the length scale of the motion is on the order of 0.1 to 1.0 millimeters. This is the length scale of a (human) micro-tumor that has enlarged to a size that will require vascularization for further growth. Moreover, some tumors grow rapidly and contain abundant complexes of ECM. We therefore hypothesize that some young, rapidly growing tumors mimic the physical properties of precirculation bird and mammalian embryos. Therefore, studies on the biophysical properties and the tissue dynamics of bird embryos, during vasculogenesis, will provide important clues to understanding the growth and vascularization of some malignant tumors.
Footnotes
Declaration of Conflicting Interests
The author(s) declared potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Institutes of Health [R01 grants HL087136 to A.C., HL085694 to B.J.R., and HL068855 to C.D.L.], the Hungarian Research Fund [OTKA K72664 to A.C.], and the G. Harold & Leila Y. Mathers Charitable Foundation [to A.C., C.D.L., B.J.R.].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
