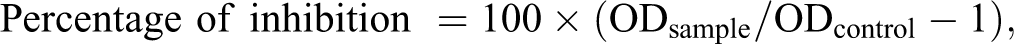Abstract
Silver nanoparticles (AgNPs) synthesized are utilized in drugs because of their pharmacological and biomedical applications and also due to their ecofriendly properties. In the present study, stable AgNPs have been synthesized from the aqueous extract of Dalbergia rostrata stem bark (DRSB), which is used both as a reducing and as a stabilizing agent. The AgNPs synthesized by ultrasonication at 25°C for 10 min were found to be stable in aqueous solution at room temperature over a period of 3 months. The quantitatively stable AgNPs formed by treating the aqueous solution of AgNO3 with the aqueous extract of the plant by reduction of Ag+ ions when monitored by UV–visible spectroscopic study revealed the surface plasmon resonance (SPR) at 425 nm. According to transmission electron micrography, the NPs were spherical and in the size range of 14 ± 4 nm. When evaluated for their anti-inflammatory and antioxidant activity by in vitro methods, AgNPs showed considerably enhanced activity compared to DRSB aqueous extract.
Introduction
Among higher plants, the family Leguminosae is a large one having more than 600 genera and 12,000 species. Plants belonging to this family vary in size from small shrubs and creepers to large tree. Dalbergia are important genus of tree, shrubs, and climbers, belonging to the family of Leguminosae and subfamily Papilionaceae, with about 120 species scattered in the tropics and subtropics and also in the temperate parts of South East Asia and North Australia.
Of the Dalbergia species available in the world, only 35 are reportedly present in India. 1,2 Dalbergia species have already been reported to possess a wide range of medicinal properties. Thorough phytochemical examination of various species of Dalbergia has resulted in the isolation and characterization of a large number of compounds like flavonoids, isoflavonoids, neoflavonoids, steroids, terpenoids, and so on; and a number of reviews have been published on the phytochemical investigations of Dalbergia species. 3–6
Flavonoids are reported to possess a variety of biological activities, and hence this group has recently gained much interest as active pharmacological agents. Flavonoids are known for their anti-inflammatory, antidiabetic, antiallergic effects, so on. They are also known for antithrombotic and vasoprotective properties, for inhibition of tumor promotion, and as protectives for gastric mucosa. Many naturally occurring flavonoids are found to be diuretics or antispasmodics. Bae et al. 7 have recently investigated the in vitro anti–Helicobacter pylori activity of a number of flavonoids.
Nanoparticle (NP) synthesis is normally carried out by various physical and chemical methods like green route facile, single-pot, lithography, laser ablation, electrodeposition, metal evaporation, UV light irradiation, sol -gel techniques. Silver nanoparticles (AgNPs) can be synthesized by reduction in solution 8–12 chemical and photochemical reaction in reverse micelles, sonochemical reduction, thermal decomposition and silver complex microwave assisted heatin. 13–15 AgNPs have been considered as an important area of research due to their unique and tunable surface plasmon resonance (SPR) and their applications in biomedical science including drug delivery, catalysis, tissue/tumor imaging, photothermal therapy, DNA detection, and protein detection. 16–19 Synthesis of AgNPs from different biological agents like microorganisms, yeast, bacteria, fungi, algae, actinomycetes, and plant extracts has been previously reported, which have potential applications in hyperthermia in cancer cells. 20,21
Nowadays, plant extracts are being used as reducing and capping agents for the synthesis of NPs, and this could be advantageous over microbial synthesis because there is no need of the elaborated process of culturing and maintaining the cell. The use of precious metal silver in nanotechnology has been on the increase due to its wide application. The control over the size and shape as well as the stability of AgNPs is important in electronics, sensor, chemistry, photography, and engineering owing to their specific optical and electrical properties. 22,23
In the present study, we have demonstrated the ability of Dalbergia rostrata stem bark (DRSB) to synthesize AgNPs without the addition of any external surfactant, capping agent, or template. Biological synthesis was adopted since it is ecofriendly. Dalbergia rostrata contains a wide range of active ingredients and research is underway regarding the evaluation of its uses as it is found to be rich in flavonoids, isoflavonoids, neoflavonoids, steroids, terpenoids, and gallic acid.
Materials and Methods
Plant Material and the Preparation of Extract by Microwave Method
D. rostrata is a delicate shrub belonging to the family Leguminosae. The stem bark and leaves of D. rostrata needed for the work were collected from Tropical Botanic Garden and Research Institute (TBGRI), Palode, Thiruvananthapuram, Kerala, India. The authenticity of the plant was confirmed by Dr. A. G. Pandurangan, Head, Department of Plant Systematics and Evolution Science, TBGRI, and a voucher specimen is deposited in TBGRI (specimen no: 26387) for future reference. Primarily the stem bark was cleaned with water, dried by pressing with blotted paper, shade dried, and finally cut into small pieces. Ten gram of stem bark in 100 mL of Millipore water was microwave irradiated for 5 min and the aqueous extract was then filtered and stored at 4°C for further experiments.
Synthesis of AgNPs
To 5 ml of DRSB aqueous extract taken in a conical flask, 50 ml of 0.1M silver nitrate solution was added drop wise with ultrasonication at 27°C. It was incubated at room temperature for 10 min and the change in color of the solution was checked periodically. The change in color of the aqueous extract from yellowish brown to dark brown indicated the formation of AgNPs using the aqueous extract of DRSB.
Purification of AgNPs
The fully reduced solution was centrifuged at 8000 rpm for 30 min. The supernatant liquid was discarded and the residue was dispersed in Millipore water. It was centrifuged four or five times to wash off any absorbed substances on the surface of the AgNPs.
Chemicals
All chemicals of analytical grade were obtained commercially and were used without further purification. AgNO3 (99.9%), 1,1-diphenyl-2-picryl-hydrazyl (DPPH), phosphate buffer, diclofenac, and ascorbic acid were purchased from Sigma-Aldrich, Mumbai, India. All the aqueous solutions were prepared using distilled deionized water.
Characterizaion
The specimen was prepared by adding AgNPs solution on to the var-coated copper grid and dried in air naturally. The morphology and composition of the product were identified using high-resolution transmission electron microscopy (HR-TEM). Images of AgNps were obtained from a JEOL JEM3010 operating at 200 kV (Icon Analytical Equipments, Mumbai, India. SPR and pH morphology was measured using UV-visible (UV-vis) spectrophotometer (Perkin Elmer Lambda 35, USA) with 1 cm quartz cell.
In Vitro Methods of Determination of Antioxidant Activity
A variety of in vitro methods are being used to determine the antioxidant activity of products and ingredients, but questions regarding whether the results have any bearing on effectiveness in the human body are leading to development of additional methods that may be more appropriate for screening potential antioxidant ingredients. In our study, we have used four methods, viz., DPPH radical scavenging, nitric oxide (NO), hydrogen peroxide (H2O2), and reducing power assay method for evaluating the antioxidant activity of the test drug.
DPPH Radical Scavenging Activity
In DPPH radical scavenging method, 0.1 mM solution of DPPH in ethanol was prepared and 1 mL of this solution was mixed with 3 mL of 0.01% sample solutions in water at different concentrations. After 30 min, the absorbance was measured at 517 nm. A decrease in absorbance of DPPH solution indicates an increase in the DPPH radical scavenging activity. 24
Reducing Power Assay
In the reducing power assay method, different concentrations of the aqueous extract of the plant in 1 mL of distilled water were mixed with phosphate buffer (2.5 mL, 0.2 M, pH 6.6) and potassium ferricyanide (2.5 mL, 1%). The mixture was incubated at 50°C for 20 min. Trichloroacetic acid (10%, 2.5 mL) was added to the mixture. A portion of the resulting mixture was mixed with ferric chloride (FeCl3; 0.1 %, 0.5 mL) and the absorbance was measured at 700 nm in a UV–vis spectrophotometer. In this method, ascorbic acid was used as the standard. 25
H2O2 Scavenging Activity
The H2O2 scavenging ability of the DRSB aqueous extract and AgNPs was determined by the method of Ruch et al. 26 A solution of H2O2 (40 mM) was prepared in phosphate buffer (pH 7.4). The AgNPs synthesized and the aqueous extracts of DRSB at varying concentrations (5, 10, 15, 20 and 25 µg/mL) were added individually to H2O2 solution (0.6 mL, 40 mM). The absorbance of the reaction mixture was recorded at 230 nm. A blank containing phosphate buffer only without H2O2 was also performed.
NO Radical Scavenging Activity 27
A stock solution of DRSB aqueous extract containing 1 mg/mL was prepared and different amounts of the stock solution (5, 10, 15, 20, and 25 µg/mL) were transferred to different test tubes and the volume was adjusted to 10 mL by the same solvent. Sodium nitro prusside, 0.2 mL (20 mM) in phosphate-buffered saline (PBS; pH 7.4) and 1.8 mL of PBS solution were added and incubated at 37°C for 3 h. One milliliter of each solution was taken and diluted with 1 mL of Griess reagent (1% sulfanilamide, 2% phosphoric acid (H3PO4), 0.1% N-1-naphthyl ethylene diamine); similarly, a blank was prepared containing the equivalent amount of reagents but without the AgNPs. The absorbance of these solutions was measured at 540 nm against the corresponding blank solutions. Ascorbic acid was used as a positive control in this experiment.
The percentage of inhibition was calculated in all the four methods for DRSB, AgNPs, and the standard using the equation
where, Ab is the absorbance of the blank, As is the absorbance for the test sample.
In Vitro Anti-Inflammatory Activity by Human Red Blood Cell Membrane Stabilization (HRBC) Method
The HRBC membrane stabilization has been used as a method to study the anti-inflammatory activity. 28 Blood collected from healthy volunteers was mixed with equal volume of sterilized Alsever solution (2% dextrose, 0.8% sodium citrate, 0.05% citric acid, and 0.42% sodium chloride in water). It was centrifuged at 3000 rpm and the packed cells were washed with isosaline (0.85%, pH 7.2) and a 10% v/v suspension was made with isosaline. The assay mixture contained the drug at various concentrations as mentioned previously, 1 mL phosphate buffer (0.15 M, pH 7.4), 2 mL of hyposaline (0.36 %), and 0.5 mL of HRBC suspension. Diclofenac sodium was used as the standard drug. Instead of hyposaline, 2 mL of distilled water was used in the control. All the assay mixtures were incubated at 37°C for 30 min and centrifuged. The hemoglobin content in the supernatant solutions was estimated at 560 nm using a spectrophotometer and the percentage of inhibition was calculated from HRBC membrane stabilization by using the formula
where ODsample is optical density of sample and ODcontrol is the optical density of control.
Catalytic Reduction of 4-Nitrophenol (4-NP)
The photocatalysis reactions were carried out in standard quartz cuvette with a 1-cm path length containing the reaction mixture, 1.4 cm3 of water, and 200 μL of 2 mmol dm–3 4-NP. Addition and proper mixing of 1 cm3 of aqueous 0.03 mol dm–3 sodium borohyride (NaBH4) and 30 μL of AgNPs solution to the reaction mixtures caused the decrease in the peak intensity of 4-NP. The reduction of 4-NP was monitored in situ using a UV–vis spectrophotometer. The reaction temperature was held constant at room temperature (20°C) to reduce thermal effects on the catalytic rate. The time for the reduction started and completion of the reaction varies and depends upon the capping of the AgNPs.
Results and Discussion
Experimental Results
Interest in drugs of herbal origin has significantly increased recently due to easy access of the medicinal plants and the less harmful side-effects of natural products. Scientific research is being conducted all over the world to determine whether plants traditionally used to treat various diseases are actually appropriate for their intended use. Antioxidants play the role of removing the free radicals by donating hydrogen to free radicals to reduce them to unreactive species. Addition of hydrogen would remove the odd electron feature that is responsible for radical reactivity. The hydrogen-donating activity was measured using DPPH radicals as hydrogen acceptor. The reducing potential of AgNPs obtained using aqueous extract of DRSB was examined by the DPPH, reducing power, H2O2, and NO antioxidant assay methods, and the results are summarized in Table 1. The results of in vitro anti-inflammatory activity by HRBC membrane stabilization method are given in Table 2.
In Vitro Antioxidant Assay of DRSB Aqueous Extract, AgNPs, and the Standard.

Note. DRSB, Dalbergia rostrata stem bark; AgNPs, silver nanoparticles; DPPH, 1,1-diphenyl-2-picryl-hydrazyl; H2O2, hydrogen peroxide; std, standard.
In Vitro Anti-Inflammatory Assay of DRSB Aqueous Extract, AgNPs, and the Standard by HRBC Method.

Note. DRSB, Dalbergia rostrata stem bark; AgNPs, silver nanoparticles; std, standard.
The reduction of silver ions could be accomplished by the phytochemicals such as flavonoids, gallic acid, or any other polyphenol/phytochelating agents present in DRSB. A possible mechanism proposed for the reduction of Ag+ is presented in Figure 1. Ag+ ions can form intermediate complexes with phenolic hydroxyls groups that are inductively oxidated to the corresponding quinones, and consequently it becomes logical to reason that the AgNPs prepared in this study should be stabilized by polyphenolic molecules through the quinones and thus Ag+ are reduced to AgNPs, 29 which are simultaneously stabilized by the correspondingly formed quinones. In our study, the reducing reaction is confirmed based on the change of color of the solution from yellowish brown to dark brown within 10 min and the SPR was centered at 425 nm. (Fig. 2). Hence the aqueous stem bark extract could be a powerful reducing agent for silver ion on ultrasonication at room temperature. The aqueous stem bark extract serves as a stabilizer for AgNPs in the proposed redox reaction. The stability of the AgNPs after 3 months was confirmed by SPR value of 425 nm (Figure 3). TEM images showed that the generated particles are nearly spherical in shape and their average size is around 14 ± 4 nm (Figure 4). The compositions of the prepared particles were further identified by Energy dispersive absorption spectroscopy (EDS) technique associated with TEM analysis and the EDS pattern recorded (Figure 5).

The possible mechanism involved in the formation and stabilization of silver nanoparticles (AgNPs).

UV–visible spectra of (A) DRSB aqueous extract and (B) AgNPs. AgNPs, silver nanoparticles; DRSB, Dalbergia rostrata stem bark.
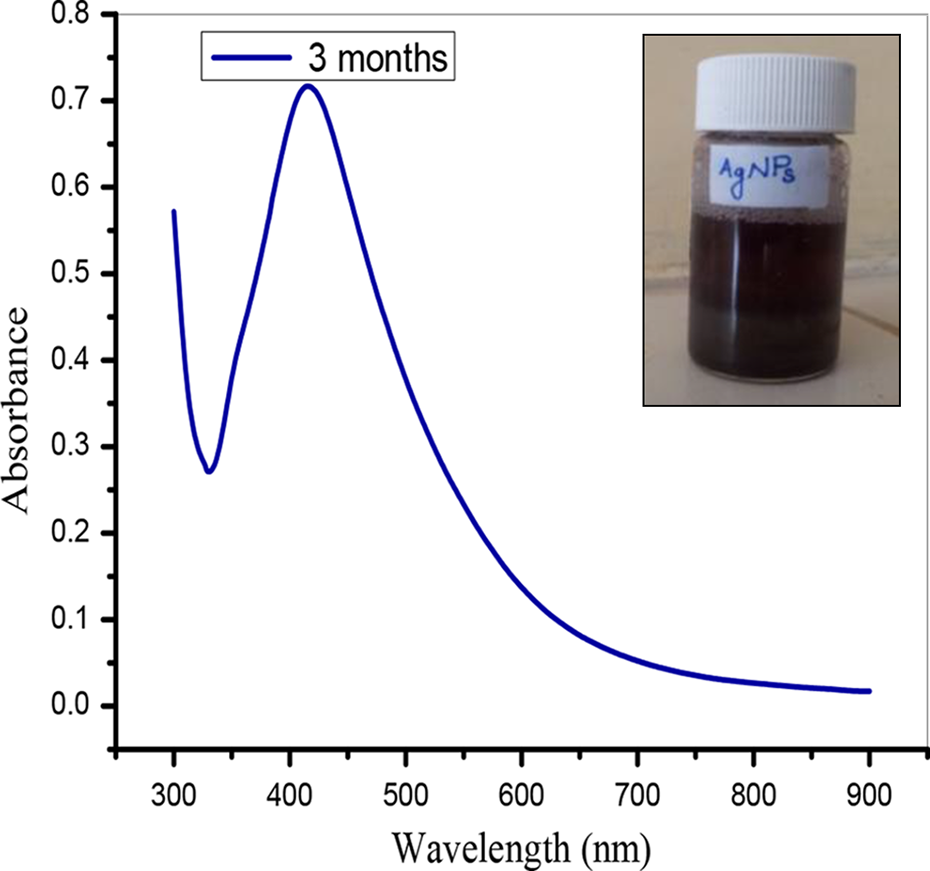
UV–visible spectra of the stable AgNPs (after 3 months). AgNPs, silver nanoparticles.

TEM analysis of spherical silver nanoparticles fabricated by DRSB aqueous extract at magnification (A)100 nm, (B) 50 nm, (C) 20 nm and (D) 10 nm. DRSB, Dalbergia rostrata stem bark; TEM, transmission electron microscopy.

Energy dispersive absorption spectroscopy (EDS) photograph of silver nanoparticles.
In Vitro Antioxidant Study of the AgNPs
Reactive oxygen species (ROS) produced by the in vitro methods mentioned above can interact with certain transition metal ions to yield a highly reactive oxidizing species (hydroxyl radicals). This antioxidant reacts further with the stable free radicals available, thereby causing their inhibition.
In the DPPH method, the antioxidant reacts with the stable DPPH (deep violet color) and converts it into 1,1-diphenyl-2-picrylhydrazine with discoloration. In the present study, the percentage of inhibition of free radicals at different concentrations ranging from 5 to 25 μg/mL for the AgNPs was calculated using ascorbic acid as the standard and the results are shown in Table 1.
Reducing power is associated with antioxidant activity, thereby serving as a significant reflection of the antioxidant activity. Drugs with reducing power indicate that they are electron donors and can reduce the oxidized intermediates of lipid peroxidation processes, so that they can act as primary and secondary antioxidants. In this assay, the yellow color of the test solution changes to various shades of green and blue, depending on the reducing power of each sample. Presence of reducing agents result in reduction of Fe3+/ferricyanide complex used to Fe2+, and by measuring the absorbance at 700 nm, the concentration of Fe3+ ion can be estimated.
In H2O2 scavenging method, when all the test samples were screened for H2O2 scavenging activity and compared with that of the standard (Figure 6), it was observed that the H2O2 scavenging activity of all the test samples increased dose dependently and the activity of the synthesized AgNPs was found to be higher than the standard as well as DRSB at the test doses.

In vitro antioxidant assay of DRSB aqueous extract, AgNPs, and the standard by DPPH, NO, H2O2 and reducing power assay methods. DRSB, Dalbergia rostrata stem bark; AgNPs, silver nanoparticles; DPPH, 1,1-diphenyl-2-picryl-hydrazyl; H2O2, hydrogen peroxide; NO, nitric oxide.
NO reacts with superoxide and forms peroxy nitrite radicals that are responsible for the inflammatory response caused by the release of prostaglandin. It is believed that repeated infections throughout early life can cause an excessive production of NO. In the present study, NO generated from sodium nitro prusside, at physiological pH 7.4, liberates nitric acid that gets converted into nitrous acid, further forming nitrite ions on contact with air. The nitrite ions when diazotized with sulfanilic acid and coupled with naphthylethylenediamine formed the pink color complex that was measured at 546 nm.
In Vitro Anti-Inflammatory Study of the AgNPs
In the present study, AgNPs at different concentrations (5, 10, 15, 20, and 25 µg/mL) showed significant stabilization toward HRBC membranes dose dependently, and the percentage inhibition of AgNPs at a concentration of 25 µg/mL was found to be the maximum and also higher than that of the standard. Standard drug diclofenac sodium showed an inhibition of 95.36%, and the results are shown in Table 2 and Figure 7.
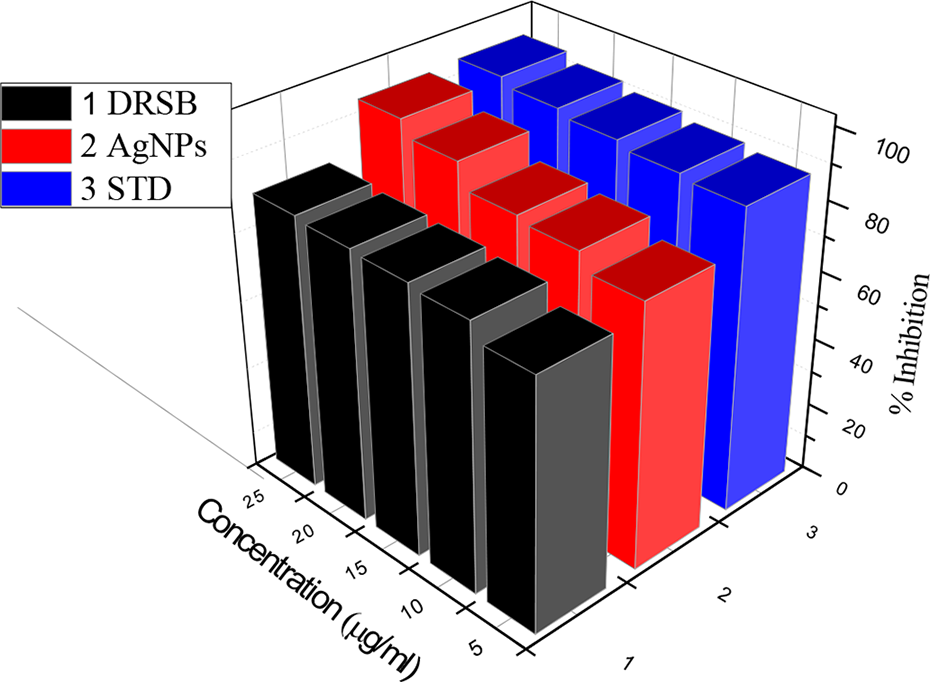
In vitro anti-inflammatory activity of DRSB aqueous extract, AgNPs, and the standard by HRBC membrane stabilization method. DRSB, Dalbergia rostrata stem bark; AgNPs, silver nanoparticles; HRBC, human red blood cell.
Catalytic Activity in 4-NP Degradation
Redox catalytic activities of fabricated AgNPs in the transformation of various aromatic organic molecules on various substrates have been explored. 30,31 The aqueous 4-NP, which shows maximum UV–vis absorbance at 317 nm, was taken for the process of remediation. When NaBH4 was added to reduce 4-NP, an intense yellow color appeared due to the formation of 4-nitrophenolate ion’s red shifting the absorption peak to 400 nm. This peak at 400 nm remained unaltered for several days in the absence of AgNPs. Normally aromatic compounds containing NO2 are inert to the reduction by NaBH4. 32,33 But with the addition of AgNPs, the yellow color fades to a colorless solution and the peak at 400 nm decreases with concomitant appearance of peak at 310 nm corresponding to the formation of 4-aminophenol ([4-AP] Figure 8). Generally, catalysts lower the activation energy of chemical reaction and increase the rate of reaction without being consumed in the process. Five different concentrations of AgNPs (5,10,15, 20 and 25 mg/L) were prepared by using deionized water. The nitro group of 4-NP was reduced to 4-AP, which was qualitatively monitored in UV–vis spectrophotometer. The formation of 4-AP was also confirmed by proton nuclear magnetic resonance spectrum.

UV- Vis absorption spectra for reduction of 4-nitrophenolate ion by AgNPs.
In this work, we have demonstrated for the first time, the complete quantitative catalytic reduction of 4-NP into 4-AP using NaBH4 and AgNPs. There are few drawbacks like restricted availability of surface area and polydispersity of AgNPs embedded in the plant aqueous extract. Though there are a few drawbacks like restricted availability of surface area and polydispersity of AgNPs embedded in the plant aqueous extract, considering the economic viability and environmental friendliness, this study may be helpful in the sustainable green biocatalytic reduction of various aromatic nitro pollutants.
Conclusions
In the present study, it has been observed that aqueous extract of DRSB is a good source for the synthesis of AgNPs by a green, high-yield, fast, and low-cost approach. AgNPs synthesized by the present method were of spherical shape and of average mean size of 14 ± 4 nm. Reduction is accomplished probably due to phytochemicals such as polyphenols, flavones, and flavonols. When evaluated for their antioxidant and anti-inflammatory activity at 5 to 25 µg/mL, the AgNPs showed enhanced activity when compared to that of the plant extract.
Footnotes
Acknowledgment
One of the authors (N. Muniyappan) thanks the UGC for the awarded of Research Fellowship for Meritorious Students to carry out this work.
Declaration of Conflicting Interests
The authors declare that there are no conflicts of interest with respect to the research work, authorship and /or publication of this article.
Funding
One of the authors (N. Muniyappan) thanks UGC, New Delhi for the award of Basic Sciences Research Fellowship for Meritorious Student and also for SAP awarded to the department of Chemistry.