Abstract
Owing to its rapid advancement in nanotechnology, latest publications (recent 5 years) related to zinc oxide (ZnO) in various aspects and applications have been reviewed. Trend has shifted to the innovation of pristine ZnO nanostructures functionalized with elemental dopants and heterojunction to improve the photoactivities on micropollutant degradation and heavy metals removal as well as the antibacterial effect towards microorganisms. The fundamentals of improved photoactivities (i.e., low recombination rate of charge carrier and effective charge transfer) and enhanced antibacterial properties (i.e., dissolution of ions and generation of reactive oxygen species) are accentuated. Subsequently, a detailed examination is undertaken to elucidate the key impacts of ZnO on actual wastewater treatment systems. This review concludes with a consolidated overview on the recent advances of ZnO for environmental wastewater decontamination and the future prospects on tailoring ZnO based on their photocatalytic and antibacterial properties to match various expectations from practical applications while minimising the adverse impacts on surrounding environment.
Keywords
Introduction
Nanotechnology is currently at the forefront of scientific research and technological development aimed at synthesizing customised nanostructured materials with unique physicochemical properties, surface structures, and quantum confinement effects. These excellent intrinsic and extrinsic properties have made nanomaterials ubiquitous in modern society and utilised for advance consumer products and multitudinous industrial applications. These include their applications as key ingredient in personal care products, paints and pigments, medical devices, electronics and electrical components, food packaging, construction materials, adsorbents and catalysts used for environmental remediation.1–8 Concurrently, the vast utilisation of these products containing nanomaterials has also placed increasing threat to water resources due to their associated diffuse and non-diffuse pollution issues. These have prompted for a critical review on the current practices surrounding the synthesis of nanostructured materials and their mode of applications, more importantly the “know-how” knowledge on controlling the extent of their dominant material properties.9,10 This complete knowledge is critical to ensure that an optimum state of nanomaterials’ properties is used for their related applications without presenting much detrimental impacts when they present in the environment as “nanopollutants”.
Among the widely used nanomaterials, zinc oxide (ZnO) is a promising candidate that finds wide applications in nanostructured material systems owing to its favourable indirect band gap, large free-exciton binding energy, excellent photocatalytic and antibacterial properties.11–14 Due to this wide range of properties and applications, ZnO is considered as a multifunctional material with significant importance in electronics, optics, environmental technology, and healthcare. As compared to titanium dioxide (TiO2), ZnO tends to absorb more quantum of light and thus, considerable amount of light could be utilised for catalytic reaction. 15 In fact, there are several drawbacks to be considered, such as the needs of UV-light activation, poor electron transfer rate, rapid recombination of electron-hole pairs (e−-h+) and low stability. Nonetheless, the material characteristics in ZnO also facilitate a high potentiality for tunable properties via the rational design of nanostructures, incorporation of foreign chemical elements, and heterojunction coupling with other semiconductors.16–18 On the other hand, the multi-faceted and tunable properties in ZnO also leads to their prevalence for wastewater decontamination encompassing the treatment of emerging organic pollutants and photo-deactivation of bacteria and other microorganisms.9,19–22 This is based on the photoexcitation mechanism of valence band (VB) electrons in ZnO resulting in the primary oxidative holes (h+) and other secondary reactive oxygen species generated, such as hydroxyl radicals (•OH), superoxide anions (•O2–) and other reactive oxygen species (ROS).
Since the first study performed by Kraeutler and Bard 23 on the photocatalytic decomposition of organic compounds using ZnO, there were many studies published which covering a range of topics from the studies on the influence of key experimental parameters (i.e., photocatalyst dosage, concentration of pollutant in aqueous solution, pH, and light intensity) to the design of visible-light responsive ZnO nanostructures for the effective photodegradation of emerging micropollutants and removal of heavy metals. A higher effective surface-to-volume ratio along with preferential exposure of polar crystal surfaces are some of the key approaches in the rational design of ZnO nanostructures, which could improve both the surface adsorption and photo-reactivity.24–26 Whilst the creation of surface vacancies in ZnO nanostructures via cationic and/or anionic doping introduces Fermi energy level intra-bands that could simultaneously narrow down the band gap and extend their light absorption into the visible-light spectrum.27–30 Another compelling approach for improving the photoactivity is through the appropriate band potential alignment between ZnO and other semiconductors, leading to the formation of heterojunction ZnO structure that facilitates an improved interfacial charge transfer process.31–33
In a different aspect, the potentiality of ZnO nanostructures in demonstrating an exceptional antibacterial ability is of immense interests for the fields of nanobiotechnology and related applications. For instance, ZnO nanostructures are playing a profound role in environmental remediations by controlling the microbial growth and outbreak, reducing microbial contamination as well as complete photo-inactivating various microorganisms of concerns.34,35 The antibacterial assessment of ZnO nanostructures towards simple model bacterial provides vital underpinning science for the following comprehensive analysis of complex microbial community within the ecosystem. From the review of scientific of literature, it was revealed that the major mechanisms for bacterial inhibitory effects in ZnO nanostructures are through the generation of intracellular ROS and dissolution of Zn2+ ions.36,37 Therefore, various surface and advanced modifications of ZnO nanomaterials can be effectively tailored to control the extent of ROS-mediated effect and leaching of metal ions towards bacterial cells. Consequently, these are causing cell lysis and death through physical attachment to cell membrane and wall, disruption of cell membrane, permeation into cell, interruption of transmembrane electron transport, denaturation of proteins, damages of mitochondria and deoxyribonucleic acid (DNA).36,38–40
Owing to the utilisation of ZnO nanomaterials in a wide range of consumer products and industrial applications, it is inevitable for the intentional and/or unintentional releases of ZnO-containing nanostructures into the wastewater and eventually enter the municipal wastewater treatment plants.41,42 At present, specifically, there are a multitude of laboratory- and field-based research being undertaken to assess the full impacts of ZnO nanostructures on the wastewater treatment systems (WTS). The presence of ZnO nanostructures in the wastewater cycle and WTS could potentially enhance or suppress their photocatalytic and antibacterial effects, and these may have a profound influence over the treatment efficiency of WTS which warrants proper scientific validation. Since the ZnO inhibitory action is generally impacting on the entire microbial community found in biological WTS, it is difficult to distinct specific antimicrobial activity to individual microorganism. 43 Additionally, further uncertainties associated with the fate and behaviour of ZnO nanostructures influenced by a series of physical and chemical transformation processes has become a paramount challenge on determining their implications on practical WTS.44–47 A systematic study on the environmental fate and toxicity of engineered nanoparticles (ENPs) funded by European Union (EU) was conducted to fill knowledge and methodological gaps on the assessment of environmental risks posed by ENPs. A series of studies was carried out to analyse the interactions of ENPs with abiotic and biotic entities, generate predictive models for examining the fate and behaviour of ENPs through wastewater treatment, determine the acute and chronic ecotoxicity of ENPs.
The number of publications on ZnO related studies has increased drastically over the past 20 years and more than 25,000 papers have been published in 2020 until lately. Many reviews were done separately on discussing the engineering of ZnO and ZnO-based nanostructures (e.g., crystal structure, lattice parameters, electronic band structure, etc.),48,49 its advanced applications in sensing devices and electronic components, 50 and photocatalytic and biological studies.51,52 However, there is a lacking of consolidated review to cover the applications of morphological-synthesized ZnO nanostructures on real world environmental concerns. Among the published works (adapted from Scopus), there are only around 3% publications which are focusing on the environmental science research scope since 2000. The environmental science includes the aspects on environmental protection and remediation, water treatment, and biological lives. Nevertheless, the key findings from these studies are not fully correlated and attuned to the currently available best management practices to fully exploit the characteristics of ZnO nanostructures for the exploitation of optimal functionality in WTS.
Thus, this critical review focuses on a large spectrum of literature published in the immediate past five years (2017–2022) on ZnO nanostructures associated with specific photocatalytic and antibacterial properties. The review first unveils by presenting a well-defined view on the state-of-the-art development of ZnO-based photocatalysts as well as their various advanced modification variants. The fundamental principles associated with the advanced modifications of pristine ZnO through rational design of nanostructures, surface functionalisation with elemental dopants, and formation of multi-component heterojunction photocatalysts are outlined. This is followed by a critical review on the latest applications of pristine and surface modified ZnO nanostructures with excellent structural design, improved crystalline structures, superior optical and electronic properties for photocatalytic applications. Following this, the antibacterial properties and efficacies as displayed by a multitude of ZnO photocatalysts are assessed against a wide spectrum of bacteria and microorganisms while their antimicrobial mechanisms are systematically reviewed. To achieve closure in the analysis, an in-depth exploration of the significant ramifications arising from the implementation of ZnO photocatalysts within practical wastewater treatment systems is presented. This scrutiny is particularly focused on the photocatalytic and antibacterial properties inherent to ZnO, offering a comprehensive examination of their effects. This review concludes with a comprehensive summary, key recommendations, and potential future prospects on synthesizing and tailoring the photocatalytic and antibacterial properties in ZnO photocatalysts to match various practical applications while minimising the key impacts on WTS and the environment.
Fundamental principles and modifications of ZnO photocatalysts for photo-responsive enhancement
ZnO photocatalysts with a broad intrinsic band gap (Eb ∼3.37 eV) that allow light absorption within the ultraviolet (UV) spectrum only, have largely restricted photo-responsiveness and are unable to be photon-activated at the visible light spectra i.e., which accounts for 32% of solar spectrum.53,54 Additionally, ZnO photocatalysts are also encountered with various materials’ drawbacks, such as possessing a very rapid recombination rate of photogenerated e−-h+ pairs as well as having low stability due to photochemical corrosion which have significantly impaired their quantum yields. 55
At present, the development of a highly tailored ZnO photocatalyst is accentuated on obtaining desirable optical band gap and interfacial structures for superior charge transfer kinetics.56–58 Owing to the aforementioned material properties drawbacks, several advanced modifications of pristine ZnO photocatalysts including the rational design of nanostructures, surface functionalisation with elemental dopants, and formation of multi-component heterojunction photocatalysts have been implemented to enhance its photoactivity.
Rational design of ZnO nanostructures
ZnO is a highly versatile and functional photocatalyst, which can be formed into various well-defined sizes, shapes, and morphologies.59–66 In this review, the controllable growth morphologies of ZnO are systematically discussed in terms of dimensional variables (i.e., zero-dimensional (0D), one-dimensional (1D), two-dimensional (2D), and three-dimensional (3D) nanostructures) with respect to the physical elongation and/or extension of particles in any direction within a space. During the rational design, this involves the morphological alterations of pristine ZnO nanomaterials while maintaining their pure states. Eventually this will lead to the variation of physicochemical properties (Figure 1), which is the main contributing factor for photoactivity enhancements.
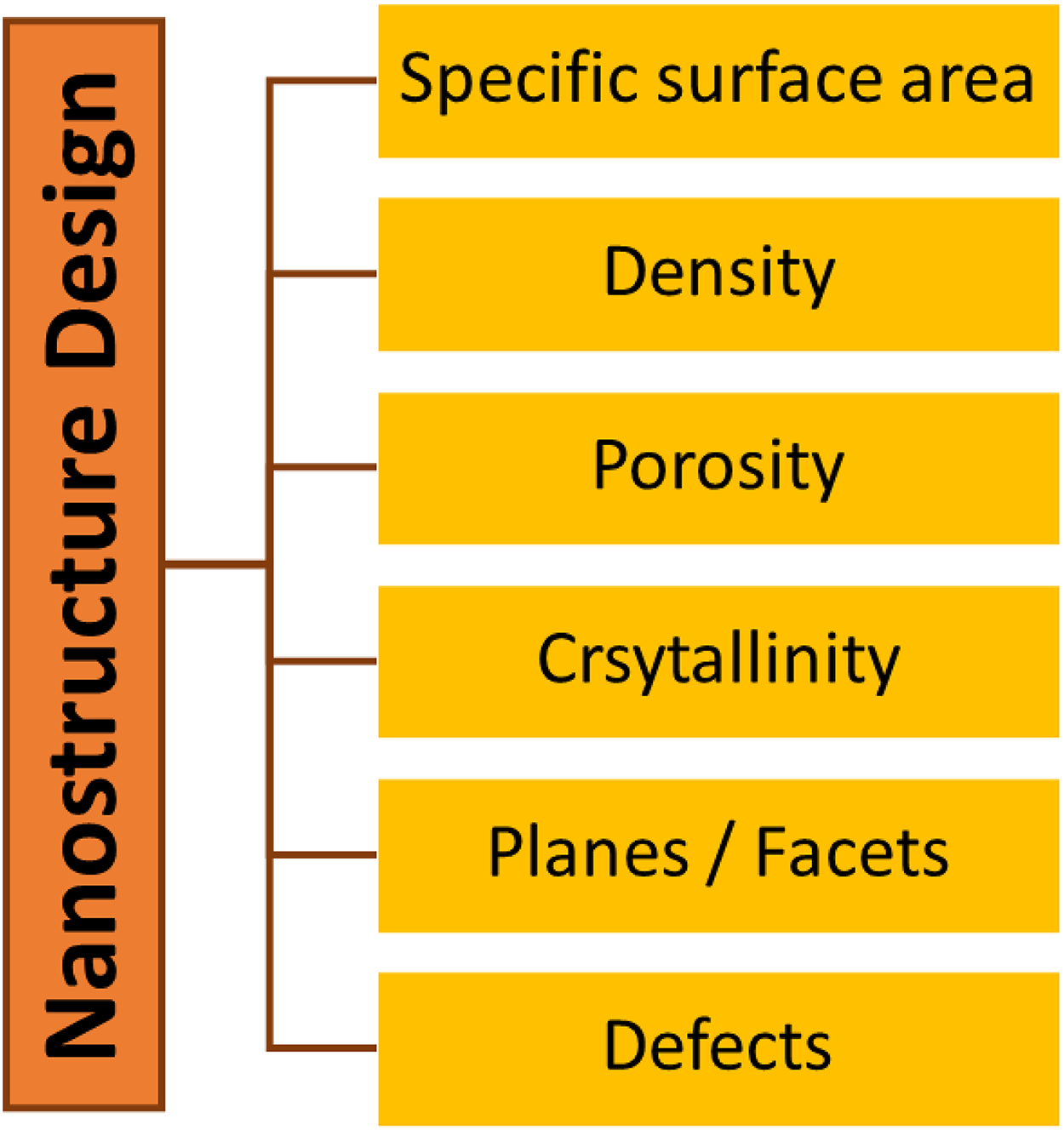
The influence of rational design approach on the changes in physicochemical properties of ZnO nanostructures.
An 0D nanostructure refers to the definite point in a space that does not have any dimensional attributes, which are made up of nearly spherical and uniform discrete particles. Various 0D-ZnO nanostructures, including quantum dots, solid nanocrystals, nanoparticles, hollow nanospheres, and core-shell particle arrays, have been fabricated and studied by many research groups due to their relatively small sizes with an abundance of active adsorption surfaces.67–70 Among the reported synthesis approaches, the solution phase method is the simplest, low-energy requirement, and its easiness in controlling the size and structural morphology.71,72 Chen et al. 73 reported the synthesis of ZnO spherical nanoparticles with tunable sizes assembled from ultrafine particles. Due to the aggregation of ultrafine particles, these ZnO nanospheres (Figure 2(a)) were fabricated by calcining polyacrylic acid (PAA)-ZnO aqueous solution at 500 °C for 4 h. In order to effectively control the degree of aggregation, a multi-stage solution phase method to obtain highly-uniform ZnO nanostructures with a narrower particle size distribution range has been reported by Yang et al.. 74 They have successfully developed monodispersed colloidal ZnO nanospheres via a two-stage solution route designated the seed solution method.
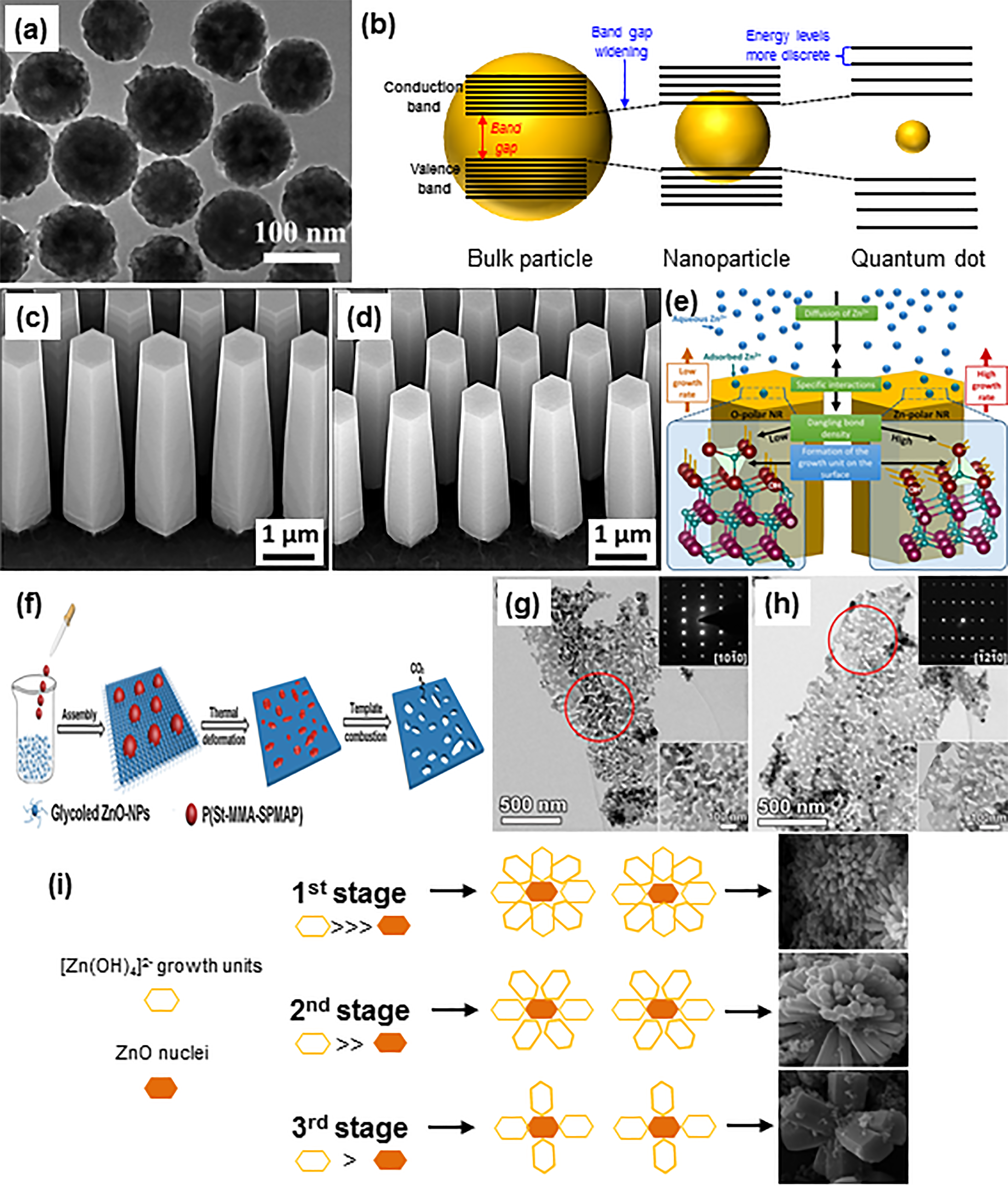
(a) TEM image of 0D-ZnO spherical nanoparticles. Reprinted with permission from ref. 73 (b) Quantum confinement effects for different particle sizes causing energy band gap widening and more discrete energy levels. FESEM images of 1D-ZnO nanorods grown at on polar (c) Zn- and (d) O-terminated surface planes. (e) Schematic representation of the possible mechanisms for the polarity-dependent nucleation and growth of ZnO nanorods. Reprinted with permission from ref. 81 (f) Schematic illustration of the 2D-ZnO nanosheets synthesis, (g) and (h) TEM images of 2D-ZnO nanosheets with their corresponding enlargement of the indicated areas in red circle (lower inset) and SAED (upper inset). Reprinted with permission from ref. 90 (i) Schematic illustration of growth mechanisms of different 3D-ZnO nanoflowers. Reprinted with permission from ref. 102
Reduced particle size of 0D-ZnO would result in multiple benefits, such as: (1) higher specific surface area for a better adsorption capacity; (2) increased particle density for improved photon absorption; and (3) reduced surface path distance in photogenerated e−-h+ pairs for efficient charge scavenging.75–77 Whilst down to the sub-nanometer range, the quantum confinement effect will dominate due to the confined particle radius-exciton (e−-h+ pairs). For ZnO material system, the quantum confinement effect occurs when the particle size is smaller than the Bohr exciton radius of bulk ZnO at 2.34 nm and this would lead to a higher band gap energy between ground and excited states. The quantum confinement effect is proven to be size-dependent as the broadening of energy band gap and blue-shift in the transition energy are observed when the size of ZnO nanostructures decreases. Such a phenomenon demonstrates the origin effects of increasing significance in exciton absorption peak, spectral widening and asymmetry of the optical phonons due to the spatial confinement of phonon in ZnO quantum dots.78,79 Figure 2(b) shows the quantum confinement effect on broadening the energy band gap with discrete energy levels for different particle sizes. In short, the advancement of 0D-ZnO nanostructures with the ease of emission wavelength and energy manipulation has proven to be highly versatile for photocatalysts design and related applications.
Theoretically, a 1D nanostructure confines to points on a line segment which have only length measurement in a space. The wurtzite structure of ZnO consists of four face terminations including two polar surfaces (i.e., Zn-terminated (0001) and O-terminated (000
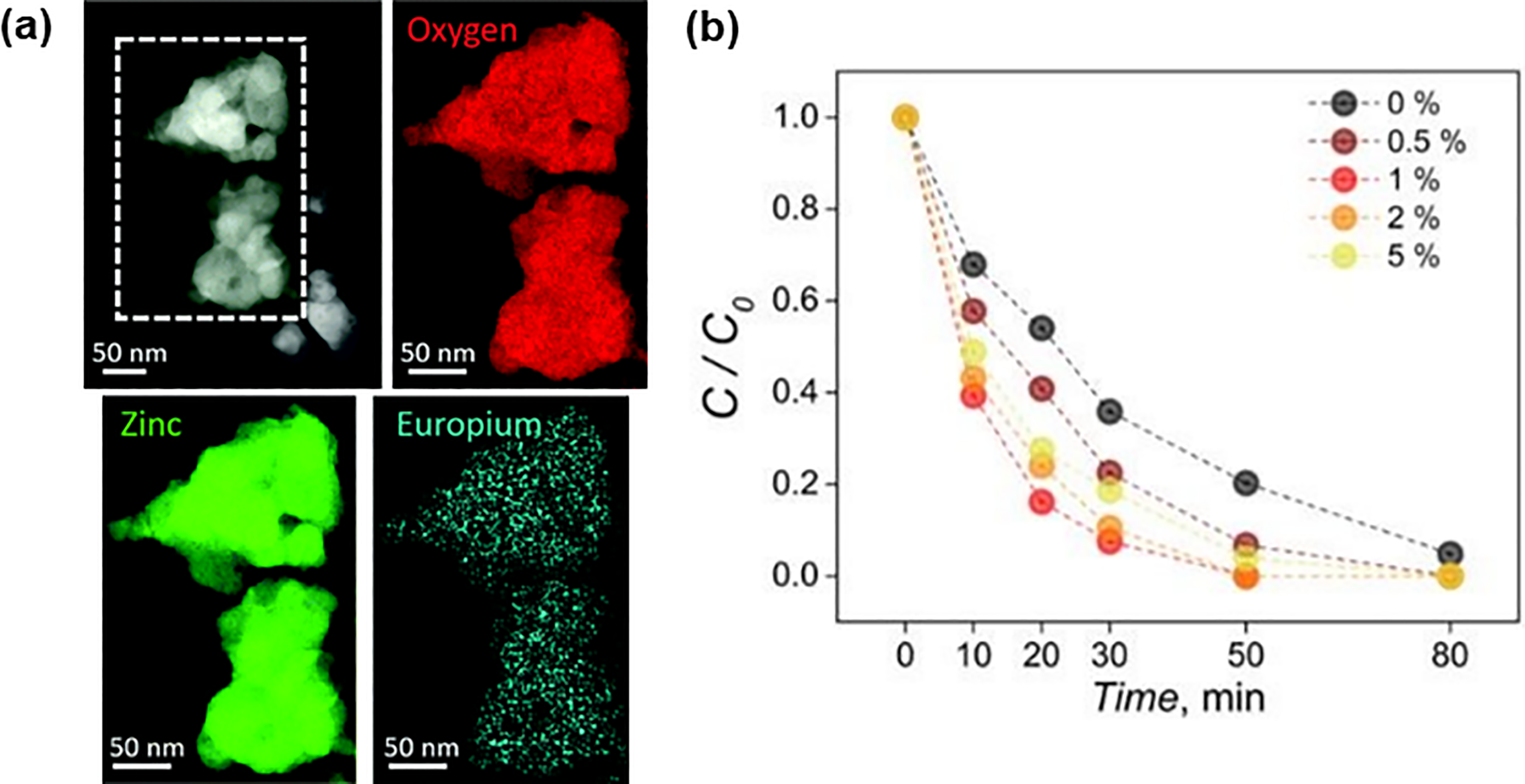
(a) STEM image and EELS elemental mapping on 1% Eu3+-doped ZnO nanosponges. (b) Photodegradation rates of RhB using Eu3+-doped ZnO nanosponges (doping 0 to 5%) upon 80 min of UV irradiation. Reprinted with permission from ref. 112
As for the 2D nanostructure, it possesses a single-layer crystallite planar structure at which the width and height dimensions are outside of the nanometric size range. This confined dimensional structure is ideal for mass transport and electron transfer investigations owing to its specific surface atomic arrangement and higher specific surface area.
90
One of the most intriguing properties for 2D nanostructures is the high-exposure degree of polarised (0001) plane which has been evolved as the main building block for many functional nanodevices.91,92 Hierarchical nanoplates, nanosheets, and nanodisks are some examples of the ultrathin-layered 2D-ZnO nanostructures which have been successfully prepared via a wide variety of physical and wet chemical methods. The introduction of intrinsic surface defects on sheet-like planar structures could enhance the performance of 2D-ZnO nanostructures in term of their photocatalytic and electrochemical responses.25,93,94 It is notable that the concentrated oxygen vacancy defects formed on the nanosheet structure with high surface-to-volume ratio would beneficially act as charge carrier traps, where the recombination rate of photogenerated e−-h+ pairs could be suppressed effectively. A comparative study on the concentration of oxygen vacancy defects that exist in the dominating exposed crystal facets (0001) and (10
A 3D nanostructure is composed of three dimensions, namely length, width, and depth, which can be further categorised into polyhedron and curved solid. Polyhedron is defined as regular solid (i.e., cube, prism, and pyramid) with straight edges, flat surfaces, and sharp vertices; whereas curved solid (i.e., cylinder, cone, and sphere) has curves and/or round edges found in the geometry. There are a number of hierarchical 3D-ZnO structures that can be self-assembled from using 1D- and 2D-ZnO nanostructures as the building blocks. The resultant 3D-ZnO structures have been extensively used in multitudinous practical applications owing to their high stability and larger surface area for enhanced reactions.65,96–98 The formation of complex 3D structures depends on the aggregation of ZnO nuclei during the initial nucleation process followed by the secondary growth process from the defects.
99
The crystal surfaces and boundaries containing defects with a lower surface energy serve as the active sites for secondary nucleation process when there are abundant Zn growth units. Besides, surface hydrophobicity and hydrophilicity are imperative in determining the assembly process of particles and therefore, influencing the final orientation outcomes of the 3D-ZnO structures. To gain an insight into the crystal growth and nucleation process for 3D particles’ aggregation structure, Shen et al.
100
computed the surface chemistry of different interface systems along with their interactions. The oriented attachment resulted from the surface hydroxylation as well as the surface phenomena at the interface of ZnO(10
Summary on the assembly of different Zno nanostructures using a wide range of precursors and synthesis methods along with their matching applications.
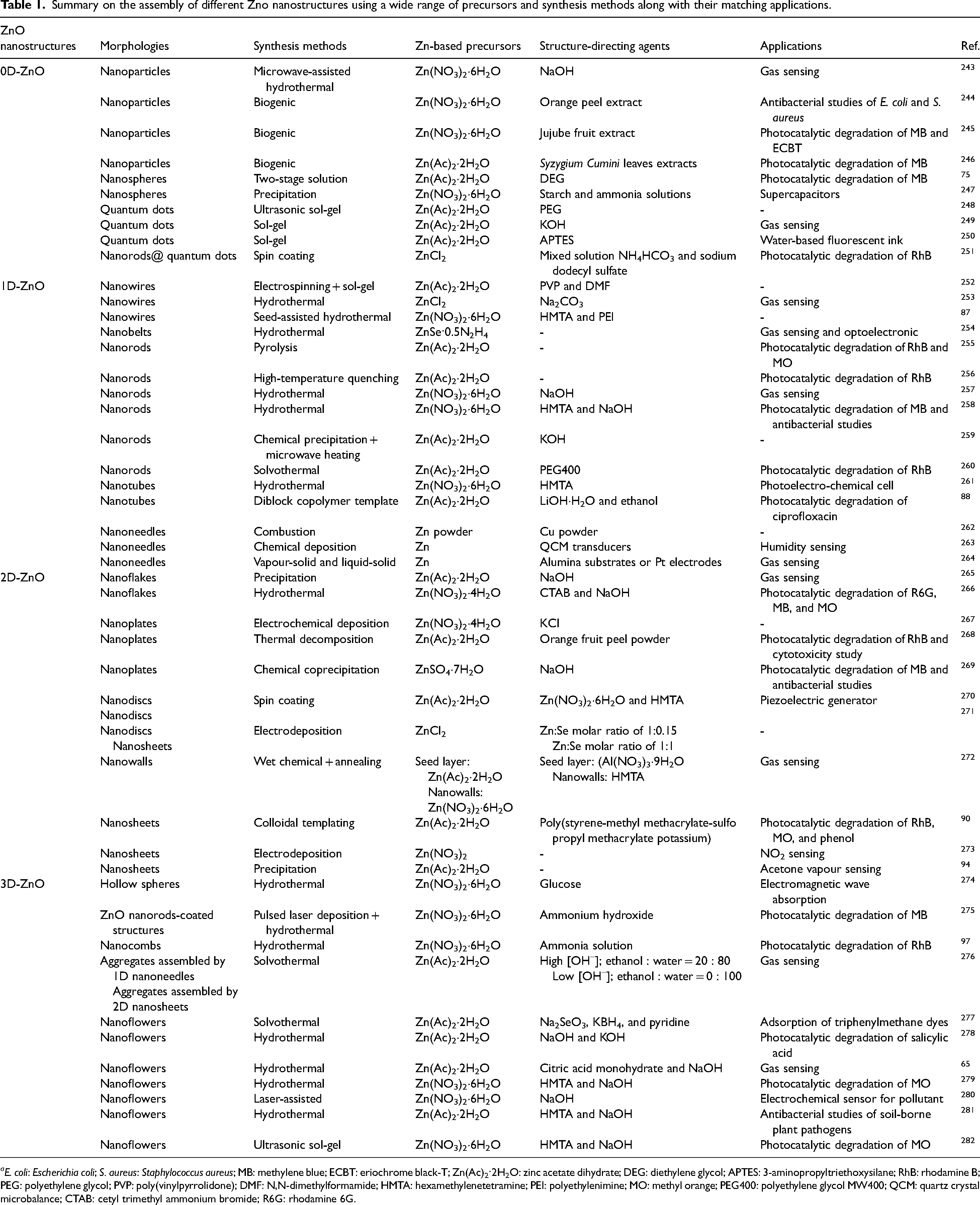
aE. coli: Escherichia coli; S. aureus: Staphylococcus aureus; MB: methylene blue; ECBT: eriochrome black-T; Zn(Ac)2·2H2O: zinc acetate dihydrate; DEG: diethylene glycol; APTES: 3-aminopropyltriethoxysilane; RhB: rhodamine B; PEG: polyethylene glycol; PVP: poly(vinylpyrrolidone); DMF: N,N-dimethylformamide; HMTA: hexamethylenetetramine; PEI: polyethylenimine; MO: methyl orange; PEG400: polyethylene glycol MW400; QCM: quartz crystal microbalance; CTAB: cetyl trimethyl ammonium bromide; R6G: rhodamine 6G.
Surface functionalisation of ZnO using elemental doping
From the basic and fundamentals of photocatalysis, one of the prime strategies to extend the light absorption ability of ZnO photocatalysts is through the systemic engineering of band gap structure. In this regard, the surface functionalisation of the ZnO photocatalysts by introducing elemental dopants enables the synchronous narrowing in the band gap structure of ZnO which allowing a significant optical shift and photon absorption into the visible light spectrum. The introduction of elemental dopants in ZnO for the alteration of its band gap structure could be achieved by three different means, which are: (1) lowering of the conduction band to the acceptor level via metal doping; (2) elevation of the valence band to the donor level via metal doping; and (3) formation of new localised energy levels (i.e., intrabands) within the band gap via non-metal doping. Therefore, two main types of elemental doping, namely metal doping and non-metal doping, are reviewed and discussed in this section.
Metal doping, with cobalt (Co), copper (Cu), iron (Fe), molybdenum (Mo), manganese (Mn), nickel (Ni) and others, has been widely explored to synthesize visible-light active ZnO photocatalysts. The incorporation of metal dopant in ZnO photocatalyst could reduce the band gap, increase the trapping sites for photogenerated charge carriers (reduced recombination rate), improve the conductivity, and accelerate the charge mobility.27,103–106 Furthermore, the metal-doped ZnO photocatalysts were found to possess a higher optical absorption intensity in the blue region which signifies that they could be well functionalised under visible light irradiation.107,108 More surface defects resulted from metal doping and a strong electronic interaction between the metal dopant and ZnO photocatalyst could also help to improve the photo-responsiveness of ZnO into the visible light spectrum. Over the years, the development of 1D-ZnO photocatalysts is of great interest owing to the ability to decorate metal dopants over its greater effective surface area. 109 Hsu and Chang 110 prepared silver (Ag)-doped ZnO nanorods coated on stainless steel wire meshes with proven excellent visible-light photoactivity and anti-photocorrosion properties. In relation to this, the measured high photodegradation rate of dye could be ascribed to the greater availability of surface area, enhanced hydrophilicity as well as the improved surface conductivity and charge mobility due to the presence of Ag dopant. Furthermore, the metal-doped ZnO nanostructures were also found to exhibit different degrees of photoactivities under different light irradiation conditions. For instance, Qiu et al. 111 found that the incorporation of Co2+ doping on 1D-ZnO nanorods resulted in the suppression of photoactivity under UV light illumination but with improved photoactivity under visible light illumination. It was concluded that the observed improvement in photoactivity under visible light illumination could be due to the roles of Co2+ ions on either trapping the e–-h+ pairs or facilitating the generation of photoinduced charge carriers.
Other than the 1D-ZnO nanostructures, ZnO with other morphologies have also been studied for the surface functionalisation with elemental doping. Marin et al. 112 synthesized the porous europium (Eu3+)-doped ZnO nanosponges for the photocatalytic degradation of rhodamine B (RhB) dye. As presented in Figure 3(a), electron energy loss spectroscopy (EELS) elemental mapping indicated a homogenous distribution of the Eu3+ dopants on the ZnO matrix. Photocatalytic results (Figure 3(b)) showed that 1% Eu3+-doped ZnO nanosponges achieved a complete RhB degradation within 50 min of UV illumination with reaction rate constant (k) = 0.088 min–1 as compared to the undoped ZnO with k = 0.036 min–1. The homogeneous distribution of the 1% Eu3+ concentration in the ZnO matrix decides the optimum performance of degradation rate. Further increment of Eu3+ concentration to 2 and 5% would cause phase segregation at the ZnO grain boundaries and surfaces. The enhanced photocatalytic performance with Eu3+ dopants could be ascribed to trapping of electrons from the conduction band of ZnO to the energetically favoured Eu3+ ions and therefore, leading to a reduction in the recombination of charge carriers. 113
Meanwhile, the non-metal doping of ZnO can narrow down its band gap through the incorporation these dopants in appropriate positions at O 2p states of ZnO lattice which help to raise the upper band edge of the valence state. Additionally, non-metal dopants including nitrogen (N), carbon (C), phosphorus (P), sulphur (S), boron (B), iodine (I), and fluorine (F) can also introduce a higher number of intrinsic defects, particularly oxygen vacancies (VO), on the surface of ZnO photocatalysts. Hence, it is apparent that these non-metal dopants would alter the geometric and electronic structures of the pristine ZnO photocatalysts resulting in changes in structural and optical properties as well as the corresponding visible-light photoactivity.114–117
Among the non-metal dopants, nitrogen is regarded as an efficient acceptor dopant into ZnO to achieve the p-type control. The fabrication of N-doped ZnO can generally be achieved via a direct synthesis route, where the ZnO powder is flowed with ammonia gas at elevated temperatures.118,119 The incorporation of N atoms within the crystal lattice of ZnO can hybridise both the N 2p and O 2p energy states leading to the formation of an intermediate energy level within the band gap, which could then reduce the light photon energy requirement for photoexcited state of ZnO. 120 UV-visible spectra and Thermogravimetry Differential Thermal Analysis (TG-DTA) analysis detected two chemical forms of N atoms: (1) site-N, in which N replaced O atoms in the ZnO crystal lattice; and (2) residue-N, which is originated from the residues of ZnO precursor and reflecting the existence of nitrogen/oxygen substitution in the N-doped ZnO nanostructures. Qin et al. 121 prepared different composite films by coupling ZnO and N-doped ZnO and subsequently evaluated their abilities over the photodegradation of humic acids. The photoactivity of the coupled ZnO and N-doped ZnO showed a synergistic improvement owing to that the photogenerated electrons will accumulate in the ZnO layer and holes will accumulate in N-doped ZnO upon illumination and thus, preventing the respective recombination process of e–-h+ pairs.
Carbon is a distinctive dopant due the ability of C dopant to provide substitutional doping for both metal and non-metal atoms to improve the photoactivity of ZnO photocatalyst. Liu et al. 122 prepared hierarchical C-doped ZnO flowers assembled from porous nanosheets obtained through the pyrolysis of analogous Zn-containing precursors. The alignment of porous nanosheets as the building blocks resulted in dense voids, while the in-situ C doping into the lattice structure of ZnO improved its visible light harvesting and thus, the improved photoactivity towards decomposition of the RhB in aqueous solution. Furthermore, DFT calculation confirmed that the C doping induced a red shift in the absorption edges of C-doped ZnO flowers. Another study has examined the effects of C doping on ZnO nanorods and ZnO nanospheres by observing a macroscopic trait over the much-improved photocatalytic reaction. 29 The outcomes from study suggested that uniform C doping on the ZnO crystals could effectively decrease the band gap energy, the formation of Zn−O and VO could improve the separation efficiency of photogenerated e−-h+ pairs and thereby, increasing the photocatalytic activity of ZnO.
Apart from N and C doping, sulphur doping can also modify the optical and electrical properties of ZnO due to large electro-negativity and Bohr radius differences between O and S atom.123,124 Adding S dopants for narrowing the band gap energy of ZnO could be helpful to utilise solar energy for the photocatalytic decomposition of organic pollutants. For instance, Patil et al. 124 fabricated S-doped ZnO through two-stage synthesis process and it exhibited 2 times better solar photocatalytic degradation of resorcinol as compared to that of pristine ZnO. A larger number of oxygen vacancies and defects found on S-doped ZnO were beneficial for photocatalytic reactions. Panda et al. 125 varied the input powers and ultrasonication modes to obtain S-doped ZnO with different surface morphologies. Ultrasound influences the nucleation process through cavitation and this causes solute thermolysis along with the formation of highly reactive radicals. Several bands in the range of 350–700 nm observed in the PL analysis also highlighted the role of defects in emission process due to sulphur incorporation. As denoted in Figure 4(a), the optimised crystal structures of 2 × 2 × 2 supercells for pure and all three doped ZnO models possessed nearly the same geometric parameters, inferring the relative stability of N, C, and S dopants inside the ZnO crystal lattice for substituting the O atoms. However, the shifting of absorption edges for both N- and C-doped ZnO into a lower energy region suggested that they had better absorption coefficients compared to S-doped and pure ZnO (Figure 4(b)). The stronger light absorption could be accredited to the N and C doping to increase the Fermi level electron density and generate vacant states into the band gap, mainly due to the nature of p-type doping. Following that, this could effectively narrow the band gap and facilitate the excitation of electrons, thereby elevating their photo-response to great extent in the visible and UV region. On the whole, akin to metal doping, non-metal dopants can act as electron scavengers and prevent the recombination of e–-h+ pairs and thus, freeing the positive h+ for the formation of hydroxyl radicals (•OH). These •OH and other oxidative species are important to participate in photocatalytic reaction to degrade hazardous compounds. Effective metal and non-metal doping of ZnO nanostructures will open a new vista in materials research to explore practical possibilities for implementation of advanced wastewater treatment technology, which is solar-driven photocatalysis of recalcitrant pollutants.
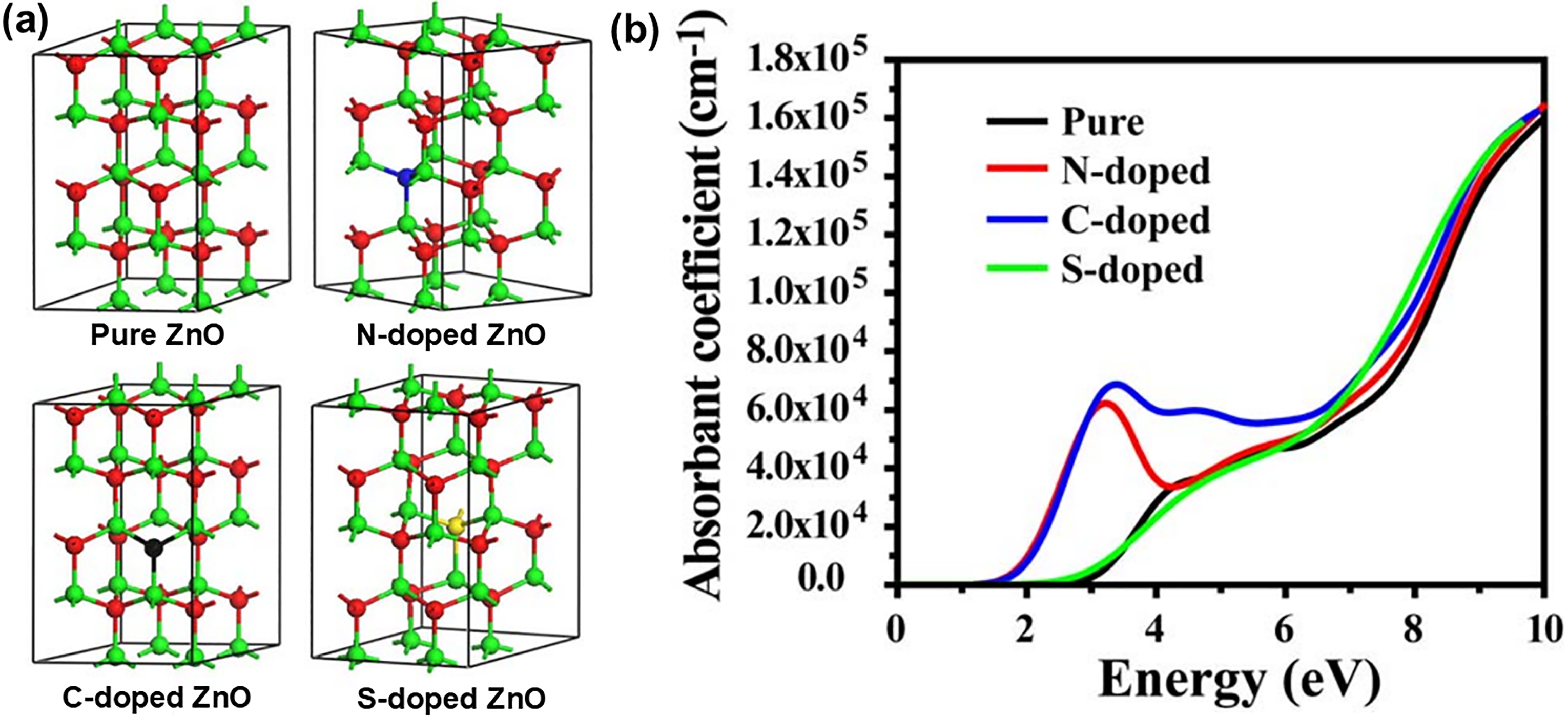
(a) Schematic illustration of the optimised crystal structures of 2 × 2 × 2 supercells for pure, N-doped, C-doped, and S-doped ZnO. Green balls represent Zn; red balls represent O; blue balls represent N; black balls represent C, and yellow balls represent S. (b) Simulated light absorption spectra of pure, N-doped, C-doped, and S-doped ZnO. Reprinted with permission from ref. 114
Heterostructuring
Heterostructuring involves the cross-transferring of photoexcited electrons and photogenerated holes between the band gap edges of two semiconductors in order to attain charge equilibrium and to improve quantum efficiency. Appropriate band gap alignment with other semiconductors through heterostructuring could effectively overcome several intrinsic drawbacks of ZnO photocatalyst, such as rapid recombination rate of photogenerated e−-h+ pairs, UV light active responsive range, and high tendency of ion dissolution.126,127 In this context, type II (staggered gap) heterostructure will be discussed in detail as this band gap alignment is the most strategic configuration with distinguished interface to enhance the charge migration and separation.
Among various types of semiconductors, tin (IV) oxide (SnO2), iron (II, III) oxide (Fe3O4), molybdenum disulfide (MoS2), zinc selenide (ZnSe), and copper sulphide (CuS) have been progressively explored to band bending at the interface of junction with different hierarchical ZnO nanostructures for the formation of type II heterostructure.32,128–130 Particularly, 1D-ZnO nanostructures have gained considerable research attention for heterostructuring with other semiconductors owing to their high surface area for light harvesting and direct transport pathway for charge transfer.131,132 Recently, Long et al.
133
synthesized triclinic iron (III) vanadate (FeVO4)-passivated ZnO heterostructured nanorods (Figure 5(a)) photoanode by virtue of spin coating followed by high temperature annealing for studying photoelectrochemical water splitting. HRTEM images in Figure 5(b) and 5(c) demonstrated the successful incorporation of ZnO nanorods (interplanar spacing = 0.260 nm corresponds to the (002) crystal plane of hexagonal wurtzite ZnO) and triclinic FeVO4 particles (interplanar spacing = 0.260 nm and 0.505 nm well-correlated to (320) and (
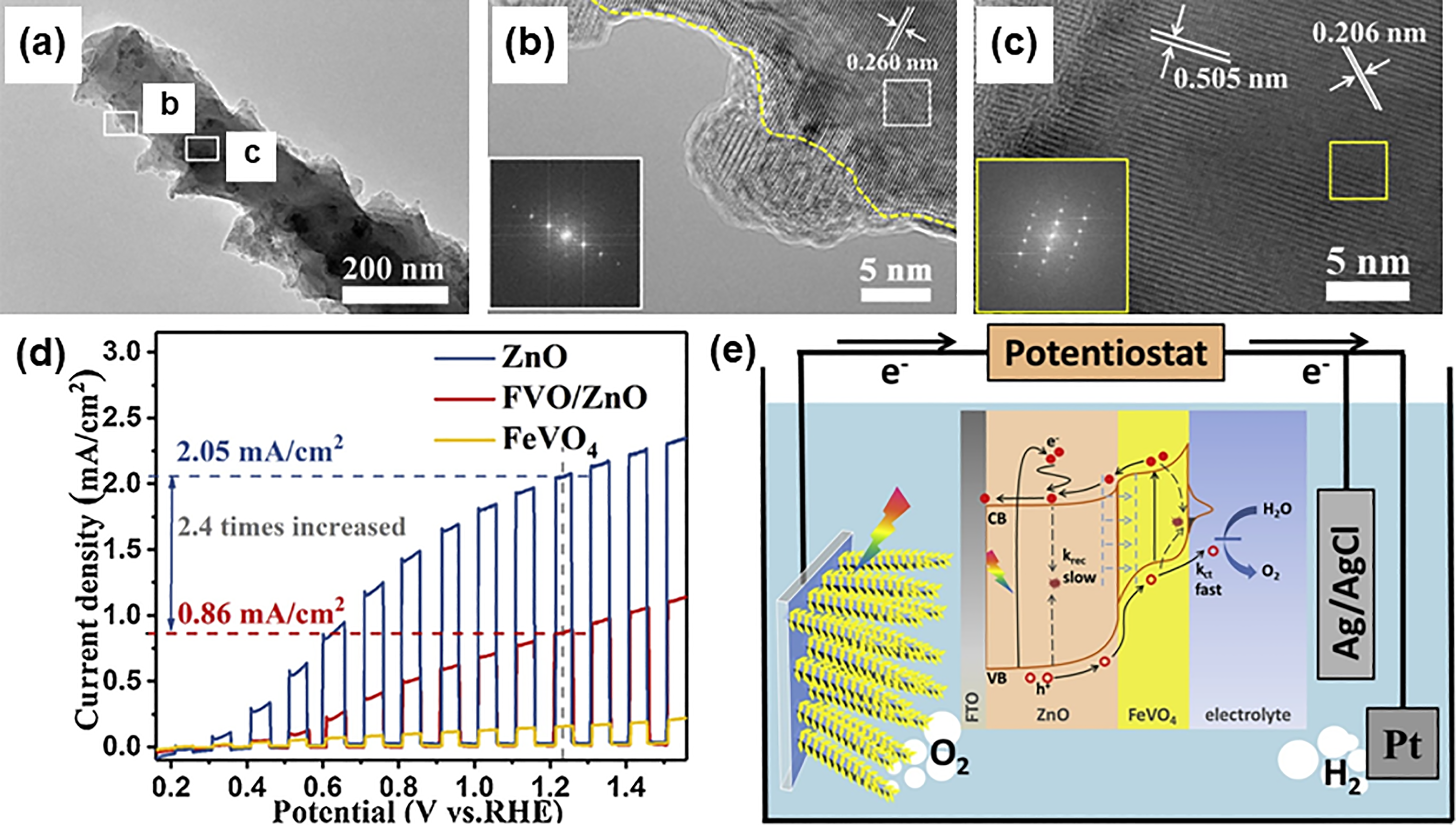
TEM image of (a) FeVO4/ZnO heterostructure. (b) and (c) HRTEM images of squared area in (a). (d) Photocurrent density against applied potential curves in sequence light on-off cycles. (e) Schematic representation of band gap alignment between ZnO and FeVO4 semiconductors for the PEC water splitting. Reprinted with permission from ref. 133
Interestingly, Wang et al. 130 fabricated a type II heterostructure consisting of four different types of semiconductors, namely Fe3O4, silicone dioxide (SiO2), ZnO, and ZnSe with a 3D morphology of multi-shell microsphere. The formation mechanism of quaternary heterostructure as indicated in Figure 6(a) clearly outlines that the Fe3O4 spherical core was initially coated with a SiO2 layer, followed by growing of ZnO nanorods, and eventually deposited with ZnSe nanoparticles. HRTEM image (Figure 6(b)) showing part of an irradiated rod structure from the microspheres confirms the decoration of ZnSe (interplanar spacing = 0.32 nm corresponding to the (111) crystal plane of cubic ZnSe) on ZnO nanorods (interplanar spacing = 0.26 nm corresponding to the (002) crystal plane of hexagonal wurtzite ZnO) forming a ZnO/ZnSe heterostructure. Based on the photocurrent analysis (Figure 6(c) and 6(d)), it reveals that Fe3O4/SiO2/ZnO/ZnSe heterostructure exhibited a better photocurrent response than Fe3O4/SiO2/ZnO sample without coating of ZnSe, under both UV and visible light irradiated conditions. The amount of ZnSe particles in the heterostructure could greatly influence the effectiveness of charge separation and migration. As shown in Figure 6(e), the plausible electron transfer pathway between the band gap edges of ZnO and ZnSe is depending on the electronegativity of semiconductors in such a way that recombination rate of photoinduced e−-h+ pairs could be decreased and more efficient charge separation.
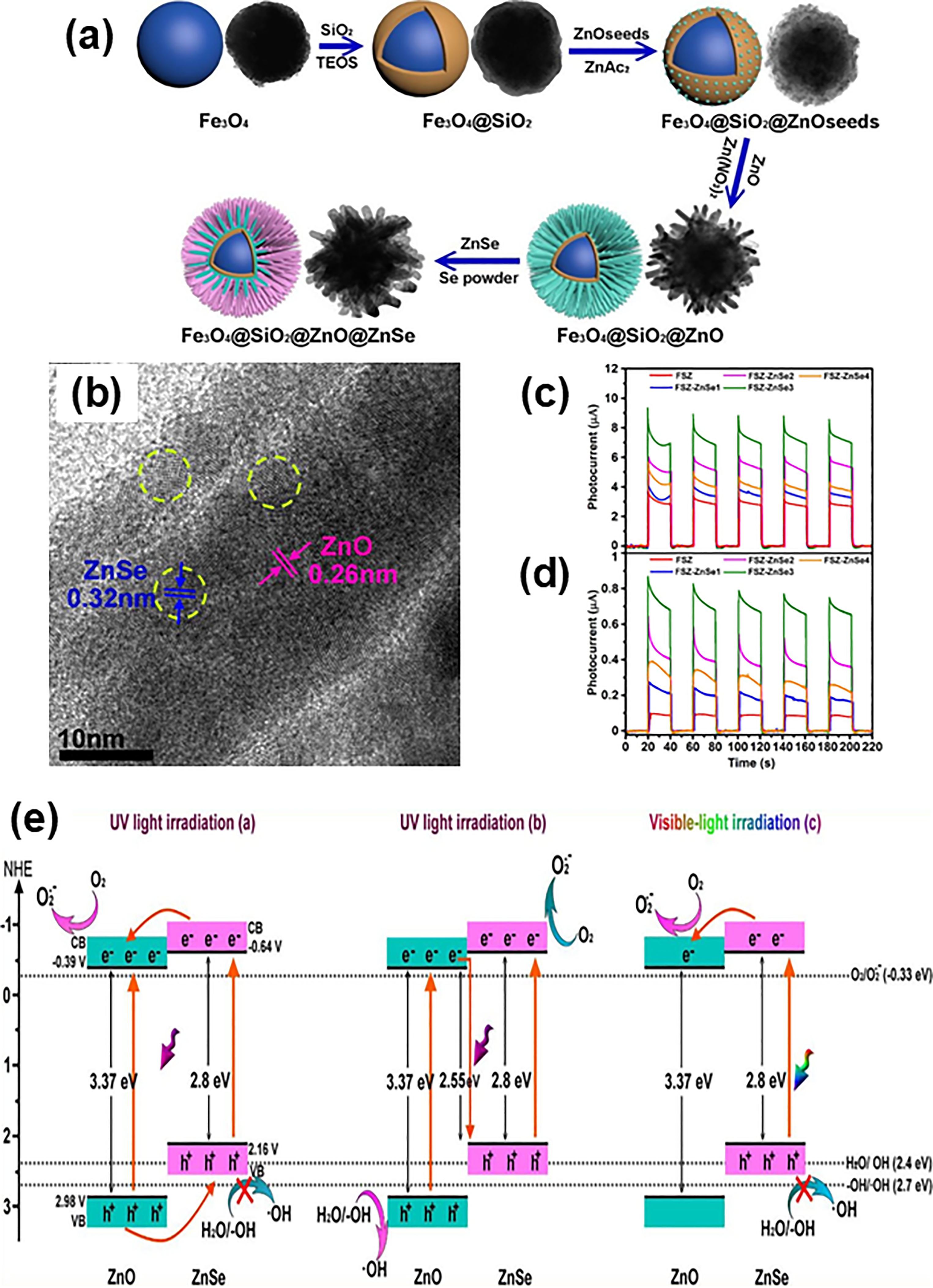
(a) Schematic diagram showing the formation pathway of Fe3O4/SiO2/ZnO/ZnSe heterostructure. (b) HRTEM image of Fe3O4/SiO2/ZnO/ZnSe heterostructure. (c) and (d) Photocurrent curves for different Fe3O4/SiO2/ZnO/ZnSe samples in sequence light on-off cycles under UV and visible light, respectively. (e) Schematic representation of possible electron transfer pathways at the interfaces of ZnO and ZnSe type II heterostructure under UV and visible light conditions. Reprinted with permission from ref. 130
Fabrication of ZnO-metal oxide semiconductor heterostructures, either derived into metal organic framework (MOF) or through MOF template route, has invigorated growing research attention.134–136 MOF has emerged as a new class of functional, highly crystalline inorganic-organic hybrid open-framework material with distinctive features of high surface area, tunable porous structure, and high nanoparticles loading capacity. Koo et al. 137 prepared heterostructure palladium (Pd)-ZnO/zinc cobaltite (ZnCo2O4) hollow spheres synthesized by using polystyrene and bimetallic metal-MOF templates. The hollow structures not only to offer high surface area and permeability, the very fine particles of Pd which were well-dispersed on ZnO/ZnCo2O4 hollow spheres could also increase the surface reaction and thus, enhancing the acetone gas sensing properties. Besides, Zhao et al. 136 transformed MOF-5 into ZnO/zinc sulphide (ZnS) heterostructure with an average size of 20 nm via alcoholysis and high temperature annealing. The ZnO/ZnS heterostructure increased the charge transfer and charge separation efficiency, therefore leading to remarkably enhanced hydrogen evolution reaction (HER) under visible light irradiation. In summary, the coupling of ZnO nanostructures with other semiconductors to form a heterostructure would enhance the photocatalytic properties of ZnO nanostructures by appropriate band gap position alignment. As such, the improved photocatalytic properties of heterostructure are attributed to the better light harvesting ability, suppression of photogenerated e−-h+ pairs recombination, effective charge carrier's separation, and low interfacial charge transfer resistance.
Photocatalytic applications of ZnO nanostructures for targeted environmental remediation
Degradation of micropollutants
Continuous release of recalcitrant micropollutants in water not only poses environmental threats, but also bring severe consequences to human and ecosystem developments.138–141 As a response, the development of efficient, economical, and sustainable removal methods for these emerging micropollutants is of vital. Heterogeneous photocatalysis, one of the advanced oxidation processes (AOPs), has been extensively explored for elimination of micropollutants and contaminants.142–144 Photocatalysis is based on the basic photophysical and photochemical principles for in-situ generation of reactive species such as •OH radicals triggered by adequate amount of photon energy, which then help to oxidise a broad range of polluting substances. Since the pioneering work reported by Kraeutler and Bard 23 on the photocatalytic decomposition of organic compounds, many important accomplishments presented throughout the years have uncovered the vast functionalities of photocatalysis as the most emerging destructive technology.51,145–147 Among the semiconducting photocatalysts, ZnO nanostructures have been widely studied in the photodegradation of aqueous phase pollutants. Table 2 summarises the recent advances in the photocatalytic degradation of micropollutants using ZnO-based nanostructures. Previously, Gouvêa et al. 148 reported the ZnO semiconductor-assisted photocatalytic degradation of different reactive dyes under UV irradiated system. As compared to TiO2, a higher adsorption capacity of ZnO encourages •OH attack on adsorbed molecules and thus, leads to a better photocatalytic efficiency. In order to improve catalyst recovery and recycle, Comparelli et al. 149 prepared immobilised organic-capped ZnO nanocrystals using non-hydrolytic and hydrolytic synthesis methods. They have examined the photocatalytic degradation rates of ZnO nanocrystals on methyl red and methyl orange (MO) in aqueous solution under UV irradiation, and collaboratively to study the pH effects towards catalysts and dyes on photocatalysis. Results indicated that surfactant-capped ZnO nanocrystals contain passivating molecules which are more resistant to photocorrosion and pH variation. Additionally, there are numerous studies highlighted the influences of ZnO particle size, specific surface area, and surface defects on the photocatalytic performance.150,151 The findings inferred that the photocatalytic degradation efficiency is improved with the increase of ZnO particle size benefited from low recombination rate of charge carrier between surface-bound hydroxyl radicals and photoexcited electrons captured by the surface oxygen deficiencies.
Representative summary of photocatalytic activity enhancement of ZnO-based nanostructures towards degradation of micropollutants.
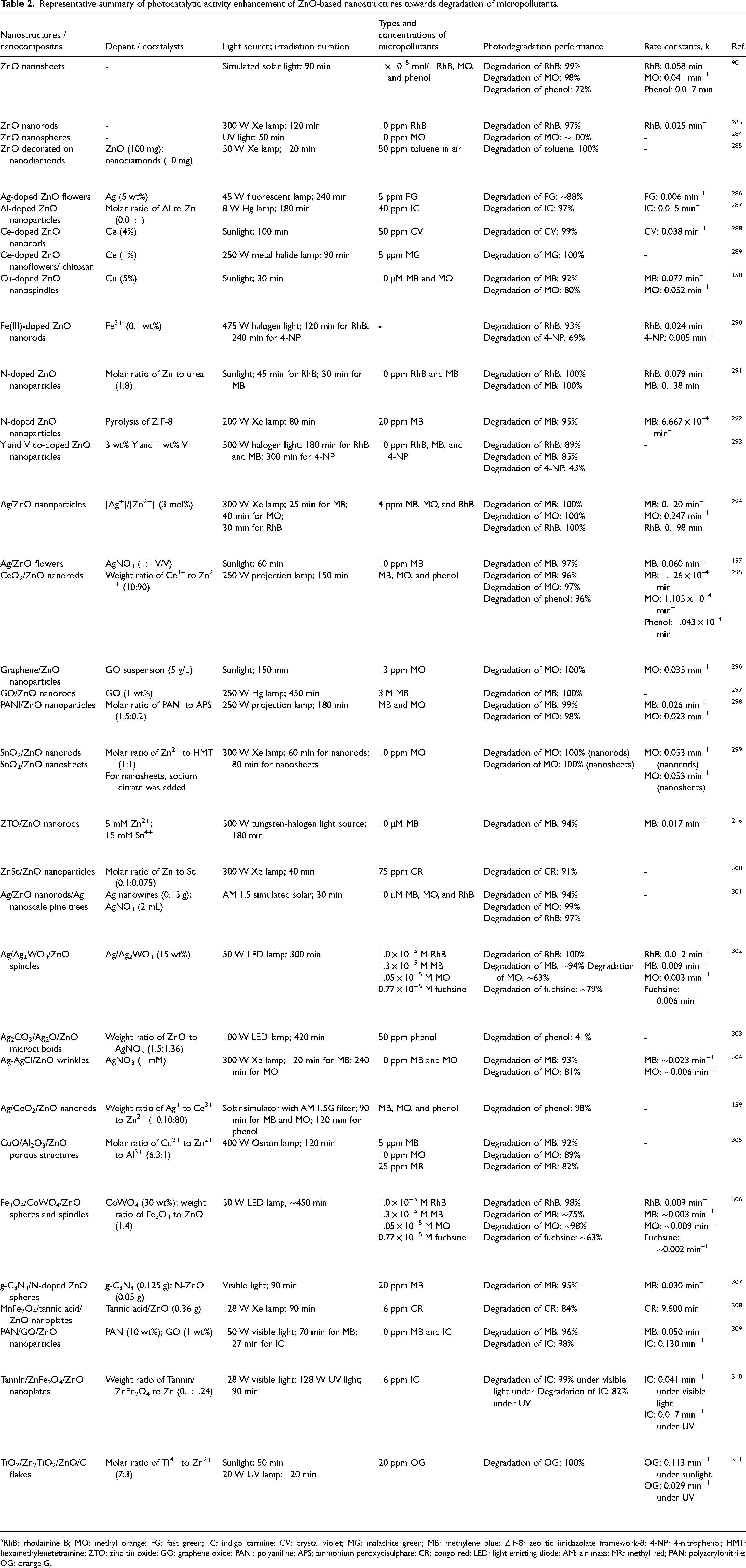
a RhB: rhodamine B; MO: methyl orange; FG: fast green; IC: indigo carmine; CV: crystal violet; MG: malachite green; MB: methylene blue; ZIF-8: zeolitic imidazolate framework-8; 4-NP: 4-nitrophenol; HMT: hexamethylenetetramine; ZTO: zinc tin oxide; GO: graphene oxide; PANI: polyaniline; APS: ammonium peroxydisulphate; CR: congo red; LED: light emitting diode; AM: air mass; MR: methyl red; PAN: polyacrylonitrile; OG: orange G.
In recent years, the advancement in the synthesis techniques of morphological-controlled ZnO nanostructures has opened an invigorating avenue for enhanced photocatalytic degradation of various emerging micropollutants. Xu et al.
152
have successfully developed 6 different morphologies of ZnO, namely cauliflower-like, truncated hexagonal conical, tubular rod-like, hourglass-like, nanorods, and spherical nanostructures via solvothermal method with different structure-directing solvents. The highest photocatalytic phenol degradation efficiency (k = 0.1496 min−1) demonstrated by cauliflower-like ZnO was deemed to be related to the effects of the crystal habits (i.e., growth of preferred structures and faceted planes) and oxygen vacancies. Besides, differences in synthesis methodologies of ZnO nanostructures could have significant influence on the semiconductor's photoresponsiveness. Saikia et al.
153
evaluated the photoactivities of ZnO nanoparticles synthesized by the colloidal precipitation method; another type of nanoparticles and flower-like shapes synthesized by hydrothermal method on the degradation of malachite green dye under solar light. Among all three synthesized samples, ZnO flowers exhibited the best photocatalytic degradation rate owing to the presence of a higher density of surface oxygen vacancies in the structures. The PL analysis showed that the stronger intensity of the green emission band at 550 nm for ZnO flowers was ascribed to the existence of singly ionised oxygen vacancies. At the surface vacancies, •O2– produced when adsorbed O2 scavenges an electron donated by photosensitised dye to the conduction band of ZnO could trigger the generation of •OH radicals (as shown in following Eqs. 1 to 5), which are essential for photodegradation.

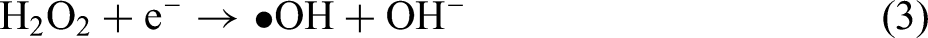


TEM images of ZnO (a) 0D nanoparticles, (b) 1D nanorods, (c) 2D nanosheets, and (d) 3D nanoflowers. (e) The photodegradation efficiency of salicylic acid using different ZnO nanostructures. (f) PL spectra and (g) Nyquist impedance plots of different ZnO nanostructures. Reprinted with permission from ref. 155
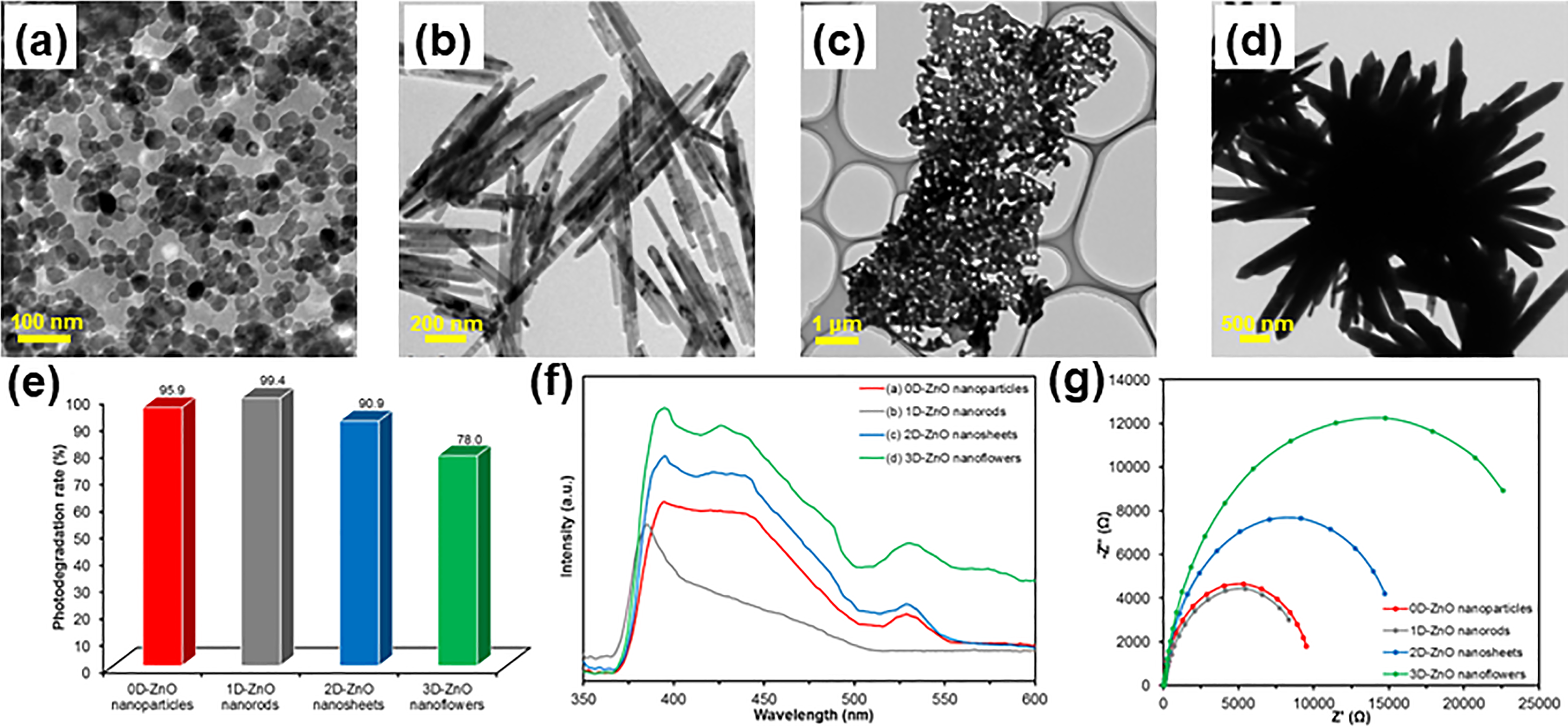
Due to the limitations of pristine ZnO nanostructures, include rapid recombination of photoinduced charge carriers and low stability during the photocatalytic reaction, hybrid nanostructures with dopants have been developed to enhance the photogenerated charge carriers separation and thus, improving the photocatalytic efficiency. Previously, Ahmad et al. 156 produced a 3D hierarchical flower-like ZnO structures functionalised with gold (Au) nanoparticles through integrated solution phase and electrochemical deposition synthesis methods. Au-ZnO hybrid nanostructure has been proved as a promising class of photocatalyst for decomposing the RhB under UV illumination. This mainly ascribed to strong electronic interaction between plasmonic Au nanoparticles and ZnO to increase the efficiency of charge separation of the photogenerated e−-h+ pairs on ZnO surfaces. Patil et al. 157 evaluated the photocatalytic performance of petal-shaped flower-like ZnO decorated with Ag nanoparticles on methylene blue (MB) degradation under natural sunlight. The enhanced photocatalytic activity of the Ag-ZnO nanostructures was attributed to the synergistic effects of a larger surface area for adsorption sites and surface plasmon resonance of Ag nanoparticles that causes intense photon scattering which is imperative for photoresponsive enhancement to direct light. Moreover, Kuriakose et al. 158 also showed the significance of improved separation of photogenerated charge carriers for an efficient sunlight-driven photocatalytic degradation of MB and MO dyes by Cu-doped ZnO nanostructures. They inferred the dependency of photoactivity on the amount of dopants as there is lattice strain present in the system. An optimum level of dopants could avoid the excessive formation of oxide layers on photocatalyst surfaces, which in turn lowering the light absorption. Instead of adding dopant, Saravanan et al. 159 synthesized a ternary nanocomposite containing Ag/cerium (IV) oxide (CeO2)/ZnO (Figure 8(a)) by a thermal decomposition method. Line dislocation linear defect as observed from HRTEM (Figure 8(b)) showed the presence of amorphous cerium (III) oxide (Ce2O3), which promotes oxygen vacancies due to the nucleation effect. This was further supported by X-ray photoelectron spectroscopy (XPS) analysis (Figure 8(c)) which confirmed the presence of both Ce4+ and Ce3+ found in the defects and amorphous phase of Ce2O3 in the nanostructure. Ce3+ and defects aided the formation of a surface state energy band of oxygen that facilitates the occurrence of oxygen adsorption, desorption, and diffusion processes and hence, greatly enhancing photocatalytic properties (Figure 8(d)). The UV-visible absorption results validated that MO (Figure 8(e)) and MB (Figure 8(f)) have been degraded completely within the irradiation time of 90 min. Lastly, they proposed the photocatalytic pathway mechanism for the ternary Ag/CeO2/ZnO nanostructure (Figure 8(g)) at which the nanostructure forms new Fermi energy level (resulted from interaction between semiconductor and metal) and contains a lot of free electrons due to the presence of Ag metals. Under visible light irradiation, free electrons are excited and transferred into the conduction band of CeO2 and ZnO. The photoexcited electrons at conduction bands and free electrons react with adsorbed oxygen molecules to form •O2–, followed by sequential reaction to generate •OH which is essential for degradation.
(a) SEM image, (b) HRTEM image, (c) hr-XPS spectrum of ce 3d, and (d) uv-visible spectra of ternary ag/CeO2/ZnO nanostructure. UV-visible absorption spectra of photocatalytic degradation of (e) MB and (f) MO using ternary Ag/CeO2/ZnO nanostructure. (g) Schematic diagram representing the electron flow and photocatalytic degradation mechanism of pollutants using ternary Ag/CeO2/ZnO nanostructure. Reprinted with permission from ref. 159
Removal of heavy metal ions
Many industrial activities increase the discharge rate of heavy metal ions, including Cr6+, As3+, Cd2+, Co2+, Ni2+, and Pb2+ to aqueous systems, which are highly toxic for biological ecosystems and human beings.160–163 Particularly, the development of nanotechnology provides a new option for the synthesis of catalysts which are low cost, high efficiency, and eco-friendly to remedy the aforementioned health and environmental issues. Among the conventional methods, adsorption process and redox reaction by ZnO-based nanostructures have come to the forefront as the advanced techniques to selectively and efficiently remove heavy metal ions from the water environment. 164 Physical adsorption of metal ions on the ZnO surfaces is usually reversible and does not require light illumination. Owing to the reversible nature of adsorption process, the adsorbents can be easily regenerated for repeated use through desorption processes.165,166 Noteworthy is that ZnO with plate-like and sheet-like nanostructures exhibited a higher adsorptive capacity over other morphologies attributed to its high specific surface area and specific affinity for heavy metal adsorption. 167
Meanwhile, the photocatalytic removal of metal ions is through the mechanisms of: (1) direct reduction of the ions by the photogenerated electrons; (2) indirect reduction by intermediates generated by hole oxidation; and (3) oxidative removal of metals. Banerjee et al. 168 synthesized different morphologies of ZnO and evaluated their photocatalytic reduction performances on Cr6+ under solar radiation. They suggested that variation in band gap energies of ZnO crystals could affect the rate of e−-h+ pair generation, followed by the production rate of hydrogen peroxide (H2O2) and methoxy radicals resulting from the photo-excited ZnO and hole scavenging, respectively on the reduction reactions of Cr6+ to Cr3+. Likewise, Chakrabarti et al. 169 reported the importance of the presence of sacrificial electron donors (i.e., methanol) on facilitating the photoreduction rate through consuming the hole and/or the •OH generated to reduce recombination. They found that about 90% reduction of Cr6+ was achieved over a reaction time of 75 min in the presence of methanol. Esmaielzadeh Kandjani et al. 170 improved the precision and selectivity of metal ions removal by fabricating surface enhanced Raman spectroscopy (SERS) active ZnO/Ag nanoarrays for combined functionalities of SERS detection and photocatalytic Hg2+ removal. Under UV irradiation, ZnO/Ag nanoarrays demonstrated an enhanced removal efficiency of Hg2+ ions to form elemental mercury (Hg0).
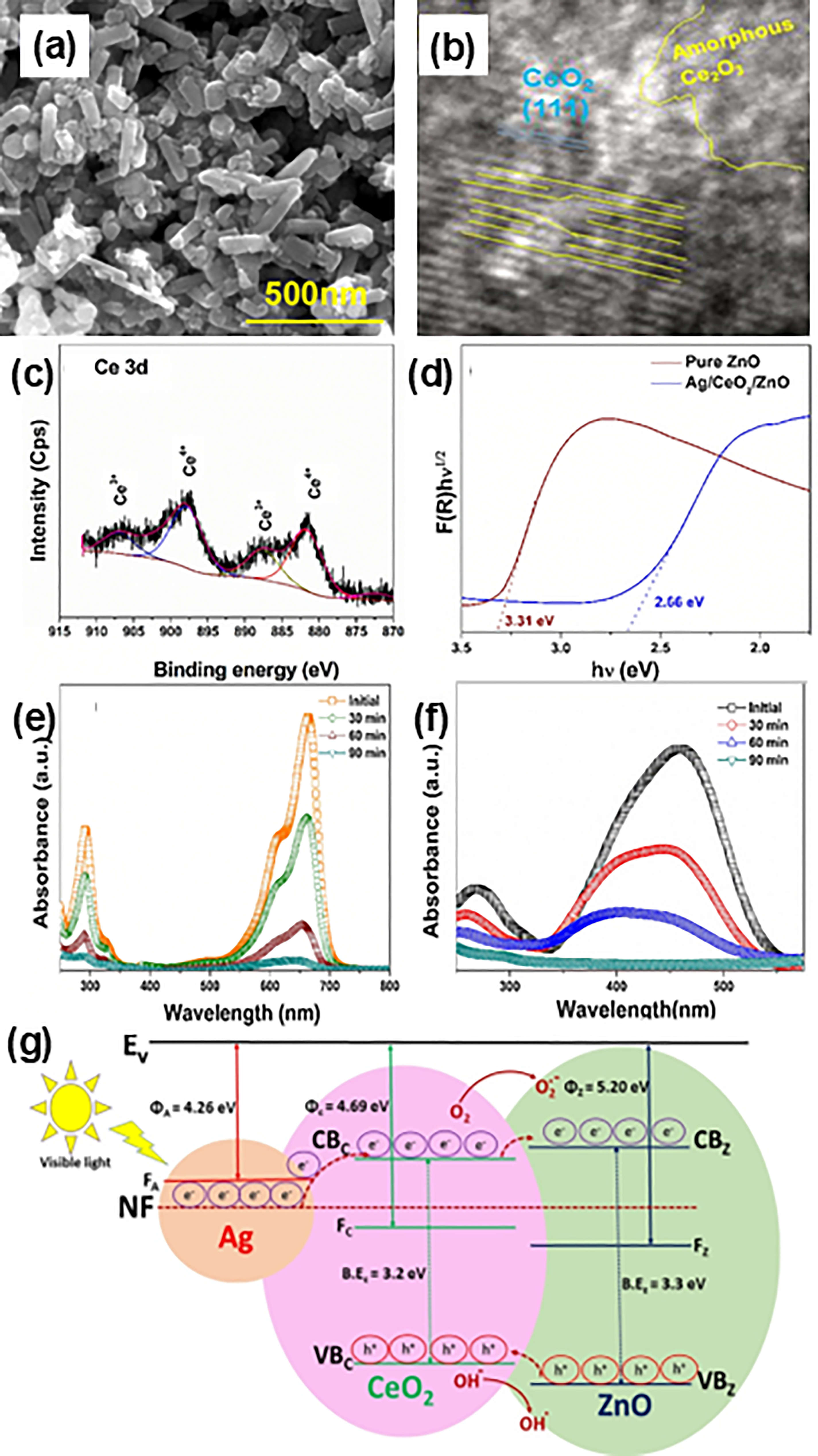
Recently, Shirzad-Siboni et al. 171 prepared ZnO nanorods immobilised on kaolin clay to study the photocatalytic reductions of Cr6+. The unique structure of ZnO/kaolin composite with improved adsorptive capacity exhibited an increment in the Cr6+ removal efficiency (from 88.3% to 98.5%) in the presence of citric acid, which could be credited to the scavenging effect on the positive holes produced during the photocatalytic process. Furthermore, Sahoo and Hota 172 prepared binary spherical structures of ZnO-zinc iron oxide (ZnFe2O4) onto amine functionalised graphene oxide (NH2-GO) surfaces as illustrated in Figure 9(a). A high surface area of NH2-GO/ZnO-ZnFe2O4 exhibited a better efficiency on Cr6+ removal where the equilibrium was at 0.05 g adsorbent dose (Figure 9(b)). Kinetic data fitting as shown in Figure 9(c) proved that the adsorptive removal of Cr6+ onto NH2-GO/ZnO-ZnFe2O4 nanocomposite surface follows a pseudo second order kinetics. The remarkable adsorption capacity of NH2-GO/ZnO-ZnFe2O4 nanocomposite is owing to the high availability of surface-active sites. In addition, the amine functional groups in the nanocomposite could form chelated complex with Cr6+ ions that enhance the electrostatic attractions.
(a) TEM image of NH2-GO/ZnO-ZnFe2O4 nanocomposite. (b) The removal efficiency of Cr(VI) at different dosages of adsorbent. (c) Pseudo second order kinetics for Cr(VI) removal by NH2-GO/ZnO-ZnFe2O4 nanocomposite. Reprinted with permission from ref. 172
Antibacterial properties of ZnO nanostructures
Gram-positive Bacteria
Gram-positive bacteria consist of three distinct cellular compartments, namely cytosol, single cytoplasmic membrane, and outer cell wall. The cell wall of Gram-positive bacteria is a peptidoglycan macromolecule with attached organic molecules, such as teichoic acids, teichuronic acids, polyphosphates, or carbohydrates. 173 Due to the emergence of antibiotic resistant bacterial infectious diseases, the antibacterial study of ZnO nanostructures on pathogenic microorganisms (i.e., Enterococcus, Staphylococcus, and Streptococcus species) has attracted ever-increasing attention.174–176 The bactericidal effect could be caused by the intrinsic toxic properties of nanomaterials, leading to cell damage and death. Over the past decade, Bacillus subtilis (B. subtilis) has been profiled as one of the best-characterised members of the Gram-positive bacteria. Several studies have indicated that ZnO nanostructures could exert biocidal action on B. subtilis and the microbial toxicity is more pronounced in the presence of light because of reactive oxygen species (ROS)-mediated effect through photo-assisted reaction.177,178 Among all, •OH is the most reactive oxygen radical that can reacts with the majority of types of molecules bonded on living cells. Other ROS like •O2– and H2O2 are weaker oxidisers with lower cell membrane permeability. However, the combination between •O2– and H2O2 can form highly toxic singlet oxygen (1O2) through the reaction of •O2– + H2O2 → OH– + •OH + 1O2. Sapkota et al. 179 evaluated the inactivation of model Gram-positive B. subtilis (TISTR 025) in MilliQ water, comparing between the dark environment and visible light photocatalysis mediated by ZnO nanorods. They concluded that the dissolution of Zn2+ ions could also be one of the mechanistic routes for bacterial inactivation under dark and light conditions. However, the enhanced antibacterial activity observed for illuminated reacting system is usually accompanied with the effect of photocatalytically mediated electron injection. The visible-light-inducement of photocatalytic microbial inactivation could primarily lead to the loss of bacterial cell membrane integrity and DNA degradation.
More recently, the antibacterial effect of nanocomposite films containing star-like ZnO has been studied in B. subtilis (Gram-positive) and Enterobacter aerogenes (Gram-negative), considering their advances in active food packaging application for extending the shelf life of food products. 178 Nanocomposites with a higher amount of ZnO possessed stronger bacterial inhibitory effect concomitant with a negligible migration rate of Zn ions from nanocomposites, which is thus considered safe for food packaging. Furthermore, ZnO nanostructures have been widely utilised in nanomedicine as antibacterial and anticancer agents. Premanathan et al. 180 investigated the antibacterial activity of ZnO nanoparticles against Gram-positive Staphylococcus aureus (S. aureus) and selective toxicity to cancerous HL60 cells. Their findings implied that the primary mechanism of ZnO nanoparticles cytotoxicity might initially involve the generation of ROS, which then were responsible for the induction of apoptosis through lipid peroxidation. Ramani et al. 181 examined the shape-dependent antibacterial activity against several model pathogens using ZnO nanostructures of various morphologies. They found that rod-like ZnO with a smaller aspect ratio and higher specific surface area could induce better antibacterial activity against Gram-positive S. aureus and other Gram-negative bacteria due to the ROS damage to cell membranes. Since then, more ZnO nanostructures (Figure 10(a) – 10(f)) have been synthesized and tested for the antibacterial properties against model Gram-positive bacteria (i.e., Streptococcus iniae (S. iniae) and Streptococcus parauberis (S. parauberis)) and Gram-negative bacteria. 182 The value of the zone of inhibition (ZOI) against the bacterial strains represented graphically in Figure 10(g) confirmed that ZnO flowers showed significant antibacterial activity with the highest ZOI against all studied pathogenic bacteria. They also deduced that Zn2+ ions dissolution and ROS generation were the major antibacterial mechanisms.
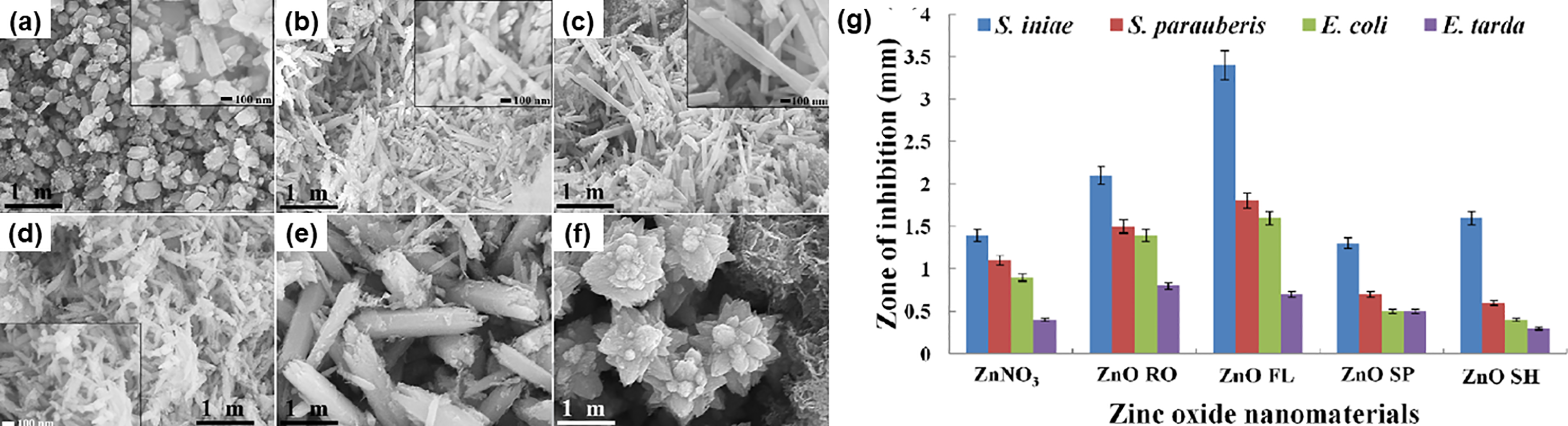
(a) – (f) SEM images of ZnO nanostructures with various surface morphologies synthesized via soft jet plasma-assisted process by varying the concentrations of ZnNO3 and NaOH. (g) The inhibition zone against different model by using different ZnO nanostructures. ( a RO: nanorods; FL: flowers; SP: spherical shape; SH: nanosheets). Reprinted with permission from ref. 182
Decoration of noble metallic elements and compounds with lower Fermi energy levels on ZnO nanostructures could enhance optical absorption capability and photoelectronic current of ZnO, and hence, promoting the production of ROS which is propitious for improved antibacterial action. Hybridisation of Ag nanoparticles with ZnO nanorod arrays grown on polymer substrates exhibited high photon harvest and significant inhibition of the growth of S. aureus. 183 Besides, Mao et al. 184 fabricated carboxymethyl cellulose (CMC) hydrogel composite incorporated with Ag/Ag@AgCl/ZnO hybrid nanostructures to cure serious infection caused by S. aureus and to accelerate wound treatment under visible light irradiation. The hydrogel system which shows controllable release of Ag+ and Zn2+ ions originating from the reversible swelling-shrinking transition due to the changes in pH has great potential in tissue repair and antibacterial application.
Gram-negative Bacteria
Unlike Gram-positive bacteria, the Gram-negative bacterial cell envelope contains an additional outer membrane composed of phospholipids and lipopolysaccharides (LPS) that faces outwards to the environment. 185 LPS increase the negative charge of the Gram-negative cell membrane and cell wall to stabilise the overall membrane structure. The cationic properties of metals and metal oxides tend to attract negatively-charged bacteria, imposing stronger bactericidal activities towards Gram-negative bacteria. To date, antimicrobial activities of nanomaterials have been extensively analysed on human pathogenic bacteria, especially Escherichia coli (E. coli) with proven significant bacterial surviving rate reduction by ZnO nanostructures.186–188 Applerot et al. 189 investigated the influence of the size of crystalline ZnO nanoparticles (ranging from the microscale to nanoscale) on the antibacterial effect towards E. coli. They validated that exposure of bacteria to the smaller size of ZnO nanoparticles results in an increased cellular internalisation of the nanoparticles and enhanced antibacterial rate. The study of electron spin resonance (ESR) is imperative to determine the nature of ROS being generated in aqueous suspensions of ZnO. ESR measurement indicated that smaller particles size produced a larger amount of •OH attributed to a higher surface-to-volume ratio of the nanoparticles reacting with water. Additionally, smaller nanoparticles would have a higher defect concentration that act as the electron trapping sites to promote ROS generation.
Brayner et al. 190 reported the effects of particle size and shape of ZnO nanostructures controlled by addition of different molecular surfactants, on the biocidal action towards E. coli strain (MG1655). It was demonstrated that ZnO nanoparticles synthesized with sodium dodecyl sulfate (SDS) inhibited the highest bacterial growth (15%) due to denaturalisation of bacterial proteins by SDS. It is discernible that a Gram-negative triple membrane disorganisation occurred after contact with diethylene glycol (DEG) and ZnO nanostructures could increase the membrane permeability, followed by encouraging the accumulation of ZnO in the bacterial membrane and cellular internalisation of these nanoparticles. A very high photoresponse rod-like ZnO nanostructure has also been tested for its potent antibacterial ability. Interestingly, Jain et al. 191 assessed the physiological effects (i.e., aspect ratio) of the ZnO nanorods on Gram-positive bacteria and Gram-negative bacteria (i.e., E. coli and Aerobacter aerogenes (A. aerogenes)). Serious damage in the morphologies of bacterial cells and a reduction in live cell count after exposure to ZnO nanorods could be caused by ZnO ionisation. Subsequently, this could increase the production rate of ROS which may further increase the pro-oxidant intracellular components and damage DNA. Lately, the direct growth of 1D-ZnO nanostructures onto textile surfaces has also been evidenced as an excellent antibacterial agent against E. coli (CCUG 3274). ZnO-coated textile has successfully reduced about 90% of the bacterial population during 3 h of incubation. 192
The distribution of ZnO nanostructures over the surface could be improved by precipitation of ZnO onto polysaccharides biopolymer owing to the strong interfacial interaction between organic and inorganic materials.193,194 The enhanced antibacterial effect observed on the Gram-negative bacteria (i.e., Pseudomonas aeruginosa (P. aeruginosa) and Klebsiella pneumoniae (K. pneumoniae)) could be accredited to the presence of oxygen vacancies in the interstitial position and the generation of H2O2 molecules which can permeate into the cell and inhibit the growth of bacteria. In a similar fashion, ZnO nanostructures decorated onto inert substrates have been regarded as one of the favourable approaches to avoid the agglomeration of nanoparticles. For instance, Ruíz-Gómez et al. 195 controlled the morphologies of ZnO, namely macropores, plate-like, and rod-like structures, on the glass substrate through electroless deposition, which acted as transparent antimicrobial coatings against Gram-negative E. coli. The ZnO films were found to exhibit high transparency in the range of the visible region, photoconductivity, and superhydrophobicity. Among various morphologies synthesized, rod-like ZnO deposited on glass film demonstrated the most significant antimicrobial performance due to preferential dimension for the contact between ZnO and bacterial surfaces, and also unique structural orientation with a higher proportion of (002) polar planes which promoted the ROS generation. Also, Fan et al. 196 evaluated bactericidal effect of E. coli by a material of MOF-derived, ZnO-doped carbon on graphene (ZnO@G) anchored on phase transformable thermal responsive brushes (TRB), yielding TRB-ZnO@G (Figure 11(a) and 11(b)). Upon near-infrared (NIR) irradiation, TRB-ZnO@G exerted strong bacterial killing efficiency (nearly 100%) on both E. coli and Gram-positive bacteria (i.e., S. aureus), as depicted in Figure 11(c). The antibacterial mechanism in Figure 11(d) illustrates the difference in hydrophilicity and hydrophobicity of TRB-ZnO@G during on and off NIR conditions. When NIR is on, hydrophobic TRB would trigger phase-to-size transformation of TRB-ZnO@G and cause the wrapping of surrounding bacteria into micrometer aggregations and therefore, leading to a localised effective bactericidal effect.
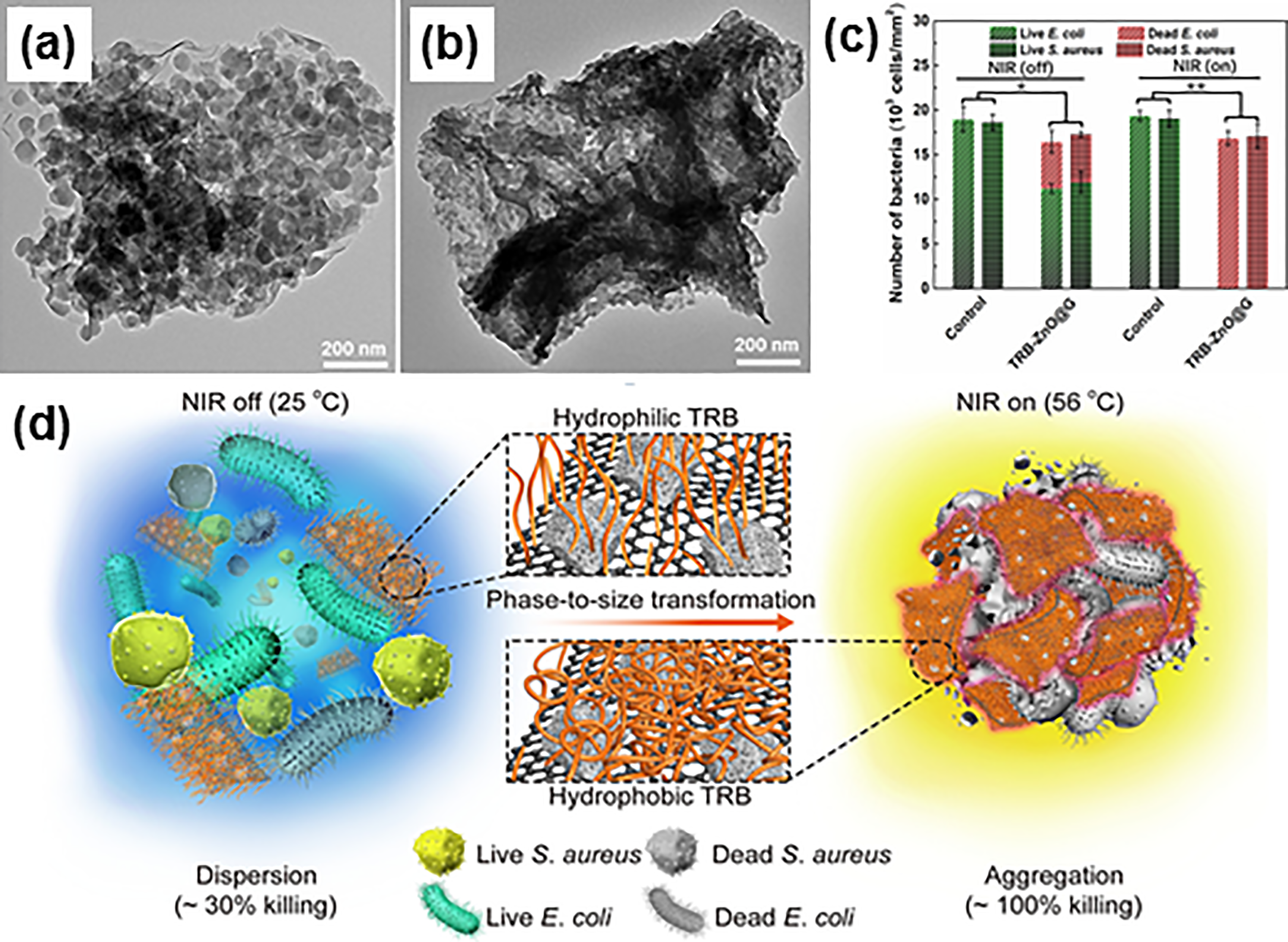
TEM images of (a) ZIF-8@GO and (b) TRB-ZnO@G. (c) Live/dead bacterial numbers with and without NIR irradiation. P-values correspond to both live and dead bacterial numbers after interaction with TRB-ZnO@G when compared to the control, ∗P < 0.05, ∗∗P < 0.005. (d) Schematic representation of the hydrophilicity and hyrdphobicity of TRB-ZnO@G as well as the phase-to-size transformation of TRB-ZnO@G causing the wrapping of bacteria into aggregations due to the hydrophobic state of TRB-ZnO@G upon NIR irradiation. Reprinted with permission from ref. 196
Advances in development of cationic and anionic doped ZnO nanostructures have gained increasing focus on their antibacterial properties and toxicology studies towards the environment. Advantages of enhanced surface area, longer absorption wavelength in a wider light region, and reduced band gap energy after adding metal and transition metal dopants improve the antibacterial performances of pure ZnO.197–199 As a matter of course, a higher bactericidal activity of chromium (Cr)-doped ZnO nanorods against Gram-negative E. coli was confirmed as there were interactions of Cr nanoparticles with S and P containing compounds to decrease cell respiration and reduction of photogenerated e–-h+ pairs recombination to produce more ROS. 200 Table 3 outlines the findings on various morphologies of ZnO and ZnO-based nanostructures for their antibacterial actions towards both Gram-positive and Gram-negative bacteria, associated with the mechanisms underlying their antimicrobial effects.
Recent reports on the antibacterial activities of ZnO-based nanostructures targeted on various model bacterial and their possible antibacterial mechanisms.
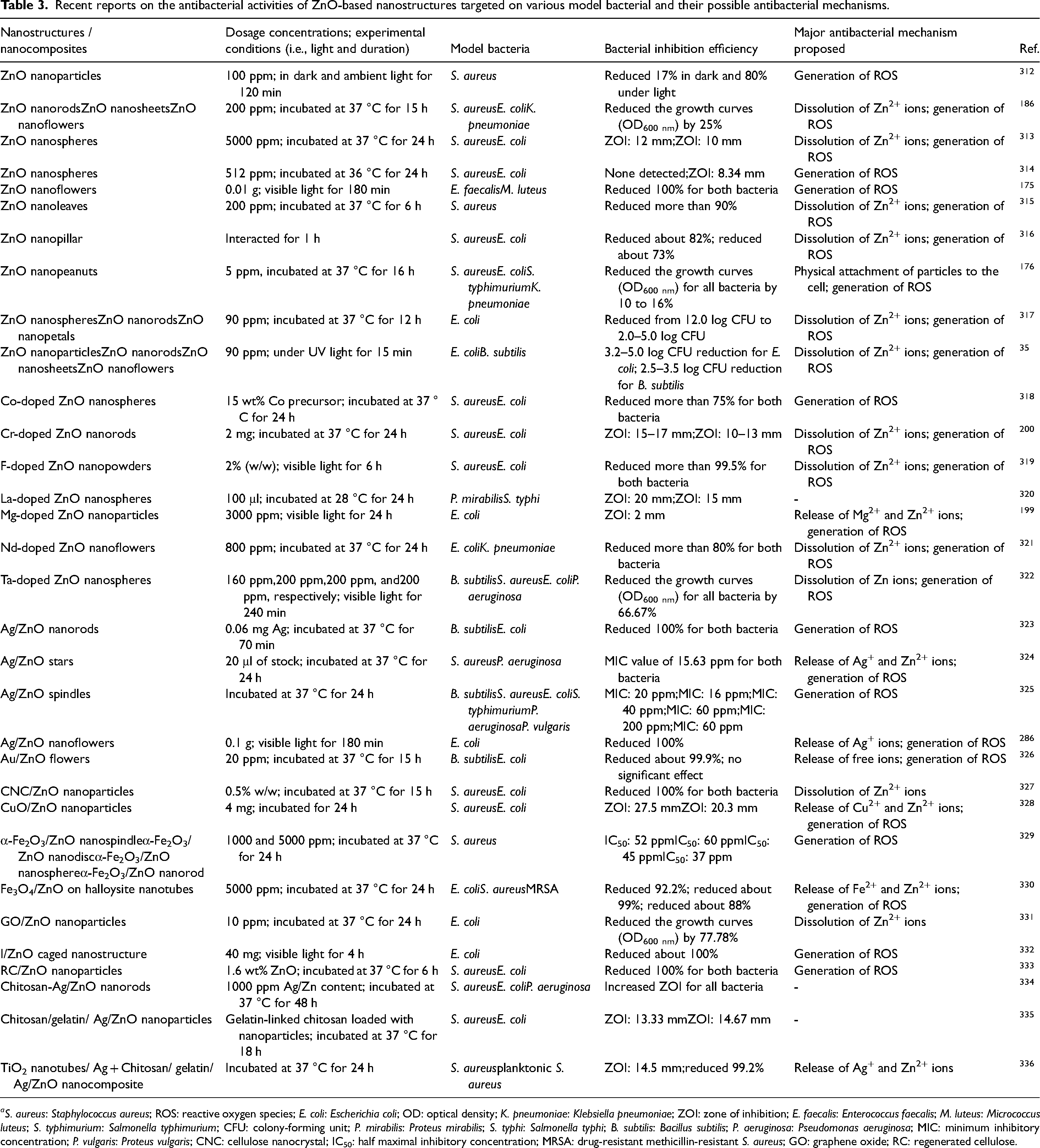
aS. aureus: Staphylococcus aureus; ROS: reactive oxygen species; E. coli: Escherichia coli; OD: optical density; K. pneumoniae: Klebsiella pneumoniae; ZOI: zone of inhibition; E. faecalis: Enterococcus faecalis; M. luteus: Micrococcus luteus; S. typhimurium: Salmonella typhimurium; CFU: colony-forming unit; P. mirabilis: Proteus mirabilis; S. typhi: Salmonella typhi; B. subtilis: Bacillus subtilis; P. aeruginosa: Pseudomonas aeruginosa; MIC: minimum inhibitory concentration; P. vulgaris: Proteus vulgaris; CNC: cellulose nanocrystal; IC50: half maximal inhibitory concentration; MRSA: drug-resistant methicillin-resistant S. aureus; GO: graphene oxide; RC: regenerated cellulose.
Impacts of ZnO nanostructures within wastewater treatment system: opportunities or threats
Treatment efficiency enhancement by photocatalytic properties
As discussed in Section 3, nanostructured ZnO photocatalysts have been extensively employed in the photocatalysis and photoelectrochemical applications for targeted environmental remediation of emerging micropollutants in source waters. ZnO is an effective catalyst for the initiation of photo-assisted reaction in order to decompose and degrade a variety of water and wastewater effluents without producing secondary and/or recalcitrant wastes in the environment.201–204 Prior to the photocatalytic process, pretreatment of the raw wastewater sources is utmost crucial step to reduce the high turbidity level so that the UV scattering and shielding stemmed from the absorption by wastewater particles can be minimized.205,206 Notably, photoelectrochemical water splitting is considered as one of the prospective solutions for energy- and environment-related issues, aiming to generate hydrogen as a clean fuel and also a form of energy source. Many efforts have been devoted to improve the efficiency of ZnO photocatalysts in splitting the water photoelectrochemically through nanostructure modifications.207–211 The light harvesting and absorption capabilities of ZnO nanostructures could be enhanced through functionalisation of ZnO that can shorten lateral charge carrier diffusion distance and increase electron mobility.208,211
In terms of environmental concerns on the treatment of wastewater effluents, reactive dyes are the most abundant and recalcitrant components found in dyeing and finishing processes of textile industry which are difficult to be removed by conventional treatment technologies.212,213 The heterogeneous photocatalysis process using ZnO nanostructures is effective to break the azo bonds (─N═N─) and aromatic rings, as well as decreasing both biochemical oxygen demand (BOD) and chemical oxygen demand (COD) values caused by the presence of organic and inorganic contaminants. Sapkal et al. 214 reported ZnO nanocrystals immobilised on the fluorine doped tin oxide (FTO) thin film for the photoelectrocatalytic treatment of textile industrial effluent under UV-A illumination. They found that ZnO nanocrystals exhibited a decolourisation rate of 93% within 180 min (calculated using American Dye Manufacturing Institute (ADMI) removal ratio) and a COD reduction of 69%. Lam et al. 215 synthesized different flower-like ZnO structures and evaluated their photocatalytic degradation on real textile wastewater. After 240 min of light-irradiated reaction, biodegradability of the treated textile wastewater was improved with high removal efficiencies for BOD5 (76%) and COD (75%). To further improve the practicability of the using AOP, development of visible-light-active ZnO-based photocatalysts is in spiraling demand to reduce the plant operation cost by maximising the usage of natural solar energy.216–218 The performance enhancement could be influenced by the alteration of physical properties (i.e., size- and surface area-related matters with the variation of ZnO and dopants compositions) and changes in optical and electrochemical properties (i.e., hindered photogenerated e−-h+ pairs recombination rate and faster charge mobility).
Not only dye contamination, environmental activists are also urging for unremitting efforts to treat the industrial effluents photocatalytically as toxic pollutants contained in wastewater streams are prone to adversely affect the treatment system, particularly the biological wastewater treatment process. Kanjwal et al. 219 evaluated the photocatalytic degradations of electrospun ZnO nanofibers and composite ZnO-nickel oxide (NiO) nanofibers on dairy effluent. Composite ZnO-NiO nanofibers increased the photocatalytic efficiency by 1.07-fold, as compared to the pristine ZnO nanofibers after 180 min of visible light irradiation. Furthermore, Wong et al. 220 examined the photocatalytic degradation of pre-treated palm oil mill effluent (POME) by various morphologies ZnO nanostructures. As high as 96% COD removal of POME by pompon-like ZnO structures was taking the merit of high surface-active sites to promote light absorption and rapid charge transfer with a better photogenerated e−-h+ pairs separation. Preparation of a porous membrane base for the attachment of ZnO-based photocatalysts is auspicious for the adsorption of pollutants on to a large active surface area. ZnO nanoparticles were decorated on granular porous natural scoria and used for the treatment of pharmaceutical and textile pollutants from wastewaters. 221 The nanocomposite achieved COD reduction rates of 41.55% and 59.25% for pharmaceutical and textile wastewaters, respectively with excellent reusability. In another study, Choi et al. 222 examined the photocatalytic water treatment efficiency of 3D Pd/ZnO nanowires populated on electrospun nanofibers. The nanowire arrays could significantly increase the surface area for pollutant adsorption, surface wettability, and UV absorption. Moreover, increasing Pd doping was found to suppress the recombination rate of photogenerated e−-h+ pairs. The Pd/ZnO nanowires nanofiber successfully removed the organic matter in water from 0.0249 (solely ZnO nanowires nanofiber) to 0.0377 mg CODCr/mg ZnO nanowires-hr. More recently, Lam et al. 223 assessed the photoelectric performance of a Z-scheme structure photoanode comprising tungsten trioxide (WO3)-loaded on hexagonal rod-like ZnO/Zn (WO3/ZnO/Zn) composite (Figure 12(a)) towards the mineralisation of sunset yellow (SSY) dye and phenol as well as actual food wastewater. As illustrated in Figure 12(b) and 12(c), a higher value in open-circuit voltage (VOC), short-circuit current (JSC), and maximum power density (Pmax) of WO3/ZnO/Zn represent that the composite exhibited more electrical energies to degrade SSY dye and phenol as compared with pristine ZnO/Zn photoanode. Following that, PL and transient photocurrent response (Figure 12(d)) analyses proved that the incorporation of WO3 in ZnO/Zn structure could significantly enhance the charge carrier separation and hinder the recombination rates. In Figure 12(e) shows the Z-scheme heterojunction mechanism delineating the migration of the photogenerated charge carrier based on the band edge position alignment between WO3 and ZnO/Zn for degrading the SSY dye and phenol. Also, WO3/ZnO/Zn achieved the removal efficiencies for COD, BOD5, ammoniacal nitrogen (NH3-N) of 56.8%, 40.8% and 42.9%, respectively for the food production wastewater.
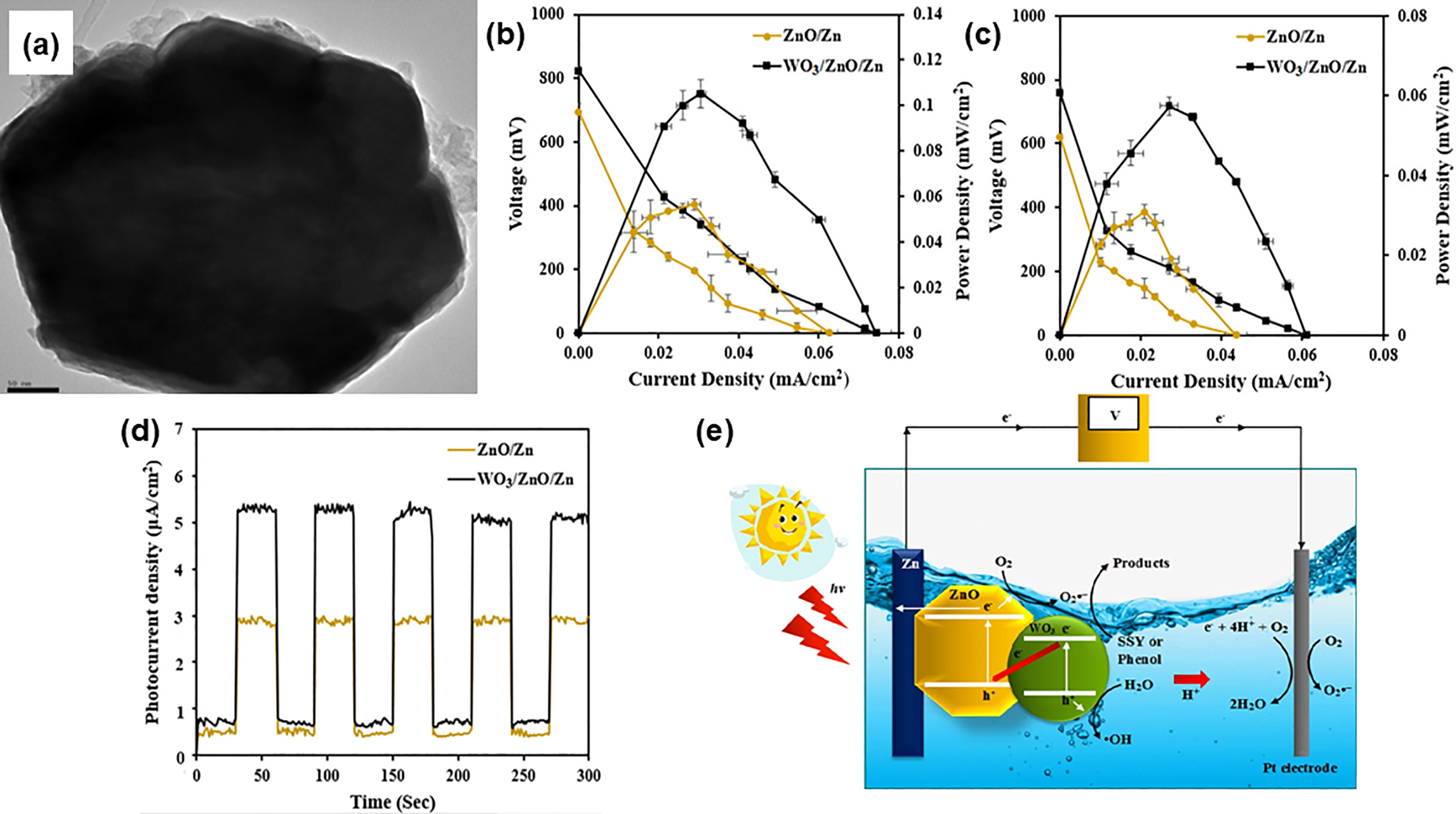
(a) TEM image of WO3/ZnO/Zn composite. I-V curves showing the degradation of (b) SSY dye and (c) phenol using pristine ZnO/Zn and WO3/ZnO/Zn under sunlight. (d) Transient photocurrent responses of pristine ZnO/Zn and WO3/ZnO/Zn. (e) Schematic diagram showing the photocatalytic degradation mechanism of SSY and phenol using WO3/ZnO/Zn upon sunlight irradiation. Reprinted with permission from ref. 223
Recent published studies on the photocatalytic degradations of different real wastewater effluents utilising ZnO-based nanostructures are tabulated in Table 4. In short, the photocatalytic treatment of real wastewater effluents using ZnO-based nanostructures holds great promise for degrading and decomposing the actively toxic contaminants that pose potential risks to the conventional wastewater treatment technologies. However, the application of ZnO-based photocatalysts in the AOPs for treatment purpose has to be monitored carefully as their pronounced antibacterial properties could equally bring a negative effect to the biological wastewater treatment system and be hazardous to the microbial-containing sludge.
Representative summary for photocatalytic treatment of different real wastewater effluents using ZnO-based nanostructures.
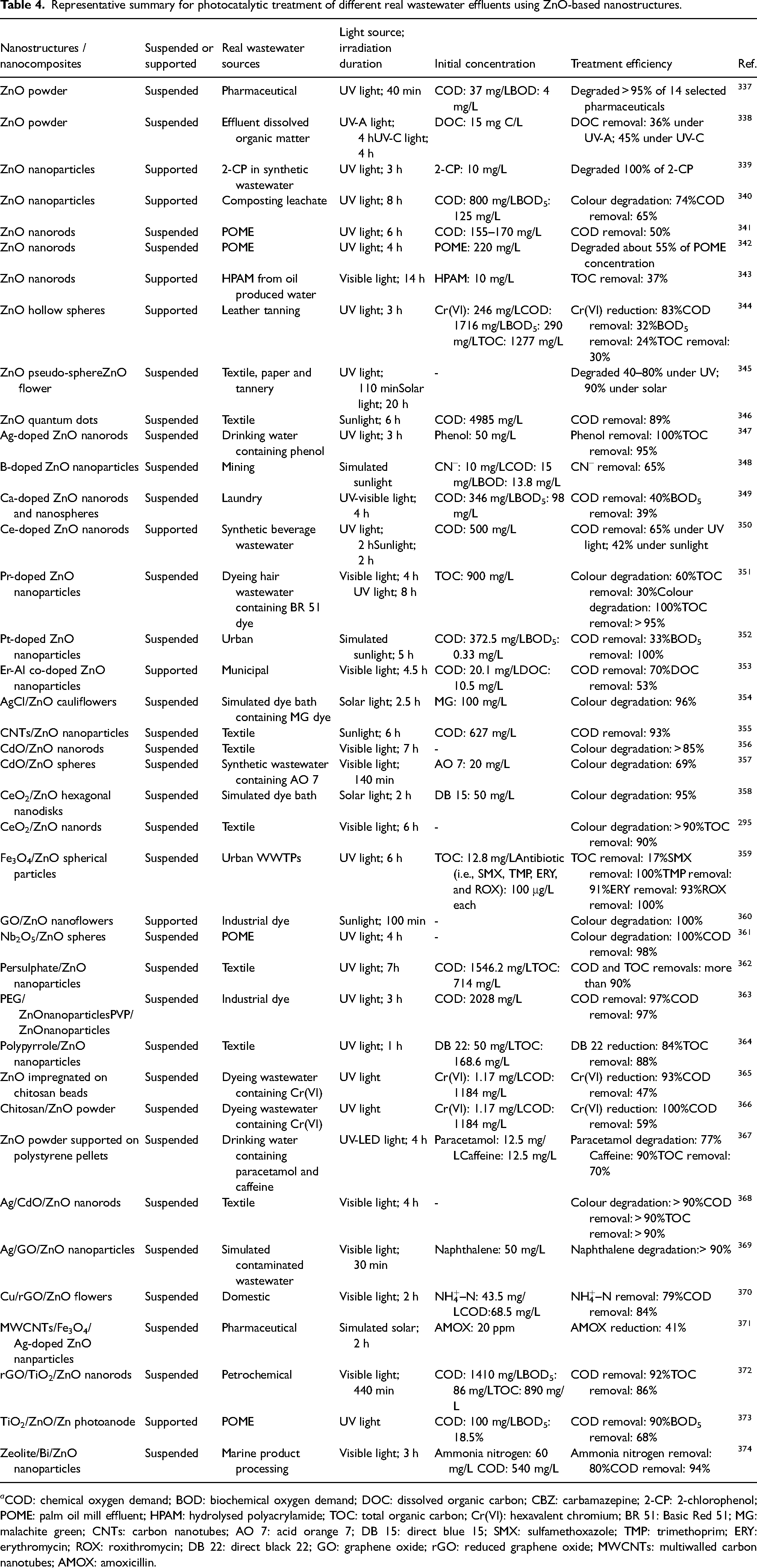
a COD: chemical oxygen demand; BOD: biochemical oxygen demand; DOC: dissolved organic carbon; CBZ: carbamazepine; 2-CP: 2-chlorophenol; POME: palm oil mill effluent; HPAM: hydrolysed polyacrylamide; TOC: total organic carbon; Cr(VI): hexavalent chromium; BR 51: Basic Red 51; MG: malachite green; CNTs: carbon nanotubes; AO 7: acid orange 7; DB 15: direct blue 15; SMX: sulfamethoxazole; TMP: trimethoprim; ERY: erythromycin; ROX: roxithromycin; DB 22: direct black 22; GO: graphene oxide; rGO: reduced graphene oxide; MWCNTs: multiwalled carbon nanotubes; AMOX: amoxicillin.
Treatment efficiency suppression by antibacterial effects
Secondary treatment is an established method to substantially decompose the organic and inorganic chemicals found in wastewater effluents by the actions of bacteria and various microorganisms. Nevertheless, the use of ZnO-based nanostructures for controlled disinfection of pathogens without harming the microbial-containing sludge in the biological wastewater treatment processes is always an arduous task to achieve. Many studies have revealed the potent inhibitory impacts and toxicity of ZnO towards biological systems, which may also greatly limit their applications for water/wastewater disinfection.224–227 Hence, it is important to monitor the raw influents and to isolate the photocatalytic treatment process with conventional wastewater treatment plants in order to prevent the entry of ZnO-based nanostructures into sewer system and leading to the deterioration of treatment process and its efficiency.
Hou et al. 228 was the first to report temporal and spatial inhibitory effects of ZnO nanoparticles on the O2 respiration activities of aerobic wastewater biofilms by using microelectrodes. At a higher ZnO dosage rate (50 mg/L), ZnO nanoparticles inhibited microbial activities only in the outer layer of the biofilms because the greatest rate of O2 respiration activity at the deeper parts (beneath 200 μm) of the biofilms. He et al. 229 examined the effects of ZnO nanoparticles loadings on the microbial community of aerobic granular sludge (AGS) based on the nitrogen and phosphorus removals, microbial activities, and extracellular polymeric substances (EPS) production. The shock loading of ZnO nanoparticles affected the transformations of nitrates and nitrites as there was a drop in ammonia removal rates (77.79%, control: 98.85%) and total inorganic nitrogen removal rates (57.43%, control: 96.65%). Intriguingly, both studies confirmed that dissolution of Zn2+ ions is the main contributor for the microbial inhibitory effects. From the phylogenetic classification of functional microbial community of the aerobic granules in sequencing batch reactor (SBR) after exposure to ZnO nanoparticles, the biological nitrogen and phosphorus removal rates that involved the activities of glycogen accumulating organisms (GAOs) ammonia oxidising bacteria (AOB), nitrite oxidising bacteria (NOB), denitrifying bacteria (DNB), phosphorus accumulating organisms (PAOs), and denitrifying PAOs (DNPAOs) were affected (either accumulating or decreasing). 230 In a related study, Wang et al. 231 reported the toxicity of different ZnO nanoparticles concentrations on the nitrification processes in activated sludge. A higher dosage of ZnO nanoparticles caused a lower oxidation rate of NH4+–N into nitrite but appeared to immediately oxidise to nitrate, suggesting that the impacts of ZnO nanoparticles on the AOB are more significant than NOB. Chen et al. 232 investigated the response of ZnO nanoparticles towards antibiotic resistance genes (ARGs) and microbial communities in estuarine water. Figure 13 presents the influences of different concentrations of ZnO nanoparticles on both the absolute and relative abundances of total ARGs (total of sul1, sul2, tetA, tetW, ermB, and qnrS) detected in estuarine water. At relatively low ZnO dosage (i.e., 0.2 and 1 mg/L), the abundances of total ARGs in water samples were significantly higher than the blank samples. Hence, ZnO nanoparticles exposure increased the ARGs abundances and also accelerated the dissemination of ARGs and thus, posing deleterious effect on public health and ecological environment.
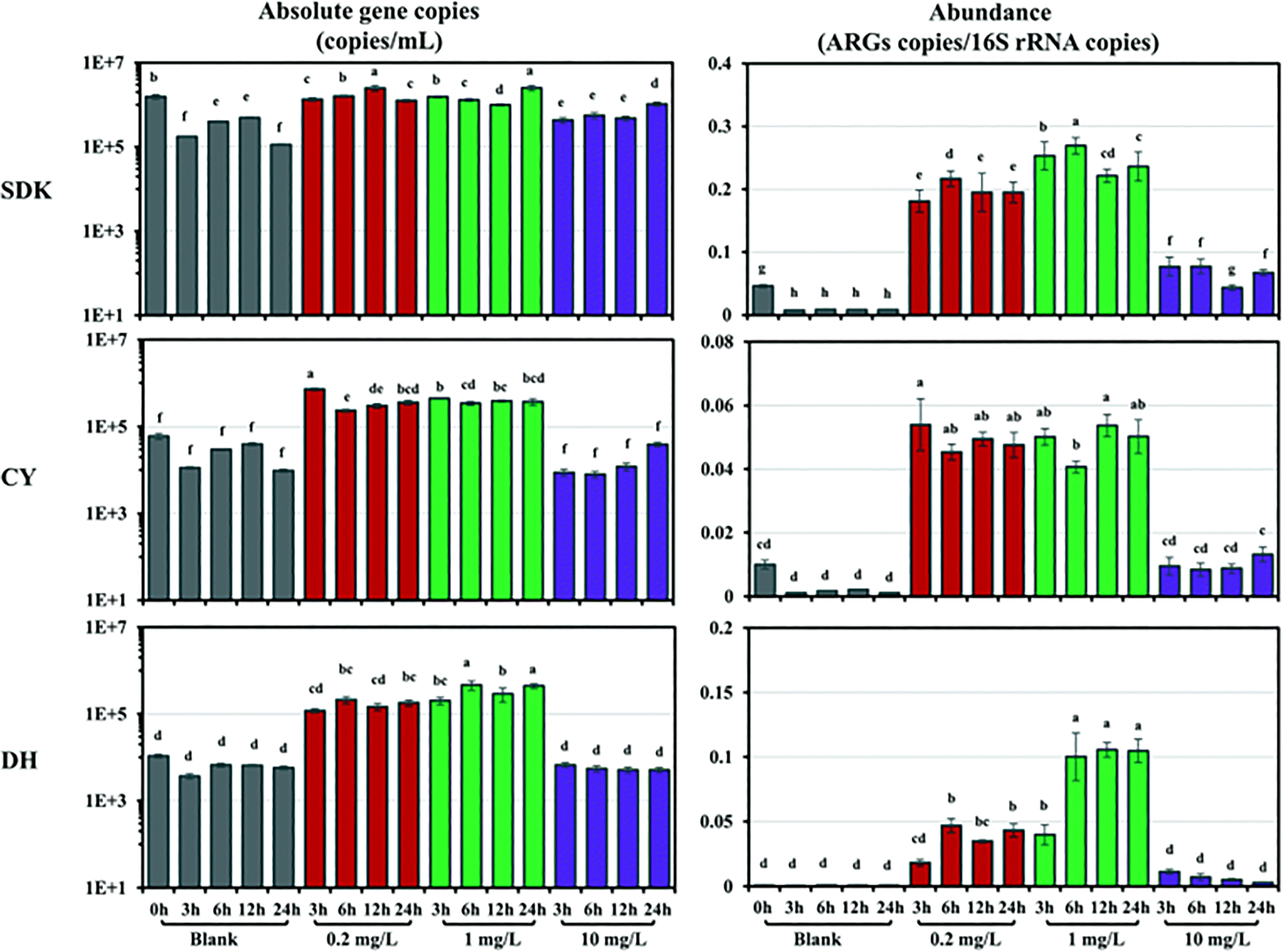
The absolute and the relative abundances of total ARGs in various estuarine water samples under different exposure concentrations of ZnO nanoparticles. The nomenclature of SDK refers to Shidongkou; CY refers to Chaoyangnongchang; DH refers to Donghainongchang. Reprinted with permission from ref. 232
The release of Zn2+ ions from ZnO nanostructures owing to its instability and dissolution process contributes to enhanced cytotoxicity.233–235 The dissolution rate of Zn2+ ions is varying to different extents depending on the external factors, such as medium pH, temperature, and light condition. The dissolution process of Zn2+ ions that are involved in acidic condition and UV-irradiated environment is delineated by the following reaction mechanisms (Eqs. 6 and 7):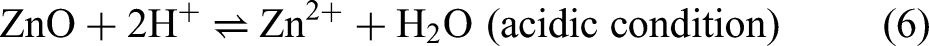
Besides, the inhibitory impact of ZnO-based nanostructures to the anaerobic wastewater treatment has also become a rising public concern. Zheng et al.
236
investigated the impacts of ZnO nanoparticles on wastewater nitrogen and phosphorus removals, and also their exposure to activated sludge which was cultured in the anaerobic-low dissolved oxygen SBR. The reductions in biological nitrogen and phosphorus removals were related to the decreased enzymatic activities of nitrate reductase (NR), exopolyphosphatase (PPX), and polyphosphate kinase (PPK). More recently, Otero-González et al.
237
evaluated the long-term effect of ZnO nanoparticles on the performance of an upflow anaerobic sludge blanket (UASB) reactor. Long-term exposure to a higher concentration of ZnO reduced acetate and propionate removals and hindered the methane production due to toxicity effect exerted on methanogens. More recently, Zhang et al.
238
studied the response of ZnO nanoparticles dosages on the granule-based anaerobic ammonium oxidation (anammox) process, which is an innovative alternative to nitrogen removal reaction by converting ammonium into dinitrogen gas through anammox bacteria that use nitrite as an electron acceptor.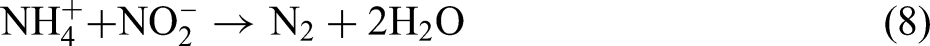
Additionally, it is crucial to understand the release and potential risks of ZnO-based nanostructures on the water/wastewater treatment system comprising of both oxic and anoxic processes. For example, Hu et al. 239 investigated the performance of an enhanced biological phosphorus removal (EBPR) system operating in an anaerobic/aerobic configuration after contact with ZnO nanoparticles. ZnO nanoparticles suppressed the phosphorus-release and phosphorus-uptake processes even with low concentration (2 mg/L). The microbial community analysed at genus level highlighted that ZnO nanoparticles reduced the abundances of Candidatus Accumulibacter (phenotype of PAOs) and Candidatus Competibacter phosphatis (phenotype of GAOs) after exposure for 23 days. Moreover, the effect of ZnO nanoparticles has also been tested on an anoxic-aerobic submerged membrane bioreactor (MBR) by characterising the soluble microbial products (SMPs) in the activated sludge. 240 In terms of the treatment efficiency, a reactor that was exposed to 50 mg/L ZnO nanoparticles demonstrated a reduction in COD removal (86.3 ± 2.3%, control: 93.4 ± 0.9%) and NH4+–N removal (65.2 ± 5.7%, control: 86.1 ± 3.6%). As illustrated in Figure 14(a) and 14(b), a higher content of SMPs (i.e., proteins, carbohydrates, and polysaccharides) and high molecular weight distribution of SMPs detected after exposure to ZnO nanoparticles signified that biomass decay and cell lysis have been substantially increased. This study also supported previous investigations stating that nanostructured materials had a stronger affinity for the biological sludge rather than dispersing in the solution and therefore, the major removal pathway for ZnO nanoparticles from wastewater is via aggregation and adsorption onto activated sludge.241,242
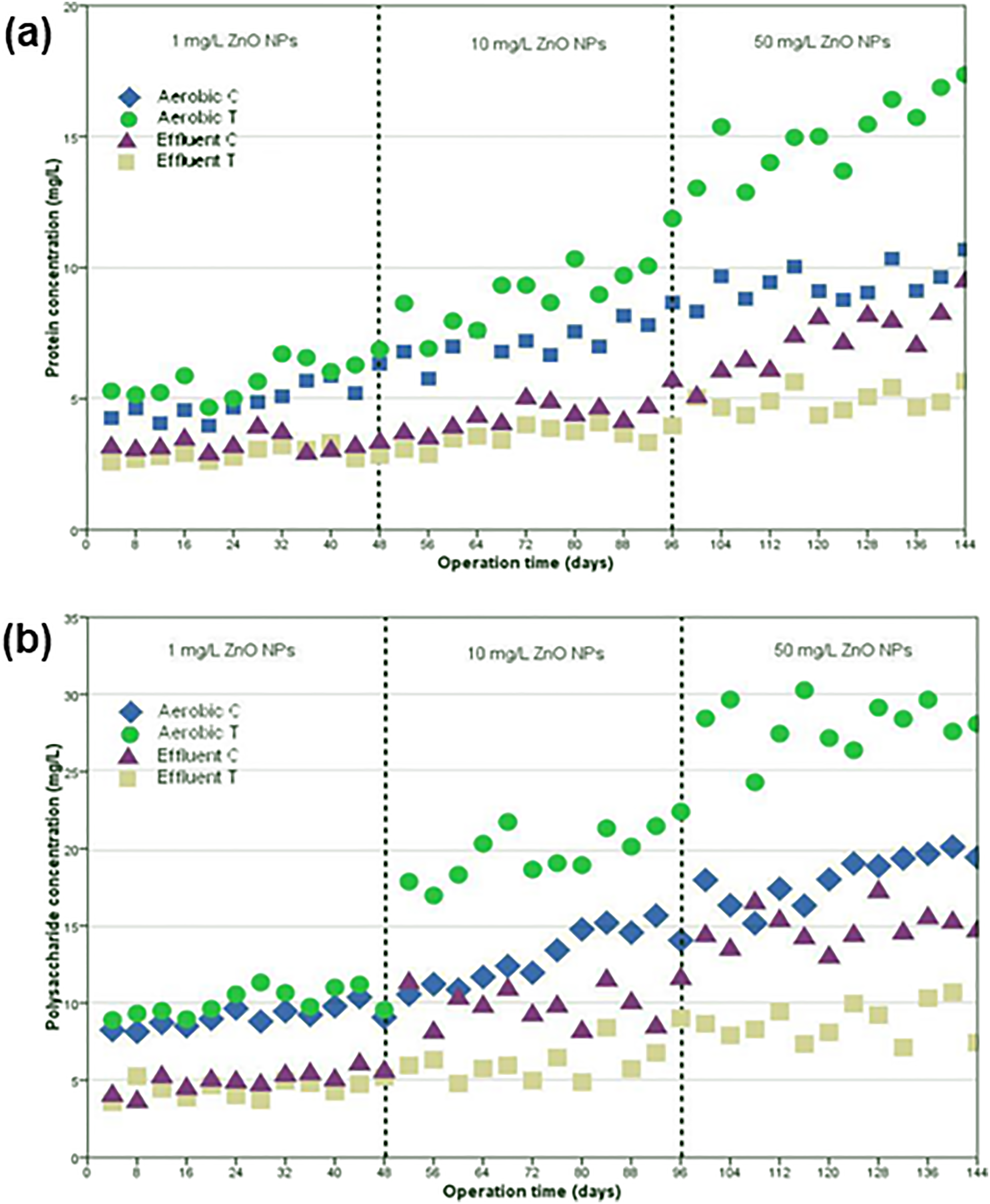
Effects of ZnO nanoparticles dosages on (a) protein concentration and (b) polysaccharide concentration. “C” represents control; and “T” represents treatment with ZnO nanoparticles. Reprinted with permission from ref. 240
Future outlook and perspectives
Over the years, extensive efforts dedicated to developing highly efficient ZnO-based nanostructures and hybrid nanocomposites for various applications and they have become more prominent owing to their excellent photocatalytic properties and potent antibacterial effects. Numerous enthralling methodologies in structural development and modification, such as nanostructure design, elemental doping, and heterojunction hybridization, have significantly elevated the responsiveness of ZnO-based nanostructures. This enhancement has positioned these semiconductor metal oxides as distinguished nanomaterials in the realms of materials science, advanced engineering, medicine and biomedicine, electrical and electronics, as well as applications related to energy and the environment. First and foremost, nanostructure design involves the controllable growth morphologies of ZnO nanostructures (i.e., 0D-, 1D-, 2D-, and 3D-nanoarchitectures) which could be easily tuned by synthesis methods, types and concentrations of precursors and additives, reaction temperatures, and pH of reacting medium. Secondly, incorporation of elemental doping, namely metal doping and non-metal doping, with ZnO nanostructures is essential to distinctly tune the optical, electrical, and magnetic properties of ZnO by governing the charge migration and separation. Lastly, matching the band alignment of ZnO with other semiconductors and/or compounds and uniting them into a heterojunction interface is one of the main criteria to synthesize an effective, functionalised photocatalyst. In general, the enhancement of the photocatalytic and antibacterial performances of pristine ZnO could be achieved through: (1) increasing the active surface areas and defect centres to encourage adsorption and reaction, (2) extending light absorption to a wider spectrum of wavelength, (3) suppressing the recombination rate of photogenerated e−-h+ pairs, and (4) increasing the charge carrier mobility.
In this critical review, we also highlighted the photocatalytic activities of improved ZnO-based nanostructures on dyes degradation, pollutant decomposition, and heavy metal ion removal. The persisting issues on discharging of recalcitrant micropollutants and highly toxic substances from households and industrial wastes to the water and wastewater streams have threatened human and environmental health. In this regard, there is a pressing need to develop economical and sustainable pre-treatment step or a stand-alone system to remove these contaminants. Heterogeneous photocatalysis utilising ZnO-based nanostructures has been considered as an effective approach that has achieved significant successes in destruction and elimination of micropollutants and azo dyes aqueous solution. Under light illuminated condition, the capability of the photocatalysts to produce a larger number of reactive species which are actively participated in the redox reaction for degradation purpose, is the key emphasis to improve pure ZnO photocatalyst which still suffer from the relatively low efficiency, low stability, and high photon energy-driven reaction. To date, there is an abundance of literature and knowledge about the enhancement techniques to resolve the aforementioned limitations by increasing surface adsorptivity, elevating the light harvesting ability, improving the recoverability and reusability. It is worth to note that adding dopants onto ZnO with distinct nanoarchitectured structures and further hybridising with other semiconductors are effective to (1) form unique structural orientation with a higher proportion of (002) polar planes that favour the ROS generation, (2) create more surface vacancies that act as charge carrier trapping site, (3) narrow down band gap energy, and (4) increase the charge transfer rate, which are all necessary for enhancing the photocatalytic efficiency of ZnO. Previous published works showed that the elemental doping is more effective than heterostructuring with the reaction rate constant of 0.05 to 0.2 min–1 (varying by dosage ratio, light irradiation power, type and concentration of studied pollutants) ascribed to a higher density of surface defects and electronic interaction that improve the photo-responsiveness.
This article also provided a critical perspective on the action of ZnO-based nanostructures as the antibacterial agents towards model Gram-positive and Gram-negative bacteria. The bacterial inhibition pathways are similar, which include physical bonding on cell membrane and cell wall, permeation into internal cell structure, interruption of transmembrane electron transport, oxidation of proteins, damage of mitochondria and DNA, and eventually cell death. These negative effects are commonly governed by two major antimicrobial mechanisms, namely the generation of ROS and leaching of dissolved Zn2+ ions. The antibacterial inhibition efficiency could achieve more than 99% reduction rate and ZOI of not lesser than 10 mm for both Gram-positive and Gram-negative bacteria. As a matter of course, the improvement of antibacterial ability of ZnO-based nanostructures could be attained by band gap engineering to enable greater light harvesting properties for triggering the generation of ROS and also through structural modification to maximise a higher density of exposed polar crystal surfaces which is propitious to the dissolution of Zn2+ ions. Although the investigation of antimicrobial pathways is vital to elucidate the fundamental enhancement and further optimisation of their antibacterial performances, concrete evidences and support on the specific bacterial motion phenomenon in a cellular environment are still scarce and more work is demanded to find out the impacts of contributing factors and how their adverse effects are exerted. Real-time visualisation on how the ZnO nanostructures interact with the microorganisms and detailed genetic analysis to identify the changes in cellular responses are essential to observe the biological impacts by ZnO.
In spite of some promising results regarding the photocatalytic treatment of wastewater effluents and toxicity to biological sludge reported thus far, the evaluation of environmental impacts of ZnO-based nanostructures are still in preliminary stage and further developments are prominently required. Notably, the wastewater treatment efficiency could either be enhanced owing to the photocatalytic properties or suppressed due to the antibacterial effect demonstrated by ZnO. For instance, a higher production rate of ROS not only was shown to increase the photo-redox efficiency for micropollutants degradation, but also caused the oxidative stress that adversely affected the microbial community in the sludge of biological wastewater treatment. This leads to a huge research upsurge by scientific professionals to evaluate the individual effects of ZnO photo-response on wastewater compartment decomposition and ZnO harmfulness to microbial diversity found in biological sludge components, and then followed by their integrated effect. More significantly, ZnO-based nanostructures demonstrated better removal rates of TOC (53.4% higher), COD (16.9% higher) and BOD5 (65.3% higher) than pristine ZnO nanostructures due to effective light absorption and rapid charge transfer. In response to this, the synergy of both experimental verification and computational simulation is mandatory for better understanding and to provide a logical framework to further advance the current state of knowledge on the photocatalysis and antibacterial performances of ZnO-based nanostructures in the real wastewater treatment application. In summary, there is a limitless scope of opportunities and challenges present to fully integrate this technology to wastewater treatment systems, as we need to further deepen our understanding of the fate and behaviour of nanomaterials. There are challenges from environmental activists calling for a worldwide moratorium on nanomaterial research and sustained functionality to accentuate their safety issue on water/wastewater treatment technologies. Finally, we are certain that this review will provide close to real scenario risk estimations and good guidelines for safeguarding the integrity of hazardous pollutants removal and targeted pathogen disinfection by ZnO nanomaterials and hence, ensuring clean water resources and long-term environmental protection.
Footnotes
Acknowledgements
Professor Dr Meng Nan Chong is highly indebted to the Royal Society-Newton Advanced Fellowship (Reference No.: NA150418) awarded to him. Dr Jang Sen Chang is also grateful to the MyBrain15 scholarship from the Ministry of Higher Education, Malaysia.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Royal Society (grant number NA150418).
