Abstract
This study presents a simple highly versatile and efficient synthesis of various β-amino carbonyl compounds in the Mannich reaction of acetophenone with aromatic aldehydes and aromatic amines in the presence of nanosulfated zirconia, nanostructured ZnO, nano-γ-alumina, and nano-ZSM-5 zeolites as the catalyst in ethanol at moderate temperature. The optical properties of the nanostructured organic molecules were studied. The advantages of methods are short reaction times, milder conditions, easy workup, and purification of products by nonchromatographic methods. The catalysts can be recovered for the subsequent reactions and reused without any appreciable loss of efficiency.
Keywords
Introduction
The Mannich reaction is a classical method for the preparation of β-amino ketones and aldehydes 1 –3 and has been one of the most important basic reactions in organic synthesis because it affords synthetically and biologically important β-amino carbonyl compounds that are important intermediates for the construction of various nitrogen-containing amino alcohols, peptides, lactams, optically active amino acids, natural products, and pharmaceuticals. 4 –18 Due to the drastic reaction conditions, severe side reactions, substrate limitations, expensive reagents or catalysts, and the long reaction time and low yield, the classical intermolecular Mannich reaction is plagued by a number of serious disadvantages. 19 –21 To overcome the drawbacks of the classic method, the Lewis acid catalyzed condensation of ketone to preformed imines is an excellent variant of the Mannich reaction. 22 –36
Hence, there is high interest in developing convenient methods for the synthesis of β-amino ketones. Some of the recent achievements in the efficient construction of this nucleus include the development of Lewis acid catalysts; however, few useful Lewis acid catalysts are developed in the past few years. 37 –40
Mannich reactions have gained popularity in synthetic chemistry over the past decades. Recently, direct Mannich reactions of aldehydes, ketones, and aryl amines have been realized via HCl, 41 –43 Lewis acids, 44 –55 praline, 56 –58 p-dodecyl benzene sulfonic acid, 59 PS-SO3H (polymer-support sulfonic acid), 60 lanthanides, 61,62 transition metal salt catalysis, 63 organocatalytic approaches, 64 –66 Yb(OiPr)3, 67 InCl3, 68,69 BiCl3, 70 phosphorodiamidic acid, 71 AuCl3–PPh3, 72 iodine, 73 aluminum nitrate, 74 sulfamic acid, 75 ceric ammonium nitrate (CAN), 76 salen–Zn complex, 77 rare-earth metal triflates, 78 –80 NbCl5, 81 Zn(OTf)2, 82 samarium(II) iodide, 83 Nano-TiO2, 84 and silica-supported AlCl3. 85 Various other promoters, such as Bronsted acid catalysts, 86,87 have also been reported to catalyze the Mannich reaction.
In the recent years, the use of nanostructured ZnO, 88 nanosulfated zirconia, 89 nano-γ-alumina, 90 and nano-ZSM-5 zeolite 91 catalysts has received considerable interest in organic synthesis. This extensive application of heterogeneous catalysts in synthetic organic chemistry can make the synthetic process more efficient from both environmental and economic point of view, 92 and catalyst used can be easily recycled.
As a part of our continuing effort toward the development of useful synthetic methodologies, 93 we report an efficient process for synthesis of various β-amino carbonyl compounds in the Mannich reaction of acetophenone with aromatic aldehydes and aromatic amines using nanostructured ZnO, nanosulfated zirconia, nano-γ-alumina, and nano-ZSM-5 zeolites as the catalyst.
Results and Discussion
First, we compared the results of β-amino carbonyl compounds synthesis from acetophenone, benzaldehyde, and aniline with the presence of nanostructured ZnO, nanosulfated zirconia, nano-γ-alumina, and nano-ZSM-5 zeolites as the catalyst (Table 1). The results showed that nanocrystalline sulfated zirconia (SZ) promoted the reaction more effectively than nanostructured ZnO, nano-γ-alumina, and nano-ZSM-5 zeolites as far as the amount of catalyst and reaction time are concerned (Table 1).
Acid-catalyzed Synthesis of β-amino Carbonyl Derivatives.a

In a systematic study (Table 1), acetophenone was added to a solution of benzaldehyde, aniline, and catalyst in ethanol, and the reaction mixture was stirred for 10 h at room temperature. Our initial experiments focused on the optimization of the amount of catalysts by using 1 equiv of benzaldehyde, 1 equiv of acetophenone, 1 equiv of aniline, and variable amount of catalysts. In an initial endeavor, a blank reaction was carried out using 1 equiv each of acetophenone, benzaldehyde, and aniline without catalyst. These were stirred at ambient temperature in ethanol. In the absence of catalyst, after 24 h, only 30% of the expected product was obtained. To further improve the yield and to optimize the reaction conditions, the same reaction was carried out in the presence of (10 mol %) of catalyst under similar conditions. A tremendous improvement was observed, and the yield of product was dramatically increased up to 90% after stirring the mixture for only 5 h.
To minimize the formation of byproducts and to achieve good yield of the desired product, the reaction is optimized by varying the amount of catalyst (5, 10, and 15 mg), an increase in the amount of nano-SZ from 5 to 15 mg increased the yield of the desired product to a great extent (83–94%, Table 2, entries 2–4). The percentage yield of the product with 5, 10, and 15 mol % of nano-ZnO as a catalyst are 75%, 90%, and 70%, respectively (Table 2, entries 5–7). For the nano-γ-alumina and nano-ZMS-5 as the catalyst, when the catalyst content was increased to 15 mg, the product yield decreased to 74% (Table 3, entry 10) and 68% (Table 2, entry 13) respectively. Therefore, it was found that the use of 10 mol % of the catalyst was sufficient to promote the reaction, and greater amounts of the catalyst did not improve the yields.
Mannich Reaction of Acetophenone, Benzaldehyde, and Aniline Catalyzed by Different Catalysts.

aYields after isolation of products.
Comparison of the Efficiency of Nanocrystalline SZ with Other Reported Catalysts in the Synthesis of β-amino Carbonyl Derivatives.a
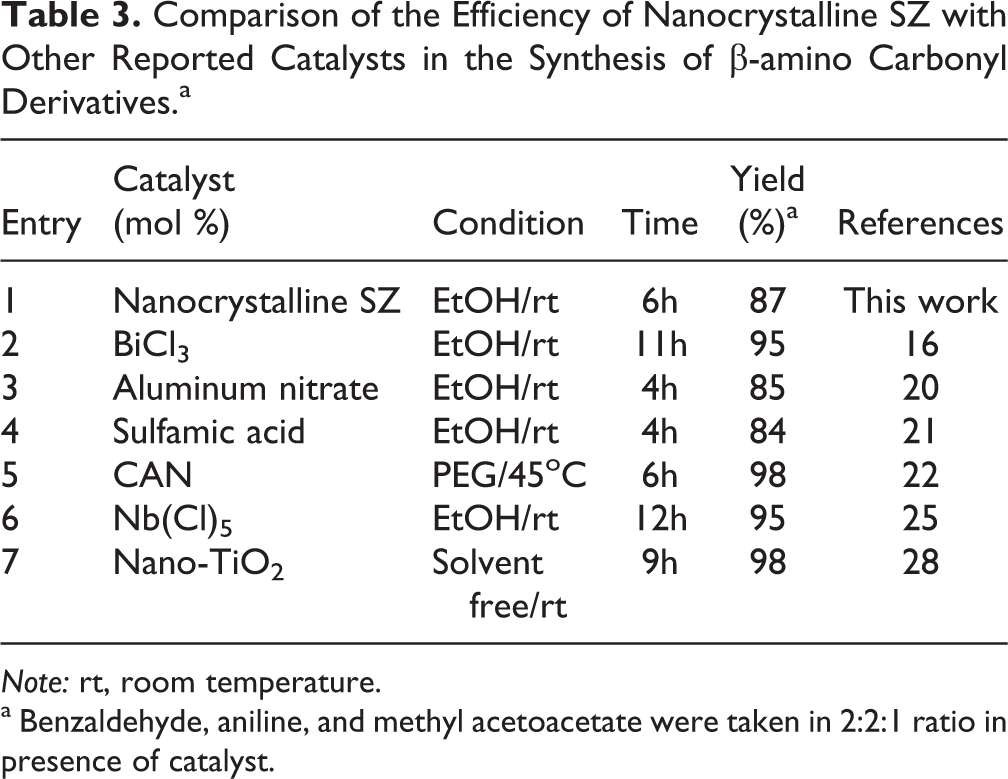
Note: rt, room temperature.
a Benzaldehyde, aniline, and methyl acetoacetate were taken in 2:2:1 ratio in presence of catalyst.
The effect of temperature was also studied. Mannich reaction was very sensitive to the reaction temperature: the high temperature could improve the reaction rate and shorten the reaction time, because the Mannich base is unstable at elevated temperature and also because one of the reactants (aldehyde) oxidizes at high temperature in the presence of catalyst (Table 1).
It was found that decreasing the temperature could accelerate the reaction and that the room temperature was an appropriate condition for one-pot three-component Mannich reaction.
Next, we examined the scope of the reaction by using various aromatic amines and aryl aldehydes. Various aldehydes bearing different substitutes, such as para-Me, MeO, OH, Cl, Br, and NO2 on the aryl rings were all suitable for the reaction. In addition, aromatic amines bearing para-Me, MeO, Cl, Br, and NO2 were also favorable to the reaction.
It is worth noting that 4-nitrobenzaldehyde catalyzed by nanocrystalline sulfated zirconia (SZ) failed to give the desired three-component Mannich reaction, whereas aromatic amine bearing electron-withdrawing substituents such as NO2 could give the desired adducts in good yields (Table 1, entries 4 and 10).
The reactivity order of these amines was 4-nitroaniline > 4-choloroaniline > 4-bromoaniline > aniline > 4-methylaniline > 4-methoxyaniline, indicating the importance of the electronic nature of the amines. When using 4-methyl aniline as a substrate, the yield of β-amino carbonyl compounds was nearly the same as that using aniline as a substrate (Entry 11).
Not only benzaldehyde but also substituted aldehydes such as 4-cholorobenzaldehyde 4-hydroxybenzaldehyde, 4-methylbenzaldehyde, and 4-methoxybenzaldehyde worked well (entries 1–7). However, when aryl aldehyde bearing electron-withdrawing substituent was subjected to the reaction under standard conditions, there was a lower yield of the desired adducts formed (Table 1, entry 4).
Table 3 compares the efficiency of present method with the efficiency of other methods in the synthesis of piperidines derivatives, respectively. As it can be seen, SZ shows better efficiency than other methods.
The effect of different solvents on reaction rate as well as yields of products was also investigated, and the results are summarized in Table 4. Acetonitrile and ethanol provided excellent yields and proved to be the solvent of choice, whereas dichloromethane afforded lower yields. The reaction in toluene afforded very poor yields while the use of THF and 1,4-dioxane could not effectively catalyze the reaction.
Effect of Solvent on the Reaction Times and Yields.

Note: THF, tetrahydrofuran.
aAcetophenone (1 mmol), aromatic aldehyde (1 mmol), and aromatic amines (1 mmol) in the presence of catalyst at room temperature in various solvents.
bReaction time monitored by thin-layer chromatography (TLC).
cIsolated yield.
One of the most important advantages of heterogeneous catalysis over the homogeneous counterpart is the possibility of reusing the catalyst by simple filtration, without loss of activity. The recovery and reusability of the catalyst was investigated in the product formation. After completion of the reaction, the catalyst was separated by filtration, washed 3 times with 5 mL acetone, then with doubly distilled water several times and dried at 110°C. Then the recovered catalyst was used in the next run.
Experimental
Instruments and Characterization
All reagents were purchased from Merck and Aldrich and used without further purification. Montmorrilonite K10 was purchased from Aldrich. Products were characterized by spectroscopy data (IR, FTIR, 1 H NMR, and 13 C NMR spectra), elemental analysis (CHN), and melting points. A JASCO FT/IR-680 PLUS spectrometer was used to record IR spectra using KBr pellets. NMR spectra were recorded on a Bruker 400 Ultrasheild NMR, and dimethyl sulfoxide (DMSO)-d6 was used as the solvent. Melting points reported were determined by open capillary method using a Galen Kamp melting point apparatus and are uncorrected. Mass spectra were recorded on a Shimadzu Gas Chromatograph Mass Spectrometer GCMS-QP5050A/Q P5000 apparatus.
Catalyst Preparation
Synthesis of Nanocrystalline Sulfated Zirconia
Nanocrystalline sulfated zirconia has been prepared by one-step sol–gel technique. 94 A typical synthesis involves the addition of concentrated sulfuric acid (1.02 mL) to zirconium n-propoxide precursor (30 wt%) followed by the hydrolysis with water. After 3 h aging at room temperature, the resulting gel was dried at 110°C for 12 h followed by calcination at 600°C for 2 h.
Synthesis of Nanostructured ZnO
Nanostructured ZnO has been prepared by one-step sol–gel technique. 95 In a typical procedure, mixtures of ethanol, diethanolamine (DEA), and zinc acetate dihydrate were prepared. The concentration of zinc acetate dihydrate in solvent was 0.2 M. The molar ratio of zinc acetate dehydrate and DEA was 1.0. The pH of the mixture was about 9. When the zinc acetate crystals were dissolved completely, sodium hydroxide (NaOH) pellets were added to the solution to increase the pH of the mixture to about 11. The resultant solution was then transferred into a Teflon-lined stainless steel autoclave that was sealed and maintained at 130°C for 24 h.
Synthesis of Nano-γ-Al2O3 Catalyst
The nano-γ-Al2O3 was prepared by sol–gel method according to a procedure described. 90 In a typical experiment, aluminum nitrate (15.614 g) was added to 400 mL of deionized water. Similarly, solution of sodium carbonate is prepared by dissolving (7.95 g) in 400 mL of deionized water; 200 mL of deionized water is taken in a 2 L capacity round-bottom flask and stirred well using magnetic stirrer. Then sodium carbonate and aluminum nitrate solutions are added to 200 mL of deionized water (from separate burettes) dropwise.
The temperature was maintained 70°C during experiment. The pH after precipitation was found to be in the range of 7.5–8.5. The mixture was stirred for 4 h. The digested precipitates were filtered and re-dispersed again in 2 L of hot deionized water, filtered and finally washed with ethanol first followed by acetone to avoid contamination of “Na” ions, and air-dried at room temperature. The dried precipitates were calcined in a furnace at 550°C for 5 h to produce nanosized γ-Al2O3 powders.
Synthesis of Nano-ZSM-5
For synthesis of nano-ZSM-5, tetrapropyl ammonium hydroxide and tetra ethyl ortho silicate were the sources of aluminum and silicon, respectively. Nano-ZSM-5 zeolite was synthesized according to the procedure described earlier. 96 The components were mixed with constant stirring. After adding all the ingredients, the solution was left to hydrolyze at room temperature for 48 h. The gel thus obtained was heated at 80°C to evaporate water and ethanol formed during reaction. The obtained solution was charged into a Teflon-lined stainless steel autoclave under pressure and static conditions at 170°C for of 48 h. The solid phase obtained was filtered, washed with distilled water several times, dried at 120°C, and then calcined at 550°C for 12 h.
Characterization
X-ray diffraction pattern were recorded on diffractometer (Philips X’pert) using CuKα radiation (λ = 1.5405 Å), angle range was between 0 and 80° (Figure 1), Crystallite size of the crystalline phase was determined from the peak of maximum intensity (2θ = 30.18) by using Scherrer formula, 97 with a shape factor (K) of 0.9, as follows: crystallite size = K.λ/W.cosθ, where W = Wb − Ws, Wb is the broadened profile width of experimental sample and Ws is the standard profile width of reference silicon sample. FT-IR spectra of the catalysts were recorded by FT-IR spectrophotometer in the range of 400–4000 cm−1 with a resolution of 4 cm−1 by mixing the sample with KBr (Figure 2).
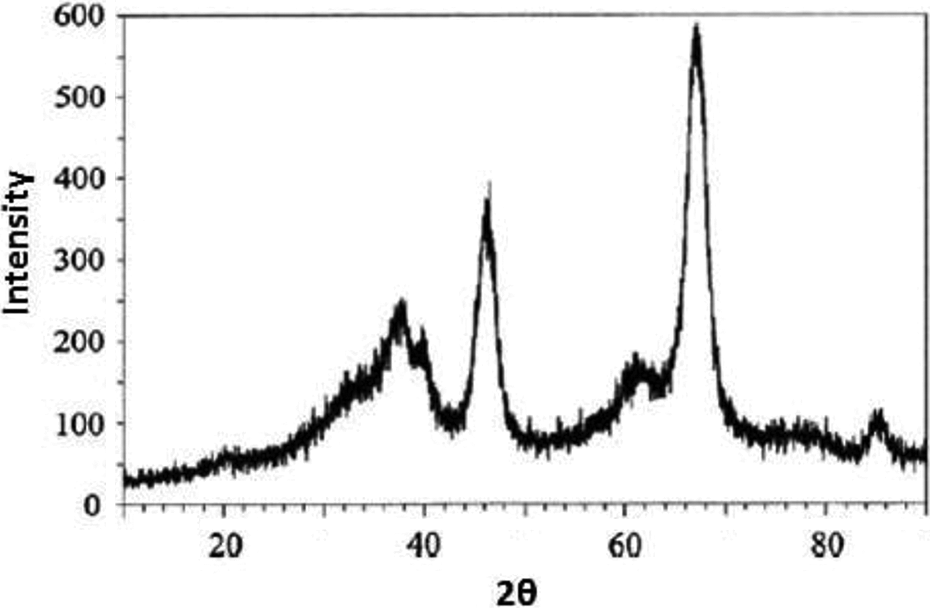
XRD pattern of nano-γ-alumina catalyst.

FT-IR spectra nano-γ-alumina catalyst.
Specific surface area, pore volume, and pore size distribution of sulfated zirconia samples calcined at 600°C were determined from N2 adsorption–desorption isotherms at 77 K (ASAP 2010 Micromeritics). Surface area was calculated by using Brunauer–Emmett–Teller (BET, adsorption isotherm) equation; pore volume and pore size distribution were calculated by Barrett–Joyner–Halenda (BJH) method. 98
The samples were degassed under vacuum at 120°C for 4 h, prior to adsorption measurement to evacuate the physisorbed moisture. The detailed imaging information about the morphology and surface texture of the catalyst was provided by scanning electron microscopy (SEM; Philips XL30 ESEM TMP), a part of the spectra data has been published in our previous work. 93
Typical Procedure
Synthesis of β-amino Carbonyl Compounds Derivatives
To a mixture of ethyl acetophenone (1 mmol), aromatic aldehyde (1 mmol), and aromatic amines (1 mmol) in ethanol (5 mL), catalyst was added and the reaction mixture was stirred at room temperature with a magnetic stirrer during the mentioned time (Table 1). When the reaction was completed as indicated by thin-layer chromatography (TLC), after completion of the reaction as indicated by TLC, the insoluble catalyst was separated by filtration. And the crude products were purified by recrystallized with ethanol to give the corresponding compounds.
Synthesis of 1,3-Diphenyl-3-(phenylamino)propan-1-one
MP 170–172°C. IR (KBr, cm−1): 3399, 3024, 2974, 1672,1598, 1515, 1295, 1080, and 512;
1
H NMR (CDCl3): 7.91–7.90 (m, 2H), 7.57–7.07 (m, 10H), 6.67–6.55 (m, 3H), 5.00 (dd, 1H),
Synthesis of 3-((4-Chlorophenyl)amino)-1,3-diphenylpropan-1-one
MP 112–114°C. IR (KBr, cm−1): 3390, 3035, 1674, 1596, 1515, 1377, 1292, 1222, 1080, 1002, 929, and 516;
1
H NMR
Conclusion
In conclusion, we have demonstrated an efficient, versatile, and convenient method for the synthesis of β-amino carbonyl compounds in the Mannich reaction of acetophenone with aromatic aldehydes and aromatic amines. A comparison of the catalytic efficiency of nanosulfated zirconia, nanostructured ZnO, nano-γ-alumina, and nano-ZSM-5 zeolites with the nanosulfated zirconia exhibiting greater activity has also been demonstrated. Compared to previously reported methods, the mild reaction conditions, high yields, easy workup, clean reaction profiles, and lower catalyst loading render this approach as an interesting alternative to the existing methods.
Footnotes
Acknowledgment
Supports from the Payame Noor University in Isfahan research council and helps of Isfahan University of technology are gratefully acknowledged. One of the authors (A. Teimouri) is also grateful to Dr. Alireza Najafi Chermahini for providing necessary facilities and encouragement during the course of this work. Thanks are also due to Mrs. Shahraki and Mr. Narimani for recording the FT-IR spectra of the compound.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
