Abstract
Background
The efficacy of adjunctive procedures targeting allergic rhinitis (AR) symptoms during septoplasty remains controversial. While radiofrequency ablation of the inferior turbinates (ITRA) is commonly performed, its impact on allergic symptoms beyond nasal obstruction is limited. Posterior nasal nerve ablation (PNNA) has recently gained attention as a promising alternative.
Objective
To compare the effects of PNNA and ITRA performed concurrently with septoplasty on allergic and obstructive nasal symptoms in patients with septal deviation and AR.
Methods
This prospective cohort study included 69 patients with septal deviation and AR who underwent septoplasty with either PNNA (n = 35) or ITRA (n = 34). Symptoms were assessed preoperatively and at 3 months postoperatively using the Mini-Rhinoconjunctivitis Quality of Life Questionnaire (MiniRQLQ), reflective Total Nasal Symptom Score (rTNSS), and Nasal Obstruction Symptom Evaluation (NOSE) scale. The rTNSS was divided into obstruction-related (rTNSS-A) and allergic (rTNSS-B) sub scores.
Results
Both groups showed significant postoperative improvement in all parameters (p < 0.001). Compared with ITRA, the PNNA group demonstrated greater reductions in MiniRQLQ (mean changes = 36.97 ± 12.4 vs 32.00 ± 11.8; p = 0.025), rTNSS-B (median changes = 4.0 vs 2.0; p < 0.001), and rTNSS (median changes = 4.0 vs 2.5; p < 0.001). Changes in rTNSS-A and NOSE scores were comparable between groups (p > 0.05). No serious adverse events occurred; transient crusting was observed in 5 ITRA and 3 PNNA patients, and mild bleeding in 2 ITRA patients.
Conclusion
Both techniques effectively improved nasal symptoms, but PNNA provided superior relief of allergic symptoms and quality-of-life gains, supporting its consideration as an adjunctive treatment in septoplasty patients with comorbid AR.
Keywords
Introduction
Allergic rhinitis (AR) is a prevalent chronic inflammatory disease of the nasal mucosa, affecting up to one-third of the global population and substantially impairing quality of life, sleep, and work productivity. 1 It is characterized by nasal obstruction, rhinorrhea, sneezing, and itching triggered by environmental allergens. Structural nasal abnormalities, particularly septal deviation, frequently coexist with AR and may exacerbate nasal obstruction, contributing to inadequate symptom control despite optimized medical therapy. 2 In such cases, surgical intervention may be considered to improve nasal airflow and overall symptom burden.
Septoplasty is the standard surgical procedure for correcting septal deviation. Adjunctive procedures addressing inferior turbinate hypertrophy—most commonly inferior turbinate radiofrequency ablation (ITRA)—are often performed in patients with concomitant AR. 3 While ITRA effectively improves nasal patency through submucosal tissue reduction, its impact on allergic symptoms beyond obstruction remains controversial. In a prospective randomized trial, Kang et al demonstrated that adding ITRA to septoplasty did not result in additional improvement in allergic symptoms such as rhinorrhea or sneezing. 4
Posterior nasal nerve ablation (PNNA) has recently emerged as a minimally invasive option for patients with medically refractory AR, targeting parasympathetic innervation of the nasal mucosa to reduce secretory and neurogenic symptom components.5,6 Clinical studies and meta-analyses have reported significant improvements in allergic symptom burden following PNN-targeted interventions, including cryotherapy and temperature-controlled radiofrequency techniques.6–9
Despite growing interest in PNNA, direct comparative data between PNN-targeted and turbinate-based adjunctive procedures remain limited, particularly in patients undergoing septoplasty for combined structural and allergic nasal obstruction. Therefore, this prospective cohort study aimed to compare the effects of PNNA and ITRA performed concomitantly with septoplasty on allergic and obstructive nasal symptoms using validated patient-reported outcome measures.
Materials and Methods
Study Design and Ethics
This prospective comparative cohort study was conducted at Hacettepe University between March 2024 and December 2024. Adult patients with septal deviation and concomitant allergic rhinitis (AR) refractory to medical therapy were enrolled and underwent septoplasty combined with either posterior nasal nerve ablation (PNNA) or inferior turbinate radiofrequency ablation (ITRA). The study was designed as a prospective observational cohort, and randomization was not performed. Patients were allocated to treatment groups based on shared decision-making between the surgeon and patient within predefined eligibility criteria, considering symptom profile and anatomical findings. To minimize selection bias, consecutive eligible patients meeting predefined inclusion criteria were enrolled, and baseline demographic and clinical characteristics were comparable between groups Table 1. The study was approved by the Hacettepe University Health Sciences Research Ethics Committee (approval number SBA 24/1230, March 4, 2024) and conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants prior to inclusion.
Baseline Questionnaire Scores of the two Groups.
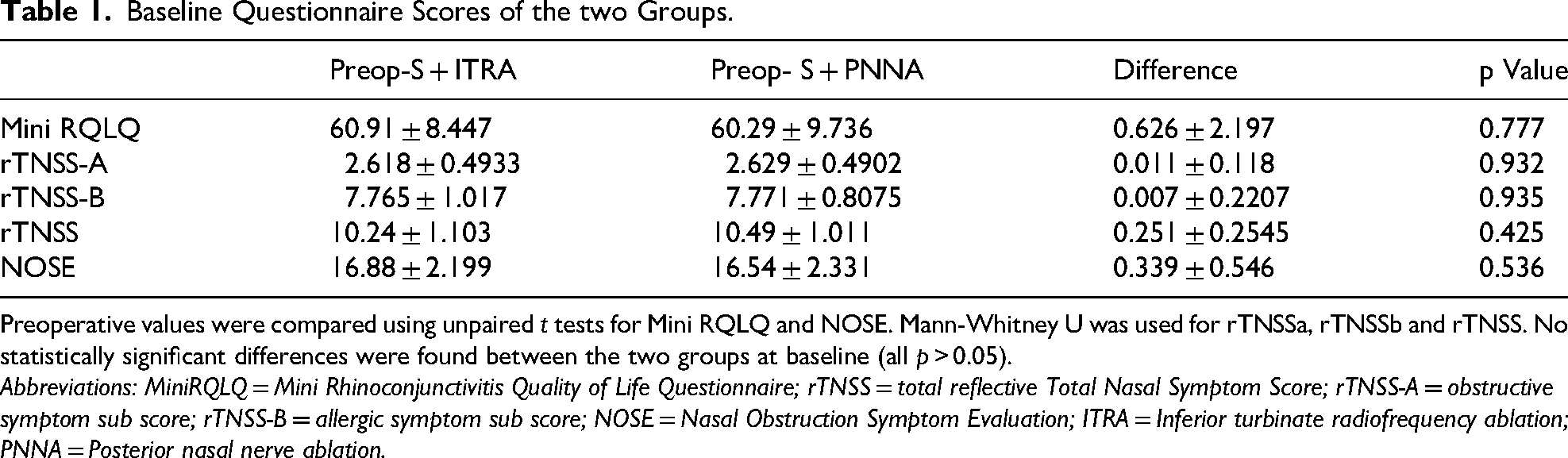
Preoperative values were compared using unpaired t tests for Mini RQLQ and NOSE. Mann-Whitney U was used for rTNSSa, rTNSSb and rTNSS. No statistically significant differences were found between the two groups at baseline (all p > 0.05).
Abbreviations: MiniRQLQ = Mini Rhinoconjunctivitis Quality of Life Questionnaire; rTNSS = total reflective Total Nasal Symptom Score; rTNSS-A = obstructive symptom sub score; rTNSS-B = allergic symptom sub score; NOSE = Nasal Obstruction Symptom Evaluation; ITRA = Inferior turbinate radiofrequency ablation; PNNA = Posterior nasal nerve ablation.
Participants
Inclusion criteria were: (1) age ≥18 years; (2) symptomatic septal deviation requiring surgical correction; (3) diagnosis of persistent, moderate-to-severe allergic rhinitis according to the Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines; and (4) persistence of typical nasal symptoms despite ≥8 weeks of optimized medical therapy consisting of daily intranasal corticosteroids and oral antihistamines. All participants continued these medications up until the time of surgery.
Allergic rhinitis diagnosis was based on both clinical and objective criteria. All patients exhibited at least two cardinal AR symptoms (rhinorrhea, sneezing, nasal itching, and/or nasal obstruction) for ≥4 days per week and ≥4 consecutive weeks and demonstrated positive skin prick test reactivity to at least one relevant aeroallergen. Baseline disease severity thresholds required a total reflective Total Nasal Symptom Score (rTNSS) ≥ 4 and a Nasal Obstruction Symptom Evaluation (NOSE) score ≥30. Serum-specific IgE testing was not routinely performed.
Postoperatively, all patients received identical standardized care, including routine nasal saline irrigation. Endoscopic nasal examinations were performed weekly during the first 3 postoperative weeks to assess mucosal healing and manage crusting as needed, followed by a scheduled evaluation at 3 months postoperatively Figure 1.
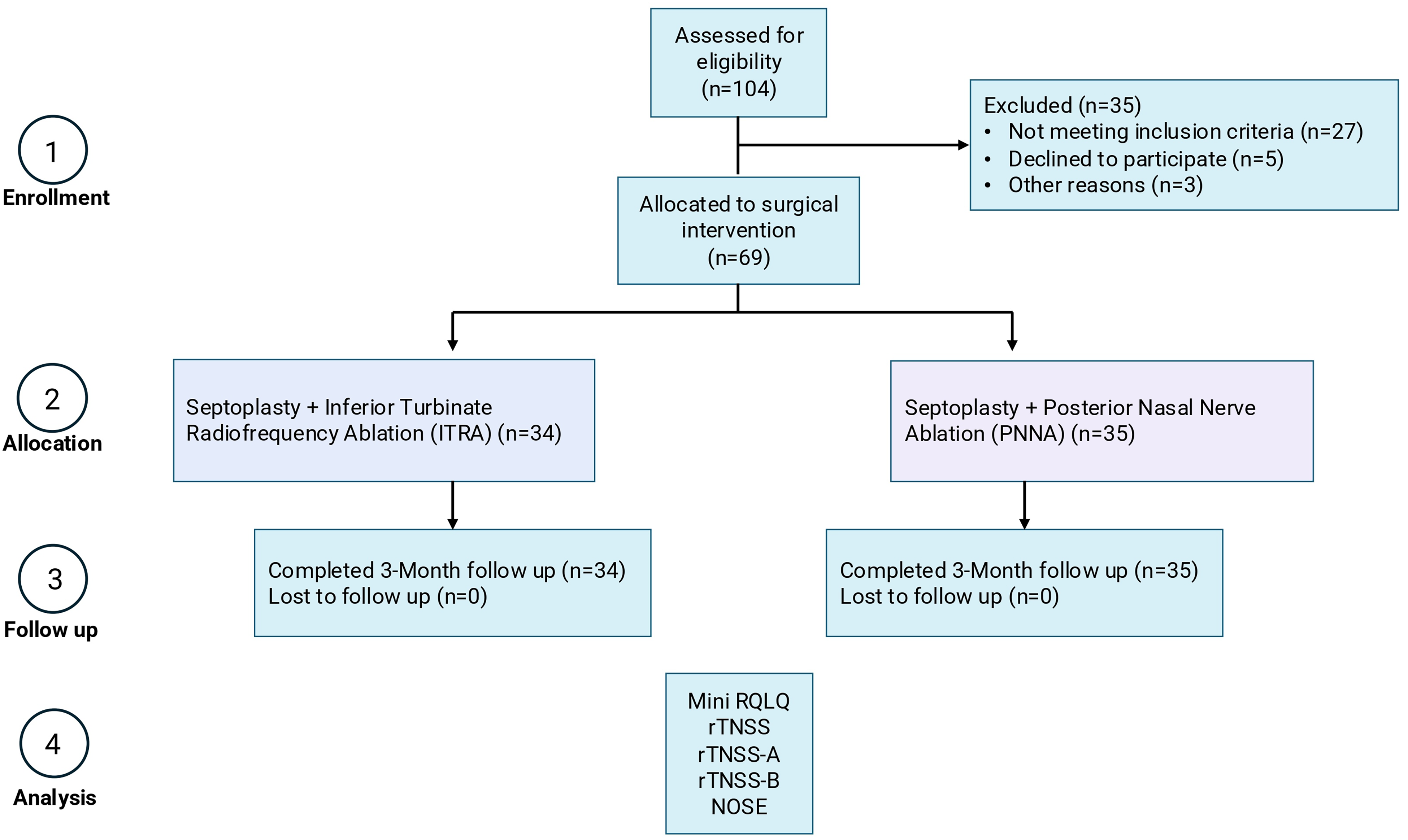
CONSORT-style flow diagram illustrating patient enrollment, exclusion, allocation to surgical intervention, follow-up, and final analysis. Consecutive adult patients with septal deviation and allergic rhinitis were screened for eligibility. Eligible patients underwent septoplasty combined with either inferior turbinate radiofrequency ablation (ITRA) or posterior nasal nerve ablation (PNNA). All patients completed the 3-month follow-up and were included in the final per-protocol analysis.
Exclusion criteria included prior nasal or sinus surgery, sinonasal polyposis, chronic rhinosinusitis requiring intervention, uncontrolled asthma, systemic inflammatory disease, pregnancy, bleeding diathesis, and inability to comply with follow-up visits.
Surgical Procedures
All operations were performed by the same surgical team under general anesthesia. Septoplasty was performed via a standard hemitransfixion incision with subperichondrial and subperiosteal flap elevation, correction of deviations, and quilting sutures.
In the S + PNNA group, the target area was localized at the posterior middle meatus, corresponding to the mucosal region overlying the posterior nasal nerve near the sphenopalatine foramen, as described by Hwang and Chang for posterior nasal nerve–targeted cryotherapy. (13,14) A bipolar cautery probe (Valleylab FT10; Medtronic-Covidien, Minneapolis, MN, USA) was applied directly to the intact mucosa at a low-power coagulation setting (≈10-15 W; generator level 8-10) for 1–2 s per application until visible localized mucosal blanching occurred, with two to three non-overlapping applications per side. Bipolar energy was selected to limit lateral thermal spread and mucosal carbonization; over-cauterization was minimized by avoiding repeated activation at the same site and areas of prominent surface vascularity. This approach was intended to target the mucosa overlying the posterior nasal nerve region and to achieve localized superficial tissue effects, analogous in application concept to commercial cryoablation or temperature-controlled radiofrequency systems, without assuming device-level equivalence. (Supplementary video)
In the S + ITRA group, submucosal radiofrequency ablation of the inferior turbinates was performed bilaterally using the Bohns RF generator (Bohns Medical, Seoul, South Korea). The RF probe was introduced longitudinally into the anterior, middle, and posterior thirds of each turbinate (2-3 passes per side, each application lasted approximately 3-4 s) with energy set to low-power coagulation (≈10-15 W), producing controlled submucosal coagulation without mucosal injury.
Outcome Measures
Primary outcome was change in allergic symptom burden, assessed by rTNSS and its allergic sub scores (rhinorrhea, sneezing, itching) at 3 months post-operation. A responder was defined a priori as a patient achieving a ≥ 30% reduction from baseline rTNSS at the 3-month postoperative assessment, consistent with previously reported clinically meaningful thresholds.
Secondary outcomes included:
NOSE score change (nasal obstruction domain); Disease-specific quality of life via Mini-Rhinoconjunctivitis Quality of Life Questionnaire (Mini-RQLQ); Adverse events (AEs) and serious AEs (procedure-related, surgical, or device-related).
Adverse events were prospectively assessed at each postoperative visit using a standardized clinical evaluation. Crusting, dryness, bleeding, infection, and synechiae were specifically assessed by endoscopic examination.
Statistical Analysis
All statistical analyses were performed using GraphPad Prism version 10.0 (GraphPad Software, San Diego, CA, USA). The Shapiro–Wilk test was applied to evaluate the normality of continuous variables. Data with normal distribution were expressed as mean ± standard deviation (SD), and non-normally distributed variables as median [interquartile range, IQR]. For within-group comparisons (pre- vs postoperative), the paired t-test was used for normally distributed data and the Wilcoxon signed-rank test for non-parametric data. For between-group comparisons of postoperative changes (Δ = postoperative − preoperative), the unpaired t-test was applied to normally distributed variables, and the Mann–Whitney U test to non-normally distributed variables. Effect sizes were calculated as eta-squared (η2) for parametric tests and rank biserial correlation (r) for non-parametric tests. A two-tailed p < 0.05 was considered statistically significant. The sample size (n = 69) was based on a prior power analysis indicating that at least 34 subjects per group were required to detect a 30% change in rTNSS with 80% power at a two-sided α = 0.05.
Results
Baseline Characteristics
A total of 69 patients (34 in the S + ITRA group and 35 in the S + PNNA group) completed the 3-month follow-up. Baseline demographic and clinical characteristics were comparable between the two groups (p > 0.05 for all). There were no significant differences in preoperative MiniRQLQ, rTNSS, rTNSS sub-scores, or NOSE scores, indicating a balanced distribution of baseline symptom severity Table 1.
Within-Group Outcomes
Both groups demonstrated significant postoperative improvement compared with baseline values. In the S + ITRA group, all scores (MiniRQLQ, rTNSS, rTNSS-A, rTNSS-B, and NOSE) improved significantly after surgery (p < 0.001 for all). Similarly, the S + PNNA group showed significant postoperative reductions in both allergic and obstructive symptom scores (p < 0.001 for all) Table 2.
Preoperative and Postoperative Symptom Scores in the ITRA and PNNA Groups.

Values with non-parametric distribution are presented as median [25th–75th percentile]. Within-group comparisons were performed using the Wilcoxon signed-rank test because most variables did not meet the assumption of normality according to the Shapiro–Wilk test. All p-values (p < 0.05 considered significant) refer to the change from preoperative to postoperative measurements within each group. Abbreviations: same as in Table 1.
Between-Group Comparisons
When postoperative changes (Δ = post − pre) were compared between groups:
rTNSS improvement was significantly greater in the S + PNNA group (median Δ = 4.0 [IQR 3.0-5.0]) than in the S + ITRA group (median Δ = 2.5 [IQR 2.0-3.0]); p < 0.001, r = 0.54 (large effect); Hodges–Lehmann median difference = 2.0 [95% CI 1.0 to 2.0] Figure 2. Allergic sub-scores (rTNSS-B) also showed greater reduction in the S + PNNA group (median Δ = 4.0 vs 2.0, p < 0.001, r = 0.52); Hodges–Lehmann median difference = 1.0 [95% CI 1.0 to 2.0]. Obstructive sub-scores (rTNSS-A) did not differ significantly between groups (p = 0.3948); Hodges–Lehmann median difference = 0.0 [95% CI 0.0 to 0.0] MiniRQLQ improvement was higher in the S + PNNA group (mean Δ = 36.97 ± 12.4) compared with S + ITRA (mean Δ = 32.00 ± 11.8), p = 0.0248, η2 = 0.073 (moderate effect); [95% CI 0.65 to 9.29]. NOSE score changes were comparable between groups (p = 0.7583, η2 = 0.001); [95% CI −1.47 to 0.68] Table 3.
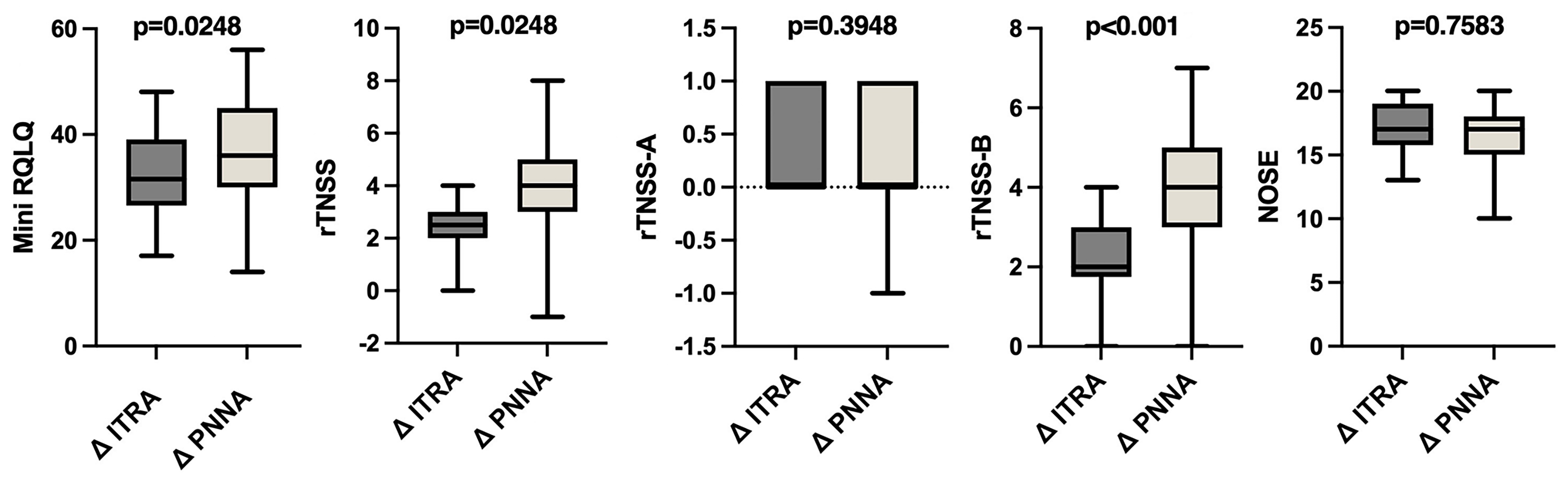
Bar graph displaying the mean ± standard deviation (SD) of postoperative change (Δ = postoperative − preoperative) in five outcome measures at 3 months: (A) MiniRQLQ, (B) rTNSS (C), rTNSS-A (obstructive domain), (D) rTNSS-B (allergic domain), and (E) NOSE. Group comparisons were performed using the unpaired t-test for normally distributed Δ (MiniRQLQ, NOSE) and the Mann–Whitney U test for non-normally distributed Δ (rTNSS, rTNSS-A, rTNSS-B). Exact p-values: MiniRQLQ p = 0.0248; rTNSS p < 0.001; rTNSS-A p = 0.395; rTNSS-B p < 0.001; NOSE p = 0.758. Sample sizes: S + ITRA (n = 34) and S + PNNA (n = 35).
Between-Group Comparison of Changes (Δ) in Outcome Measures.
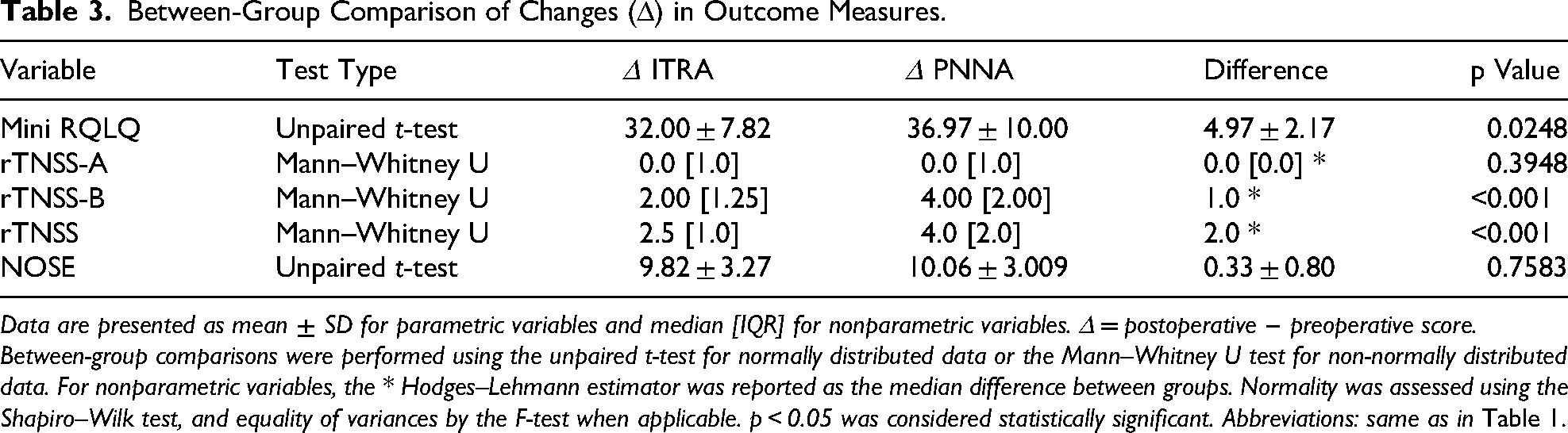
Data are presented as mean ± SD for parametric variables and median [IQR] for nonparametric variables. Δ = postoperative − preoperative score. Between-group comparisons were performed using the unpaired t-test for normally distributed data or the Mann–Whitney U test for non-normally distributed data. For nonparametric variables, the * Hodges–Lehmann estimator was reported as the median difference between groups. Normality was assessed using the Shapiro–Wilk test, and equality of variances by the F-test when applicable. p < 0.05 was considered statistically significant. Abbreviations: same as in Table 1.
Responder analysis
Using a predefined threshold of ≥30% reduction in rTNSS, the proportion of responders was significantly higher in the S + PNNA group compared with the S + ITRA group
The effect size analysis demonstrated a moderate treatment effect for PNNA on MiniRQLQ improvement (η2 = 0.073, d ≈ 0.55), whereas changes in NOSE and rTNSS-A scores showed negligible effects (η2 < 0.01, r = 0.10). In contrast, the PNNA group exhibited large effect sizes for rTNSS (r = 0.54) and rTNSS-B (r = 0.52), indicating clinically meaningful advantage over ITRA in overall symptom reduction.
Adverse Events
No major adverse events were observed in either group. Minor postoperative events were recorded prospectively. In the S + ITRA group, two patients (5.9%) experienced mild postoperative bleeding, and five patients (14.7%) developed transient crusting. In the S + PNNA group, three patients (8.6%) developed mild crusting. No cases of synechiae, infection, or persistent nasal dryness were identified on endoscopic follow-up. All events resolved with conservative management.
Discussion
Allergic rhinitis (AR) remains one of the most prevalent chronic inflammatory disorders of the upper airway, affecting nearly one-third of the global population and substantially impairing quality of life, sleep, and daily functioning. 1 Surgical correction of septal deviation by septoplasty is often required in patients with persistent nasal obstruction unresponsive to medical therapy, and various adjunctive techniques have been developed to enhance postoperative outcomes.2,3 Among these, inferior turbinate radiofrequency ablation (ITRA) has been widely adopted to reduce turbinate hypertrophy and improve airflow; however, its benefit in controlling allergic symptoms beyond mechanical obstruction remains controversial. 4 In contrast, posterior nasal nerve ablation (PNNA) has emerged as a targeted approach that attenuates parasympathetic innervation to the nasal mucosa, thereby diminishing glandular hypersecretion and neurogenic inflammation.5,6
This study is among the first to directly compare these two adjunctive techniques performed concurrently with septoplasty in a prospective design. Both ITRA and PNNA performed concomitantly with septoplasty led to significant improvement in nasal obstruction and quality-of-life indices. Nevertheless, PNNA provided greater reductions in allergic symptom scores (rTNSS-B and Mini-RQLQ) than ITRA, whereas improvements in obstructive scores (rTNSS-A and NOSE) were comparable between groups. Importantly, responder analysis demonstrated that PNNA was associated with a markedly higher likelihood of achieving clinically meaningful improvement, reinforcing that the observed reductions in symptom scores translate into tangible patient-level benefit. These findings suggest that PNNA, by addressing the neurogenic component of AR, may offer more comprehensive symptomatic control when performed alongside septoplasty in patients with concurrent structural deviation and allergic disease.
The present findings align with and extend previous evidence concerning surgical management of persistent allergic rhinitis. Kang et al 4 previously reported that adding inferior turbinate radiofrequency ablation (ITRA) to septoplasty provided no additional improvement in allergic symptoms such as sneezing or rhinorrhea, despite marked relief in nasal obstruction. This suggests that while ITRA effectively reduces turbinate volume and airflow resistance, it fails to modify the underlying neurogenic hypersecretion that drives allergic manifestations. Conversely, procedures targeting the posterior nasal nerve (PNN) have consistently demonstrated substantial benefit in controlling both obstructive and secretory components of AR. Early descriptions of endoscopic posterior nasal nerve (PNN) neurectomy date back to Kikawada [2007], and subsequent series of PNN-targeted procedures—including cryoablation and temperature-controlled radiofrequency—have reported significant reductions in nasal symptoms and improved quality of life in chronic rhinitis.10–14 Similarly, a recent meta-analysis by Balai et al (2023) confirmed that endoscopic PNN ablation provides durable improvement in allergic symptom control and quality of life compared to medical therapy alone. 15
Our results corroborate these observations by demonstrating that PNNA produced significantly greater improvement in allergic symptom-related indices (rTNSS-B and Mini-RQLQ) compared with ITRA when both were performed adjunctively to septoplasty. Importantly, the magnitude of improvement in Mini-RQLQ exceeded established minimal clinically important difference (MCID) thresholds, and responder analysis based on a ≥ 30% reduction in total rTNSS further supports the clinical relevance of the observed effects. To our knowledge, this is among the first prospective clinical studies directly comparing PNNA and ITRA performed concurrently with septoplasty, providing novel evidence on their differential effects in patients with coexisting allergic and structural nasal pathology.
The greater improvement in allergic symptom scores observed with PNNA may be related to its targeted neurophysiologic mechanism. The posterior nasal nerve conveys parasympathetic fibers regulating nasal glandular secretion and mucosal vascular tone, and ablation near the sphenopalatine foramen attenuates this pathway, reducing mucosal hyperreactivity and secretion.1,5,6 In contrast, inferior turbinate radiofrequency ablation primarily induces submucosal tissue coagulation and volume reduction, improving airflow but exerting limited influence on neurogenic inflammatory pathways.4,16,17, This mechanistic distinction may account for the comparable improvement in obstructive symptoms between groups and the greater reductions in allergic symptom burden and quality-of-life measures observed with PNNA.
Both procedures were well tolerated, with only minor, self-limited postoperative events. Transient crusting occurred in 14.7% of S + ITRA and 8.6% of S + PNNA patients, while mild postoperative bleeding was observed in 5.9% of the S + ITRA group; all resolved without intervention. No cases of synechiae, infection, prolonged dryness, epistaxis, or atrophic rhinitis were noted in either group. These findings are consistent with previously reported complication rates for turbinate radiofrequency and posterior nasal nerve procedures, which typically range from 5% to 15%.6,7,16 Prior meta-analyses have similarly demonstrated a low incidence of adverse events following endoscopic or cryoablative PNN interventions, supporting the overall safety of PNNA when performed with appropriate technique and energy settings.6,7
The clinical implications of these findings are relevant for optimizing adjunctive surgical strategies in patients with septal deviation and coexisting allergic rhinitis. While septoplasty remains the primary intervention for structural nasal obstruction, the addition of a procedure targeting mucosal hyperreactivity may further improve symptom control in selected patients. The present results suggest that posterior nasal nerve ablation may be particularly beneficial in individuals with persistent rhinorrhea, sneezing, or nasal itching despite optimized medical therapy, whereas turbinate-based interventions primarily address mechanical obstruction. Incorporating PNNA at the time of septoplasty may allow simultaneous management of structural and neurogenic components of disease, potentially reducing the need for staged interventions. However, larger randomized studies with longer follow-up and objective outcome measures are required to define optimal patient selection and assess long-term durability.
This study has several limitations. The relatively small, single-center cohort and non-randomized design may limit generalizability and allow for residual confounding despite prospective enrollment and comparable baseline characteristics. Follow-up was limited to three months, and outcomes relied primarily on patient-reported measures without adjunctive objective assessments. Variability in turbinate remodeling after ITRA, potential confounding effects of septoplasty itself, and the absence of a combined PNNA + ITRA arm may also have influenced symptom outcomes. Nevertheless, this prospective comparison provides clinically relevant evidence suggesting greater allergic symptom improvement with PNNA as an adjunct to septoplasty.
Conclusion
In patients with septal deviation and concomitant allergic rhinitis, both posterior nasal nerve ablation (PNNA) and inferior turbinate radiofrequency ablation (ITRA) performed with septoplasty resulted in significant improvements in nasal obstruction and quality of life. PNNA, however, was associated with greater improvement in allergic symptom burden and a higher proportion of patients achieving clinically meaningful symptom reduction. These findings suggest that PNNA may provide additional benefit in addressing the neurogenic and secretory components of allergic rhinitis. Further randomized studies with longer follow-up are warranted to confirm the durability and generalizability of these results.
Supplemental Material
Footnotes
Ethics Approval
The study was approved by the Hacettepe University Health Sciences Research Ethics Committee (approval number SBA 24/1230, date: 04/03/2025) and conducted in accordance with the Declaration of Helsinki.
Informed Consent
Written informed consent was obtained from all participants prior to inclusion.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and analyzed during the current study are not publicly available due to patient privacy and institutional policy but are available from the corresponding author on reasonable request.
Meeting Information
This study was presented at the 30th Congress of the European Rhinologic Society (ERS 2025) on 23 June 2025, in Budapest, Hungary.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
