Abstract
The discovery that Notch, a key regulator of cell fate determination, is functional in the vasculature has greatly improved our understanding of differentiation and specialization of vessels. Notch signaling has been proven to be critical for arterial specification, sprouting angiogenesis, and vessel maturation. In newly forming vascular sprouts, Notch promotes the distinction between the leading “tip” endothelial cell and the growing “stalk” cell, the endothelial cells that eventually form a new capillary. Notch signaling has also been implicated in vessel stability by regulating vascular mural cell function. More recently, macrophages carrying an activated Notch have been implicated in shaping the course of new sprout formation. Tumor vessels abide by similar principles and use Notch signaling in similar ways. An exciting discovery, made by several researchers, shows that blocking Notch function in tumor vasculature provides a means by which to suppress tumor growth. The authors discuss the developmental and physiological role of Notch in the vasculature and apply this knowledge to an overview of how Notch targeting in the tumor environment can affect tumor angiogenesis and growth.
The Notch signaling pathway functions to modulate cell fate decisions during development and adult life.1,2 Notch proteins (Notch1, Notch2, Notch3, Notch4) are single-pass transmembrane receptors that interact with 1 of 5 membrane-bound ligands: Jagged1, Jagged2, and Delta-likes Dll1, Dll3, and Dll4. At the cell surface, Notch is presented as an extracellular and intracellular heterodimeric receptor held together by noncovalent bonds. 3 Ligand binding to the extracellular peptide alters the receptor confirmation, leading to 2 proteolytic cleavages. ADAM (a disintegrin and metalloprotease domain) proteases cleave Notch outside the cell adjacent to the plasma membrane, followed by a presenillin/gamma-secretase proteolytic cleavage within the transmembrane, which releases the intracellular domain of Notch. The Notch intracellular domain (Notch-ICD) is then free to transit to the nucleus, where it forms a transcriptional activating complex with CSL/RBP-J (CBF-1, Suppressor of Hairless, LAG-1) and Mastermind. 4 The Notch/CSL transcriptional complex induces the expression of the Hairy/Enhancer of Split (HES) and HES-related genes (HEY, CHF, HRT, HESR). 5 The HES and HEY genes encode basic-helix-loop-helix transcriptional repressors that serve as important Notch signaling effectors.6,7
In the 1990s, researchers discovered that Notch1 8 and Notch4 9 are expressed in endothelium. Subsequently, it came to be appreciated that endothelial cells also express the Notch ligands, Dll1, 10 Dll4,11,12 and Jagged1. 13 More recently, an emerging picture is that Notch coordinates vascular endothelial growth factor (VEGF)–mediated angiogenesis.14-16 In fact, the role of VEGF and Notch in vascular development is exemplified by the fact that haploinsufficiency of VEGF17,18 or the Notch ligand, Dll4,19-21 results in early embryonic lethality due to disrupted angiogenesis.
Extensive analysis of mouse models with altered Notch signaling has further advanced our understanding of the role of Notch in developmental angiogenesis. Loss of
Notch signaling directly and indirectly regulates a cohort of genes involved in angiogenesis, including the VEGF receptors21,28-32 and EphrinB2.5,33-36 Notch indirectly inhibits VEGFR-2 expression via Hey2 induction,
28
whereas Notch activation induces VEGFR-1 transcripts and protein levels.21,29-31 However, it is not clear whether VEGFR-1 induction occurs due to direct Notch/CSL transcriptional control. Notch-dependent induction of VEGFR-1 may be pro- or anti-angiogenic, as VEGFR-1 has been shown to be both an agonist and antagonist of VEGF/VEGFR-2 signaling.
37
Notch signaling has also been shown to directly induce the expression of VEGFR-3.
32
In cultured blood endothelial cells, Notch/CSL binds and activates the
Notch and Arterial/Venous Specification
One of the earliest roles for Notch in the developing embryo is to direct arterial and venous endothelial cell determination. Endothelial cells are thought to originate in the mesoderm from a precursor cell termed the angioblast.
39
The angioblast goes on to differentiate into endothelial cells that acquire either an arterial or venous identity based on their expression of EphrinB2 or EphB4, respectively. In the nervous system, the EphrinB2/EphB4 signaling system has repulsive function; accordingly, EphrinB2/EphB4 signaling may have a similar role in the vasculature.
Only recently have we begun to appreciate the mechanism by which Notch regulates venous/arterial specification in vascular development. In zebrafish and mammals, a common precursor vessel segregates into the first embryonic artery (dorsal aorta) and vein (cardinal vein).
40
This process is dependent on both Dll4/Notch1 regulating EphrinB2/EphB4 signaling. In zebrafish, inhibition of Notch signaling with either a gamma-secretase inhibitor (GSI) or
In mice, when Notch4 signaling was constitutively activated in the endothelium, the dorsal aorta was enlarged, whereas the diameter of the cardinal vein was reduced. 41 The enlargement of the dorsal aorta was observed prior to vascular smooth muscle cell recruitment and was independent of endothelial cell proliferation. Instead, the authors demonstrated an increase in EphrinB2- and Dll4-positive cells in the dorsal aorta and a concomitant reduction of EphB4- positive cells in the cardinal vein. Similarly, endothelial-specific overexpression of Dll4 correlates with a loss of venous endothelial EphB4 expression and a gain in arterial endothelial EphrinB2 in the cardinal vein. 36 In contrast, disruption of Dll4/Notch1 signaling had the opposite phenotype, observed as an increase in EphB4+ endothelial cells in the cardinal vein and reduced EphrinB2+ endothelial cells in the dorsal aorta.33,41 Taken together, these data suggest that activation of Notch promotes endothelial cell differentiation toward an arterial identity, thereby ensuring proper segregation of arteries and veins.
It has recently come to light that Notch and EphrinB2 signaling also have independent functions in vascular development. At E9.5, both loss and gain of function
Later in development, arterial endothelial cells have been shown to require Dll1 to maintain their cellular identity. Dll1 expression is first observed in the mouse at E13.5 in the vascular endothelium, where it is restricted to the arteries and absent from the veins and capillaries.
25
Notch and Sprouting Angiogenesis
Insight into the function for Notch in sprouting angiogenesis and endothelial cell identity has come from studies of murine retinal angiogenesis (Figure 1). The work of Betsholtz, Eichmann, Wiegand, and others has helped to dramatically refine our understanding of Notch function in vascular biology by identifying 2 endothelial cell types essential for sprouting angiogenesis: the “tip” cell and the “stalk” cell.21,42,43 The “tip” cell, located at the leading edge of an angiogenic sprout, is highly migratory as evident by the presence of filopodia and expresses high levels of VEGFR-2, VEGFR-3, and platelet-derived growth factor-B. In the retina, the tip cells are thought to require high levels of VEGFR-2 to migrate toward a VEGF gradient produced by the underlying astrocyte plane. 44 The “stalk” cell, located adjacent to the tip cell, is proliferative and lumenizing, and it expresses high levels of VEGFR-1. The cellular mechanisms that differentiate a lumenizing stalk cell from a proliferative stalk cell have yet to be elucidated.
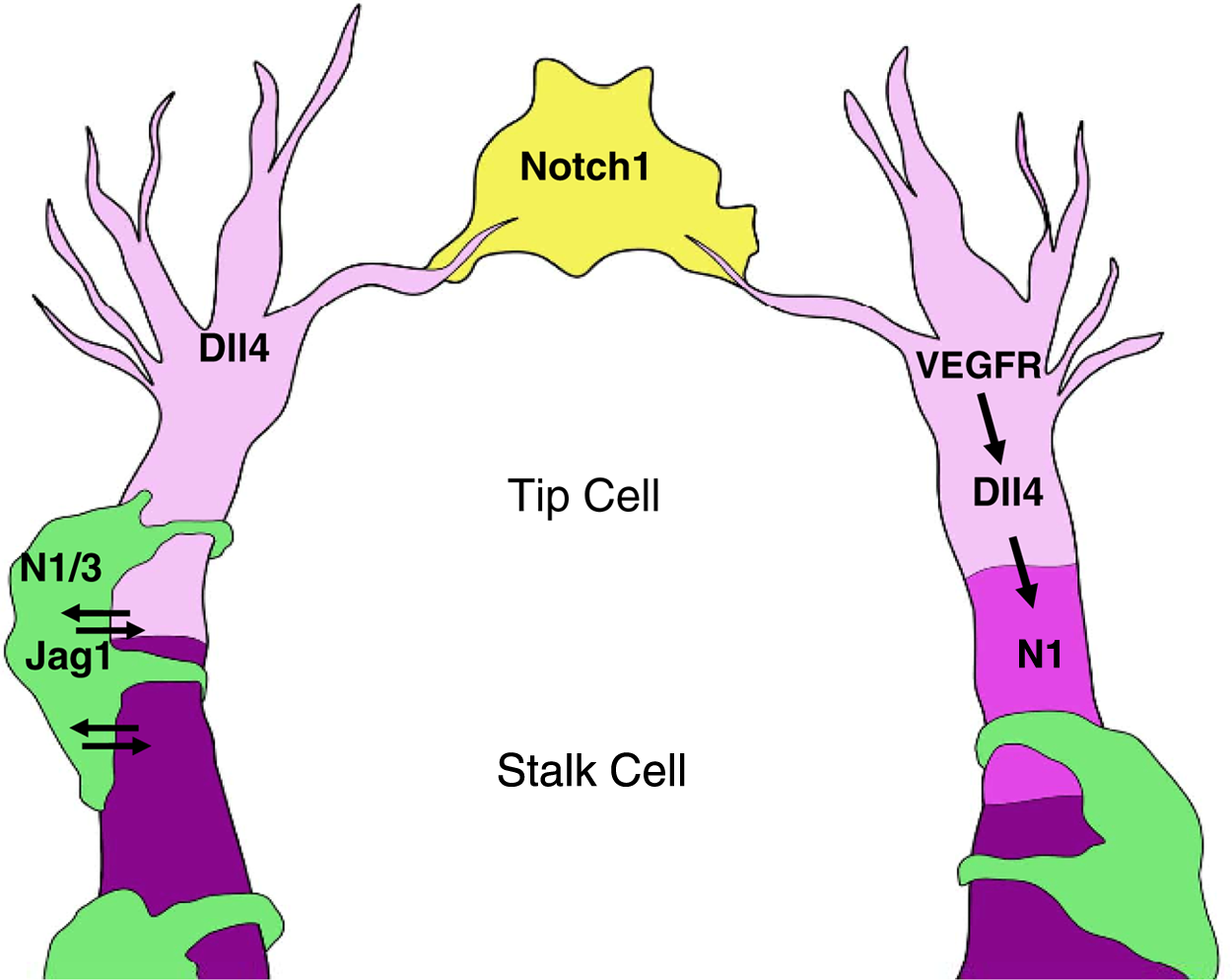
Notch signaling in sprouting angiogenesis. In an angiogenic sprout, the endothelial tip cell (light pink) expresses high levels of Dll4 in response to VEGFR-2 activation. Dll4 in the tip cell activates Notch1 in the neighboring endothelial stalk cell (dark pink). A Notch1-positive macrophage (yellow) is located between 2 endothelial tip cells undergoing anastomosis. It is proposed that the Notch-positive macrophage acts as a “bridge” between 2 Dll4 positive tip cells. Notch/Jagged1 signaling regulates endothelial and mural cell (green) interactions. N1 = Notch1; N3 = Notch3; Jag1 = Jagged1.
Analysis of the retinal vasculature of
In retinal angiogenesis, it has become clear that proper levels and localization of VEGFR-1, VEGFR-2, and VEGFR-3 are critical for tip cell identity and function.38,44 Inhibition of Vefgr-2 signaling in the retina results in decreased vessel density characterized by a reduction in endothelial cells with filopodia, suggesting a loss of the migratory tip cell identity. This phenotype was enhanced by additional inhibition of Vegfr-3. In contrast, inhibition of Vegfr-1 results in an increase in tip cells.
44
Using computational modeling and sprouting assays with genetically mosaic cells, the Gerhardt group found that endothelial cells are in constant competition for the tip cell position at the leading edge of an angiogenic front. 48 They propose that endothelial tip cell identity is transitory and dependent on environmental cues. Notch regulation of VEGF receptor expression downstream of Dll4 is critical in determining the dynamic flux between tip and stalk cell identity. This process appears to be in constant flux, with cellular contact between neighboring endothelial cells functioning as a dynamic process, subjecting cells to varying levels of Notch signaling and thus differential VEGF receptor expression. An understanding of this dynamic process helps to align the current model of high expression of Notch in the proliferative stalk cell with the findings that Notch signaling inhibits endothelial cell proliferation. It may be that high Notch signaling lasts only for a brief moment in a stalk cell to allow for momentary tip cell identity of its cellular neighbor during sprouting angiogenesis. However, once tip versus stalk cell identity has been briefly established, tight regulation of Notch signaling would allow for endothelial cell proliferation of stalk cells. Recently, Sirt1 has been shown to inhibit Notch signaling in the stalk cell and thus provide a mechanism by which Notch signaling can be fine-tuned. 49 Sirt1, a deacetylase, was shown to deacytelate the Notch1-ICD to promote its ubiquitin- mediated degradation and thereby downregulate Notch-mediated gene transcription. To fully understand the role of Notch in regulating sprouting angiogenesis, future work will need to focus on the cellular mechanisms that modify Notch signaling to allow for the dynamic cellular identities of the nascent sprout.
Notch and Vascular Mural Cell Function
The production of new, functional blood-carrying vessels during angiogenesis requires sprouting endothelial cells to undergo lumenization followed by appropriate maturation. Vessel maturation is dependent on the recruitment of mural cells to nascent vessels. As their name suggests, mural cells make up the vessel wall that supports the underlying endothelial cells, thereby functioning as critical components of vessel stability. In this review, we define mural cells as both pericytes and vSMCs.
It is well established that mural cells promote vessel stability by signaling to endothelial cells to inhibit their proliferation and promote survival. 50 Pericytes and vSMCs are cells of mesodermal origin and are believed to arise from the same cell lineage. However, the source of mural cells is still somewhat controversial in the field. It has been proposed that pericytes and vSMCs make up a differentiation continuum of the same cell type. Accordingly, pericytes and vSMCs are distinguished by their differential tissue distribution, their localization within the vessel wall, and their function. Capillaries are lined solely by pericytes, where they have a distinct morphology; pericytes appear to be wrapped around capillaries with multiple cellular protrusions and often lacking association with other pericytes. On the other hand, vSMCs are found in tight association with each other and are found in single or multiple layers around larger vessels. In addition, unlike vSMCs, which are exclusively found outside the endothelial basement membrane, pericytes share a basement membrane with the endothelium through which they can make direct contact with endothelial cells. 51
The intimate association of mural cells and endothelial cells provides an opportunity for Notch signaling between the two cell types. Notch may function either through homotypic mural cell contact or through direct cell contact between mural cells and endothelial cells, providing a mechanism by which one cell type can regulate the function of another. Mural cells express Notch1, Notch2, and Notch3 and the Notch ligands Jagged1 and Dll4.52,53 In embryonic and retinal angiogenesis, endothelial
Analysis of mural cell phenotypes of Notch mutant mice provides further support for a role of Notch in endothelial/mural cell interactions. Despite being viable and fertile, adult
The recruitment of mural cells to nascent blood vessels involves a variety of signaling mechanisms, including transforming growth factor–β (TGF-β), angiopoietin-Tie2, and the platelet-derived growth factor (PDGF) signaling pathway. PDGF signaling functions to promote mural cell migration and proliferation. During angiogenesis, spouting endothelial cells secrete the ligand PDGF-B to promote the proliferation and migration of PDFGR-β mural cells to the nascent vessels. Accordingly, genetic mouse models in which either
Another way that Notch may regulate mural cell coverage is through promotion of endothelial-to-mesenchymal transition (EMT). EMT has been shown to be an important process during cardiovascular development, where it has been implicated in both cardiac leaflet and endocardial cushion formation. 64 A role for Notch in regulating EMT is made possible by the fact that both endothelial cells and mesenchymal cells such as pericytes and vascular smooth muscles cells express Notch1, Notch2, and Jagged1. Furthermore, Notch can directly target and promote the expression of genes important for mesenchymal cell differentiation such as PDGFR-β and alpha smooth muscle actin (αSMA).62,65 In fact, Dll4 signaling has been shown to be required by bone marrow–derived cells to activate expression of pericyte/vascular smooth muscle–specific cell markers. 66 Expression of the intracellular domain of Notch1 in cultured endothelial cells was shown to induce morphological and functional changes that were characteristic of a mesenchymal phenotype. 64 In addition to regulating mural cell recruitment and differentiation, future work may show that Notch contributes to mural cell coverage by altering cellular identity. Understanding this process could have great therapeutic potential by providing an additional source of vascular support cells in pathological settings defined by mural cell loss.
Notch1 Function in Macrophage Recruitment and Macrophage-Mediated Angiogenesis
Macrophages arise from the myeloid lineage and can act to establish unique microenvironments in which specialized cellular functions can occur. Accordingly, macrophages have the potential to regulate endothelial cell function. Until recently, studies of Notch signaling in the myeloid lineage have focused on differentiation of progenitor cells.67,68 However, Notch signaling can also be activated in monocytes and macrophages, suggesting a role for Notch in mature cells of the myeloid lineage. Macrophages express Notch1, Notch2, and Notch4, as well as Dll4, Jagged1, and Jagged2.68-70 Direct cell contact between macrophages and endothelial cells could therefore result in active Notch signaling to regulate the function and identity of either cell type.
To describe the diversity of their activation states and function, a simplified paradigm of macrophage polarization toward an M1 “classic” or M2 “alternative” activation state is useful. 71 M1 macrophages are induced in response to lipopolysaccharide (LPS) and interferon-gamma (IFN-γ) and produce inducible nitric oxide synthase (iNOS) and cytokines such as tumor necrosis factor alpha (TNF-α), interleukin (IL)–1, IL-6, and IL-12, 72 whereas M2 macrophages are induced in response to IL-4, IL-10, or IL-13 and generally counteract the proinflammatory activity of M1 macrophages. 72 In addition to inhibiting inflammation, M2 macrophages can promote cellular responses that are distinct from the inflammatory response. For instance, M2 macrophages are thought to function in angiogenic regulation. M2 macrophages are characterized by expression of arginase-1 and mannose receptor and stimulate cell proliferation, collagen production, angiogenesis, and tissue remodeling. 72 Production of pro-angiogenic factors is generally associated with M2 macrophages, which can produce VEGF, fibroblast growth factor–1 (FGF-1), angiopoietin-1 (Ang1), and matrix metalloproteinases.72,73 However, the inflammatory cytokines, IL-6 and TNF-α, that are released by M1 macrophages can also be pro-angiogenic.74,75 Thus, stimulation of angiogenesis is a function of macrophages that is not necessarily limited to a particular differentiation state.
Expression of Notch proteins is upregulated in response to inflammatory stimuli such as LPS and IFN-γ.31,69,76 Notch may also interact with the nuclear factor–kappa B (NF-κB) and mitogen-activated protein kinase (MAPK) pathways, which have long been known to be essential for the inflammatory response of macrophages.76-78 Thus, Notch may modulate the activity of other signaling pathways to fine-tune macrophage responses to environmental cues. Indeed, our group and others have shown that Notch regulates macrophage response to inflammatory stimuli. Macrophages mutant for Notch signaling have decreased expression of IL-6, IL-12, and TNF-α in response to LPS/IFN-γ stimulation.31,76
Macrophages are recruited to sites of inflammation and active angiogenesis via VEGFR-1 activation, which may also regulate their angiogenic function at these sites.79-82 This shows that VEGF, although most notable for its effect on endothelial cells to promote angiogenesis, can also affect macrophage function. It has also been proposed that VEGF stimulates inflammatory lymphangiogenesis via recruitment and function of macrophages. 83 In addition to VEGF, there is increasing evidence that Ang2, via Tie2 expressed on macrophages, plays an important role in their pro-angiogenic activity, particularly in tumor models. 84 Therefore, macrophages affect angiogenesis in a variety of contexts via distinct signaling pathways.
Previous studies show that macrophages are closely associated with sprouting endothelial cells in the developing postnatal mouse retina. 85 In this setting, macrophages appear to form bridges between neighboring endothelial cell sprouts to facilitate anastomosis. 86 As such, macrophage-deficient mice have reduced branching in the retinal vascular plexus.85,87 Macrophages may also affect the directionality of endothelial cell sprouts by promoting sprouting both forward and sideways to form the vascular plexus. 88 VEGF-mediated endothelial sprouting and macrophage-mediated anastomosis appear to be distinct steps in angiogenesis.86,88 In fact, macrophage regulation of vessel fusion is secondary to VEGF-mediated endothelial cell induction, as the number of tip cells and filopodia is comparable in both macrophage-deficient and wild-type mice. 87 The mechanism by which macrophages mediate tip cell branching remains unclear. It is likely that communication between macrophages and endothelial cells elicits the release of angiogenic factors from both cell types to mediate anastomosis. The factors involved and their cellular source remain to be determined.
Recent
Notch regulates macrophage expression of Vegfr-1, 31 suggesting that Notch signaling is important for macrophage recruitment and function in tissues with increased expression of VEGF—namely, those undergoing inflammation or angiogenesis. There may not be a need for a distinction between inflammatory and angiogenic tissue, as inflammation occurs in vascularized tissue, and conversely, inflammation necessitates remodeling of blood vessels that are often damaged or destroyed by the inflammatory process. 90 Wounds, tumors, and many other pathological processes are characterized by increased expression of VEGF that acts as a chemoattractant for inflammatory cells.37,91 Notch-regulated VEGFR-1 expression may also mediate macrophage function at these sites. Because of their ability to extend many long and complex filopodia, macrophages are ideally suited to interact directly with other cells in their environment but also to act as “adaptors” between different cells. Currently, models of sprouting angiogenesis largely overlook the role that macrophages play in this process. Notch signaling in macrophages during angiogenesis may therefore complicate anti-angiogenic therapeutic strategies, which largely focus on Notch signaling only in endothelial cells. The role of Notch in macrophages and their interaction with endothelial cells should be recognized as important for mediating endothelial cell behavior.
Future work may further elucidate a role for Notch signaling in defining the activation state of differentiated macrophages in a variety of cellular and tissue contexts. Our data and that of others suggest that Notch plays a role in defining the activation state of differentiated macrophages. However, previous studies have also suggested that Notch signaling may promote commitment of hematopoetic progenitor cells to the myeloid fate, 67 implying that Notch signaling is active in both early and later phases of myeloid differentiation. This concept raises the question of how Notch signaling can affect myeloid cell phenotypes in such divergent ways. Because macrophages express multiple Notch proteins, it is possible that the relative expression of different receptors may dictate distinct phenotypes and downstream functions. The many combinations of Notch ligands and receptors and the spatial and temporal regulation of Notch ligand and receptor expression can mediate differences in Notch signaling activity depending on context. 92
Notch and Tumor Angiogenesis
Pathological angiogenesis relies on similar mechanisms and angiogenic factors as those involved in physiological angiogenesis, including hypoxia and VEGF signaling. When a tissue lacks sufficient oxygen, a series of coordinated signal transduction cascades are initiated and culminate in blood vessel recruitment into the hypoxic tissue. Under normal conditions, angiogenic factors and corresponding cellular functions usually recede when vascular perfusion allows sufficient oxygen to reach the tissue. However, pathological angiogenesis, such as tumor angiogenesis, is persistent and unresolved due to an incessant supply of tumor-derived angiogenic factors and inflammatory cytokines. It is well established that growth and metastasis of solid tumors are crucially dependent on development of the new vasculature.
VEGF signaling is the best-characterized signaling pathway in angiogenesis. VEGF-A is upregulated by the transcription factor hypoxia-inducible factor (HIF) and binds to 2 receptor tyrosine kinases, VEGFR-1 and VEGFR-2. Several studies demonstrate that VEGF-A is overexpressed in many human tumors. 93 Therefore, it is not surprising that inhibition of angiogenesis and development of the VEGF blockade have been considered an attractive strategy to treat human cancer (Table 1). VEGF inhibitors, including Food and Drug Administration (FDA)–approved bevacizumab (Avastin), have been shown to reduce tumor angiogenesis and tumor growth and are now validated for cancer therapy. 94 However, different tumors reportedly exhibit widely varying susceptibility to VEGF inhibition. This phenomenon may occur due to alternative angiogenic signals that rescue tumor vasculature and allow tumor growth despite VEGF blockade.
Anti-Angiogenesis Agents
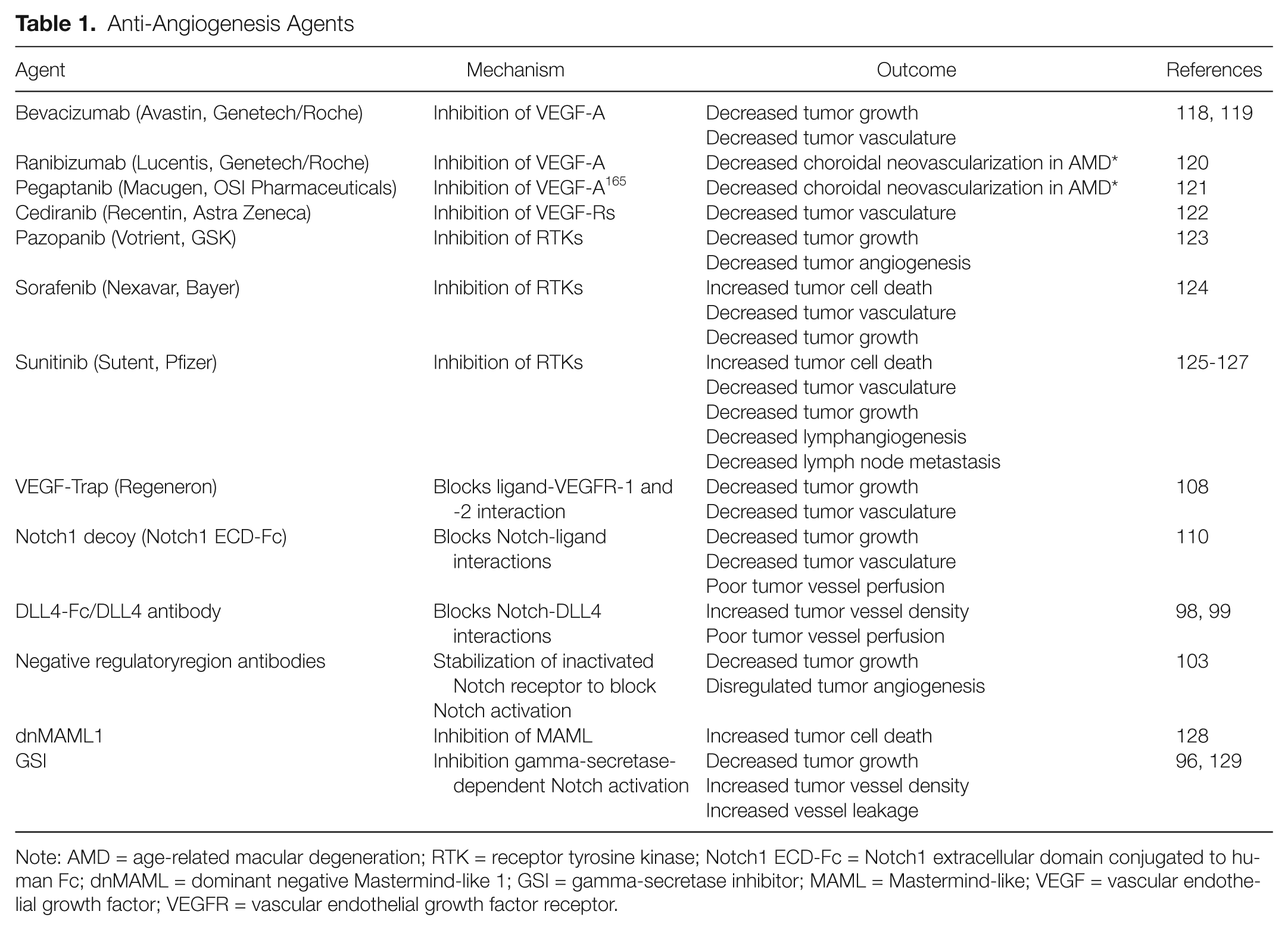
Note: AMD = age-related macular degeneration; RTK = receptor tyrosine kinase; Notch1 ECD-Fc = Notch1 extracellular domain conjugated to human Fc; dnMAML = dominant negative Mastermind-like 1; GSI = gamma-secretase inhibitor; MAML = Mastermind-like; VEGF = vascular endothelial growth factor; VEGFR = vascular endothelial growth factor receptor.
A role for Notch signaling is well documented in both hematological malignancies and solid tumors. 95 Notch has been demonstrated to act as either a tumor suppressor or an oncogene in a context-dependent manner. In addition, several lines of evidence suggest that Notch also plays an important role in tumor angiogenesis. As previously discussed, Notch receptors and ligands are widely expressed in endothelial and perivascular cells, but they are also shown to be upregulated in several types of tumor cells. Therefore, Notch, like VEGF, has become a focus for the development of targeted therapeutics by pharmaceutical companies. Several GSIs have been developed and evaluated in phase I clinical trials. However, the GSI-induced toxic effects of goblet cell metaplasia and diarrhea make the reagent unpromising for cancer treatment. 96 Interestingly, Real and colleagues 97 demonstrated that glucocorticoids protected GSI-treated mice from developing goblet cell hyperplasia, which supported the possibility of glucocorticoids plus GSIs in the treatment of T cell acute lymphoblastic leukemia (T-ALL).
Tumor vasculature and tumor cells have also been shown to overexpress the Notch ligand Dll4.
47
Genetic studies have shown that reduction of Dll4 results in hypersprouting phenotypes and disruption of the vasculature.
33
Accordingly, several anti-DLL4 agents have been developed and have entered phase I clinical trials. DLL4 blockade resulted in decreased tumor growth due to poorly perfused, nonfunctional tumor vasculature.98-100 Treatment with either a DLL4-neutralizing antibody or a soluble form of DLL4, DLL4-Fc, caused an increase in sprouting angiogenesis and endothelial tip cell number in tumors. In addition, DLL4-blocking agents have been shown to lack the adverse effect of goblet cell metaplasia, which is a dose- limiting factor for GSIs.
99
Thus, DLL4 inhibitors appeared to be a promising agent for cancer therapy. However, it has been shown that chronic Dll4 inhibition can cause pathological activation of endothelial cells and consequently vascular neoplasms.
101
Liver pathology has also been documented with chronic treatment of DLL4 antibodies in several animal models. The observation of these chronic effects of DLL4-neutralizing antibodies was further supported by genetic studies of Notch1 as a potential tumor suppressor.
102
Loss of
To avoid the severe side affects associated with pan-Notch inhibitors such as GSIs, Notch receptor–specific blocking antibodies have been developed. Antibodies that block ligand binding to either Notch1 or Notch2 have been shown to decrease tumor volume in mouse xenograft models. 103 Consistent with DLL4 blockade, these tumors had increased vascular density, as measured by an increase in CD31 staining. Furthermore, treatment with Notch1 antibody increased vessel sprouting and vascular density in the retina. These findings suggest that Notch-specific blockade can inhibit tumor growth by disrupting angiogenesis. Encouragingly, treatment with only Notch1 or Notch2 blocking antibody alone has been found to be nontoxic in the mouse.
Nevertheless, anti-angiogenesis still remains an attractive strategy for treatment of solid tumors. More studies have shown that, in addition to the endothelium, perivascular cells also play an important role in angiogenesis, and they can be dysregulated in pathological angiogenesis.50,104,105 Liu and colleagues 55 showed that endothelial cells and mural cells communicate with each other through NOTCH3 and JAGGED1. In addition, JAGGED1 expressed in tumor cells has been shown to stimulate Notch-dependent angiogenesis. 106 Inflammatory cells, including tumor-associated macrophages (TAMs), release a number of pro-angiogenic cytokines, such as VEGF-A, TNF-α, and basic fibroblast growth factor (bFGF). 107 Therefore, it is possible that the next generation of Notch-inhibiting agents will be the ones that target Notch activity in perivascular cells rather than endothelial cells. Recent data support the ideas that individual Notch receptors and ligands may differentially regulate sprouting angiogenesis, vascular remodeling, and endothelial–mural cell interaction. By blocking specific Notch signaling pathways, the adverse effects of activated endothelial cells and vascular neoplasms may be reduced or even avoided.
Protein-based signaling pathway inhibitors have also been studied as anti-angiogenic agents in tumors. These peptide molecules contain the active domain of the protein and thereby block receptor- ligand interaction. These constructs include VEGF-Trap, 108 dominant negative Mastermind, 109 and Notch1 decoy. 110 VEGF-Trap is a protein consisting of regions of the extracellular domains of VEGFR-1 and VEGFR-2 that prevents activation of the VEGF receptor by sequestering its ligands. 108 When purified protein is injected twice weekly, VEGF-Trap inhibited tumor growth and decreased vascular density in various mouse tumor models, including mouse melanoma, rat glioma, and human sarcoma cells. Dominant negative Mastermind-like-1 (dnMAML) binds to the CSL and Notch-ICD complex to prevent Notch target gene transcription. dnMAML is primarily designed to target cancer stem cells by inducing apoptosis in Notch-driven cancers such as T-ALL. 109 However, in mouse tumor xenograft models, osteosarcoma cells expressing dnMAML have decreased tumor growth compared to control. 111 The Notch1 decoy consists of the extracellular domain of Notch1 and can block the activity of several Notch ligands, including Jagged1, Dll1, and Dll4. Notch1 decoy therefore blocks Notch activation. However, tumors expressing the decoy do not show an increase in vascular density, which is a characteristic outcome of treatment with GSIs and DLL4 antibodies. Instead, when tumors expressing Jagged1 were treated with N1 decoy, they displayed reduced and disrupted endothelial networks and a significant reduction in vSMC recruitment. Thus, the Notch1 decoy, in contrast to Dll4 blockade, is a truly anti-angiogenic compound. The Notch1 decoy blocks not only Dll4-specific Notch activation but also the activity of other ligands, suggesting that inhibition of other Notch ligands should be considered as therapeutic targets.
Although Notch inhibitors are still preclinical, the monoclonal VEGF antibody bevacizumab is clinically available and represents a potential for combination therapy. As discussed above, some tumors are responsive to VEGF inhibition, although others are unresponsive and become resistant over time.112,113 This is most likely due to compensation by other angiogenic pathways that maintain the tumor vasculature. In patients with metastatic breast cancer, treatment with anti-VEGF therapy did not show a statistically significant increase in overall survival benefit. 114 However, combination anti-angiogenic therapy may be useful in overcoming this resistance. In preclinical tumor models, VEGF therapy in combination with chemotherapy or other inhibitors has additive effects on inhibition of tumor growth when compared to monotherapy. In a mouse tumor model of Ewing’s sarcoma, soluble VEGFR-1 peptide-based inhibitor combined with cyclophosphamide had an enhanced effect on decreasing tumor size. 115 Combined inhibition of placental growth factor (PlGF) using anti-PlGF and anti-VEGFR-2 or soluble VEGFR-2 peptide also showed a more pronounced inhibitory effect on tumor growth. 116 Combination therapy that includes DLL4 inhibition has also been proven to decrease tumor size. In xenograft mouse model tumors, anti-DLL4 antibody in combination with chemotherapy (paclitaxel) caused an enhanced decrease in tumor size and reduced the number of cancer stem cells. 100
In tumor models, VEGF inhibition has been reported to directly affect Notch signaling and vice versa. VEGF inhibition in the mouse Lewis lung carcinoma tumor model correlates with a decrease in Dll4 expression in tumor vessels. 98 Also, human neuroblastoma tumor xenografts in mice treated with VEGFR-2 blocking antibody had upregulated Jagged1 protein expression and displayed increased Notch1 activation. 117 Given these findings, coupling agents that block the VEGF pathway with agents targeting the Notch pathway may result in enhanced treatment efficacy. Combined therapy may therefore overcome the issue of tumor escape that is associated with single-agent treatments. Evidence of this benefit was demonstrated when anti-DLL4 antibody was combined with VEGF inhibition in human lung carcinoma and mouse leukemia tumor models. In both studies, combined therapy resulted in enhanced tumor growth inhibition as compared to either monotherapy. 99 It has also been shown that human sarcoma xenograft tumors in mice that are unresponsive to VEGF inhibition are instead responsive to DLL4 blocking antibody. 98 These data further exemplify the crosstalk between VEGF and the Notch signaling pathway. Clearly, the field of Notch pathway inhibitors is a new and exciting area in cancer research, and additional study of their use both alone and in combination with chemotherapy or other biological agents should be explored.
Footnotes
Declaration of Conflicting Interests
The authors declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the following grants from the National Institutes of Health: T32DK07328 (N.M.K. and J.G.), R01 CA136673 (C.J.S.), F31HL090032-01 (H.O.R.), and R01 HL62454 (J.K.).
