Abstract
Tumor cells have devised several strategies to block the mitochondrial pathway of apoptosis despite endogenous or pharmacological cues to die. This process of cell death proceeds through the coordinated regulation of multiple anti-apoptotic and pro-apoptotic BCL-2 family proteins that ultimately impinge on the integrity of the outer mitochondrial membrane. Once compromised, mitochondria release pro-apoptotic factors to promote caspase activation and the apoptotic phenotype. Within the BCL-2 family exists a subclass of pro-apoptotic members termed the BH3-only proteins, which directly and/or indirectly functionally regulate the remaining anti- and pro-apoptotic BCL-2 proteins to compromise mitochondria and engage apoptosis. The focus of this review is to discuss the cellular and pharmacological regulation of the BH3-only proteins to gain a better understanding of the signaling pathways and agents that regulate this class of proteins. As the BH3-only proteins increase cellular sensitivity to pro-apoptotic agents such as chemotherapeutics, numerous small-molecule BH3 mimetics have been developed and are currently in various phases of clinical trials. Toward the end of the review, the discovery and application of the small-molecule BH3 mimetics will be discussed.
Introduction
Apoptosis is a physiological form of programmed cell death that is essential for metazoan development and tissue homeostasis. Aberrations in apoptotic signaling are implicated in numerous disease states, including autoimmunity, cancer, and degenerative disorders.1-5 Importantly, alterations within the apoptotic pathway contribute to tumorigenesis and confer resistance to not only physiological apoptotic stimuli but therapeutic regimens as well. Apoptotic signaling can initiate from outside the cell via plasma membrane receptors (referred to as the
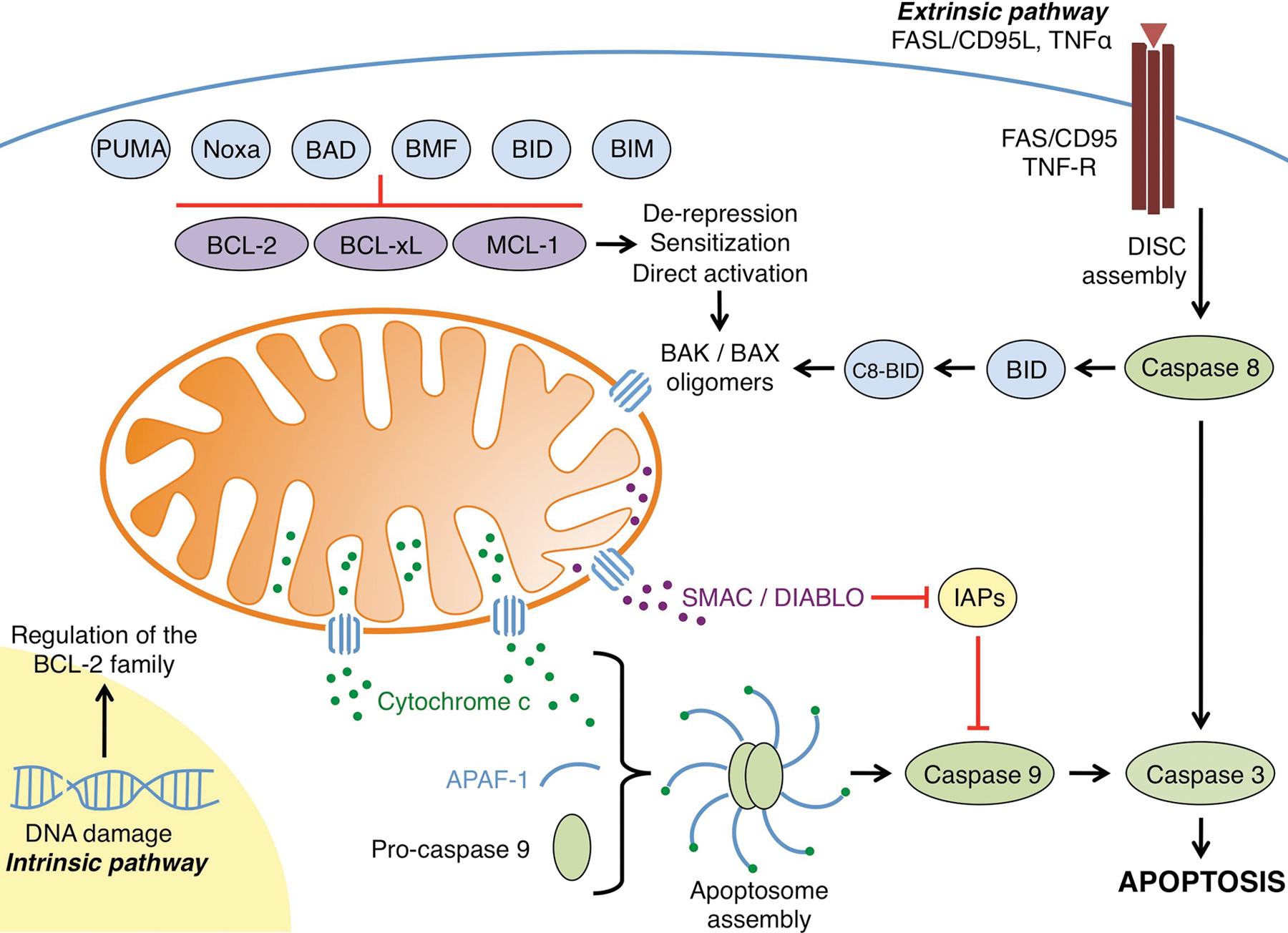
The regulation of apoptosis by the BCL-2 family. The intrinsic pathway is initiated by cellular stress signals such as DNA damage and growth factor/cytokine withdrawal. These intrinsic signals lead to the transcriptional upregulation and/or activation of pro-apoptotic BH3-only proteins such as BIM, BID, BAD, BMF, Noxa, and PUMA. These proteins are able to bind anti-apoptotic members of the family (e.g., BCL-2, BCL-xL, and/or MCL-1) and inhibit their activity. In addition to binding and inhibiting anti-apoptotic proteins, direct activator BH3-only proteins (e.g., BID and BIM) also bind and activate the effector molecules BAK and BAX. Once activated, BAK and BAX homo-oligomerize and subsequently form pores in the outer mitochondrial membrane, leading to mitochondrial outer membrane permeabilization. Pro-apoptotic proteins such as cytochrome c and SMAC/DIABLO are subsequently released from the intermembrane space into the cytosol. Cytochrome c forms a complex with APAF-1 and pro-caspase 9, whereas SMAC/DIABLO binds the inhibitors of apoptosis proteins (IAPs), which normally bind and inhibit initiator as well as effector caspases. Both steps allow for the dimerization and activation of caspase 9, subsequent cleavage and activation of effector caspase 3, and the apoptotic phenotype. The extrinsic apoptotic pathway is activated by extracellular signals such as death ligands, including FASL/CD95L and tumor necrosis factor (TNF) α. Binding of these ligands to their receptors (FAS and TNF receptor, respectively) causes receptor trimerization and subsequent recruitment of several factors that form the death-inducing signaling complex (DISC). The DISC activates initiator caspase 8, which subsequently activates effector caspase 3. Crosstalk can occur between the extrinsic and intrinsic pathways following the DISC assembly. Activated caspase 8 is able to cleave and activate the pro-apoptotic BH3-only protein BID, leading to C8-BID, which can simultaneously initiate the intrinsic apoptotic pathway through effector molecule activation.
The BCL-2 family comprises a network of related proteins that are defined by their α-helical composition of up to 4 BCL-2 homology domains (BH) and are divided into 3 classes: the anti-apoptotic BCL-2 proteins (e.g., BCL-2, BCL-xL, and MCL-1), the pro-apoptotic BCL-2 effectors (e.g., BAK and BAX), and the pro-apoptotic BH3-only proteins (e.g., BID, BIM, and PUMA) (Figure 2). 10 The founding member of the family is the anti-apoptotic protein BCL-2, which was identified as a molecular hallmark of follicular B cell lymphoma at the chromosomal translocation t (14; 18). 11 Since its discovery as an oncogene, the function of BCL-2 has been defined as preventing cell death as opposed to inducing cell proliferation. 12 Subsequently, numerous anti- and pro-apoptotic members have been identified, and each functionally cooperates within the BCL-2 family. In general, the transcriptional and posttranslational regulation of individual members, along with the numerous BCL-2 family protein-protein interactions, regulates the integrity of the outer mitochondrial membrane (OMM). Following pro-apoptotic treatment or accumulated stress, anti-apoptotic BCL-2 proteins become functionally inhibited, whereas pro-apoptotic BCL-2 proteins are enabled to permeabilize the OMM, through the process of mitochondrial outer membrane permeabilization (MOMP). MOMP occurs when proteolipid pores are created in the OMM, allowing for the release of mitochondrial proteins, such as cytochrome c and SMAC/DIABLO (second mitochondria-derived activator of caspase/direct inhibitors of apoptosis protein [IAP]–binding protein with low pI) from the mitochondrial intermembrane space into the cytosol, where they directly promote caspase activation and rapid apoptosis (Figure 1).
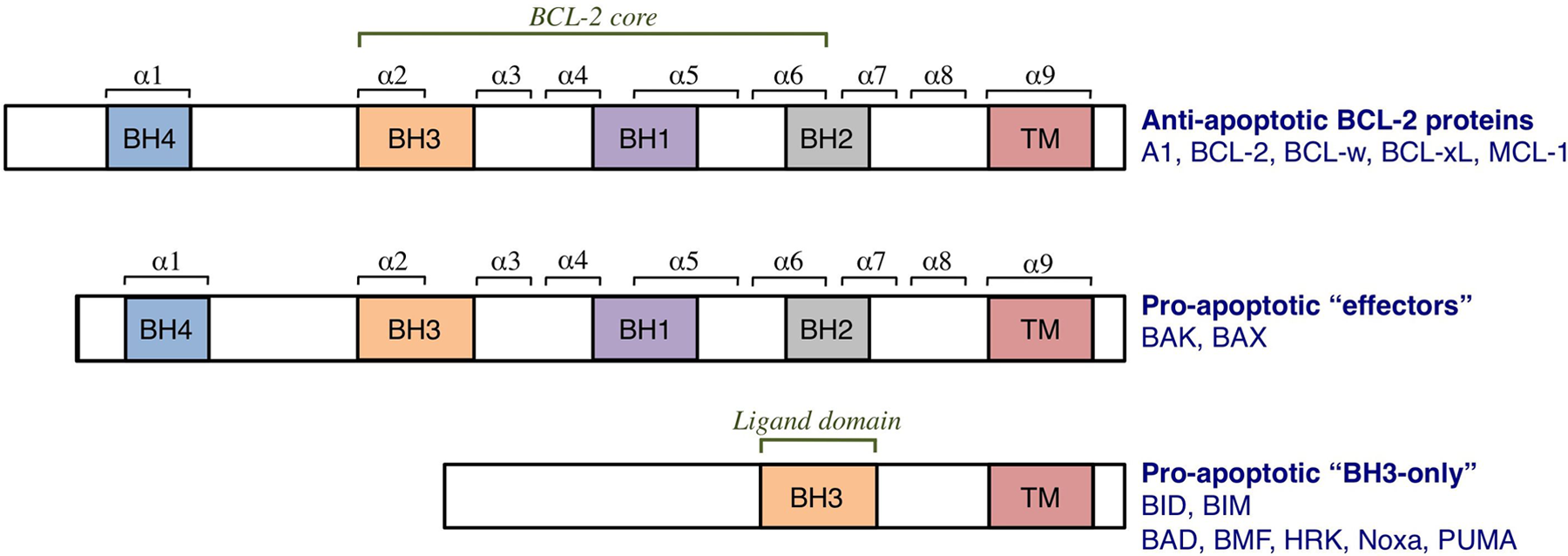
The BCL-2 family of proteins. The family is divided into anti- and pro-apoptotic members. The anti-apoptotic members, which include A1, BCL-2, BCL-xL, BCL-w, and MCL-1, share homology in all 4 BCL-2 homology (BH) domains (BH1-4). BCL-2 homology domains 1 to 3 comprise the BCL-2 structural core and create the hydrophobic groove. It is this “BCL-2 groove” that is the binding site for the BH3 domains of pro-apoptotic members. The pro-apoptotic members are subdivided into “effector” proteins and “BH3-only” proteins. The effector proteins also contain homology domains 1 to 4, whereas the BH3-only proteins contain only one BH domain, the BH3, which binds the anti-apoptotic proteins. Many BCL-2 proteins also contain a hydrophobic transmembrane domain (TM).
Here we review the basic mechanisms that control BCL-2 family function, MOMP, and apoptosis in the context of tumor cell development and resistance to anti-tumor treatments. Finally, we discuss the role of pharmacologically regulating the BCL-2 family of proteins to enhance anti-tumor strategies and patient survival.
The BCL-2 Family
The anti-apoptotic BCL-2 proteins
The anti-apoptotic BCL-2 proteins are responsible for maintaining the integrity of the OMM. They are globular proteins that contain BH domains 1 to 4 (Figure 2). The members of this subfamily include A1 (BCL-2-related gene A1), BCL-2, BCL-xL (BCL-2-related gene, long isoform), BCL-w, and MCL-1 (myeloid cell leukemia 1). These proteins are generally found at the OMM but can also be localized to the endoplasmic reticulum (ER) membrane and in the cytosol. 13 The anti-apoptotic BCL-2 proteins preserve OMM integrity by directly binding to both classes of pro-apoptotic BCL-2 proteins (i.e., the BH3-only and effector proteins), which prevents them from cooperating to induce MOMP and subsequent apoptosis. The anti-apoptotic members share a conserved “BCL-2 core” structure composed of 6 alpha (α) helices that results in the formation of a hydrophobic groove. Although the anti-apoptotic BCL-2 proteins demonstrate conserved sequence and/or structural homologies, slight variations within this groove afford unique binding profiles between the anti-apoptotic and pro-apoptotic proteins. Likewise, many of the pharmacologic inhibitors developed to target the anti-apoptotic BCL-2 proteins are able to distinguish between the anti-apoptotic members due to variations within this groove and therefore have different binding specificities to these proteins (Table 1).
BCL-2 Family Inhibitors
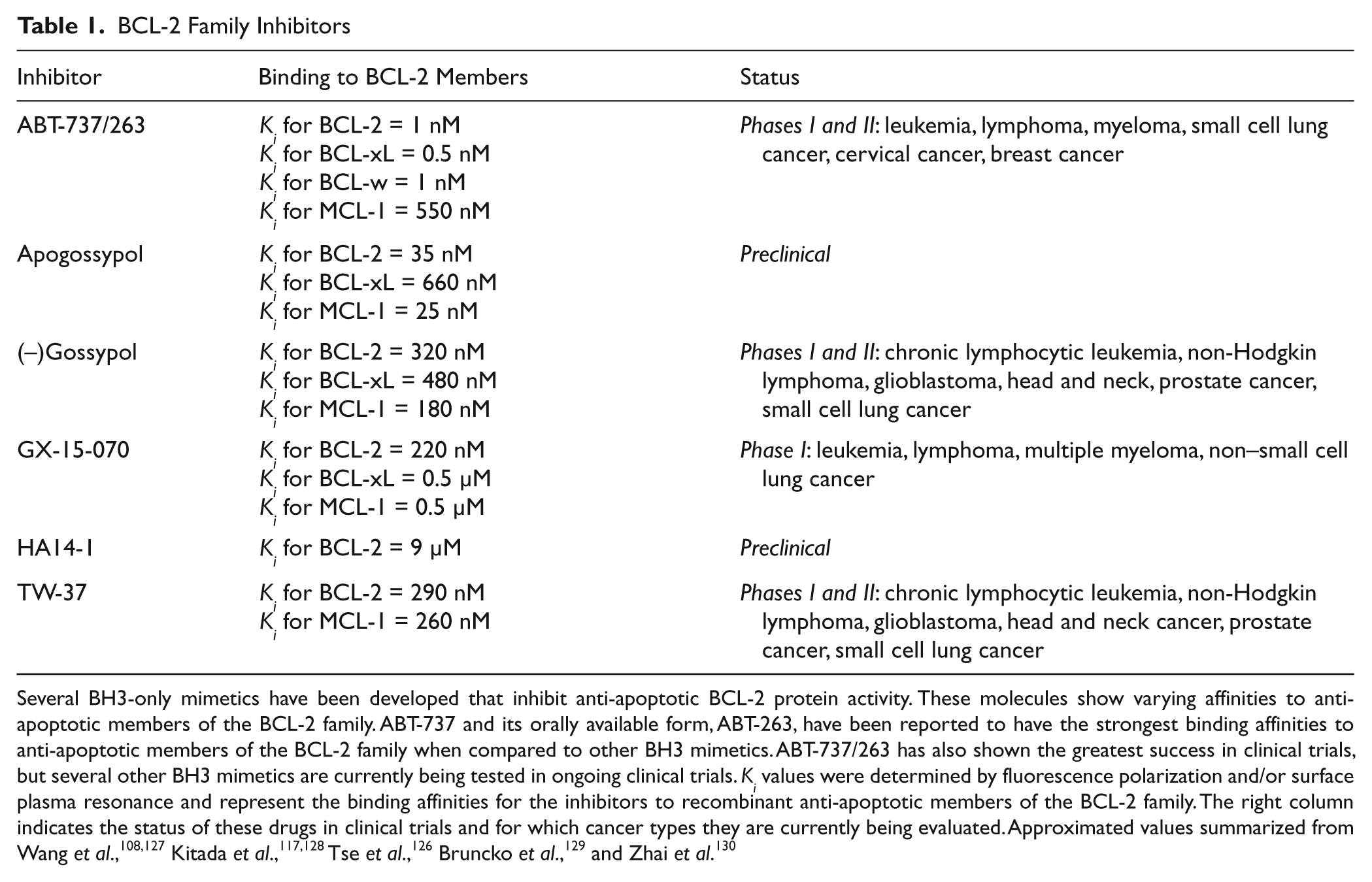
Several BH3-only mimetics have been developed that inhibit anti-apoptotic BCL-2 protein activity. These molecules show varying affinities to anti-apoptotic members of the BCL-2 family. ABT-737 and its orally available form, ABT-263, have been reported to have the strongest binding affinities to anti-apoptotic members of the BCL-2 family when compared to other BH3 mimetics. ABT-737/263 has also shown the greatest success in clinical trials, but several other BH3 mimetics are currently being tested in ongoing clinical trials.
The pro-apoptotic BCL-2 effector proteins
The effectors are globular proteins that were originally characterized to contain BH domains 1 to 3 but recently have been suggested to also contain a BH4 domain.
14
There are two well-characterized effector proteins: BCL-2 antagonist killer 1 (BAK) and BCL-2-associated X protein (BAX), both of which can independently permeabilize the OMM to promote MOMP.15-18 Although BAK and BAX are largely redundant in their ability to affect MOMP, their localizations are different prior to pro-apoptotic stimulation. BAK is primarily localized to the OMM via a trans-membrane region in α9, but a fraction may also be found at the ER. In contrast, BAX is normally cytosolic and must re-localize to the OMM to induce MOMP. During unstressed cellular conditions, BAK and BAX exist as monomeric species, yet following sufficient pro-apoptotic stimulation, BAK and BAX “activate,” leading to their homo-oligomerization at the OMM to enable MOMP. BAK and BAX are essential for MOMP to proceed, as genetic evidence from
The pro-apoptotic BH3-only proteins
Another subclass within the pro-apoptotic BCL-2 members is the BH3-only proteins, which include BAD (BCL-2 antagonist of cell death), BID (BH3 interacting domain death agonist), BIM (BCL-2 interacting mediator of cell death), BMF (BCL-2 modifying factor), PUMA (p53 upregulated modulator of apoptosis), and Noxa. Within this subclass, all members share homology with only a single domain, the BH3, which is defined as L-x-x-x-x-D (L = leucine, x = any amino acid, D = aspartic acid). Nearly all the BH3-only proteins are intrinsically unstructured proteins and become structured only after binding to a globular member of the BCL-2 family. 22 The major exception to the BH3-only proteins is BID, which demonstrates the conserved BCL-2 core structural motif. Despite sharing a BH3 domain, which itself is not well conserved within this subclass, the remaining flanking sequences exhibit little similarity, which may underscore the unique expression, regulation, and function of the BH3-only proteins.
The BH3-only proteins are often subdivided into two groups, depending on their ability to interact with the anti-apoptotic and effector proteins. One subgroup of BH3-only proteins, termed the
The second group of BH3-only proteins is the sensitizers/ de-repressors. Although these proteins lack the ability to directly activate BAK or BAX, they inhibit the anti-apoptotic BCL-2 network to promote MOMP. Sensitizer/de-repressor BH3-only proteins include BAD, BMF, Hrk (Harakiri), Noxa, and PUMA.29-33 These proteins bind and inhibit the hydrophobic groove of anti-apoptotic BCL-2 members, thereby preventing any future associations between the anti-apoptotics and pro-apoptotics (e.g., direct activators or effectors) (Figure 3). For example, when the hydrophobic groove of BCL-2 is unoccupied and BMF is subsequently expressed and bound to BCL-2, BMF has now “sensitized” the cell to future direct activators (and therefore BAK/BAX activation, MOMP, and apoptosis) as the BCL-2·BMF complex cannot neutralize BID or BIM activity. Another situation is when a cell harbors a complex between BCL-2 and a direct activator (e.g., BCL-2·BID); when additional BH3-only proteins are encountered, such as PUMA, they can displace BID to promote MOMP; this is referred to as “de-repression” (Figure 3). The BH3-only proteins demonstrate preferences as to which anti-apoptotic BCL-2 proteins they bind. Although BID, BIM, and PUMA are able to bind all the anti-apoptotic BCL-2 members, BAD, Noxa, and BMF are more selective within the anti-apoptotic BCL-2 repertoire. It is unlikely that one pro-apoptotic BCL-2 protein is sufficient to engage MOMP, but rather the combined interactions between the anti-apoptotic BCL-2 repertoire and the BH3-only proteins establish the threshold and trigger for BAK/BAX activation and MOMP.
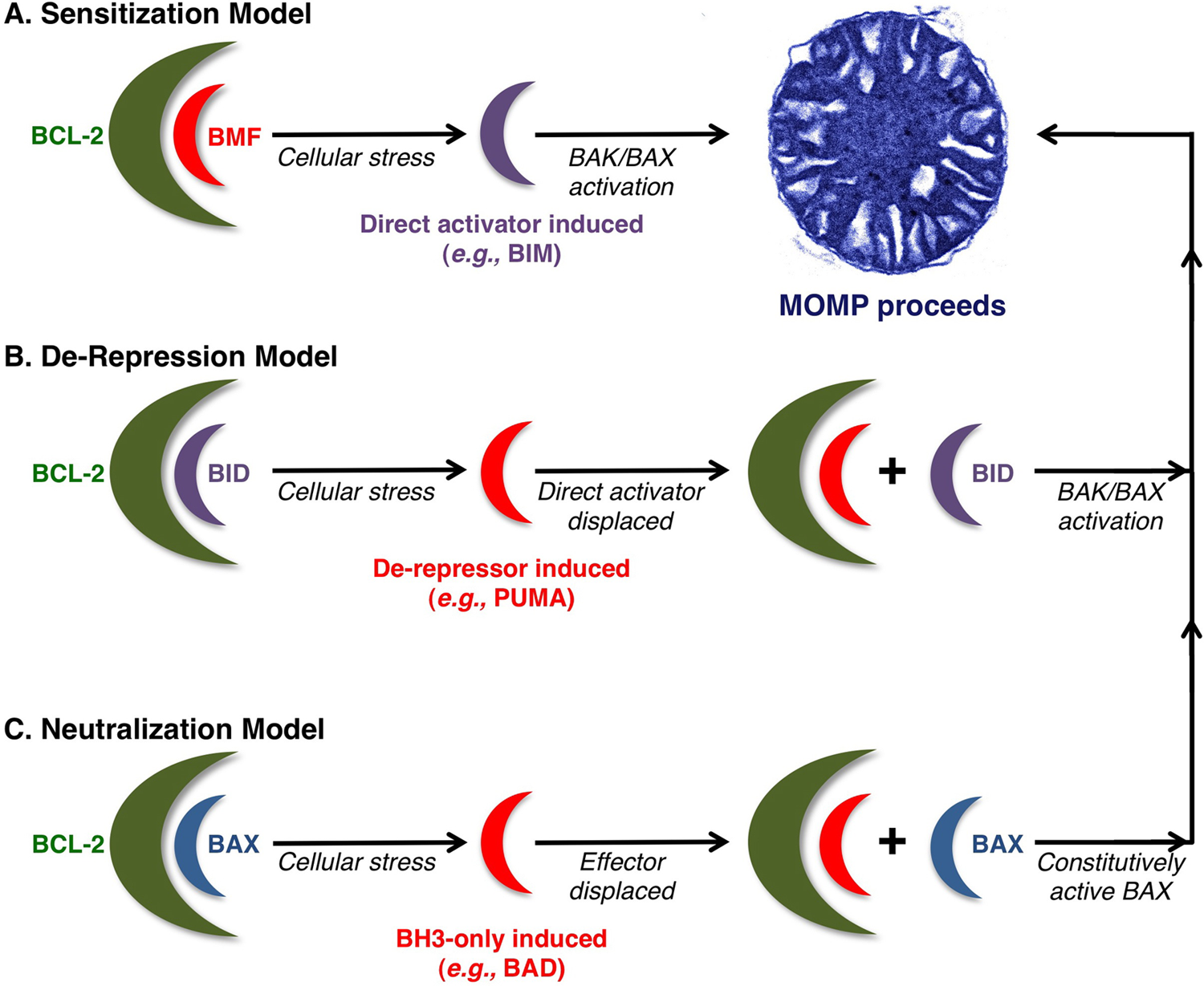
Current models for BH3-only protein functions. (
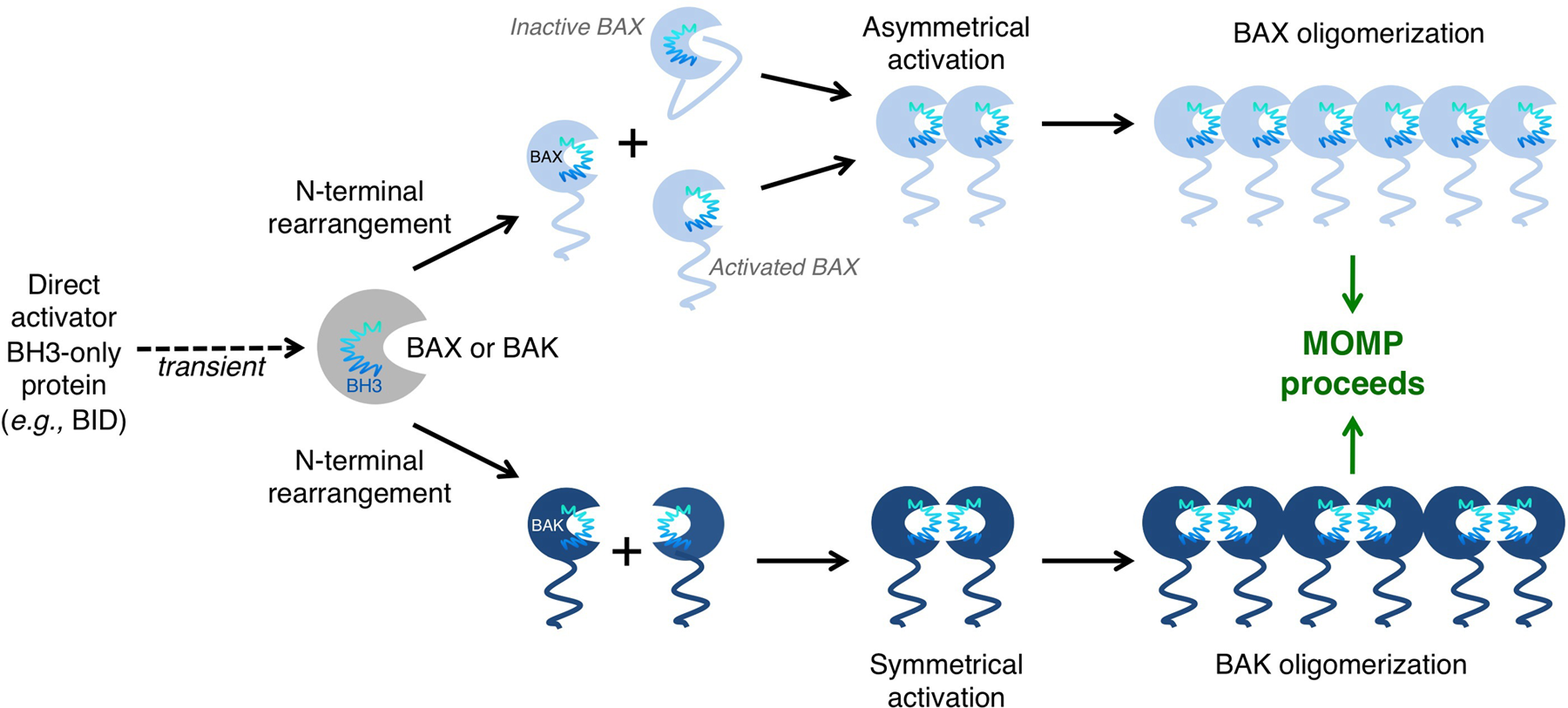
BAK/BAX activation and oligomerization promote mitochondrial outer membrane permeabilization (MOMP). BAK and BAX can be activated through interaction with direct activator BH3-only proteins and the outer mitochondrial membrane. These interactions induce conformational changes that lead to N-terminal rearrangement and the exposure of the BAK/BAX BH3 domain. In the case of BAK, rearrangement leads to the transient exposure of its BH3 domain, which subsequently binds into the hydrophobic groove of another BAK monomer. BAK dimers are able to form multimers through an α6-α6 interaction. High molecular weight species of BAK form pores in the outer mitochondrial membrane (OMM) and induce MOMP. A similar mechanism exists for BAX activation. N-terminal rearrangement of BAX leads to exposure of its BH3 domain, as well as releasing α5, α6, and α9 from the hydrophobic groove. Exposure of these α-helices allows BAX to insert into the OMM. A single BAX molecule can “propagate” this activation signal through binding of its exposed BH3 domain to inactive BAX molecules. Many activated BAX monomers form high molecular weight species once in the OMM to promote MOMP.
It is worth noting that the exact mechanism(s) by which the BH3-only proteins interact within the BCL-2 family network to promote BAK/BAX activation, MOMP, and apoptosis are still somewhat debated. There are several models explaining how the anti-apoptotic and pro-apoptotic BCL-2 family proteins interact to lead to the activation of the effectors, and these are illustrated in Figure 3. Despite these controversies, one consistent and unquestionable aspect is the indispensable role for the BH3-only proteins in regulating MOMP and apoptosis.
Activation of BAK and BAX
When coordinated direct activator and de-repressor/sensitizer BH3-only protein function has superseded the anti-apoptotic BCL-2 repertoire, the cellular response is to engage BAK and/or BAX activation, MOMP, and apoptosis. The activation of BAK and BAX is ultimately regulated through complex protein-protein interactions between the direct activator BH3-only proteins and BAK/BAX at the OMM, leading to its disruption. Recently, a better understanding of the mechanisms leading to BAK/BAX activation has emerged. As previously mentioned, BAK constitutively localizes to mitochondria, via α9 insertion within the OMM. Once activated by BID or BIM, a conformational change leads to the rearrangement of the amino-terminus, as well as subsequent exposure of the BAK BH3 domain. This exposed BH3 domain subsequently binds the hydrophobic groove of another BAK monomer, thereby forming a BAK “groove-to-groove” dimer. This dimer is able to form multimers with other BAK dimers through an α6-α6 interaction (Figure 4). 34
As discussed, BAX shuttles between the cytoplasm and mitochondria in healthy cells, which maintain mitochondrial network dynamics.35,36 Recent literature suggests that BCL-xL plays a role in BAX retro-translocation from the OMM to the cytoplasm, thereby preventing mitochondrial localization and activation of BAX.
37
However, in the presence of an apoptotic stimulus, the binding of direct activators to BAX induces amino terminal rearrangements and subsequent BH3 exposure, promoting the release of α9 from the hydrophobic groove, mitochondrial localization, and permeabilization of the OMM. It is thought that BAX α5, α6, and α9 insert into the OMM and that the activation of a single BAX molecule can propagate the activation of other BAX molecules via the exposed BAX BH3 domains to drive BAX oligomerization (Figure 4).
38
A recent publication by Kim
The BH3-Only Proteins: A Closer Look
Cellular stress leads to the transcriptional and posttranscriptional activation of distinct BH3-only proteins, which in turn inhibits the anti-apoptotic repertoire directly, leading to BAK and BAX activation. Although the BH3-only proteins are indispensable for the mitochondrial pathway of apoptosis, evidence suggests they also regulate other cellular pathways such as metabolism, inflammation, and autophagy (for a comprehensive review, see Chipuk
The BH3-Only Proteins and Tumor-Promoting Signaling Pathways
BAD (BCL-2 antagonist of cell death)
This BH3-only protein was originally identified by Yang
BAD-deficient mice are developmentally normal and viable, but the mice spontaneously develop diffuse large B cell lymphoma within 15 months of life.
49
The observation that the
BID (BH3 interacting domain death agonist)
BID is the only BH3-only protein that is mainly activated through the extrinsic death receptor pathway. This occurs via caspase-8-mediated cleavage generating the active molecule termed
BIM (BCL-2 interacting mediator of cell death)
Identified by O’Conner
At the transcriptional level, BIM expression is regulated by the forkhead transcription factor, Foxo3a, which binds and activates the
Decreased BIM expression is reported in several human malignancies, including B cell lymphoma and colon cancer, and is associated with poor patient survival.76,77 Interestingly,
BMF (BCL-2 modifying factor)
The BH3-only member, BMF, was originally identified by Puthalakath
The role of BMF in tumorigenesis still remains to be fully elucidated, but there have been several reports suggesting that the protein may play a role in suppressing tumor development. Using the
PUMA (p53 upregulated modulator of apoptosis)
Shortly following its initial discovery, PUMA was identified as a transcriptional target of p53 through global gene expression profiling in a p53-inducible cell line, SAOS-2-p53.30,84,94 PUMA was also identified by a Y2H screen as a BCL-2 binding partner and was thus named BBC3 (BCL2–binding component 3). Once transcriptionally activated, the
PUMA possesses potent pro-apoptotic activity as a result of its ability to directly interact with all anti-apoptotic members of the BCL-2 family. PUMA mainly localizes to mitochondria (since it is bound to the anti-apoptotics) and functions to promote BAK and BAX activation through de-repression and sensitization of the anti-apoptotics sequestering them (Figure 3).26,91,92 Interestingly, recent reports suggest that PUMA can also directly activate BAK and BAX through transient binding that leads to their homo-oligomerization, although the results are in contrast to numerous reports.39,91-93 Despite playing such an indispensable role in p53-dependent and p53-independent apoptosis,
Noxa (Latin for “damage”)
Noxa was originally identified as a phorbol myristate acetate responsive gene from an adult T cell leukemia library, but the function of the protein remained elusive.
100
A decade after its initial identification, Noxa was defined as a p53-inducible gene that encoded for a BH3-only protein that played a role in DNA damage and hypoxia-induced apoptosis.31,101 Even though Noxa does play a role in p53-mediated apoptosis, it is less essential than PUMA, possibly due to Noxa having a higher selectivity for the anti-apoptotic BCL-2 proteins (i.e., Noxa only antagonizes the activity of MCL-1, compared to PUMA, which inhibits all the anti-apoptotic BCL-2 proteins). Similarly to PUMA, Noxa can also be activated through p53-independent mechanisms. Under hypoxic conditions, the basic helix-loop-helix transcription factor HIF-1α is released from sequestration in the cytoplasm and re-localizes to the nucleus, where it transcriptionally activates the expression of several genes, including
The BH3-Only Proteins and Chemotherapy
It is important to note that although alterations in BH3-only protein expression have been reported in several cancers, such as colon and lung cancer, this is not a common phenotype. In contrast, the regulation of anti-apoptotic members is more widely demonstrated in many cancer types, including those that show no aberrant changes in BH3-only expression. This may be a mechanism selected for by tumor cells to block the activity of several BH3-only proteins and BAK/BAX, along with additional metabolic and survival advantages conferred by increased anti-apoptotic BCL-2 protein expression. Based on these observations, anti-apoptotic members of the BCL-2 family are attractive targets for the treatment of numerous cancers. As will be discussed below, several strategies have been adopted to develop such therapies. One of the most successful attempts to create such therapeutics is evident in BH3 mimetic molecules that inhibit anti-apoptotic BCL-2 protein activity, thereby enhancing BH3-only function and promoting MOMP.
One of the first successful attempts to pharmacologically regulate the BCL-2 family was using Oblimersen, an 18-mer antisense oligonucleotide designed to target the first 6 codons of
HA14-1 is a compound identified via
In 2007, Gemin X published the first pan anti-apoptotic BCL-2 protein inhibitor, Obatoclax.
111
This was one of the first compounds designed that not only inhibited BCL-2 and BCL-xL but MCL-1 and presumably other anti-apoptotics as well—possibly the reason it conferred low resistance in patients. Obatoclax entered into clinical trials and was found to be well tolerated in patients with hematological and myeloid malignancies.112,113 However, the exact mechanism of action of this compound has recently been called into question by several studies. Similar to reports regarding HA14-1, Konopleva
BH3 mimetics have also been derived from natural molecules, as is the case with gossypol. It is a natural polyphenol derived from the cottonseed plant. It was historically used as a male contraceptive, but due to reports of permanent infertility as well as high toxicity, further studies in the context of contraception were abandoned. Following this, several labs began studying the potential effects of gossypol on cancer, and it was subsequently found that the (–) enantiomer exhibits stronger apoptotic effects compared to the (+) enantiomer. 116 As a BH3 mimetic, (–) gossypol is able to bind BCL-2, BCL-xL, and MCL-1 although with affinities that are much lower than of the BH3-only proteins to these proteins. Gossypol derivatives have been generated, and these include Apogossypol, TM-106, and TW-37. These derivatives, TW-37 in particular, exhibit stronger binding affinities to the anti-apoptotics while also conferring less toxicity.117-119 TW-37 was the first BCL-2 inhibitor to enter the clinic. It has shown promise in inducing apoptosis and overcoming resistance in cases of B cell lymphoma, chronic lymphocytic leukemia, and multiple myeloma. 120
Despite these many attempts to generate potent BH3 mimetics, none have proven as effective at binding the anti-apoptotics as the BH3-only proteins themselves. Given the importance of the BH3 domain in the interaction between the pro-apoptotic proteins and the anti-apoptotics, Letai
ABT-737, ABT-263
The most successful and potent BH3 mimetic compound generated to date is a compound developed by Abbott, termed ABT-737. In 2005, Oltersdorf
Perspectives
Our discussions centered on how the BCL-2 family of proteins functions to regulate tumorigenesis, with a particular emphasis on BH3-only protein signaling. As the BCL-2 family defines the balance between life and death, a modern understanding of the cellular mechanisms that control its function is crucial to establish how stressed cells undergo apoptosis—and how cancer cells resist pro-apoptotic signals to ensure survival. Throughout, we suggest that numerous BCL-2 family members functionally cooperate to maintain cellular survival, and similar multipartner interactions are essential for apoptosis to proceed. These interactions appear to be directly regulated by numerous signaling cascades, many of which are commonly abrogated in cancer cells. From this, it comes as no surprise that inhibiting the cellular signaling pathways that promote survival along with lowering the threshold for apoptosis to proceed represents the ideal regimen for pharmacologically targeting cancer cells. The development of small-molecule inhibitors to the anti-apoptotic BCL-2 proteins is a milestone in modern chemotherapeutic strategies, and complementing these drugs with specific drugs to silence pro-survival pathways is already showing signs of clinical success. As we further dissect the mechanisms of the BCL-2 family and apoptotic pathways, novel therapeutic targets will certainly emerge and it is hoped provide further benefits to patients.
Footnotes
Acknowledgements
We apologize for not being able to discuss and cite all the relevant literature due to space restrictions. We would like to thank members of the Chipuk Laboratory for useful discussions and feedback on this review.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The laboratory is funded by the National Institutes of Health [grant R01CA157740] and is supported in part by a research grant from the March of Dimes Foundation [5-FY11-74].
