Abstract
Latin America hosts extraordinary biological diversity but remains underrepresented in global biomedical research, underscoring the need for robust biobanking infrastructures. This work provides an updated snapshot of Chilean biobanks, based on a national survey exploring their current capacities and challenges. Nine active biobanks were identified across 5 of Chile’s 16 regions, the majority concentrated in Santiago. Collectively, they store over 640,000 biospecimens from nearly 49,000 participants, predominantly oncological. While standardized protocols for sample management are broadly implemented by Chilean biobanks, data management practices are not yet well-developed, as only a few centers have adopted internationally recognized standards. Governance structures vary considerably and often lack formal written documentation. Financial sustainability relies mainly on institutional support, competitive grants, and modest cost recovery. Although Chilean biobanks contribute to research and training, measuring productivity remains challenging due to underreported acknowledgments and limited post-transfer traceability. Overall, our analysis suggests a bottom-up development of Chilean biobanks in the absence of dedicated legislation or strategic governmental policies. This overview shows that Chile’s biobanks hold considerable potential for strengthening translational research and health equity, particularly if further support enables expansion into underrepresented regions. By integrating these infrastructures into higher education, clinical care, and broader regional collaborations, biobanks can help leverage Chilean genetic diversity and address health disparities. With greater governmental prioritization, a cohesive regulatory framework, and collaboration as a key strength, biobanks could enhance interaction with global networks and further strengthen Latin America’s overall contribution to biomedical innovation.
Keywords
Introduction
In recent years, scientific and technological advances, particularly in -omics and big data analysis, have expanded our understanding of biological variability and improved probabilistic predictions. 1 These developments are driving a shift from reactive medicine to 4P medicine: predictive, preventive, personalized, and participatory, aimed at preserving health and delivering the right treatment to the right person.2,3
Addressing these goals in translational research requires overcoming challenges of reproducibility and reliability,4–6 which depend on access to large numbers of comparable samples and data generated under harmonized, high-quality, and standardized protocols regularly updated.7,8 These logistical requirements and expertise often exceed the capacity of individual laboratories.
As widely acknowledged in scientific literature9–13 and mainstream media,14,15 biobanks have played a pivotal role in advancing basic and translational biomedical research over the past two decades.
Yet, gaps in population diversity and the limited development of biobanks in Latin America contribute to scientific underrepresentation and the inequitable application of biomedical solutions based on nonlocal data.16–18 While progress has been made in reducing Eurocentric bias—especially for Asian populations—Latin American groups remain underrepresented.19,20 The absence of biobank infrastructure also limits access to international funding, as global research agendas are progressively shaped by large-scale consortia that rely on harmonized, diverse datasets and require standardization, data interoperability, and robust governance. These missing capacities are both cause and consequence of scientific marginalization, delaying Latin America’s integration into global research. Addressing this requires updated, detailed regional diagnostics.
A recent systematic review reported 44 biobanks in Latin America, mostly of them in Chile, Mexico, Argentina, and Brazil. However, it also highlighted the limited literature on the subject, with only 29 relevant publications identified. 21 This study aims to help fill that gap by providing an overview of Chile’s current biobanking landscape.
Ethical and Legal Framework
International ethical guidelines for human research mandate the guarantee of dignity, autonomy, and privacy for participants. Accordingly, Chilean biobanks adhere to the Declaration of Helsinki, 22 the Council for International Organizations of Medical Sciences (CIOMS) Ethical Guidelines, 23 and the Declaration of Taipei, 24 recognizing their role as social guarantors of rights in the donation, custody, and transfer of human biological material and data. This responsibility goes beyond specimen and data storage, requiring sustained efforts to ensure ethical, efficient, and socially beneficial use.25–27 Biobanks must therefore build competence, install capacity, and operate under international standards that guarantee quality, accountability, transparency, and access in compliance with Findable, Accessible, Interoperable, and Reusable (FAIR) principles.28–30 Accordingly, Chilean biobanks follow international technical guidelines, including International Society for Biological and Environmental Repositories (ISBER) Best Practices, 31 the Common Minimum Technical Standards for Biobanks, 32 and ISO 20387:2018, 33 to ensure security and traceability. However, full implementation is still constrained by challenges in infrastructure, human resources, funding, and essential services.
In Chile, biobanks lack specific legislation, adhering instead to general regulations that, while applicable, are insufficient to foster biobank development under a clear legal framework adapted to the local context. Law No. 20.120 34 (2006) regulates research involving human subjects and the genome, and prohibits cloning. It outlines general principles for handling and storing biological samples, operationally complemented by Supreme Decree No. 114 of 2011. 35 However, it does not establish standards for biobank operation.
Law No. 19.628 on the Protection of Private Life 36 generally regulates personal data processing but has been widely criticized for its inadequate protection of genomic and biometric data, particularly in the digital era of biomedical research. Since 2017, a legislative reform has been under discussion to strengthen regulation by incorporating new categories of sensitive data (including for research purposes) and establishing a supervisory Data Protection Agency. The new Law No. 21.719 will take effect in December 2026, 37 bringing Chilean regulation closer to international standards, such as the European Union’s General Data Protection Regulation (GDPR). 38
Other laws, such as Law No. 21.258 (National Cancer Law), 39 Law No. 19.970 (National DNA Registry System), 40 and Law No. 20.584 (Rights and Duties of Individuals in Relation to Healthcare Actions), 41 tangentially impact biobanking activities. The first promotes cancer research, human capital training, and establishes the National Cancer Registry. The second, though focused on forensic investigations, sets standards for collecting biological samples, potentially adaptable to broader scientific contexts. The third regulates research involving individuals unable to provide informed consent, such as minors, unconscious people, or those with neurodegenerative diseases.
Within this legal context, which lacks specific provisions for access, governance, usage, or international transfers, biobanks rely on institutional material transfer agreements, international standards, and best practices. Yet, the weakness of the regulatory framework constrains both the consolidation of the role of Chilean biobanks and their integration into global research efforts.
Since 2014, academia, government agencies, and private organizations have identified regulatory gaps, developed proposals, implemented harmonization strategies aligned with international standards and promoted a national biobank network. Most of these efforts remain stalled in consultative phases, lacking the political momentum to become public policy, reflecting biobanking’s low priority in Chile’s legislative agenda.
General Description of Chilean Biobanks
In this context, Chilean biobanking evolved in a sometimes-discontinuous way but nonetheless developed across various institutions, driven by researchers and professionals who took the initiative. The first biobank in Chile—the Tumor Biobank of the Universidad de la Frontera—was established in 2003 in Temuco. Since then, other initiatives have emerged but faced irregular activity due to funding constraints, institutional transitions, or leadership changes. Some, such as the Clínica Alemana de Santiago, Complejo Asistencial Hospital Sótero del Río, Hospital del Salvador, Hospital Luis Tisné, and Hospital San Borja Arriarán, eventually ceased functioning when project-based funding ended. In several cases, no updated information on the fate of samples could be retrieved, though some may remain stored in hospital laboratories. An exception is San Borja Arriarán, whose materials were formally transferred to the Biobanco de Tejidos y Fluidos de la Universidad de Chile. At Sótero del Río, a biobank project was fully planned but never implemented, and the allocated funds were ultimately redirected.
This fragmentation has hindered the ability to maintain an accurate and updated snapshot of the national biobanking landscape, as reflected in the differences reported across previous studies.21,42
To address this gap, this study aims to identify and characterize currently active Chilean biobanks. The analysis, based on institutional self-reports and publicly available information, was ethically considered exempt from ethics committee review in accordance with Chilean regulations and international guidelines, as it did not involve human participants, patient data, or biological samples. Methodologically, since no official registry exists, a Google Forms questionnaire was developed to gather updated information. The survey included 79 questions grouped into 10 sections (Table 1), covering organizational aspects.
Thematic Areas of the Biobank Characterization Form in Chile

The form and study invitation were sent via email to directors or coordinators of centers that, to our knowledge, were active. This included biobanks recruiting participants and those engaged in institutionally supported implementation processes, such as developing protocols or governance. Centers were identified through ongoing collaborations, prior interactions, and internet or publication searches. Given the absence of a national registry and the self-reported nature of data, some existing or emerging biobanks may not have been captured by our approach. Of the 16 centers invited, 7 did not complete the form: 2 were no longer operational, 3 expressed interest in future development without a current institutional project, 1 held sample collections but did not identify as a biobank, and 1 did not respond. The remaining 9 centers replied, declared themselves active biobanks, and completed the form. Their names, institutions, abbreviations, location, and biobank type are listed in Table 2.
Active Chilean Biobanks in 2025
Figure 1 illustrates the geographical distribution of Chilean biobanks. The Ministry of Science, Technology, Knowledge, and Innovation groups the 16 regions into 5 macrozones 43 : North, Central, Central-South, South, and Austral. Active biobanks are found in only five regions, with very limited presence outside the Central macrozone. The Metropolitan Region, especially Santiago, hosts most biobanks (56%, n = 6), which are also the most consolidated in staff, infrastructure, and collections (icons in Fig. 1), highlighting an important geographic gap. Considering this strong centralization, the presence of biobanks in remote regions is noteworthy, though still at an early stage.
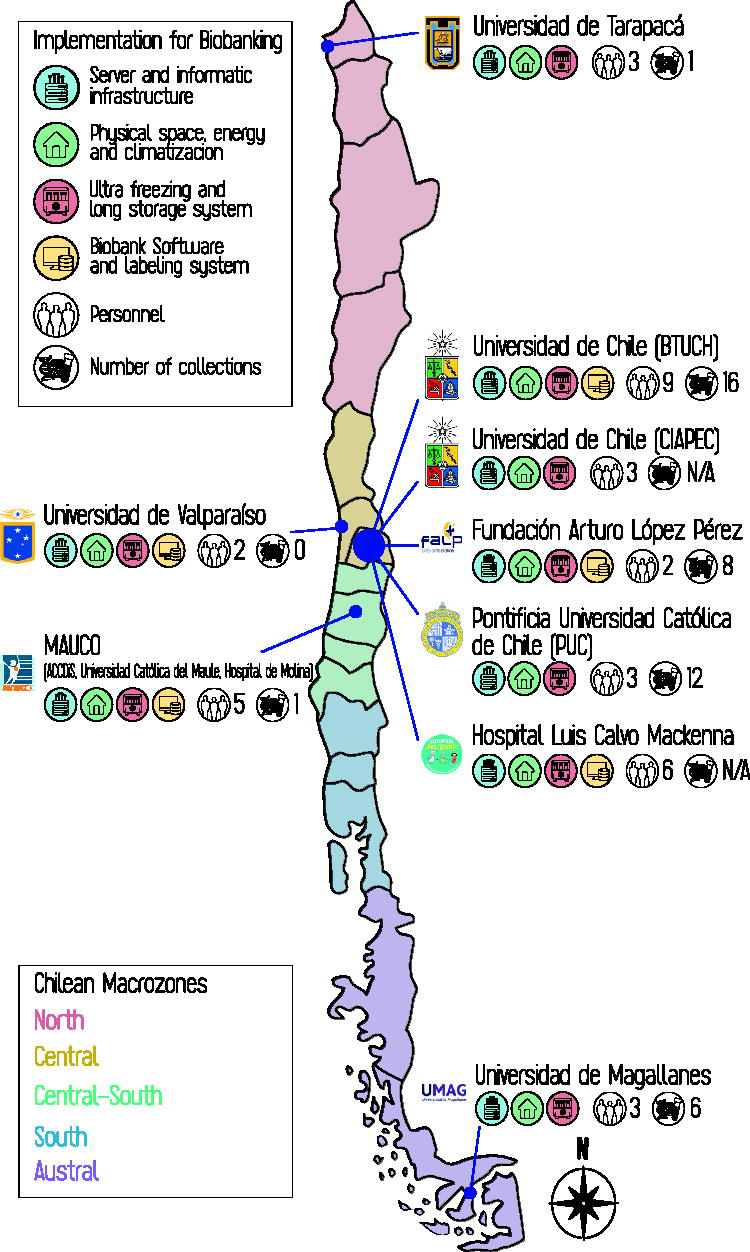
Geographic distribution, resources, and capabilities of Chilean biobanks. The current landscape of Chilean biobanks (2025) is shown, highlighting their geographic location along with details of their resources, capabilities, and number of collections, represented through colored icons (legend in the upper left corner). The numbers indicate the quantity of personnel and collections, respectively. In the case of CIAPEC-BB and HLCM-BB, their biobanks reported having samples, but they are not organized into formal collections.
A general analysis provides initial insight into the current landscape. In this study, a collection refers to a set of biological specimens and associated data organized around a specific research objective, pathology, or population group. A single biobank may host one or multiple collections depending on its focus, internal organization, and operational trajectory.
Nationally, there are 44 biobank collections, with 16 (36.4%) at BTUCH. Along with the 12 (27.3%) at PUC-BB, these 2 hold 63.7% of the country’s collections. Including the eight (18.2%) at FALP-BB, the three account for 81.9% nationwide. This centralization, though varying across biobanks, is also observed in the number of samples and participants, analyzed later (see the section “Collections, Samples, and Data Practices in Chilean Biobanks”). Personnel distribution is somewhat less concentrated: BTUCH, HLCM-BB, and MAUCO-BB employ 55.5% of biobank staff. Still, only 36 people work directly in biobanking nationwide.
Regarding operational infrastructure, all biobanks have adequate physical space, ultra-freezing storage, and general information technology infrastructure. However, the lack of biobank-specific software and labeling systems in four of nine centers is the main implementation challenge. Unlike other resources adaptable from clinical or laboratory settings, these systems are highly specialized, often expensive, and require targeted investment and technical expertise, which may explain their limited implementation.
In summary, the territorial distribution of biobanks in Chile reveals a strong concentration, mirroring the broader centralization of the country’s scientific infrastructure. At the same time, it highlights the need to expand into other regions to address diverse local health challenges. For example, lung cancer is a major concern in the North, while gastric and gallbladder cancer prevail in the South. 44 This disparity underscores the need to mitigate the concentration of research and health investment that exacerbates inequalities in access to biomedical resources across the country. 45
Figures 2 and 3 provide details on Chilean biobanks, including general and organizational characteristics. Most are housed within universities, with only two in health care facilities. A similar distribution applies to public–private affiliation (Fig. 2A). Most biobanks are identified as project-driven, meaning that at least some collections were established in responding to specific research project requirements. This often implies that informed consent restricts sample use to the project’s original objectives, but not always: projects and biobanks can agree to set priority conditions for use or create parallel collections allowing future utilization beyond the original project. These policies promote biobank development by leaving samples, data, and installed capabilities within them, while enabling researchers to benefit from biobank expertise in sample and data management. Nevertheless, most biobanks (77.7%) operate under a broad informed consent model, either alone or alongside project-specific consent, with only two operating solely under the latter (Fig. 3F).
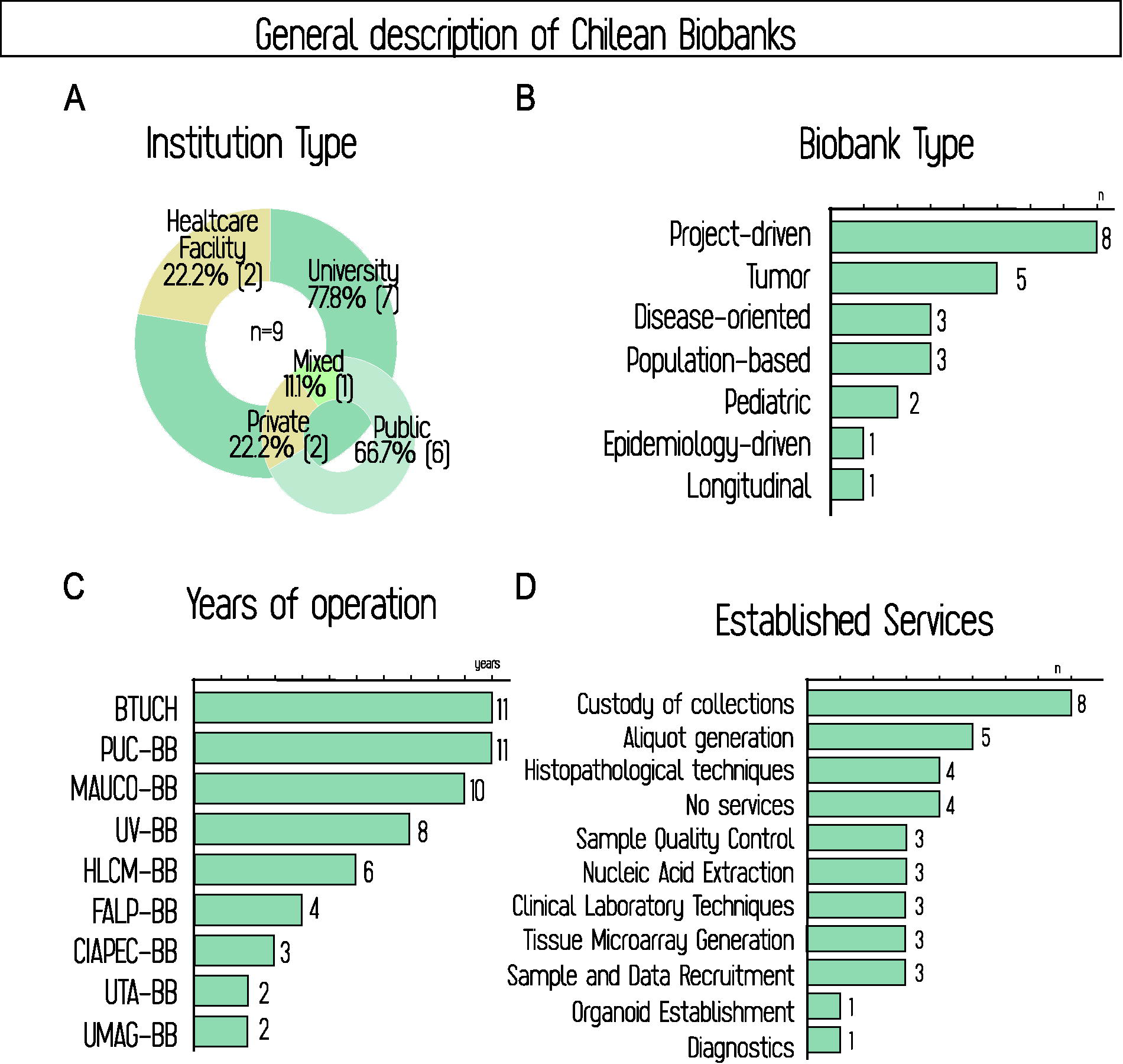
General description of Chilean biobanks, including institution type
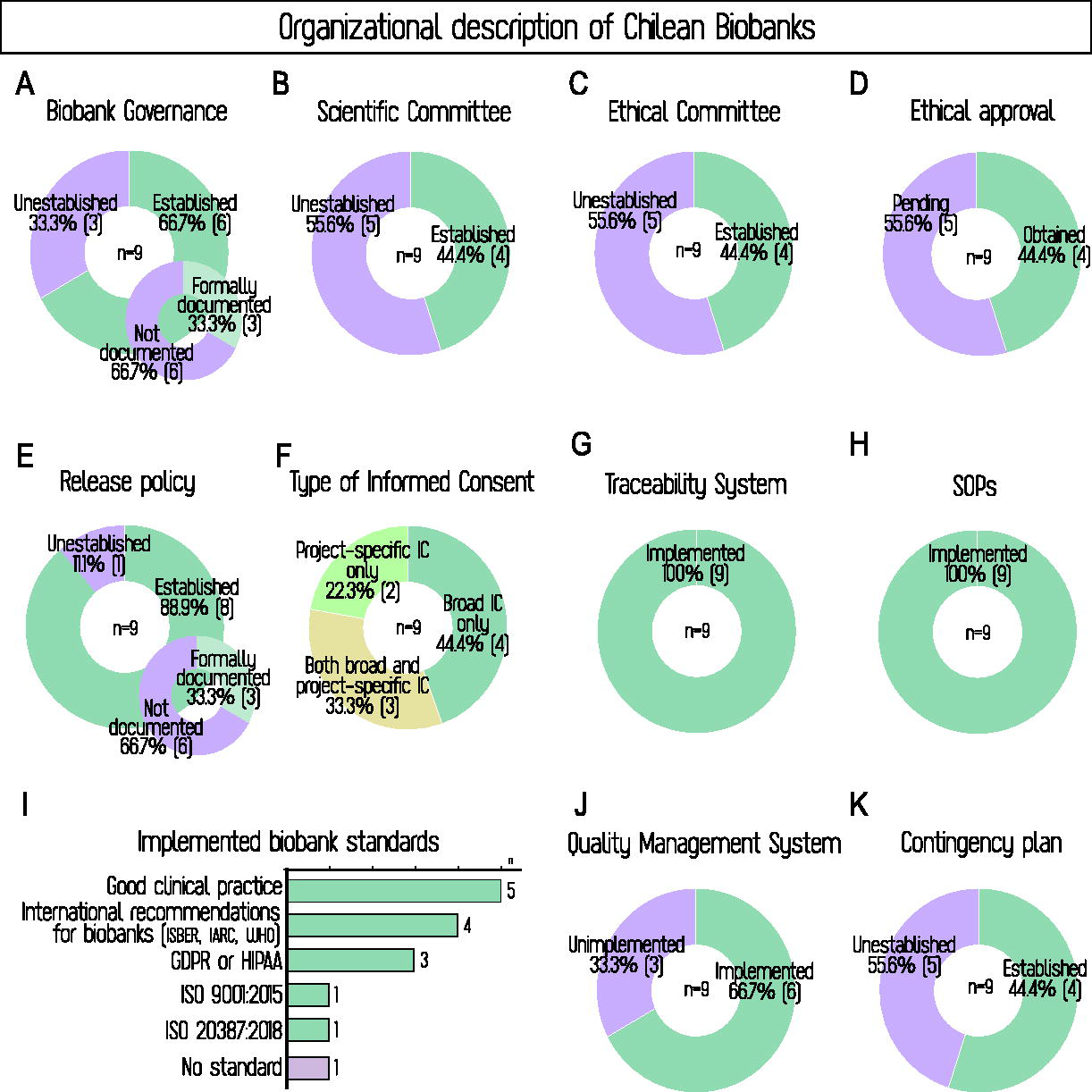
Organizational overview of Chilean biobanks. Panels illustrate governance structures
Aside from specific projects, Chilean biobanks are primarily disease-oriented, with a predominance of tumor biobanks. While population-based cohorts exist at CIAPEC-BB and are emerging in Magallanes, the only fully established population biobank is MAUCO-BB, a longitudinal study following 9462 adult residents of Molina since 2014, aimed at analyzing the natural history of chronic diseases in this agricultural community 46 (Fig. 2B).
Most Chilean biobanks have operated for less than 10 years, except BTUCH, PUC-BB, and MAUCO-BB, which reached this age in December 2024 (Fig. 2C). This is relevant when considering that four out of nine (44.4%) do not offer external services, while others have gradually developed some, mainly custodianship and aliquoting. Only the most established biobanks offer advanced services, including molecular and histological procedures, as well as participant recruitment. Organoid cultures remain emerging, currently implemented by only one biobank nationwide (Fig. 2D).
Regardless of services offered, biobank maturity depends on organizational structure and the integration of ethical, legal, and social considerations (ELSI) throughout operations. A clear structure with policies and procedures formally documented in regulations and records is essential.47,48 The most important organizational document is the governance, which defines formal and informal policies, processes, and structures. It includes the role of scientific and ethical committees, the organizational chart, mission, vision, and mechanisms to ensure quality, transparency, accountability, and oversight.28,29
Six surveyed biobanks reported having governance, of which three had it documented—meaning that one-third of Chilean biobanks have a formal written structure, another third have undocumented frameworks, and the remaining third lack governance entirely (Fig. 3A). Figure 3B–D shows that about half of the biobanks report a scientific committee, an advisory ethics committee, and formal ethical approval. These elements do not consistently overlap across the same set of biobanks, despite similar overall proportions. Of the three biobanks with documented governance, two had both committees and approval, while the third had none. Additionally, two biobanks without documented governance reported a scientific committee and ethical approval, but no ethics committee. In contrast, all biobanks but one have policies for releasing samples and data, although only one-third have them formally documented (Fig. 3E). These results highlight the heterogeneity in how governance structures and policies have been implemented across Chilean biobanks.
The unconsolidated organizational maturity is also reflected in the extent of implementation of biobank management standards. All centers reported implemented traceability systems and standard operating procedures (Fig. 3G and H), and nearly all adhere to international biobanking standards (Fig. 3I). However, implementation drops to two-thirds for quality management systems and to half for contingency plans (Fig. 3J and K).
Overall, Figures 2 and 3 show Chilean biobanking as a landscape of heterogeneous development. Despite progress in structure, traceability, and services, challenges persist in governance, international standards, and policy documentation. This reflects an ongoing process toward a more robust and harmonized system.
Collections, Samples, and Data Practices in Chilean Biobanks
Beyond geographic, infrastructural, and organizational features, we analyzed collections, sample types, their origin and use, and management systems to assess biobank heterogeneity, functionality, interoperability, and stakeholder engagement in biomedical research.
The top panel of Figure 4 shows data on collection type, participant numbers, and biospecimens. Figure 4A illustrates the diversity of collections—referring to the variety of purposes, pathologies, and participant groups—and their number per type. This usually corresponds to the number of biobanks holding each type, with some exceptions: PUC-BB reported separate collections for cholelithiasis and gallbladder cancer, and for colon and rectal cancer, which were grouped together in the figure. In total, 18 distinct collection types were reported.
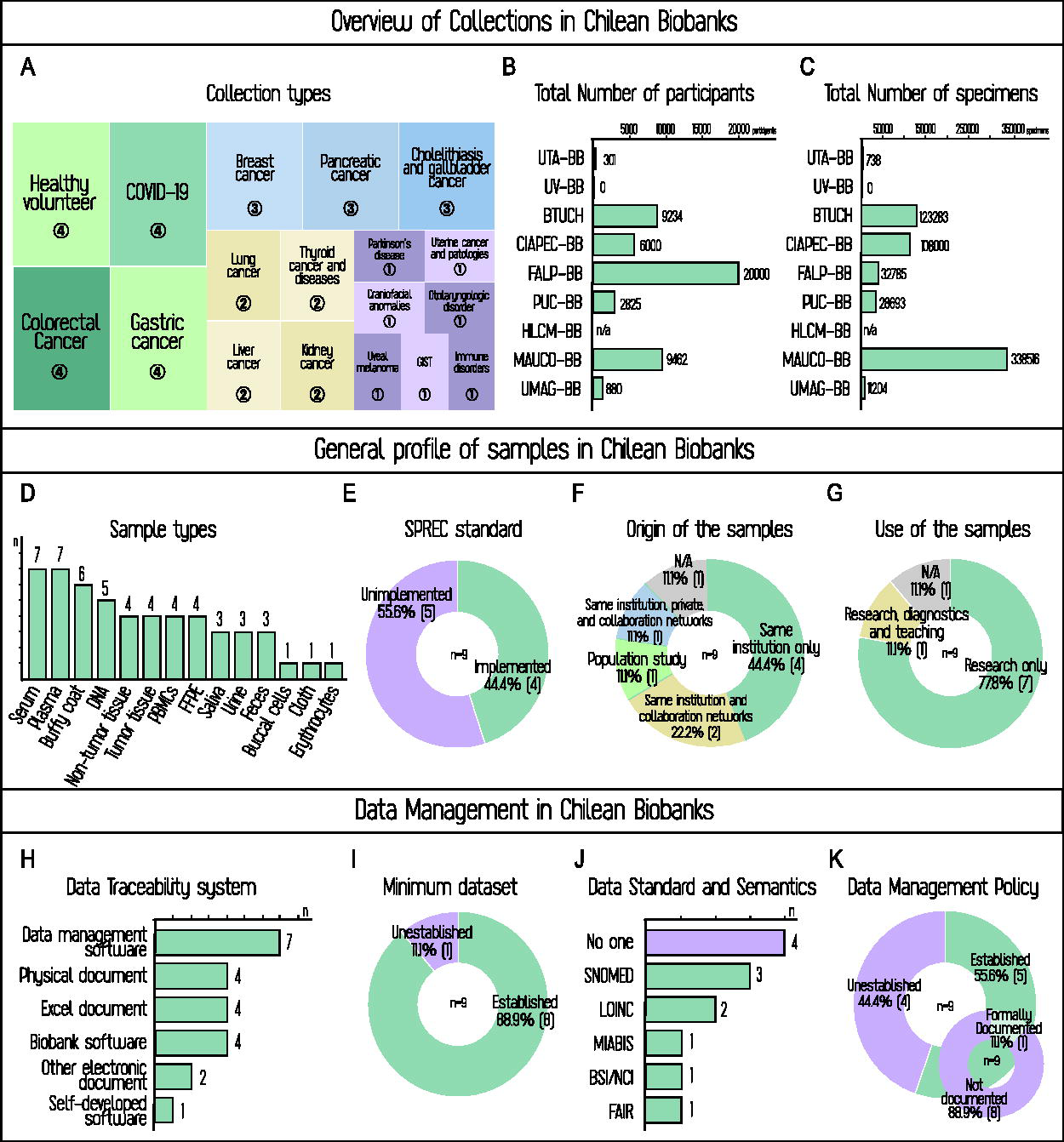
Operational overview of Chilean biobanks. Top panel. General overview presenting data on collections, participants, and biospecimens generated by Chilean biobanks.
Several COVID-19 collections stem from the collaborative creation of Chile’s first national biorepository, developed under biobank standards during the pandemic. 49 BTUCH, together with the Universities of Tarapacá and Magallanes, participated in this self-organized, multicenter initiative, which prompted the latter two to transition into full biobanks and six additional centers to explore similar transitions. Of the four declared COVID-19 collections, three belong to this network harmonized for interoperability and are now available through the C19-GenoNet Biobank Network Catalog—Chile’s first interoperable, multicenter shared catalog (https://redcovid.uchile.cl).
Oncological collections are the most frequent after COVID-19, with 12 out of 18 (66.6%) being cancer-related. Their distribution generally aligns with the most common cancers in Chile, based on 2024 Center for Cancer Prevention and Control (CECAN) and 2022 Global Cancer Observatory (GLOBOCAN) data.50,51 However, relevant gaps remain: prostate and testicular cancers (second and eighth most common) are not represented. Conversely, thyroid cancer—outside the top 10—is represented by two collections; at least one includes other thyroid pathologies, which are more common in the population. 52 The prominence of gallbladder and biliary tract cancer collections, along with those focused on cholelithiasis, is particularly notable. Their presence reflects the importance of ongoing research in this area, as Chile has the second-highest global incidence of gallbladder cancer, with cholelithiasis recognized as a major risk factor.53–56
The four non-oncological collections (22.2%) include Parkinson’s disease, craniofacial anomalies, and immune and otolaryngologic disorders. This distribution reflects a predominant oncological focus, alongside expanding research interests.
Figure 4B and C shows participant and biospecimen totals per biobank, revealing expected variation reflecting their historical and operational peculiarities. Nationwide, 48,972 participants and 701,015 biospecimens (including aliquots and sample types) were recorded. The first four biobanks (MAUCO-BB, BTUCH, PUC-BB, FALP-BB) hold 84.8% of participants and 74.6% of biospecimens, confirming operational and geographic centralization. On average, Chilean biobanks store 14.3 specimens per donor. These ratios may also be influenced by the disease focus, as certain conditions tend to require specific specimen types, but are equally shaped by biobank characteristics and the type and design of each collection. MAUCO-BB, a longitudinal population biobank, has the highest ratio (35.8), reflecting repeated sampling points per participant. In contrast, BTUCH and PUC-BB, with diverse collections, report values closer to the national average (13.4 and 10.2, respectively).
Among the biobanks with lower ratios, UTA-BB and FALP-BB report values of 2.5 and 1.6, respectively. Given that FALP-BB is a well-established biobank and UTA-BB an emerging one, their similarly low specimen-to-donor ratios may reflect different causes: a strategic operational model for FALP-BB and capacity constraints for UTA-BB.
Two notable cases are CIAPEC-BB and UV-BB. CIAPEC-BB’s high participant and biospecimen numbers align with its cohort-based model and exclusive use of project-specific informed consent (Fig. 3F). Despite being a young biobank, it has implemented governance structures, pre-analytical quality standards (Fig. 4G), and other organizational milestones, though it has yet to adopt international biobanking standards (Fig. 3I). In contrast, UV-BB, although not recently established, has struggled to expand and consolidate because unstable health care partnerships limited patient access. Nevertheless, it has managed to retain archival formalin-fixed paraffin-embedded material, but no validated inventory was available at the time of the survey. Therefore, quantitative data could not be reported. Conversely, FALP-BB, one of two biobanks housed in a health care facility (an oncology nonprofit), benefits from direct patient access, supporting a large participant base and multiple collections despite its lower specimen-to-donor ratio.
The central panel of Figure 4D–G offers an overview of sample management, illustrating the complexity of Chilean biobank operations—from stored sample types to quality standards, sources, and uses. Figure 4D shows that the most common sample types are serum, plasma, and buffy coat, likely due to their ease of collection, processing, and versatility for proteomic, metabolomic, and genomic studies. 57 Blood-derived samples predominate, with an average of four sample types per biobank, followed by tissue samples, which are particularly relevant for oncological research. Saliva, urine, and feces are less represented but hold growing potential, especially for microbiome studies.
Expanding on processing capacities, more than half of Chilean biobanks have established the capability to extract and store DNA, positioning themselves for genomic studies, personalized medicine, and molecular diagnostics. The data indicate a good level of diversification and processing capacity—averaging 5.9 sample types per biobank—especially in multi-collection centers. Collaboration plays a key role in expanding technical capacities in emerging biobanks.
Consistent with earlier observations (Fig. 3J), the challenges in standardization and quality control are also reflected in sample management. A key issue is that nearly half of the biobanks have not implemented the Standard Preanalytical Code (SPREC) quality standard, 58 which is crucial given the high sensitivity of sample quality to pre-analytical handling and the need for standardized procedures to enable interoperability at national and international levels.
Regarding sample origin (Fig. 4F), most specimens are sourced exclusively from the host institution; only three biobanks report collaboration with networks or private entities. This strong institutional dependence reflects a closed ecosystem of largely individual initiatives. Such fragmentation limits interoperability, expansion, visibility, and collection impact. 7
Sample use, like sample origin, shows low complexity in terms of interaction with end users. Nearly all biobanks report exclusive use for research, except BTUCH, which also supports teaching and diagnosis. Given that most are housed in universities, a stronger educational role would be expected. Yet, the formative potential of biobanks remains largely unrealized, constraining the development of specialized personnel, despite the need for advanced training and ongoing education, both essential for operational success and innovation in biobanking. 59
While the central panel (Fig. 4D–G) focused on sample management, highlighting operational strengths and persistent gaps, the bottom panel turns to data management, offering insight into processing structures, standard adoption, and the existence of documented data policies across Chilean biobanks. Rigorously structuring data associated with biospecimens is pivotal in transitioning from storage-only to biobanking. 60 Figure 4H shows that all biobanks have implemented some form of data traceability, though the type and complexity of software tools vary significantly. Advanced systems—such as self-developed software (Syspath), biobank LIMs (Noraybanks®, BSI), and data management platforms (REDCap, OpenClinica, Medrio)—coexist with simpler, nonspecialized tools such as Microsoft Excel® or even physical records. This variation may stem from financial constraints, software costs, training needs, and differences in collection size, complexity, and intended use, as smaller collections may rely on less sophisticated systems, in a context where Chilean regulations do not mandate advanced data infrastructures and where a national electronic health record system was only legislated in 2024. 61
Despite these limitations, nearly all biobanks have established a minimum dataset (Fig. 4I), a fundamental step toward harmonization and consistency in data collection and management.62,63 Regarding data standards and semantics (Fig. 4J), implementation remains delayed and lags behind progress in sample management. Of the nine biobanks, four (44.4%) have not adopted any standard or semantic framework, and four have implemented only one, with SNOMED-CT most commonly used. Only BTUCH follows multiple standards, including Systematized Nomenclature of Medicine—Clinical Terms (SNOMED-CT), Logical Observation Identifiers Names and Codes (LOINC), Minimum Information About BIobank data Sharing (MIABIS), and FAIR. In this area, Chilean biobanks face a key and urgent challenge to achieve internationally recognized standards and enable interoperability. Despite these shortcomings, Figure 4K shows ongoing efforts to close this gap. At least 55.6% (five out of nine) have a data management policy in place, but only one has it documented. This reflects growing awareness but highlights the need for formalization.
In summary, Chilean biobanks have progressed in data management, partly driven by the recent expansion of data sciences. However, this progress remains limited, reflecting the legacy of earlier biorepository models—focused on biological sample storage—and the resulting predominance of professionals from biological and health sciences, with limited involvement from informatics experts. Yet biobanking is inherently interdisciplinary, requiring integration across biological, clinical, bioinformatics, ethical, and social domains. Advancing toward inter- and transdisciplinary models is essential to enhance scientific impact, operational efficiency, and integration with health care and society.13,64
Biobank Workforce Development
The success of a biobank heavily depends on its human resources. 59 However, most analyses focus on infrastructure and technology, often overlooking the crucial role of trained personnel in managing these resources and ensuring their functionality. Given the complexity of biobank processes, inadequate training and suboptimal performance can significantly impact the operation, administration, and long-term sustainability.33,65,66
In Chile, biobanks engage in training activities at varying levels, with notable differences between them. The most frequently reported is internal training, carried out by six out of the nine biobanks (66.7%), showing a widespread effort to strengthen internal capabilities. Some biobanks also train personnel within their institutions or extend their training externally. However, most (six out of nine, 66.7%) conduct only one of these types of training, and one biobank reported no training at all.
The training of future biobank experts requires postgraduate specialization programs to increase the number of trained professionals in the country and to strengthen the discipline. Currently, no formal programs of this kind exist in Chile, but established biobanks offer direct training and have sought collaborations with academic institutions to develop formal educational programs. It is expected that these efforts will soon yield tangible results.
Beyond training programs for biobank personnel, it is essential to integrate biobanking as a discipline into the curricula of biomedical and health sciences degree programs at universities. This serves two equally important objectives: training future professionals who will work in biobanks and those who will work with biobanks. Preparing new generations of researchers and professionals who can correctly understand and navigate the technical and ethical complexities of working with biological samples and data is crucial, particularly in the era of 4P medicine.
Chilean biobanks have trained 101 individuals so far, but BTUCH and PUC-BB account for 79.2% of trainees (50 and 30, respectively), as they are the only biobanks regularly offering courses and internships for undergraduate, postgraduate, and postdoctoral students. Other centers report significantly lower numbers, ranging from one to five trained individuals each. This disparity underscores the need for national strategies to balance and expand biobank training. Strengthening educational initiatives would not only enhance biobank operations but also enrich academic education, fostering a more rigorous and ethical approach to biomedical research.
However, this goal faces significant challenges. Each biobank has unique needs, requiring professionals with both adaptability and specialized knowledge tailored to their specific context. 65 This is particularly relevant in Chile, where centralization and the country’s geographic and population diversity create both challenges and opportunities. 42
Sustainability in Chilean Biobanks
As biobanks evolve in complexity, they must ensure sustainability across operational, social, and financial domains. 67 Initially, they primarily rely on government or institutional funding, but these sources are often limited and vulnerable to cuts.68,69 Long-term financial stability requires diversifying funding sources, incorporating public and institutional contributions, research grants, cost recovery, private partnerships, and philanthropy.67,70
Cost recovery models, typically implemented early, involve tiered pricing based on user type. However, this strategy alone is insufficient, particularly in academic biobanks, where high fees are complicated. 71 Cost recovery generally covers only 5%–25% of research biobank revenue, rarely exceeding 40%, and in some cases represents just 1% of total funding.70,71
While a detailed economic analysis is beyond this study’s scope, data from BTUCH and PUC-BB using the funding categories proposed by Doucet et al. 70 provide insight into Chile’s biobank funding landscape. To enable structured comparison, the analysis focused on BTUCH and PUC-BB, the only institutions that could provide data and staff support to restructure information into comparable categories from heterogeneous sources. Unlike these university biobanks, FALP-BB belongs to a private health care facility, a context where allocating resources for research entails difficulties, hindering inclusion in this analysis. In light of the sensitivity surrounding financial data, we present the BTUCH and PUC-BB results narratively to reflect the descriptive scope and avoid overstating precision. During the analyzed 2021–2023 window, both biobanks relied exclusively on university support, competitive grants, and cost recovery, with no private funding. University contributions comprised 66.4% and 88.0% of total income at BTUCH and PUC-BB, respectively. Research grants contributed 20.4% for BTUCH and 6.7% for PUC-BB, while cost recovery represented 13.2% and 5.2%, respectively.
Regarding expenditure, BTUCH reported an annual operational cost of ∼$640,000 USD, while PUC-BB reported ∼$310,000 USD. BTUCH manages 4.3 times more samples than PUC-BB, which results in a per-specimen cost that is 45.3% lower than PUC-BB’s, suggesting that larger volumes allow for broader cost distribution. Analyzing the expenditure by category, sample management accounted for 28.2% of BTUCH’s total costs, compared with 7.6% at PUC-BB, leading to a per-specimen cost 1.67 times higher at BTUCH in this category. Human resources were the largest expense in both biobanks, at 59.8% of BTUCH’s costs and 90.7% of PUC-BB’s. On a per-unit basis, BTUCH’s personnel costs were 29.88% lower than PUC-BB’s, reflecting differing resource allocation strategies.
Among smaller expenditures, both PUC-BB Biobank and BTUCH allocated approximately 1% to infrastructure. Meanwhile, BTUCH spent 9.8% on equipment, a category with no reported PUC-BB expenses. BTUCH also allocated 1.5% to biobank network maintenance, which was absent at PUC-BB. Other expenses accounted for 0.7% at BTUCH and 0.9% at PUC-BB.
Each biobank operates differently, and differences in expenditure distribution and cost structures suggest that BTUCH, by handling a larger sample volume, might be starting to benefit from economies of scale. In contrast, PUC-BB appears to follow a more human-resource-intensive model. However, these estimates reflect only direct operational expenses and exclude indirect costs covered by host universities, such as physical space, institutional infrastructure, basic utilities, insurance, and cleaning and maintenance services. These enabling resources are substantial and crucial for biobank sustainability, and their reduction could compromise core operations. 71 Overlooking such costs underestimates the actual resources required to maintain a functional and sustainable biobank.
Productivity of the Chilean Biobank
While securing funding is crucial, long-term biobank sustainability also relies on demonstrating scientific and societal impact.67,72,73 Biobank productivity is multidimensional, encompassing scientific output, researcher training, and integration into public health policies.72,74 Each biobank follows a unique mission and model, influencing its contributions. Thus, applying uniform productivity metrics may underestimate their role in biomedical research.73,75
Broader indicators have been proposed, beyond sample distribution, including scientific publications, collaborative networks, data and sample reuse, professional training, and policy influence.72–74,76 However, tracking biobank productivity remains challenging, as contributions are not formally acknowledged and systematically underreported, especially in scientific publications.72,73,75
This study compiles data reported by BTUCH and PUC-BB regarding the 2021–2023 period, representing an initial effort to harmonize the recording of biobank productivity, although significant information gaps remain. Despite their operational differences and distinct objectives, the data from BTUCH and PUC-BB allow for the identification of general trends and challenges in accessibility, collaboration, sample distribution, scientific output, and training.
BTUCH recorded 339 sample requests, while PUC-BB recorded 874. However, the two biobanks differ in some sample release modalities. For example, while BTUCH receives requests and releases samples mainly in batches and considers the release to the same project as a single session, PUC-BB has a “short release” modality that distributes individual samples per release, used, for example, in the establishment of organoids. These differences reduce the possibility of directly comparing the numbers between the two biobanks. Despite these differences, the new user incorporation was stable and comparable: BTUCH reported 9, 10, and 4 new users in 2021, 2022, and 2023, respectively, compared with PUC-BB’s 5, 5, and 3. BTUCH reported 7 national and 4 international collaborations, while PUC-BB reported 3 national and 10 international partnerships.
Regarding sample distribution, BTUCH released 3709 biospecimens and PUC-BB released 875 between 2021 and 2023. When these numbers are considered in relation to each biobank’s total stored biospecimens, BTUCH had collected 50,058 samples by the end of 2022—yielding a release proportion of 7.41%—while PUC-BB, with 18,688 samples, reached 4.68%. This multiyear proportion was considered a starting point for internal monitoring. Based on this assessment, both institutions identified a release proportion above 10% as a target to guide progress in the current phase.
A second parameter that supports internal monitoring by helping to identify collections with particularly high or low levels of usage is the per-collection release percentage. For example, in the case of colorectal cancer, BTUCH released 755 biospecimens out of 5402 stored (14.0%), while PUC-BB released 156 out of 4740 (3.3%), despite both having similarly sized collections. COVID-19-related samples at BTUCH also showed high release proportions (12.7%), reflecting strong research demand during the pandemic. At PUC-BB, the gallbladder cancer collection, although smaller (731 samples), reached an even higher proportion (20.5%). While not designed for inter-biobank benchmarking, these numbers illustrate how this parameter can help biobanks align collection management with evolving institutional priorities. Additional contrasts—such as the number of sample types distributed (10 at BTUCH vs. 2 at PUC-BB)—may also offer insights into the reach of collections. Moving forward, tracking these proportions over time, monitoring trends annually, and incorporating other related metrics could strengthen internal monitoring and guide future planning.
Recognition in scientific publications also varies. Although neither biobank has a formal recognition policy in place, both have developed internal practices to monitor acknowledgments and are gradually moving toward more structured mechanisms, including defined conditions and requirements for such attribution. BTUCH reported that 9 publications used its resources without acknowledgment, 12 mentioned the biobank in the methods or acknowledgments section, 3 included a biobank staff member as an author, and 4 incorporated coauthorship along with a mention in methods or acknowledgments. PUC-BB could not track publications using its samples without acknowledgment but reported 4 publications mentioning the biobank and 33 including at least one biobank member as a coauthor.
For training and outreach, BTUCH recorded 33 conference presentations featuring its samples or data and 8 conferences dedicated to biobank-related topics. PUC-BB reported 11 presentations in the latter category. In both cases, presentations about biobanking were delivered by biobank staff.
Measuring biobank productivity remains a challenge. While current efforts enhance systematization, strengthening reporting mechanisms and increasing acknowledgment in publications, conferences, and training programs is necessary. Additionally, structured mechanisms for returning experimental results to biobanks are lacking, limiting the potential to enrich collections with complementary data.72,74 Chile’s scientific ecosystem has yet to establish a strong culture of acknowledgment for biobanks, restricting their visibility and limiting their impact. Developing more structured practices would improve productivity evaluation and reinforce biobank sustainability.
Discussion and Conclusions
Chile hosts a relatively large number of biobanks for Latin America, with a predominance of tumor-focused collections and a strong concentration in the capital. However, many previously reported biobanks have disappeared or failed to consolidate and expand over time, highlighting the challenges of financial instability, limited institutional support, and dependence on specific institutional leadership.21,77,78
Only a few Chilean biobanks are fully structured according to the discipline’s evolution, while others are newly established or just beginning to align with international standards. Despite this discontinuity, biobank proliferation reflects significant installed capacity and sustained interest. These developments have occurred without a national policy or legal framework, resulting in a bottom-up creation driven by biobanks and researchers from academic and health care institutions interested in supporting biobanks. Determining whether this pattern is unique to Chile or reflects broader Latin American trends would be valuable.
While biobanks vary in organizational maturity, progress has been made in traceability and biospecimen standardization. However, challenges persist in data management and interoperability, as standardized systems for biobank integration and biomedical infrastructure connections remain limited. Additionally, governance formalization and documentation require strengthening to ensure transparency, sustainability, and international alignment.
Sustainability remains a major concern. The financial structures of Chile’s most established biobanks reflect a transition into what the literature describes as the Biobank 2.0 stage, which is characterized by a shift from accumulation to quality, operational consolidation, and a progressive move from heavy reliance on core institutional funding toward more diversified funding strategies. 69 However, stable, long-term funding mechanisms remain absent, creating uncertainty, particularly for emerging biobanks. A deeper analysis would be valuable to assess the replicability of proposed models within the Chilean context, particularly considering the previously discussed bottom-up development.
In terms of productivity, Chilean biobanks are generating outputs across multiple dimensions. However, this study has highlighted the need for post-transfer traceability mechanisms, as their absence hinders an accurate evaluation of the biobanks’ impact within the biomedical ecosystem.
Despite these challenges, Chilean biobanks have shown strong collaborative maturity, enabling interinstitutional coordination and joint initiatives to structure and consolidate the biobanking ecosystem while preventing fragmentation. Multiple funding applications have been submitted to the Chilean National Agency for Research and Development (ANID) for a national biobank network and a tumor biobank network. However, existing funding programs do not prioritize long-term, open-access platforms like biobanks, preventing these initiatives from securing sustained support and hindering their full development.
Nonetheless, bottom-up initiatives have achieved tangible results like the aforementioned National COVID-19 Biobank Network 49 and the CECAN Biobank Network—a consortium of seven centers under the CECAN—which is finalizing governance, ELSI policy standardization, operational harmonization, and catalog software before initiating sample and data collection.
Additionally, starting in 2018, various collaborative efforts among experts led to the drafting of a regulatory proposal for the operation of biobanks for biomedical research in Chile. 79 Unfortunately, the regulation has not yet been formalized or legally enacted. Nevertheless, these initiatives reinforced interinstitutional integration and strengthened the national biobank community. Cooperation is particularly relevant in regional areas, where it can help promote a more equitable distribution of resources and infrastructure.
Chilean biobanking is evolving, with significant potential and opportunities ahead, but it needs to be incorporated into national research and public health policies. The maturation of biobanks will depend on their ability to address challenges in formalization, standardization, and post-transfer traceability while integrating into international networks, diversifying funding sources, and enhancing visibility and societal engagement. However, achieving a sustainable model that fully realizes its translational potential also requires structural support and formal recognition of its value within the national and international biomedical ecosystem, ensuring its impact on improving health and quality of life.
Authors’ Contributions
A.C. and I.A.S. contributed to conceptualization. I.A.S. wrote the original draft. G.D. and D.I.R. conducted investigation, data curation, and formal analysis, with I.A.S. also contributing to formal analysis and visualization. Y.E.-P., C.B., E.A., C.S., M.F.-G., J.C.R., A.C., and C.C. were responsible for validation. S.C.-Z., Y.E.-P., C.B., E.A., C.S., M.F.-G., A.C., G.D., I.A.S., and D.I.R. contributed to writing—review and editing. A.C. provided supervision. All authors have read and agreed to the published version of the article.
Footnotes
Acknowledgments
The authors extend their deepest gratitude to all the participants in the Chilean Biobanks collections for their generous contribution of information and samples, which are vital for advancing science for the common good in Chile. They also sincerely thank the academic, technical, and professional staff for their dedication and support, especially Ian Reyes (PUC Biobank); Michelle Mattar Araos (Universidad de Valparaíso); Macarena Arellano Sepúlveda (CIAPEC); Jorge Carrasco (Hospital Luis Calvo Mackenna); Patricia Faundez Riquelme (Universidad de Tarapacá); Daniela Zapata Contreras (Universidad de Magallanes); and from the MAUCO Biobank: Catterina Ferreccio (Founder and former Director), Claudia Marco, Miguel Carrera, and Katherine Brito.
Author Disclosure Statement
The authors declare no conflict of interest.
Funding Information
This work was supported by the Chilean National Agency for Research and Development—ANID (Grant: ACT210085); by the CECAN ANID/FONDAP 152220002; by the School of Medicine, Pontificia Universidad Católica de Chile; by the Faculty of Medicine, Universidad de Chile; Hospital Clínico de la Universidad de Chile; and by the Universidad de Atacama.
